Melatonin Pharmacokinetics Following Oral Administration in Preterm Neonates
Abstract
:1. Introduction
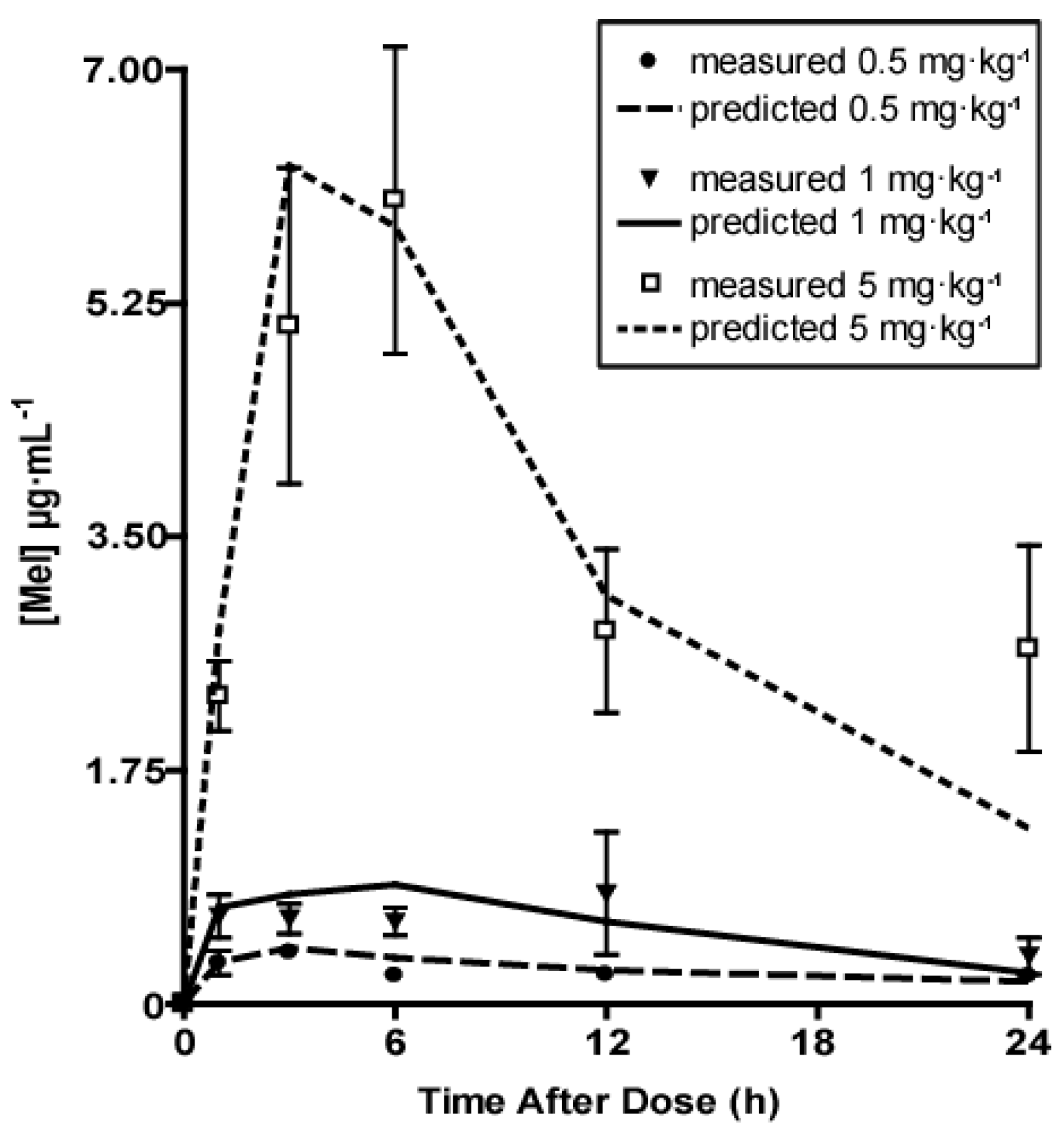
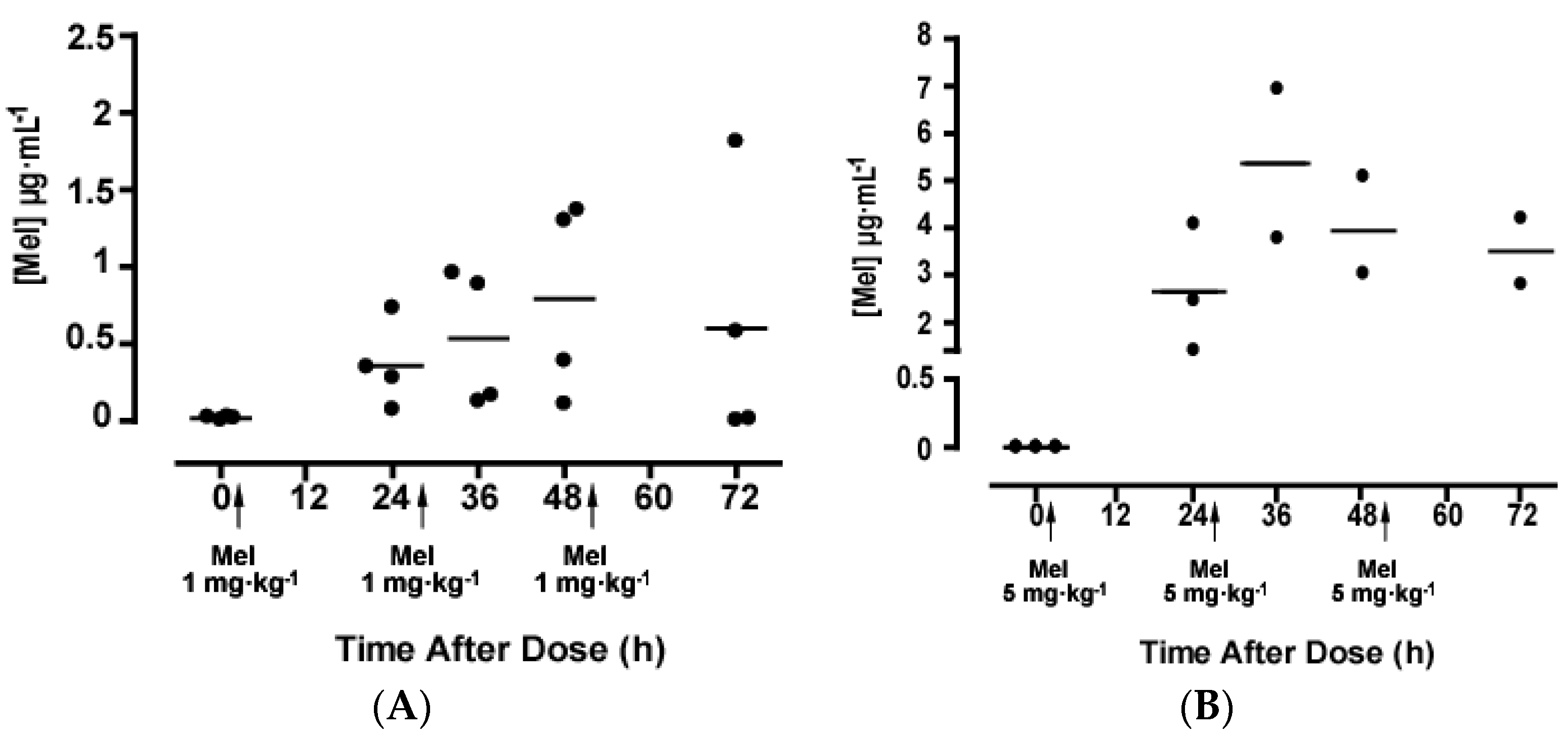
2. Results
2.1. Melatonin Concentration Assay Validation
2.2. Melatonin Pharmacokinetics
3. Discussion
4. Materials and Methods
4.1. Study Population
- (a)
- inclusion criteria: gestational age <37 weeks; normal liver function test (i.e., serum bilirubin, alkaline phosphatase, serum glutamic-oxaloacetic transaminase, serum glutamic pyruvic transaminase, etc.), normal kidney function test (i.e., serum creatinin levels, blood urea nitrogen); presence of an indwelling vascular catheter;
- (b)
- exclusion criteria: obvious congenital malformation; oliguria (<1 mL·kg−1·h−1) during preceding eight hours; documented infection. Demographic and clinical data are reported in Table 1.
4.2. Dose and Medication
4.3. Melatonin Concentration Assay
4.4. Pharmacokinetic Analysis
- (i)
- elimination half-life (T1/2)
- (ii)
- time of peak concentration (Tmax)
- (iii)
- maximal concentration (Cmax)
- (iv)
- area under the curve (AUC)
- (iv)
- area under the first moment curve (AUMC)
- (vi)
- mean residence time (MRT)
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Soleimani, F.; Zaheri, F.; Abdi, F. Long-term neurodevelopmental outcomes after preterm birth. Iran. Red Crescent Med. J. 2014, 16, e17965. [Google Scholar] [CrossRef] [PubMed]
- National Institute for Clinical Excellence. Therapeutic Hypothermia with Intracorporeal Temperature Monitoring for Hypoxic Perinatal Brain Injury. Guidance NICE Guidelines. Interventional Procedures. 2010. Available online: http://www.nice.org.uk/nicemedia/live/11315/48809/48809.pdf (accessed on 26 May 2010).
- Reiter, R.J.; Tan, D.X.; Fuentes-Broto, L. Melatonin: A multitasking molecule. Prog. Brain Res. 2010, 181, 127–151. [Google Scholar] [PubMed]
- Jacob, S.; Poeggeler, B.; Weishaupt, J.H.; Siren, A.L.; Hardeland, R.; Bahr, M.; Ehrenreich, H. Melatonin as a candidate compound for neuroprotection in amyotrophic lateral sclerosis (als): High tolerability of daily oral melatonin administration in als patients. J. Pineal Res. 2002, 33, 186–187. [Google Scholar] [CrossRef] [PubMed]
- Pappolla, M.A.; Chyan, Y.J.; Poeggeler, B.; Frangione, B.; Wilson, G.; Ghiso, J.; Reiter, R.J. An assessment of the antioxidant and the antiamyloidogenic properties of melatonin: Implications for alzheimer’s disease. J. Neural Transm. 2000, 107, 203–231. [Google Scholar] [CrossRef] [PubMed]
- Srinivasan, V.; Cardinali, D.P.; Srinivasan, U.S.; Kaur, C.; Brown, G.M.; Spence, D.W.; Hardeland, R.; Pandi-Perumal, S.R. Therapeutic potential of melatonin and its analogs in parkinson’s disease: Focus on sleep and neuroprotection. Ther. Adv. Neurol Disord 2011, 4, 297–317. [Google Scholar] [CrossRef] [PubMed]
- Escribano, B.M.; Colin-Gonzalez, A.L.; Santamaria, A.; Tunez, I. The role of melatonin in multiple sclerosis, huntington’s disease and cerebral ischemia. CNS Neurol. Disord. Drug Targets 2014, 13, 1096–1119. [Google Scholar] [CrossRef] [PubMed]
- Balduini, W.; Carloni, S.; Perrone, S.; Bertrando, S.; Tataranno, M.L.; Negro, S.; Proietti, F.; Longini, M.; Buonocore, G. The use of melatonin in hypoxic-ischemic brain damage: An experimental study. J. Matern. Fetal Neonatal Med. 2012, 25, 119–124. [Google Scholar] [CrossRef] [PubMed]
- Carloni, S.; Albertini, M.C.; Galluzzi, L.; Buonocore, G.; Proietti, F.; Balduini, W. Melatonin reduces endoplasmic reticulum stress and preserves sirtuin 1 expression in neuronal cells of newborn rats after hypoxia-ischemia. J. Pineal Res. 2014, 57, 192–199. [Google Scholar] [CrossRef] [PubMed]
- Carloni, S.; Perrone, S.; Buonocore, G.; Longini, M.; Proietti, F.; Balduini, W. Melatonin protects from the long-term consequences of a neonatal hypoxic-ischemic brain injury in rats. J. Pineal Res. 2008, 44, 157–164. [Google Scholar] [CrossRef] [PubMed]
- Carloni, S.; Riparini, G.; Buonocore, G.; Balduini, W. Rapid modulation of the silent information regulator 1 by melatonin after hypoxia-ischemia in the neonatal rat brain. J. Pineal Res. 2017, 63, e12434. [Google Scholar] [CrossRef] [PubMed]
- Moretti, R.; Zanin, A.; Pansiot, J.; Spiri, D.; Manganozzi, L.; Kratzer, I.; Favero, G.; Vasiljevic, A.; Rinaldi, V.E.; Pic, I.; et al. Melatonin reduces excitotoxic blood-brain barrier breakdown in neonatal rats. Neuroscience 2015, 311, 382–397. [Google Scholar] [CrossRef] [PubMed]
- Signorini, C.; Ciccoli, L.; Leoncini, S.; Carloni, S.; Perrone, S.; Comporti, M.; Balduini, W.; Buonocore, G. Free iron, total f-isoprostanes and total f-neuroprostanes in a model of neonatal hypoxic-ischemic encephalopathy: Neuroprotective effect of melatonin. J. Pineal Res. 2009, 46, 148–154. [Google Scholar] [CrossRef] [PubMed]
- Sinha, B.; Wu, Q.; Li, W.; Tu, Y.; Sirianni, A.C.; Chen, Y.; Jiang, J.; Zhang, X.; Zhou, S.; Chen, W.; et al. Protection of melatonin in experimental models of newborn hypoxic-ischemic brain injury through mt1 receptor. J. Pineal Res. 2017. [Google Scholar] [CrossRef] [PubMed]
- Ozdemir, D.; Uysal, N.; Gonenc, S.; Acikgoz, O.; Sonmez, A.; Topcu, A.; Ozdemir, N.; Duman, M.; Semin, I.; Ozkan, H. Effect of melatonin on brain oxidative damage induced by traumatic brain injury in immature rats. Physiol. Res. 2005, 54, 631–637. [Google Scholar] [PubMed]
- Carloni, S.; Favrais, G.; Saliba, E.; Albertini, M.C.; Chalon, S.; Longini, M.; Gressens, P.; Buonocore, G.; Balduini, W. Melatonin modulates neonatal brain inflammation through endoplasmic reticulum stress, autophagy, and mir-34a/silent information regulator 1 pathway. J. Pineal Res. 2016, 61, 370–380. [Google Scholar] [CrossRef] [PubMed]
- Miller, S.L.; Yan, E.B.; Castillo-Melendez, M.; Jenkin, G.; Walker, D.W. Melatonin provides neuroprotection in the late-gestation fetal sheep brain in response to umbilical cord occlusion. Dev. Neurosci. 2005, 27, 200–210. [Google Scholar] [CrossRef] [PubMed]
- Welin, A.K.; Svedin, P.; Lapatto, R.; Sultan, B.; Hagberg, H.; Gressens, P.; Kjellmer, I.; Mallard, C. Melatonin reduces inflammation and cell death in white matter in the mid-gestation fetal sheep following umbilical cord occlusion. Pediatr. Res. 2007, 61, 153–158. [Google Scholar] [CrossRef] [PubMed]
- Fulia, F.; Gitto, E.; Cuzzocrea, S.; Reiter, R.J.; Dugo, L.; Gitto, P.; Barberi, S.; Cordaro, S.; Barberi, I. Increased levels of malondialdehyde and nitrite/nitrate in the blood of asphyxiated newborns: Reduction by melatonin. J. Pineal Res. 2001, 31, 343–349. [Google Scholar] [CrossRef] [PubMed]
- Gitto, E.; Reiter, R.J.; Amodio, A.; Romeo, C.; Cuzzocrea, E.; Sabatino, G.; Buonocore, G.; Cordaro, V.; Trimarchi, G.; Barberi, I. Early indicators of chronic lung disease in preterm infants with respiratory distress syndrome and their inhibition by melatonin. J. Pineal Res. 2004, 36, 250–255. [Google Scholar] [CrossRef] [PubMed]
- Gitto, E.; Reiter, R.J.; Sabatino, G.; Buonocore, G.; Romeo, C.; Gitto, P.; Bugge, C.; Trimarchi, G.; Barberi, I. Correlation among cytokines, bronchopulmonary dysplasia and modality of ventilation in preterm newborns: Improvement with melatonin treatment. J. Pineal Res. 2005, 39, 287–293. [Google Scholar] [CrossRef] [PubMed]
- Aly, H.; Elmahdy, H.; El-Dib, M.; Rowisha, M.; Awny, M.; El-Gohary, T.; Elbatch, M.; Hamisa, M.; El-Mashad, A.R. Melatonin use for neuroprotection in perinatal asphyxia: A randomized controlled pilot study. J. Perinatol. 2015, 35, 186–191. [Google Scholar] [CrossRef] [PubMed]
- Buscemi, N.; Vandermeer, B.; Hooton, N.; Pandya, R.; Tjosvold, L.; Hartling, L.; Vohra, S.; Klassen, T.P.; Baker, G. Efficacy and safety of exogenous melatonin for secondary sleep disorders and sleep disorders accompanying sleep restriction: Meta-analysis. BMJ 2006, 332, 385–393. [Google Scholar] [CrossRef] [PubMed]
- Gitto, E.; Marseglia, L.; Manti, S.; D’Angelo, G.; Barberi, I.; Salpietro, C.; Reiter, R.J. Protective role of melatonin in neonatal diseases. Oxid. Med. Cell. Longev. 2013, 2013, 980374. [Google Scholar] [CrossRef] [PubMed]
- Harpsoe, N.G.; Andersen, L.P.; Gogenur, I.; Rosenberg, J. Clinical pharmacokinetics of melatonin: A systematic review. Eur. J. Clin. Pharmacol. 2015, 71, 901–909. [Google Scholar] [CrossRef] [PubMed]
- Yeleswaram, K.; McLaughlin, L.G.; Knipe, J.O.; Schabdach, D. Pharmacokinetics and oral bioavailability of exogenous melatonin in preclinical animal models and clinical implications. J. Pineal Res. 1997, 22, 45–51. [Google Scholar] [CrossRef] [PubMed]
- Gibbs, F.P.; Vriend, J. The half-life of melatonin elimination from rat plasma. Endocrinology 1981, 109, 1796–1798. [Google Scholar] [CrossRef] [PubMed]
- Merchant, N.M.; Azzopardi, D.V.; Hawwa, A.F.; McElnay, J.C.; Middleton, B.; Arendt, J.; Arichi, T.; Gressens, P.; Edwards, A.D. Pharmacokinetics of melatonin in preterm infants. Br. J. Clin. Pharmacol. 2013, 76, 725–733. [Google Scholar] [CrossRef] [PubMed]
- Sanchez-Barcelo, E.J.; Mediavilla, M.D.; Tan, D.X.; Reiter, R.J. Clinical uses of melatonin: Evaluation of human trials. Curr. Med. Chem. 2010, 17, 2070–2095. [Google Scholar] [CrossRef] [PubMed]
- Gitto, E.; Romeo, C.; Reiter, R.J.; Impellizzeri, P.; Pesce, S.; Basile, M.; Antonuccio, P.; Trimarchi, G.; Gentile, C.; Barberi, I.; et al. Melatonin reduces oxidative stress in surgical neonates. J. Pediatr. Surg. 2004, 39, 184–189. [Google Scholar] [CrossRef] [PubMed]
- Nair, A.B.; Jacob, S. A simple practice guide for dose conversion between animals and human. J. Basic Clin. Pharm. 2016, 7, 27–31. [Google Scholar] [CrossRef] [PubMed]
- Gitto, E.; Karbownik, M.; Reiter, R.J.; Tan, D.X.; Cuzzocrea, S.; Chiurazzi, P.; Cordaro, S.; Corona, G.; Trimarchi, G.; Barberi, I. Effects of melatonin treatment in septic newborns. Pediatr. Res. 2001, 50, 756–760. [Google Scholar] [CrossRef] [PubMed]
- Aldhous, M.; Franey, C.; Wright, J.; Arendt, J. Plasma concentrations of melatonin in man following oral absorption of different preparations. Br. J. Clin. Pharmacol. 1985, 19, 517–521. [Google Scholar] [CrossRef] [PubMed]
- Cardinali, D.P.; Lynch, H.J.; Wurtman, R.J. Binding of melatonin to human and rat plasma proteins. Endocrinology 1972, 91, 1213–1218. [Google Scholar] [CrossRef] [PubMed]
- Ehrnebo, M.; Agurell, S.; Jalling, B.; Boreus, L.O. Age differences in drug binding by plasma proteins: Studies on human foetuses, neonates and adults. Eur. J. Clin. Pharmacol. 1971, 3, 189–193. [Google Scholar] [CrossRef] [PubMed]
- Lerman, J.; Strong, H.A.; LeDez, K.M.; Swartz, J.; Rieder, M.J.; Burrows, F.A. Effects of age on the serum concentration of alpha 1-acid glycoprotein and the binding of lidocaine in pediatric patients. Clin. Pharmacol. Ther. 1989, 46, 219–225. [Google Scholar] [CrossRef] [PubMed]
- Kearns, G.L.; Abdel-Rahman, S.M.; Alander, S.W.; Blowey, D.L.; Leeder, J.S.; Kauffman, R.E. Developmental pharmacology—Drug disposition, action, and therapy in infants and children. N. Engl. J. Med. 2003, 349, 1157–1167. [Google Scholar] [CrossRef] [PubMed]
- Paakkonen, T.; Makinen, T.M.; Leppaluoto, J.; Vakkuri, O.; Rintamaki, H.; Palinkas, L.A.; Hassi, J. Urinary melatonin: A noninvasive method to follow human pineal function as studied in three experimental conditions. J. Pineal Res. 2006, 40, 110–115. [Google Scholar] [CrossRef] [PubMed]
- Kovacs, J.; Brodner, W.; Kirchlechner, V.; Arif, T.; Waldhauser, F. Measurement of urinary melatonin: A useful tool for monitoring serum melatonin after its oral administration. J. Clin. Endocrinol. Metab. 2000, 85, 666–670. [Google Scholar] [CrossRef] [PubMed]
- Tateishi, T.; Nakura, H.; Asoh, M.; Watanabe, M.; Tanaka, M.; Kumai, T.; Takashima, S.; Imaoka, S.; Funae, Y.; Yabusaki, Y.; et al. A comparison of hepatic cytochrome p450 protein expression between infancy and postinfancy. Life Sci. 1997, 61, 2567–2574. [Google Scholar] [CrossRef]
- Sonnier, M.; Cresteil, T. Delayed ontogenesis of cyp1a2 in the human liver. Eur. J. Biochem. 1998, 251, 893–898. [Google Scholar] [CrossRef] [PubMed]
- Aldridge, A.; Aranda, J.V.; Neims, A.H. Caffeine metabolism in the newborn. Clin. Pharmacol. Ther. 1979, 25, 447–453. [Google Scholar] [CrossRef] [PubMed]
- McCarver, D.G.; Hines, R.N. The ontogeny of human drug-metabolizing enzymes: Phase ii conjugation enzymes and regulatory mechanisms. J. Pharmacol. Exp. Ther. 2002, 300, 361–366. [Google Scholar] [CrossRef] [PubMed]
- Bueva, A.; Guignard, J.P. Renal function in preterm neonates. Pediatr. Res. 1994, 36, 572–577. [Google Scholar] [CrossRef] [PubMed]
- Wang, A.Q.; Wei, B.P.; Zhang, Y.; Wang, Y.J.; Xu, L.; Lan, K. An ultra-high sensitive bioanalytical method for plasma melatonin by liquid chromatography-tandem mass spectrometry using water as calibration matrix. J. Chromatogr. B Analyt. Technol. Biomed. Life Sci. 2011, 879, 2259–2264. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Huo, M.; Zhou, J.; Xie, S. Pksolver: An add-in program for pharmacokinetic and pharmacodynamic data analysis in microsoft excel. Comput. Methods Programs Biomed. 2010, 99, 306–314. [Google Scholar] [CrossRef] [PubMed]
- Dean, R.B.; Dixon, W.J. Simplified Statistics for Small Numbers of Observations. Anal. Chem. 1951, 23, 636–638. [Google Scholar] [CrossRef]
- Thrasher Research Fund, University of Florida. Melatonin as a Neuroprotective Therapy in Neonates With HIE Undergoing Hypothermia; National Library of Medicine: Bethesda, MD, USA, 2015. Available online: https://clinicaltrials.gov/show/NCT02621944 (accessed on 19 October 2015).
Sample Availability: Samples of the compounds are available from the authors. |
| Demographic and Clinical Data | Group-1 | Group-2 | Group-3 | |||
|---|---|---|---|---|---|---|
| Mean | Range | Mean | Range | Mean | Range | |
| Age (weeks) | 31 | 26–33 | 33 | 27–36 | 30 | 28–33 |
| Sex (M/F) | 3:3 | 4:0 | 1:4 | |||
| Birth Weight (g) | 1461 | 780–2150 | 2185 | 1050–3200 | 1803 | 1100–2830 |
| Apgar 1’ | 7 | 4–8 | 7 | 5–8 | 8 | 8–9 |
| Apgar 5’ | 8 | 7–9 | 9 | 8–9 | 9 | 8–9 |
| MV 1 (h) | 14 | 0–98 | 18 | 0–120 | 0 | 0 |
| Patient | A | B | C * | D | E | F | Mean ± SE |
|---|---|---|---|---|---|---|---|
| Kel | 0.06 | 0.07 | 0.05 | 0.11 | 0.04 | 0.05 | 0.07 ± 0.01 |
| Ka | 0.07 | 0.36 | 0.28 | 0.55 | 0.38 | 1.20 | 0.51 ± 0.19 |
| T1/2 (h) | 10.51 | 9.44 | 12.35 | 6.20 | 15.51 | 13.03 | 10.94 ± 1.58 |
| AUC (μg·mL−1·h) | 7.74 | 15.27 | 0.307 | 4.12 | 14.54 | 10.75 | 10.48 ± 2.09 |
| AUMC (μg·mL−1·h) | 203.03 | 367.01 | 8.90 | 67.19 | 460.67 | 251.90 | 269.96 ± 31.19 |
| Tmax (h) | 3.40 | 5.50 | 7.2 | 3.60 | 6.30 | 2.70 | 4.30 ± 0.68 |
| Cmax (µg·mL−1) | 0.32 | 0.58 | 0.008 | 0.30 | 0.40 | 0.59 | 0.44 ± 0.06 |
| MRT (h) | 26.20 | 24.03 | 28.97 | 16.29 | 31.66 | 23.41 | 24.32 ± 2.48 |
| Patient | G | H | I | L | Mean ± SE |
|---|---|---|---|---|---|
| Kel | 0.03 | 0.16 | 0.36 | 0.06 | 0.15 ± 0.07 |
| Ka | 2.08 | 0.84 | 1.83 | 2.75 | 1.88 ± 0.40 |
| T1/2 (h) | 20.81 | 4.07 | 1.88 | 10.73 | 9.37 ± 4.25 |
| AUC (μg·mL−1·h) | 37.58 | 28.54 | 3.95 | 18.96 | 22.26 ± 7.19 |
| AUMC (μg·mL−1·h) | 1386.85 | 348.18 | 31.42 | 344.95 | 527.85 ± 295.81 |
| Tmax (h) | 2.25 | 6.10 | 1.88 | 1.40 | 2.91 ± 1.08 |
| Cmax (µg·mL−1) | 0.88 | 1.78 | 0.50 | 0.97 | 1.03 ± 0.27 |
| MRT (h) | 36.89 | 12.19 | 7.94 | 18.18 | 18.80 ± 6.39 |
| Patient | M | N | O | P | Q | Mean ± SE |
|---|---|---|---|---|---|---|
| Kel | 0.13 | 0.12 | 0.06 | 0.11 | 0.06 | 0.09 ± 0.01 |
| Ka | 0.67 | 0.61 | 2.83 | 0.11 | 0.14 | 0.87 ± 0.50 |
| T1/2 (h) | 5.19 | 5.66 | 11.45 | 6.03 | 11.58 | 7.98 ± 1.45 |
| AUC (μg·mL−1·h) | 53.87 | 89.21 | 245.24 | 94.66 | 110.59 | 118.77 ± 33.02 |
| AUMC (μg·mL−1·h) | 787.32 | 1411.78 | 4300.18 | 1100.50 | 2063.28 | 1932.61 ± 628.40 |
| Tmax (h) | 3.01 | 3.30 | 1.40 | 8.70 | 7.10 | 4.70 ± 1.37 |
| Cmax (µg·mL−1) | 3.54 | 5.20 | 12.25 | 8.21 | 6.02 | 7.04 ± 1.50 |
| MRT (h) | 14,61 | 15.82 | 17.53 | 11.62 | 18.65 | 15.64 ± 1.22 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Carloni, S.; Proietti, F.; Rocchi, M.; Longini, M.; Marseglia, L.; D’Angelo, G.; Balduini, W.; Gitto, E.; Buonocore, G. Melatonin Pharmacokinetics Following Oral Administration in Preterm Neonates. Molecules 2017, 22, 2115. https://doi.org/10.3390/molecules22122115
Carloni S, Proietti F, Rocchi M, Longini M, Marseglia L, D’Angelo G, Balduini W, Gitto E, Buonocore G. Melatonin Pharmacokinetics Following Oral Administration in Preterm Neonates. Molecules. 2017; 22(12):2115. https://doi.org/10.3390/molecules22122115
Chicago/Turabian StyleCarloni, Silvia, Fabrizio Proietti, Marco Rocchi, Mariangela Longini, Lucia Marseglia, Gabriella D’Angelo, Walter Balduini, Eloisa Gitto, and Giuseppe Buonocore. 2017. "Melatonin Pharmacokinetics Following Oral Administration in Preterm Neonates" Molecules 22, no. 12: 2115. https://doi.org/10.3390/molecules22122115
APA StyleCarloni, S., Proietti, F., Rocchi, M., Longini, M., Marseglia, L., D’Angelo, G., Balduini, W., Gitto, E., & Buonocore, G. (2017). Melatonin Pharmacokinetics Following Oral Administration in Preterm Neonates. Molecules, 22(12), 2115. https://doi.org/10.3390/molecules22122115








