The Blood-Brain Barrier Permeability of Six Indole Alkaloids from Uncariae Ramulus Cum Uncis in the MDCK-pHaMDR Cell Monolayer Model
Abstract
:1. Introduction
2. Results and Discussion
2.1. Validation of the MDCK-pHaMDR Cell Monolayer
2.2. Validation of High-Performance Liquid Chromatography Analysis Method
2.3. Bidirectional Transport Determination
2.3.1. Bidirectional Transport of Alkaloids 1–6
2.3.2. Intracellular Accumulation and Recovery
2.3.3. The Time Course and Concentration-Dependence of Permeation of Alkaloids 1–6
2.3.4. The Relationship between Permeability and Drug Properties
2.4. The Interactions between Alkaloids and P-gp
2.4.1. Verapamil Inhibition on the Efflux of Alkaloids 1 and 3
2.4.2. Molecular Docking of 1–6 to P-gp
3. Experimental Section
3.1. Assayed Alkaloids
3.2. Chemicals and Reagents
3.3. HPLC Analysis
3.4. HPLC Method Validation
3.5. Culture of MDCK-pHaMDR Cells
3.6. Transport Experiments on MDCK-pHaMDR Cell Monolayer
3.6.1. Bidirectional Transport Experiments of Alkaloids 1–6
3.6.2. Time- and Concentration-Dependent Transport Experiments of Alkaloids 1–6
3.6.3. Verapamil Inhibition of Transport
3.6.4. Sample Preparations
3.6.5. Data Analysis
3.7. Statistical Analysis
3.8. Molecular Docking
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Chinese Pharmacopoeia Commission. Pharmacopoeia of the People’s Republic of China; China Medical Science and Technology Press: Beijing, China, 2015; Volume I, p. 257. [Google Scholar]
- Mo, Z.X.; Xu, D.D. New advances in clinical application of Uncariae Ramulus Cum Uncis. Lishizhen Med. Mater. Med. Res. 2006, 17, 684–685. [Google Scholar]
- Pan, H.Q.; Yang, W.Z.; Zhang, Y.B.; Yang, M.; Feng, R.H.; Wu, W.Y.; Guo, D.A. An integrated strategy for the systematic characterization and discovery of new indole alkaloids from Uncaria rhynchophylla by UHPLC/DAD/LTQ-Orbitrap-MS. Anal. Bioanal. Chem. 2015, 407, 6057–6070. [Google Scholar] [CrossRef] [PubMed]
- Yuan, D.; Ma, B.; Wu, C.F.; Yang, J.Y.; Zhang, L.J.; Liu, S.K.; Wu, L.J.; Kano, Y. Alkaloids from the leaves of Uncaria rhynchophylla and their inhibitory activity on NO production in lipopolysaccharide-activated microglia. J. Nat. Prod. 2008, 71, 1271–1274. [Google Scholar] [CrossRef] [PubMed]
- Jung, H.Y.; Nam, K.N.; Woo, B.C.; Kim, K.P.; Kim, S.O.; Lee, E.H. Hirsutine, an indole alkaloid of Uncaria rhynchophylla, inhibits inflammation-mediated neurotoxicity and microglial activation. Mol. Med. Rep. 2013, 7, 154–158. [Google Scholar] [CrossRef] [PubMed]
- Shimada, Y.; Goto, H.; Itoh, T.; Sakakibara, I.; Kubo, M.; Sasaki, H.; Terasawa, K. Evaluation of the protective effects of alkaloids isolated from the hooks and stems of Uncaria sinensis on glutamate-induced neuronal death in cultured cerebellar granule cells from rats. J. Pharm. Pharmacol. 1999, 51, 715–722. [Google Scholar] [CrossRef] [PubMed]
- Kawakami, Z.; Kanno, H.; Ikarashi, Y.; Kase, Y. Yokukansan, a kampo medicine, protects against glutamate cytotoxicity due to oxidative stress in PC12 cells. J. Ethnopharmacol. 2011, 134, 74–81. [Google Scholar] [CrossRef] [PubMed]
- Xian, Y.F.; Mao, Q.Q.; Wu, J.C.; Su, Z.R.; Chen, J.N.; Lai, X.P.; Ip, S.P.; Lin, Z.X. Isorhynchophylline treatment improves the amyloid-β-induced cognitive impairment in rats via inhibition of neuronal apoptosis and tau protein hyperphosphorylation. J. Alzheimer’s Dis. 2014, 39, 331–346. [Google Scholar]
- Wu, Y.T.; Lin, L.C.; Tsai, T.H. Determination of rhynchophylline and hirsutine in rat plasma by UPLC-MS/MS after oral administration of Uncaria rhynchophylla extract. Biomed. Chromatogr. 2014, 28, 439–445. [Google Scholar] [CrossRef] [PubMed]
- Wang, W.; Ma, C.M.; Hattori, M. Metabolism of isorhynchophylline in rats detected by LC-MS. J. Pharm. Pharm. Sci. 2010, 13, 27–37. [Google Scholar] [CrossRef] [PubMed]
- Wang, W.; Luo, S.Z.; Chen, Y.P.; Li, B.; Hattori, M. Effective separation and simultaneous determination of corynoxeine and its metabolites in rats by high-performance liquid chromatography with tandem mass spectrometry and application to pharmacokinetics and in vivo distribution in main organs. Anal. Sci. 2016, 32, 705–707. [Google Scholar] [CrossRef] [PubMed]
- Chen, Q.Z.; Zeng, C.Q.; Liu, L.; Zhu, D.; Chen, Q.L.; Liu, S.L.; Huang, B.Y. Pharmacokinetics and tissue distribution of isocorynoxeine in rats. Asia-Pac. Tradit. Med. 2016, 12, 18–21. [Google Scholar]
- Kushida, H.; Fukutake, M.; Tabuchi, M.; Katsuhara, T.; Nishimura, H.; Ikarashi, Y.; Kanitani, M.; Kase, Y. Simultaneous quantitative analyses of indole and oxindole alkaloids of Uncaria Hook in rat plasma and brain after oral administration of the traditional Japanese medicine Yokukansan using high-performance liquid chromatography with tandem mass spectrometry. Biomed. Chromatogr. 2013, 27, 1647–1656. [Google Scholar] [CrossRef] [PubMed]
- Abbott, N.J. Prediction of blood-brain barrier permeation in drug discovery from in vivo, in vitro and in silico models. Drug Discov. Today Technol. 2004, 1, 407–416. [Google Scholar] [CrossRef] [PubMed]
- Shityakov, S.; Salmas, R.E.; Durdagi, S.; Salvador, E.; Papai, K.; Yanez-Gascon, M.J.; Perez-Sanchez, H.; Puskas, I.; Roewer, N.; Forster, C.; et al. Characterization, in vivo evaluation, and molecular modeling of different propofol-cyclodextrin complexes to assess their drug delivery potential at the blood-brain barrier level. J. Chem. Inf. Model. 2016, 56, 1914–1922. [Google Scholar] [CrossRef] [PubMed]
- Shityakov, S.; Salvador, E.; Pastorin, G.; Forster, C. Blood-brain barrier transport studies, aggregation, and molecular dynamics simulation of multiwalled carbon nanotube functionalized with fluorescein isothiocyanate. Int. J. Nanomed. 2015, 10, 1703–1713. [Google Scholar] [CrossRef] [PubMed]
- Cecchelli, R.; Dehouck, B.; Descamps, L.; Fenart, L.; Buee-Scherrer, V.; Duhem, C.; Lundquist, S.; Rentfel, M.; Torpier, G.; Dehouck, M.P. In vitro model for evaluating drug transport across the blood-brain barrier. Adv. Drug Deliv. Rev. 1999, 36, 165–178. [Google Scholar] [CrossRef]
- Wang, Q.; Rager, J.D.; Weinstein, K.; Kardos, P.S.; Dobson, G.L.; Li, J.B.; Hidalgo, I.J. Evaluation of the MDR-MDCK cell line as a permeability screen for the blood-brain barrier. Int. J. Pharm. 2005, 288, 349–359. [Google Scholar] [CrossRef] [PubMed]
- Bicker, J.; Alves, G.; Fortuna, A.; Falcao, A. Blood-brain barrier models and their relevance for a successful development of CNS drug delivery systems: A review. Eur. J. Pharm. Biopharm. 2014, 87, 409–432. [Google Scholar] [CrossRef] [PubMed]
- Hellinger, E.; Veszelka, S.; Toth, A.E.; Walter, F.; Kittel, A.; Bakk, M.L.; Tihanyi, K.; Hada, V.; Nakagawa, S.; Duy, T.D.H.; et al. Comparison of brain capillary endothelial cell-based and epithelial (MDCK-MDR1, Caco-2, and VB-Caco-2) cell-based surrogate blood-brain barrier penetration models. Eur. J. Pharm. Biopharm. 2012, 82, 340–351. [Google Scholar] [CrossRef] [PubMed]
- Pastan, I.; Gottesman, M.M.; Ueda, K.; Lovelace, E.; Rutherford, A.V.; Willingham, M.C. A retrovirus carrying an MDR1 cDNA confers multidrug resistance and polarized expression of P-glycoprotein in MDCK cells. Proc. Natl. Acad. Sci. USA 1988, 85, 4486–4490. [Google Scholar] [CrossRef] [PubMed]
- Yang, Y.F.; Xu, W.; Song, W.; Ye, M.; Yang, X.W. Transport of twelve coumarins from Angelicae Pubescentis Radix across a MDCK-pHaMDR cell monolayer—An in vitro model for blood-brain barrier permeability. Molecules 2015, 20, 11719–11732. [Google Scholar] [CrossRef] [PubMed]
- Wu, N.; Xu, W.; Cao, G.Y.; Yang, Y.F.; Yang, X.B.; Yang, X.W. The blood-brain barrier permeability of lignans and malabaricones from the seeds of Myristica fragrans in the MDCK-pHaMDR cell monolayer model. Molecules 2016, 21, 134. [Google Scholar] [CrossRef] [PubMed]
- Madgula, V.L.M.; Avula, B.; Reddy, N.V.L.; Khan, K.A.; Khan, S.I. Transport of decursin and decursinol angelate across Caco-2 and MDR-MDCK cell monolayers: In vitro models for intestinal and blood-brain barrier permeability. Planta Med. 2007, 73, 330–335. [Google Scholar] [CrossRef] [PubMed]
- U.S. Department of Health and Human Services; Food and Drug Administration; Center for Drug Evaluation and Research; Center for Veterinary Medicine. Guidance for Industry, Bioanalytical Method Validation. 2013. Available online: http://www.fda.gov/ucm/groups/fdagov-public/@fdagov-drugs-gen/documents/document/ucm368107.pdf (accessed on 27 September 2017).
- Clark, D.E. Rapid calculation of polar molecular surface area and its application to the prediction of transport phenomena. 2. Prediction of blood–brain barrier penetration. J. Pharm. Sci. 1999, 88, 815–821. [Google Scholar] [CrossRef] [PubMed]
- Hou, T.J.; Xu, X.J. Recent development and application of virtual screening in drug discovery: An overview. Curr. Pharm. Des. 2004, 10, 1011–1033. [Google Scholar] [CrossRef] [PubMed]
- Li, J.Z.; Jaimes, K.F.; Aller, S.G. Refined structure of mouse P-glycoprotein. Protein Sci. 2014, 23, 34–46. [Google Scholar] [CrossRef] [PubMed]
- Karim, N.; Khan, I.; Abdelhalim, A.; Abdel-Halim, H.; Hanrahan, J.R. Molecular docking and antiamnesic effects of nepitrin isolated from Rosmarinus officinalis on scopolamine-inuced memory impairment in mice. Biomed. Pharmacother. 2017, 96, 700–709. [Google Scholar] [CrossRef] [PubMed]
- Yang, Y.F.; Wu, N.; Yang, X.W. Establishment of MDCK-pHaMDR cell model and standard operation procedure for assessing blood-brain barrier permeability of chemical components of traditional Chinese medicine. China J. Chin. Mater. Med. 2016, 41, 2706–2711. [Google Scholar]
Sample Availability: Samples of the compounds 1–6 are not available from the authors. |
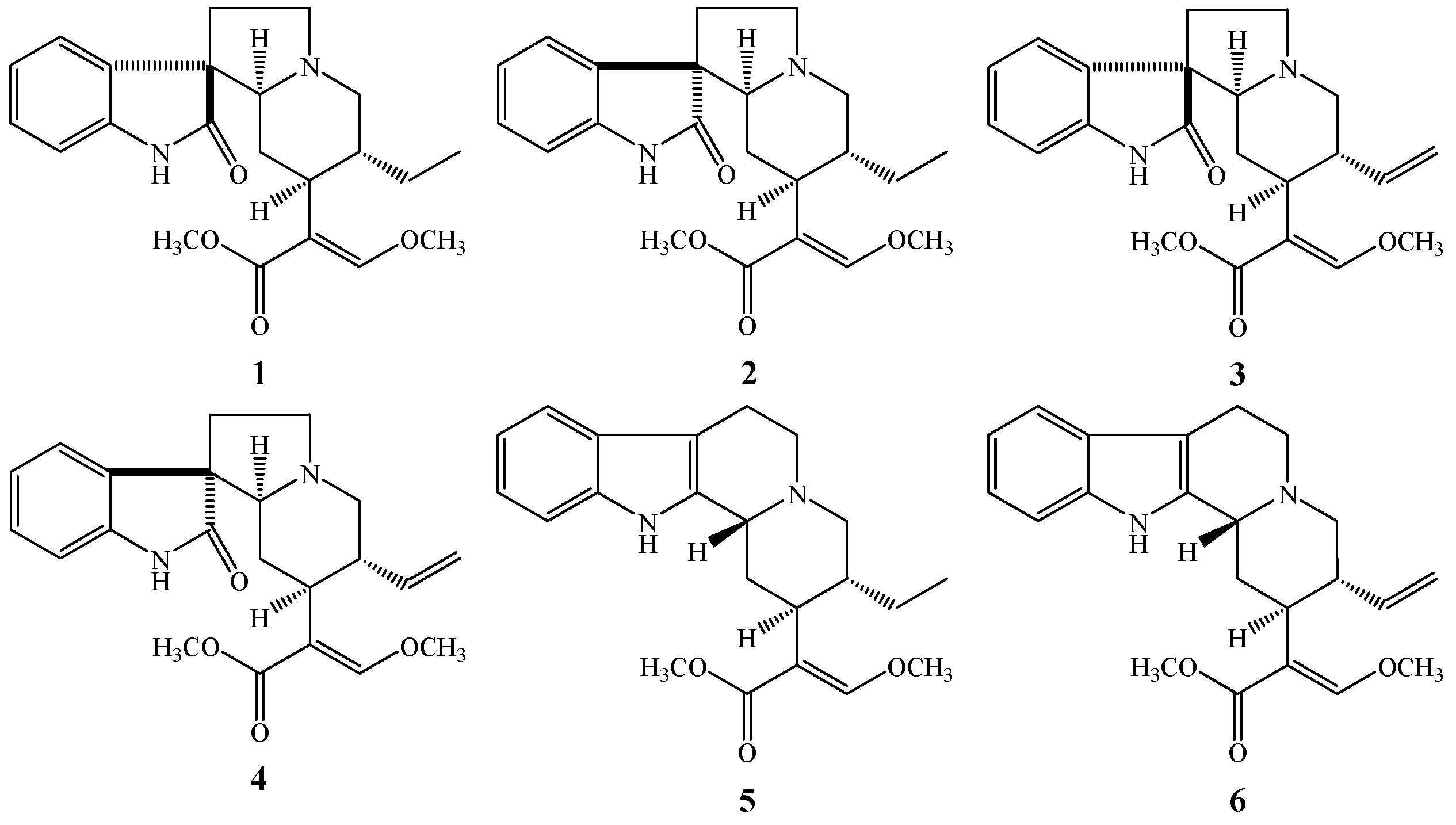
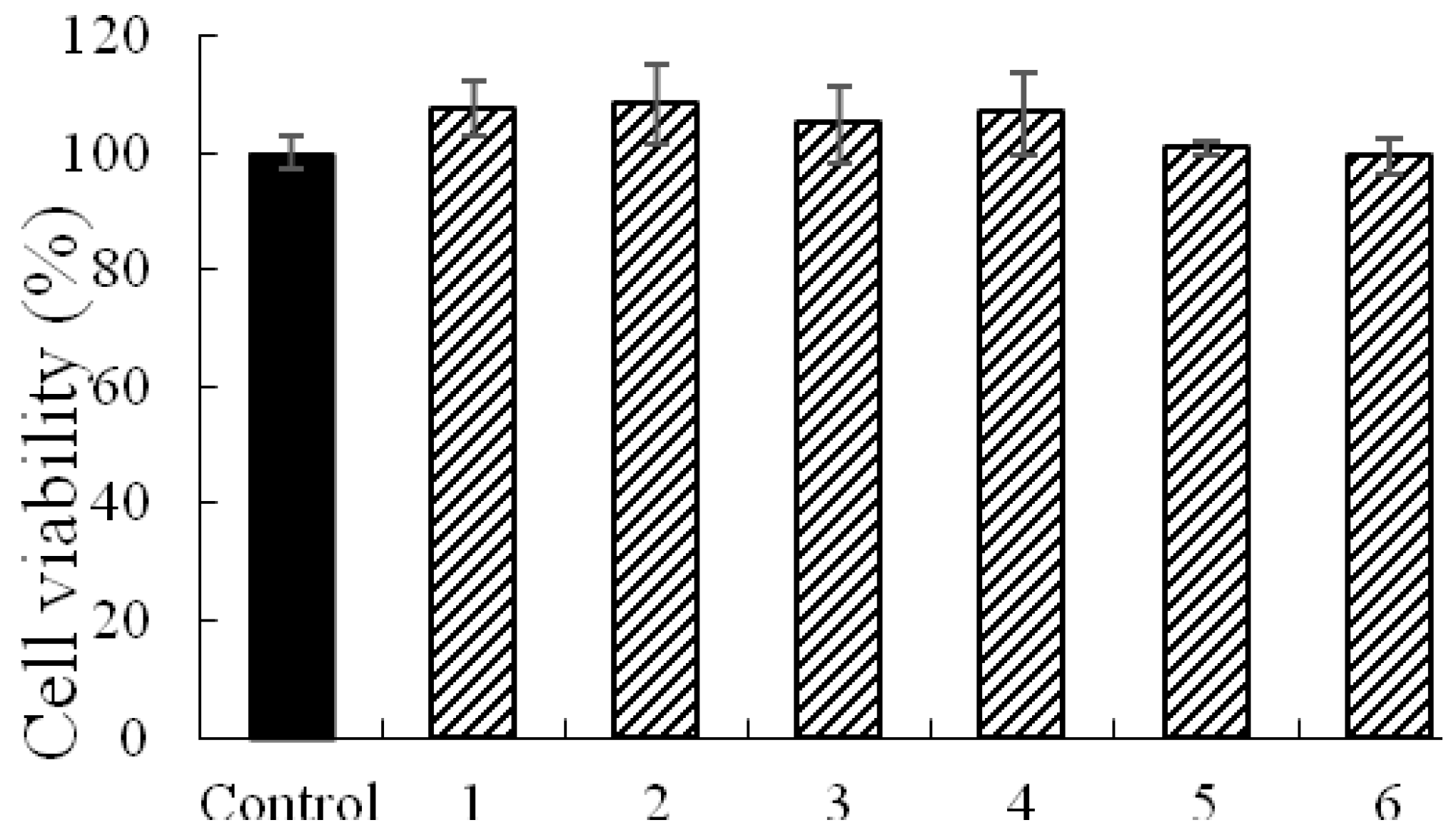
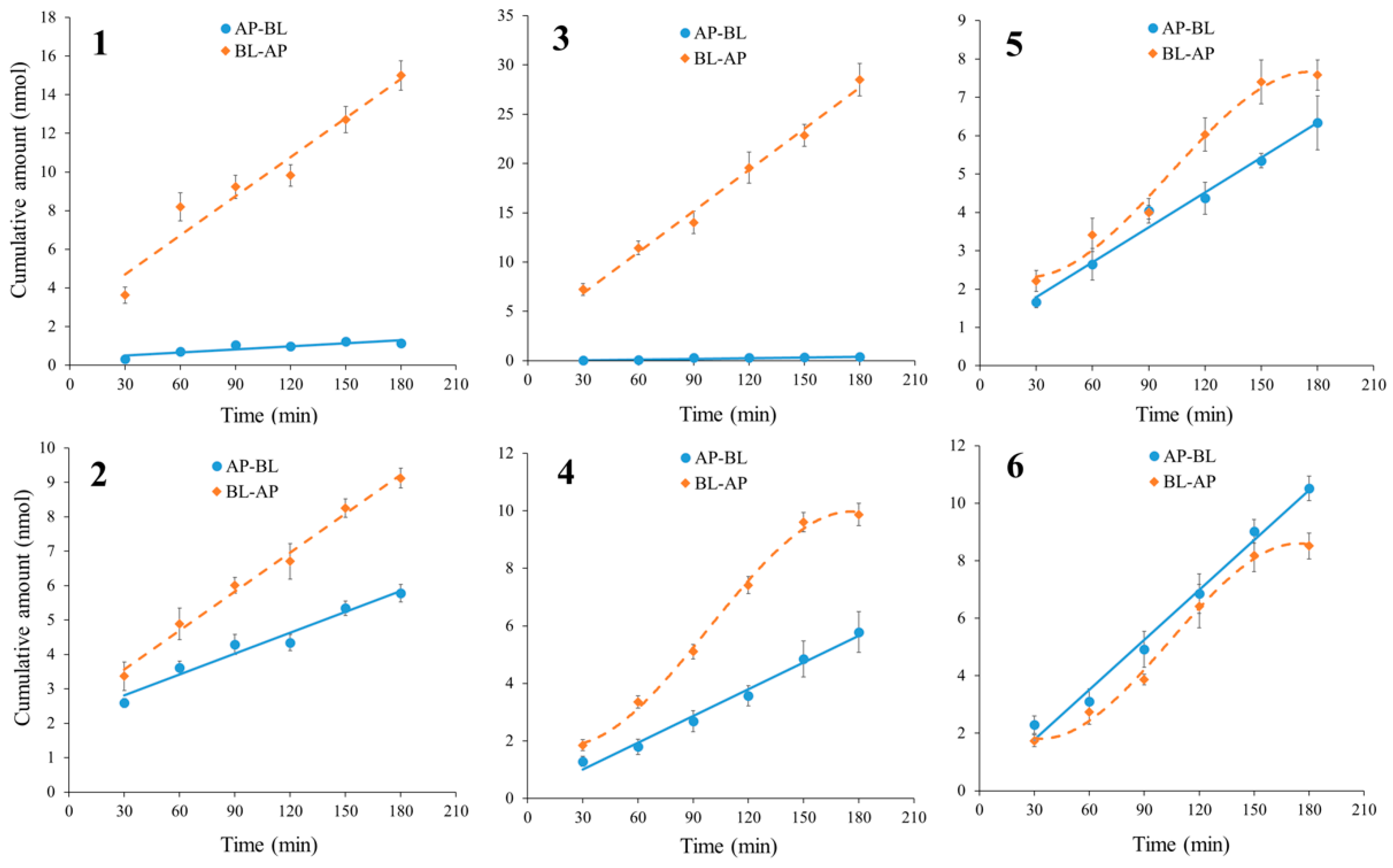

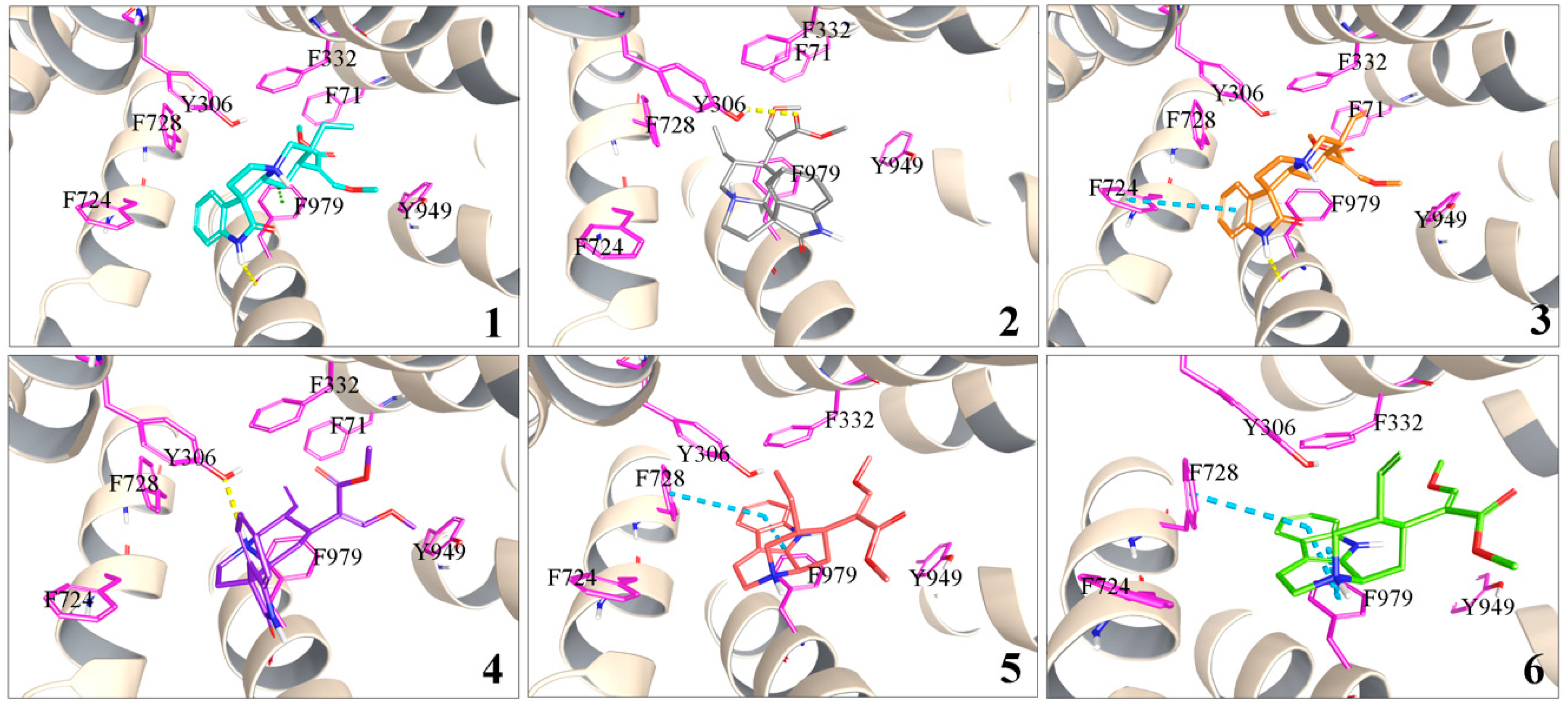
| Analytes | Papp AP→BL (×10−7 cm/s) | Papp BL→AP (×10−7 cm/s) | Efflux Ratio |
|---|---|---|---|
| Rh123 | 8.43 ± 1.95 | 57.99 ± 2.59 | 6.88 |
| Rh123 + verapamil | 12.70 ± 2.13 * | 17.07 ± 0.99 ** | 1.34 |
| Analytes | Regression Equation | r2 | Linear Range |
|---|---|---|---|
| 1 | y = 0.1327x − 0.3365 | 0.9979 | 1–150 µM |
| 2 | y = 0.1301x − 0.1076 | 0.9980 | 1–150 µM |
| 3 | y = 0.1109x + 0.0141 | 0.9989 | 1–150 µM |
| 4 | y = 0.1235x + 0.1916 | 0.9993 | 1–150 µM |
| 5 | y = 0.2634x − 0.0056 | 0.9997 | 1–150 µM |
| 6 | y = 0.1965x − 0.2061 | 0.9961 | 1–150 µM |
| Analytes | Concentration (μM) | Precision (%) | Accuracy (%) | Recovery (%) | Stability (%) | |
|---|---|---|---|---|---|---|
| Intraday | Interday | |||||
| 1 | 5 | 0.19 | 6.29 | 107.11 ± 6.23 | 103.65 ± 6.59 | 96.32 ± 3.30 |
| 60 | 0.56 | 6.41 | 100.94 ± 5.99 | 93.19 ± 5.70 | 96.86 ± 4.64 | |
| 120 | 0.11 | 4.84 | 104.81 ± 5.06 | 99.78 ± 4.89 | 102.59 ± 3.38 | |
| 2 | 5 | 0.40 | 10.34 | 111.75 ± 3.75 | 100.83 ± 3.23 | 108.52 ± 1.08 |
| 60 | 0.08 | 6.53 | 101.71 ± 6.59 | 94.44 ± 6.13 | 100.08 ± 2.26 | |
| 120 | 0.25 | 4.28 | 103.89 ± 4.32 | 95.94 ± 3.40 | 105.21 ± 3.28 | |
| 3 | 5 | 0.57 | 7.33 | 113.34 ± 5.49 | 108.14 ± 5.22 | 106.67 ± 5.82 |
| 60 | 0.07 | 6.66 | 105.42 ± 7.03 | 99.04 ± 6.60 | 102.37 ± 1.65 | |
| 120 | 0.44 | 5.75 | 105.18 ± 6.05 | 101.37 ± 5.83 | 101.52 ± 2.26 | |
| 4 | 5 | 0.15 | 5.40 | 106.75 ± 2.56 | 110.07 ± 4.54 | 107.15 ± 5.06 |
| 60 | 0.26 | 5.70 | 104.25 ± 3.15 | 95.09 ± 2.14 | 102.72 ± 1.97 | |
| 120 | 0.42 | 5.58 | 102.26 ± 2.55 | 95.88 ± 1.50 | 102.02 ± 2.43 | |
| 5 | 5 | 4.63 | 12.43 | 110.3 ± 3.79 | 104.9 ± 3.60 | 106.1 ± 3.57 |
| 60 | 0.21 | 5.68 | 102.2 ± 5.81 | 89.71 ± 5.10 | 101.9 ± 6.51 | |
| 120 | 0.25 | 6.40 | 106.5 ± 6.81 | 100.7 ± 6.44 | 100.5 ± 4.86 | |
| 6 | 5 | 1.16 | 3.67 | 112.2 ± 1.91 | 112.7 ± 2.33 | 106.89 ± 3.01 |
| 60 | 1.64 | 8.87 | 101.6 ± 8.83 | 93.75 ± 8.32 | 103.80 ± 5.92 | |
| 120 | 2.33 | 2.40 | 99.37 ± 2.36 | 96.60 ± 2.31 | 102.23 ± 4.19 | |
| Analytes | Papp AP→BL b (×10−6 cm/s) | Papp BL→AP c (×10−6 cm/s) | Efflux Ratio d | MW | Log D e (pH = 7.35) |
|---|---|---|---|---|---|
| 1 | 4.37 ± 0.37 (1) | 42.33 ± 2.48 (2) | 9.67 | 384.47 | 0.94 |
| 2 | 17.73 ± 1.20 | 27.73 ± 0.93 | 1.56 | 384.47 | 0.94 |
| 3 | 1.21 ± 0.14 (3) | 49.67 ± 4.71 (4) | 40.94 | 382.45 | 1.05 |
| 4 | 11.10 ± 1.49 | 20.66 ± 1.03 | 1.86 | 382.45 | 1.05 |
| 5 | 15.06 ± 1.32 | 14.11 ± 0.73 | 0.94 | 368.47 | 1.59 |
| 6 | 14.87 ± 1.83 | 11.35 ± 1.80 | 0.76 | 366.45 | 1.61 |
| 1 + verapamil | 22.24 ± 6.41 * | 18.13 ± 1.09 ▲ | 0.82 | ||
| 3 + verapamil | 17.25 ± 2.45 ♦ | 17.54 ± 0.82 △ | 1.02 |
| Analytes | AP→BL | BL→AP | ||
|---|---|---|---|---|
| Intracellular Accumulation (%) | Recovery Rate (%) | Intracellular Accumulation (%) | Recovery Rate (%) | |
| 1 | 0.36 ± 0.05 | 99.91 ± 1.15 | 0.40 ± 0.04 | 102.18 ± 0.47 |
| 2 | 0.24 ± 0.04 | 94.32 ± 1.43 | 0.37 ± 0.06 | 100.84 ± 1.89 |
| 3 | n.d. b | 96.36 ± 1.17 | n.d. b | 99.60 ± 1.57 |
| 4 | n.d. b | 92.96 ± 1.40 | n.d. b | 98.47 ± 0.82 |
| 5 | 16.82 ± 0.32 | 89.29 ± 6.15 | 6.19 ± 0.73 | 104.3 ± 1.94 |
| 6 | 19.27 ± 0.52 | 88.89 ± 5.99 | 7.52 ± 0.43 | 104.0 ± 1.17 |
| Compounds | 1 | 2 | 3 | 4 | 5 | 6 |
|---|---|---|---|---|---|---|
| log P | 2.18 | 2.18 | 1.73 | 1.73 | 3.22 | 2.62 |
| PSA | 67.87 | 67.87 | 67.87 | 67.87 | 54.56 | 54.56 |
| log BB | −0.53 | −0.53 | −0.60 | −0.60 | −0.18 | −0.27 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhang, Y.-N.; Yang, Y.-F.; Xu, W.; Yang, X.-W. The Blood-Brain Barrier Permeability of Six Indole Alkaloids from Uncariae Ramulus Cum Uncis in the MDCK-pHaMDR Cell Monolayer Model. Molecules 2017, 22, 1944. https://doi.org/10.3390/molecules22111944
Zhang Y-N, Yang Y-F, Xu W, Yang X-W. The Blood-Brain Barrier Permeability of Six Indole Alkaloids from Uncariae Ramulus Cum Uncis in the MDCK-pHaMDR Cell Monolayer Model. Molecules. 2017; 22(11):1944. https://doi.org/10.3390/molecules22111944
Chicago/Turabian StyleZhang, Yi-Nan, Yan-Fang Yang, Wei Xu, and Xiu-Wei Yang. 2017. "The Blood-Brain Barrier Permeability of Six Indole Alkaloids from Uncariae Ramulus Cum Uncis in the MDCK-pHaMDR Cell Monolayer Model" Molecules 22, no. 11: 1944. https://doi.org/10.3390/molecules22111944
APA StyleZhang, Y.-N., Yang, Y.-F., Xu, W., & Yang, X.-W. (2017). The Blood-Brain Barrier Permeability of Six Indole Alkaloids from Uncariae Ramulus Cum Uncis in the MDCK-pHaMDR Cell Monolayer Model. Molecules, 22(11), 1944. https://doi.org/10.3390/molecules22111944




