A Rapid and Simple TLC-Densitometric Method for Assay of Clobetasol Propionate in Topical Solution †
Abstract
:1. Introduction
2. Results and Discussion
2.1. Procedures
Optimization of Chromatographic Conditions
2.2. Validation Data
2.2.1. Specificity
2.2.2. Linearity
2.2.3. LOD and LOQ
2.2.4. Accuracy
2.2.5. Precision
2.2.6. Robustness
2.2.7. Analysis of Formulation (Topical Solution)
3. Experimental Section
3.1. Apparatus
3.2. Chemicals and Solvents
3.3. Pharmaceutical Preparation
3.4. Materials
3.5. Preparation of Standard Stock Solutions
3.6. Sample Preparation
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Jacob, S.E.; Steele, T. Corticosteroid classes: A quick reference guide including patch test substances and cross-reactivity. J. Am. Acad. Dermatol. 2006, 54, 723–727. [Google Scholar] [CrossRef] [PubMed]
- Makin, H.L.J.; Gower, D.B. Steroids Analysis, 2nd ed.; Springer: New York, NY, USA, 2010. [Google Scholar]
- Janiec, W. Compendium of Pharmacology, 4th ed.; PZWL: Warsaw, Poland, 2016. [Google Scholar]
- Brazzini, B.; Pimpinelli, N. New and established topical corticosteroids in dermatology: Clinical pharmacology and therapeutic use. Am. J. Clin. Dermatol. 2002, 3, 47–58. [Google Scholar] [CrossRef] [PubMed]
- Giannotti, B.; Pimpinelli, N. Topical corticosteroids. Which drug and when? Drugs 1992, 44, 65–71. [Google Scholar] [CrossRef] [PubMed]
- Stein, L. Clinical studies of a new vehicle formulation for topical corticosteroids in the treatment of psoriasis. J. Am. Acad. Dermatol. 2005, 53, 39–49. [Google Scholar] [CrossRef] [PubMed]
- Rajabi-Estarabadi, A.; Hasanzadeh, H.; Taheri, A.; Feldman, S.R.; Firooz, A. The efficacy of short-term clobetasol lotion in the treatment of scalp psoriasis. J. Dermatol. Treat. 2017, 6, 1–5. [Google Scholar] [CrossRef] [PubMed]
- ICH Harmonised Tripartite Guideline: Validation of Analytical Procedures: Text and Methodology, Q2(R1), ICH. 2005. Available online: http://www.ich.org/products/guidelines/efficacy/article/efficacy-guidelines.html (accessed on 10 September 2017).
- Fontana, M.C.; Bastos, M.O.; Beck, R.C.R. Development and validation of a fast RP-HPLC method for the determination of clobetasol propionate in topical nanocapsule suspensions. J. Chromatogr. Sci. 2010, 48, 637–640. [Google Scholar] [CrossRef] [PubMed]
- Turabi, Z.M.; Khatatbeh, O.A. Simultaneous determination of clobetasol (as propionate) and chlorocresol in cream by stability indicating RP-HPLC method. Int. J. Pharm. Sci. Drug Res. 2014, 6, 140–144. [Google Scholar]
- Gagliardi, L.; De Orsi, D.; Manna, F.; Tonelli, D. HPLC determination of clobetasol propionate in cosmetic products. J. Liq. Chromatogr. Relat. Technol. 2000, 23, 355–362. [Google Scholar] [CrossRef]
- Jakasaniya, M.A.; Shah, J.S.; Maheswari, D.G. Simultaneous estimation of clobetasol propionate and fusidic acid in cream dosage form by reversed phase high performance liquid chromatographic method. Pharmacophore 2014, 5, 231–238. [Google Scholar]
- Badilli, U.; Amasya, G.; Tarimci, N. Simultaneous determination of clobetasol propionate and calcipotriol in a novel fixed dose emulgel formulation by LC-UV. Chromatographia 2013, 76, 133–140. [Google Scholar] [CrossRef]
- Dyderski, S.; Grzeskowiak, E.; Mrzyglod, A. Pharmaceutical availability of clobetasol-17-propionate from cream and ointment. Acta Pol. Pharm. Drug Res. 2001, 58, 435–438. [Google Scholar]
- Kamberi, M.; Fu, K.; Lu, J.; Chemaly, G.M.; Feder, D. A sensitive high-throughput HPLC assay for simultaneous determination of everolimus and clobetasol propionate. J. Chromatogr. Sci. 2008, 46, 23–29. [Google Scholar] [PubMed]
- Manoharan, G. Development and validation of a stability-indicating RP-HPLC method for the estimation of clobetasol propionate in bulk and ointment dosage form. Eur. J. Pharm. Med. Res. 2017, 4, 119–126. [Google Scholar]
- Bhuyian, M.H.U.; Rashid, M.H.A.; Islam, A.F.M.A.; Tareque, I. Development and validation of method for determination of clobetasol propionate and salicylic acid from pharmaceutical dosage form by HPLC. Br. J. Pharm. Res. 2015, 7, 375–385. [Google Scholar] [CrossRef]
- Patel, B.; Raj, H.; Jain, V.; Sutariya, V.; Bhatt, M. Development and validation of reversed phase—High performance liquid chromatography method for clobetasol propionate and salicylic acid in its pharmaceutical dosage forms. Pharma Sci. Monit. 2014, 5 (Suppl. 1), 374–385. [Google Scholar]
- Golubovic, J.B.; Otasevic, B.M.; Protic, A.D.; Stankovic, A.M.; Zecevic, M.L. Liquid chromatography/tandem mass spectrometry for simultaneous determination of undeclared corticosteroids in cosmetic creams. Rapid Commun. Mass Spectrom. 2015, 29, 2319–2327. [Google Scholar] [CrossRef] [PubMed]
- Fauzee, A.F.; Walker, R.B. Forced degradation studies of clobetasol 17-propionate in methanol, propylene glycol, as bulk drug and cream formulations by RP-HPLC. J. Sep. Sci. 2013, 36, 849–856. [Google Scholar] [CrossRef] [PubMed]
- Nam, Y.S.; Kwon, I.K.; Lee, K.B. Monitoring of clobetasol propionate and betamethasone dipropionate as undeclared steroids in cosmetic products manufactured in Korea. Forensic Sci. Int. 2011, 210, 144–148. [Google Scholar] [CrossRef] [PubMed]
- Damle, M.C.; Polawar, A.R. Stability indicating HPTLC method for the estimation of clobetasol propionate in presence of alkali induced degradation product. Int. J. PharmTech Res. 2014, 6, 1914–1925. [Google Scholar]
- Bassuoni, Y.F.; Elzanfaly, E.S.; Essam, H.M.; El-Sayed Zaazaa, H. Development and validation of stability indicating TLC densitometric and spectrophotometric methods for determination of clobetasol propionate. Bull. Fac. Pharm. 2016, 54, 165–174. [Google Scholar] [CrossRef]
- Malani, P.S.; Raj, H.A.; Jain, V.C. Development and validation of analytical method for simultaneous estimation of miconazole nitrate and clobetasol propionate in cream by HPTLC method. J. Pharm. Sci. Monit. 2014, 5, 386–399. [Google Scholar]
- Polish Pharmacopoeia; Polish Pharmaceutical Society: Warsaw, Poland, 2014.
- The United States Pharmacopeia, 34th ed.; United States Pharmacopeial Convention: Rockville, MD, USA, 2011.
- Altemimi, A.; Lightfoot, D.A.; Kinsel, M.; Watson, D.G. Employing response surface methodology for the optimization of ultrasound assisted extraction of lutein and β-carotene from spinach. Molecules 2015, 20, 6611–6625. [Google Scholar] [CrossRef] [PubMed]
- Altemimi, A.; Watson, D.G.; Kinsel, M.; Lightfoot, D.A. Simultaneous extraction, optimization, and analysis of flavonoids and polyphenols from peach and pumpkin extracts using a TLC-densitometric method. Chem. Cent. J. 2015, 9, 39. [Google Scholar] [CrossRef] [PubMed]
- Sobanska, A.W.; Pyzowski, J.; Brzezinska, E. SPE/TLC/densitometric quantification of selected synthetic food dyes in liquid foodstuffs and pharmaceutical preparations. J. Anal. Methods Chem. 2017, 2017, 9528472. [Google Scholar] [CrossRef] [PubMed]
Sample Availability: Not Available. |

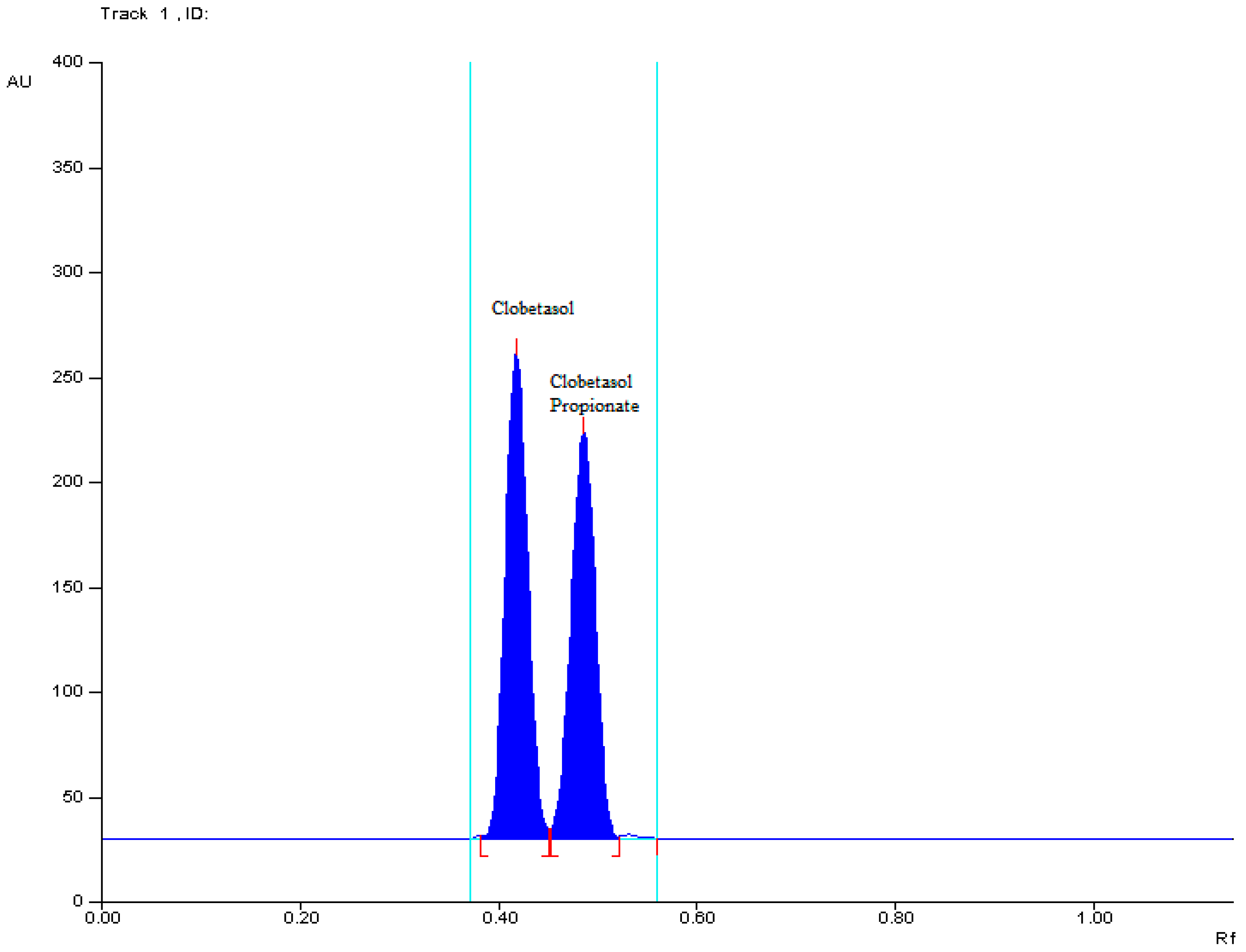

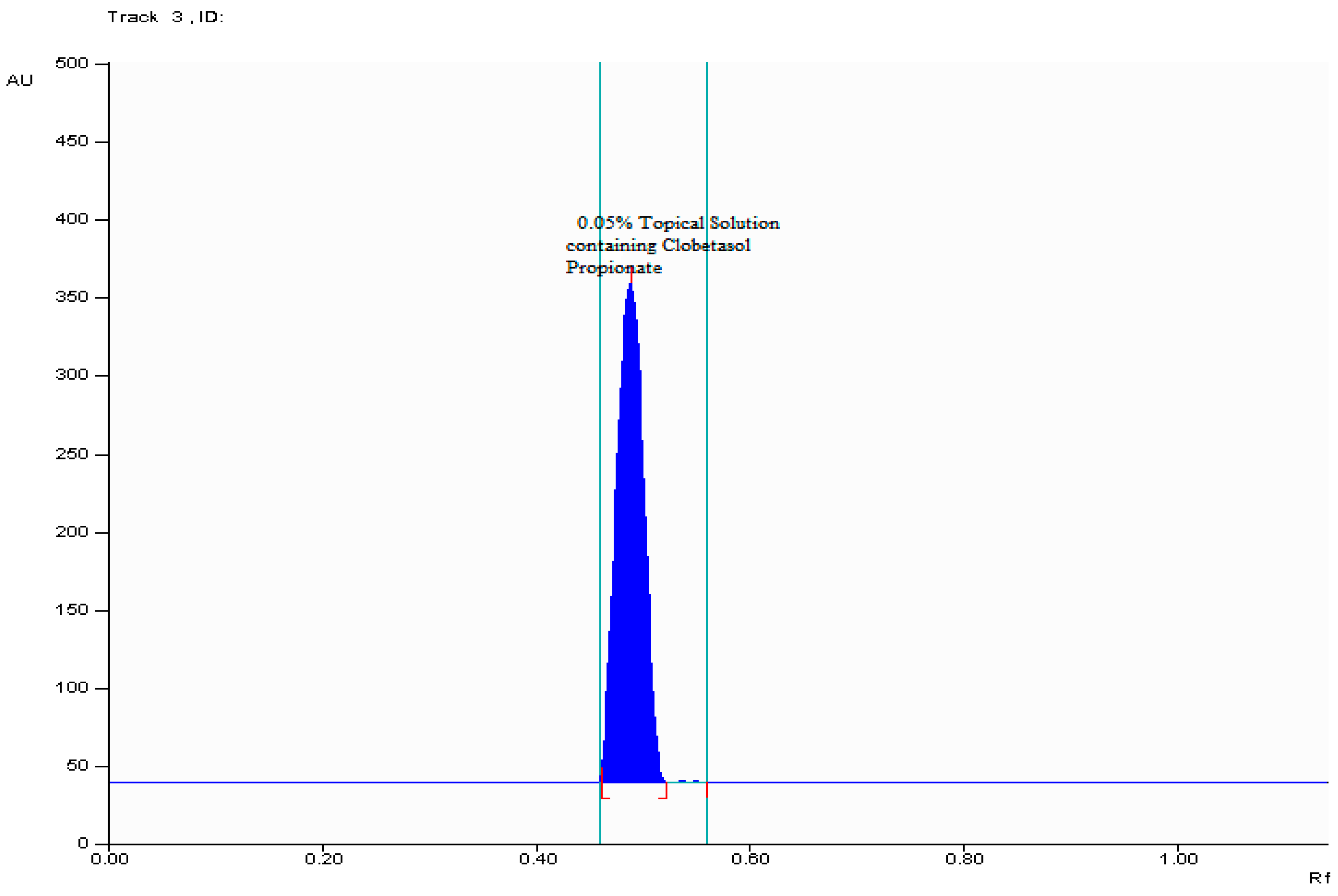
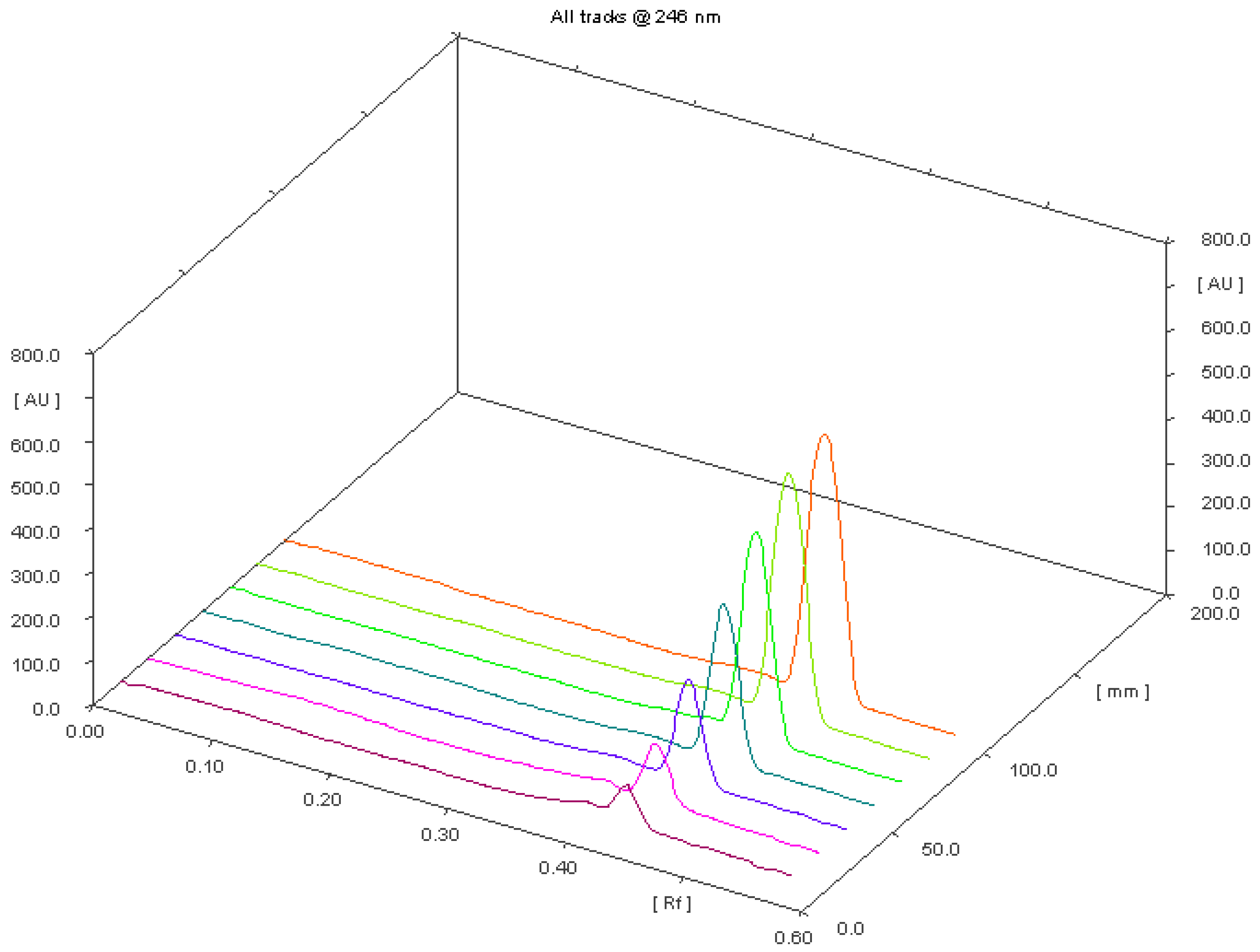
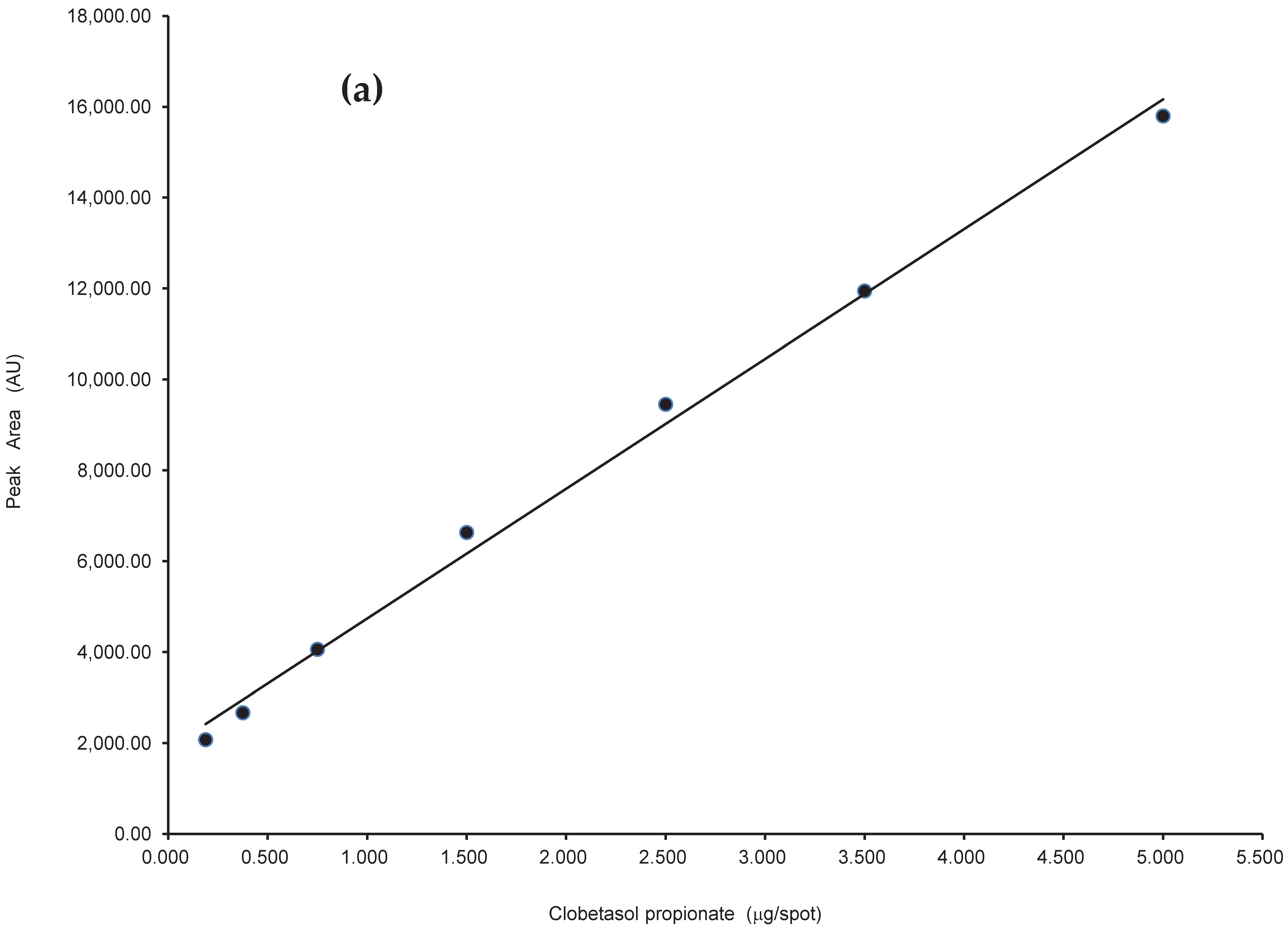

| Parameter | Results |
|---|---|
| ΔRF | 0.06 |
| RS | 1.25 |
| α | 1.27 |
| RFα | 1.13 |
| Parameter | Results |
|---|---|
| Wavelength (nm) | 246 |
| Linearity range (μg/spot) | 0.188–5 |
| Slope (b) | 2858.18 |
| Standard deviation of the slope (Sb) | 87.84 |
| Coefficient of variation (CV, %) of slope | 3.08 |
| Intercept (a) | 1877.43 |
| Standard deviation of the intercept (Sa) | 226.36 |
| Correlation coefficient (r) | 0.998 |
| Standard deviation of residuals (Sy/x) | 385.19 |
| F | 1058.85 |
| Significance level (p) | 0 |
| Limit of detection (LOD) in μg/spot | 0.061 |
| Limit of quantification (LOQ) in μg/spot | 0.186 |
| Nominal Amount of Clobetasol Propionate in Drug (mg/mL) | Amountof Pure Clobetasol Propionate Added (mg) | Total Amount of Clobetasol Propionate in Drug Sample (mg/mL) | Average Amount of Clobetasol Propionate Found in Drug Sample (mg/mL) | ±SD | CV (%) | Recovery (%) |
|---|---|---|---|---|---|---|
| 0.50 | 0.25 (50%) | 0.75 | 0.74 | 0.006 | 0.81 | 98.7 |
| 0.50 | 0.50 (100%) | 1.00 | 1.01 | 0.008 | 0.78 | 101.0 |
| 0.50 | 1.00 (150%) | 1.50 | 1.49 | 0.005 | 0.36 | 99.3 |
| Intra-Day Precision (Repeatability, n = 3) | |||
| Amount of Clobetasol Propionate (µg/spot) | Measured Amount ± SD | CV (%) | Recovery (%) |
| 1.00 | 1.01 ± 0.008 | 0.79 | 101.0 |
| 1.50 | 1.48 ± 0.014 | 0.94 | 98.7 |
| 2.00 | 2.03 ± 0.010 | 0.49 | 101.5 |
| Intra-Day Precision (Intermediate Precision, n = 3) | |||
| Amount of Clobetasol Propionate (µg/spot) | Measured Amount ± SD | CV (%) | Recovery (%) |
| 1.00 | 0.98 ± 0.009 | 0.92 | 98.0 |
| 1.50 | 1.49 ± 0.006 | 0.40 | 99.3 |
| 2.00 | 1.97 ± 0.023 | 1.17 | 98.5 |
| Method Parameters | CV (%) of Peak Area | Assay of Clobetasol Propionate (%) |
|---|---|---|
| Chamber saturation time (±10 min) | 0.75 | 101.5 |
| Volume of mobile phase (varied ±10 mL) | 1.48 | 111.8 |
| Development distance (±10 mm) | 1.31 | 109.1 |
| Temperature of activation of chromatographic plates (±10 °C) | 0.87 | 101.3 |
| Time from spotting to chromatography (±10 min) | 0.97 | 104.8 |
| Parameter | Data |
|---|---|
| Number of determinations | 6 |
| The label claim of clobetasol propionate in (mg/mL) | 0.50 |
| Average amount of clobetasol propionate (mg/mL) | 0.49 |
| Minimum amount of clobetasol propionate (mg/mL) | 0.47 |
| Maximum amount of clobetasol propionate (mg/mL) | 0.51 |
| Standard deviation (SD) | 0.008 |
| Coefficient of variation (CV, %) | 1.63 |
| Confidence interval of arithmetic mean with confidence level equal 95% (mg/mL) | 0.49 ± 0.02 |
| Amount of clobetasol propionate (%) in relation to the label claim | 98.0% |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Dolowy, M.; Kozik, V.; Bak, A.; Jampilek, J.; Barbusinski, K.; Thomas, M.; Pyka-Pajak, A. A Rapid and Simple TLC-Densitometric Method for Assay of Clobetasol Propionate in Topical Solution. Molecules 2017, 22, 1888. https://doi.org/10.3390/molecules22111888
Dolowy M, Kozik V, Bak A, Jampilek J, Barbusinski K, Thomas M, Pyka-Pajak A. A Rapid and Simple TLC-Densitometric Method for Assay of Clobetasol Propionate in Topical Solution. Molecules. 2017; 22(11):1888. https://doi.org/10.3390/molecules22111888
Chicago/Turabian StyleDolowy, Malgorzata, Violetta Kozik, Andrzej Bak, Josef Jampilek, Krzysztof Barbusinski, Maciej Thomas, and Alina Pyka-Pajak. 2017. "A Rapid and Simple TLC-Densitometric Method for Assay of Clobetasol Propionate in Topical Solution" Molecules 22, no. 11: 1888. https://doi.org/10.3390/molecules22111888
APA StyleDolowy, M., Kozik, V., Bak, A., Jampilek, J., Barbusinski, K., Thomas, M., & Pyka-Pajak, A. (2017). A Rapid and Simple TLC-Densitometric Method for Assay of Clobetasol Propionate in Topical Solution. Molecules, 22(11), 1888. https://doi.org/10.3390/molecules22111888










