Surface-Relief Gratings in Halogen-Bonded Polymer–Azobenzene Complexes: A Concentration-Dependence Study
Abstract
1. Introduction
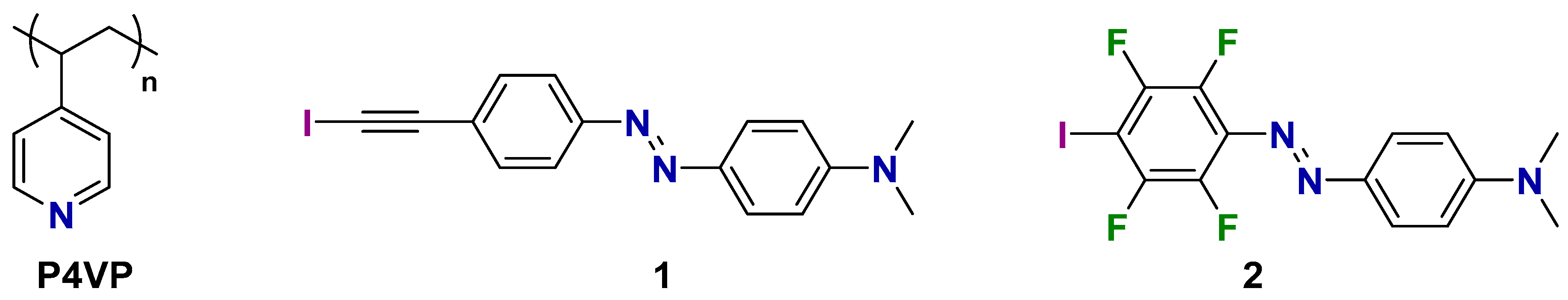
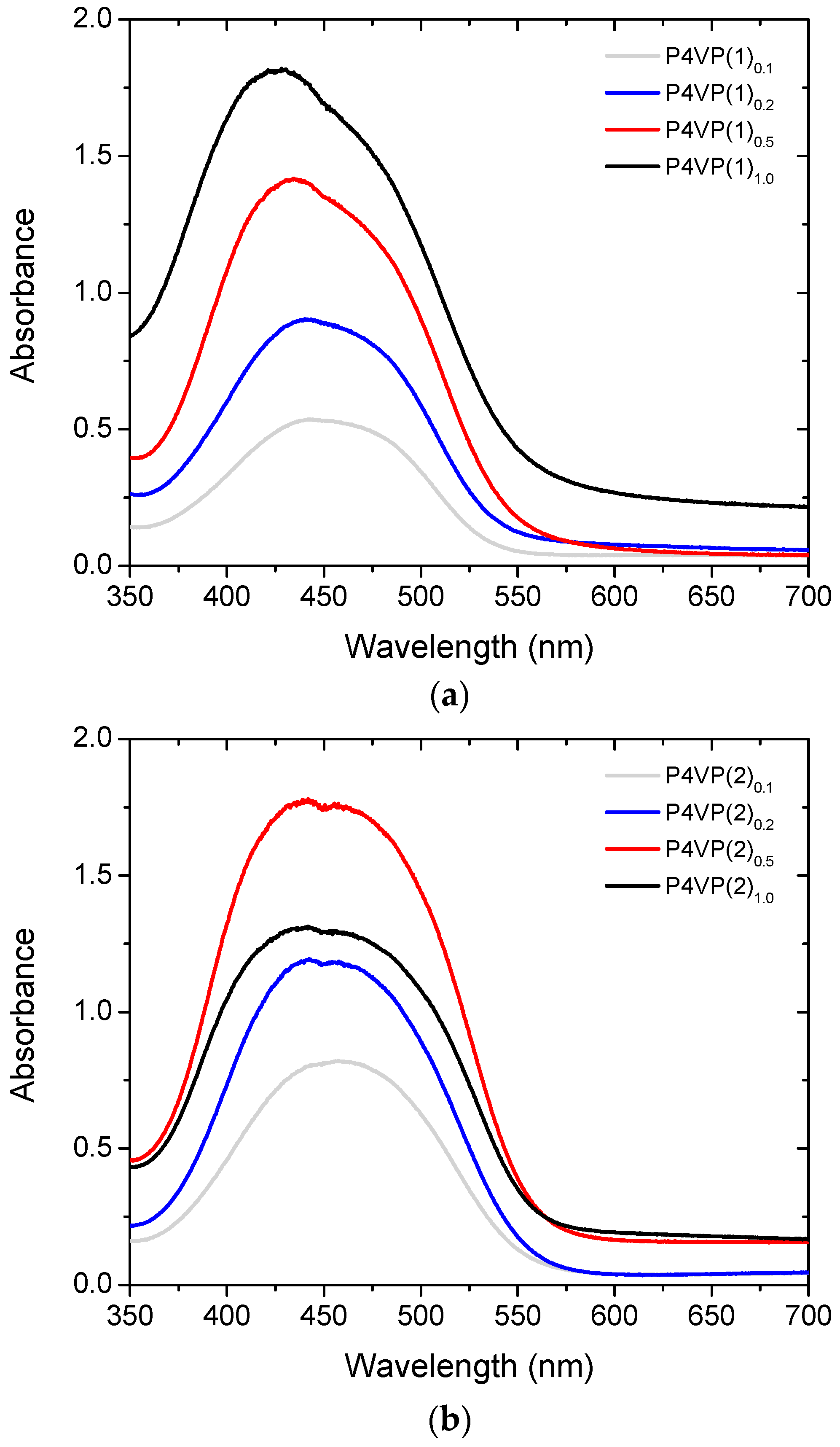
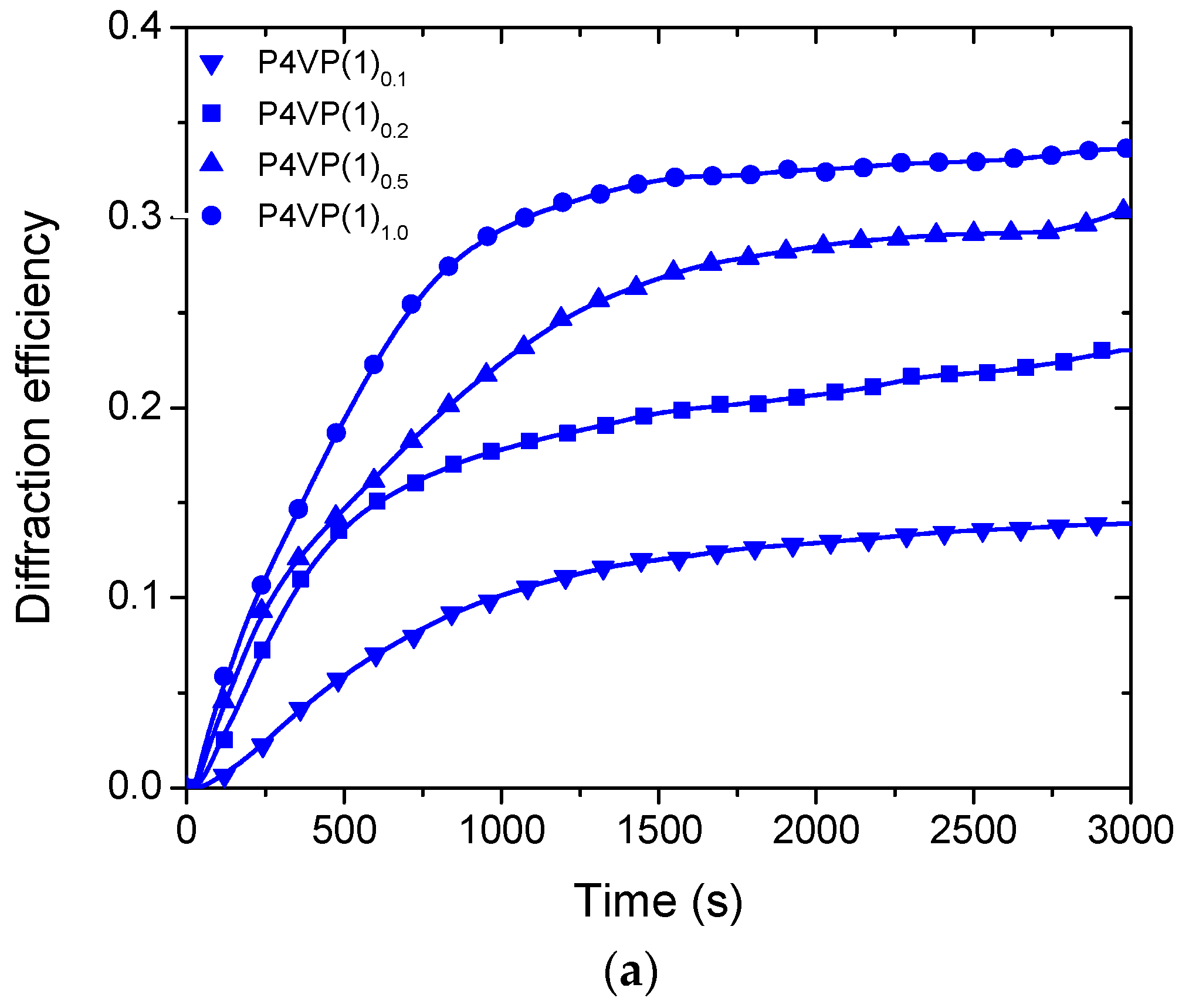
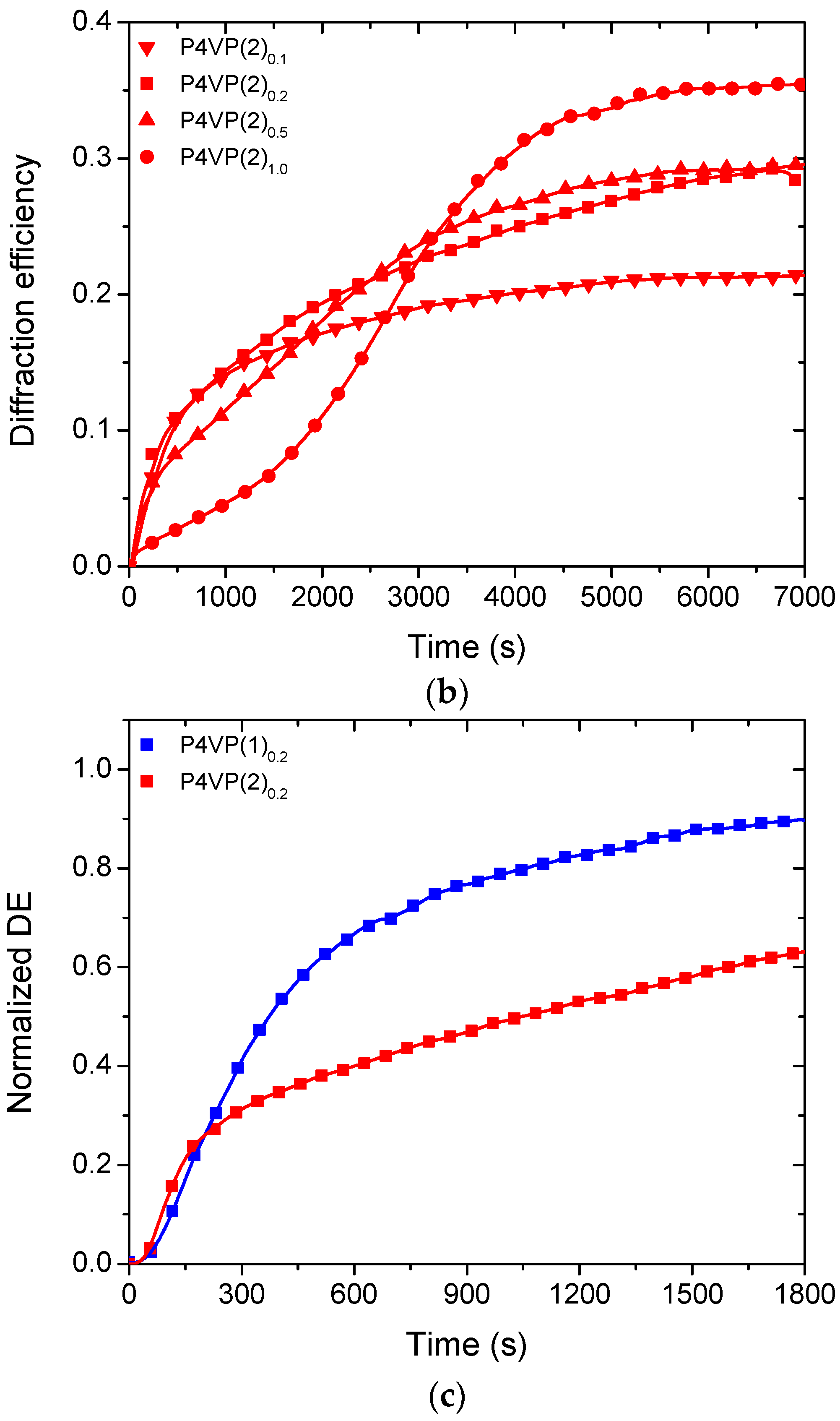
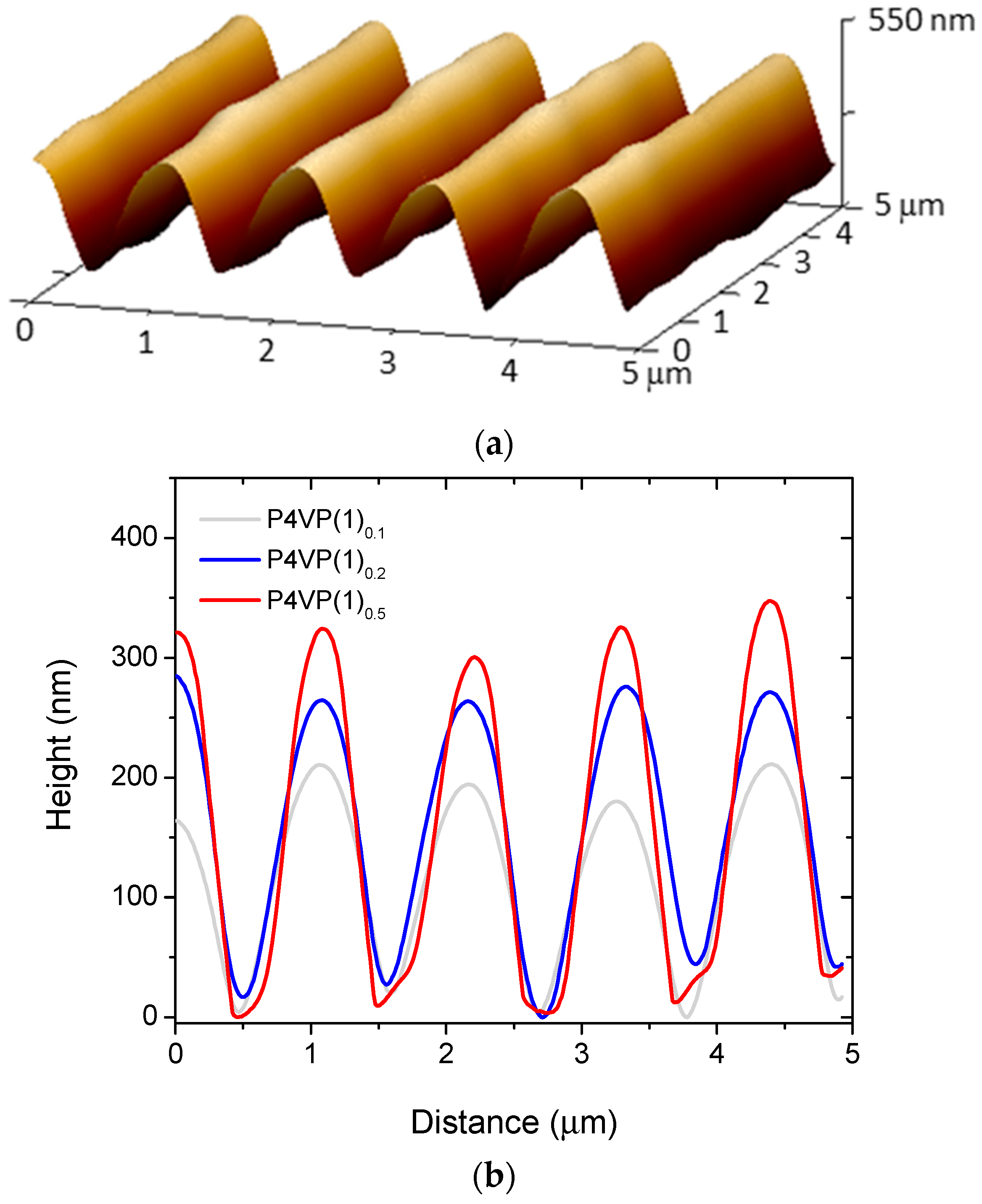
2. Results and Discussion
3. Materials and Methods
4. Conclusions and Outlook
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Zhao, Y.; Ikeda, T. Smart Light-Responsive Materials: Azobenzene-Containing Polymers and Liquid Crystals; Wiley: Hoboken, NJ, USA, 2009. [Google Scholar]
- Rochon, P.; Batalla, E.; Natansohn, A. Optically induced surface gratings on azoaromatic polymer films. Appl. Phys. Lett. 1995, 66, 136–138. [Google Scholar] [CrossRef]
- Kim, D.Y.; Tripathy, S.K.; Li, L.; Kumar, J. Laser-induced holographic surface relief gratings on nonlinear optical polymer films. Appl. Phys. Lett. 1995, 66, 1166–1168. [Google Scholar] [CrossRef]
- Ubukata, T.; Isoshima, T.; Hara, M. Wavelength-programmable organic distributed-feedback laser based on a photoassisted polymer-migration system. Adv. Mater. 2005, 17, 1630–1633. [Google Scholar] [CrossRef]
- Vapaavuori, J.; Ras, R.H.A.; Kaivola, M.; Bazuin, C.G.; Priimagi, A. From partial to complete optical erasure of azobenzene-polymer gratings: The effect of molecular weight. J. Mater. Chem. C 2015, 3, 11011–11016. [Google Scholar] [CrossRef]
- Lee, S.; Kang, H.S.; Park, J. Directional photofluidization lithography: Micro/nanostructural evolution by photofluidic motions of azobenzene materials. Adv. Mater. 2012, 24, 2069–2103. [Google Scholar] [CrossRef] [PubMed]
- Priimagi, A.; Shevchenko, A. Azopolymer-based micro- and nanopatterning for photonic applications. J. Polym. Sci. Part B Polym. Phys. 2014, 52, 163–182. [Google Scholar] [CrossRef]
- Toshchevikov, V.; Ilnytskyi, J.; Saphiannikova, M. Photoisomerization kinetics and mechanical stress in azobenzene-containing materials. J. Phys. Chem. Lett. 2017, 8, 1094–1098. [Google Scholar] [CrossRef] [PubMed]
- Juan, M.L.; Plain, J.; Bachelot, R.; Royer, P.; Gray, S.K.; Wiederrecht, G.P. Multiscale model for photoinduced molecular motion in azo polymers. ACS Nano 2009, 3, 1573–1579. [Google Scholar] [CrossRef] [PubMed]
- Toshchevikov, V.; Saphiannikova, M.; Heinrich, G. Microscopic theory of light-induced deformation in amorphous side-chain azobenzene polymers. J. Phys. Chem. B 2009, 113, 5032–5045. [Google Scholar] [CrossRef] [PubMed]
- Fiorini, C.; Prudhomme, N.; De Veyrac, G.; Maurin, I.; Raimond, P.; Nunzi, J.M. Molecular migration mechanism for laser induced surface relief grating formation. Synth. Met. 2000, 115, 121–125. [Google Scholar] [CrossRef]
- Labarthet, F.L.; Buffeteau, T. Analyses of the diffraction efficiencies, birefringence, and surface relief gratings on azobenzene-containing polymer films. J. Phys. Chem. C 1998, 102, 2654–2662. [Google Scholar] [CrossRef]
- Zettsu, N.; Ogasawara, T.; Mizoshita, N.; Nagano, S.; Seki, T. Photo-triggered surface relief grating formation in supramolecular liquid crystalline polymer systems with detachable azobenzene units. Adv. Mater. 2008, 20, 516–521. [Google Scholar] [CrossRef]
- Kulikovska, O.; Goldenberg, L.M.; Stumpe, J. Supramolecular azobenzene-based materials for optical generation of microstructures. Chem. Mater. 2007, 19, 3343–3348. [Google Scholar] [CrossRef]
- Zhang, Q.; Wang, X.; Barrett, C.J.; Bazuin, C.G. Spacer-free ionic dye-polyelectrolyte complexes: Influence of molecular structure on liquid crystal order and photoinduced motion. Chem. Mater. 2009, 21, 3216–3227. [Google Scholar] [CrossRef]
- Wang, X.; Vapaavuori, J.; Wang, X.; Sabat, R.G.; Pellerin, C.; Bazuin, C.G. Influence of supramolecular interaction type on photoresponsive azopolymer complexes: A surface relief grating formation study. Macromolecules 2016, 49, 4923–4934. [Google Scholar] [CrossRef]
- Gao, J.; He, Y.; Liu, F.; Zhang, X.; Wang, Z.; Wang, X. Azobenzene-containing supramolecular side-chain polymer films for laser-induced surface relief gratings. Chem. Mater. 2007, 19, 3877–3881. [Google Scholar] [CrossRef]
- Priimagi, A.; Lindfors, K.; Kaivola, M.; Rochon, P. Efficient surface-relief gratings in hydrogen-bonded polymer–azobenzene complexes. ACS Appl. Mater. Interfaces 2009, 1, 1183–1189. [Google Scholar] [CrossRef] [PubMed]
- Priimagi, A.; Cavallo, G.; Forni, A.; Gorynsztejn-Leben, M.; Kaivola, M.; Metrangolo, P.; Milani, R.; Shishido, A.; Pilati, T.; Resnati, G.; et al. Halogen bonding versus hydrogen bonding in driving self-assembly and performance of light-responsive supramolecular polymers. Adv. Funct. Mater. 2012, 22, 2572–2579. [Google Scholar] [CrossRef]
- Saccone, M.; Dichiarante, V.; Forni, A.; Goulet-Hanssens, A.; Cavallo, G.; Vapaavuori, J.; Terraneo, G.; Barrett, C.J.; Resnati, G.; Metrangolo, P.; et al. Supramolecular hierarchy among halogen and hydrogen bond donors in light-induced surface patterning. J. Mater. Chem. C 2015, 3, 759–768. [Google Scholar] [CrossRef]
- Fukuda, T.; Matsuda, H.; Shiraga, T.; Kimura, T.; Kato, M.; Viswanathan, N.K.; Kumar, J.; Tripathy, S.K. Photofabrication of surface relief grating on films of azobenzene polymer with different dye functionalization. Macromolecules 2000, 33, 4220–4225. [Google Scholar] [CrossRef]
- Vapaavuori, J.; Priimagi, A.; Kaivola, M. Photoinduced surface-relief gratings in films of supramolecular polymer-bisazobenzene complexes. J. Mater. Chem. 2010, 20, 5260–5264. [Google Scholar] [CrossRef]
- Koskela, J.E.; Vapaavuori, J.; Hautala, J.; Priimagi, A.; Faul, C.F.J.; Kaivola, M.; Ras, R.H.A. Surface-relief gratings and stable birefringence inscribed using light of broad spectral range in supramolecular polymer-bisazobenzene complexes. J. Phys. Chem. C 2012, 116, 2363–2370. [Google Scholar] [CrossRef]
- Koskela, J.E.; Vapaavuori, J.; Ras, R.H.A.; Priimagi, A. Light-driven surface patterning of supramolecular polymers with extremely low concentration of photoactive molecules. ACS Macro Lett. 2014, 3, 1196–1200. [Google Scholar] [CrossRef]
- Saccone, M.; Cavallo, G.; Metrangolo, P.; Resnati, G.; Priimagi, A. Halogen-bonded photoresponsive materials. Top. Curr. Chem. 2014, 359, 147–166. [Google Scholar]
- Clark, T.; Hennemann, M.; Murray, J.S.; Politzer, P. Halogen bonding: The σ-hole. J. Mol. Model. 2006, 13, 291–296. [Google Scholar] [CrossRef] [PubMed]
- Desiraju, G.R.; Shing Ho, P.; Kloo, L.; Legon, A.C.; Marquardt, R.; Metrangolo, P.; Politzer, P.; Resnati, G.; Rissanen, K. Definition of the halogen bond. Pure Appl. Chem. 2013, 85, 1711–1713. [Google Scholar] [CrossRef]
- Politzer, P.; Murray, J.S.; Clark, T. Halogen bonding: An electrostatically-driven highly directional noncovalent interaction. Phys. Chem. Chem. Phys. 2010, 12, 7748–7757. [Google Scholar] [CrossRef] [PubMed]
- Cavallo, G.; Metrangolo, P.; Milani, R.; Pilati, T.; Priimagi, A.; Resnati, G.; Terraneo, G. The halogen bond. Chem. Rev. 2016, 116, 2478–2601. [Google Scholar] [CrossRef] [PubMed]
- Aakeröy, C.B.; Fasulo, M.; Schultheiss, N.; Desper, J.; Moore, C. Structural competition between hydrogen bonds and halogen bonds. J. Am. Chem. Soc. 2007, 129, 13772–13773. [Google Scholar] [CrossRef] [PubMed]
- Chudzinski, M.G.; McClary, C.A.; Taylor, M.S. Anion receptors composed of hydrogen- and halogen-bond donor groups: Modulating selectivity with combinations of distinct noncovalent interactions. J. Am. Chem. Soc. 2011, 133, 10559–10567. [Google Scholar] [CrossRef] [PubMed]
- Barrett, C.J.; Natansohn, A.L.; Rochon, P.L. Mechanism of optically inscribed high-efficiency diffraction gratings in azo polymer films. J. Phys. Chem. 1996, 100, 8836–8842. [Google Scholar] [CrossRef]
- Vapaavuori, J.; Valtavirta, V.; Alasaarela, T.; Mamiya, J.-I.; Priimagi, A.; Shishido, A.; Kaivola, M. Efficient surface structuring and photoalignment of supramolecular polymer–azobenzene complexes through rational chromophore design. J. Mater. Chem. 2011, 21, 15437–15441. [Google Scholar] [CrossRef]
- Vapaavuori, J.; Heikkinen, I.T.S.; Dichiarante, V.; Resnati, G.; Metrangolo, P.; Sabat, R.G.; Bazuin, C.G.; Priimagi, A.; Pellerin, C. Photomechanical energy transfer to photopassive polymers through hydrogen and halogen bonds. Macromolecules 2015, 48, 7535–7542. [Google Scholar] [CrossRef]
- Ten Brinke, G.; Ikkala, O. Smart materials based on self-assembled hydrogen-bonded comb-shaped supramolecules. Chem. Rec. 2004, 4, 219–230. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Y.; Thorkelsson, K.; Mastroianni, A.J.; Schilling, T.; Luther, J.M.; Rancatore, B.J.; Matsunaga, K.; Jinnai, H.; Wu, Y.; Poulsen, D.; et al. Small-molecule-directed nanoparticleassembly towards stimuli-responsive nanocomposites. Nat. Mater. 2009, 8, 979–985. [Google Scholar] [CrossRef] [PubMed]
- Milani, R.; Houbenov, N.; Fernandez-Palacio, F.; Cavallo, G.; Luzio, A.; Haataja, J.; Giancane, G.; Saccone, M.; Priimagi, A.; Metrangolo, P.; et al. Hierarchical self-assembly of halogen-bonded block copolymer complexes into upright cylindrical domains. Chem 2017, 2, 417–426. [Google Scholar] [CrossRef] [PubMed]
- Salonen, L.M.; Ellermann, M.; Diederich, F. Aromatic rings in chemical and biological recognition: Energetics and structures. Angew. Chem. Int. Ed. 2011, 50, 4808–4842. [Google Scholar] [CrossRef] [PubMed]
- Patrick, C.R.; Prosser, G.S. A molecular complex of benzene and hexafluorobenzene. Nature 1960, 187, 1021. [Google Scholar] [CrossRef]
- Řezáč, J.; Riley, K.E.; Hobza, P. Benchmark calculations of noncovalent interactions of halogenated molecules. J. Chem. Theory Comput. 2012, 8, 4285–4292. [Google Scholar] [CrossRef] [PubMed]
- Brown, D.; Natansohn, A.; Rochon, P. Azo Polymers for Reversible Optical Storage. 5. Orientation and Dipolar Interactions of Azobenzene Side Groups in Copolymers and Blends Containing Methyl Methacrylate Structural Units. Macromolecules 1995, 28, 6116–6123. [Google Scholar] [CrossRef]
- Iftime, G.; Fisher, L.; Natansohn, A.; Rochon, P. Photoinduced birefringence in copolymers containing Disperse Red 1 and styrene. Can. J. Chem. 2000, 78, 409–414. [Google Scholar] [CrossRef]
- Virkki, M.; Kauranen, M.; Priimagi, A. Different chromophore concentration dependence of photoinduced birefringence and second-order susceptibility in all-optical poling. Appl. Phys. Lett. 2011, 99, 183309. [Google Scholar] [CrossRef]
- Priimagi, A.; Shevchenko, A.; Kaivola, M.; Rodriguez, F.J.; Kauranen, M.; Rochon, P. High and stable photoinduced anisotropy in guest–host polymer mediated by chromophore aggregation. Opt. Lett. 2010, 35, 1813–1815. [Google Scholar] [CrossRef] [PubMed]
- Kim, D.Y.; Li, L.; Jiang, X.L.; Shivshankar, V.; Kumar, J.; Tripathy, S.K. Polarized laser induced holographic surface relief gratings on polymer films. Macromolecules 1995, 28, 8835–8839. [Google Scholar] [CrossRef]
- Lagugné-Labarthet, F.; Buffeteau, T.; Sourisseau, C. Inscription of holographic gratings using circularly polarized light: Influence of the optical setup on the birefringence and surface relief grating properties. Appl. Phys. B 2002, 74, 129–137. [Google Scholar] [CrossRef]
Sample Availability: Small quantities of the compounds are available from the authors. |


| Sample | Film Thickness | Diffraction Efficiency | Modulation Depth |
|---|---|---|---|
| P4VP(1)0.1 | 380 nm | 0.14 | 180 nm |
| P4VP(1)0.2 | 300 nm | 0.23 | 245 nm |
| P4VP(1)0.5 | 263 nm | 0.30 | 315 nm |
| P4VP(1)1.0 | 223 nm | 0.34 | 315 nm |
| P4VP(2)0.1 | 397 nm | 0.22 | 330 nm |
| P4VP(2)0.2 | 407 nm | 0.28 | 360 nm |
| P4VP(2)0.5 | 278 nm | 0.29 | 390 nm |
| P4VP(2)1.0 | (330 nm) 1 | 0.35 | (125 nm) 1 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Stumpel, J.E.; Saccone, M.; Dichiarante, V.; Lehtonen, O.; Virkki, M.; Metrangolo, P.; Priimagi, A. Surface-Relief Gratings in Halogen-Bonded Polymer–Azobenzene Complexes: A Concentration-Dependence Study. Molecules 2017, 22, 1844. https://doi.org/10.3390/molecules22111844
Stumpel JE, Saccone M, Dichiarante V, Lehtonen O, Virkki M, Metrangolo P, Priimagi A. Surface-Relief Gratings in Halogen-Bonded Polymer–Azobenzene Complexes: A Concentration-Dependence Study. Molecules. 2017; 22(11):1844. https://doi.org/10.3390/molecules22111844
Chicago/Turabian StyleStumpel, Jelle E., Marco Saccone, Valentina Dichiarante, Ossi Lehtonen, Matti Virkki, Pierangelo Metrangolo, and Arri Priimagi. 2017. "Surface-Relief Gratings in Halogen-Bonded Polymer–Azobenzene Complexes: A Concentration-Dependence Study" Molecules 22, no. 11: 1844. https://doi.org/10.3390/molecules22111844
APA StyleStumpel, J. E., Saccone, M., Dichiarante, V., Lehtonen, O., Virkki, M., Metrangolo, P., & Priimagi, A. (2017). Surface-Relief Gratings in Halogen-Bonded Polymer–Azobenzene Complexes: A Concentration-Dependence Study. Molecules, 22(11), 1844. https://doi.org/10.3390/molecules22111844





