UPLC-PDA-Q/TOF-MS Profile of Polyphenolic Compounds of Liqueurs from Rose Petals (Rosa rugosa)
Abstract
:1. Introduction
2. Results and Discussion
2.1. Polyphenolic Compounds in the Tested Rosa rugosa Petals
2.2. Polyphenolic Compounds in the Liqueurs Obtained From Rosa rugosa Petals
3. Experiment
3.1. Chemicals
3.2. Plant Material
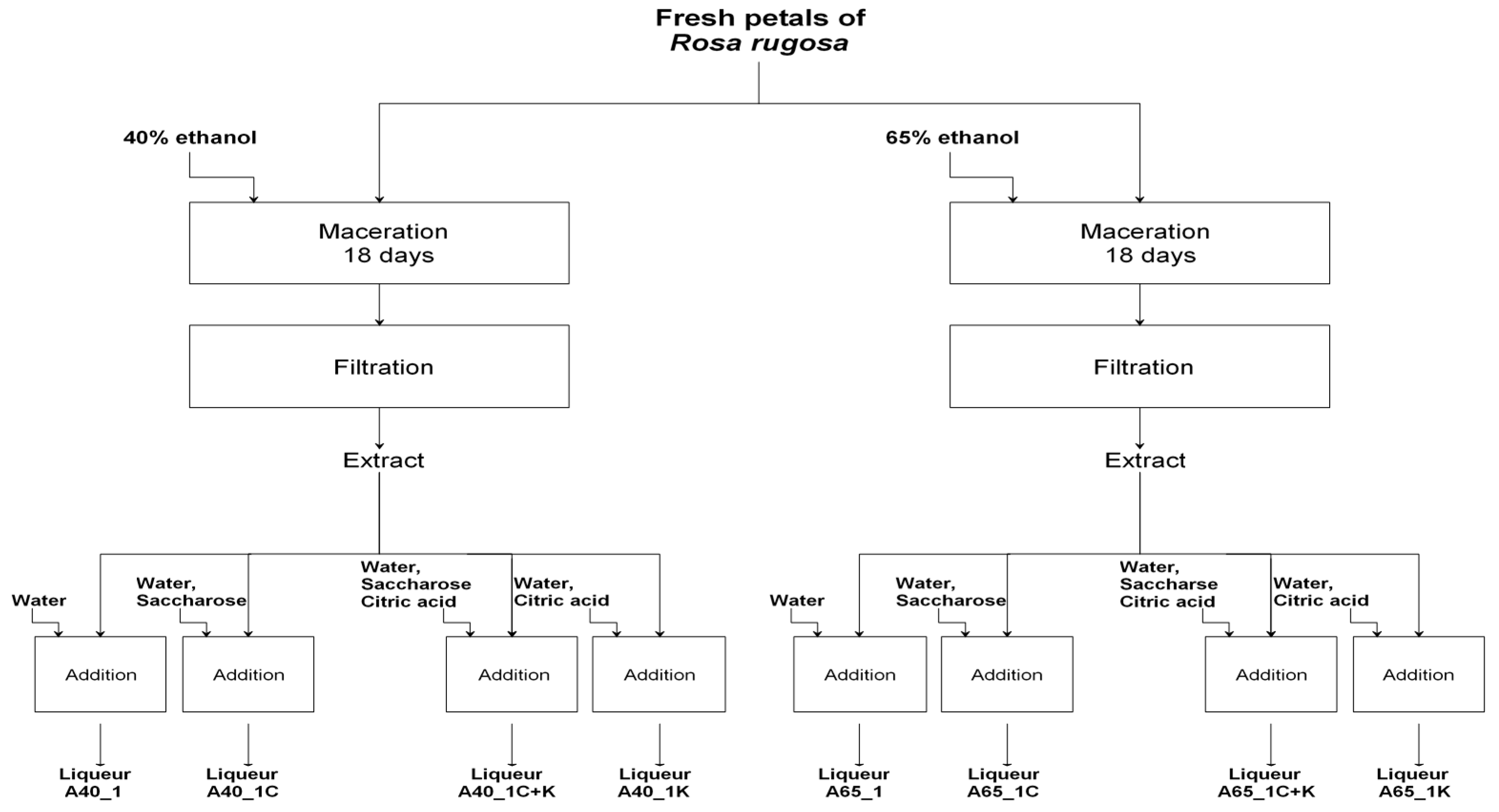
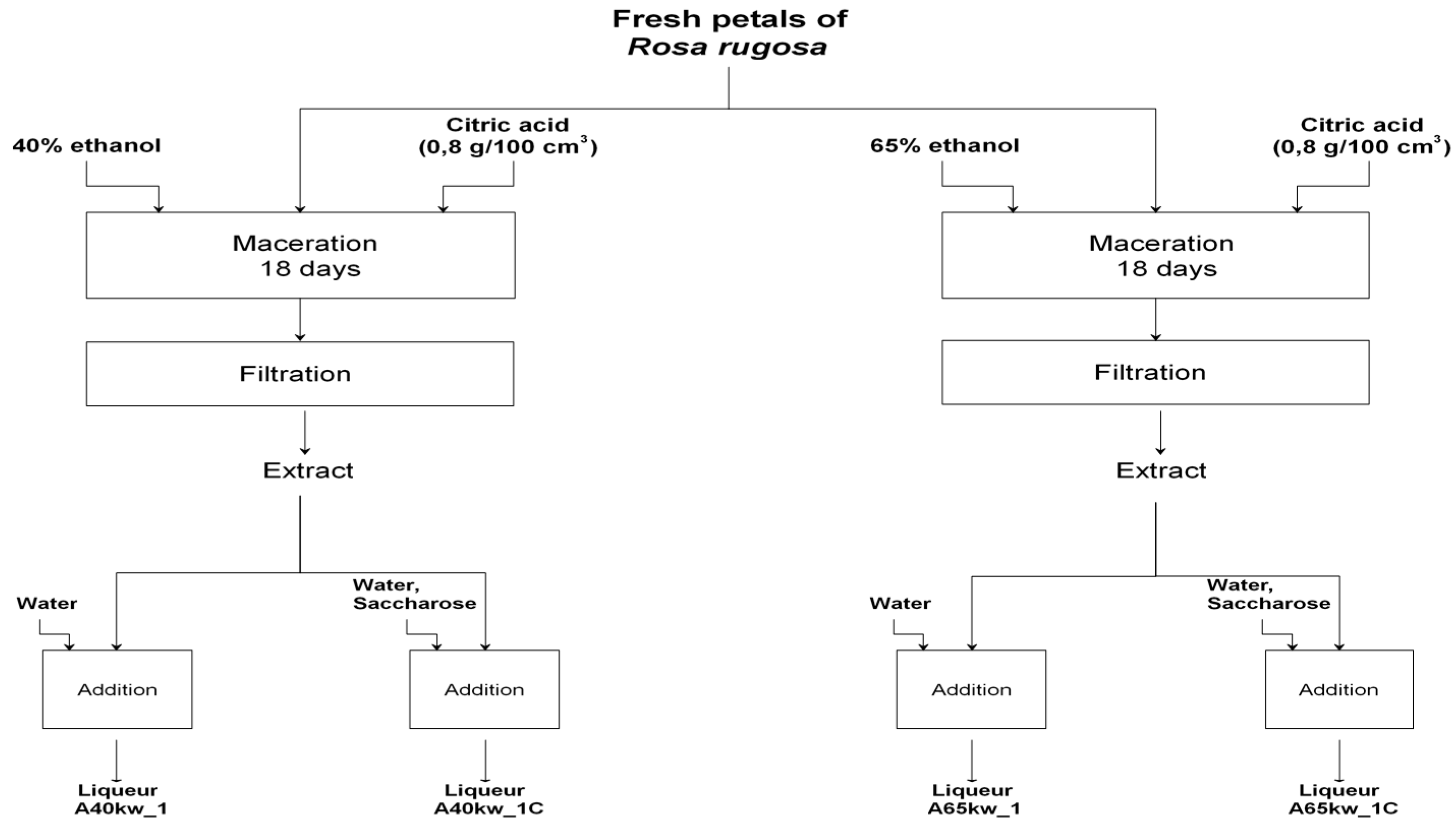
3.3. Liqueurs Preparation
3.4. Qualitative and Quantitative Analysis of Polyphenolic Compounds by UPLC-PDA-ESI-QTOF-MS
3.5. Method Validation
3.5.1. Linearity
3.5.2. LODs and LOQs
3.5.3. Precision, Repeatability and Stability
3.6. Statistical Analysis
4. Conclusions
Author Contributions
Conflicts of Interest
References
- Sanches-Rangel, J.C.; Benavides, J.; Heredia, J.B.; Cisneros-Zevallos, L.; Jacobo-Velazquez, D.A. The Folin—Ciocalteu assay revisited: Improvement of its specificity for total phenolic content determination. Anal. Methods 2013, 5, 5990–5999. [Google Scholar] [CrossRef]
- Päivärinta, E.; Pajari, A.-M.; Torronen, R.; Mutanen, M. Ellagic acid and natural sources of ellagitannins as possible chemopreventive agents against intestinal tumorigenesis in the min mouse. Nutr. Cancer 2006, 54, 79–83. [Google Scholar] [CrossRef] [PubMed]
- Tokarska-Guzik, B. The expansion of some alien plant species (neophytes) in Poland. In Plant Invasions: Ecological Treats and Management Solutions; Child, L.E., Brock, J.H., Brundu, G., Prach, K., Pysek, P., Wade, P.M., Wiliamson, M., Eds.; Backhuys Publishers: Leiden, The Netherlands, 2003; pp. 147–167. [Google Scholar]
- Hashidoko, Y. The phytochemistry of Rosa rugose. Phytochemistry 1996, 43, 535–549. [Google Scholar] [CrossRef]
- Jung, H.J.; Nam, J.H.; Choi, J.; Lee, K.T.; Park, H.J. 19α-hydroxyursane-type triterpenoids: Antinociceptive anti-inflammatory principles of the roots of Rosa rugosa. Biol. Pharm. Bull. 2005, 28, 101–104. [Google Scholar] [CrossRef] [PubMed]
- Razungles, A.; Oszmiański, J.; Sapis, J.-C. Determination of carotenoids in fruits of Rosa sp. (Rosa canina and Rosa rugosa) and of chokeberry (Aronia melanocarpa). J. Food Sci. 1989, 54, 774–775. [Google Scholar] [CrossRef]
- Ochir, S.; Park, B.; Nishizawa, M.; Kanazawa, T.; Funaki, M.; Yamagishi, T. Simultaneous determination of hydrolysable tannins in the petals of Rosa rugosa and allied plants. J. Nat. Med. 2010, 64, 383–387. [Google Scholar] [CrossRef] [PubMed]
- Asres, K.; Seyoum, A.; Veeresham, C.; Bucar, F.; Gibbons, S. Naturally derived anti-HIV agents. Phytother. Res. 2005, 19, 557–581. [Google Scholar] [CrossRef] [PubMed]
- Fu, M.; Ng, T.B.; Jiang, Y.; Pi, Z.F.; Liu, Z.K.; Li, L.; Liu, F. Compounds from rose (Rosa rugosa) flowers with human immunodeficiency virus type 1 reverse transcriptase inhibitory activity. J. Pharm. Pharmacol. 2006, 58, 1275–1280. [Google Scholar] [CrossRef] [PubMed]
- Ng, T.B.; He, J.S.; Niu, S.M.; Zhao, L.; Pi, Z.F.; Shao, W.; Liu, F. A gallic acid derivative and polysaccharides with antioxidant activity from the rose (Rosa rugosa) flowers. J. Pharm. Pharmacol. 2004, 56, 537–545. [Google Scholar] [CrossRef] [PubMed]
- Paixão, N.; Perestrelo, R.; Marques, J.C.; Câmara, J.S. Relationship between antioxidant capacity and total phenolic content of red, rose and white wines. Food Chem. 2007, 105, 204–214. [Google Scholar] [CrossRef]
- Mikanagi, Y.; Saito, N.; Yokoi, M.; Tatsuzawa, F. Anthocyanins in flower of genus rosa, sections Cinnamomeae (= Rosa), Chinenses, Gallicanae and some modern garden roses. Biochem. Syst. Ecol. 2000, 28, 887–902. [Google Scholar] [CrossRef]
- Nowak, R.; Olech, M.; Pecio, Ł.; Oleszek, W.; Los, R.; Malm, A.; Rzymowska, J. Cytoxic, antioxidant, antimicrobial properties and chemical composition of rose petals. J. Sci. Food Agric. 2014, 94, 560–567. [Google Scholar] [CrossRef] [PubMed]
- Liu, L.; Tang, D.; Zhao, H.; Xin, X.; Aisa, H.A. Hypoglycemic effect of the polyphenols rich extract from Rose rugosa Thunb on high fat diet and STZ induced diabetic rats. J. Ethnopharmacol. 2017, 200, 174–181. [Google Scholar] [CrossRef] [PubMed]
- Ng, T.B.; Gao, W.; Li, L.; Niu, S.M.; Zhao, L.; Liu, J.; Shi, L.S.; Fu, M.; Liu, F. Rose (Rosa rugosa)—Flower extract increases the activities of antioxidant enzymes and their gene expression and reduces lipid peroxidation. Biochem. Cell Biol. 2005, 83, 78–85. [Google Scholar] [CrossRef] [PubMed]
- Cho, E.J.; Yokozawa, T.; Rhyu, D.Y.; Kim, H.Y. The inhibitory effects of 12 medicinal plants and their component compounds on lipid peroxidation. Am. J. Chin. Med. 2003, 31, 907–917. [Google Scholar] [CrossRef] [PubMed]
- Assunção, M.; Santos-Marques, M.J.; de Freitas, V.; Carvalho, F.; Andrade, J.P.; Lukoyanov, N.V.; Paula-Barbosa, M.M. Red wine antioxidants protect hippocampal neurons against ethanol-induced damage: A biochemical, morphological and behavioral study. Neuroscience 2007, 146, 1581–1592. [Google Scholar] [CrossRef] [PubMed]
- Imark, C.; Kneubühl, M.; Bodmer, S. Occurrence and activity of natural antioxidants in herbal spiritus. Innov. Food Sci. Emerg. 2000, 1, 239–243. [Google Scholar] [CrossRef]
- Landbo, A.K.; Meyer, A.S. Enzyme—Assisted extraction of antioxidative phenols from black currant juice press residues (Ribas nigrum). J. Agric. Food Chem. 2001, 49, 3169–3177. [Google Scholar] [CrossRef] [PubMed]
- Li, F.; Mao, Y.D.; Wang, Y.F.; Raza, A.; Qiu, L.P.; Xu, X.Q. Optimisation of ultrasonic-assisted enzymatic extraction conditions for improving total phenolic content, antioxidant and antitumor activities in vitro from Trapa quadrispinosa Roxb. Residues. Molecules 2017, 22, 396. [Google Scholar] [CrossRef] [PubMed]
- Altemimi, A.; Choudhary, R.; Watson, D.G.; Lightfoot, D.A. Effects of ultrasonic treatments on the polyphenol and antioxidant content of spinach extracts. Ultrason. Sonchem. 2015, 24, 247–255. [Google Scholar] [CrossRef] [PubMed]
- Dranca, F.; Oroian, M. Optimization of ultrasound-assisted extraction of total monomeric anthocyanin (TMA) and total phenolic content (TPC) from eggplant (Solanummelongena L.) peel. Ultrason. Sonchem. 2016, 31, 637–646. [Google Scholar] [CrossRef] [PubMed]
- Vinatoru, M.; Mason, T.J.; Calinescu, I. Ultrasonically assisted extraction (UAE) and microwave assisted extraction (MAE) of functional compounds from plant materials. TRAC Trends Anal. Chem. 2017, 97, 159–178. [Google Scholar] [CrossRef]
- Yeo, J.; Shahidi, F. Effect of hydrothermal processing on changes of insoluble-bound phenolics of lentils. J. Funct. Foods 2017. [Google Scholar] [CrossRef]
- Wanyo, P.; Meeso, N.; Siriamornpun, S. Effects of different treatments on the antioxidant properties and phenolic compounds of rice bran and rice husk. Food Chem. 2014, 157, 457–463. [Google Scholar] [CrossRef] [PubMed]
- Sokół-Łętowska, A.; Kucharska, A.Z.; Wińska, K.; Szumny, A.; Nawirska-Olszańska, A.; Mizgier, P.; Wyspiańska, D. Comosition and antioxidant activity of red fruit liqueurs. Food Chem. 2014, 157, 533–539. [Google Scholar] [CrossRef] [PubMed]
- Wilson, E.; Hagerman, A. Ellagitannins in plant are hydrolysed to release ellagic acid, which conforms a colored product upon reaction with nitrous acid. J. Agric. Food Chem. 1990, 38, 1678–1683. [Google Scholar] [CrossRef]
- Hartzfel, W.P.; Forkner, R.; Hunter, M.D.; Hagerman, A.E. Determination of hydrolizable tannins (Gallotannins and Ellagitannins) after reaction with potassium iodate. J. Agric. Food Chem. 2002, 50, 1785–1790. [Google Scholar] [CrossRef]
- Mochado, T.B.; Leal, I.C.R.; Amaral, A.C.F.; Santos, K.R.N.; da Silva, M.G.; Kuster, R.M. Antimicrobial ellagitannins of Punica granatum fruits. J. Braz. Chem. Soc. 2002, 13, 606–610. [Google Scholar] [CrossRef]
- Andrade, P.; Ferreres, F.; Gil, M.I. Determination of phenolic compounds in honeys with different floralorigin by capillary zone electrophoresis. Food Chem. 1997, 60, 79–84. [Google Scholar] [CrossRef]
- Doussot, F.; Pardon, P.; Dedier, J.; De Jesc, B. Individual, species and geographic origin influence on cooperage oak extractible content (Qersus robur L. and Quersus petrea Liebl). Analusis 2000, 28, 960–965. [Google Scholar] [CrossRef]
- Lee, J.H.; Talcott, S.T. Ellagic acid and ellagitannins affect on sedimentation in muscadine juice and wine. J. Agric. Food Chem. 2002, 50, 3971–3976. [Google Scholar] [CrossRef] [PubMed]
- Määttä, K.R.; Kamal-Eldin, A.; Törrönen, A.R. HPLC analysis of phenolic compounds in berries with diode array and electrospray ionization MS detection ribes species. J. Agric. Food Chem. 2003, 51, 6736–6744. [Google Scholar] [CrossRef] [PubMed]
- Teixeira Lde, L.; Bertoldi, F.C.; Lajolo, F.M.; Hassimotto, N.M. Identification of ellagitannins and flavonoids from Eugenia brasilienses Lam. (Grumixama) by HPLC-ESI-MS/MS. J. Agric. Food Chem. 2015, 63, 5417–5427. [Google Scholar] [CrossRef] [PubMed]
- Schmidt, J. Negative ion electrospray high-resolution tandem mass spectrometry of polyphenols. Mass Spectrom. 2016, 51, 33–43. [Google Scholar] [CrossRef] [PubMed]
- Marzouk, M.S.; Moharram, F.A.; Gamal-Eldeen, A.; Damlakhy, I.M. Spectroscopic Identifi cation of New Ellagitannins and a Trigalloylglucosylkaempferol from an Extract of Euphorbia cotinifolia L. with Antitumour and Antioxidant Activity. Z. Naturforsch. 2012, 67, 151–162. [Google Scholar]
- Ga Hee, J.; Heon Woong, K.; Min Ki, L.; So Young, J.; Ram, B.A.; Dong Jin, L.; Jung Bong, K. Characterization and quantification of flavonoid glycosides in the Prunus genus by UPLC-DAD-QTOF/MS. Saudi J. Biol. Sci. 2016. [Google Scholar] [CrossRef]
- Borràs, E.; Ferré, J.; Boqué, R.; Mestres, M.; Aceña, L.; Busto, O. Data fusion methodologies for food and beverage authentication and quality assessment—A review. Anal. Chim. Acta 2015, 891, 1–14. [Google Scholar]
- Ajila, C.M.; Brar, S.K.; Verma, M.; Tyagi, R.D.; Godbout, S.; Valéro, J.R. Extraction and analysis of polyphenols: Recent trends. Crit. Rev. Biotechnol. 2011, 31, 227–249. [Google Scholar] [CrossRef] [PubMed]
- Panche, A.N.; Diwan, A.D.; Chandra, S.R. Flavonoids: An overview. J. Nutr. Sci. 2016, 5, 1–15. [Google Scholar] [CrossRef] [PubMed]
- Nowak, R. Determination of ellagic acid in pseudofruits of some species of roses. Acta Pol. Pharm. 2006, 63, 289–292. [Google Scholar] [PubMed]
- Teleszko, M.; Wojdyło, A.; Oszmiański, J. Zawartość kwasu elagowego i spolimeryzowanych proantocyjanidyn w pseudoowocach wybranych gatunków róż. Zywn. Nauka Technol. Ja. 2012, 5, 37–46. [Google Scholar]
- Kumar, N.; Bhandari, P.; Singh, B.; Bari, S.S. Antioxidant activity and ultra-performance LC-electrospray ionization-quadrupole time-of-flight mass spectrometry for phenolics-based fingerprinting of Rose species: Rosa damascena, Rosa bourboniana and Rosa brunonii. Food Chem. Toxicol. 2009, 47, 361–367. [Google Scholar] [CrossRef] [PubMed]
- Schmitzer, V.; Veberic, R.; Osterc, G.; Stampar, F. Changes in the phenolic concentration durin flower development of rose ‘KORcrisett’. J. Am. Soc. Hort. Sci. 2009, 134, 491–496. [Google Scholar]
- Velioglu, Y.S.; Mazza, G. Characterization of flavonoides in petals of Rosa damascena by HPLC and spectral analysis. J. Agric. Food Chem. 1991, 39, 463–467. [Google Scholar] [CrossRef]
- Pinelo, M.; Rubilar, M.; Sineiro, J.; Nuńez, M.J. A thermal treatment to increase the antioxidant capacity of natural phenols: Catechine and grape extract Cases. Eur. Food Res. Technol. 2005, 221, 284–290. [Google Scholar] [CrossRef]
- Montoro, P.; Tuberoso, C.I.; Piacente, S.; Perrone, A.; De Feo, V.; Cabras, P.; Pizza, C. Stability and antioxidant activity of polyphenols in extracts of Myrtus communis L. berries used for the preparation of myrtle liqueur. J. Pharmaceut. Biomed. 2006, 41, 1614–1619. [Google Scholar] [CrossRef] [PubMed]
- Alamprese, C.; Pompei, C. Influence of processing variables on some characteristic of nocino liqueur. Food Chem. 2005, 92, 203–209. [Google Scholar] [CrossRef]
- Kucharska, A.Z.; Sokół-Łętowska, A.; Hudko, J.; Nawirska-Olszańska, A. Influence of the preparation procedure on the antioxidant activity and color of liqueurs from cornelian cherry (Cornus mas L.). J. Nutr. Health Food Sci. 2007, 57, 343–347. [Google Scholar]
- Latti, A.K.; Jaakola, L.; Riihinen, K.R.; Kainulainen, P.S. Anthocyanin and flavonol variation in bog bilberries (Vaccinium uliginosum L.) in Finland. J. Agric. Food Chem. 2010, 58, 427–433. [Google Scholar] [CrossRef] [PubMed]
- Mohdaly, A.A.A.; Sarhan, M.A.; Smetanska, I.; Mahmound, A. Antioxidant properties of various solvent extracts of potato peels, sugar beet pulp and sesame cake. J. Sci. Food Agric. 2010, 90, 218–226. [Google Scholar] [CrossRef] [PubMed]
- Singh, A.P.; Wilson, T.; Luthria, D.; Freeman, M.R.; Scott, R.M.; Bilenker, D.; Shaha, S.; Somasundaram, S.; Vorsa, N. LC-MS-MS characterisation of curry leaf flavonols and antioxidant activity. Food Chem. 2011, 127, 80–85. [Google Scholar] [CrossRef]
- Metivier, R.P.; Francis, F.J.; Clydesdale, F.M. Solvent extraction of anthocyanins from wine pomace. J. Food Sci. 1980, 45, 1099–1100. [Google Scholar] [CrossRef]
- Oszmiański, J.; Kolniak-Ostek, J.; Wojdyło, A. Application of ultra performance liquid chromatography-photodiode detector-quadrupole/time of flight-mass spectrometry (UPLC-PDA-Q/TOF-MS) method for the characterization of phenolic compounds of Lepidium sativum L. sprouts. Eur. Food Res. Technol. 2013, 236, 699–706. [Google Scholar] [CrossRef]
Sample Availability: Samples of the compounds are not available from the authors. |
| Compound | tR UPLC-MS (min) | [M − H]− (m/z) | [MS2] (m/z) | Content (mg/100 g FW) |
|---|---|---|---|---|
| Flavonols | ||||
| Myricetin 3,5-di-O-glucoside | 4.07 | 641.1755 | 479.1206; 317.0670 | 82.00 ± 2.54 |
| Quercetin 3,4-di-O-glucoside | 5.85 | 625.1386 | 300.0277 | 158.62 ± 13.80 |
| Kaempferol 3,4-di-O-glucoside | 6.75 | 609.1432 | 284.0348 | 40.56 ± 0.58 |
| Quercetin 3-O-glucosyl-xyloside | 6.84 | 595.1262 | 300.0277 | 100.63 ± 1.12 |
| Isorhamnetin 3-O-glucoside | 7.36 | 477.1022 | 315.0476; 271.0413 | 6.85 ± 0.05 |
| Unknown quercetin derivatives | 7.58 | 1087.0920 | 301.0354 | 10.28 ± 0.18 |
| Kaempferol 3,7-di-O-rhamnoside | 7.96 | 579.1329 | 284.0313 | 6.42 ± 0.00 |
| Quercetin 3-O-rhamnoside | 8.5 | 447.0916 | 300.0277 | 7.33 ± 0.20 |
| Flavan-3-ols | ||||
| (+)-catechin | 3.19 | 289.0688 | 169.0136 | 177.60 ± 6.22 |
| Phenolic acids | ||||
| Ellagic acid a | 6.63 | 300.9999 | – | 47.30 ± 6.58 |
| Hydrolysable tannins | ||||
| Ellagitannins | ||||
| Sanguine H-2 | 5.33 | 1103.0829 | 935.0815; 300.9999 | 165.45 ± 4.60 |
| Ellagitannin 1 | 5.42 | 860.0810 | 785.0868; 300.9999 | 72.57 ± 2.60 |
| Ellagitannin 2 | 5.79 | 860.0870 | 785.0868; 300.9999 | 21.41 ± 1.61 |
| Ellagitannin 3 | 5.89 | 937.0917 | 465.0684; 300.9999 | 1071.81 ± 29.30 |
| Ellagitannin 4 | 6.19 | 1105.1028 | 1061.1268; 300.9999 | 185.77 ± 1.30 |
| Gallotannins | ||||
| Isomer galloyl-bis-HHDP glucose | 6.36 | 935.0815 | 433.0352; 300.9999 | 20.83 ± 7.58 |
| Sum of phenolic compounds | 2175.43 | |||
| Compound (mg/100 cm3) | Liqueurs Code | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| A40_1 | A40_1K | A40_1C | A40_1C + K | A40kw_1 | A40kw_1C | |||||||
| Time of Seasoning (Days) | ||||||||||||
| 0 | 120 | 0 | 120 | 0 | 120 | 0 | 120 | 0 | 120 | 0 | 120 | |
| Myricetin 3,5-di-O-glucoside | 4.21 ± 1.11 | 4.03 ± 0.12 | 4.51 ± 0.71 | 4.40 ± 0.68 | 4.48 ± 0.66 | 4.27 ± 0.97 | 4.65 ± 0.64 | 4.33 ± 1.12 | 4.85 ± 0.80 | 4.63 ± 0.36 | 4.90 ± 0.49 | 4.85 ± 0.53 |
| Quercetin 3,4-di-O-glucoside | 8.31 ± 0.40 | 7.98 ± 0.71 | 8.92 ± 0.95 | 8.65 ± 1.30 | 8.82 ± 0.17 | 8.48 ± 0.22 | 8.90 ± 0.47 | 8.37 ± 0.18 | 9.00 ± 0.38 | 8.85 ± 0.75 | 9.05 ± 0.75 | 8.65 ± 0.64 |
| Kaempferol 3,4-di-O-glucoside | 1.75 ± 0.12 | 1.58 ± 0.46 | 2.22 ± 0.07 | 2.19 ± 0.19 | 2.16 ± 0.02 | 2.07 ± 0.27 | 2.21 ± 1.08 | 2.15 ± 0.16 | 2.35 ± 0.11 | 2.25 ± 0.56 | 2.36 ± 0.05 | 2.18 ± 0.06 |
| Quercetin 3-O-glucosyl-xyloside | 5.21 ± 0.01 | 4.79 ± 0.12 | 5.44 ± 0.12 | 5.27 ± 0.30 | 5.50 ± 0.11 | 5.23 ± 0.10 | 5.45 ± 0.19 | 5.22 ± 0.36 | 5.80 ± 0.92 | 5.59 ± 0.11 | 5.75 ± 0.29 | 5.65 ± 0.29 |
| Isorhamnetin 3-O-glucoside | 0.24 ± 0.03 | 0.16 ± 0.04 | 0.30 ± 0.06 | 0.28 ± 0.07 | 0.30 ± 0.00 | 0.27 ± 0.07 | 0.29 ± 0.21 | 0.25 ± 0.02 | 0.29 ± 0.53 | 0.27 ± 0.00 | 0.30 ± 0.03 | 0.29 ± 0.03 |
| Unknown quercetin derivatives | 0.51 ± 0.12 | 0.48 ± 0.13 | 0.55 ± 0.10 | 0.50 ± 0.06 | 0.55 ± 0.04 | 0.52 ± 0.03 | 0.57 ± 0.11 | 0.52 ± 0.20 | 0.52 ± 0.00 | 0.48 ± 0.02 | 0.50 ± 0.08 | 0.18 ± 0.08 |
| Kaempferol 3,7-di-O-rhamnoside | 0.20 ± 0.05 | 0.18 ± 0.04 | 0.28 ± 0.05 | 0.27 ± 0.02 | 0.28 ± 0.02 | 0.25 ± 0.05 | 0.28 ± 0.01 | 0.25 ± 0.03 | 0.30 ± 0.03 | 0.29 ± 0.01 | 0.30 ± 0.07 | 0.28 ± 0.07 |
| Quercetin 3-O-rhamnoside | 0.33 ± 0.09 | 0.30 ± 0.01 | 0.36 ± 0.06 | 0.33 ± 0.10 | 0.38 ± 0.03 | 0.36 ± 0.16 | 0.39 ± 0.07 | 0.36 ± 0.06 | 0.32 ± 0.11 | 0.30 ± 0.07 | 0.29 ± 0.00 | 0.28 ± 0.01 |
| (+)-catechin | 8.22 ± 0.36 | 7.54 ± 0.36 | 8.50 ± 0.89 | 8.35 ± 1.22 | 8.50 ± 0.20 | 8.29 ± 0.22 | 8.45 ± 0.72 | 8.21 ± 0.42 | 10.03 ± 1.27 | 9.95 ± 0.92 | 10.07 ± 1.01 | 9.98 ± 0.49 |
| Sanguine H-2 | - | - | - | - | - | - | - | - | 9.55 ± 0.29 | 9.20 ± 0.75 | 9.60 ± 0.15 | 9.50 ± 0.24 |
| Ellagitannin 1 | - | - | - | - | - | - | - | - | 2.40 ± 0.06 | 2.25 ± 0.07 | 2.61 ± 0.23 | 2.53 ± 0.32 |
| Ellagitannin 2 | - | - | - | - | - | - | - | - | 1.06 ± 0.23 | 1.00 ± 0.06 | 1.07 ± 0.07 | 1.05 ± 0.23 |
| Ellagitannin 3 | 54.27 ± 3.72 | 51.22 ± 1.22 | 56.43 ± 3.21 | 56.10 ± 5.10 | 56.50 ± 3.11 | 54.83 ± 2.22 | 56.33 ± 1.33 | 54.13 ± 1.06 | 50.72 ± 1.72 | 48.11 ± 2.10 | 51.11 ± 1.11 | 50.51 ± 2.49 |
| Ellagitannin 4 | 5.50 ± 0.70 | 5.07 ± 0.55 | 5.63 ± 0.13 | 5.60 ± 0.66 | 5.65 ± 0.18 | 5.30 ± 0.15 | 5.40 ± 0.31 | 5.27 ± 0.27 | 9.78 ± 0.24 | 9.31 ± 1.04 | 9.85 ± 0.63 | 9.60 ± 0.57 |
| Isomer galloilo-bis-HHDP glucose | 0.21 ± 0.09 | 0.19 ± 0.13 | 0.31 ± 0.14 | 0.28 ± 0.08 | 0.29 ± 0.09 | 0.28 ± 0.14 | 0.28 ± 0.10 | 0.28 ± 0.08 | 1.15 ± 0.32 | 1.08 ± 0.01 | 1.10 ± 0.08 | 1.07 ± 0.27 |
| Ellagic acid | 4.50 ± 0.23 | 4.15 ± 0.11 | 4.68 ± 0.52 | 4.40 ± 0.14 | 4.83 ± 0.40 | 4.58 ± 0.52 | 4.45 ± 0.96 | 4.42 ± 0.24 | 9.70 ± 0.68 | 9.22 ± 0.36 | 9.88 ± 0.75 | 9.60 ± 0.88 |
| Sum of phenolic compounds | 93.46 | 87.67 | 98.13 | 96.62 | 98.24 | 94.73 | 97.65 | 93.76 | 117.82 | 112.78 | 118.74 | 116.20 |
| Compound (mg/100 cm3) | Liqueurs Code | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| A65_1 | A65_1K | A65_1C | A65_1C + K | A65kw_1 | A65kw_1C | |||||||
| Time of Seasoning (Days) | ||||||||||||
| 0 | 120 | 0 | 120 | 0 | 120 | 0 | 120 | 0 | 120 | 0 | 120 | |
| Myricetin 3,5-di-O-glucoside | 4.33 ± 0.42 | 4.15 ± 0.47 | 4.45 ± 1.15 | 4.28 ± 1.87 | 4.55 ± 1.20 | 4.34 ± 0.52 | 4.55 ± 0.60 | 4.23 ± 0.42 | 4.80 ± 0.71 | 4.78 ± 1.19 | 5.10 ± 1.94 | 4.70 ± 1.18 |
| Quercetin 3,4-di-O-glucoside | 8.63 ± 0.46 | 8.27 ± 0.87 | 8.67 ± 0.42 | 8.32 ± 0.60 | 8.87 ± 0.87 | 8.45 ± 0.11 | 8.95 ± 0.58 | 8.35 ± 0.36 | 9.03 ± 0.52 | 8.81 ± 0.29 | 9.10 ± 0.71 | 9.48 ± 0.32 |
| Kaempferol 3,4-di-O-glucoside | 2.29 ± 0.14 | 2.16 ± 0.08 | 2.25 ± 0.85 | 2.19 ± 0.96 | 2.27 ± 0.68 | 2.25 ± 0.64 | 2.28 ± 0.43 | 2.20 ± 0.72 | 2.40 ± 0.36 | 2.37 ± 0.67 | 2.41 ± 0.55 | 2.39 ± 0.09 |
| Quercetin 3-O-glucosyl-xyloside | 5.57 ± 0.45 | 5.31 ± 0.68 | 5.55 ± 0.12 | 5.44 ± 0.83 | 5.67 ± 0.37 | 5.50 ± 0.10 | 5.58 ± 1.42 | 5.40 ± 0.83 | 5.90 ± 0.17 | 5.75 ± 0.85 | 5.85 ± 1.03 | 5.76 ± 0.91 |
| Isorhamnetin 3-O-glucoside | 0.20 ± 0.06 | 0.18 ± 0.07 | 0.21 ± 0.15 | 0.16 ± 0.01 | 0.19 ± 0.01 | 0.17 ± 0.00 | 0.18 ± 0.08 | 0.16 ± 0.00 | 0.35 ± 0.04 | 0.33 ± 0.03 | 0.33 ± 0.00 | 0.30 ± 0.08 |
| Unknown quercetin derivatives | 0.36 ± 0.11 | 0.32 ± 0.03 | 0.36 ± 0.06 | 0.35 ± 0.00 | 0.36 ± 0.04 | 0.34 ± 0.08 | 0.35 ± 0.15 | 0.32 ± 0.07 | 0.50 ± 0.00 | 0.48 ± 0.12 | 0.47 ± 0.24 | 0.43 ± 0.12 |
| Kaempferol 3,7-di-O-rhamnoside | 0.19 ± 0.03 | 0.18 ± 0.05 | 0.20 ± 0.06 | 0.19 ± 0.02 | 0.21 ± 0.07 | 0.20 ± 0.00 | 0.20 ± 0.05 | 0.18 ± 0.11 | 0.33 ± 0.17 | 0.31 ± 0.09 | 0.31 ± 0.07 | 0.28 ± 0.00 |
| Quercetin 3-O-rhamnoside | 0.38 ± 0.17 | 0.36 ± 0.05 | 0.39 ± 0.04 | 0.37 ± 0.11 | 0.38 ± 0.12 | 0.37 ± 0.09 | 0.40 ± 0.06 | 0.38 ± 0.06 | 0.32 ± 0.02 | 0.30 ± 0.00 | 0.30 ± 0.03 | 0.28 ± 0.00 |
| (+)-catechin | 8.59 ± 0.75 | 8.15 ± 0.79 | 8.78 ± 0.28 | 8.38 ± 1.41 | 8.88 ± 1.15 | 8.52 ± 1.32 | 8.71 ± 0.21 | 8.40 ± 0.94 | 10.20 ± 1.10 | 9.95 ± 0.77 | 10.10 ± 0.11 | 9.95 ± 0.17 |
| Sanguine H-2 | - | - | - | - | - | - | - | - | 9.43 ± 1.48 | 9.05 ± 0.56 | 9.68 ± 0.83 | 9.55 ± 0.84 |
| Ellagitannin 1 | - | - | - | - | - | - | - | - | 3.50 ± 0.70 | 3.30 ± 0.72 | 3.38 ± 0.38 | 3.16 ± 0.30 |
| Ellagitannin 2 | - | - | - | - | - | - | - | - | 0.98 ± 0.09 | 0.94 ± 0.06 | 1.05 ± 0.48 | 0.96 ± 0.23 |
| Ellagitannin 3 | 53.77 ± 2.37 | 51.15 ± 1.47 | 54.70 ± 2.15 | 52.55 ± 1.42 | 55.85 ± 1.30 | 53.22 ± 1.67 | 56.20 ± 1.38 | 52.30 ± 1.77 | 48.70 ± 0.59 | 46.90 ± 2.21 | 49.99 ± 1.79 | 45.92 ± 2.48 |
| Ellagitannin 4 | 6.09 ± 0.87 | 5.83 ± 0.09 | 6.11 ± 0.52 | 5.75 ± 0.34 | 6.38 ± 0.34 | 6.09 ± 0.90 | 6.25 ± 0.55 | 5.80 ± 0.29 | 9.38 ± 0.39 | 9.05 ± 0.21 | 9.55 ± 0.73 | 8.80 ± 0.64 |
| Isomer galloilo-bis-HHDP glucose | 0.41 ± 0.07 | 0.37 ± 0.05 | 0.42 ± 0.01 | 0.40 ± 0.09 | 0.40 ± 0.04 | 0.38 ± 0.00 | 0.43 ± 0.19 | 0.39 ± 0.06 | 1.15 ± 0.04 | 1.10 ± 0.54 | 1.15 ± 0.15 | 1.00 ± 0.00 |
| Ellagic acid | 4.29 ± 0.21 | 4.15 ± 0.18 | 4.35 ± 0.35 | 4.18 ± 0.12 | 4.40 ± 0.85 | 4.18 ± 0.89 | 4.45 ± 0.42 | 4.15 ± 0.38 | 10.95 ± 0.65 | 10.55 ± 0.01 | 10.78 ± 0.18 | 9.92 ± 0.26 |
| Sum of phenolic compounds | 95.10 | 90.58 | 96.44 | 92.56 | 98.41 | 94.01 | 98.53 | 92.26 | 117.92 | 113.97 | 119.55 | 112.88 |
| Compound Name | Rt (min) | λ (nm) | Calibration Curve | R2 | Concentration Range (µg/mL) | LOD (µg/mL) | LOQ (µg/mL) | Precision (RSD%) | Repeatability (RSD%, n= 3) | Stability (RSD%, n = 9) | |
|---|---|---|---|---|---|---|---|---|---|---|---|
| Intraday (n = 3) | Interday (n = 9) | ||||||||||
| (+)-Catechin | 3.19 | 280 | y = 1565.9x + 2243 | 0.9999 | LOQ-385 | 0.17 | 0.6 | 1.98 | 3.05 | 1.38 | 2.89 |
| Ellagic acid | 6.63 | 320 | y = 324.92x − 918.5 | 0.9994 | LOQ-100 | 0.15 | 0.5 | 1.01 | 2.95 | 1.15 | 2.65 |
| Isorhamnetin 3-O-glucoside | 7.36 | 360 | y = 20062x − 7082.8 | 0.9998 | LOQ-120 | 0.06 | 0.2 | 1.55 | 3.33 | 1.41 | 2.55 |
| Kaempferol 3-O-galactoside | 8.60 | 360 | y = 12057x − 1922.4 | 0.9997 | LOQ-110 | 0.07 | 0.2 | 1.30 | 2.99 | 1.20 | 2.79 |
| Myricetin 3-O-glucoside | 8.43 | 360 | y = 798.26x + 9241.3 | 0.9998 | LOQ-180 | 0.10 | 0.4 | 0.85 | 1.99 | 1.00 | 2.32 |
| Quercetin 3-O-glucoside | 8.22 | 360 | y = 11,923x + 8188 | 0.9999 | LOQ-180 | 0.03 | 0.1 | 0.87 | 2.02 | 1.00 | 2.88 |
| Sanguine H-2 | 5.33 | 254 | y = 31,926.5x − 1576.2 | 0.9994 | LOQ-200 | 0.02 | 0.6 | 0.90 | 2.11 | 0.08 | 1.13 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cendrowski, A.; Ścibisz, I.; Kieliszek, M.; Kolniak-Ostek, J.; Mitek, M. UPLC-PDA-Q/TOF-MS Profile of Polyphenolic Compounds of Liqueurs from Rose Petals (Rosa rugosa). Molecules 2017, 22, 1832. https://doi.org/10.3390/molecules22111832
Cendrowski A, Ścibisz I, Kieliszek M, Kolniak-Ostek J, Mitek M. UPLC-PDA-Q/TOF-MS Profile of Polyphenolic Compounds of Liqueurs from Rose Petals (Rosa rugosa). Molecules. 2017; 22(11):1832. https://doi.org/10.3390/molecules22111832
Chicago/Turabian StyleCendrowski, Andrzej, Iwona Ścibisz, Marek Kieliszek, Joanna Kolniak-Ostek, and Marta Mitek. 2017. "UPLC-PDA-Q/TOF-MS Profile of Polyphenolic Compounds of Liqueurs from Rose Petals (Rosa rugosa)" Molecules 22, no. 11: 1832. https://doi.org/10.3390/molecules22111832
APA StyleCendrowski, A., Ścibisz, I., Kieliszek, M., Kolniak-Ostek, J., & Mitek, M. (2017). UPLC-PDA-Q/TOF-MS Profile of Polyphenolic Compounds of Liqueurs from Rose Petals (Rosa rugosa). Molecules, 22(11), 1832. https://doi.org/10.3390/molecules22111832








