Synthesis and 2D-QSAR Study of Active Benzofuran-Based Vasodilators
Abstract
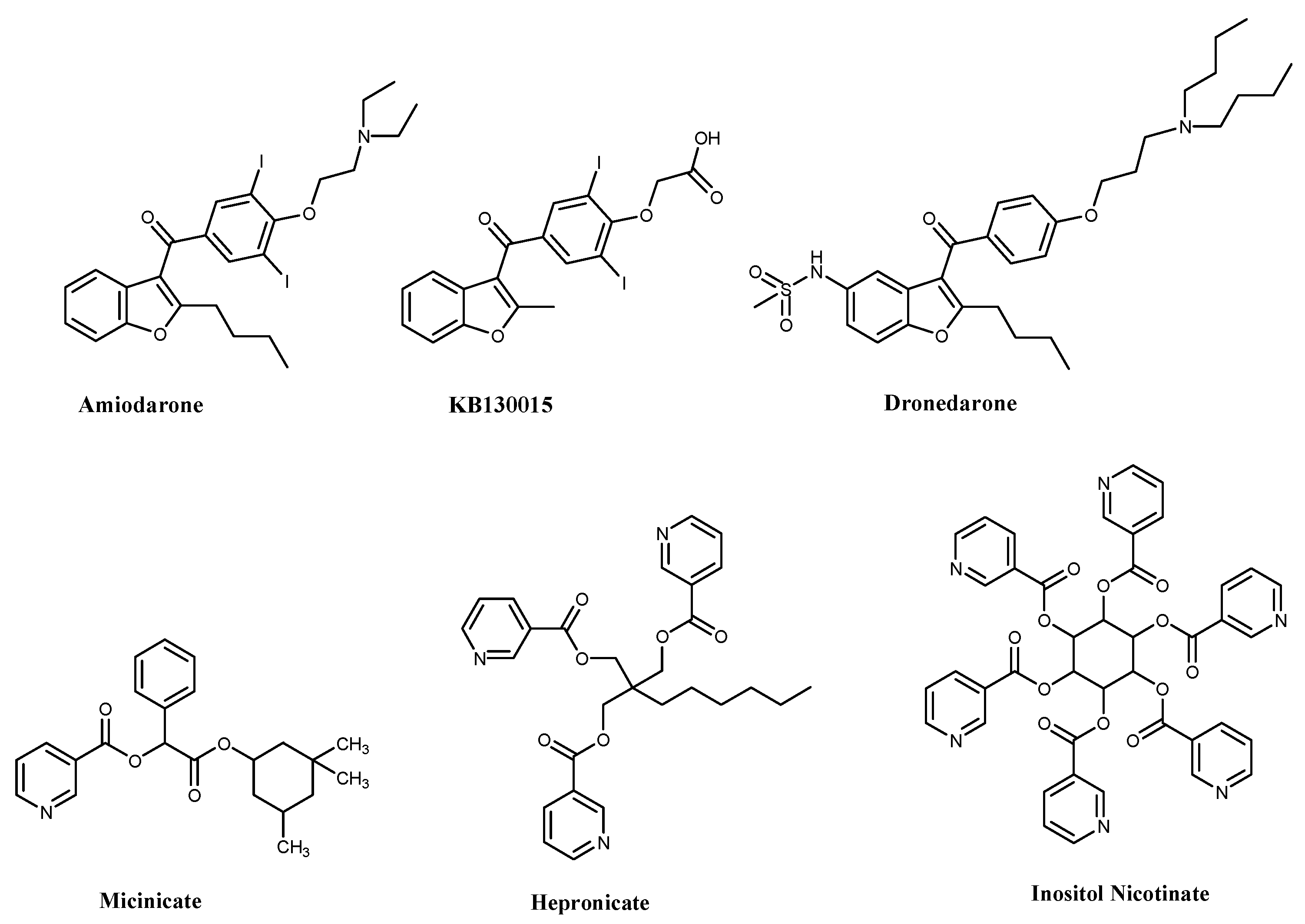
:1. Introduction
2. Results and Discussion
2.1. Chemistry
2.2. Biological Evaluation
2.2.1. Vasodilation Properties
2.2.2. Toxicological Bioassay
3. Experimental Section
3.1. General Information
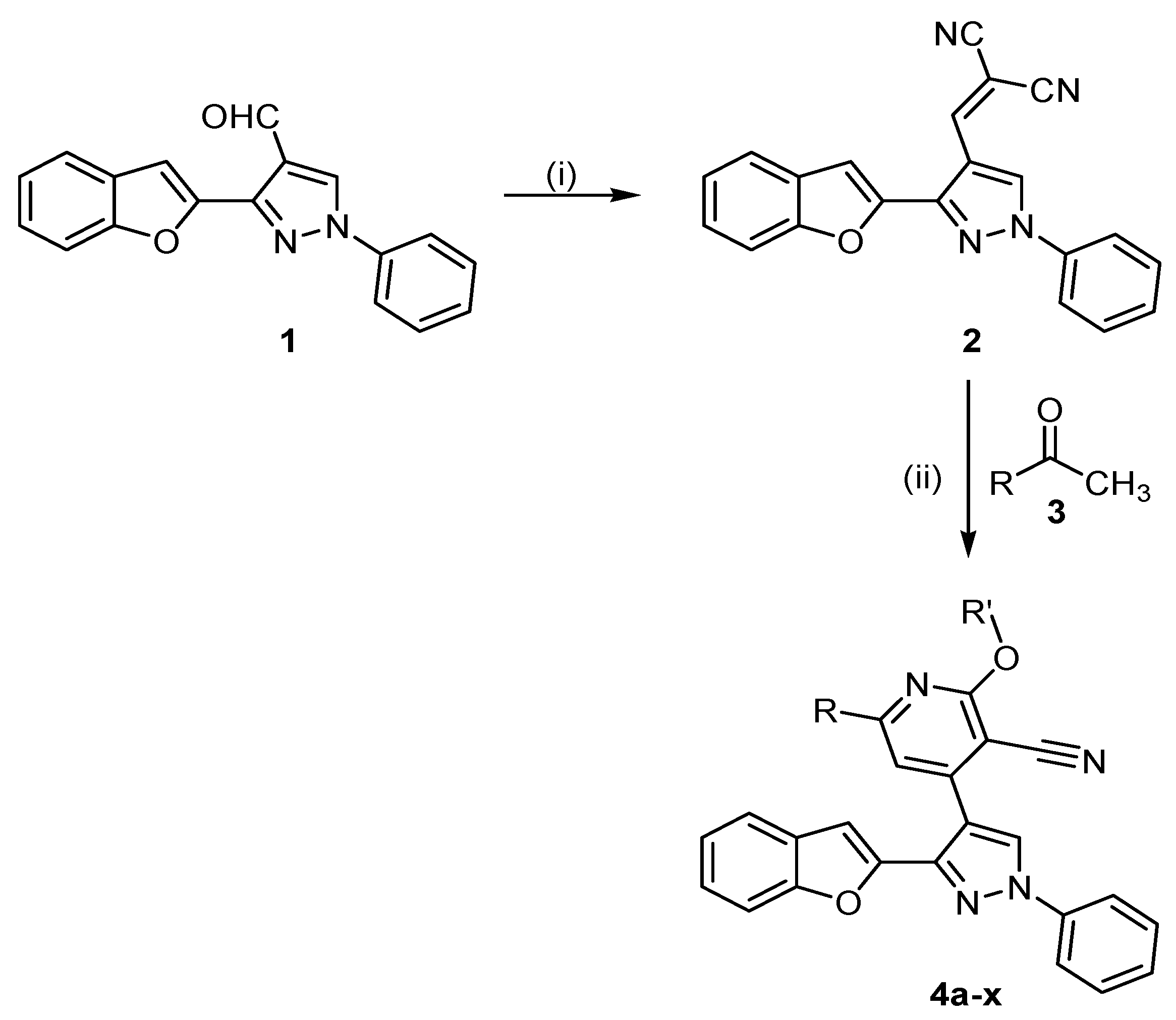
3.1.1. Synthesis of 2-((3-(Benzofuran-2-yl)-1-phenyl-1H-pyrazol-4-yl)methylene)malononitrile (2)
3.1.2. Synthesis of 2-Alkoxy-4-(3-(benzofuran-2-yl)-1-phenyl-1H-pyrazol-4-yl)-6-phenylpyridine-3-carbonitriles (4a–x)
General Procedure
3.2. Vasodilation Activity Screening
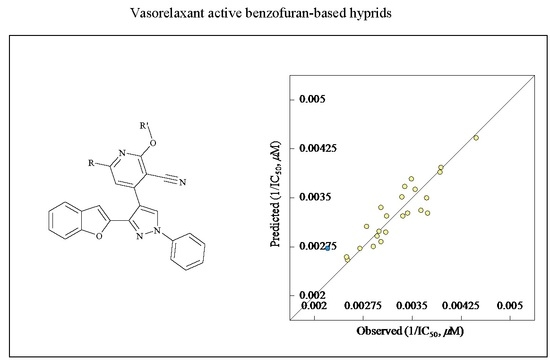
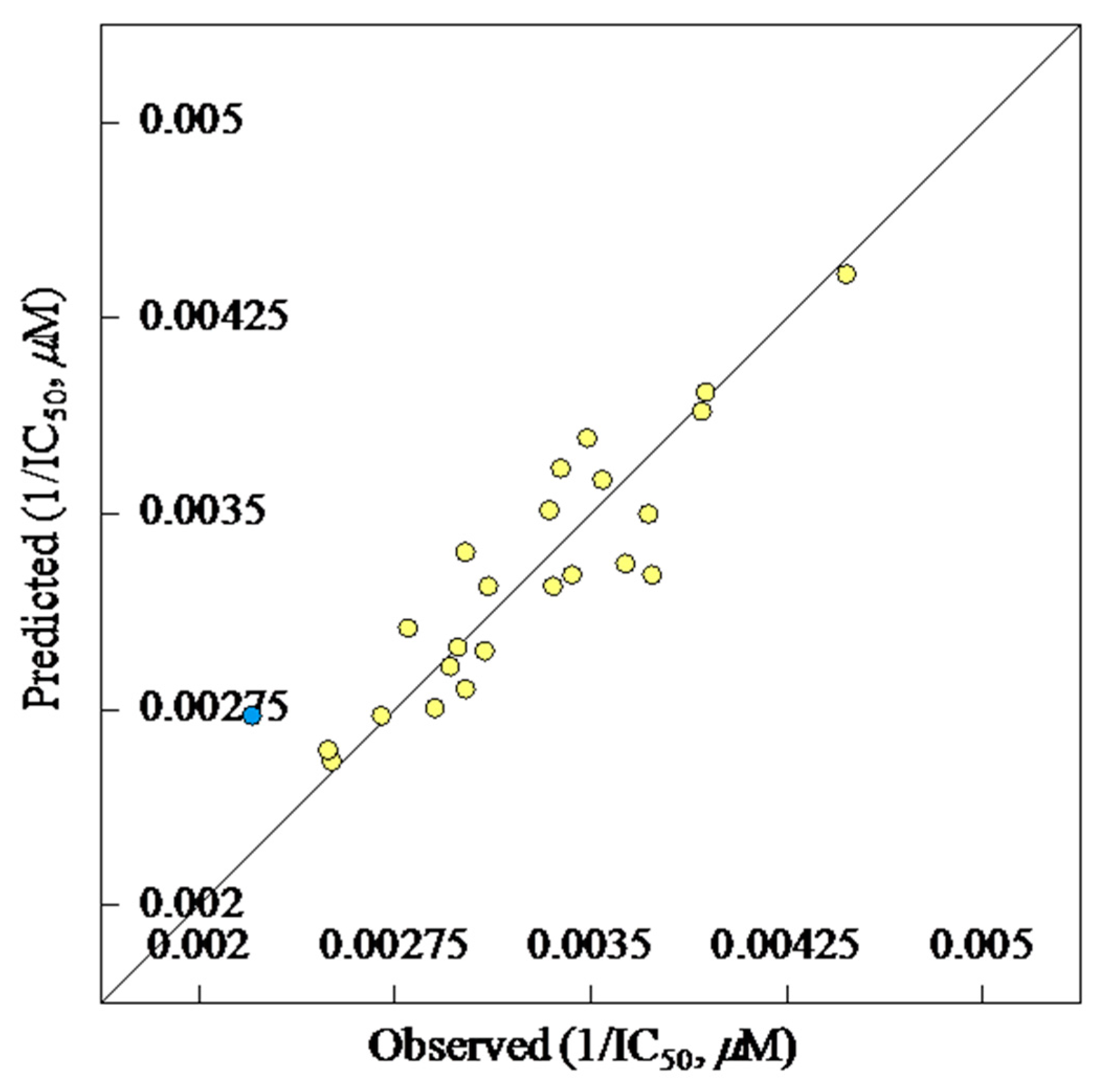
2D-QSAR Study
3.3. Toxicological Bioassay
4. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Garovic, V.D.; Hayman, S.R. Hypertension in pregnancy: An emerging risk factor for cardiovascular disease. Nat. Clin. Pract. Nephrol. 2007, 3, 613–622. [Google Scholar] [CrossRef] [PubMed]
- Kummerle, A.E.; Raimundo, J.M.; Leal, C.M.; Da Silva, G.S.; Balliano, T.L.; Pereira, M.A.; De Simone, C.A.; Sudo, R.T.; Zapata-Sudo, G.; Fraga, C.A.; et al. Studies towards the identification of putative bioactive conformation of potent vasodilator arylidene N-acylhydrazone derivatives. Eur. J. Med. Chem. 2009, 44, 4004–4009. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization. Media Centre. Available online: http://www.who.int/mediacentre/news/releases/2012/world_health_statistics_20120516/en/4004-4009 (accessed on 14 February 2016).
- Da Silva, V.J.; Viana, P.C.; Alves, R.M.; Salgado, H.C.; Montano, N.; Fazan, R.J. Antihypertensive action of amiodarone in spontaneously hypertensive rats. Hypertension 2001, 38, 597–601. [Google Scholar] [CrossRef]
- Almeida, M.R.; Lima, E.O.; da Silva, V.J.; Campos, M.G.; Antunes, L.M.; Salman, A.K.; Dias, F.L. Genotoxic studies in hypertensive and normotensive rats treated with amiodarone. Mutat. Res. 2008, 657, 155–159. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization. Cardiovascular Disease. Available online: http://www.who.int/topics/cardiovascular_diseases/en/ (accessed on 22 March 2016).
- Kashyap, M.K.; Yadav, V.; Sherawat, B.S.; Jain, S.; Kumari, S.; Khullar, M.; Sharma, P.C.; Nath, R. Different antioxidants status, total antioxidant power and free radicals in essential hypertension. Mol. Cell. Biochem. 2005, 277, 89–99. [Google Scholar] [CrossRef] [PubMed]
- Wang, R.; Shamloul, R.; Wang, X.; Meng, Q.; Wu, L. Sustained normalization of high blood pressure in spontaneously hypertensive rats by implanted hemin pump. Hypertension 2006, 48, 685–692. [Google Scholar] [CrossRef] [PubMed]
- Carlsson, B.; Singh, B.N.; Temciuc, M.; Nilsson, S.; Li, Y.-L.; Mellin, C.; Malm, J. Synthesis and preliminary characterization of a novel antiarrhythmic compound (KB130015) with an improved toxicity profile compared with amiodarone. J. Med. Chem. 2002, 45, 623–630. [Google Scholar] [CrossRef] [PubMed]
- Mubagwa, K.; Macianskiene, R.; Viappiani, S.; Gendviliene, V.; Carlsson, B.; Brandts, B. KB130015, a new amiodarone derivative with multiple effects on cardiac ion channels. Cardiovasc. Drug Rev. 2003, 21, 216–235. [Google Scholar] [CrossRef] [PubMed]
- Guiraudou, P.; CosnierPucheu, S.C.; Gayraud, R.; Gautier, P.; Roccon, A.; Herbert, J.M.; Nisato, D. Involvement of nitric oxide in amiodarone- and dronedarone-induced coronary vasodilation in guinea pig heart. Eur. J. Pharmacol. 2004, 496, 119–127. [Google Scholar] [CrossRef] [PubMed]
- Kodama, I.; Kamiya, K.; Toyama, J. Amiodarone: Ionic and cellular mechanisms of action of the most promising class III agent. Am. J. Cardiol. 1999, 84, 20–28. [Google Scholar] [CrossRef]
- Grossmann, M.; Dobrev, D.; Kirch, W. Amiodarone causes endothelium-dependent vasodilation in human hand veins in vivo. Clin. Pharmacol. Ther. 1998, 64, 302–311. [Google Scholar] [CrossRef]
- Massie, B.M.; Fisher, S.G.; Deedwania, P.C.; Singh, B.N.; Fletcher, R.D.; Singh, S.N. Effect of amiodarone on clinical status and left ventricular function in patients with congestive heart failure. Circulation 1996, 93, 2128–2134. [Google Scholar] [CrossRef] [PubMed]
- Gessner, G.; Heller, R.; Hoshi, T.; Heinemann, S.H. The amiodarone derivative 2-methyl-3-(3,5-diiodo-4-carboxymethoxybenzyl)benzofuran (KB130015) opens large- conductance Ca2+-activated K+ channels and relaxes vascular smooth muscle. Eur. J. Pharm. 2007, 555, 185–193. [Google Scholar] [CrossRef] [PubMed]
- Hanafy, D.A.; Chen, Y.; Chang, S.; Lu, Y.; Lin, Y.; Kao, Y.; Chen, S.; Chen, Y. Different effects of dronedarone and amiodarone on pulmonary vein electrophysiology, mechanical properties and H2O2-induced arrhythmogenicity. Eur. J. Pharm. 2013, 702, 103–108. [Google Scholar] [CrossRef] [PubMed]
- Stewart, S.; Hart, C.L.; Hole, D.J.; McMurray, J.J. A population-based study of the long-term risks associated with atrial fibrillation: 20-year follow-up of the Renfrew/Paisley study. Am. J. Med. 2002, 113, 359–364. [Google Scholar] [CrossRef]
- Hayta, S.A.; Arisoy, M.; Arpaci, O.T.; Yildiz, I.; Aki, E.; Ozkan, S.; Kaynak, F. Synthesis, antimicrobial activity, pharmacophore analysis of some new 2-(substitutedphenyl/benzyl)-5-[(2-benzofuryl)carboxamido]benzoxazoles. Eur. J. Med. Chem. 2008, 43, 2568–2578. [Google Scholar] [CrossRef] [PubMed]
- Manna, K.; Agrawal, Y.K. Microwave assisted synthesis of new indophenazine 1,3,5-trisubstruted pyrazoline derivatives of benzofuran and their antimicrobial activity. Bioorg. Med. Chem. Lett. 2009, 19, 2688–2692. [Google Scholar] [CrossRef] [PubMed]
- Xie, Y.; Kumar, D.; Bodduri, V.D.; Tarani, P.S.; Zhao, B.; Miao, J.; Jang, K.; Shin, D. Microwave-assisted parallel synthesis of benzofuran-2-carboxamide derivatives bearing anti-inflammatory, analgesic and antipyretic agents. Tetrahed. Lett. 2014, 55, 2796–2800. [Google Scholar] [CrossRef]
- El-Sawy, E.R.; Ebaid, M.S.; Abo-Salem, H.M.; Al-Sehemi, A.G.; Mandour, A.H. Synthesis, anti-inflammatory, analgesic and anticonvulsant activities of some new 4,6-dimethoxy-5- (heterocycles)benzofuran starting from naturally occurring. Arab. J. Chem. 2014, 7, 914–923. [Google Scholar] [CrossRef]
- Thevenin, M.; Thoret, S.; Grellier, P.; Dubois, J. Synthesis of polysubstitutedbenzofuran derivatives as novel inhibitors of parasitic growth. Bioorg. Med. Chem. 2013, 21, 4885–4892. [Google Scholar] [CrossRef] [PubMed]
- Feher, D.; Barlow, R.S.; McAtee, J.; Hemscheidt, T.K. Highly brominated antimicrobial metabolites from a marine Pseudoalteromonas sp. J. Nat. Prod. 2010, 73, 1963–1966. [Google Scholar] [CrossRef] [PubMed]
- Galal, S.A.; Abd El-All, A.S.; Abdallah, M.M.; El-Diwani, H.I. Synthesis of potent antitumor and antiviral benzofuran derivatives. Bioorg. Med. Chem. Lett. 2009, 19, 2420–2628. [Google Scholar] [CrossRef] [PubMed]
- Xie, F.; Zhu, H.; Zhang, H.; Lang, Q.; Tang, L.; Huang, Q.; Yu, L. In vitro and in vivo characterization of a benzofuran derivative, a potential anticancer agent, as a novel Aurora B kinase inhibitor. Eur. J. Med. Chem. 2015, 89, 310–319. [Google Scholar] [CrossRef] [PubMed]
- Kleemann, A.; Engel, J.; Kutscher, B.; Reichert, D. Pharmaceutical Substances: Syntheses Patents Applications, 3rd ed.; Thieme, Stuttgart: New York, NY, USA, 1999. [Google Scholar]
- Girgis, A.S.; Kalmouch, A.; Ellithey, M. Synthesis of novel vasodilatory active nicotinate esters with amino acid function. Bioorg. Med. Chem. 2006, 14, 8488–8494. [Google Scholar] [CrossRef] [PubMed]
- Nofal, Z.M.; Srour, A.M.; El-Eraky, W.I.; Saleh, D.O.; Girgis, A.S. Rational design, synthesis and QSAR study of vasorelaxant active 3-pyridinecarbonitriles incorporating 1H-benzimidazol-2-yl function. Eur. J. Med. Chem. 2013, 63, 14–21. [Google Scholar] [CrossRef] [PubMed]
- Hassan, G.S.; Abdel Rahman, D.E.; Saleh, D.O.; Abdel Jaleel, G.A. Benzofuran–Morpholinomethyl–Pyrazoline Hybrids as a New Class of Vasorelaxant Agents: Synthesis and Quantitative Structure–Activity Relationship Study. Chem. Pharm. Bull. 2013, 62, 1238–1251. [Google Scholar] [CrossRef]
- Matsui, A.; Matsuo, H.; Takanaga, H.; Sasaki, S.; Maeda, M.; Sawada, Y. Prediction of catalepsies induced by amiodarone, aprindine and procaine: Similarity in conformation of diethylaminoethyl side chain. J. Pharmacol. Exp. Ther. 1998, 287, 725–732. [Google Scholar] [PubMed]
- Srour, A.M.; Abd El-Karim, S.S.; Saleh, D.O.; El-Eraky, W.I.; Nofal, Z.M. Rational design, synthesis and 2D-QSAR study of novel vasorelaxant active benzofuran-pyridine hybrids. Bioorg. Med. Chem. Lett. 2016, 26, 2557. [Google Scholar] [CrossRef] [PubMed]
- El-Zahar, M.I.; Abd El-Karim, S.S.; Anwar, M.M. Synthesis and cytotoxicity screening of some novel benzofuranoyl-pyrazole derivatives against liver and cervix carcinoma cell lines. S. Afr. J. Chem. 2009, 62, 189–199. [Google Scholar]
- Oliferenko, P.V.; Oliferenko, A.A.; Girgis, A.S.; Saleh, D.O.; Srour, A.M.; George, R.F.; Pillai, G.G.; Panda, C.S.; Hall, C.D.; Katritzky, A.R. Synthesis, bioassay, and molecular field topology analysis of diverse vasodilatoryheterocycles. J. Chem. Inf. Model. 2014, 54, 1103–1116. [Google Scholar] [CrossRef] [PubMed]
- University of Florida 2001–2005, CODESSA PRO. Available online: http://www.codessa-pro.com/index.htm (accessed on 20 July 2017).
- Panda, S.S.; Liaqat, S.; Girgis, A.S.; Samir, A.; Hall, C.D.; Katritzky, A.R. Novel antibacterial active quinolone–fluoroquinolone conjugates and 2D-QSAR studies. Bioorg. Med. Chem. Lett. 2015, 25, 3816–3821. [Google Scholar] [CrossRef] [PubMed]
- Girgis, A.S.; Pand, S.S.; Ahmed Farag, I.S.; El-Shabiny, A.M.; Moustafa, A.M.; Pillai, G.G.; Panda, C.S.; Hall, C.D.; Katritzky, A.R. Synthesis, and QSAR analysis of anti-oncological active spiro-alkaloids. Org. Biomol. Chem. 2015, 13, 1741–1753. [Google Scholar] [CrossRef] [PubMed]
- Girgis, A.S.; Mishriky, N.; Farag, A.M.; El-Eraky, W.I.; Farag, H. Synthesis of new 3-pyridinecarboxylates of potential vasodilation properties. Eur. J. Med. Chem. 2008, 43, 1818–1827. [Google Scholar] [CrossRef] [PubMed]
- Raj, P.A.; Suddendra, G.; Shakeel, A.S.; Girish, M. Synthesis of new benzofuran derivatives and their in vitro evaluation for antimicrobial activity. IJDFR 2012, 3, 135–147. [Google Scholar]
- Girgis, A.S.; Panda, S.S.; Srour, A.M.; Farag, H.; Ismail, N.M.; Elgendy, A.M.; Abdel-Aziz, M.; Katritzky, A.R. Rational design, synthesis and molecular modeling studies of novel anti-oncological alkaloids against melanoma. Org. Biomol. Chem. 2015, 13, 6619–6633. [Google Scholar] [CrossRef] [PubMed]
- Girgis, A.S.; Panda, S.S.; Aziz, M.N.; Steel, P.J.; Hall, C.D.; Katritzky, A.R. Rational design, synthesis, and 2D-QSAR study of anti-oncological alkaloids against hepatoma and cervical carcinoma. RSC Adv. 2015, 5, 28554–28569. [Google Scholar] [CrossRef]
- Girgis, A.S.; Panda, S.S.; Shalaby, E.M.; Mabied, A.F.; Steel, P.J.; Hall, C.D.; Katritzky, A.R. Regioselective synthesis and theoretical studies of an anti-neoplastic fluoro-substituted dispiro-oxindole. RSC Adv. 2015, 5, 14780–14787. [Google Scholar] [CrossRef]
- Girgis, A.S.; Aziz, M.N.; Shalaby, E.M.; Saleh, D.O.; Mishriky, N.; El-Eraky, W.I.; Farag, I.S. Molecular structure studies of novel bronchodilatory active 4-azafluorenes. Z. Kristallog. 2016, 231, 179–187. [Google Scholar] [CrossRef]
- Girgis, A.S.; Mabied, A.F.; Stawinski, J.; Hegazy, L.; George, R.F.; Farag, H.; Shalaby, E.M.; Farag, I.S. Synthesis and DFT studies of an antitumor active spiro-oxindole. N. J. Chem. 2015, 39, 8017–8027. [Google Scholar] [CrossRef]
- Shalaby, E.M.; Girgis, A.S.; Moustafa, A.M.; ElShaabiny, A.M.; El-Gendy, B.E.; Mabied, A.F.; Farag, I.S. Regioselective synthesis, stereochemical structure, spectroscopic characterization and geometry optimization of dispiro[3H-indole-3,2′-pyrrolidine-3′,3″-piperidines]. J. Mol. Struct. 2014, 1075, 327–334. [Google Scholar] [CrossRef]
- Tiwari, A.D.; Panda, S.S.; Girgis, A.S.; Sahu, S.; George, R.F.; Srour, A.M.; La Starza, B.; Asiri, A.M.; Hall, C.D.; Katritzky, A.R. Microwave assisted synthesis and QSAR study of novel NSAID acetaminophen conjugates with amino acid linkers. Org. Biomol. Chem. 2014, 12, 7238–7249. [Google Scholar] [CrossRef] [PubMed]
Sample Availability: Samples of which all the compounds are available from the authors. |
| 3b; R = 4-ClC6H4 | 4b; R = Ph |
| 3c; R = 4-BrC6H4 | 4c; R = 4-ClC6H4 |
| 3d; R = 4-FC6H4 | 4d; R = 4-ClC6H4 |
| 3e; R = 4-H3CC6H4 | 4e; R = 4-BrC6H4 |
| 3f; R = 4-H3COC6H4 | 4f; R = 4-BrC6H4 |
| 3g; R = 1,2,3,4-Tetrahydronaphthalen-6-yl | 4g; R = 4-FC6H4 |
| 3h; R = 2-Pyrrolyl | 4h; R = 4-FC6H4 |
| 3i; R = 2-Furanyl | 4i; R = 4-H3CC6H4 |
| 3j; R = 2-Thienyl | 4j; R = 4-H3CC6H4 |
| 3k; R = 2-Pyridinyl | 4k; R = 4-H3COC6H4 |
| 3l; R = 1-Methyl-1H-benzo[d]imidazol-2-yl | 4l; R = 4-H3COC6H4 |
| 4m; R = 1,2,3,4-Tetrahydronaphthalen-6-yl, R′ = Me | |
| 4n; R = 1,2,3,4-Tetrahydronaphthalen-6-yl, R′ = Et | |
| 4o; R = 2-Pyrrolyl, R′ = Me | |
| 4p; R = 2- Pyrrolyl, R′ = Et | |
| 4q; R = 2-Furanyl, R′ = Me | |
| 4r; R = 2-Furanyl, R′ = Et | |
| 4s; R = 2-Thienyl, R′ = Me | |
| 4t; R = 2-Thienyl, R′ = Et | |
| 4u; R = 2-Pyridinyl, R′ = Me | |
| 4v; R = 2-Pyridinyl, R′ = Et | |
| 4w;R = 1-Methyl-1H-benzo[d]imidazol-2-yl, R′ = Me | |
| Entry | Compound | R | R′ | Potency (IC50), mM |
|---|---|---|---|---|
| 1 | 4a | Ph | Me | 0.281 |
| 2 | 4b | Ph | Et | 0.343 |
| 3 | 4c | 4-ClC6H4 | Me | 0.295 |
| 4 | 4d | 4-ClC6H4 | Et | 0.397 |
| 5 | 4e | 4-BrC6H4 | Me | 0.253 |
| 6 | 4f | 4-BrC6H4 | Et | 0.267 |
| 7 | 4g | 4-FC6H4 | Me | 0.275 |
| 8 | 4h | 4-FC6H4 | Et | 0.330 |
| 9 | 4i | 4-H3CC6H4 | Me | 0.330 |
| 10 | 4j | 4-H3CC6H4 | Et | 0.452 |
| 11 | 4k | 4-H3COC6H4 | Me | 0.322 |
| 12 | 41 | 4-H3COC6H4 | Et | 0.291 |
| 13 | 4m | 1,2,3,4-Tetrahydronaphthalen-6-yl | Me | 0.286 |
| 14 | 4n | 1,2,3,4-Tetrahydronaphthalen-6-yl | Et | 0.337 |
| 15 | 4o | 2-Pyrrolyl | Me | 0.356 |
| 16 | 4p | 2-Pyrrolyl | Et | 0.400 |
| 17 | 4q | 2-Furanyl | Me | 0.321 |
| 18 | 4r | 2-Furanyl | Et | 0.254 |
| 19 | 4s | 2-Thienyl | Me | 0.268 |
| 20 | 4t | 2-Thienyl | Et | 0.298 |
| 21 | 4u | 2-Pyridinyl | Me | 0.333 |
| 22 | 4v | 2-Pyridinyl | Et | 0.370 |
| 23 | 4w | 1-Methyl-1H-benzo[d]imidazol-2-yl | Me | 0.223 |
| 24 | 4x | 1-Methyl-1H-benzo[d]imidazol-2-yl | Et | 0.299 |
| 25 | Amiodarone.HCl | - | - | 0.300 |
| Entry | ID | Coefficient | s | t | Descriptor |
|---|---|---|---|---|---|
| N = 24, n= 4, R2 = 0.816, R2cvOO = 0.731, R2cvMO = 0.772, F = 21.103, s2 = 6.191 × 10−8 | |||||
| 1 | 0 | 1.464 | 0.244 | 6.009 | Intercept |
| 2 | D1 | 0.0004 | 4.751 × 10−5 | 7.789 | Max. e–e repulsion for bond C–O |
| 3 | D2 | −7.1825 × 10−6 | 1.975 × 10−6 | −3.637 | WNSA-1 Weighted PNSA (PNSA1*TMSA/1000) (MOPAC PC) |
| 4 | D3 | −0.255 | 0.045 | −5.670 | FHACA Fractional HACA (HACA1/TMSA) (MOPAC PC) |
| 5 | D4 | −0.0043 | 0.001 | −6.241 | Max. e–n attraction for bond C–N |
| 1/IC50 (μM) = 1.464 + (0.0004 × D1) − [(7.1825 × 10−6) × D2] − (0.255 × D3) − (0.0043 × D3) | |||||
| Entry | Compd. | R | R′ | Observed IC50, μM | Estimated IC50, μM | Error |
|---|---|---|---|---|---|---|
| 1 | 4a | Ph | Me | 281 | 276 | 5 |
| 2 | 4b | Ph | Et | 343 | 363 | −20 |
| 3 | 4c | 4-ClC6H4 | Me | 295 | 272 | 23 |
| 4 | 4d | 4-ClC6H4 | Et | 397 | 392 | 5 |
| 5 | 4e | 4-BrC6H4 | Me | 253 | 253 | 0 |
| 6 | 4f | 4-BrC6H4 | Et | 267 | 307 | −40 |
| 7 | 4g | 4-FC6H4 | Me | 275 | 303 | −28 |
| 8 | 4h | 4-FC6H4 | Et | 330 | 354 | −24 |
| 9 | 4i | 4-H3CC6H4 | Me | 330 | 299 | 31 |
| 10 | 4j | 4-H3CC6H4 | Et | 452 | 369 | 83 |
| 11 | 4k | 4-H3COC6H4 | Me | 322 | 337 | −15 |
| 12 | 41 | 4-H3COC6H4 | Et | 291 | 307 | −16 |
| 13 | 4m | 1,2,3,4-Tetrahydronaphthalen-6-yl | Me | 286 | 265 | 21 |
| 14 | 4n | 1,2,3,4-Tetrahydronaphthalen-6-yl | Et | 337 | 344 | −7 |
| 15 | 4o | 2-Pyrrolyl | Me | 268 | 286 | −18 |
| 16 | 4p | 2-Pyrrolyl | Et | 298 | 311 | −13 |
| 17 | 4q | 2-Furanyl | Me | 321 | 310 | 11 |
| 18 | 4r | 2-Furanyl | Et | 254 | 257 | −3 |
| 19 | 4s | 2-Thienyl | Me | 356 | 328 | 28 |
| 20 | 4t | 2-Thienyl | Et | 400 | 386 | 14 |
| 21 | 4u | 2-Pyridinyl | Me | 333 | 336 | −3 |
| 22 | 4v | 2-Pyridinyl | Et | 370 | 369 | 1 |
| 23 | 4w | 1-Methyl-1H-benzo[d]imidazol-2-yl | Me | 223 | 227 | −4 |
| 24 | 4x | 1-Methyl-1H-benzo[d]imidazol-2-yl | Et | 299 | 285 | 14 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Khalifa, N.M.; Srour, A.M.; Abd El-Karim, S.S.; Saleh, D.O.; Al-Omar, M.A. Synthesis and 2D-QSAR Study of Active Benzofuran-Based Vasodilators. Molecules 2017, 22, 1820. https://doi.org/10.3390/molecules22111820
Khalifa NM, Srour AM, Abd El-Karim SS, Saleh DO, Al-Omar MA. Synthesis and 2D-QSAR Study of Active Benzofuran-Based Vasodilators. Molecules. 2017; 22(11):1820. https://doi.org/10.3390/molecules22111820
Chicago/Turabian StyleKhalifa, Nagy M., Aladdin M. Srour, Somaia S. Abd El-Karim, Dalia O. Saleh, and Mohamed A. Al-Omar. 2017. "Synthesis and 2D-QSAR Study of Active Benzofuran-Based Vasodilators" Molecules 22, no. 11: 1820. https://doi.org/10.3390/molecules22111820
APA StyleKhalifa, N. M., Srour, A. M., Abd El-Karim, S. S., Saleh, D. O., & Al-Omar, M. A. (2017). Synthesis and 2D-QSAR Study of Active Benzofuran-Based Vasodilators. Molecules, 22(11), 1820. https://doi.org/10.3390/molecules22111820