Phytotherapeutics: The Emerging Role of Intestinal and Hepatocellular Transporters in Drug Interactions with Botanical Supplements
Abstract
1. Introduction
2. Oral Absorption of Coadministered Botanical Ingredients and Drugs
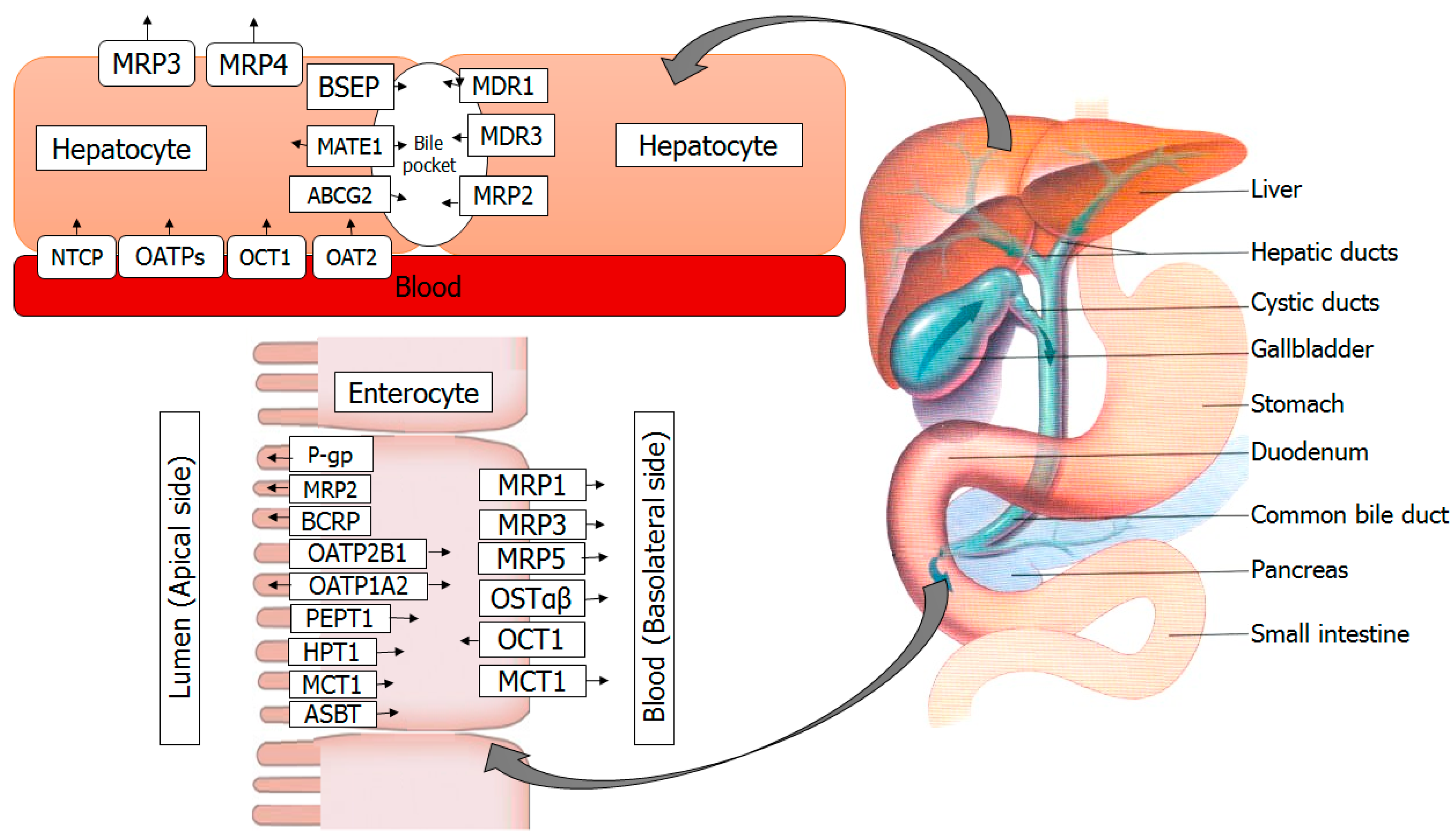
3. Transporters in the Intestine
4. Transporters in the Liver
5. Botanical Supplement-Mediated Cellular Uptake and Efflux
6. Selected Botanical Supplements and their Interactions
6.1. Citrus paradise (Grapefruit) Juice
6.2. Citrus sinensis (Orange) Juice
6.3. Malus pumila (Apple) Juice
6.4. Silybum marianum (Milk Thistle)
6.5. Camellia sinensis Leaves (Green Tea)
6.6. Glycine max Merrill (Soybean)
6.7. Hypericum perforatum (St. John’s Wort)
6.8. Cynara scolymus (Artichoke)
6.9. Resveratrol
6.10. Anthocyans
6.11. Other Botanical Supplements
7. Conclusions
Conflicts of Interest
References
- De Lima Toccafondo Vieira, M.; Huang, S.M. Botanical-drug interactions: A scientific perspective. Planta Med. 2012, 78, 1400–1415. [Google Scholar] [CrossRef] [PubMed]
- Calitz, C.; du Plessis, L.; Gouws, C.; Steyn, D.; Steenekamp, J.; Muller, C.; Hamman, S. Herbal hepatotoxicity: Current status, examples, and challenges. Exp. Opin. Drug Metab. Toxicol. 2015, 11, 1551–1565. [Google Scholar] [CrossRef] [PubMed]
- STICKEL, F.; SHOUVAL, D. Hepatotoxicity of herbal and dietary supplements: An update. Arch. Toxicol. 2015, 89, 851–865. [Google Scholar] [CrossRef] [PubMed]
- Domitrovic, R.; Potocnjak, I. A comprehensive overview of hepatoprotective natural compounds: Mechanism of action and clinical perspectives. Arch. Toxicol. 2016, 90, 39–79. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Paxton, J.W. The effects of flavonoids on the ABC transporters: Consequences for thepharmacokinetics of substrate drugs. Exp. Opin. Drug Metab. Toxicol. 2013, 9, 267–285. [Google Scholar] [CrossRef] [PubMed]
- Haefeli, W.E.; Carls, A. Drug interactions with phytotherapeutics in oncology. Exp. Opin. Drug Metab. Toxicol. 2014, 10, 359–377. [Google Scholar] [CrossRef] [PubMed]
- Hussain, M.M. Intestinal lipid absorption and lipoprotein formation. Curr. Opin. Lipidol. 2014, 25, 200–206. [Google Scholar] [CrossRef] [PubMed]
- Palacin, M.; Estevez, R.; Bertran, J.; Zorzano, A. Molecular biology of mammalian plasma membrane amino acid transporters. Physiol. Rev. 1998, 78, 969–1054. [Google Scholar] [PubMed]
- Poncet, N.; Taylor, P.M. The role of amino acid transporters in nutrition. Curr. Opin. Clin. Nutr. Metab. Care 2013, 16, 57–65. [Google Scholar] [CrossRef] [PubMed]
- Wright, E.M. Glucose transport families SLC5 and SLC50. Mol. Asp. Med. 2013, 34, 183–196. [Google Scholar] [CrossRef] [PubMed]
- Mueckler, M.; Thorens, B. The SLC2 (GLUT) family of membrane transporters. Mol. Asp. Med. 2013, 34, 121–138. [Google Scholar] [CrossRef] [PubMed]
- Rui, L. Energy metabolism in the liver. Compr. Physiol. 2014, 4, 177–197. [Google Scholar] [PubMed]
- Khan, F.A.; Maalik, A.; Murtaza, G. Inhibitory mechanism against oxidative stress of caffeic acid. J. Food Drug Anal. 2016, 24, 695–702. [Google Scholar] [CrossRef] [PubMed]
- Tenore, G.C.; Daglia, M.; Ciampaglia, R.; Novellino, E. Exploring the nutraceutical potential of polyphenols from black, green and white tea infusions—An overview. Curr. Pharm. Biotechnol. 2015, 16, 265–271. [Google Scholar] [CrossRef] [PubMed]
- Murtaza, G.; Sajjad, A.; Mehmood, Z.; Shah, S.H.; Siddiqi, A.R. Possible Molecular Targets for Therapeutic Applications of Caffeic Acid Phenethyl Ester in Inflammation and Cancer. J. Food Drug Anal. 2015, 23, 11–18. [Google Scholar] [CrossRef] [PubMed]
- Stieger, B.; Mahdi, Z.M.; Jager, W. Intestinal and Hepatocellular Transporters: Therapeutic Effects and Drug Interactions of Herbal Supplements. Annu. Rev. Pharmacol. Toxicol. 2017, 57, 399–416. [Google Scholar] [CrossRef] [PubMed]
- Stieger, B.; Meier, P.J. Pharmacogenetics of drug transporters in the enterohepatic circulation. Pharmacogenomics 2017, 12, 611–631. [Google Scholar] [CrossRef] [PubMed]
- Seeff, L.B.; Bonkovsky, H.L.; Navarro, V.J.; Wang, G. Herbal products and the liver: A review of adverse effects and mechanisms. Gastroenterology 2015, 148, 517–532. [Google Scholar] [CrossRef] [PubMed]
- Kramer, R. Functional principles of solute transport systems: Concepts and perspectives. Biochim. Biophys. Acta 1994, 1185, 1–34. [Google Scholar] [CrossRef]
- Kobayashi, D.; Nozawa, T.; Imai, K.; Nezu, J.; Tsuji, A.; Tamai, I. Involvement of human organicanion transporting polypeptide OATP-B (SLC21A9) in pH-dependent transport across intestinal apical membrane. J. Pharmacol. Exper. Ther. 2003, 306, 703–708. [Google Scholar] [CrossRef] [PubMed]
- Govindarajan, R.; Bakken, A.H.; Hudkins, K.L.; Lai, Y.; Casado, F.J.; Pastor-Anglada, M.; Tse, C.M.; Hayashi, J.; Unadkat, J.D. In situ hybridization and immunolocalization of concentrative and equilibrative nucleoside transporters in the human intestine, liver, kidneys, and placenta. Am. J. Physiol. 2007, 293, R1809–R1822. [Google Scholar] [CrossRef] [PubMed]
- Groneberg, D.A.; Doring, F.; Eynott, P.R.; Fischer, A.; Daniel, H. Intestinal peptide transport: Ex vivo uptake studies and localization of peptide carrier PEPT1. Am. J. Physiol. 2001, 281, G697–G704. [Google Scholar]
- Dawson, P.A.; Hubbert, M.L.; Rao, A. Getting them OST from OST: Role of organic solute transporter, OSTα-OSTβ, in bile acid and steroid metabolism. Biochim. Biophys. Acta 2010, 1801, 994–1004. [Google Scholar] [CrossRef] [PubMed]
- Kalapos-Kovacs, B.; Magda, B.; Jani, M.; Fekete, Z.; Szabo, P.T.; Antal, I.; Krajcsi, P.; Klebovich, I. Multiple ABC transporters efflux baicalin. Phytother. Res. 2015, 29, 1987–1990. [Google Scholar] [CrossRef] [PubMed]
- Van de Wetering, K.; Feddema, W.; Helms, J.B.; Brouwers, J.F.; Borst, P. Targeted metabolomics identifies glucuronides of dietary phytoestrogens as a major class of MRP3 substrates in vivo. Gastroenterology 2009, 137, 1725–1735. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Lu, J.; Paxton, J.W. The role of ABC and SLC transporters in the pharmacokinetics of dietary and herbal phytochemicals and their interactions with xenobiotics. Curr. Drug Metab. 2012, 13, 624–639. [Google Scholar] [CrossRef] [PubMed]
- Planas, J.M.; Alfaras, I.; Colom, H.; Juan, M.E. The bioavailability and distribution of trans-resveratrol are constrained by ABC transporters. Arch. Biochem. Biophys. 2012, 527, 67–73. [Google Scholar] [CrossRef] [PubMed]
- Berger, W.; Micksche, M.; Elbling, L. Effects of multidrug resistance-related ATP-binding-cassette transporter proteins on the cytoskeletal activity of cytochalasins. Exp. Cell Res. 1997, 237, 307–317. [Google Scholar] [CrossRef] [PubMed]
- He, S.M.; Li, C.G.; Liu, J.P.; Chan, E.; Duan, W.; Zhou, S.F. Disposition pathways and pharmacokinetics of herbal medicines in humans. Curr. Med. Chem. 2010, 17, 4072–4113. [Google Scholar] [CrossRef] [PubMed]
- De Leeuw, A.M.; Brouwer, A.; Knook, D.L. Sinusoidal endothelial cells of the liver: Fine structure and function in relation to age. J. Electron Microsc. Tech. 1990, 14, 218–236. [Google Scholar] [CrossRef] [PubMed]
- Burckhardt, G. Drug transport by organic anion transporters (OATs). Pharmacol. Ther. 2012, 136, 106–130. [Google Scholar] [CrossRef] [PubMed]
- Wlcek, K.; Stieger, B. ATP-binding cassette transporters in liver. BioFactors 2014, 40, 188–198. [Google Scholar] [CrossRef] [PubMed]
- Zhang, L.; Zuo, Z.; Lin, G. Intestinal and hepatic glucuronidation of flavonoids. Mol. Pharmacol. 2007, 4, 833–845. [Google Scholar] [CrossRef] [PubMed]
- Motohashi, H.; Inui, K. Multidrug and toxin extrusion family SLC47: Physiological, pharmacokinetic and toxicokinetic importance of MATE1 and MATE2-K. Mol. Asp. Med. 2013, 34, 661–668. [Google Scholar] [CrossRef] [PubMed]
- Stieger, B. The role of the sodium-taurocholate cotransporting polypeptide (NTCP) and of the bilesalt export pump (BSEP) in physiology and pathophysiology of bile formation. Handb. Exp. Pharmacol. 2011, 201, 205–259. [Google Scholar]
- Pauli-Magnus, C.; Meier, P.J.; Stieger, B. Genetic determinants of drug-induced cholestasis andintrahepatic cholestasis of pregnancy. Semin. Liver Dis. 2010, 30, 147–159. [Google Scholar] [CrossRef] [PubMed]
- Meng, Q.; Liu, K. Pharmacokinetic Interactions Between Herbal Medicines and Prescribed Drugs: Focus on Drug Metabolic Enzymes and Transporters. Curr. Drug Metab. 2014, 15, 791–807. [Google Scholar] [CrossRef] [PubMed]
- Liu, C.X.; Yi, X.L.; Si, D.Y.; Xiao, X.F.; He, X.; Li, Y.Z. Herb-drug Interactions Involving Drug Metabolizing Enzymes and Transporters. Curr. Drug Metab. 2011, 12, 835–849. [Google Scholar] [CrossRef] [PubMed]
- Tamaki, H.; Satoh, H.; Hori, S.; Ohtani, H.; Sawada, Y. Inhibitory effects of herbal extracts on breast cancer resistance protein (BCRP) and structure-inhibitory potency relationship of isoflavonoids. Drug Metab. Pharmacokinet. 2010, 25, 170–179. [Google Scholar] [CrossRef] [PubMed]
- Ge, S.; Yin, T.; Xu, B.; Gao, S.; Hu, M. Curcumin Affects Phase II Disposition of Resveratrol through Inhibiting Efflux Transporters MRP2 and BCRP. Pharm. Res. 2016, 23, 590–592. [Google Scholar] [CrossRef] [PubMed]
- Zhang, S.; Yang, X.; Morris, M.E. Flavonoids are inhibitors of breast cancer resistance protein (ABCG2)-mediated transport. Mol. Pharmacol. 2004, 65, 1208–1216. [Google Scholar] [CrossRef] [PubMed]
- Perez, M.; Otero, J.A.; Barrera, B.; Prieto, J.G.; Merino, G.; Alvarez, A.I. Inhibition of ABCG2/BCRP transporter by soy isoflavones genistein and daidzein: Effect on plasma and milk levels of danofloxacin in sheep. Vet. J. 2013, 196, 203–208. [Google Scholar] [CrossRef] [PubMed]
- Hajda, J.; Rentsch, K.M.; Gubler, C.; Steinert, H.; Stieger, B.; Fattinger, K. Garlic extract induces intestinal P-glycoprotein, but exhibits no effect on intestinal and hepatic CYP3A4 in humans. Eur. J. Pharm. Sci. 2010, 41, 729–735. [Google Scholar] [CrossRef] [PubMed]
- Dresser, G.K.; Bailey, D.G.; Leake, B.F.; Schwarz, U.I.; Dawson, P.A.; Freeman, D.J.; Kim, R.B. Fruit juices inhibit organic anion transporting polypeptide-mediated drug uptake to decrease the oral availability of fexofenadine. Clin. Pharmacol. Ther. 2002, 71, 11–20. [Google Scholar] [CrossRef] [PubMed]
- Durr, D.; Stieger, B.; Kullak-Ublick, G.A.; Rentsch, K.M.; Steinert, H.C.; Meier, P.J.; Fattinger, K. St John’s Wort induces intestinal P-glycoprotein/MDR1 and intestinal and hepatic CYP3A4. Clin. Pharmacol. Ther. 2000, 68, 598–604. [Google Scholar] [CrossRef] [PubMed]
- Cermanova, J.; Kadova, Z.; Zagorova, M.; Hroch, M.; Tomsik, P.; Nachtigal, P.; Kudlackova, Z.; Pavek, P.; Dubecka, M.; Ceckova, M.; et al. Boldine enhances bile production in rats via osmotic and farnesoid X receptor dependent mechanisms. Toxicol. Appl. Pharmacol. 2015, 285, 12–22. [Google Scholar] [CrossRef] [PubMed]
- Qiang, F.; Kang, K.W.; Han, H.K. Repeated dosing of piperine induced gene expression of P-glycoprotein via stimulated pregnane-X-receptor activity and altered pharmacokinetics of diltiazem in rats. Biopharm. Drug Dispos. 2012, 33, 446–454. [Google Scholar] [CrossRef] [PubMed]
- Rigalli, J.P.; Ciriaci, N.; Arias, A.; Ceballos, M.P.; Villanueva, S.S.; Luquita, M.G.; Mottino, A.D.; Ghanem, C.I.; Catania, V.A.; Ruiz, M.L. Regulation of multidrug resistance proteins by genistein in a hepatocarcinoma cell line: Impact on sorafenib cytotoxicity. PLoS ONE 2015, 10, e0119502. [Google Scholar] [CrossRef] [PubMed]
- Foster, B.C.; Foster, M.S.; Vandenhoek, S.; Krantis, A.; Budzinski, J.W.; Arnason, J.T.; Gallicano, K.D.; Choudri, S. An in vitro evaluation of human cytochrome P450 3A4 and P-glycoprotein inhibition by garlic. J. Pharm. Pharm. Sci. 2001, 4, 176–184. [Google Scholar] [PubMed]
- Berginc, K.; Milisav, I.; Kristl, A. Garlic flavonoids and organosulfur compounds: Impact on the hepatic pharmacokinetics of saquinavir and darunavir. Drug Metab. Pharmacokinet. 2010, 25, 521–530. [Google Scholar] [CrossRef] [PubMed]
- Berginc, K.; Zakelj, S.; Ursic, D.; Kristl, A. Aged garlic extract stimulates p-glycoprotein and multidrug resistance associated protein 2 mediated effluxes. Biol. Pharm. Bull. 2009, 32, 694–699. [Google Scholar] [CrossRef] [PubMed]
- Patel, J.; Buddha, B.; Dey, S.; Pal, D.; Mitra, A.K. In vitro interaction of the HIV protease inhibitor ritonavir with herbal constituents: Changes in P-gp and CYP3A4 activity. Am. J. Ther. 2004, 11, 262–277. [Google Scholar] [CrossRef] [PubMed]
- Kitagawa, S.; Nabekura, T.; Kamiyama, S. Inhibition of P-glycoprotein function by tea catechins in KB-C2 cells. J. Pharm. Pharmacol. 2004, 56, 1001–1005. [Google Scholar] [CrossRef] [PubMed]
- Anuchapreeda, S.; Leechanachai, P.; Smith, M.M.; Ambudkar, S.V.; Limtrakul, P.N. Modulation of P-glycoprotein expression and function by curcumin in multidrug-resistant human KB cells. Biochem. Pharmacol. 2002, 64, 573–582. [Google Scholar] [CrossRef]
- Romiti, N.; Pellati, F.; Nieri, P.; Benvenuti, S.; Adinolfi, B.; Chieli, E. P-Glycoprotein inhibitory activity of lipophilic constituents of Echinacea pallida roots in a human proximal tubular cell line. Planta Med. 2008, 74, 264–266. [Google Scholar] [CrossRef] [PubMed]
- Nabekura, T.; Yamaki, T.; Ueno, K.; Kitagawa, S. Inhibition of P-glycoprotein and multidrug resistance protein 1 by dietary phytochemicals. Cancer Chemother. Pharmacol. 2008, 62, 867–873. [Google Scholar] [CrossRef] [PubMed]
- Hellum, B.H.; Nilsen, O.G. In vitro inhibition of CYP3A4 metabolism and P-glycoprotein-mediated transport by trade herbal products. Basic Clin. Pharmacol. Toxicol. 2008, 102, 466–475. [Google Scholar] [CrossRef] [PubMed]
- Perloff, M.D.; von Moltke, L.L.; Stormer, E.; Shader, R.I.; Greenblatt, D.J. Saint John’s wort: An in vitro analysis of P-glycoprotein induction due to extended exposure. Br. J. Pharmacol. 2001, 134, 1601–1608. [Google Scholar] [CrossRef] [PubMed]
- Etheridge, A.S.; Black, S.R.; Patel, P.R.; So, J.; Mathews, J.M. An in vitro evaluation of cytochrome P450 inhibition and P-glycoprotein interaction with goldenseal, Ginkgo biloba, grape seed, milk thistle, and ginseng extracts and their constituents. Planta Med. 2007, 73, 731–741. [Google Scholar] [CrossRef] [PubMed]
- Demeule, M.; Brossard, M.; Turcotte, S.; Regina, A.; Jodoin, J.; Beliveau, R. Diallyl disulfide, a chemopreventive agent in garlic, induces multidrug resistance-associated protein 2 expression. Biochem. Biophys. Res. Commun. 2004, 324, 937–945. [Google Scholar] [CrossRef] [PubMed]
- Wlcek, K.; Koller, F.; Ferenci, P.; Stieger, B. Hepatocellular organic anion-transporting polypeptides (OATPs) and multidrug resistance-associated protein 2 (MRP2) are inhibited by silibinin. Drug Metab. Dispos. 2013, 41, 1522–1528. [Google Scholar] [CrossRef] [PubMed]
- Roth, M.; Timmermann, B.N.; Hagenbuch, B. Interactions of green tea catechins with organic anion-transporting polypeptides. Drug Metab. Dispos. 2011, 39, 920–926. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Wolkoff, A.W.; Morris, M.E. Flavonoids as a novel class of human organic anion-transporting polypeptide OATP1B1 (OATP-C) modulators. Drug Metab. Dispos. 2005, 33, 1666–1672. [Google Scholar] [CrossRef] [PubMed]
- Imanaga, J.; Kotegawa, T.; Imai, H.; Tsutsumi, K.; Yoshizato, T.; Ohyama, T.; Shirasaka, Y.; Tamai, I.; Tateishi, T.; Ohashi, K. The effects of the SLCO2B1 c.1457C > T polymorphism and apple juice on the pharmacokinetics of fexofenadine and midazolam in humans. Pharmacogenet. Genom. 2011, 21, 84–93. [Google Scholar] [CrossRef]
- Shirasaka, Y.; Shichiri, M.; Mori, T.; Nakanishi, T.; Tamai, I. Major active components in grapefruit, orange, and apple juices responsible for OATP2B1-mediated drug interactions. J. Pharm. Sci. 2013, 102, 280–288. [Google Scholar] [CrossRef] [PubMed]
- Fuchikami, H.; Satoh, H.; Tsujimoto, M.; Ohdo, S.; Ohtani, H.; Sawada, Y. Effects of herbal extracts on the function of human organic anion-transporting polypeptide OATP-B. Drug Metab. Dispos. 2006, 34, 577–582. [Google Scholar] [CrossRef] [PubMed]
- Rebello, S.; Zhao, S.; Hariry, S.; Dahlke, M.; Alexander, N.; Vapurcuyan, A.; Hanna, I.; Jarugula, V. Intestinal OATP1A2 inhibition as a potential mechanism for the effect of grapefruit juice on aliskiren pharmacokinetics in healthy subjects. Eur. J. Clin. Pharmacol. 2012, 68, 697–708. [Google Scholar] [CrossRef] [PubMed]
- Reddy, P.; Ellington, D.; Zhu, Y.; Zdrojewski, I.; Parent, S.J.; Harmatz, J.S.; Derendorf, H.; Greenblatt, D.J.; Browne, K., Jr. Serum concentrations and clinical effects of atorvastatin in patients taking grapefruit juice daily. Br. J. Clin. Pharmacol. 2011, 72, 434–441. [Google Scholar] [CrossRef] [PubMed]
- Lilja, J.J.; Backman, J.T.; Laitila, J.; Luurila, H.; Neuvonen, P.J. Itraconazole increases but grapefruit juice greatly decreases plasma concentrations of celiprolol. Clin. Pharmacol. Ther. 2003, 73, 192–198. [Google Scholar] [CrossRef] [PubMed]
- Edwards, D.J.; Fitzsimmons, M.E.; Schuetz, E.G.; Yasuda, K.; Ducharme, M.P.; Warbasse, L.H.; Woster, P.M.; Schuetz, J.D.; Watkins, P. 6′,7′-Dihydroxybergamottin in grapefruit juice and Seville orange juice: Effects on cyclosporine disposition, enterocyte CYP3A4, and P-glycoprotein. Clin. Pharmacol. Ther. 1999, 65, 237–244. [Google Scholar] [CrossRef]
- Lown, K.S.; Bailey, D.G.; Fontana, R.J.; Janardan, S.K.; Adair, C.H.; Fortlage, L.A.; Brown, M.B.; Guo, W.; Watkins, P.B. Grapefruit juice increases felodipine oral availability in humans by decreasing intestinal CYP3A protein expression. J. Clin. Investig. 1997, 99, 2545–2553. [Google Scholar] [CrossRef] [PubMed]
- Bailey, D.G.; Spence, J.D.; Munoz, C.; Arnold, J.M. Interaction of citrus juices with felodipine and nifedipine. Lancet 1991, 337, 268–269. [Google Scholar] [CrossRef]
- Kupferschmidt, H.H.; Fattinger, K.E.; Ha, H.R.; Follath, F.; Krahenbuhl, S. Grapefruit juice enhances the bioavailability of the HIV protease inhibitor saquinavir in man. Br. J. Clin. Pharmacol. 1998, 45, 355–359. [Google Scholar] [CrossRef] [PubMed]
- Lilja, J.J.; Neuvonen, M.; Neuvonen, P.J. Effects of regular consumption of grapefruit juice on the pharmacokinetics of simvastatin. Br. J. Clin. Pharmacol. 2004, 58, 56–60. [Google Scholar] [CrossRef] [PubMed]
- Tapaninen, T.; Neuvonen, P.J.; Niemi, M. Orange and apple juice greatly reduce the plasma concentrations of the OATP2B1 substrate aliskiren. Br. J. Clin. Pharmacol. 2011, 71, 718–726. [Google Scholar] [CrossRef] [PubMed]
- Lilja, J.J.; Juntti-Patinen, L.; Neuvonen, P.J. Orange juice substantially reduces the bioavailability of the beta-adrenergic-blocking agent celiprolol. Clin. Pharmacol. Ther. 2004, 75, 184–190. [Google Scholar] [CrossRef] [PubMed]
- Jeon, H.; Jang, I.J.; Lee, S.; Ohashi, K.; Kotegawa, T.; Ieiri, I.; Cho, J.-Y.; Yoon, S.H.; Shin, S.-G.; Yu, K.-S.; et al. Apple juice greatly reduces systemic exposure to atenolol. Br. J. Clin. Pharmacol. 2013, 75, 172–179. [Google Scholar] [CrossRef] [PubMed]
- Yamsani, S.K.; Yamsani, M.R. Effect of silymarin pretreatment on the bioavailability of domperidone in healthy human volunteers. Drug Metabol. Drug Interact. 2014, 29, 261–267. [Google Scholar] [CrossRef] [PubMed]
- Han, Y.; Guo, D.; Chen, Y.; Chen, Y.; Tan, Z.R.; Zhou, H.H. Effect of silymarin on the pharmacokinetics of losartan and its active metabolite E-3174 in healthy Chinese volunteers. Eur. J. Clin. Pharmacol. 2009, 65, 585–591. [Google Scholar] [CrossRef] [PubMed]
- Rajnarayana, K.; Reddy, M.S.; Vidyasagar, J.; Krishna, D.R. Study on the influence of silymarin pretreatment on metabolism and disposition of metronidazole. Arzneimittelforschung 2004, 54, 109–113. [Google Scholar] [CrossRef] [PubMed]
- Han, Y.; Guo, D.; Chen, Y.; Tan, Z.R.; Zhou, H.H. Effect of continuous silymarin administration on oral talinolol pharmacokinetics in healthy volunteers. Xenobiotica 2009, 39, 694–699. [Google Scholar] [CrossRef] [PubMed]
- Chow, H.H.; Hakim, I.A.; Vining, D.R.; Crowell, J.A.; Cordova, C.A.; Chew, W.M.; Xu, M.J.; Hsu, C.H.; Ranger-Moore, J.; Alberts, D.S. Effects of repeated green tea catechin administration on human cytochrome P450 activity. Cancer Epidemiol. Biomark. Prev. 2006, 15, 2473–2476. [Google Scholar] [CrossRef] [PubMed]
- Misaka, S.; Yatabe, J.; Muller, F.; Takano, K.; Kawabe, K.; Glaeser, H.; Yatabe, M.S.; Onoue, S.; Werba, J.P.; Watanabe, H.; et al. Green tea ingestion greatly reduces plasma concentrations of nadolol in healthy subjects. Clin. Pharmacol. Ther. 2014, 95, 432–438. [Google Scholar] [CrossRef] [PubMed]
- Cambria-Kiely, J.A. Effect of soy milk on warfarin efficacy. Ann. Pharmacother. 2002, 36, 1893–1896. [Google Scholar] [CrossRef] [PubMed]
- Johne, A.; Schmider, J.; Brockmoller, J.; Stadelmann, A.M.; Stormer, E.; Bauer, S.; Scholler, G.; Langheinrich, M.; Roots, I. Decreased plasma levels of amitriptyline and its metabolites on comedication with an extract from St. John’s wort (Hypericum perforatum). J. Clin. Psychopharmacol. 2002, 22, 46–54. [Google Scholar] [CrossRef] [PubMed]
- Van Strater, A.C.; Bogers, J.P. Interaction of St John’s wort (Hypericum perforatum) with clozapine. Int. Clin. Psychopharmacol. 2012, 27, 121–124. [Google Scholar] [CrossRef] [PubMed]
- Barone, G.W.; Gurley, B.J.; Ketel, B.L.; Lightfoot, M.L.; Abul-Ezz, S.R. Drug interaction between St. John’s wort and cyclosporine. Ann. Pharmacother. 2000, 34, 1013–1016. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.; Hamman, M.A.; Huang, S.M.; Lesko, L.J.; Hall, S.D. Effect of St John’s wort on the pharmacokinetics of fexofenadine. Clin. Pharmacol. Ther. 2002, 71, 414–420. [Google Scholar] [CrossRef] [PubMed]
- Piscitelli, S.C.; Burstein, A.H.; Chaitt, D.; Alfaro, R.M.; Falloon, J. Indinavir concentrations and St John’s wort. Lancet 2000, 355, 547–548. [Google Scholar] [CrossRef]
- Pathak, A.; Leger, P.; Bagheri, H.; Senard, J.M.; Boccalon, H.; Montastruc, J.L. Garlic interaction with fluindione: A case report. Therapie 2003, 58, 380–381. [Google Scholar] [CrossRef] [PubMed]
- Gorski, J.C.; Huang, S.M.; Pinto, A.; Hamman, M.A.; Hilligoss, J.K.; Zaheer, N.A.; Desai, M.; Miller, M.; Hall, S.D. The effect of echinacea (Echinacea purpurea root) on cytochrome P450 activity in vivo. Clin. Pharmacol. Ther. 2004, 75, 89–100. [Google Scholar] [CrossRef] [PubMed]
- Markowitz, J.S.; Donovan, J.L.; Lindsay DeVane, C.; Sipkes, L.; Chavin, K.D. Multiple-dose administration of Ginkgo biloba did not affect cytochrome P-450 2D6 or 3A4 activity in normal volunteers. J. Clin. Psychopharmacol. 2003, 23, 576–581. [Google Scholar] [CrossRef] [PubMed]
- Kim, K.A.; Park, P.W.; Park, J.Y. Short-term effect of quercetin on the pharmacokinetics of fexofenadine, a substrate of P-glycoprotein, in healthy volunteers. Eur. J. Clin. Pharmacol. 2009, 65, 609–614. [Google Scholar] [CrossRef] [PubMed]
- Uchida, S.; Yamada, H.; Li, X.D.; Maruyama, S.; Ohmori, Y.; Oki, T.; Watanabe, H.; Umegaki, K.; Ohashi, K.; Yamada, S. Effects of Ginkgo biloba extract on pharmacokinetics and pharmacodynamics of tolbutamide and midazolam in healthy volunteers. J. Clin. Pharmacol. 2006, 46, 1290–1298. [Google Scholar] [CrossRef] [PubMed]
- Fan, L.; Mao, X.Q.; Tao, G.Y.; Wang, G.; Jiang, F.; Chen, Y.; Li, Q.; Zhang, W.; Lei, H.P.; Hu, D.L.; et al. Effect of Schisandra chinensis extract and Ginkgo biloba extract on the pharmacokinetics of talinolol in healthy volunteers. Xenobiotica 2009, 39, 249–254. [Google Scholar] [CrossRef] [PubMed]
- Yin, O.Q.; Tomlinson, B.; Waye, M.M.; Chow, A.H.; Chow, M.S. Pharmacogenetics and herb-drug interactions: Experience with Ginkgo biloba and omeprazole. Pharmacogenetics 2004, 14, 841–850. [Google Scholar] [CrossRef] [PubMed]
- Malati, C.Y.; Robertson, S.M.; Hunt, J.D.; Chairez, C.; Alfaro, R.M.; Kovacs, J.A.; Penzak, S.R. Influence of Panax ginseng on cytochrome P450 (CYP)3A and P-glycoprotein (P-gp) activity in healthy participants. J. Clin. Pharmacol. 2012, 52, 932–979. [Google Scholar] [CrossRef] [PubMed]
- Hanley, M.J.; Cancalon, P.; Widmer, W.W.; Greenblatt, D.J. The effect of grapefruit juice on drug disposition. Exp. Opin. Drug Metab. Toxicol. 2011, 7, 267–286. [Google Scholar] [CrossRef] [PubMed]
- Seden, K.; Dickinson, L.; Khoo, S.; Back, D. Grapefruit-drug interactions. Drugs 2010, 70, 2373–2407. [Google Scholar] [CrossRef] [PubMed]
- Takanaga, H.; Ohnishi, A.; Matsuo, H.; Sawada, Y. Inhibition of vinblastine efflux mediated by P-glycoprotein by grapefruit juice components in Caco-2 cells. Biol. Pharm. Bull. 1998, 21, 1062–1066. [Google Scholar] [CrossRef] [PubMed]
- De Castro, W.V.; Mertens-Talcott, S.; Derendorf, H.; Butterweck, V. Grapefruit juice–drug interactions: Grapefruit juice and its components inhibit P-glycoprotein (ABCB1) mediated transport of talinolol in Caco-2 cells. J. Pharm. Sci. 2007, 96, 2808–2817. [Google Scholar] [CrossRef] [PubMed]
- Soldner, A.; Christians, U.; Susanto, M.; Wacher, V.J.; Silverman, J.A.; Benet, L.Z. Grapefruit juice activates P-glycoprotein-mediated drug transport. Pharm. Res. 1999, 16, 478–485. [Google Scholar] [CrossRef] [PubMed]
- Panchagnula, R.; Bansal, T.; Varma, M.V.; Kaul, C.L. Co-treatment with grapefruit juice inhibits while chronic administration activates intestinal P-glycoprotein-mediated drug efflux. Pharmazie 2005, 60, 922–927. [Google Scholar] [PubMed]
- Parker, R.B.; Yates, C.R.; Soberman, J.E.; Laizure, S.C. Effects of grapefruit juice on intestinal P-glycoprotein: Evaluation using digoxin in humans. Pharmacotherapy 2003, 23, 979–987. [Google Scholar] [CrossRef] [PubMed]
- An, G.; Mukker, J.K.; Derendorf, H.; Frye, R.F. Enzyme- and transporter-mediated beverage-drug interactions: An update on fruit juices and green tea. J. Clin. Pharmacol. 2015, 55, 1313–1331. [Google Scholar] [CrossRef] [PubMed]
- Shirasaka, Y.; Suzuki, K.; Nakanishi, T.; Tamai, I. Differential effect of grapefruit juice on intestinal absorption of statins due to inhibition of organic anion transporting polypeptide and/or P-glycoprotein. J. Pharm. Sci. 2011, 100, 3843–3853. [Google Scholar] [CrossRef] [PubMed]
- Mitsunaga, Y.; Takanaga, H.; Matsuo, H.; Naito, M.; Tsuruo, T.; Ohtani, H.; Sawada, Y. Effect of bioflavonoids on vincristine transport across blood-brain barrier. Eur. J. Pharmacol. 2000, 395, 193–201. [Google Scholar] [CrossRef]
- Honda, Y.; Ushigome, F.; Koyabu, N.; Morimoto, S.; Shoyama, Y.; Uchiumi, T.; Kuwano, M.; Ohtani, H.; Sawada, Y. Effects of grapefruit juice and orange juice components on P-glycoprotein- and MRP2-mediated drug efflux. Br. J. Pharmacol. 2004, 143, 856–864. [Google Scholar] [CrossRef] [PubMed]
- Malhotra, S.; Bailey, D.G.; Paine, M.F.; Watkins, P.B. Seville orange juice-felodipine interaction: Comparison with dilute grapefruit juice and involvement of furocoumarins. Clin. Pharmacol. Ther. 2001, 69, 14–23. [Google Scholar] [CrossRef] [PubMed]
- Shirasaka, Y.; Shichiri, M.; Murata, Y.; Mori, T.; Nakanishi, T.; Tamai, I. Long-lasting inhibitory effect of apple and orange juices, but not grapefruit juice, on OATP2B1-mediated drug absorption. Drug Metab. Dispos. 2013, 41, 615–621. [Google Scholar] [CrossRef] [PubMed]
- Dolton, M.J.; Roufogalis, B.D.; McLachlan, A.J. Fruit juices as perpetrators of drug interactions: The role of organic anion-transporting polypeptides. Clin. Pharmacol. Ther. 2012, 92, 622–630. [Google Scholar] [CrossRef] [PubMed]
- Saller, R.; Melzer, J.; Reichling, J.; Brignoli, R.; Meier, R. An updated systematic review of the pharmacology of silymarin. Forsch. Komplement. 2007, 14, 70–80. [Google Scholar] [CrossRef]
- Simanek, V.; Kren, V.; Ulrichova, J.; Vicar, J.; Cvak, L. Silymarin: What is in the name...? An appeal for a change of editorial policy. Hepatology 2000, 32, 442–444. [Google Scholar] [CrossRef] [PubMed]
- Saller, R.; Brignoli, R.; Melzer, J.; Meier, R. An updated systematic review with meta-analysis for the clinical evidence of silymarin. Forsch. Komplement. 2008, 15, 9–20. [Google Scholar] [CrossRef] [PubMed]
- Loguercio, C.; Festi, D. Silybin and the liver: From basic research to clinical practice. World J. Gastroenterol. 2011, 17, 2288–2301. [Google Scholar] [CrossRef] [PubMed]
- Ganzert, M.; Felgenhauer, N.; Schuster, T.; Eyer, F.; Gourdin, C.; Zilker, T. Silibinin und Kombination von Silibinin und Penicillin im Vergleich [Amatoxin poisoning—comparison of silibinin with a combination of silibinin and penicillin]. Dtsch. Med. Wochenschr. 2008, 133, 2261–2267. [Google Scholar] [CrossRef] [PubMed]
- Garcia, J.; Costa, V.M.; Carvalho, A.; Baptista, P.; de Pinho, P.G.; de Lourdes Bastos, M.; Carvalho, F. Amanita phalloides poisoning: Mechanisms of toxicity and treatment. Food Chem. Toxicol. 2015, 86, 41–55. [Google Scholar] [CrossRef] [PubMed]
- Murtaza, G.; Latif, U.; Najam-Ul-Haq, M.; Sajjad, A.; Karim, S.; Akhtar, M.; Hussain, I. Resveratrol: An active natural compound in red wines for health. J. Food Drug Anal. 2013, 21, 1–12. [Google Scholar]
- Letschert, K.; Faulstich, H.; Keller, D.; Keppler, D. Molecular characterization and inhibition ofamanitin uptake into human hepatocytes. Toxicol. Sci. 2006, 91, 140–149. [Google Scholar] [CrossRef] [PubMed]
- Fehrenbach, T.; Cui, Y.; Faulstich, H.; Keppler, D. Characterization of the transport of the bicyclic peptide phalloidin by human hepatic transport proteins. Naunyn Schmiedeberg’s Arch. Pharmacol. 2013, 368, 415–420. [Google Scholar] [CrossRef] [PubMed]
- Meier-Abt, F.; Faulstich, H.; Hagenbuch, B. Identification of phalloidin uptake systems of rat and human liver. Biochim. Biophys. Acta 2004, 1664, 64–69. [Google Scholar] [CrossRef] [PubMed]
- Gundala, S.; Wells, L.D.; Milliano, M.T.; Talkad, V.; Luxon, B.A.; Neuschwander-Tetri, B.A. The hepatocellular bile acid transporter Ntcp facilitates uptake of the lethal mushroom toxin α-amanitin. Arch. Toxicol. 2004, 78, 68–73. [Google Scholar] [CrossRef] [PubMed]
- Ferenci, P.; Scherzer, T.M.; Kerschner, H.; Rutter, K.; Beinhardt, S.; Hofer, H.; Schöniger-Hekele, M.; Holzmann, H.; Steindl-Munda, P. Silibinin is a potent antiviralagent in patients with chronic hepatitis C not responding to pegylated interferon/ribavirin therapy. Gastroenterology 2008, 135, 1561–1567. [Google Scholar] [CrossRef] [PubMed]
- Biermer, M.; Berg, T. Rapid suppression of hepatitis C viremia induced by intravenous silibinin plus ribavirin. Gastroenterology 2009, 137, 390–391. [Google Scholar] [CrossRef] [PubMed]
- Rutter, K.; Scherzer, T.M.; Beinhardt, S.; Kerschner, H.; Stattermayer, A.F.; Hofer, H.; Popow-Kraupp, T.; Steindl-Munda, P.; Ferenci, P. Intravenous silibinin as ‘rescue treatment’ for on-treatment non-responders to pegylated interferon/ribavirin combination therapy. Antivir. Ther. 2011, 16, 1327–1333. [Google Scholar] [CrossRef] [PubMed]
- Braun, D.L.; Rauch, A.; Aouri, M.; Durisch, N.; Eberhard, N.; Anagnostopoulos, A.; Ledergerber, B.; Müllhaupt, B.; Metzner, K.J.; Decosterd, L.; et al. A lead-in with silibinin prior to triple therapy translates into favorable treatment outcomes in difficult-to-treat HIV/hepatitis C coinfected patients. PLoS ONE 2015, 10, e0133028. [Google Scholar] [CrossRef] [PubMed]
- Stieger, B.; Heger, M.; de Graaf, W.; Paumgartner, G.; van Gulik, T. The emerging role of transport systems in liver function tests. Eur. J. Pharmacol. 2012, 675, 1–5. [Google Scholar] [CrossRef] [PubMed]
- Misaka, S.; Kawabe, K.; Onoue, S.; Werba, J.P.; Giroli, M.; Watanabe, H.; Yamada, S. Green tea extract affects the cytochromeP450 3A activity and pharmacokinetics of simvastatin in rats. Drug Metab. Pharmacokinet. 2013, 28, 514–518. [Google Scholar] [CrossRef] [PubMed]
- Jager, W.; Winter, O.; Halper, B.; Salamon, A.; Sartori, M.; Gajdzik, L.; Hamilton, G.; Theyer, G.; Graf, J.; Thalhammer, T. Modulation of liver canalicular¨ transport processes by the tyrosine-kinase inhibitor genistein: Implications of genistein metabolism in the rat. Hepatology 1997, 26, 1467–1476. [Google Scholar] [CrossRef] [PubMed]
- Cheng, T.O. Potential interaction between soy milk and warfarin. Am. Fam. Physician 2004, 70, 1231. [Google Scholar] [PubMed]
- Kasper, S.; Caraci, F.; Forti, B.; Drago, F.; Aguglia, E. Efficacy and tolerability of Hypericum extract for the treatment of mild to moderate depression. Eur. Neuropsychopharmacol. 2010, 20, 747–765. [Google Scholar] [CrossRef] [PubMed]
- Nahrstedt, A.; Butterweck, V. Lessons learned from herbal medicinal products: The example of St. John’s wort. J. Nat. Prod. 2010, 73, 1015–1021. [Google Scholar] [CrossRef] [PubMed]
- Butterweck, V.; Schmidt, M. St. John’s wort: Role of active compounds for its mechanism of action and efficacy. Wien. Med. Wochenschr. 2007, 157, 356–361. [Google Scholar] [CrossRef] [PubMed]
- Schmidt, M.; Butterweck, V. The mechanisms of action of St. John’s wort: An update. Wien. Med. Wochenschr. 2015, 165, 229–235. [Google Scholar] [CrossRef] [PubMed]
- Zhou, S.; Chan, E.; Pan, S.Q.; Huang, M.; Lee, E.J. Pharmacokinetic interactions of drugs with St. John’s wort. J. Psychopharmacol. 2004, 18, 262–276. [Google Scholar] [CrossRef] [PubMed]
- Russo, E.; Scicchitano, F.; Whalley, B.J.; Mazzitello, C.; Ciriaco, M.; Esposito, S.; Patanè, M.; Upton, R.; Pugliese, M.; Chimirri, S.; et al. Hypericum perforatum: Pharmacokinetic, mechanism of action, tolerability, and clinical drug–drug interactions. Phytother. Res. 2014, 28, 643–655. [Google Scholar] [CrossRef] [PubMed]
- Moore, L.B.; Goodwin, B.; Jones, S.A.; Wisely, G.B.; Serabjit-Singh, C.J.; Willson, T.M.; Collins, J.L.; Kliewer, S.K. St. John’s wort induces hepatic drug metabolism through activation of the pregnane X receptor. Proc. Natl. Acad. Sci. USA 2000, 97, 7500–7502. [Google Scholar] [CrossRef] [PubMed]
- Obach, R.S. Inhibition of human cytochrome P450 enzymes by constituents of St. John’s wort, an herbal preparation used in the treatment of depression. J. Pharmacol. Exp. Ther. 2000, 294, 88–95. [Google Scholar] [PubMed]
- Ruschitzka, F.; Meier, P.J.; Turina, M.; Luscher, T.F.; Noll, G. Acute heart transplant rejection due to Saint John’s wort. Lancet 2000, 355, 548–549. [Google Scholar] [CrossRef]
- Breidenbach, T.; Kliem, V.; Burg, M.; Radermacher, J.; Hoffmann, M.W.; Klempnauer, J. Profound drop of cyclosporin A whole blood trough levels caused by St. John’s wort (Hypericum perforatum). Transplantation 2000, 69, 2229–2230. [Google Scholar] [CrossRef] [PubMed]
- Ernst, E. St. John’s wort supplements endanger the success of organ transplantation. Arch. Surg. 2002, 137, 316–319. [Google Scholar] [CrossRef] [PubMed]
- Borrelli, F.; Izzo, A.A. Herb–drug interactions with St. John’s wort (Hypericum perforatum): An update on clinical observations. AAPS J. 2009, 11, 710–727. [Google Scholar] [CrossRef] [PubMed]
- Rahimi, R.; Abdollahi, M. An update on the ability of St. John’s wort to affect the metabolism ofother drugs. Exp. Opin. Drug Metab. Toxicol. 2012, 8, 691–708. [Google Scholar] [CrossRef] [PubMed]
- Mannel, M. Drug interactions with St. John’s wort: Mechanisms and clinical implications. Drug Saf. 2004, 27, 773–797. [Google Scholar] [CrossRef] [PubMed]
- Posadzki, P.; Watson, L.; Ernst, E. Herb–drug interactions: An overview of systematic reviews. Br. J. Clin. Pharmacol. 2013, 75, 603–618. [Google Scholar] [CrossRef] [PubMed]
- Pan, S.Y.; Litscher, G.; Gao, S.H.; Zhou, S.F.; Yu, Z.L.; Chen, H.Q.; Zhang, S.F.; Tang, M.K.; Sun, J.N.; Ko, K.M. Historical perspective of traditional indigenous medical practices: The current renaissance and conservation of herbal resources. Evid.-Based Complement. Altern. Med. 2014, 2014, 525340. [Google Scholar] [CrossRef] [PubMed]
- Pan, S.Y.; Litscher, G.; Chan, K.; Yu, Z.L.; Chen, H.Q.; Ko, K.M. Traditional medicines in the world: Where to go next? Evid. Based Complement. Alternat. Med. 2014, 2014, 739895. [Google Scholar] [CrossRef] [PubMed]
- Hong, M.; Li, S.; Tan, H.Y.; Wang, N.; Tsao, S.W.; Feng, Y. Current status of herbal medicines inchronic liver disease therapy: The biological effects, molecular targets and future prospects. Int. J. Mol. Sci. 2015, 16, 28705–28745. [Google Scholar] [CrossRef] [PubMed]
- Seeff, L.B.; Curto, T.M.; Szabo, G.; Everson, G.T.; Bonkovsky, H.L.; Dienstag, J.L.; Shiffman, M.L.; Lindsay, K.L.; Lok, A.S.; Di Bisceglie, A.M.; et al. Herbal product use bypersons enrolled in the Hepatitis C Antiviral Long-Term Treatment Against Cirrhosis (HALT-C) Trial. Hepatology 2008, 47, 605–612. [Google Scholar] [CrossRef] [PubMed]
- Struppler, A.; Rossler, H. Choleretic effect of artichoke extract. Med. Monatsschrift 1957, 11, 221–223. (In German) [Google Scholar]
- Kraft, K. Artichoke leaf extract—Recent findings reflecting effects on lipid metabolism, liver and gastrointestinal tracts. Phytomedicine 1997, 4, 369–378. [Google Scholar] [CrossRef]
- Preziosi, P.; Loscalzo, B. Pharmacological properties of 1, 4 dicaffeylquinic acid, the active principle of Cynara scolimus. Arch. Int. Pharmacodyn. Ther. 1958, 117, 63–80. [Google Scholar] [PubMed]
- Kirchhoff, R.; Beckers, C.; Kirchhoff, G.M.; Trinczek-Gartner, H.; Petrowicz, O.; Reimann, H.J. Increase in choleresis by means of artichoke extract. Phytomedicine 1994, 1, 107–115. [Google Scholar] [CrossRef]
- Salem, M.B.; Affes, H.; Ksouda, K.; Dhouibi, R.; Sahnoun, Z.; Hammami, S.; Zeghal, K.M. Pharmacological studies of artichoke leaf extract and their health benefits. Plant Foods Hum. Nutr. 2015, 70, 441–453. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Z.Y.; Si, D.Y.; Yi, X.L.; Liu, C.X. Inhibitory effect of medicinal plant-derived carboxylic acids on the human transporters hOAT1, hOAT3, hOATP1B1, and hOATP2B1. Chin. J. Nat. Med. 2014, 12, 131–138. [Google Scholar] [CrossRef]
- Tang, L.; Li, Y.; Chen, W.Y.; Zeng, S.; Dong, L.N.; Peng, X.J.; Jiang, W.; Hu, M.; Liu, Z.Q. Breast cancer resistance protein-mediated efflux of luteolin glucuronides in HeLa cells overexpressing UDP-glucuronosyltransferase 1A9. Pharm. Res. 2014, 31, 847–860. [Google Scholar] [CrossRef] [PubMed]
- Tome-Carneiro, J.; Larrosa, M.; Yánez-Gascõn, M.J.; Dávalos, A.; Gil-Zamorano, J.; Gonzálvez, M.; García-Almagro, F.J.; Ruiz Ros, J.A.; Tomás-Barberán, F.A.; Espín, J.C.; et al. One-year’ supplementation with a grape extract containing resveratrol modulates inflammatory-related microRNAs and cytokines expression in peripheral blood mononuclear cells of type 2 diabetes and hypertensive patients with coronary artery disease. Pharmacol. Res. 2013, 72, 69–82. [Google Scholar] [PubMed]
- Wahab, A.; Gao, K.; Jia, C.; Zhang, F.; Tian, G.; Murtaza, G.; Chen, J. Significance of resveratrol in clinical management of chronic diseases. Moleicules 2017, 22, 1329. [Google Scholar] [CrossRef] [PubMed]
- Urpí-Sarda, M.; Jàuregui, O.; Lamuela-Raventós, R.M.; Jaeger, W.; Miksits, M.; Covas, M.I.; Andres-Lacueva, C. Uptake of diet resveratrol into the human low-density lipoprotein. Identification and quantification of resveratrol metabolites by liquid chromatography coupled with tandem mass spectrometry. Anal. Chem. 2005, 77, 3149–3155. [Google Scholar] [CrossRef] [PubMed]
- Walle, T.; Hsieh, F.; DeLegge, M.H.; Oatis, J.E., Jr.; Walle, U.K. High absorption but very low bioavailability of oral resveratrol in humans. Drug Metab. Dispos. 2004, 32, 1377–1382. [Google Scholar] [CrossRef] [PubMed]
- Van de Wetering, K.; Burkon, A.; Feddema, W.; Bot, A.; de Jonge, H.; Somoza, V.; Borst, P. Intestinal breast cancer resistance protein (BCRP)/Bcrp1 and multidrug resistance protein 3 (MRP3)/Mrp3 are involved in the pharmacokinetics of resveratrol. Mol. Pharmacol. 2009, 75, 876–885. [Google Scholar] [CrossRef] [PubMed]
- Henry, C.; Vitrac, X.; Decendit, A.; Ennamany, R.; Krisa, S.; Merillon, J.M. Cellular uptake and efflux of trans-piceid and its aglycone trans-resveratrol on the apical membrane of human intestinal Caco-2 cells. J. Agric. Food Chem. 2005, 53, 798–803. [Google Scholar] [CrossRef] [PubMed]
- Al-Abd, A.M.; Mahmoud, A.M.; El-Sherbiny, G.A.; El-Moselhy, M.A.; Nofal, S.M.; El-Latif, H.A.; El-Eraky, W.I.; El-Shemy, H.A. Resveratrol enhances the cytotoxic profile of docetaxel and doxorubicin in solid tumour cell lines in vitro. Cell Prolif. 2011, 44, 591–601. [Google Scholar] [CrossRef] [PubMed]
- Jia, Y.; Liu, Z.; Huo, X.; Wang, C.; Meng, Q.; Liu, Q.; Sun, H.; Sun, P.; Yang, X.; Shu, X.; et al. Enhancement effect of resveratrol on the intestinal absorption of bestatin by regulating PEPT1, MDR1 and MRP2 in vivo and in vitro. Int. J. Pharm. 2015, 495, 588–598. [Google Scholar] [CrossRef] [PubMed]
- Zhu, Y.; He, W.; Gao, X.; Li, B.; Mei, C.; Xu, R.; Chen, H. Resveratrol overcomes gefitinib resistance by increasing the intracellular gefitinib concentration and triggering apoptosis, autophagy and senescence in PC9/G NSCLC cells. Sci. Rep. 2015, 5, 17730. [Google Scholar] [CrossRef] [PubMed]
- Patel, K.R.; Brown, V.A.; Jones, D.J.; Britton, R.G.; Hemingway, D.; Miller, A.S.; West, K.P.; Booth, T.D.; Perloff, M.; Crowell, J.A.; et al. Clinical pharmacology of resveratrol and its metabolites in colorectal cancer patients. Cancer Res. 2010, 70, 7392–7399. [Google Scholar] [CrossRef] [PubMed]
- Burkon, A.; Somoza, V. Quantification of free and protein-bound trans-resveratrol metabolites and identification of trans-resveratrol-C/O-conjugated diglucuronides—Two novel resveratrol metabolites in human plasma. Mol. Nutr. Food Res. 2008, 52, 549–557. [Google Scholar] [CrossRef] [PubMed]
- Chow, H.H.; Garland, L.L.; Hsu, C.H.; Vining, D.R.; Chew, W.M.; Miller, J.A.; Perloff, M.; Crowell, J.A.; Alberts, D.S. Resveratrol modulates drug- and carcinogen-metabolizing enzymes in a healthy volunteer study. Cancer Prev. Res. 2010, 3, 1168–1175. [Google Scholar] [CrossRef] [PubMed]
- Srovnalova, A.; Svecarova, M.; Zapletalova, M.K.; Anzenbacher, P.; Bachleda, P.; Anzenbacherova, E.; Dvorak, Z. Effects of anthocyanidins and anthocyanins on the expression and catalytic activities of CYP2A6, CYP2B6, CYP2C9, and CYP3A4 in primary human hepatocytes and human liver microsomes. J. Agric. Food Chem. 2014, 62, 789–797. [Google Scholar] [CrossRef] [PubMed]
- Szotaková, B.; Bártíkova, H.; Hlaváćcovă, J.; Bouśovă, I.; Skálová, L. Inhibitory effect of anthocyanidinś on hepatic glutathione S-transferase, UDP-glucuronosyltransferase and carbonyl reductase activities in rat and human. Xenobiotica 2013, 43, 679–685. [Google Scholar] [CrossRef] [PubMed]
- Dreiseitel, A.; Oosterhuis, B.; Vukman, K.V.; Schreier, P.; Oehme, A.; Locher, S.; Hajak, G.; Sand, P.G. Berry anthocyanins and anthocyanidins exhibit distinct affinities for the efflux transporters BCRP and MDR1. Br. J. Pharmacol. 2009, 158, 1942–1950. [Google Scholar] [CrossRef] [PubMed]
- Srinivas, N.R. Cranberry juice ingestion and clinical drug-drug interaction potentials: Review of case studies and perspectives. J. Pharm. Pharm. Sci. 2013, 16, 289–303. [Google Scholar] [CrossRef] [PubMed]
- Hamann, G.L.; Campbell, J.D.; George, C.M. Warfarin-cranberry juice interaction. Ann. Pharmacother. 2011, 45, e17. [Google Scholar] [CrossRef] [PubMed]
- Riha, J.; Brenner, S.; Srovnalova, A.; Klameth, L.; Dvorak, Z.; Jäger, W.; Thalhammer, T. Effects of anthocyans on the expression of organic anion transporting polypeptides (SLCOs/OATPs) in primary human hepatocytes. Food Funct. 2015, 6, 772–779. [Google Scholar] [CrossRef] [PubMed]
- Hansen, T.S.; Nilsen, O.G. Echinacea purpurea and P-glycoprotein drug transport in Caco-2 cells. Phytother. Res. 2009, 23, 86–91. [Google Scholar] [CrossRef] [PubMed]
- Velpandian, T.; Jasuja, R.; Bhardwaj, R.K.; Jaiswal, J.; Gupta, S.K. Piperine in food: Interference in the pharmacokinetics of phenytoin. Eur. J. Drug Metab. Pharmacokinet. 2001, 26, 241–247. [Google Scholar] [CrossRef] [PubMed]
Sample Availability: Not Available. |
| Transporter | Botanicals and Their Effect | References | |
|---|---|---|---|
| BCRP | Inhibitor | Cimicifuga racemosa extract | [39] |
| Curcumin (Curcuma longa flavonoid) | [40] | ||
| Kaempferol (Gingko biloba flavonoid) | [41] | ||
| Naringenin (Grapefruit flavonoid) | [41] | ||
| Genistein (Glycine max flavonoid) | [42] | ||
| Silybum marianum extract | [39] | ||
| MDR1 | Inducer | Garlic extract | [43] |
| Grapefruit juice | [44] | ||
| Hypericum perforatum extract | [45] | ||
| Bodine (Peumus boldo flavonoid) | [46] | ||
| Piperine (Piper nigrum flavonoid) | [47] | ||
| Genistein (Glycine max flavonoid) | [48] | ||
| Inhibitor | Garlic extract | [49] | |
| Tangeretin, nobiletin, rutin, allicin (Garlic flavonoid) | [50,51,52] | ||
| epigallocatechin gallate, epicatechin gallate, quercetin (Camellia sinensis flavonoid) | [53] | ||
| Curcumin (Curcuma longa flavonoid) | [54] | ||
| Echinacea extract | [55] | ||
| Glycyrrhetinic acid (Glycyrrhiza glabra flavonoid) | [56] | ||
| Gingko biloba extract | [57] | ||
| Hypericum perforatum extract | [58] | ||
| Panax ginseng extract | [59] | ||
| MRP1 | Inhibitor | Glycyrrhetinic acid (Glycyrrhiza glabra flavonoid) | [56] |
| MRP2 | Inducer | Garlic extract | [51] |
| Diallyl disulfide (Garlic flavonoid) | [60] | ||
| Bodine (Peumus boldo flavonoid) | [46] | ||
| Genistein (Glycine max flavonoid) | [48] | ||
| Inhibitor | Curcumin (Curcuma longa flavonoid) | [40] | |
| Silybin A + silybin B (Silybum marianum flavonoids) | [61] | ||
| OATP1A2 | Inhibitor | Camellia sinensis extract, epicatechin gallate, epigallocatechin gallate | [62] |
| Grapefruit juice | [44] | ||
| Orange juice | [44] | ||
| OATP1B1 | Inhibitor | Epigallocatechin gallate, epicatechin gallate (Camellia sinensis flavonoid) | [62] |
| Genistein (Glycine max flavonoid) | [63] | ||
| Silybin A + silybin B (Silybum marianum flavonoids) | [61] | ||
| OATP1B3 | Inhibitor | Silybin A + silybin B (Silybum marianum flavonoids) | [61] |
| OATP2B1 | Inhibitor | Apple juice | [64] |
| Apple juice, phloridzin, phloretin | [64,65] | ||
| Camellia sinensis extract, epicatechin gallate, epigallocatechin gallate | [66] | ||
| Cimicifuga racemosa extract | [66] | ||
| Echinacea extract | [66] | ||
| Kaempferol (Gingko biloba flavonoid) | [66] | ||
| Grapefruit juice | [44] | ||
| Orange juice | [44] | ||
| Glycine max extract | [66] | ||
| Silybin A + silybin B (Silybum marianum flavonoids) | [61] | ||
| NTCP | Inhibitor | Apple juice | [44] |
| BSEP | Inducer | Bodine (Peumus boldo flavonoid) | [46] |
| Drugs | Botanical Supplements | Effect on Transporters/Enzymes | Pharmacokinetic Change | Refs. | ||
|---|---|---|---|---|---|---|
| AUC | Cmax | PR | ||||
| Aliskiren | Grapefruit juice | ↓ intestinal OATP1A2 | ↓ | ↓ | - | [67] |
| Atorvastatin | ↓ CYP3A4 | - | ↑ | - | [68] | |
| Celiprolol | ↓ OATPs | ↓ | ↓ | - | [69] | |
| Cyclosporine | ↓ CYP3A4 | ↑ | ↑ | - | [70] | |
| Felodipine | ↓ CYP3A4 | ↑ | ↑ | ↑ | [71] | |
| Fexofenadine | ↓ intestinal OATP1A2 | ↓ | ↓ | - | [44] | |
| Nifedipine | ↓ CYPs | ↑ | ↑ | - | [72] | |
| Saquinavir | ↓ CYP3A4 | ↑ | ↑ | - | [73] | |
| Simvastatin | ↓ CYP3A4 | ↑ | ↑ | - | [74] | |
| Aliskiren | Orange juice | ↓ OATP2B1 | ↓ | ↓ | ↓ | [75] |
| Celiprolol | ↓ OATPs | ↓ | ↓ | - | [76] | |
| Fexofenadine | ↓ OATP1A2 | ↓ | ↓ | - | [44] | |
| Aliskiren | Apple juice | ↓ OATP2B1 | ↓ | ↓ | ↓ | [75] |
| Atenolol | ↓ OATP2B1 | ↓ | ↓ | - | [77] | |
| Fexofenadine | ↓ OATP1A2 | ↓ | ↓ | - | [44] | |
| Fexofenadine | ↓ OATP2B1 | ↓ | ↓ | - | [67] | |
| Domperidone | Silybum marianum | ↓ CYP3A4, ↓ MDR1 | ↑ | ↑ | - | [78] |
| Losartan | ↓ CYP3A4 | ↑ | ↑ | - | [79] | |
| Metronidazole | ↑ MDR1, ↑ CYP3A4 | ↓ | ↓ | - | [80] | |
| Talinol | ↓ MDR1 | ↑ | ↑ | - | [81] | |
| Buspirone | Camelia sinensis | ↓ CYP3A4 | ↓ | - | - | [82] |
| Nadolol | ↓ OATP1A2 | ↓ | ↓ | ↓ | [83] | |
| Warfarin | Soybean | ↓ OATPs | - | - | ↓ | [84] |
| Amitriptyline | Hypericum perforatum | ↑ MDR1, ↑ CYP3A4 | ↓ | ↓ | - | [85] |
| Clozapine | ↑ MDR1, ↑ CYPs | - | ↓ | - | [86] | |
| Cyclosporine | ↑ MDR1, ↑ CYP3A4 | ↓ | - | - | [87] | |
| Digoxin | ↑ MDR1, ↑ CYP3A4 | ↓ | ↓ | - | [45] | |
| Fexofenadine (single dose) | ↓ MDR1 | ↑ | ↑ | - | [88] | |
| Fexofenadine (multiple dosing) | ↓ MDR1 | ↓ | ↓ | - | [88] | |
| Indinavir | ↑ MDR1, ↑ CYP3A4 | ↓ | - | - | [89] | |
| Fluindione | Allium sativum | ↑ CYPs | - | - | ↓ | [90] |
| Saquinavir | ↑ intestinal MDR1 | ↓ | ↓ | [43] | ||
| Midazolam | Echinacea pupurea | ↑ hepatic CYP3A | ↓ | - | - | [91] |
| Alprazolam | Gingko biloba | ↓ CYP3A4 | ↓ | - | - | [92] |
| Midazolam | ↑ CYP3A | ↓ | ↓ | - | [93] | |
| Midazolam | ↓ CYP3A4 | ↑ | - | - | [94] | |
| Talinol | ↓MDR1 | ↓ | ↓ | - | [95] | |
| Omeprazole | ↑ CYP2C19 | ↓ | - | - | [96] | |
| Midazolam | Panax ginseng | ↑ CYP3A | ↓ | ↓ | - | [97] |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Murtaza, G.; Ullah, N.; Mukhtar, F.; Nawazish, S.; Muneer, S.; Mariam. Phytotherapeutics: The Emerging Role of Intestinal and Hepatocellular Transporters in Drug Interactions with Botanical Supplements. Molecules 2017, 22, 1699. https://doi.org/10.3390/molecules22101699
Murtaza G, Ullah N, Mukhtar F, Nawazish S, Muneer S, Mariam. Phytotherapeutics: The Emerging Role of Intestinal and Hepatocellular Transporters in Drug Interactions with Botanical Supplements. Molecules. 2017; 22(10):1699. https://doi.org/10.3390/molecules22101699
Chicago/Turabian StyleMurtaza, Ghulam, Naveed Ullah, Farah Mukhtar, Shamyla Nawazish, Saiqa Muneer, and Mariam. 2017. "Phytotherapeutics: The Emerging Role of Intestinal and Hepatocellular Transporters in Drug Interactions with Botanical Supplements" Molecules 22, no. 10: 1699. https://doi.org/10.3390/molecules22101699
APA StyleMurtaza, G., Ullah, N., Mukhtar, F., Nawazish, S., Muneer, S., & Mariam. (2017). Phytotherapeutics: The Emerging Role of Intestinal and Hepatocellular Transporters in Drug Interactions with Botanical Supplements. Molecules, 22(10), 1699. https://doi.org/10.3390/molecules22101699




