Aspalathin Reverts Doxorubicin-Induced Cardiotoxicity through Increased Autophagy and Decreased Expression of p53/mTOR/p62 Signaling
Abstract
:1. Introduction
2. Results
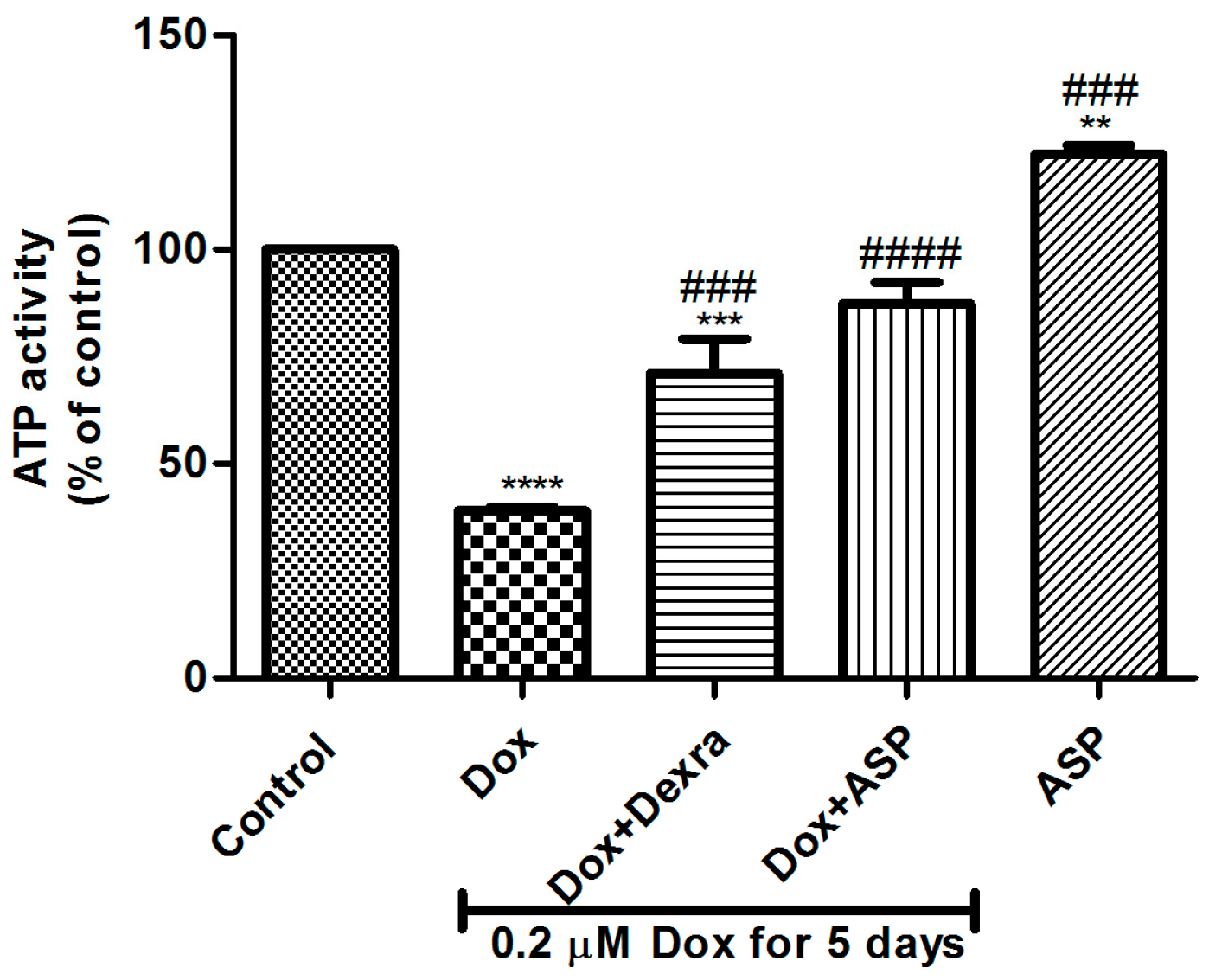
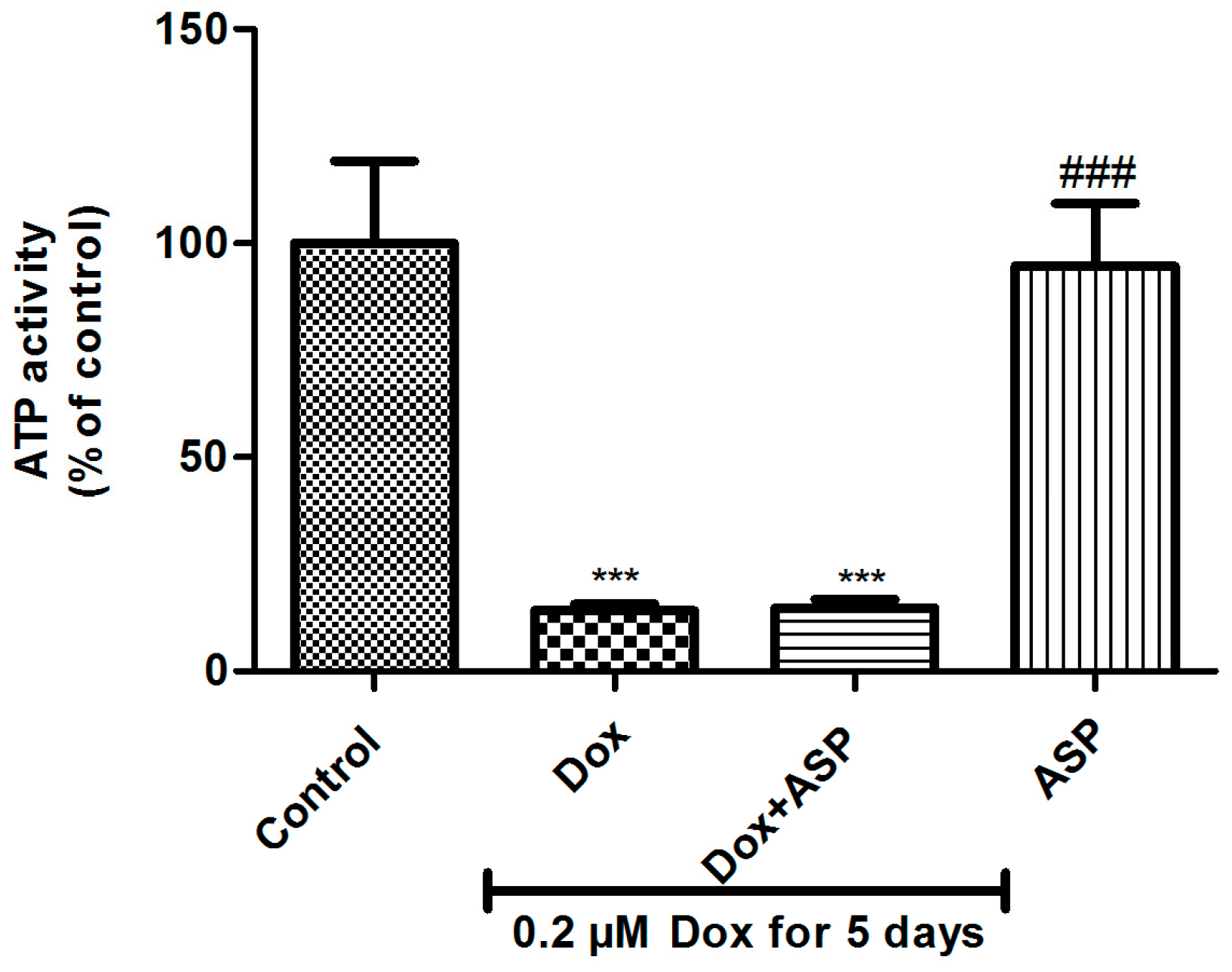
2.1. The Effect of ASP Treatment on ATP Activity as a Measurement of Metabolic Activity in H9c2 Cells
2.2. ASP Decrease Caspase 3/7 Activity and Reverts Dox-Induced Inhibition of Autophagy in H9c2 Cells
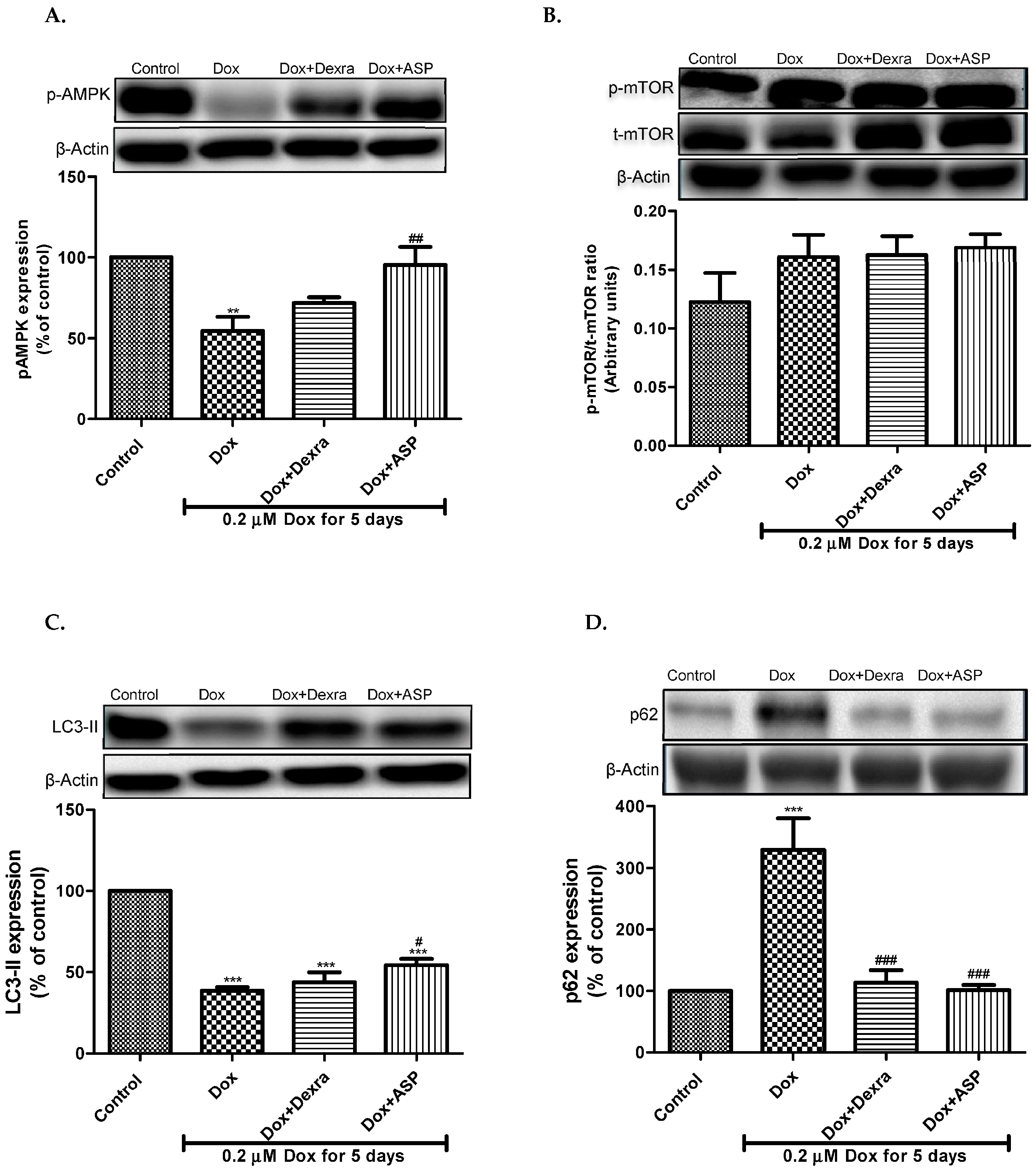
2.2.1. The Effect of ASP on p-AMPK Expression
2.2.2. The Effect of ASP on the Expression of mTOR
2.2.3. The Effect of ASP on the Expression of LC3-II
2.2.4. The Effect of ASP on the Expression of p62
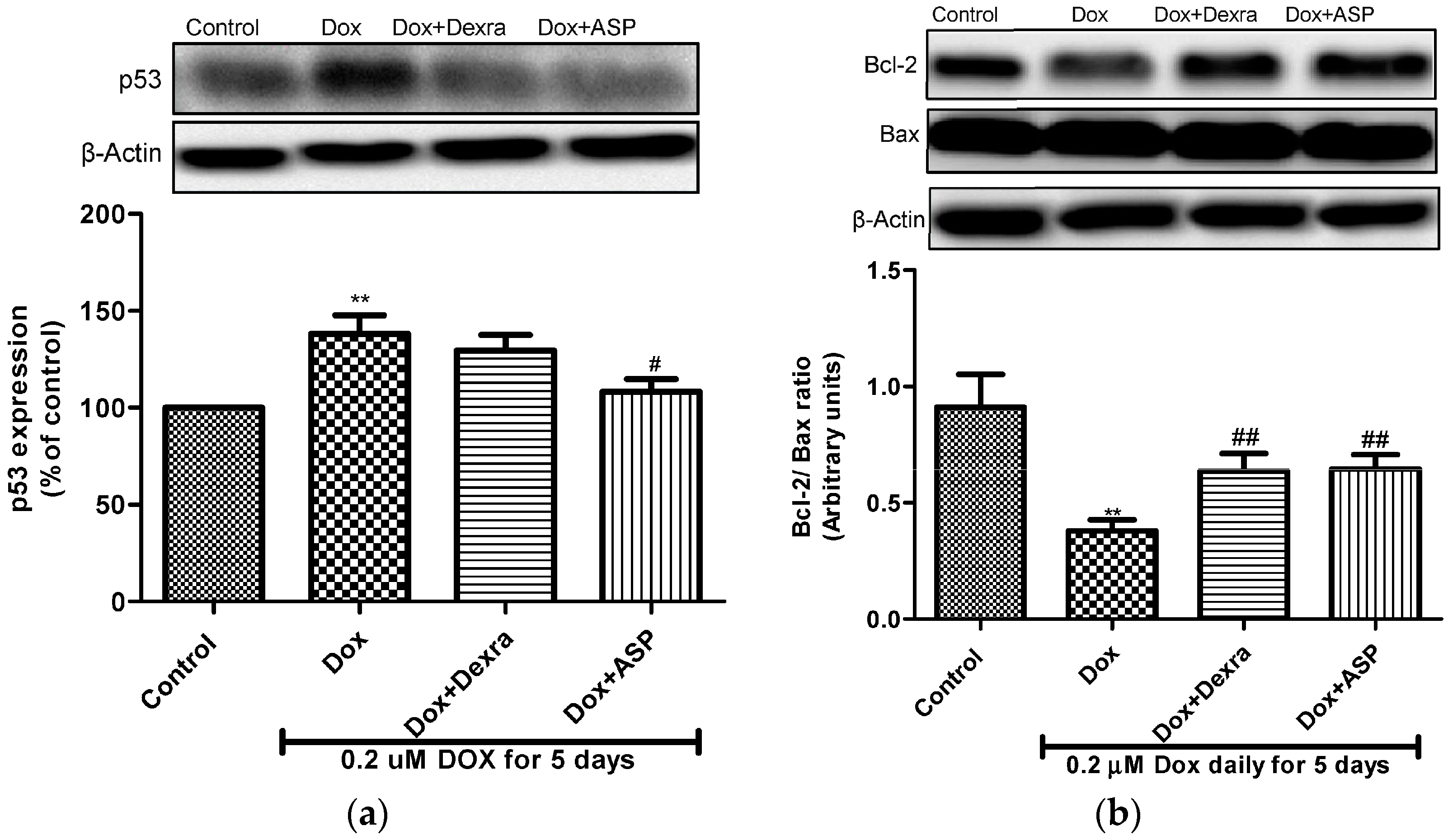
2.2.5. The Effect of ASP on p53 Expression
2.2.6. The Effect of ASP on the Bcl-2/Bax Ratio
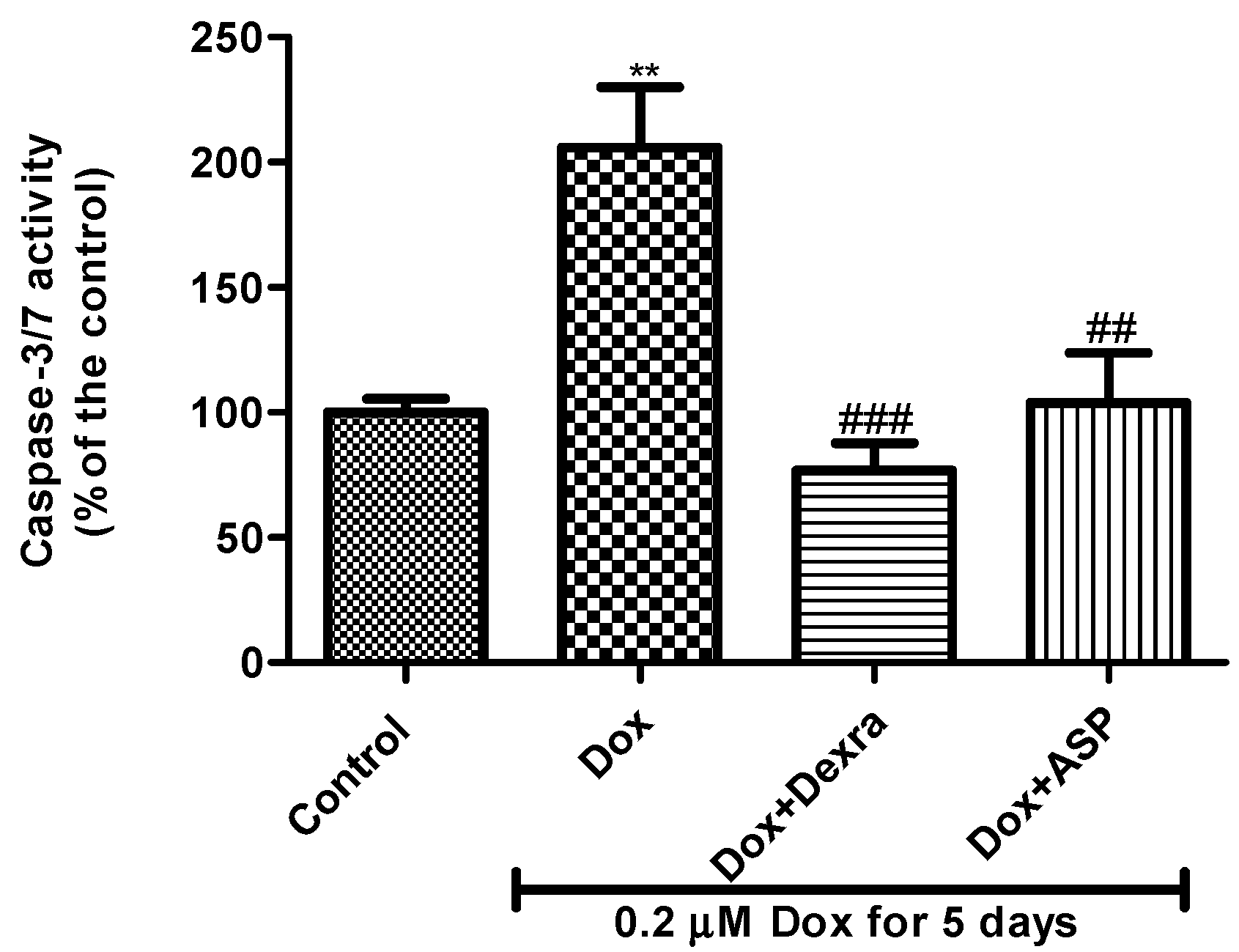
2.2.7. The Effect of ASP on Capase-3/7 Activity
2.3. Transcriptional Regulation of Autophagy Gene in H9c2 Cardiomyoblasts
The Effect of ASP on the Activation of Autophagy Genes
2.4. The Effect of ASP Co-Treatment on the Efficacy of Dox to Increase Apoptosis in Caov-3 Ovarian Cancer Cells
2.4.1. The Effect of ASP Co-Treatment on Caov-3 Cell Viability
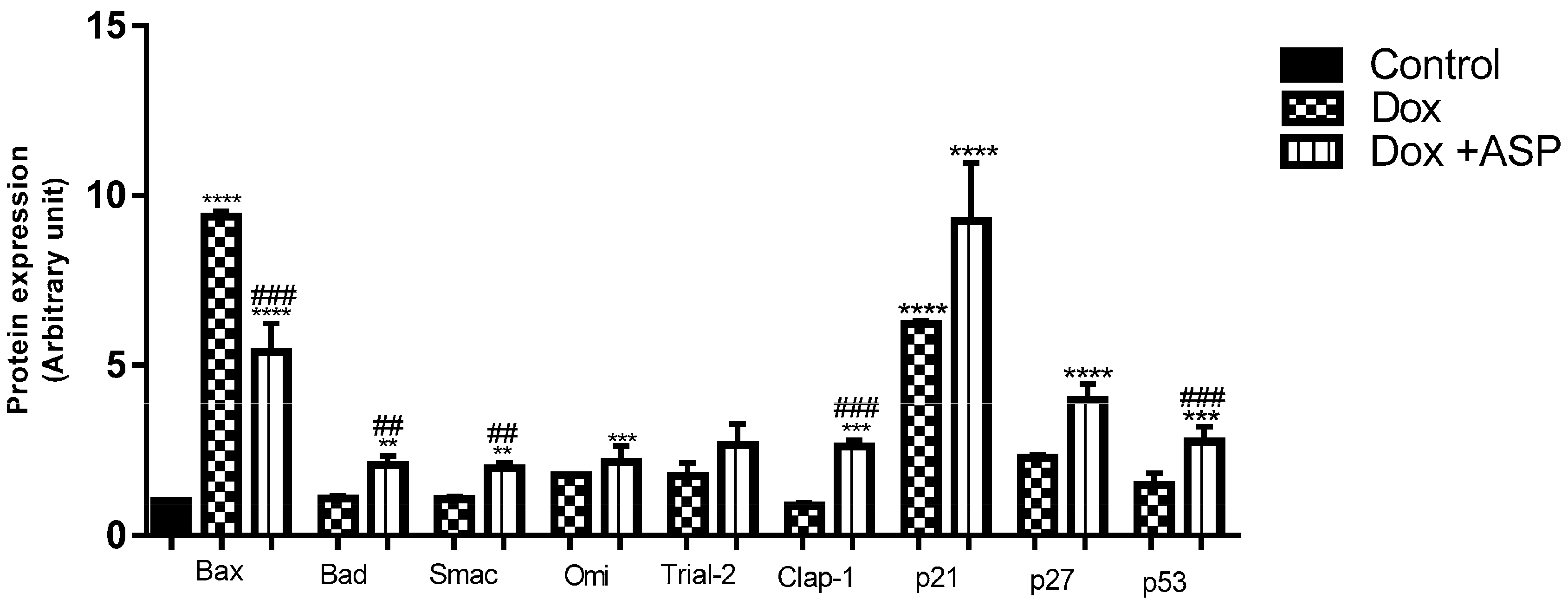
2.4.2. The Effect of ASP Co-Treatment on the Expression of Pro-Apoptotic Proteins
3. Discussion
4. Materials and Methods
4.1. Aspalathin Preparation for Cell Culture
4.2. Doxorubicin (Dox) and Dexrazoxane (Dexra) Dose
4.3. Cell Culture and Treatment of H9c2 Cardiomyoblasts and Caov3 Ovarian Carcinoma Cells with Aspalathin
4.4. ATP Assay
4.5. Western Blot Analysis
4.6. Caspase 3/7 Activity
4.7. mRNA Expression Analysis
4.8. Proteome Profiler Human Apoptosis Array
4.9. Statistical Analysis
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Sui, X.; Han, W.; Pan, H. P53-induced autophagy and senescence. Oncotarget 2015, 6, 11723–11724. [Google Scholar] [CrossRef] [PubMed]
- Maiuri, M.C.; Zalckvar, E.; Kimchi, A.; Kroemer, G. Self-eating and self-killing: Crosstalk between autophagy and apoptosis. Nat. Rev. Mol. Cell Biol. 2007, 8, 741–752. [Google Scholar] [CrossRef] [PubMed]
- Marino, G.; Niso-Santano, M.; Baehrecke, E.H.; Kroemer, G. Self-consumption: The interplay of autophagy and apoptosis. Nat. Rev. Mol. Cell Biol. 2014, 15, 81–94. [Google Scholar] [CrossRef] [PubMed]
- Thorburn, A. Apoptosis and autophagy: Regulatory connections between two supposedly different processes. Apop. Int. J. Progr. Cell Death 2008, 13, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Bjorkoy, G.; Lamark, T.; Pankiv, S.; Overvatn, A.; Brech, A.; Johansen, T. Monitoring autophagic degradation of p62/sqstm1. Methods Enzymol. 2009, 452, 181–197. [Google Scholar] [PubMed]
- Nikoletopoulou, V.; Markaki, M.; Palikaras, K.; Tavernarakis, N. Crosstalk between apoptosis, necrosis and autophagy. Biochim. Biophys. Acta 2013, 1833, 3448–3459. [Google Scholar] [CrossRef] [PubMed]
- Octavia, Y.; Tocchetti, C.G.; Gabrielson, K.L.; Janssens, S.; Crijns, H.J.; Moens, A.L. Doxorubicin-induced cardiomyopathy: From molecular mechanisms to therapeutic strategies. J. Mol. Cell. Cardiol. 2012, 52, 1213–1225. [Google Scholar] [CrossRef] [PubMed]
- DeSantis, C.E.; Lin, C.C.; Mariotto, A.B.; Siegel, R.L.; Stein, K.D.; Kramer, J.L.; Alteri, R.; Robbins, A.S.; Jemal, A. Cancer treatment and survivorship statistics, 2014. CA A Cancer J. Clin. 2014, 64, 252–271. [Google Scholar] [CrossRef] [PubMed]
- Saeed, M.F.; Premecz, S.; Goyal, V.; Singal, P.K.; Jassal, D.S. Catching broken hearts: Pre-clinical detection of doxorubicin and trastuzumab mediated cardiac dysfunction in the breast cancer setting. Can. J. Physiol. Pharmacol. 2014, 92, 546–550. [Google Scholar] [CrossRef] [PubMed]
- Jain, D.; Russell, R.R.; Schwartz, R.G.; Panjrath, G.S.; Aronow, W. Cardiac complications of cancer therapy: Pathophysiology, identification, prevention, treatment, and future directions. Curr. Cardiol. Rep. 2017, 19, 36. [Google Scholar] [CrossRef] [PubMed]
- Gu, J.; Hu, W.; Song, Z.P.; Chen, Y.G.; Zhang, D.D.; Wang, C.Q. Resveratrol-induced autophagy promotes survival and attenuates doxorubicin-induced cardiotoxicity. Int. Immunopharmacol. 2016, 32, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Wang, S.; Song, P.; Zou, M.H. Amp-activated protein kinase, stress responses and cardiovascular diseases. Clin. Sci. 2012, 122, 555–573. [Google Scholar] [CrossRef] [PubMed]
- Liu, M.H.; Lin, X.-L.; Guo, D.M.; Zhang, Y.; Yuan, C.; Tan, T.P.; Chen, Y.D.; Wu, S.J.; Ye, Z.F.; He, J. Resveratrol protects cardiomyocytes from doxorubicin-induced apoptosis through the ampk/p53 pathway. Mol. Med. Rep. 2015, 13, 1281–1286. [Google Scholar] [CrossRef] [PubMed]
- Dickey, J.S.; Gonzalez, Y.; Aryal, B.; Mog, S.; Nakamura, A.J.; Redon, C.E.; Baxa, U.; Rosen, E.; Cheng, G.; Zielonka, J.; et al. Mito-tempol and dexrazoxane exhibit cardioprotective and chemotherapeutic effects through specific protein oxidation and autophagy in a syngeneic breast tumor preclinical model. PLoS ONE 2013, 8, e70575. [Google Scholar] [CrossRef] [PubMed]
- Tebbi, C.K.; London, W.B.; Friedman, D.; Villaluna, D.; De Alarcon, P.A.; Constine, L.S.; Mendenhall, N.P.; Sposto, R.; Chauvenet, A.; Schwartz, C.L. Dexrazoxane-associated risk for acute myeloid leukemia/myelodysplastic syndrome and other secondary malignancies in pediatric hodgkin's disease. J. Clin. Oncol. Off. J. Am. Soc. Clin. Oncol. 2007, 25, 493–500. [Google Scholar] [CrossRef] [PubMed]
- Johnson, R.; Dludla, P.; Joubert, E.; February, F.; Mazibuko, S.; Ghoor, S.; Muller, C.; Louw, J. Aspalathin, a dihydrochalcone c-glucoside, protects h9c2 cardiomyocytes against high glucose induced shifts in substrate preference and apoptosis. Mol. Nutr. Food Res. 2016, 60, 922–934. [Google Scholar] [CrossRef] [PubMed]
- Johnson, R.; Dludla, P.V.; Muller, C.J.; Huisamen, B.; Essop, M.F.; Louw, J. The transcription profile unveils the cardioprotective effect of aspalathin against lipid toxicity in an in vitro h9c2 model. Molecules 2017, 22. [Google Scholar] [CrossRef] [PubMed]
- Snijman, P.W.; Joubert, E.; Ferreira, D.; Li, X.C.; Ding, Y.; Green, I.R.; Gelderblom, W.C. Antioxidant activity of the dihydrochalcones aspalathin and nothofagin and their corresponding flavones in relation to other rooibos ( aspalathus linearis ) flavonoids, epigallocatechin gallate, and trolox. J. Agric. Food Chem. 2009, 57, 6678–6684. [Google Scholar] [CrossRef] [PubMed]
- Hale, A.N.; Ledbetter, D.J.; Gawriluk, T.R.; Rucker, I.I.I.E.B. Autophagy: Regulation and role in development. Autophagy 2013, 9, 951–972. [Google Scholar] [CrossRef] [PubMed]
- Fulda, S.; Gorman, A.M.; Hori, O.; Samali, A. Cellular stress responses: Cell survival and cell death. Int. J. Cell Biol. 2010, 2010, 23. [Google Scholar] [CrossRef] [PubMed]
- Cao, Y.; Shen, T.; Huang, X.; Lin, Y.; Chen, B.; Pang, J.; Li, G.; Wang, Q.; Zohrabian, S.; Duan, C.; et al. Astragalus polysaccharide restores autophagic flux and improves cardiomyocyte function in doxorubicin-induced cardiotoxicity. Oncotarget 2017, 8, 4837–4848. [Google Scholar] [CrossRef] [PubMed]
- Lee, K.H.; Cho, H.; Lee, S.; Woo, J.S.; Cho, B.H.; Kang, J.H.; Jeong, Y.M.; Cheng, X.W.; Kim, W. Enhanced-autophagy by exenatide mitigates doxorubicin-induced cardiotoxicity. Int. J. Cardiol. 2017, 232, 40–47. [Google Scholar] [CrossRef] [PubMed]
- Shabalala, S.; Muller, C.J.F.; Louw, J.; Johnson, R. Polyphenols, autophagy and doxorubicin-induced cardiotoxicity. Life Sci. 2017, 180, 160–170. [Google Scholar] [CrossRef] [PubMed]
- Sishi, B.J.; Loos, B.; van Rooyen, J.; Engelbrecht, A.M. Autophagy upregulation promotes survival and attenuates doxorubicin-induced cardiotoxicity. Biochem. Pharmacol. 2013, 85, 124–134. [Google Scholar] [CrossRef] [PubMed]
- Kobayashi, S.; Volden, P.; Timm, D.; Mao, K.; Xu, X.; Liang, Q. Transcription factor gata4 inhibits doxorubicin-induced autophagy and cardiomyocyte death. J. Biol. Chem. 2010, 285, 793–804. [Google Scholar] [CrossRef] [PubMed]
- Lu, L.; Wu, W.; Yan, J.; Li, X.; Yu, H.; Yu, X. Adriamycin-induced autophagic cardiomyocyte death plays a pathogenic role in a rat model of heart failure. Int. J. Cardiol. 2009, 134, 82–90. [Google Scholar] [CrossRef] [PubMed]
- Jain, M.V.; Paczulla, A.M.; Klonisch, T.; Dimgba, F.N.; Rao, S.B.; Roberg, K.; Schweizer, F.; Lengerke, C.; Davoodpour, P.; Palicharla, V.R.; et al. Interconnections between apoptotic, autophagic and necrotic pathways: Implications for cancer therapy development. J. Cell. Mol. Med. 2013, 17, 12–29. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.W.; Park, S.; Takahashi, Y.; Wang, H.G. The association of ampk with ulk1 regulates autophagy. PLoS ONE 2010, 5, e15394. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.; Kundu, M.; Viollet, B.; Guan, K.L. Ampk and mtor regulate autophagy through direct phosphorylation of ulk1. Nat. Cell Biol. 2011, 13, 132–141. [Google Scholar] [CrossRef] [PubMed]
- Chen, K.; Xu, X.; Kobayashi, S.; Timm, D.; Jepperson, T.; Liang, Q. Caloric restriction mimetic 2-deoxyglucose antagonizes doxorubicin-induced cardiomyocyte death by multiple mechanisms. J. Biol. Chem. 2011, 286, 21993–22006. [Google Scholar] [CrossRef] [PubMed]
- Kang, Y.J.; Chen, Y.; Epstein, P.N. Suppression of doxorubicin cardiotoxicity by overexpression of catalase in the heart of transgenic mice. J. Biol. Chem. 1996, 271, 12610–12616. [Google Scholar] [CrossRef] [PubMed]
- Quinsay, M.N.; Thomas, R.L.; Lee, Y.; Gustafsson, Å.B. Bnip3-mediated mitochondrial autophagy is independent of the mitochondrial permeability transition pore. Autophagy 2010, 6, 855–862. [Google Scholar] [CrossRef] [PubMed]
- He, C.; Klionsky, D.J. Regulation mechanisms and signaling pathways of autophagy. Annu. Rev. Genet. 2009, 43, 67–93. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.-W.; Shi, J.; Li, Y.-J.; Wei, L. Cardiomyocyte death in doxorubicin-induced cardiotoxicity. Arch. Immunol. Ther. Exp. 2009, 57, 435–445. [Google Scholar] [CrossRef] [PubMed]
- Dludla, P.V.; Muller, C.J.; Joubert, E.; Louw, J.; Essop, M.F.; Gabuza, K.B.; Ghoor, S.; Huisamen, B.; Johnson, R. Aspalathin protects the heart against hyperglycemia-induced oxidative damage by up-regulating nrf2 expression. Molecules 2017, 22, 129. [Google Scholar] [CrossRef] [PubMed]
- Chacko, S.M.; Nevin, K.G.; Dhanyakrishnan, R.; Kumar, B.P. Protective effect of p-coumaric acid against doxorubicin induced toxicity in h9c2 cardiomyoblast cell lines. Toxicol. Rep. 2015, 2, 1213–1221. [Google Scholar] [CrossRef]
- Goldswain, T.L.; Lacerda, L.; Sishi, B.J.N. The (un)safe and Risk(y) Sides of Doxorubicin-Induced Cardiotoxicity. Electronic Thesis, Stellenbosch University, Stellenbosch, South Africa, 2014. [Google Scholar]
- Deng, S.; Yan, T.; Jendrny, C.; Nemecek, A.; Vincetic, M.; Godtel-Armbrust, U.; Wojnowski, L. Dexrazoxane may prevent doxorubicin-induced DNA damage via depleting both topoisomerase ii isoforms. BMC Cancer 2014, 14, 842. [Google Scholar] [CrossRef] [PubMed]
Sample Availability: Samples of the compound aspalathin is available from the authors. |

| Genes | Dox | Dox + Dexra | Dox + ASP |
|---|---|---|---|
| Atg5 | ↓1.81 ± 0.19 * | ↑1.24 ± 0.24 | ↑1.18 ± 0.25 # |
| Atg7 | ↓1.56 ± 0.2 * | ↑1.02 ± 0.28 # | ↑1.06± 0.30 ## |
| Beclin1 | ↓1.61 ± 0.21 * | ↑1.13 ± 0.25 # | ↑1.03± 0.27 # |
| BNIP3 | ↓1.38 ± 0.23 | ↑1.04 ± 0.33 | ↑1.17± 0.4 |
| Fox01 | ↓1.40 ± 0.24 | ↑1.14 ± 0.31 | ↑1.18± 0.32 # |
| AMPK | ↓1.72 ± 0.29 | ↑1.38 ± 0.41 | ↑1.2± 0.34 |
| mTOR | ↑1.30 ± 0.21 | ↓2.24 ± 0.16 | ↓1.39± 0.19 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Johnson, R.; Shabalala, S.; Louw, J.; Kappo, A.P.; Muller, C.J.F. Aspalathin Reverts Doxorubicin-Induced Cardiotoxicity through Increased Autophagy and Decreased Expression of p53/mTOR/p62 Signaling. Molecules 2017, 22, 1589. https://doi.org/10.3390/molecules22101589
Johnson R, Shabalala S, Louw J, Kappo AP, Muller CJF. Aspalathin Reverts Doxorubicin-Induced Cardiotoxicity through Increased Autophagy and Decreased Expression of p53/mTOR/p62 Signaling. Molecules. 2017; 22(10):1589. https://doi.org/10.3390/molecules22101589
Chicago/Turabian StyleJohnson, Rabia, Samukelisiwe Shabalala, Johan Louw, Abidemi Paul Kappo, and Christo John Frederick Muller. 2017. "Aspalathin Reverts Doxorubicin-Induced Cardiotoxicity through Increased Autophagy and Decreased Expression of p53/mTOR/p62 Signaling" Molecules 22, no. 10: 1589. https://doi.org/10.3390/molecules22101589
APA StyleJohnson, R., Shabalala, S., Louw, J., Kappo, A. P., & Muller, C. J. F. (2017). Aspalathin Reverts Doxorubicin-Induced Cardiotoxicity through Increased Autophagy and Decreased Expression of p53/mTOR/p62 Signaling. Molecules, 22(10), 1589. https://doi.org/10.3390/molecules22101589








