Abstract
The combined synergistic effects of copper (Cu2+) and sulfur dioxide (SO2) on the formation of hydrogen sulfide (H2S) in Verdelho and Shiraz wine samples post-bottling was studied over a 12-month period. The combined treatment of Cu2+ and SO2 significantly increased H2S formation in Verdelho wines samples that were not previously treated with either Cu2+ or SO2. The formation of H2S produced through Cu2+ mediated reactions was likely either: (a) directly through the interaction of SO2 with either Cu2+ or H2S; or (b) indirectly through the interaction of SO2 with other wine matrix compounds. To gain better understanding of the mechanisms responsible for the significant increases in H2S concentration in the Verdelho samples, the interaction between Cu2+ and SO2 was studied in a model wine matrix with and without the presence of a representative thiol quenching compound (4-methylbenzoquinone, 4MBQ). In these model studies, the importance of naturally occurring wine compounds and wine additives, such as quinones, SO2, and metal ions, in modulating the formation of H2S post-bottling was demonstrated. When present in equimolar concentrations a 1:1 ratio of H2S- and SO2-catechol adducts were produced. At wine relevant concentrations, however, only SO2-adducts were produced, reinforcing that the competition reactions of sulfur nucleophiles, such as H2S and SO2, with wine matrix compounds play a critical role in modulating final H2S concentrations in wines.
1. Introduction
The origin and management of volatile sulfur compounds (VSCs) in wine is a topic that concerns many winemakers, as these compounds have a significant impact on wine aroma as well as wine quality [1]. Volatile sulfur compounds such as hydrogen sulfide (H2S) contribute negatively to wine quality and are considered a wine fault when present at concentrations greater than its odor threshold (OT) value of 1.1–1.6 μg/L [2]. As one of the main compounds contributing to “reduced” characters in wine, H2S imparts aromas of rotten egg and sewage when present in high concentrations, however, when present in low concentrations H2S can add complexity to wine aroma [2]. While the release and subsequent perception of H2S into the wine headspace is affected by the wine matrix, the contribution of individual wine matrix components on VSC perception, such as H2S, has not yet been determined [3].
The reaction pathways for H2S formation post-bottling are not yet as well defined as those during fermentation, however, certain key factors affecting H2S concentration post-bottling have recently been elucidated. It is known that decreased oxygen concentrations during fermentation as well as during wine storage conditions are associated with increased H2S concentrations in wines post-bottling [4,5,6]. Franco-Luesma and Ferreira [7] have recently shown that H2S is present in both free and bound forms in wine and the percentage of free and bound forms are related to the trace metal content of the wine. According to Franco-Luesma and Ferreira, both de novo formation of H2S from precursor compounds as well as the release of H2S from metal complexes contribute to the final concentration of H2S formation in wines post-bottling, with release from metal complexes responsible for the majority of H2S produced in red wines, and de novo formation responsible for the majority of H2S produced in white wines and rosé wines [7]. Additionally, Franco-Luesma suggests that the release of free H2S and MeSH from bound sources are a function of a decrease in redox potential of wine [8].
A number of studies have also highlighted the important catalytic role of metals such as Cu2+, Fe3+, Mn2+, Al3+, and Zn2+ ions in VSC formation in wine [4,9,10,11]. Metal ions naturally occur in wines at trace concentrations and their final concentrations in finished wine are influenced by the use of metal containing pesticides and herbicides, processing aids, and winemaking equipment. However, the addition of Cu2+ (copper can exist in both Cu+ and Cu2+ oxidation states in wine; thus, in this manuscript, “Cu2+” is used when the authors refer to the oxidation state in which copper was added to the wine) to wines through the use of copper sulfate as a fining agent applied to decrease reductive aromas in wine, remains the main use of metals that results in increased residual concentration of the metal post-bottling [5]. While the mechanism of copper fining is not yet well defined, Kreitman et al. [12] recently provided insights into the mechanisms through which Cu2+ interacts with thiols such as H2S in wine-like conditions saturated with oxygen. The manner in which residual copper sulfide (CuS) may affect wine aroma has recently been studied by Clark et al., who firstly showed that once formed, CuS is not easily removed from white wine and that tartaric acid significantly affects CuS precipitation [13]; and secondly, that in both model and finished wines CuS was not measurable as labile copper but that oxidation as well as the loss of volatile thiol compounds through volatilization affects the ratio of labile copper to CuS [14]. These studies demonstrated that when copper and H2S are present in wines, non-labile CuS will be produced that cannot be removed through filtering and that changes in the wine conditions will affect the ability of copper to remain active/labile to mediate reactions. The ability of copper to mediate H2S formation reactions have been shown in wines where the addition of Cu2+ was associated with significantly increased H2S concentration in wines during bottle aging [4,10,15]. Given that a major source of Cu2+ in finished wines is within the control of the winemaker, the timing and amount of Cu2+ can be used to manage H2S formation.
Another important winemaking intervention is the use of sulfur dioxide (SO2). The dosage and timing of SO2 addition are similarly key activities under winemaking control and may impact H2S formation. Sulfur dioxide naturally occurs in wines at very low concentrations (10–30 mg/L [16]), however, the main contributor to the final concentration in commercial wines is added SO2 at concentrations in the range of 50–200 mg/L. Sulfur dioxide exists in free and bound forms, with the majority of free SO2 present as bisulfite ions (HSO3−) at normal wine pH (all of the different species of sulfur dioxide in equilibrium found in wine, which includes the molecular sulfur dioxide, bisulfite, and sulfite, will be referred to generically as “SO2” throughout the text). Sulfur dioxide plays a critical role in the prevention of microbial spoilage, and is important as an antioxidant, through reaction with hydrogen peroxide (H2O2) generated from oxygen via a series of redox steps, as well as with quinones to regenerate polyphenols [1,17,18,19,20]. It is also known that SO2 influences the loss of thiol compounds post-bottling [21,22]. It has also been proposed that SO2 may act as a source of VSCs. Rankine suggested the reduction of SO2 through the interaction of metal ions such as manganese and zinc with tartaric and malic acids during fermentation [23]. No evidence for the post-bottling formation of H2S via the metal catalyzed reduction of SO42− or SO32− during low oxygen conditions has yet been shown, even though this pathway has been proposed by Ribéreau-Gayon [24] and Lopes et al. [25]. Another possible pathway through which SO2 can be involved in the modulation of “reductive” VSCs post-bottling is through reactions with wine compounds, such as quinones. Nikolantonaki et al. [26] described the reaction and kinetics between certain o-quinones and nucleophilic thiols and has shown that the addition of SO2 directly influenced the rates and the yields of all of the o-quinone sulfur adducts. Research has demonstrated, using model wine studies, that sulfites, ascorbic acid, and/or GSH can act as sacrificial nucleophiles, suppressing varietal thiol consumption during wine aging [21,22]. This competing reaction between H2S, methanethiol (MeSH), glutathione, and SO2 has not yet been demonstrated in real wines.
The separate effects that Cu2+ and SO2 additions may have on H2S formation in wines post-bottling have been discussed above, however, Cu2+ and SO2 are usually both present in wine and may have synergistic effects via wine matrix compounds. Given that the final concentrations of Cu2+ and SO2 in finished wines, as well as timing of the addition of Cu2+ and SO2, are under a winemaker’s control it is important to understand the synergistic interactions between wine additives such as Cu2+ and SO2 and the effects that these compounds may have on H2S formation. The interactions between Cu2+ and SO2 and its effects on H2S formation have not previously been researched in a structured way in real wines, and this synergistic “Cu2+ + SO2” interaction may be an additional key contributing factor in the accumulation of H2S post-bottling. The aim of the present study was to investigate the synergistic effects of the combined treatment of Cu2+ and SO2 on H2S formation in a white wine (Verdelho) and a red wine (Shiraz). Some of the factors modulating the formation of H2S via Cu2+ and SO2 interactions during wine storage under low oxygen conditions were also studied.
2. Results
2.1. Dissolved Oxygen
The dissolved oxygen (DO) was monitored over the course of the experiment using PreSens oxygen sensors applied to representative colorless vials (in triplicate) for a control sample (blank wine) and for each of the treatments (Cu2+, SO2, and “Cu2+ + SO2”) for the Verdelho (n = 12) and for the Shiraz (n = 12) wine samples. After two months of storage under low oxygen conditions, both Verdelho and Shiraz samples had consumed all available oxygen and samples can be considered oxygen-free. All samples were stored at room temperature (22 °C) in 19 L post mix Cornelius kegs (Ambar technology, Alexandria, NSW, Australia). The kegs were flushed with nitrogen gas (N2 (g)) to <1 ppb and maintained at a slight positive pressure of 1 psi N2 (g) to prevent any oxygen ingress into the keg storage systems, as described by Viviers et al. [10]. As such all wine samples were not exposed to oxygen during storage and only exposed to normal atmospheric oxygen at the point of analysis.
2.2. Formation of Hydrogen Sulfide in Wine
2.2.1. The Effect of Cu2+ on H2S Formation
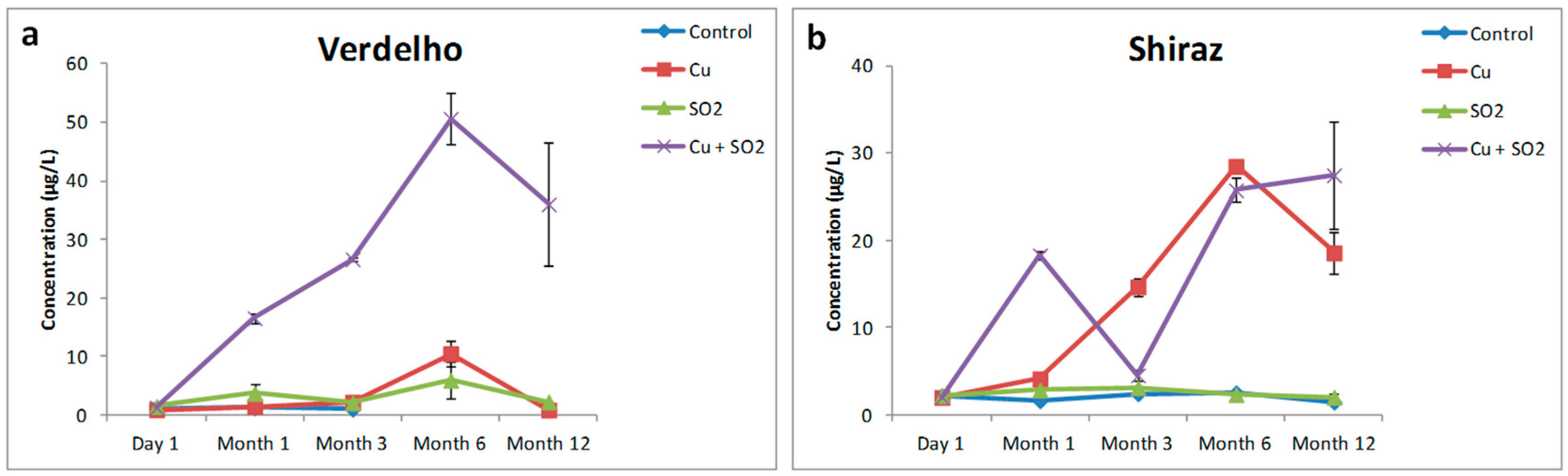
Copper treatment did not result in an overall significant effect on H2S formation in the Verdelho samples. Significant increases in H2S concentration were measured after six months of anaerobic storage associated with Cu2+ treatment, however, the overall Cu2+ effect, as measured over the course of the twelve months, was not significant for H2S formation (Figure 1). Hydrogen sulfide is a reactive nucleophile and it has been shown to react readily with quinones in wine-like solutions [21] which may explain the decrease in H2S concentration after the initial increase measured after six months of storage. Only in the Shiraz wine samples did Cu2+ treatment alone result in an overall significant effect on H2S formation. All Shiraz wine samples treated with Cu2+ displayed significant increases in H2S concentration, irrespective of the other additional treatments (Figure 1b, Table 1). Multi-way analysis of variance (ANOVA) confirmed that the increased H2S concentration was significantly correlated with the Cu2+ addition and not associated with interactions between Cu2+ and “SO2” in the “Cu2+ + SO2” treatment.
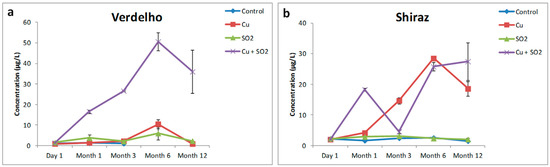
Figure 1.
The effects of Cu2+, SO2, and “Cu2+ + SO2” on the formation of H2S in (a) Verdelho and (b) Shiraz wine samples that were stored under nitrogen (N2) and measured over the course of 12 months.

Table 1.
Effects of Cu2+, SO2, and “Cu2+ + SO2” treatments on H2S formation in Verdelho and Shiraz wine samples.
The effect of residual Cu2+ on H2S formation is well established [4,10,27]. All the precursors to H2S have not yet been identified, however, wines with elevated residual Cu2+ concentrations post-bottling risk the formation of H2S during bottle maturation.
2.2.2. The Effect of SO2 on H2S Formation
The treatment of both Verdelho and Shiraz wine samples with SO2 alone had no significant effect on H2S concentration (Figure 1a,b). No increased H2S concentrations were measured by treating Verdelho or Shiraz wines with SO2 alone, suggesting that for the wines investigated in the current study, SO2 is not capable of significantly increasing H2S concentrations when present in wines without Cu2+.
2.2.3. The Effects of Cu2+ and SO2 Interaction on H2S Formation
The combined Cu2+ and SO2 treatment in the Verdelho wine samples resulted in significant and large increases in H2S concentrations. For the Verdelho samples more H2S was measured in samples treated with “Cu2+ + SO2” than by treating samples with Cu2+ alone (Figure 1a). Using multi-way ANOVA it was possible to separate the effects of Cu2+ treatment from the combined Cu2+ and SO2 effects, with the increase in H2S concentration significantly associated with “Cu2+ + SO2” treatment in the Verdelho samples (p < 0.001, Table 1). The concentration of H2S produced in Verdelho samples treated with “Cu2+ + SO2” was 36.01 (±18.1) µg/L compared to the 0.879 (±0.028) µg/L of H2S measured in the control samples after 12 months of storage under low oxygen conditions, which is a significant increase in H2S concentration.
Significantly increased H2S concentrations were also measured in the Shiraz samples treated with “Cu2+ + SO2” (Figure 1b). By making use of multi-way ANOVA it was clear that these increases were only associated with a significant effect of Cu2+ (p < 0.001, Table 1) and not associated with the combined Cu2+ and SO2 treatments (Table 1, Figure 1b). If the evolution of H2S in Shiraz samples is considered over the course of the 12 months of storage in a low oxygen environment, the trends of H2S formation for Cu2+ treated samples and samples treated with both Cu2+ and SO2 are remarkably different (Figure 1b). Shiraz samples treated with only Cu2+ displayed a steady increase in H2S concentrations from Month 1 to Month 6, followed by a decrease to Month 12, whereas samples treated with Cu2+ and SO2 displayed an initial increase from Day 0 to Month 1, followed by a significant decrease in H2S concentrations at Month 3, which was then followed by increased H2S concentrations from Month 6 through to Month 12. The exact mechanism for these different H2S evolution trends are not clear, however, this demonstrates the complex underlying interactions of different wine compounds with one another and how the addition of SO2 to a wine already containing Cu2+ could have significant implications on H2S evolution. Previous studies have also shown the non-linear increase in thiols such as H2S and MeSH evolution over time [4,5,10], which suggest that compounds such as H2S and MeSH may be dynamically bound and released by other wine compounds after bottling and during storage.
2.3. Interaction of Cu2+ and SO2 in Model Wine
2.3.1. Formation of H2S in Model Wine
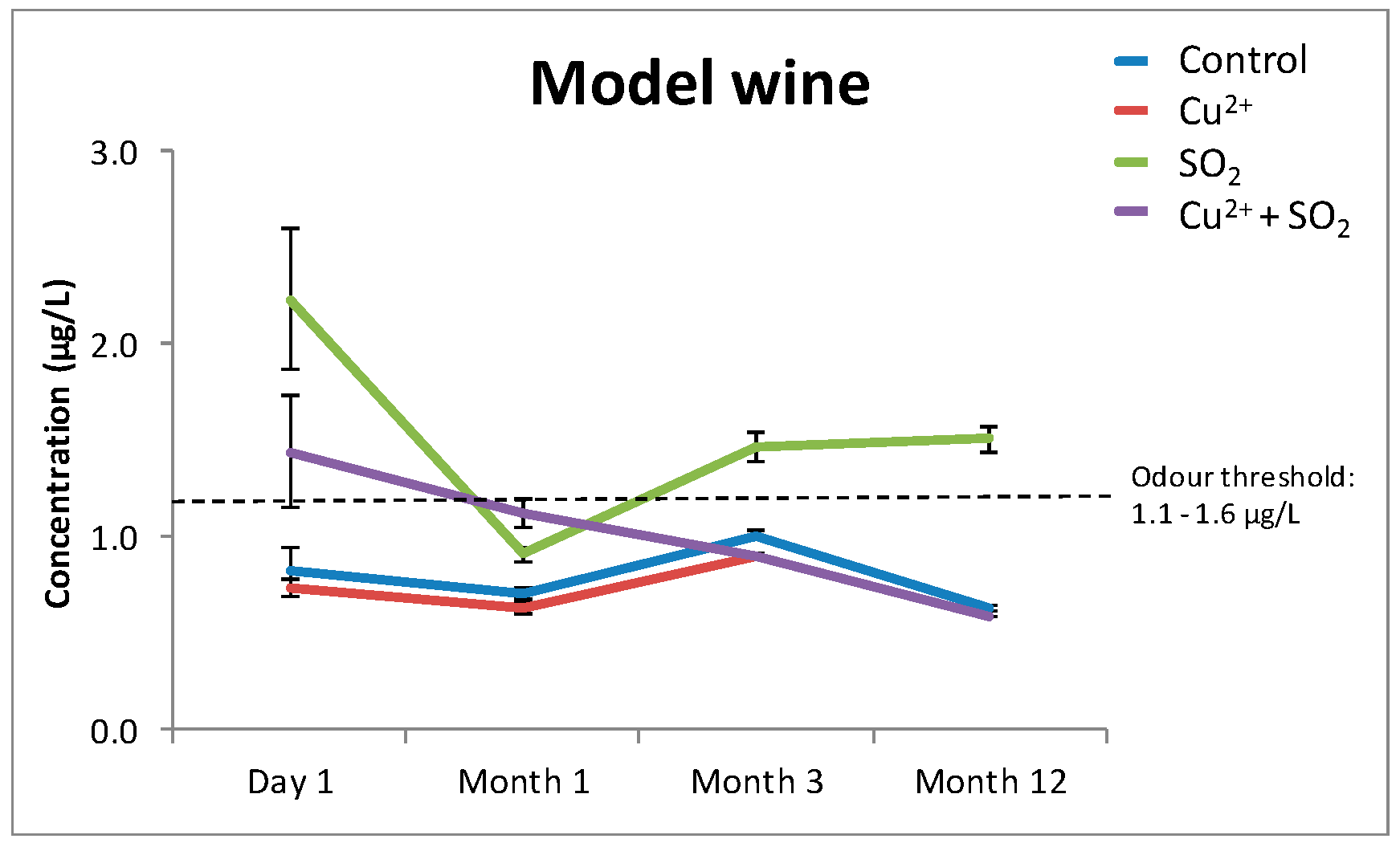
In an effort to identify some of the potential mechanisms involved in modulating the significantly increased H2S concentrations in Verdelho samples treated with “Cu2+ + SO2”, the experiment was repeated in a model wine containing only water, ethanol (12%), and tartaric acid (10 g/L). Samples treated with “Cu2+ + SO2” did not produce similar significant increases in H2S concentrations as was measured in the Verdelho samples treated with “Cu2+ + SO2” (Figure 2). While there were some changes in H2S concentrations in SO2 treated samples (Figure 2), the increases were small compared to the significantly increased H2S concentrations measured in Verdelho samples treated with “Cu2+ + SO2” (Figure 1a).
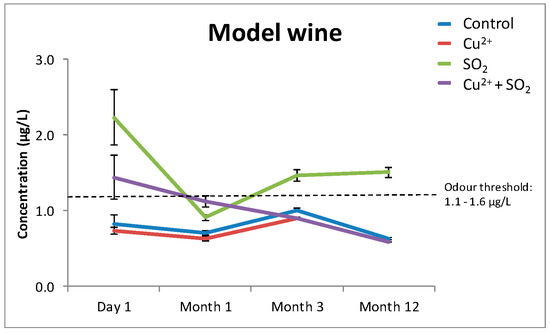
Figure 2.
The effects of Cu2+, SO2, and “Cu2+ + SO2” on the concentration of H2S in a model wine matrix that did not contain any wine compounds, such as polyphenols or tannins, that could modulate the formation of H2S. The odor threshold of H2S (1.1–1.6 µg/L [28]) is indicated with the dashed line parallel to the x-axis. Samples were stored under nitrogen (N2) and measured over the course of 12 months.
From this experiment, it is clear the increases measured in H2S associated with “Cu2+ + SO2” treatment require other wine matrix components.
2.3.2. Reactions between H2S and SO2 with a Model Wine Compound/Thiol Quenching Compound
It is known that certain wine matrix compounds, such as quinones, for example, have the ability to readily react with thiols in wine [21] and thus play an important role in modulating the final concentrations of thiols in wines. To investigate the competition reactions between H2S and SO2 with wine matrix compounds capable of reacting with thiols, a model quinone (4-methylbenzoquinone (4MBQ)) was synthesized and H2S and SO2 were added to 4MBQ to produce adducts of 4MBQ with H2S and SO2. The sulfur containing nucleophiles were first reacted in excess concentration (4 mM) relative to 4MBQ (1 mM) and the reaction products tentatively identified using high pressure liquid chromatography (HPLC), liquid chromatography-mass spectrometry (LCMS), liquid chromatography-high resolution mass spectrometry (LC-HRMS), and comparison of the mass spectra with published results [21]. All the isomers produced as reaction products from reacting 4MBQ with H2S will from here on be referred to as “H2S-4MC” adducts. Similarly, all the isomers produced as reaction products from reacting 4MBQ with SO2 will from here on be referred to as “SO2-4MC”. Synthesized H2S-4MC and SO2-4MC adducts were used as standards for retention time. The main reaction products are listed in Table 2. The high reactivity of the SO2 and H2S in relation to the 4MBQ resulted in a high number of isomeric compounds being produced, and for the purpose of this study, only the major isomeric compounds were considered.

Table 2.
Mass spectral data and retention times of adducts of 4-methyl benzoquinone with H2S and SO2.
Competition Reactions between H2S and SO2 with a Model Wine Quinone
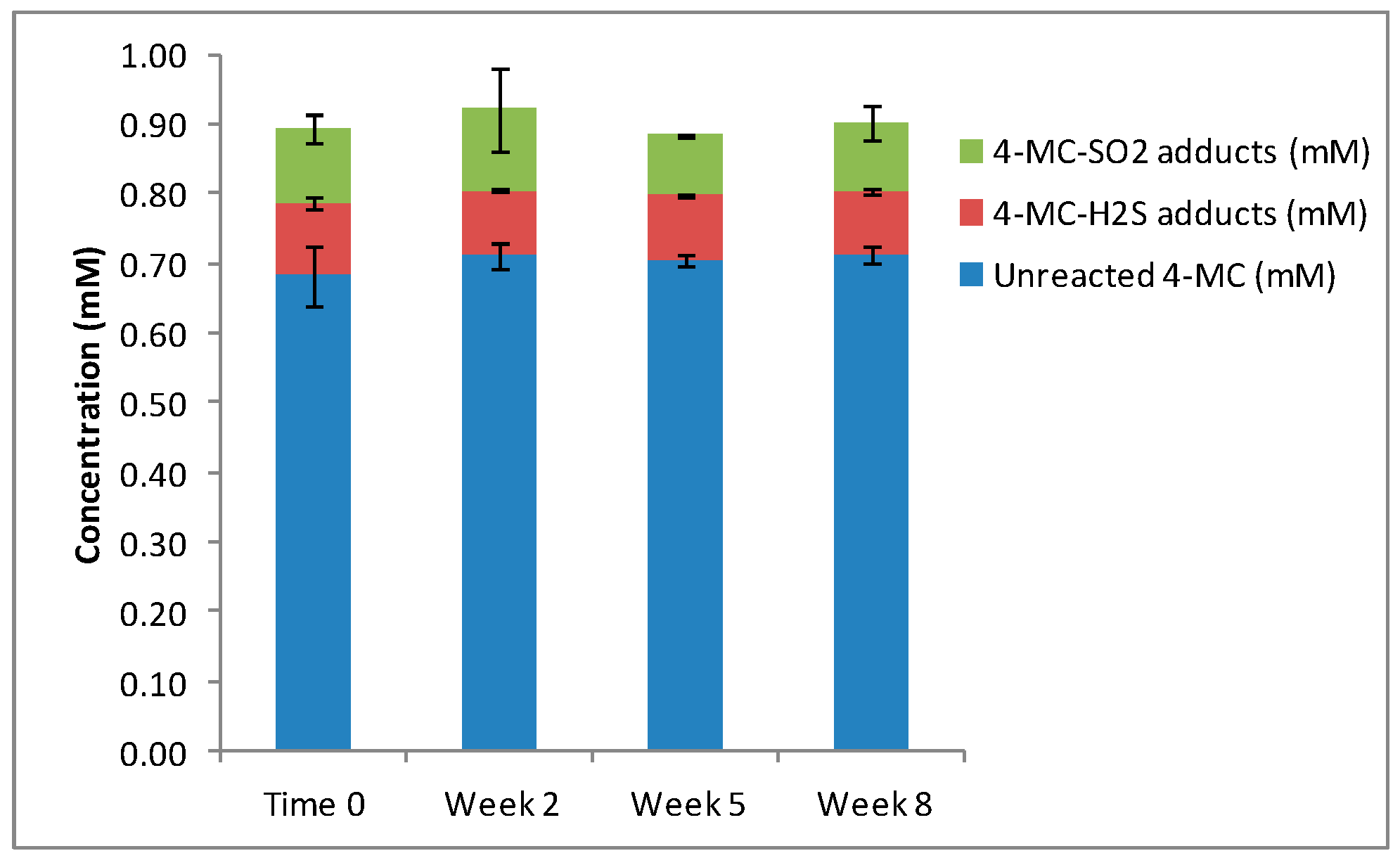
When 4-methylcatechol (4MC) was oxidized to 4MBQ, the HPLC and LCMS analyses showed only approximately 20% of the 4MBQ reacted with the nucleophiles, while the remaining 4MBQ fraction was converted back to 4MC. This incomplete conversion of 4MC to 4MBQ is agreement with previous studies [21]. When 4MBQ (1 mM) was incubated with H2S (4 mM), three main isomers were formed at a ratio of 5:3:1, and when 4MBQ (1 mM) was incubated with SO2 (4 mM), four main isomers were formed at a ratio of 2:1:3:1 (Table 2). When 4MBQ (1 mM) was added to model wine containing both sulfur nucleophiles in equimolar concentrations of 2 mM respectively, the same concentration of H2S-4MC and SO2-4MC adducts were measured (Figure 3), with a peak ratio of 4:1:3:1 for H2S-4MC and the three main SO2-4MC adducts. There were no statistical differences in the total concentrations of H2S-4MC and SO2-4MC adducts produced (p = 0.193). The distribution of the H2S-4MC and SO2-4MC adducts remain approximately the same throughout the eight weeks of the experiment (Figure 3).
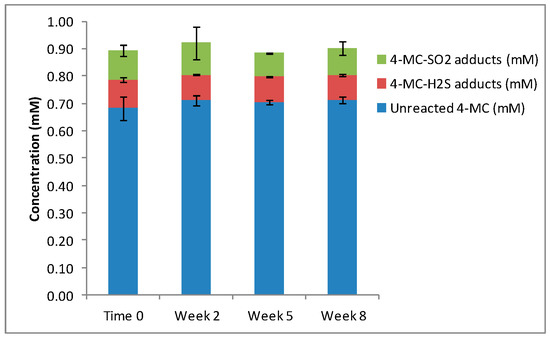
Figure 3.
The reaction products measured when 4MBQ was added to model wine containing equimolar concentrations of H2S and SO2.
This experiment demonstrated that when H2S and SO2 are present in equimolar concentrations, both sulfur nucleophiles compete equally for binding to the quinone and a 1:1 ratio of H2S-4MC and SO2-4MC adducts are produced.
Competition Reactions between a Model Wine Quinone with H2S and SO2 at Wine Relevant Concentrations
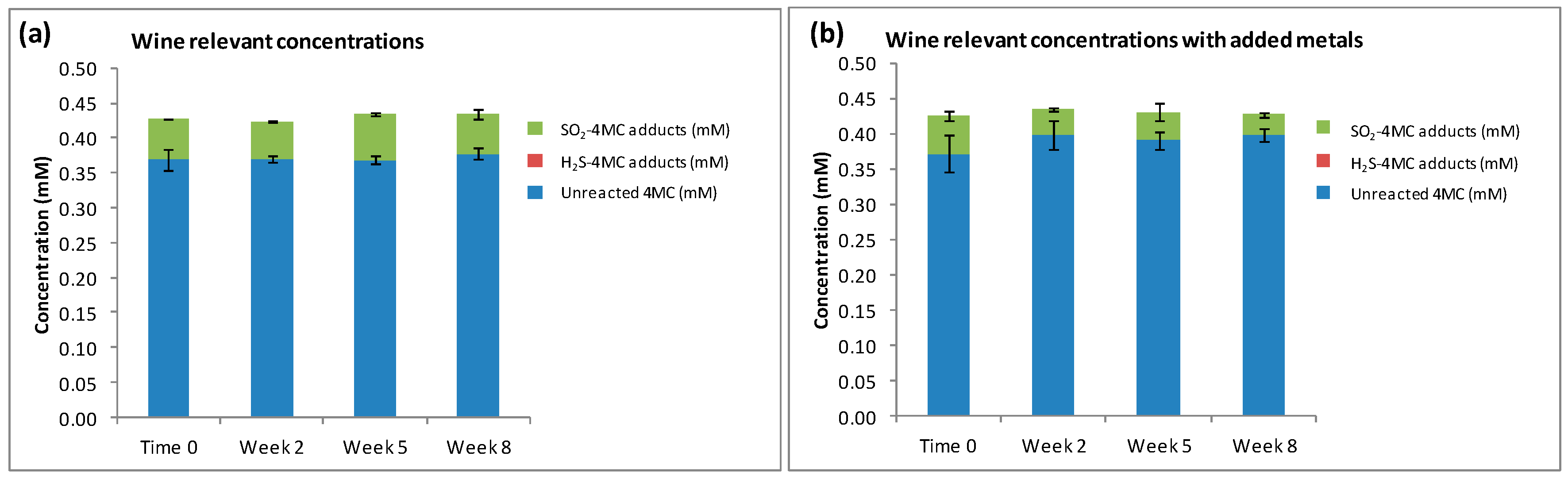
When 4MC was oxidized to 4MBQ, and added to model wine containing H2S and SO2 at wine relevant concentrations (4MC, 0.45 mM; H2S, 0.39 µM; SO2, 0.23 mM), SO2 preferentially reacted with 4MBQ to form a mixture of SO2-4MC adducts (Figure 4a) even though the spiked H2S was available in the sample to react with 4MBQ (see H2S concentration data in Figure S1). The peak ratio for the three main SO2-4MC adducts was 1:5:1. Considering that H2S and SO2 have approximately identical first order rate constants when reacting with 4MBQ [21], it was anticipated that the SO2-4MC adducts would be the main 4-MC adducts produced, but this experiment confirmed that it was indeed the case. This experiment demonstrated that when H2S and SO2 are present in wine relevant concentrations of 0.39 µM H2S and 0.23 mM SO2, the SO2-4MC adducts dominate and no H2S-4MC adducts can be measured. This suggests that thiol quenching compounds may fully react with sulfur nucleophiles that are present in higher concentrations, such as SO2, and thus allow the accumulation of sulfur nucleophiles present in lower concentrations, such as H2S, for example.
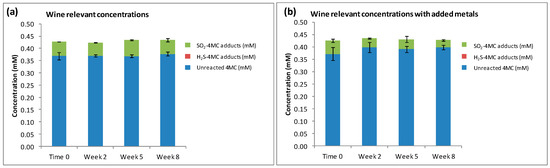
Figure 4.
The reaction products measured when 4MBQ (0.45 mM) was added to model wine containing: (a) wine relevant concentrations of H2S (0.39 µM) and SO2 (0.23 mM); and (b) wine relevant concentrations of H2S (0.39 µM), SO2 (0.23 mM), as well as Cu2+ (0.5 mg/L) and Fe3+ (4.0 mg/L).
The concentrations of SO2-4MC adducts measured in samples with added Cu2+ and Fe3+ were significantly decreased when compared with the samples without any added metal ions (p = 0.019) (Figure 4b), and a peak ratio of 1:2:1 was measured for the three main SO2-4MC adducts. This decrease in SO2-4MC adducts may be attributed to the interaction between SO2 and the metal ions when reacting with oxygen as described by Danilewicz [29]. No H2S-4MC adducts were measured in samples with added metals, even though a small concentration of H2S was measured at Time 0 (Figure S1). No H2S was observed in the samples post Time 0, which may be attributed to the reaction between Cu2+ and H2S, a reaction through which CuS complexes are produced, preventing H2S from reacting with the quinone to produce H2S-4MC adducts.
3. Discussion
3.1. Impact of Oxygen on the Formation of H2S from Cu2+ + SO2 Interaction in This Study
DO was monitored over the course of the experiment using PreSens oxygen sensors applied to representative colorless vials (in triplicate) for a control sample (blank wine) and for each of the treatments (Cu2+, SO2, and “Cu2+ + SO2”) for the Verdelho (n = 12) and for the Shiraz (n = 12) wine samples. The rate of oxygen consumption varied between the Verdelho and Shiraz wine samples, with the Shiraz samples consuming the available oxygen faster than the Verdelho samples. The rate of oxygen consumption also varied between the Cu2+ and SO2 treated wine samples (Figure S2). Verdelho samples treated with SO2 displayed the fastest rate of oxygen consumption, and samples treated with Cu2+ also displayed a faster oxygen consumption rate than samples without any added Cu2+ (Figure S2a). The interaction between oxygen and SO2 in wine, the differences in oxygen consumption rates between red and white wine, and the effect of metals on the oxygen consumption rate in wine is in agreement with trends observed by other authors [17,19,24,29]. After two months of storage under low oxygen conditions, both Verdelho and Shiraz samples had consumed all available oxygen and samples can be considered to be effectively oxygen-free (Figure S2a,b).
3.2. Formation of H2S Associated with Cu2+ + SO2 Treatment
The role of the interactive effects of residual Cu2+ and free SO2 in the modulation of H2S concentrations in wines post-bottling has not previously been considered. A significant source of Cu2+ in wines is through intentional addition during copper fining remediation treatments, and the dosage and the timing of these Cu2+ additions are determined by the winemaker. Similarly, SO2 can be added pre-fermentation, post-fermentation, during maturation and before bottling. In this study, wines were not previously treated with Cu2+ or SO2. The first additions of Cu2+ and SO2 for the Verdelho and Shiraz wines represented an “at-bottling” treatment for both Cu2+ and SO2. In the Verdelho samples, treatment with “Cu2+ + SO2” significantly increased H2S concentrations after 12 months of anaerobic storage. Concentrations of up to 36.01 (±18.1) µg/L H2S were measured in the “Cu2+ + SO2” treated Verdelho samples compared to the 0.879 (±0.028) µg/L of H2S in the control samples. This study has demonstrated that in wines produced without early Cu2+ and SO2 additions, but treated with these additives before bottling may increase the risk of developing elevated H2S concentrations post-bottling. The high concentration of free SO2 available due to late SO2 treatment just before bottling may explain why the interactive effects of Cu2+ and SO2 were associated with such significant increased H2S concentrations in Verdelho wines.
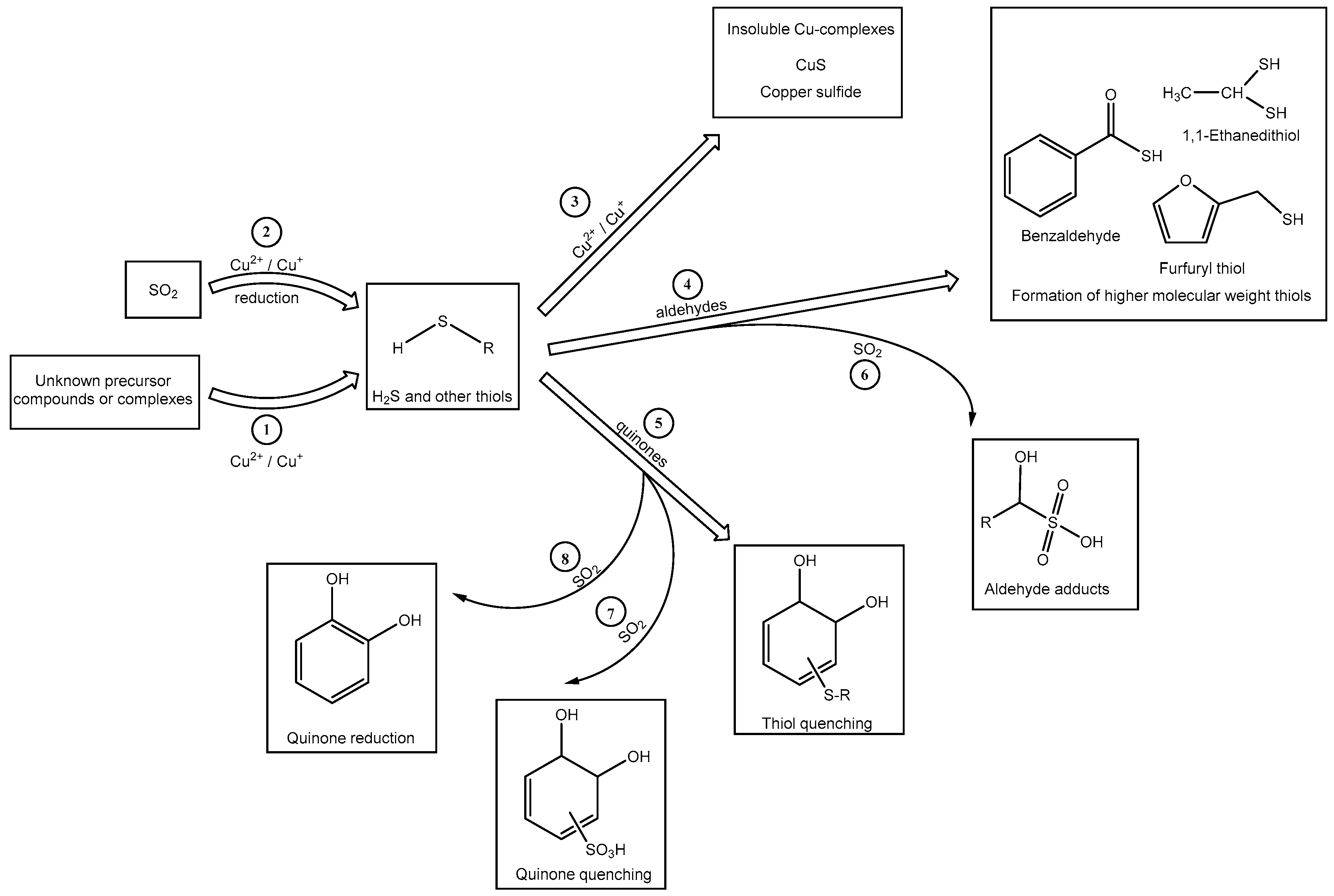
The exact mechanism for the formation of H2S due to the treatment of Verdelho samples with “Cu2+ + SO2” is not yet known (Figure 1). It is possible that in the samples spiked with “Cu2+ and SO2”, H2S is produced through a Cu2+ mediated reaction (Scheme 1, (1)) from unknown precursors naturally present in the wine, either as complexes or in molecular precursor forms. The produced H2S can be quenched through addition to either wine quinones or aldehydes [Scheme 1, (4) and (5)]. However, when a high concentration of free SO2 is available, competing reactions between SO2 and H2S for quinones/aldehydes may occur (Scheme 1, (6), (7), and (8)). It is known that nucleophilic addition reactions compete with the two electron reduction of quinones by SO2 [22]. The first-order rate constants for the addition of SO2 or H2S to quinones are practically identical [21], thus the concentration of these nucleophiles determines the preferential addition of SO2 or H2S to quinone molecules. Given that SO2 was added in mM concentrations, whereas H2S was produced in µM concentrations, the addition of SO2 to quinone molecules or the reduction of the quinones via SO2 may be the favored reaction (Scheme 1, (7)). The reaction of bisulfite with wine quinones to produce the sulfonic acid derivative or that reduces the quinone back to its corresponding diphenolic compound [30], may prevent the quenching of thiols and thereby indirectly increase H2S concentrations in the Verdelho samples. Another possible quenching reaction could be through the addition of H2S to wine carbonyl compounds such as acetaldehyde, a compound known to readily react with thiols [31]. The availability of free SO2 and the formation of H2S through Cu2+ mediated reactions would lead to competition reactions between these two sulfur containing molecules for the available carbonyl compound (Scheme 1, (4)), which could result in a higher concentration of H2S in samples treated with SO2 than in treatments without SO2. Another explanation may be the suggested reduction of SO2 by Cu2+ that results in the direct formation of H2S (Scheme 1, (2)), a proposed mechanism for wine post-bottling by Ribéreau-Gayon and Lopes et al. [24,25].
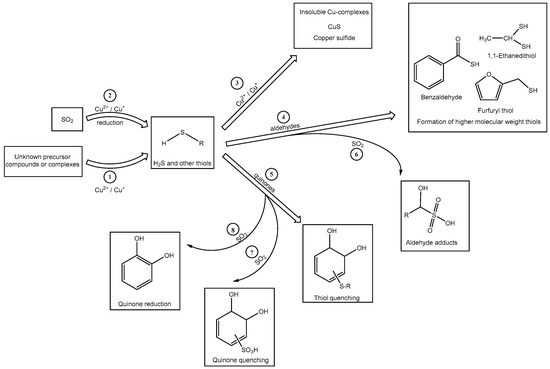
Scheme 1.
Possible mechanisms for the formation of H2S and other low molecular weight thiol compounds and the interaction between Cu2+, SO2, and “Cu2+ + SO2” with wine compounds such as aldehydes and o-quinones, during H2S formation.
The benefits of treating wine with SO2 offsets the risk of developing “reduced” aromas, and this study demonstrated that even late SO2 additions close to bottling did not pose a risk of increased H2S unless there was a high concentration of residual Cu2+ present in the wines. This study emphasizes the “Cu2+ + SO2” interaction effect and that increased free SO2 concentrations combined with elevated residual Cu2+ concentrations in wines post-bottling poses a risk of increasing H2S concentrations post-bottling. As such it is important to ensure that residual Cu2+ concentrations, which can be controlled by the winemaker, are kept as low as possible in finished wine.
In the Shiraz samples treated with “Cu2+ + SO2” the significant increases in H2S concentrations were associated with Cu2+ treatment (p < 0.001, Table 1) and not associated with the combined Cu2+ and SO2 treatments (Table 1, Figure 1b). It is important to note that the Shiraz wine had approximately 50% less free SO2 than the Verdelho wine, and in red wine a significant proportion of the free SO2 is loosely bound to anthocyanins and other phenolic compounds [17] and as such may not be as available for participation in reactions associated with increased H2S concentrations. In the Verdelho wine samples, a higher concentration of free SO2 was available to be involved in reactions associated with the increase in H2S. This difference in free SO2 concentrations may have contributed to the varying effects of Cu2+ and “Cu2+ + SO2” measured in these two red and white wines. A previous report [10] also showed the varying effect of Cu2+ on H2S formation in Chardonnay relative to Shiraz wines and demonstrated that H2S increased in Shiraz samples spiked with Cu2+ when stored in a low oxygen environment, but not in Chardonnay samples treated in the same way. The opposing Cu2+ effects seen in this study for the Verdelho wine samples relative to Shiraz wine samples again illustrates the complexity of the reactions taking place in wines with different matrices.
3.3. Mechanisms Modulating H2S Formation
The experiments described in Section 2.3.1 where the “Cu2+ + SO2” treatment was applied to model wine demonstrated that no H2S was produced in model wine containing only water, EtOH, tartaric acid, Cu2+, and SO2. This shows that other wine matrix compounds were necessary to produce the significantly increased H2S concentrations measured in the Verdelho samples treated with “Cu2+ + SO2”. The increased H2S concentration measured in Verdelho samples treated with both “Cu2+ + SO2” may be the result of the interaction of wine compounds such as wine quinones, aldehydes or any other wine compounds that possess the ability to react with H2S.
The quinone (4MBQ) of a representative wine polyphenol (4MC) was added to model wine and the competition reactions between H2S and SO2 were studied at equimolar concentrations as well as at wine relevant concentrations of H2S and SO2. It is known that H2S and SO2 have similar first order rate constants [21] and when H2S and SO2 were added at wine relevant concentrations with SO2 concentrations significantly greater than H2S concentrations, the SO2-quinone adducts were the major reaction product produced. This experiments showed that concentrations in which sulfur nucleophiles are present in wine would determine the major quinone-adduct produced in the case of sulfur nucleophiles with similar first order rate constants. When H2S and SO2 are involved in such competition reactions at wine relevant concentrations, fewer H2S molecules will be quenched through reactions with quinones, leading to an indirect increase in H2S concentrations (Scheme 1 (5) and (7)). These results suggest that the effects of Cu2+ and SO2 may operate independently on separate pathways, but the effects of the additives are cumulative, leading to an increased H2S concentration.
This study has shown that the presence of wine additives, such as Cu2+ and SO2, play a fundamental role in the modulation of H2S profiles in both red and white wine post-bottling. Although the exact reaction pathway for the formation of H2S associated with the combination of Cu2+ and SO2 is not yet known, there are a few possible mechanisms. When a higher ratio of free SO2 to H2S is available, it can form adducts with wine quinones or reduce them back to their corresponding phenols, and in doing so prevent the quenching of the newly formed thiols. Competing reactions between H2S and SO2 could also result in a smaller percentage of H2S lost through, for example, reactions with acetaldehyde. This could indirectly lead to a relative increase in thiol concentration in the treatments with a high concentration of carbonyl compounds. Another explanation may be the suggested reduction of SO2 by Cu2+ that might lead to the formation of H2S, as proposed by Ribéreau-Gayon [24] and Lopes et al. [25], although studies in our model wine have not supported this.
Recent work by Franco-Luesma and Ferreira proposed two methods to access H2S production from “loosely bound” vs. “de novo” formation [32]. In this study a method that is comparable with the “total” H2S quantification method was applied that relied on fully releasing all the H2S into the wine headspace through the addition of 2 g of salt into 10 mL of wine [28]. As such the H2S loosely bound were fully released, measured and quantified in all samples at the first measurement at Day 0. The same method was used to analyze the samples over a 12-month period and increased H2S concentrations were statistically significantly associated with the combined “Cu2+ + SO2” treatment and H2S concentration was shown to increase over time in samples, indicating de novo formation of H2S associated with “Cu2+ + SO2” treatment.
The interactive effects of Cu2+ and SO2 have not previously been explored as a significant factor associated with the accumulation of H2S in wines post-bottling. The majority of commercial wines are treated with SO2 and many contain Cu2+ incorporated through uptake from the soil, pickup during the winemaking process, or by direct addition of Cu2+ through remediation treatments later in production. Fully understanding the timing effects of early and/or late SO2 and Cu2+ additions will help determine whether a wine could be placed at risk of developing elevated H2S concentrations later during storage.
4. Materials and Methods
4.1. Materials
Amberlyst A26 hydroxide form, ethanol (99.5%), ethylmethyl sulfide (EMS, 96%), iron (III) sulfate hydrate (97%), 4-methylcatechol (4MC, 95%), periodic acid (99%), potassium hydrogen tartrate (99%), potassium metabisulfite (PMS, 98%), and sodium hydrosulfide hydrate (NaSH·xH2O, 71%) were obtained from Sigma-Aldrich (Castle Hill, NSW, Australia). Propyl thioacetate (99.7%) was obtained from Lancaster Synthesis (Jomar Bioscience, Kensington, SA, Australia). Acetonitrile (ACN, gradient grade for liquid chromatography), methanol (MeOH, 99.8% by GC), ortho-phosphoric acid (85%), tartaric acid, tetrahydrofuran (THF), and sodium chloride were obtained from Merck (Frenchs Forest, NSW, Australia). Water was obtained from a Milli-Q purification system (Millipore, North Ryde, NSW, Australia). Copper (II) sulfate pentahydrate (99%) was purchased from Ajax Chemicals (Sydney, NSW, Australia). Formic acid (98%–100%) was purchased from Rowe Scientific (Lonsdale, SA, Australia).
4.2. Wine Samples
Certified organic Verdelho wine from the 2012 vintage and Shiraz wine from the 2010 vintage, produced in South Eastern Australia, were obtained from a local winery. Neither wine was treated with either SO2 or Cu2+ during winemaking. Analyses of the chemical compositions of the two base wines were conducted by The Australian Wine Research Institute (AWRI) Analytical Service (ISO 17025 Laboratory, Adelaide, Australia) and are as follows: pH 3.33, 5.8 g/L residual sugars,12.63% (v/v) alcohol, 0.85 g/L volatile acidity (as acetic acid), 6.1 g/L titratable acidity (as tartaric acid), <4 mg/L free SO2 and <4 mg/L total SO2 for the Verdelho wine; and pH 3.46, 0.7 g/L residual sugars, 14.0% (v/v) alcohol, 0.37 g/L volatile acidity (as acetic acid), 6.6 g/L titratable acidity (as tartaric acid), <4 mg/L free SO2 and <4 mg/L total SO2 for the Shiraz wine.
4.3. Chemical Analyses
4.3.1. Oxygen Measurement
Colorless 20 mL crimp top vials (Chromacol, Part of Thermo Fisher Scientific Inc., Scoresby, VIC, Australia) were fitted with PreSens Pst6 oxygen sensors (Presens, Regensburg, Germany) and filled with the three treatments (Cu2+, SO2, and “Cu2+ + SO2”) and control samples (without any treatments) in triplicate for the Verdelho (n = 12) and Shiraz wine (n = 12) and were used to measure DO during storage of the wines. Samples were stored at 20 °C under light free conditions and under nitrogen (N2) as described by Viviers et al. [10]. Oxygen measurements were carried out using a PreSens Fibox 3 trace v3 oxygen meter, with the limit of detection when using a PtS6 sensor of 1 ppb oxygen. (Presens, Regensburg, Germany).
4.3.2. Metal Quantifications
Base wines were analyzed for their metal concentrations by Flinders Analytical, Flinders University (Adelaide, Australia) using an Agilent 7500 cx inductively coupled plasma mass spectrometers (Agilent Technologies, Tokyo, Japan) as described in Thiel et al. [33]. Stock solutions of Cu2+ were prepared volumetrically in an anaerobic hood using degassed water (Milli-Q), and then measured for Cu2+ concentrations. The concentrations of total Cu2+ in the base Verdelho and Shiraz wines, as well as the spiked total Cu2+ concentrations in the Verdelho and Shiraz wines are given in Table 3. The stock solution of Fe3+ was prepared as for Cu2.

Table 3.
Concentrations of Cu2+ and SO2 in the base wines, spiked samples and average metal and SO2 concentrations for Australian wines.
4.3.3. Determination of SO2
Base wines and the SO2 stock solution prepared for addition to the samples were analyzed for their free and total SO2 concentrations using the Lachat flow injection analysis (FIA) system by the AWRI Analytical Service (ISO 17025 Laboratory, Adelaide, Australia). Fresh stock solutions of SO2 were prepared volumetrically in an anaerobic hood by dissolving PMS in degassed water (Milli-Q) on the day of the SO2 addition and discarded after use. The concentrations of SO2 in the base Verdelho and Shiraz wines, as well as the spiked SO2 concentrations in the Verdelho, Shiraz, and model wines are given in Table 3.
4.3.4. Preparation of Reaction Products of H2S and SO2 with 4-Methylbenzoquinone
The preparation of 4MBQ by periodate resin was as described by Jongberg et al. [37] and Nikolantonaki et al. [21], with 4MBQ used immediately after synthesis. The adducts of SO2 and H2S, used for retention time reference standards and for the comparison of mass spectral data, were synthesized as follows: 4MBQ (1.1 mM) was prepared as described by Nikolantonaki et al. [21] and dissolved in cold (4 °C) model wine (12% EtOH, 10 g/L tartaric acid, pH 3.4) containing SO2 (4 mM) or H2S (4 mM). The reaction mixture was stirred at 20 °C for 15 min. All reaction products were characterized using LC-HRMS (Table 2), as well as comparing their mass spectra with published spectral data [21].
The competition reaction between SO2 and H2S with 4MBQ was studied in three separate experiments. First, synthesized 4MBQ (1 mM) was added to cold (4 °C) model wine (12% EtOH, 10 g/L tartaric acid, pH 3.4) that already contained SO2 (2 mM) and H2S (2 mM) at equimolar concentrations. The second and third experiments were conducted at wine relevant concentrations, where 4MBQ (0.45 mM) [38,39] was added to cold (4 °C) model wine (12% EtOH, 10 g/L tartaric acid, pH 3.4) that already contained either (2) SO2 (0.23 mM) [36] and H2S (0.39 µM) [28]; or (3) SO2 (0.23 mM) [36] and H2S (0.39 µM) [28] with added Cu2+ (0.5 mg/L) [34] and Fe3+ (4.0 mg/L) [34]. In all three experiments, the reaction mixtures were stirred at 20 °C for 15 min. The reaction between the nucleophiles and 4MBQ was followed using HPLC. Additionally, residual H2S and SO2 concentrations were measured using GC-SCD and FIA, respectively.
4.3.5. Analysis of 4-Methylbenzoquinone Adducts Using Liquid Chromatography
The reaction between 4MBQ with SO2 and H2S was monitored and the reaction products quantified using the high pressure liquid chromatography (HPLC) as described in Mercurio et al. [40] with slight modification. Briefly, samples were analyzed using an Agilent 1100 LC (Agilent, Mulgrave, VIC, Australia) with a Phenomenex Synergi Hydro-RP C18 column (150 mm × 2 mm, 4 μm) at 25 °C. Solvent A consisted of 1% ACN and 1.5% phosphoric acid in H2O, and solvent B was 80:20 ACN/Solvent A, for gradient elution at a flow rate of 0.4 mL/min: 0 min (0% solvent B), 10 min (5% solvent B), 15 min (20% solvent B), 17 min (100% solvent B), and 30 min (0% solvent B).
The reaction products of 4MBQ with SO2 and H2S were quantified using LCMS as well as LC-HRMS. An Agilent 1200 HPLC system (Forest Hill, Victoria, Australia) equipped with binary pump (1290 model), degasser, autosampler, column oven and diode array detector (DAD) was used. A 5 µL aliquot of a sample was injected and separated on a 150 × 2 mm i.d., 4 µm, 80 Å, Synergi Hydro-RP column connected with a 4 × 2 mm i.d. guard column packed with the same material (Phenomenex, Lane Cove, NSW, Australia). The column temperature was maintained at 20 °C during the HPLC run. A binary gradient with mobile phases consisting of 0.5% formic acid in water (solvent A) and 0.5% formic acid in acetonitrile (solvent B) was used. The elution conditions were as follows: a flow rate of 400 µL/min, a linear gradient of solvent B held at 0% for 5 min, from 0 to 5% in 5 min, from 5% to 20% in 5 min, from 20% to 100% in 2 min and from 100% to 0% for 13 min. A 5 min re-equilibration time at 0% of solvent B was programmed between runs. The effluent from the column was monitored by DAD acquiring absorbance at wavelengths of 254, 280 and 320 nm, followed by introduction to a mass spectrometer through an electrospray (ESI) interface.
For LCMS analysis, a 4000 QTRAP mass spectrometer equipped with a Turbo ion source (Sciex, Mt Waverly, Victoria, Australia) was connected to the outlet of DAD with a PEEK tubing (0.13 mm i.d.). Data acquisition and processing were performed using Analyst software 1.6.2 (Sciex, Mt Waverley, VIC, Australia). ESI negative ion mass spectra were recorded in scan mode ranging from m/z 50 to m/z 1000 with a scan time of 1 s and step size of 0.1 Da. Nitrogen was used for curtain, nebulizer and heated turbo (500 °C) gases set at 20 psi, 50 psi and 50 psi, respectively. The electrospray and declustering potentials were set at −4200 V and −50 V, respectively.
The LC-HRMS analysis was performed on an Agilent HPLC 1200 coupled to a Bruker microTOF-Q II (Bruker Singapore, The Helios, Singapore). Mass calibration was performed on each file using Bruker Daltonic’s DataAnalysis v4.1 “Enhanced Quadratic” calibration method (Bruker Singapore, The Helios, Singapore). All other data analysis including chemical formula predictions was done using Bruker Daltonic’s DataAnalysis 4.3. Data were acquired using ESI negative ionisation in scan mode from m/z 50 to 1650 with a scan speed of 2 spectra/s. Nitrogen was used for the nebulizer and dry gas (200 °C) which were set at 2 bar and 7 L/min, respectively. The capillary voltage was 3500 V and endplate offset was −500 V. DAD acquisition range was 200–600 nm.
4.3.6. Gas Chromatography Coupled to Sulfur Chemiluminescence Detection
All H2S measurements in the Verdelho and Shiraz base wines, as well as the treated samples, were determined using an Agilent 355 sulfur chemiluminescence detector (SCD) coupled to an Agilent 6890A gas chromatograph (Forest Hill, VIC, Australia). Experimental and analytical parameters, as well as the analysis method were as described by Siebert et al. [28] without modification.
4.4. Sample Preparation and Analysis
Preparation of Verdelho, Shiraz, and Model Wine Samples Spiked with Cu2+ + SO2
Wine samples and all stock solutions were prepared in a low oxygen atmosphere by making use of an anaerobic bag (Aldrich® AtmosBag, Sigma-Aldrich, Castle Hill, NSW, Australia) fitted with a PreSens Pst6 oxygen sensor (Presens, Regensburg, Germany). The bag was flushed down to <1 ppb oxygen with N2 (g) at the start of the sample preparation and a slight positive pressure was maintained to prevent oxygen ingress into the bag during experimental setup. The oxygen concentration in the anaerobic bag was continuously monitored and maintained at <15 ppb. The Verdelho, Shiraz, and model (12% EtOH, 10 g/L tartaric acid, pH = 3.4) base wines were placed inside the anaerobic bag and sub-sampled by placing 10 mL of wine into a 20 mL crimp top amber vial (Chromacol, Thermo Fisher Scientific Inc., Scoresby, VIC, Australia). The Verdelho and Shiraz wines were each divided into four treatments repeated in triplicate (n = 12): (1) Cu2+ treatment (1 mg/L for Verdelho, Shiraz and model wines); (2) SO2 treatment (Verdelho: free 87.8 mg/L, total 100.0 mg/L; Shiraz: free 40.8 mg/L, total 60 mg/L; model wine: free 99.67 mg/L, total 100 mg/L); (3) “Cu2+ + SO2” treatment, where the above mentioned levels of Cu2+ and SO2 were combined into one treatment for Verdelho, Shiraz, and model wines; and (4) the control Verdelho, Shiraz, and model wine samples were not treated with either Cu2+ or SO2 (Table 3).
Stock solutions of Cu2+, and SO2 were prepared in such a manner that the high level of each metal was added by spiking 50 µL of the appropriate stock solution to the 10 mL wine sample using a pipette, avoiding significant dilution effects. After the samples were prepared with the four treatments, they were sealed with 20 mm magnetic crimp caps with 8 mm center and blue PTFE/silicon septa (118 mm) (Grace Davison Discovery Sciences, Rowville, VIC, Australia) and stored at room temperature (20 °C) in 19 L post mix Cornelius kegs (Ambar technology, Alexandria, NSW, Australia). The lid of each keg was fitted with a PreSens Pst6 oxygen sensor (Presens, Regensburg, Germany). The kegs were flushed with N2 (g) until they reached <1 ppb oxygen and maintained at a slight positive pressure of 1 psi N2 (g) to prevent any oxygen ingress into the keg storage systems. The kegs were continuously monitored and not allowed to exceed an oxygen measurement of 25 ppb, and flushed with N2 (g) to <1 ppb if increased oxygen concentrations were measured.
Verdelho, Shiraz, and model wine samples were analyzed for their VSC concentrations immediately after spiking with metals (Day 0), and then following 1 month (Month 1), 3 months (Month 3), 6 months (Month 6) and 12 months (Month 12) of storage under low oxygen conditions.
4.5. Statistical Analyses
All significance tests (Student’s t-test, ANOVAs and Tukey analyses) were conducted using GraphPad Prism statistics software (v6.04 GraphPad Software Inc., La Jolla, CA, USA). Multi-way analysis of variance (ANOVA) and Dunnett multiple comparison post-hoc tests were used to determine which treatments were associated with significant effects on the formation of H2S over the course of the 12-month experiment, and statistical significance was assigned if p < 0.05. All values are represented as mean ± standard deviation (SD).
Supplementary Materials
The following are available online at http://www.mdpi.com/1420-3049/21/9/1214/s1, Figure S1: Residual H2S concentrations measured in samples that replicated wine-like conditions, with 4MBQ at 0.45 mM, H2S at 0.39 µM, and SO2 0.23 mM.; as well as the 4MBQ (0.45 mM), H2S (0.39 µM), and SO2 (0.23 mM) with added Cu2+ (0.5 mg/L) and Fe3+ (4.0 mg/L), Figure S2: Dissolved oxygen consumption measured from Day 0 to Month 12 for: (a) Verdelho samples; and (b) Shiraz samples.
Acknowledgments
The authors would like to thank Cory Black (The Australian Wine Research Institute) for his support and guidance in the synthesis of the SO2-4MC and H2S-4MC adducts, Yoji Hayasaka (The Australian Wine Research Institute) for LCMS analysis, and Natoiya Lloyd of Metabolomics Australia (South Australian Metabolomics Facility, The Australian Wine Research Institute, Adelaide, SA, Australia) for high resolution accurate mass analysis. The authors would also like to thank Geoff Scollary (The University of Melbourne) for critical review of the manuscript. The AWRI is a member of the Wine Innovation Cluster in Adelaide. The work was supported by Australia’s grape growers and winemakers through their investment body, the Wine Australia, with matching funds from the Australian Government.
Author Contributions
Mark Smith conceived the idea; Marlize Bekker designed and performed the experiments and analyzed the data; and Eric Wilkes, Paul Smith, and Mark Smith contributed to data interpretation and all authors were involved in the writing of the paper.
Conflicts of Interest
The authors declare no conflict of interest.
Abbreviations
The following abbreviations are used in this manuscript:
| 4MBQ | 4-methylbenzoquinone |
| 4MC | 4-Methylcatechol |
| ANOVA | Analysis of variance |
| AVG | Average |
| AWRI | Australian Wine Research Institute |
| DO | Dissolved oxygen |
| GSH | Glutathione |
| H2O2 | Hydrogen peroxide |
| H2S | Hydrogen sulfide |
| HPLC | High pressure liquid chromatography |
| LCMS | Liquid chromatography-mass spectrometry |
| LC-HRMS | Liquid chromatography-high resolution mass spectrometry |
| MeSH | Methanethiol |
| MeOH | Methanol |
| OT | Odor threshold |
| PMS | Potassium hydrogen tartrate |
| SO2 | Sulfur dioxide |
| SD | Standard deviation |
| THF | Tetrahydrofuran |
| VSCs | Volatile sulfur compounds |
References
- Smith, M.E.; Bekker, M.Z.; Smith, P.A.; Wilkes, E.N. Sources of volatile sulfur compounds in wine. Aust. J. Grape Wine Res. 2015, 21, 705–712. [Google Scholar] [CrossRef]
- Siebert, T.E.; Bramley, B.; Solomon, M.R. Hydrogen sulfide: Aroma detection threshold study in white and red wine. AWRI Tech. Rev. 2009, 183, 14–16. [Google Scholar]
- Villamor, R.R.; Ross, C.F. Wine matrix compounds affect perception of wine aromas. Annu. Rev. Food Sci. Technol. 2013, 4, 1–20. [Google Scholar] [CrossRef] [PubMed]
- Ugliano, M.; Kwiatkowski, M.; Vidal, S.; Capone, D.; Siebert, T.; Dieval, J.B.; Aagaard, O.; Waters, E.J. Evolution of 3-mercaptohexanol, hydrogen sulfide, and methyl mercaptan during bottle storage of Sauvignon blanc wines. Effect of glutathione, copper, oxygen exposure, and closure-derived oxygen. J. Agric. Food Chem. 2011, 59, 2564–2572. [Google Scholar] [CrossRef] [PubMed]
- Bekker, M.Z.; Day, M.P.; Holt, H.; Wilkes, E.; Smith, P.A. Effect of oxygen exposure during fermentation on volatile sulfur compounds in Shiraz wine and a comparison of strategies for remediation of reductive character. Aust. J. Grape Wine Res. 2016, 22, 24–35. [Google Scholar] [CrossRef]
- Day, M.P.; Schmidt, S.A.; Smith, P.A.; Wilkes, E.N. Use and impact of oxygen during winemaking. Aust. J. Grape Wine Res. 2015, 21, 693–704. [Google Scholar] [CrossRef]
- Franco-Luesma, E.; Ferreira, V. Reductive off-odors in wines: Formation and release of H2S and methanethiol during the accelerated anoxic storage of wines. Food Chem. 2016, 199, 42–50. [Google Scholar] [CrossRef] [PubMed]
- Franco-Luesma, E.; Ferreira, V. Formation and release of H2S, methanethiol, and dimethylsulfide during the anoxic storage of wines at room temperature. J. Agric. Food Chem. 2016, 64, 6317–6326. [Google Scholar] [CrossRef] [PubMed]
- Nedjma, M.; Hoffmann, N. Hydrogen sulfide reactivity with thiols in the presence of copper(II) in hydroalcoholic solutions cognac brandies: Formation of symmetrical and unsymmetrical dialkyl trisulfides. J. Agric. Food Chem. 1996, 44, 3935–3938. [Google Scholar] [CrossRef]
- Viviers, M.Z.; Smith, M.E.; Wilkes, E.; Smith, P. Effects of five metals on the evolution of hydrogen sulfide, methanethiol, and dimethyl sulfide during anaerobic storage of Chardonnay and Shiraz wines. J. Agric. Food Chem. 2013, 61, 12385–12396. [Google Scholar] [CrossRef] [PubMed]
- Walker, M.D. The influence of metal-ions on concentrations of flavor-active sulfur-compounds measured in beer using dynamic headspace sampling. J. Sci. Food Agric. 1995, 67, 25–28. [Google Scholar] [CrossRef]
- Kreitman, G.Y.; Danilewicz, J.C.; Jeffery, D.W.; Elias, R.J. Reaction mechanisms of metals with hydrogen sulfide and thiols in model wine. Part 1: Copper-catalyzed oxidation. J. Agric. Food Chem. 2016, 64, 4095–4104. [Google Scholar] [PubMed]
- Clark, A.C.; Grant-Preece, P.; Cleghorn, N.; Scollary, G.R. Copper(II) addition to white wines containing hydrogen sulfide: Residual copper concentration and activity. Aust. J. Grape Wine Res. 2015, 21, 30–39. [Google Scholar] [CrossRef]
- Clark, A.C.; Kontoudakis, N.; Barril, C.; Schmidtke, L.M.; Scollary, G.R. Measurement of labile copper in wine by medium exchange stripping potentiometry utilising screen printed carbon electrodes. Talanta 2016, 154, 431–437. [Google Scholar] [CrossRef] [PubMed]
- Bekker, M.Z.; Mierczynska-Vasilev, A.; Smith, P.A.; Wilkes, E.N. The effects of pH and copper on the formation of volatile sulfur compounds in Chardonnay and Shiraz wines post-bottling. Food Chem. 2016, 207, 148–156. [Google Scholar] [CrossRef] [PubMed]
- Jackson, R.S. Wine Science Principles and Applications, 3rd ed.; Elsevier Inc.: San Diego, CA, USA, 2008; p. 307. [Google Scholar]
- Danilewicz, J.C. Mechanism of autoxidation of polyphenols and participation of sulfite in wine: Key role of iron. Am. J. Enol. Vitic. 2011, 62, 319–328. [Google Scholar] [CrossRef]
- Elias, R.J.; Waterhouse, A.L. Controlling the fenton reaction in wine. J. Agric. Food Chem. 2010, 58, 1699–1707. [Google Scholar] [CrossRef] [PubMed]
- Danilewicz, J.C. Reactions involving iron in mediating catechol oxidation in model wine. Am. J. Enol. Vitic. 2013, 64, 316–324. [Google Scholar] [CrossRef]
- Waterhouse, A.L.; Laurie, V.F. Oxidation of wine phenolics: A critical evaluation and hypotheses. Am. J. Enol. Vitic. 2006, 53, 306–313. [Google Scholar]
- Nikolantonaki, M.; Waterhouse, A.L. A method to quantify quinone reaction rates with wine relevant nucleophiles: A key to the understanding of oxidative loss of varietal thiols. J. Agric. Food Chem. 2012, 60, 8484–8491. [Google Scholar] [CrossRef] [PubMed]
- Nikolantonaki, M.; Magiatis, P.; Waterhouse, A.L. Measuring protection of aromatic wine thiols from oxidation by competitive reactions vs wine preservatives with ortho-quinones. Food Chem. 2014, 163, 61–67. [Google Scholar] [CrossRef] [PubMed]
- Rankine, B.C. Making Good Wine; Pan Macmillan Australia Pt Limited: Sydney, Australia, 2004; pp. 96–99. [Google Scholar]
- Ribereau-Gayon, P.; Glories, Y.; Maujean, A.; Dubourdieu, D. Handbok of Enology: The Chemistry of Wine Stabilization and Treatments, 2nd ed.; John Wiley & Sons Ltd.: Chichester, UK, 2006; Volume 2, pp. 103, 393. [Google Scholar] [CrossRef]
- Lopes, P.; Silva, M.A.; Pons, A.; Tominaga, T.; Lavigne, V.; Saucier, C.; Darriet, P.; Teissedre, P.L.; Dubourdieu, D. Impact of oxygen dissolved at bottling and transmitted through closures on the composition and sensory properties of a sauvignon blanc wine during bottle storage. J. Agric. Food Chem. 2009, 57, 10261–10270. [Google Scholar] [CrossRef] [PubMed]
- Nikolantonaki, M.; Jourdes, M.; Shinoda, K.; Teissedre, P.L.; Quideau, S.; Darriet, P. Identification of adducts between an odoriferous volatile thiol and oxidized grape phenolic compounds: Kinetic study of adduct formation under chemical and enzymatic oxidation conditions. J. Agric. Food Chem. 2012, 60, 2647–2656. [Google Scholar] [CrossRef] [PubMed]
- Ugliano, M. Oxygen contribution to wine aroma evolution during bottle aging. J. Agric. Food Chem. 2013, 61, 6125–6136. [Google Scholar] [CrossRef] [PubMed]
- Siebert, T.E.; Solomon, M.R.; Pollnitz, A.P.; Jeffery, D.W. Selective determination of volatile sulfur compounds in wine by gas chromatography with sulfur chemiluminescence detection. J. Agric. Food Chem. 2010, 58, 9454–9462. [Google Scholar] [CrossRef] [PubMed]
- Danilewicz, J.C. Interaction of sulfur dioxide, polyphenols, and oxygen in a wine-model system: Central role of iron and copper. Am. J. Enol. Vitic. 2007, 58, 53–60. [Google Scholar]
- Danilewicz, J.C.; Seccombe, J.T.; Whelan, J. Mechanism of interaction of polyphenols, oxygen, and sulfur dioxide in model wine and wine. Am. J. Enol. Vitic. 2008, 59, 128–136. [Google Scholar]
- Rauhut, D.; Kurbel, H.; Dittrich, H.H. Sulfur compounds and their influence on wine quality. Wein Wiss. 1993, 48, 214–218. [Google Scholar]
- Franco-Luesma, E.; Ferreira, V. Quantitative analysis of free and bonded forms of volatile sulfur compouds in wine. Basic methodologies and evidences showing the existence of reversible cation-complexed forms. J. Chromatogr. A 2014, 1359, 8–15. [Google Scholar] [CrossRef] [PubMed]
- Thiel, G.; Geisler, G.; Blechschmidt, I.; Danzer, K. Determination of trace elements in wines and classification according to their provenance. Anal. Bioanal. Chem. 2004, 378, 1630–1636. [Google Scholar] [CrossRef] [PubMed]
- Martin, A.E.; Watling, R.J.; Lee, G.S. The multi-element determination and regional discrimination of Australian wines. Food Chem. 2012, 133, 1081–1089. [Google Scholar] [CrossRef]
- Analytical Requirements for the Export of Australian Wine. The Australian Wine Research Institute, 2016. Available online: http://www.awri.com.au/industry_support/regulatory_assistance/export_requirements/ (accessed on 29 June 2016).
- Peterson, G.F.; Kirrane, M.; Hill, N.; Agapito, A. A comprehensive survey of the total sulfur dioxide concentration of American wines. Am. J. Enol. Vitic. 2000, 51, 189–191. [Google Scholar]
- Jongberg, S.; Gislason, N.E.; Lund, M.N.; Skibsted, L.H.; Waterhouse, A.L. Thiol-quinone adduct formation in myofibrillar proteins detected by lc-ms. J. Agric. Food Chem. 2011, 59, 6900–6905. [Google Scholar] [CrossRef] [PubMed]
- Fisher, U.; Noble, A.C. The effect of ethanol, catechin concetration, and pH on sourness and bitterness of wine. Am. J. Enol. Vitic. 1994, 45, 6–10. [Google Scholar]
- Komes, D.; Ulrich, D.; Kovacevic, G.; Lovric, T. Study of phenolic and volatile composition of white wine during fermentaion and a short time of storage. Vitis 2007, 46, 77–84. [Google Scholar]
- Mercurio, M.D.; Dambergs, R.G.; Herderich, M.J.; Smith, P.A. High throughput analysis of red wine and grape phenolics-adaptation and validation of methyl cellulose precipitable tannin assay and modified Somers color assay to a rapid 96 well plate format. J. Agric. Food Chem. 2007, 55, 4651–4657. [Google Scholar] [CrossRef] [PubMed]
- Sample Availability: Not Available.
© 2016 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license ( http://creativecommons.org/licenses/by/4.0/).