Synthesis and Antimicrobial Characterization of Half-Calycanthaceous Alkaloid Derivatives
Abstract
:1. Introduction
2. Results and Discussion
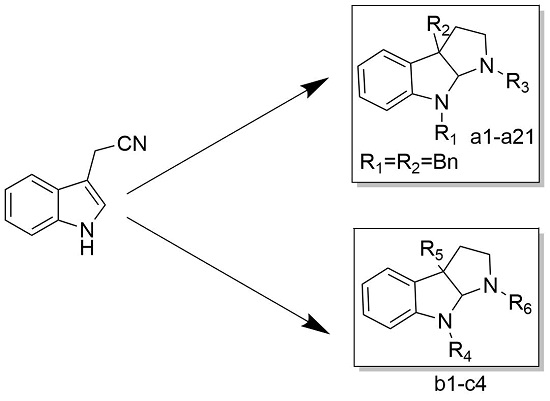
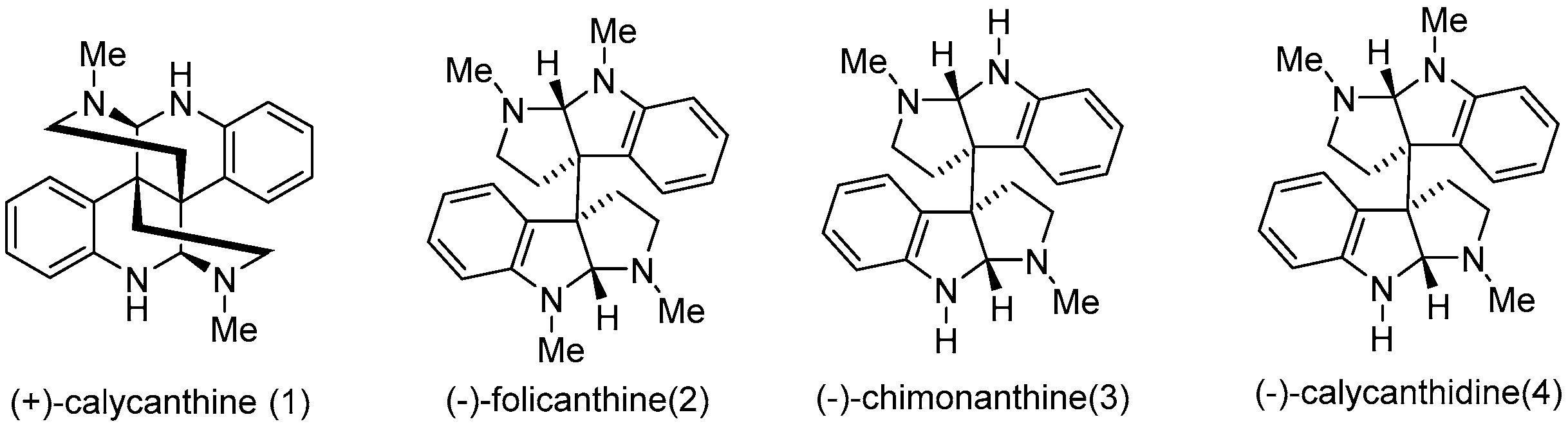
2.1. Design and Synthesis of Calycanthaceous Alkaloids Analogues
2.2. Antimicrobial Acitivity
3. Materials and Methods
3.1. Instruments and Chemicals
3.2. Synthesis
3.2.1. Synthesis of 2-(2-Oxoindolin-3-yl)acetonitrile (2)
3.2.2. Synthesis of 2-(1,3-Dibenzyl-2-oxoindolin-3-yl)acetonitrile (3)
3.2.3. Synthesis of 3a,8-Dibenzyl-1,2,3,3a,8,8a-hexahydropyrrolo[2,3-b]indole (4)
3.2.4. Synthesis of Compounds a1–a21
3.2.5. Synthesis of 2-(1-Ethyl-1H-indol-3-yl)acetonitrile (5)
3.2.6. Synthesis of 2-(1-Ethyl-2-oxoindolin-3-yl)acetonitrile (6)
3.2.7. Synthesis of 2-(1,3-Diethyl-2-oxoindolin-3-yl)acetonitrile (7)
3.2.8. Synthesis of 3a,8-Diethyl-1,2,3,3a,8,8a-hexahydropyrrolo[2,3-b]indole (8)
3.2.9. Synthesis of Compounds b1–c4
3.3. Biological Activity
4. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- He, D.; Jian, W.; Liu, X.; Shen, H.; Song, S. Synthesis, biological evaluation, and structure-activity relationship study of novel stilbene derivatives as potential fungicidal agents. J. Agric. Food Chem. 2015, 63, 1370–1377. [Google Scholar] [CrossRef] [PubMed]
- Lin, L.; Mulholland, N.; Wu, Q.Y.; Beattie, D.; Huang, S.W.; Irwin, D.; Clough, J.; Gu, Y.C.; Yang, G.F. Synthesis and antifungal activity of novel sclerotiorin analogues. J. Agric. Food Chem. 2012, 60, 4480–4491. [Google Scholar] [CrossRef] [PubMed]
- Newman, D.J.; Cragg, G.M. Natural products as sources of new drugs over the last 25 years. J. Nat. Prod. 2007, 70, 461–477. [Google Scholar] [CrossRef] [PubMed]
- Walter, M.W. Structure-based design of agrochemicals. J. Nat. Prod. 2002, 19, 278–291. [Google Scholar] [CrossRef]
- Xiang, L.; Zhao, K.; Chen, L. Molecular cloning and expression of Chimonanthus praecox farnesyl pyrophosphate synthase gene and its possible involvement in the biosynthesis of floral volatile sesquiterpenoids. Plant Physiol. Biochem. 2010, 48, 845–850. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.W.; Li, S.K.; Ji, Z.Q.; Hu, Z.N.; Wu, W.J. Synthesis and insecticidal activity of novel dimers of celangulin-V and podophyllotoxin. Chem. Nat. Compd. 2009, 45, 507–510. [Google Scholar]
- Kozomara, B.; Vinterhalter, B.; Radojevic, L.; Vinterhalter, D. In vitro propagation of Chimonanthus praecox (L.), a winter flowering ornamental shrub. In Vitro Cell Dev. Biol. Plant 2008, 44, 142–147. [Google Scholar] [CrossRef]
- Zhang, J.; Cui, L.; Li, L.; Hu, Z.; Zhang, Q.; Hu, Z.; Wu, W. Synthesis and insecticidal activities of novel nitrogenous derivatives of celangulin-V. Nat. Prod. Commun. 2014, 9, 745–748. [Google Scholar] [PubMed]
- Liu, Y.; Liu, Y.; Xiao, H.; Gao, R.; Tian, X. Synthesis and insecticidal activities of novel derivatives of podophyllotoxin: Part XII. Pestic. Biochem. Physiol. 2008, 91, 116–121. [Google Scholar] [CrossRef]
- Lv, J.S.; Zhang, L.L.; Chu, X.Z.; Zhou, J.F. Chemical composition, antioxidant and antimicrobial activity of the extracts of the flowers of the Chinese plant Chimonanthus praecox. Nat. Prod. Res. 2012, 26, 1363–1367. [Google Scholar] [CrossRef] [PubMed]
- Gui, R.Y.; Liang, W.W.; Yang, S.X.; Llu, L.; Qin, J.C. Chemical composition, antifungal activity and toxicity of essential oils from the leaves of Chimonanthus praecox located at two different geographical origins. Asian J. Chem. 2014, 26, 4445–4448. [Google Scholar]
- Zhang, J.W.; Gao, J.M.; Xu, T.; Zhang, X.C.; Ma, Y.T.; Jarussophon, S.; Konishi, Y. Antifungal activity of alkaloids from the seeds of Chimonanthus praecox. Chem. Biodivers. 2009, 6, 838–845. [Google Scholar] [CrossRef] [PubMed]
- Araki, T.; Manabe, Y.; Fujioka, K.; Yokoe, H.; Kanematsu, M.; Yoshida, M.; Shishido, K. Total syntheses of (±)-folicanthine and (±)-chimonanthine via a double intramolecular carbamoylketene-alkene [2+2] cycloaddition. Tetrahedron Lett. 2013, 54, 1012–1014. [Google Scholar] [CrossRef]
- Ruiz-Sanchis, P.; Savina, S.A.; Albericio, F.; Álvarez, M. Structure, bioactivity and synthesis of natural products with hexahydropyrrolo[2,3-b]indole. Chem. Eur. J. 2011, 17, 1388–1408. [Google Scholar] [CrossRef] [PubMed]
- Hino, T.; Yamada, S.I. Total synthesis of (±)-folicanthine. Tetrahedron Lett. 1963, 4, 1757–1760. [Google Scholar] [CrossRef]
- Hall, E.S.; McCapra, F.; Scott, A.I. Biogenetic-type synthesis of the calycanthaceous alkaloids. Tetrahedron 1967, 23, 4131–4141. [Google Scholar] [CrossRef]
- Fang, C.L.; Horne, S.; Taylor, N.; Rodrigo, R. Dimerization of a 3-substituted oxindole at c-3 and its application to the synthesis of (±)-folicanthine. J. Am. Chem. Soc. 1994, 116, 9480–9486. [Google Scholar] [CrossRef]
- Li, Y.X.; Wang, H.X.; Ali, S.; Xia, X.F.; Liang, Y.M. Iodine-mediated regioselective C2-amination of indoles and a concise total synthesis of (±)-folicanthine. Chem. Commun. 2012, 48, 2343–2345. [Google Scholar] [CrossRef] [PubMed]
- Peng, Y.; Luo, L.; Yan, C.S.; Zhang, J.J.; Wang, Y.W. Ni-catalyzed reductive homocoupling of unactivated alkyl bromides at room temperature and its synthetic application. J. Org. Chem. 2013, 78, 10960–10967. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.; Movassaghi, M. Biogenetically-inspired total synthesis of epidithiodiketopiperazines and related alkaloids. Acc. Chem. Res. 2015, 48, 1159–1171. [Google Scholar] [CrossRef] [PubMed]
- Xu, J.B.; Cheng, K.J. Studies on the alkaloids of the Calycanthaceae and their syntheses. Molecules 2015, 20, 6715–6738. [Google Scholar] [CrossRef] [PubMed]
- Movassaghi, M.; Schmidt, M.A. Concise Total synthesis of (−)-calycanthine, (+)-chimonanthine, and (+)-folicanthine. Angew. Chem. Int. Edit. 2007, 46, 3725–3728. [Google Scholar] [CrossRef] [PubMed]
- Verotta, L.; Orsini, F.; Sbacchi, M.; Scheildler, M.A.; Amador, T.A.; Elisabetsky, E. Synthesis and antinociceptive activity of chimonanthines and pyrrolidinoindoline-type alkaloids. Bioorg. Med. Chem. 2002, 10, 2133–2142. [Google Scholar] [CrossRef]
- Overman, L.E.; Paone, D.V.; Stearns, B.A. Direct Stereo- and Enantiocontrolled synthesis of vicinal stereogenic quaternary carbon centers. Total syntheses of meso- and (−)-Chimonanthine and (+)-Calycanthine. J. Am. Chem. Soc. 1999, 121, 7702–7703. [Google Scholar] [CrossRef]
- Overman, L.E.; Paone, D.V.; Stearns, B.A. ChemInform abstract: Direct stereo and enantiocontrolled synthesis of vicinal stereogenic quaternary carbon centers. Total syntheses of meso- and (−)-Chimonanthine and (+)-Calycanthine. Cheminform 2010, 30, 6905–6919. [Google Scholar] [CrossRef]
- Ma, M.F.; Feng, J.L.; Li, R.X.; Chen, S.W. Synthesis of Hexahypyrrole[2,3-b]indole Derivatives. CN 104250254 A, 31 December 2014. [Google Scholar]
- Rivera-Becerril, E.; Joseph-Nathan, P.; Pérez-Álvarez, V.M.; Morales-Ríos, M.S. Synthesis and biological evaluation of (−)- and (+)-Debromoflustramine B and its analogues as selective butyrylcholinesterase inhibitors. J. Med. Chem. 2008, 51, 5271–5284. [Google Scholar] [CrossRef] [PubMed]
- Kawasaki, T.; Ogawa, A.; Terashima, R.; Saheki, T.; Ban, N.; Sekiguchi, H.; Sakaguchi, K.; Sakamoto, M. Synthesis of diversely functionalized Hexahydropyrrolo[2,3-b]indoles using domino reactions, Olefination, isomerization and Claisen rearrangement followed by reductive cyclization. J. Org. Chem. 2005, 70, 2957–2966. [Google Scholar] [CrossRef] [PubMed]
- Feng, N.; Ye, W.; Wu, P.; Huang, Y.; Xie, H.; Wei, X. Two New Antifungal Alkaloids Produced by Streptoverticillium morookaense. J. Antibiot. 2007, 60, 179–183. [Google Scholar] [CrossRef] [PubMed]
- Zhang, C.; Ondeyka, J.; Guan, Z.; Dietrich, L.; Burgess, B.; Wang, J.; Singh, S.B. Isolation, structure an biological activities of platensimycin B4 from Streptomyces platensis. J. Antibiot. 2009, 62, 699–702. [Google Scholar] [CrossRef] [PubMed]
- Sample Availability: Samples of the compounds are available from the authors.

| Compounds | MIC μg/mL | MBC μg/mL | ||||
|---|---|---|---|---|---|---|
| B. cereus | S. aureus | S. epidermidis | B. cereus | S. aureus | S. epidermidis | |
| a1 | − | 256 | 62.5 | − | − | 125 |
| a2 | − | 256 | 62.5 | − | − | 125 |
| a3 | 125 | − | − | 256 | − | − |
| a4 | − | − | 62.5 | − | − | 125 |
| a5 | − | − | − | − | − | − |
| a6 | 256 | − | − | − | − | − |
| a7 | 31.25 | 256 | 62.5 | 62.5 | − | 125 |
| a8 | 62.5 | 256 | 125 | 125 | − | 256 |
| a9 | 125 | − | − | 256 | − | − |
| a10 | 125 | − | − | 256 | − | − |
| a11 | 31.25 | 125 | 62.5 | 62.5 | 256 | 125 |
| a12 | 15.63 | − | − | 31.25 | − | − |
| a13 | − | − | − | − | − | − |
| a14 | 256 | − | − | − | − | − |
| a15 | 256 | − | − | − | − | − |
| a16 | 62.5 | − | − | 125 | − | − |
| a17 | − | − | − | − | − | − |
| a18 | − | − | − | − | − | − |
| a19 | − | 256 | 62.5 | − | − | 125 |
| a20 | 125 | 256 | 125 | 256 | − | 256 |
| a21 | − | − | − | − | − | − |
| b1 | 62.5 | 125 | 125 | 125 | 256 | 256 |
| b2 | 31.25 | 125 | 125 | 62.5 | 256 | 256 |
| b3 | 7.81 | 125 | 125 | 15.63 | 256 | 256 |
| b4 | 31.25 | 256 | 125 | 62.5 | − | 256 |
| c1 | 62.5 | 125 | 62.5 | 125 | 256 | 125 |
| c2 | 125 | 125 | 62.5 | 256 | 256 | 125 |
| c3 | 62.5 | 125 | 125 | 125 | 256 | 256 |
| c4 | 62.5 | 31.25 | 31.25 | 125 | 62.5 | 62.5 |
| gentamicin | − | 62.5 | 15.63 | − | 125 | 31.25 |
| streptomycin | 1.96 | 15.63 | 31.25 | 3.9 | 31.25 | 62.5 |
| Compounds | MIC μg/mL | MBC μg/mL | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| E.c. | S.t. | S.f. | E.s. | P.a. | R.s. | E.c. | S.t. | S.f. | E.s. | P.a. | R.s. | |
| a1 | 125 | 31.25 | 62.5 | 62.5 | 256 | 62.5 | 256 | 62.5 | 125 | 125 | − | 125 |
| a2 | 125 | 125 | 125 | 125 | 125 | − | 256 | 256 | 256 | 256 | 250 | − |
| a3 | − | − | − | − | 125 | 125 | − | − | − | − | 250 | 256 |
| a4 | − | 125 | 125 | − | 125 | 7.81 | − | 256 | 256 | − | 250 | 15.63 |
| a5 | − | − | − | − | 125 | 125 | − | − | − | − | 250 | 256 |
| a6 | − | − | − | − | 62.5 | 256 | − | − | − | − | 125 | − |
| a7 | 62.5 | 125 | − | 125 | 62.5 | 7.81 | 125 | 256 | − | 256 | 125 | 15.63 |
| a8 | 62.5 | 125 | 125 | 125 | 62.5 | 31.25 | 125 | 256 | 256 | 256 | 125 | 62.5 |
| a9 | − | − | − | − | 125 | 15.63 | − | − | − | − | 250 | 31.25 |
| a10 | − | − | − | − | 62.5 | 125 | − | − | − | − | 125 | 256 |
| a11 | 62.5 | 125 | 62.5 | 125 | 62.5 | 15.63 | 125 | 256 | 125 | 256 | 125 | 31.25 |
| a12 | − | 125 | 125 | − | 125 | 7.81 | − | 256 | 256 | − | 250 | 15.63 |
| a13 | − | 125 | 125 | − | − | − | − | 256 | 256 | − | − | − |
| a14 | − | 125 | 125 | − | 62.5 | 256 | − | 256 | 256 | − | 125 | − |
| a15 | − | 125 | 125 | − | 125 | 256 | − | 256 | 256 | − | 250 | − |
| a16 | − | 125 | 256 | − | 62.5 | 125 | − | 256 | − | − | 125 | 256 |
| a17 | − | 62.5 | 62.5 | − | 62.5 | 62.5 | − | 125 | 125 | − | 125 | 125 |
| a18 | − | − | − | − | 7.81 | 125 | − | − | − | − | 15.63 | 256 |
| a19 | 62.5 | 125 | 62.5 | 125 | 15.63 | 256 | 125 | 256 | 125 | 256 | 31.25 | − |
| a20 | 62.5 | 125 | − | 125 | 31.25 | 62.5 | 125 | 256 | − | 256 | 62.5 | 125 |
| a21 | − | 125 | 62.5 | − | 125 | − | − | 256 | 125 | − | 250 | − |
| b1 | 62.5 | 125 | 62.5 | − | 125 | 256 | 125 | 256 | 125 | − | 250 | − |
| b2 | 62.5 | 125 | 125 | 125 | 125 | − | 125 | 256 | 256 | 256 | 250 | − |
| b3 | 62.5 | 125 | 62.5 | 62.5 | 62.5 | 125 | 125 | 256 | 125 | 125 | 125 | 265 |
| b4 | 62.5 | − | − | 62.5 | 7.81 | 125 | 125 | − | − | 125 | 15.63 | 256 |
| c1 | 125 | 125 | − | 125 | 31.25 | 125 | 256 | 256 | − | 256 | 62.5 | 256 |
| c2 | 125 | 125 | − | 125 | 15.63 | 125 | 256 | 256 | − | 256 | 31.25 | 256 |
| c3 | 125 | 62.5 | − | 62.5 | 15.63 | 256 | 256 | 125 | − | 125 | 31.25 | − |
| c4 | 31.25 | 62.5 | 31.25 | 16.5 | 62.5 | 1.96 | 62.5 | 125 | 62.5 | 31.25 | 125 | 3.9 |
| gentamicin | 125 | 125 | 256 | 256 | − | − | 256 | 256 | − | − | − | − |
| streptomycin | 125 | 125 | 62.5 | 7.81 | 1.96 | 3.9 | 256 | 256 | 125 | 15.63 | 3.9 | 7.81 |
| Compounds | MIC(µg/mL) | MFC(µg/mL) | ||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| P.ca. | V.d. | F.s. | C.o. | P.c. | C.j. | A.sf. | A.s. | C.l. | F.o. | A.n. | P.ca. | V.d. | F.s. | C.o. | P.c. | C.j. | A.sf. | A.s. | C.l. | F.o. | A.n. | |
| a1 | 31.25 | − | 256 | 125 | − | 125 | 256 | 256 | − | − | 62.5 | 62.5 | − | − | 256 | − | 256 | − | − | − | − | 125 |
| a2 | 256 | − | 256 | 125 | − | 62.5 | 256 | − | − | 256 | − | − | − | − | 256 | − | 125 | − | − | − | − | − |
| a3 | 256 | − | − | − | − | − | 256 | 256 | 62.5 | 125 | 256 | − | − | − | − | − | − | − | − | 125 | 256 | − |
| a4 | − | − | − | − | − | − | 256 | 256 | 62.5 | 15.63 | 256 | − | − | − | − | − | − | − | − | 125 | 31.25 | − |
| a5 | − | − | 256 | − | − | − | − | − | − | − | 256 | − | − | − | − | − | − | − | − | − | − | − |
| a6 | − | − | 256 | − | − | − | − | − | − | 125 | 125 | − | − | − | − | − | − | − | − | − | 256 | 256 |
| a7 | − | 256 | 256 | − | − | 125 | − | 256 | 125 | 31.25 | 256 | − | − | − | − | − | 256 | − | − | 256 | 62.5 | − |
| a8 | − | 256 | − | − | − | 256 | − | 125 | 125 | 62.5 | 125 | − | − | − | − | − | − | − | 256 | 256 | 125 | 256 |
| a9 | − | − | − | − | − | − | − | 256 | − | 256 | 256 | − | − | − | − | − | − | − | − | − | − | − |
| a10 | − | − | − | − | − | 125 | − | − | − | 125 | 256 | − | − | − | − | − | 256 | − | − | − | 256 | − |
| a11 | − | 125 | − | − | − | 256 | − | − | − | 125 | 256 | − | 256 | − | − | − | − | − | − | − | 256 | − |
| a12 | − | − | − | 256 | − | − | − | 256 | − | 62.5 | 125 | − | − | − | − | − | − | − | − | − | 125 | 256 |
| a13 | − | − | − | − | − | − | − | − | − | 256 | 125 | − | − | − | − | − | − | − | − | − | − | 256 |
| a14 | − | − | − | − | − | − | − | − | − | 125 | 256 | − | − | − | − | − | − | − | − | − | 256 | − |
| a15 | − | − | 256 | − | − | − | − | − | − | 62.5 | 256 | − | − | − | − | − | − | − | − | − | 125 | − |
| a16 | − | − | 256 | − | − | 256 | − | − | − | 125 | 125 | − | − | − | − | − | − | − | − | − | 256 | 256 |
| a17 | 256 | − | 125 | 256 | − | − | − | − | − | 256 | 125 | − | − | 256 | − | − | − | − | − | − | − | 256 |
| a18 | 256 | − | 256 | 256 | − | 256 | − | − | 125 | 125 | 62.5 | − | − | − | − | − | − | − | − | 256 | 256 | 125 |
| a19 | 256 | − | 256 | 256 | − | − | − | − | − | 125 | 256 | − | − | − | − | − | − | − | − | − | 256 | − |
| a20 | − | − | 125 | 256 | − | − | − | − | − | 125 | 62.5 | − | − | 256 | − | − | − | − | − | − | 256 | 125 |
| a21 | − | − | − | − | − | − | − | − | − | 125 | 125 | − | − | − | − | − | − | − | − | − | 256 | 256 |
| b1 | 125 | − | 256 | 256 | 125 | − | 256 | − | − | 125 | 125 | 256 | − | − | − | 256 | − | − | − | − | 256 | 256 |
| b2 | 62.5 | 125 | 256 | 125 | 125 | 125 | 256 | 125 | 125 | 256 | 62.5 | 125 | 256 | − | 256 | 256 | 256 | − | 256 | 256 | − | 125 |
| b3 | 125 | 125 | − | 256 | 125 | − | 256 | − | 125 | 125 | 62.5 | 256 | 256 | − | − | 256 | − | − | − | 256 | 256 | 125 |
| b4 | 125 | − | 256 | 256 | − | − | − | − | − | 125 | 62.5 | 256 | − | − | − | − | − | − | − | − | 256 | 125 |
| c1 | 62.5 | − | − | 125 | 125 | 256 | − | − | 125 | 62.5 | 125 | 125 | − | − | 256 | 256 | − | − | − | 125 | 256 | |
| c2 | 31.25 | − | − | 125 | 125 | − | 62.5 | 256 | 125 | 31.25 | 31.25 | 62.5 | − | − | 256 | 256 | − | 125 | − | 256 | 62.5 | 62.5 |
| c3 | 256 | − | 256 | 256 | 125 | − | 125 | − | − | 256 | 125 | − | − | − | − | 256 | − | 256 | − | − | − | 256 |
| c4 | 31.25 | 62.5 | 256 | 62.5 | 62.5 | − | 15.63 | − | − | 31.25 | 125 | 62.5 | 125 | − | 125 | 125 | − | 31.25 | − | − | 62.5 | 256 |
| A. | 3.9 | 62.5 | 256 | 125 | 15.63 | 256 | 7.81 | − | 7.81 | 125 | 0.97 | 7.81 | 125 | − | 256 | 31.25 | − | 15.63 | − | 15.63 | 256 | 1.93 |
| C. | 1.95 | 256 | 125 | 0.97 | 0.97 | 62.5 | 0.97 | − | − | 7.81 | − | 3.9 | − | 256 | 1.95 | 1.95 | 125 | 1.95 | − | − | 15.63 | − |
© 2016 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zheng, S.; Zhou, X.; Xu, S.; Zhu, R.; Bai, H.; Zhang, J. Synthesis and Antimicrobial Characterization of Half-Calycanthaceous Alkaloid Derivatives. Molecules 2016, 21, 1207. https://doi.org/10.3390/molecules21091207
Zheng S, Zhou X, Xu S, Zhu R, Bai H, Zhang J. Synthesis and Antimicrobial Characterization of Half-Calycanthaceous Alkaloid Derivatives. Molecules. 2016; 21(9):1207. https://doi.org/10.3390/molecules21091207
Chicago/Turabian StyleZheng, Shaojun, Xinping Zhou, Shixun Xu, Rui Zhu, Hongjin Bai, and Jiwen Zhang. 2016. "Synthesis and Antimicrobial Characterization of Half-Calycanthaceous Alkaloid Derivatives" Molecules 21, no. 9: 1207. https://doi.org/10.3390/molecules21091207
APA StyleZheng, S., Zhou, X., Xu, S., Zhu, R., Bai, H., & Zhang, J. (2016). Synthesis and Antimicrobial Characterization of Half-Calycanthaceous Alkaloid Derivatives. Molecules, 21(9), 1207. https://doi.org/10.3390/molecules21091207