Immobilization of α-Amylase from Anoxybacillus sp. SK3-4 on ReliZyme and Immobead Supports
Abstract
:1. Introduction
2. Results
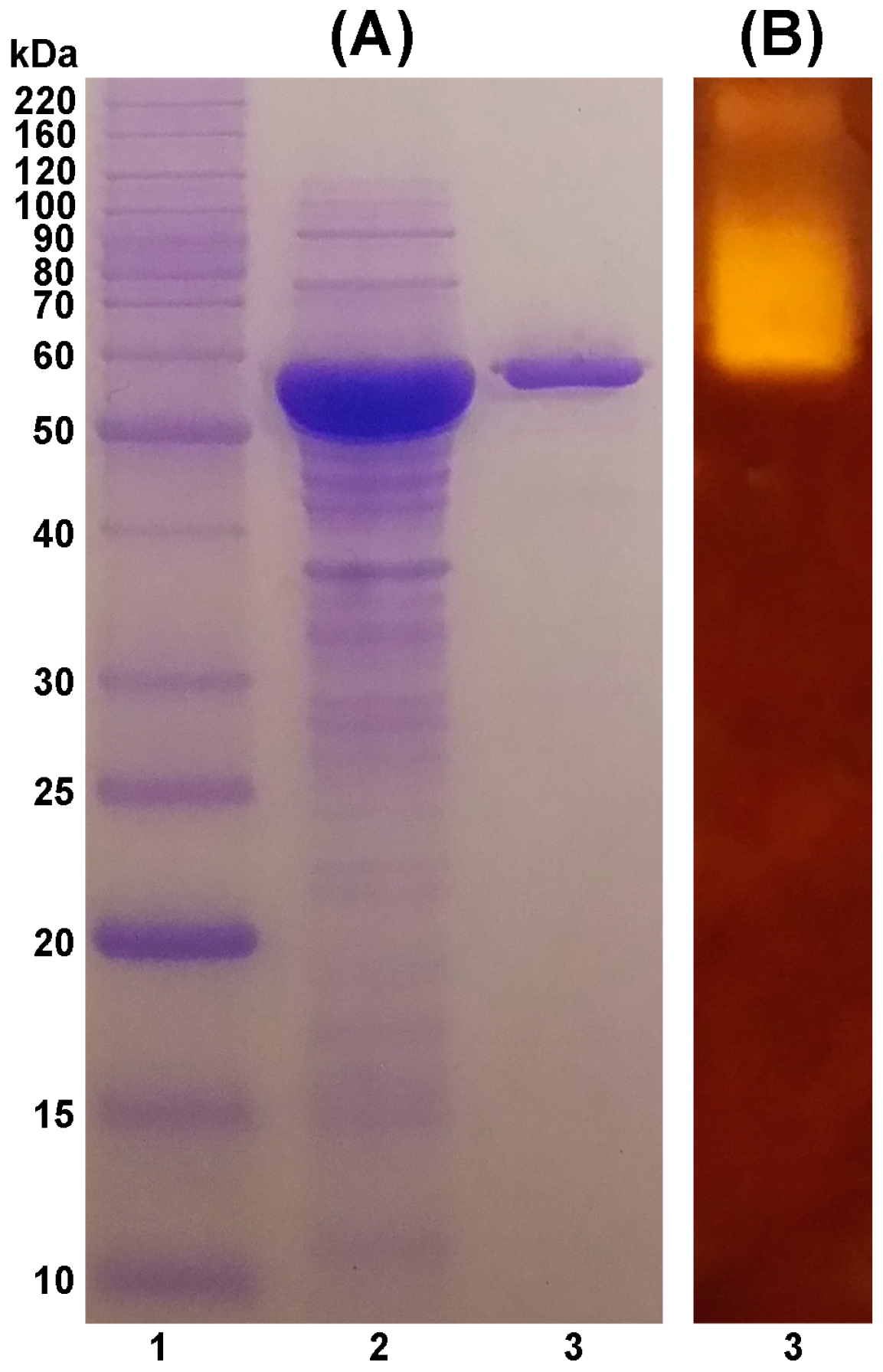
2.1. Purification of TASKA
2.2. Optimization of the Immobilization Conditions
2.2.1. Optimum pH and Temperature
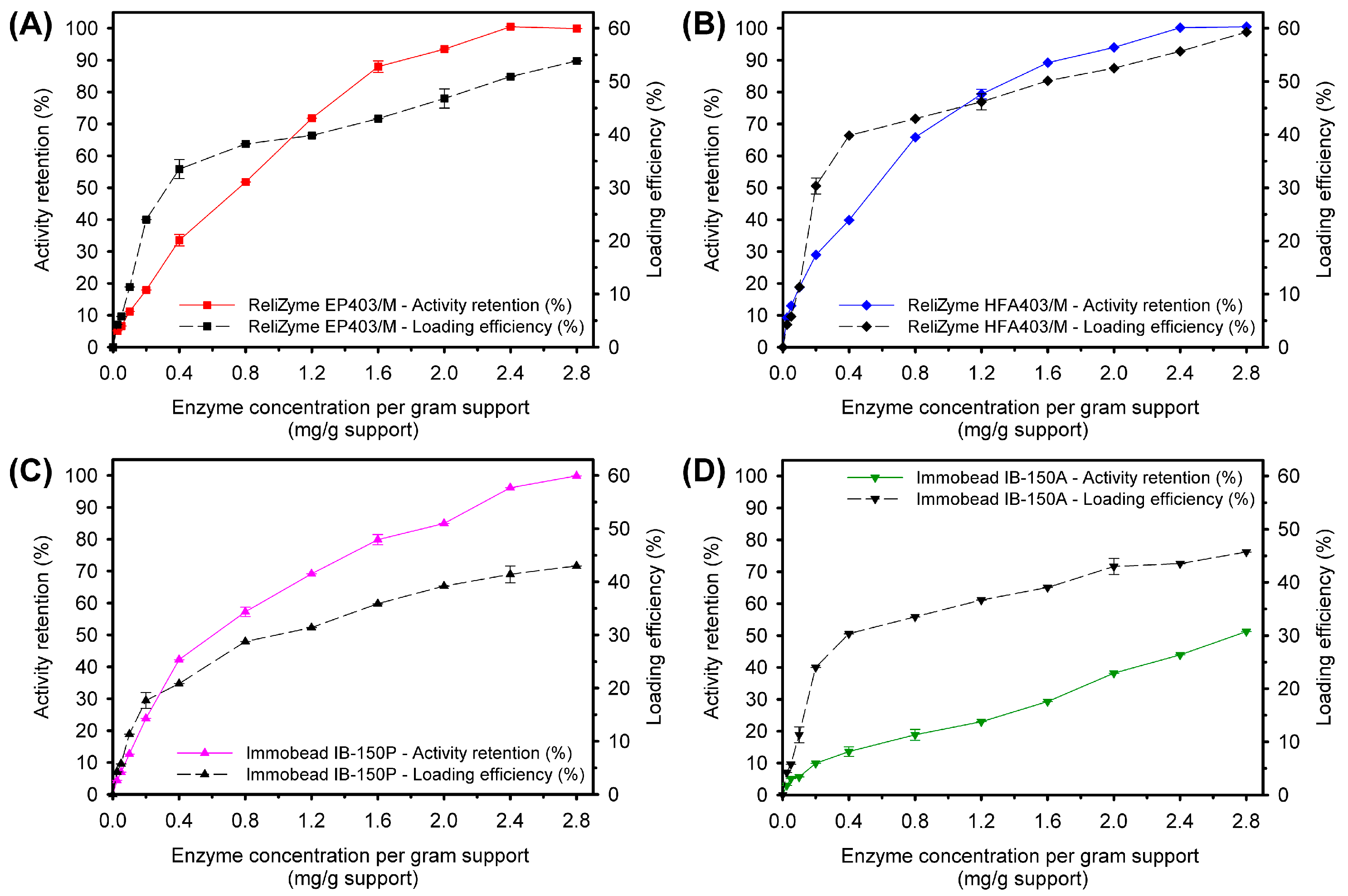
2.2.2. Protein-Loading Efficiency
2.2.3. Activity Retention and Recovery
2.3. Characterization of Free and Immobilized TASKA
2.3.1. Biochemical Characterization
2.3.2. Analysis of Reaction Products
3. Discussion
3.1. Properties of ReliZyme and Immobead Supports
3.2. Optimization of Immobilization Conditions
3.3. Characterization of Free and Immobilized TASKA
4. Materials and Methods
4.1. Materials
4.2. Expression of TASKA
4.3. Purification of TASKA
4.4. Enzyme Activity and Protein-Concentration Assays
4.5. TASKA Immobilization on Different Supports
4.5.1. Immobilization Method
4.5.2. Optimization of Immobilization Conditions
Optimum pH and Temperature
Loading Efficiency
Activity Retention and Recovery
4.6. Characterization of Free and Immobilized TASKA
4.6.1. Biochemical Characterization
4.6.2. Analysis of Reaction Products
4.7. Statistical Analysis
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
Abbreviations
| ASKA | α-Amylase from Anoxybacillus sp. SK3-4 |
| BCA | Bicinchoninic acid |
| BSA | Bovine serum albumin |
| DNS | 3,5-Dinitrosalicylic acid |
| G1 | Glucose |
| G2 | Maltose |
| G3 | Maltotriose |
| G4 | Maltotetraose |
| G5 | Maltopentaose |
| G6 | Maltohexaose |
| G7 | Maltoheptaose |
| GH13 | Glycoside hydrolase 13 |
| HPLC-ELSD | High performance liquid chromatography with an evaporative light scattering detector |
| LB | Luria-Bertani |
| MWCO | Molecular weight cut-off |
| Ni-NTA | Nickel-nitrilotriacetic |
| SDS-PAGE | Sodium dodecyl sulfate-polyacrylamide gel electrophoresis |
| SLM | Standard liters per min |
| TASKA | Truncated α-amylase from Anoxybacillus sp. SK3-4 |
References
- Goh, K.M.; Kahar, U.M.; Chai, Y.Y.; Chong, C.S.; Chai, K.P.; Ranjani, V.; Illias, R.M.; Chan, K.-G. Recent discoveries and applications of Anoxybacillus. Appl. Microbiol. Biotechnol. 2013, 97, 1475–1488. [Google Scholar] [CrossRef] [PubMed]
- Urbieta, M.S.; Donati, E.R.; Chan, K.-G.; Shahar, S.; Sin, L.L.; Goh, K.M. Thermophiles in the genomic era: Biodiversity, science, and applications. Biotechnol. Adv. 2015, 33, 633–647. [Google Scholar] [CrossRef] [PubMed]
- Belduz, A.O.; Canakci, S.; Chan, K.-G.; Kahar, U.M.; Chan, C.S.; Yaakop, A.S.; Goh, K.M. Genome sequence of Anoxybacillus ayderensis AB04T isolated from the Ayder hot spring in Turkey. Stand. Genomic. Sci. 2015, 10, 70. [Google Scholar] [CrossRef] [PubMed]
- Lim, Y.L.; Chan, K.-G.; Ee, R.; Belduz, A.O.; Canakci, S.; Kahar, U.M.; Yaakop, A.S.; Goh, K.M. Complete genome of the potential thermozyme producer Anoxybacillus gonensis G2T isolated from the Gönen hot springs in Turkey. J. Biotechnol. 2015, 212, 65–66. [Google Scholar] [CrossRef] [PubMed]
- Chai, Y.Y.; Kahar, U.M.; Salleh, M.M.; Illias, R.M.; Goh, K.M. Isolation and characterization of pullulan-degrading Anoxybacillus species isolated from Malaysian hot springs. Environ. Technol. 2012, 33, 1231–1238. [Google Scholar] [CrossRef] [PubMed]
- Chan, C.S.; Chan, K.-G.; Tay, Y.-L.; Chua, Y.-H.; Goh, K.M. Diversity of thermophiles in a Malaysian hot spring determined using 16S rRNA and shotgun metagenome sequencing. Front. Microbiol. 2015, 6, 177. [Google Scholar] [CrossRef] [PubMed]
- Goh, K.M.; Gan, H.M.; Chan, K.-G.; Chan, G.F.; Shahar, S.; Chong, C.S.; Kahar, U.M.; Chai, K.P. Analysis of Anoxybacillus genomes from the aspects of lifestyle adaptations, prophage diversity, and carbohydrate metabolism. PLoS ONE 2014, 9, e90549. [Google Scholar] [CrossRef] [PubMed]
- Chai, K.P.; Othman, N.F.B.; Teh, A.-H.; Ho, K.L.; Chan, K.-G.; Shamsir, M.S.; Goh, K.M.; Ng, C.L. Crystal structure of Anoxybacillus α-amylase provides insights into maltose binding of a new glycosyl hydrolase subclass. Sci. Rep. 2016, 6, 23126. [Google Scholar] [CrossRef] [PubMed]
- Chai, Y.Y.; Rahman, R.N.Z.R.A.; Illias, R.M.; Goh, K.M. Cloning and characterisation of two new thermostable and alkalitolerant α-amylases from the Anoxybacillus species that produce high levels of maltose. J. Ind. Microbiol. Biotechnol. 2012, 39, 731–741. [Google Scholar] [CrossRef] [PubMed]
- Ranjani, V.; Janeček, Š.; Chai, K.P.; Shahir, S.; Rahman, R.N.Z.R.A.; Chan, K.-G.; Goh, K.M. Protein engineering of selected residues from conserved sequence regions of a novel Anoxybacillus α-amylase. Sci. Rep. 2014, 4, 5850. [Google Scholar] [CrossRef] [PubMed]
- Kahar, U.M.; Chan, K.-G.; Salleh, M.M.; Hii, S.M.; Goh, K.M. A high molecular-mass Anoxybacillus sp. SK3-4 amylopullulanase: Characterization and its relationship in carbohydrate utilization. Int. J. Mol. Sci. 2013, 14, 11302–11318. [Google Scholar] [CrossRef] [PubMed]
- Kahar, U.M.; Ng, C.L.; Chan, K.-G.; Goh, K.M. Characterization of a type I pullulanase from Anoxybacillus sp. SK3-4 reveals an unusual substrate hydrolysis. Appl. Microbiol. Biotechnol. 2016, 100, 6291–6307. [Google Scholar] [CrossRef] [PubMed]
- Kahar, U.M.; Salleh, M.M.; Goh, K.M. Medium optimisation for pullulanase production from Anoxybacillus species using experimental design. Indian J. Biotechnol. 2014, 13, 89–97. [Google Scholar]
- Muniandy, K.; Kahar, U.M.; Chong, C.S.; Chai, Y.Y.; Goh, P.H.; Goh, K.M. Application of statistical experimental design for optimization of novel α-amylase production by Anoxybacillus species. J. Biol. Sci. 2013, 13, 605–613. [Google Scholar]
- Bertoldo, C.; Antranikian, G. Starch-hydrolyzing enzymes from thermophilic archaea and bacteria. Curr. Opin. Chem. Biol. 2002, 6, 151–160. [Google Scholar] [CrossRef]
- Synowiecki, J.; Grzybowska, B.; Zdziebło, A. Sources, properties and suitability of new thermostable enzymes in food processing. Crit. Rev. Food Sci. Nutr. 2006, 46, 197–205. [Google Scholar] [CrossRef] [PubMed]
- Janeček, Š.; Kuchtová, A.; Petrovičová, S. A novel GH13 subfamily of α-amylases with a pair of tryptophans in the helix α3 of the catalytic TIM-barrel, the LPDlx signature in the conserved sequence region V and a conserved aromatic motif at the C-terminus. Biologia 2015, 70, 1284–1294. [Google Scholar] [CrossRef]
- Van der Maarel, M.J.E.C. Starch-processing enzymes. In Enzymes in Food Technology, 2nd ed.; Whitehurst, R.J., Oort, M.V., Eds.; Wiley-Blackwell Publishing Ltd.: Hoboken, NJ, USA, 2009; pp. 322–324. [Google Scholar]
- McAuliffe, J.C. Industrial enzymes and biocatalysis. In Handbook of Industrial Chemistry and Biotechnology, 12th ed.; Kent, J.A., Ed.; Springer: London, UK, 2012; Volumes 1 and 2, pp. 1183–1227. [Google Scholar]
- Mateo, C.; Grazú, V.; Pessela, B.C.C.; Montes, T.; Palomo, J.M.; Torres, R.; López-Gallego, F.; Fernández-Lafuente, R.; Guisán, J.M. Advances in the design of new epoxy supports for enzyme immobilization–stabilization. Biochem. Soc. Trans. 2007, 35, 1593–1601. [Google Scholar] [CrossRef] [PubMed]
- Brady, D.; Jordaan, J. Advances in enzyme immobilisation. Biotechnol. Lett. 2009, 31, 1639–1650. [Google Scholar] [CrossRef] [PubMed]
- Singh, R.K.; Tiwari, M.K.; Singh, R.; Lee, J.-K. From protein engineering to immobilization: Promising strategies for the upgrade of industrial enzymes. Int. J. Mol. Sci. 2013, 14, 1232–1277. [Google Scholar] [CrossRef] [PubMed]
- Eş, I.; Vieira, J.D.G.; Amaral, A.C. Principles, techniques, and applications of biocatalyst immobilization for industrial application. Appl. Microbiol. Biotechnol. 2015, 99, 2065–2082. [Google Scholar] [CrossRef] [PubMed]
- Rodrigues, R.C.; Ortiz, C.; Berenguer-Murcia, Á.; Torres, R.; Fernández-Lafuente, R. Modifying enzyme activity and selectivity by immobilization. Chem. Soc. Rev. 2013, 42, 6290–6307. [Google Scholar] [CrossRef] [PubMed]
- Hernandez, K.; Fernández-Lafuente, R. Control of protein immobilization: Coupling immobilization and site-directed mutagenesis to improve biocatalyst or biosensor performance. Enzyme Microb. Technol. 2011, 48, 107–122. [Google Scholar] [CrossRef] [PubMed]
- Vaghari, H.; Jafarizadeh-Malmiri, H.; Mohammadlou, M.; Berenjian, A.; Anarjan, N.; Jafari, N.; Nasiri, S. Application of magnetic nanoparticles in smart enzyme immobilization. Biotechnol. Lett. 2016, 38, 223–233. [Google Scholar] [CrossRef] [PubMed]
- Sheldon, R.A.; Pelt, S.v. Enzyme immobilisation in biocatalysis: Why, what and how. Chem. Soc. Rev. 2013, 42, 6223–6235. [Google Scholar] [CrossRef] [PubMed]
- Secundo, F. Conformational changes of enzymes upon immobilisation. Chem. Soc. Rev. 2013, 42, 6250–6261. [Google Scholar] [CrossRef] [PubMed]
- Hwang, E.T.; Gu, M.B. Enzyme stabilization by nano/microsized hybrid materials. Eng. Life Sci. 2013, 13, 49–61. [Google Scholar] [CrossRef]
- Garcia-Galan, C.; Berenguer-Murcia, Á.; Fernández-Lafuente, R.; Rodrigues, R.C. Potential of different enzyme immobilization strategies to improve enzyme performance. Adv. Synth. Catal. 2011, 353, 2885–2904. [Google Scholar] [CrossRef]
- Fernández-Lafuente, R. Stabilization of multimeric enzymes: Strategies to prevent subunit dissociation. Enzyme Microb. Technol. 2009, 45, 405–418. [Google Scholar] [CrossRef]
- Min, K.; Yoo, Y.J. Recent progress in nanobiocatalysis for enzyme immobilization and its application. Biotechnol. Bioprocess Eng. 2014, 19, 553–567. [Google Scholar] [CrossRef]
- Mateo, C.; Palomo, J.; Fernández-Lorente, G.; Guisán, J.M.; Fernández-Lafuente, R. Improvement of enzyme activity, stability and selectivity via immobilization techniques. Enzyme Microb. Technol. 2007, 40, 1451–1463. [Google Scholar] [CrossRef]
- Hormigo, D.; de la Mata, I.; Acebal, C.; Arroyo, M. Immobilized aculeacin A acylase from Actinoplanes utahensis: Characterization of a novel biocatalyst. Bioresour. Technol. 2010, 101, 4261–4268. [Google Scholar] [CrossRef] [PubMed]
- Ghazi, I.; Segura, A.G.D.; Fernández-Arrojo, L.; Alcalde, M.; Yates, M.; Rojas-Cervantes, M.L.; Plou, F.J.; Ballesteros, A. Immobilisation of fructosyltransferase from Aspergillus aculeatus on epoxy-activated Sepabeads EC for the synthesis of fructo-oligosaccharides. J. Mol. Catal. B: Enzym. 2005, 35, 19–27. [Google Scholar] [CrossRef]
- Bayraktar, H.; Serilmez, M.; Karkaş, T.; Çelem, E.B.; Önal, S. Immobilization and stabilization of α-galactosidase on Sepabeads EC-EA and EC-HA. Int. J. Biol. Macromol. 2011, 49, 855–860. [Google Scholar] [CrossRef] [PubMed]
- Torres, P.; Batista-Viera, F. Immobilization of β-galactosidase from Bacillus circulans onto epoxy-activated acrylic supports. J. Mol. Catal. B Enzym. 2012, 74, 230–235. [Google Scholar] [CrossRef]
- Torres, R.; Mateo, C.; Fernández-Lorente, G.; Ortiz, C.; Fuentes, M.; Palomo, J.M.; Guisan, J.M.; Fernández-Lafuente, R. A novel heterofunctional epoxy-amino sepabeads for a new enzyme immobilization protocol: Immobilization-stabilization of α-galactosidase from Aspergillus oryzae. Biotechnol. Prog. 2003, 19, 1056–1060. [Google Scholar] [CrossRef] [PubMed]
- Esposito, M.; Pierro, P.D.; Dejonghe, W.; Mariniello, L.; Porta, R. Enzymatic milk clotting activity in artichoke (Cynara scolymus) leaves and alpine thistle (Carduus defloratus) flowers. Immobilization of alpine thistle aspartic protease. Food Chem. 2016, 204, 115–121. [Google Scholar] [CrossRef] [PubMed]
- Bonet-Ragel, K.; Canet, A.; Benaiges, M.D.; Valero, F. Synthesis of biodiesel from high FFA alperujo oil catalysed by immobilised lipase. Fuel 2015, 161, 12–17. [Google Scholar] [CrossRef]
- Çelem, E.B.; Önal, S. Immobilization of phytase on epoxy-activated Sepabead EC-EP for the hydrolysis of soymilk phytate. J. Mol. Catal. B Enzym. 2009, 61, 150–156. [Google Scholar] [CrossRef]
- Mateo, C.; Abian, O.; Fernández-Lorente, G.; Pedroche, J.; Fernández-Lafuente, R.; Guisan, J.M.; Tam, A.; Daminati, M. Epoxy sepabeads: A novel epoxy support for stabilization of industrial enzymes via very intense multipoint covalent attachment. Biotechnol. Prog. 2002, 18, 629–634. [Google Scholar] [CrossRef] [PubMed]
- Balcão, V.M.; Vila, M.M.D.C. Structural and functional stabilization of protein entities: State-of-the-art. Adv. Drug Deliv. Rev. 2015, 93, 25–41. [Google Scholar] [CrossRef] [PubMed]
- Turková, J.; Bláha, K.; Malaníková, M.; Vancurová, D.; Svec, F.; Kálal, J. Methacrylate gels with epoxide groups as supports for immobilization of enzymes in pH range 3–12. Biochim. Biophys. Acta. 1978, 524, 162–169. [Google Scholar] [CrossRef]
- Guzik, U.; Hupert-Kocurek, K.; Wojcieszyńska, D. Immobilization as a strategy for improving enzyme properties-application to oxidoreductases. Molecules 2014, 19, 8995–9018. [Google Scholar] [CrossRef] [PubMed]
- Stepankova, V.; Bidmanova, S.; Koudelakova, T.; Prokop, Z.; Chaloupkova, R.; Damborsky, J. Strategies for stabilization of enzymes in organic solvents. ACS Catal. 2013, 3, 2823–2836. [Google Scholar] [CrossRef]
- Barbosa, O.; Ortiz, C.; Berenguer-Murcia, Á.; Torres, R.; Rodrigues, R.C.; Fernandez-Lafuente, R. Strategies for the one-step immobilization-purification of enzymes as industrial biocatalysts. Biotechnol. Adv. 2015, 33, 435–456. [Google Scholar] [CrossRef] [PubMed]
- DiCosimo, R.; McAuliffe, J.; Poulose, A.J.; Bohlmann, G. Industrial use of immobilized enzymes. Chem. Soc. Rev. 2013, 42, 6437–6474. [Google Scholar] [CrossRef] [PubMed]
- Liese, A.; Hilterhaus, L. Evaluation of immobilized enzymes for industrial applications. Chem. Soc. Rev. 2013, 42, 6236–6249. [Google Scholar] [CrossRef] [PubMed]
- Cantone, S.; Ferrario, V.; Corici, L.; Ebert, C.; Fattor, D.; Spizzoa, P.; Gardossi, L. Efficient immobilisation of industrial biocatalysts: Criteria and constraints for the selection of organic polymeric carriers and immobilisation methods. Chem. Soc. Rev. 2013, 42, 6262–6276. [Google Scholar] [CrossRef] [PubMed]
- Contesini, F.J.; de Alencar Figueira, J.; Kawaguti, H.Y.; de Barros Fernandes, P.C.; de Oliveira Carvalho, P.; da Graça Nascimento, M.; Sato, H.H. Potential applications of carbohydrases immobilization in the food industry. Int. J. Mol. Sci. 2013, 14, 1335–1369. [Google Scholar] [CrossRef] [PubMed]
- Resolution ResAP (2004) 3 on Ion Exchange and Absorbant Resins Used in the Processing of Foodstuffs. Available online: http://www.coe.int/soc-sp (accessed on 28 January 2009).
- Cowan, D.A.; Fernández-Lafuente, R. Enhancing the functional properties of thermophilic enzymes by chemical modification and immobilization. Enzyme Microb. Technol. 2011, 49, 326–346. [Google Scholar] [CrossRef] [PubMed]
- Pyykkö, P.; Atsumi, M. Molecular double-bond covalent radii for elements Li–E112. Chem. Eur. J. 2009, 15, 12770–12779. [Google Scholar] [CrossRef] [PubMed]
- Bayne, L.; Ulijn, R.V.; Halling, P.J. Effect of pore size on the performance of immobilised enzymes. Chem. Soc. Rev. 2013, 42, 9000–9010. [Google Scholar] [CrossRef] [PubMed]
- Radhakrishna, M.; Grimaldi, J.; Belfort, G.; Kumar, S.K. Stability of proteins inside a hydrophobic cavity. Langmuir 2013, 29, 8922–8928. [Google Scholar] [CrossRef] [PubMed]
- Shewale, S.D.; Pandit, A.B. Hydrolysis of soluble starch using Bacillus licheniformis α-amylase immobilized on superporous CELBEADS. Carbohydr. Res. 2007, 342, 997–1008. [Google Scholar] [CrossRef] [PubMed]
- Pandya, P.H.; Jasra, R.V.; Newalkar, B.L.; Bhatt, P.N. Studies on the activity and stability of immobilized α-amylase in ordered mesoporous silicas. Microporous Mesoporous Mater. 2005, 77, 67–77. [Google Scholar] [CrossRef]
- Siso, M.I.G.; Graber, M.; Condoret, J.-S.; Combes, D. Effect of diffusional resistances on the action pattern of immobilized alpha-amylase. J. Chem. Tech. Biotechnol. 1990, 48, 185–200. [Google Scholar] [CrossRef]
- Kvesitadze, G.I.; Dvali, M.S. Immobilization of mold and bacterial amylases on silica carriers. Biotechnol. Bioeng. 1982, 24, 1765–1772. [Google Scholar] [CrossRef] [PubMed]
- Tüzmen, N.; Kalburcu, T.; Denizli, A. α-Amylase immobilization onto dye attached magnetic beads: Optimization and characterization. J. Mol. Catal. B Enzym. 2012, 78, 16–23. [Google Scholar] [CrossRef]
- Ivanova, V.; Dobreva, E.; Legoy, M.D. Characteristics of immobilized thermostable amylases from two Bacillus lichenifomis strains. Acta Biotechnol. 1998, 18, 339–351. [Google Scholar] [CrossRef]
- Qi, X.; Tester, R.F. Effect of native starch granule size on susceptibility to amylase hydrolysis. Starch/Stärke 2016, 68, 1–4. [Google Scholar] [CrossRef]
- Tester, R.F.; Karkalas, J.; Qi, X. Starch—composition, fine structure and architecture. J. Cereal Sci. 2004, 39, 151–165. [Google Scholar] [CrossRef]
- Bertoft, E.; Piyachomkwan, K.; Chatakanonda, P.; Sriroth, K. Internal unit chain composition in amylopectins. Carbohydr. Polym. 2008, 74, 527–543. [Google Scholar] [CrossRef]
- Kim, J.-K.; Park, J.-K.; Kim, H.-K. Synthesis and characterization of nanoporous silica support for enzyme immobilization. Colloids. Surf. A 2004, 241, 113–117. [Google Scholar] [CrossRef]
- Yang, S.-J.; Lee, H.-S.; Park, C.-S.; Kim, Y.-R.; Moon, T.-W.; Park, K.-H. Enzymatic analysis of an amylolytic enzyme from the hyperthermophilic archaeon Pyrococcus furiosus reveals its novel catalytic properties as both an α-amylase and a cyclodextrin-hydrolyzing enzyme. Appl. Environ. Microbiol. 2004, 70, 5988–5995. [Google Scholar] [CrossRef] [PubMed]
- Miller, G.L. Use of dinitrosalicylic acid reagent for determination of reducing sugar. Anal. Chem. 1959, 31, 426–428. [Google Scholar] [CrossRef]
- Long, J.; Jiao, A.; Wei, B.; Wu, Z.; Zhang, Y.; Xu, X.; Jin, Z. A novel method for pullulanase immobilized onto magnetic chitosan/Fe3O4 composite nanoparticles by in situ preparation and evaluation of the enzyme stability. J. Mol. Catal. B Enzym. 2014, 109, 53–61. [Google Scholar] [CrossRef]
- Sample Availability: Samples of the compounds are not available from the authors.



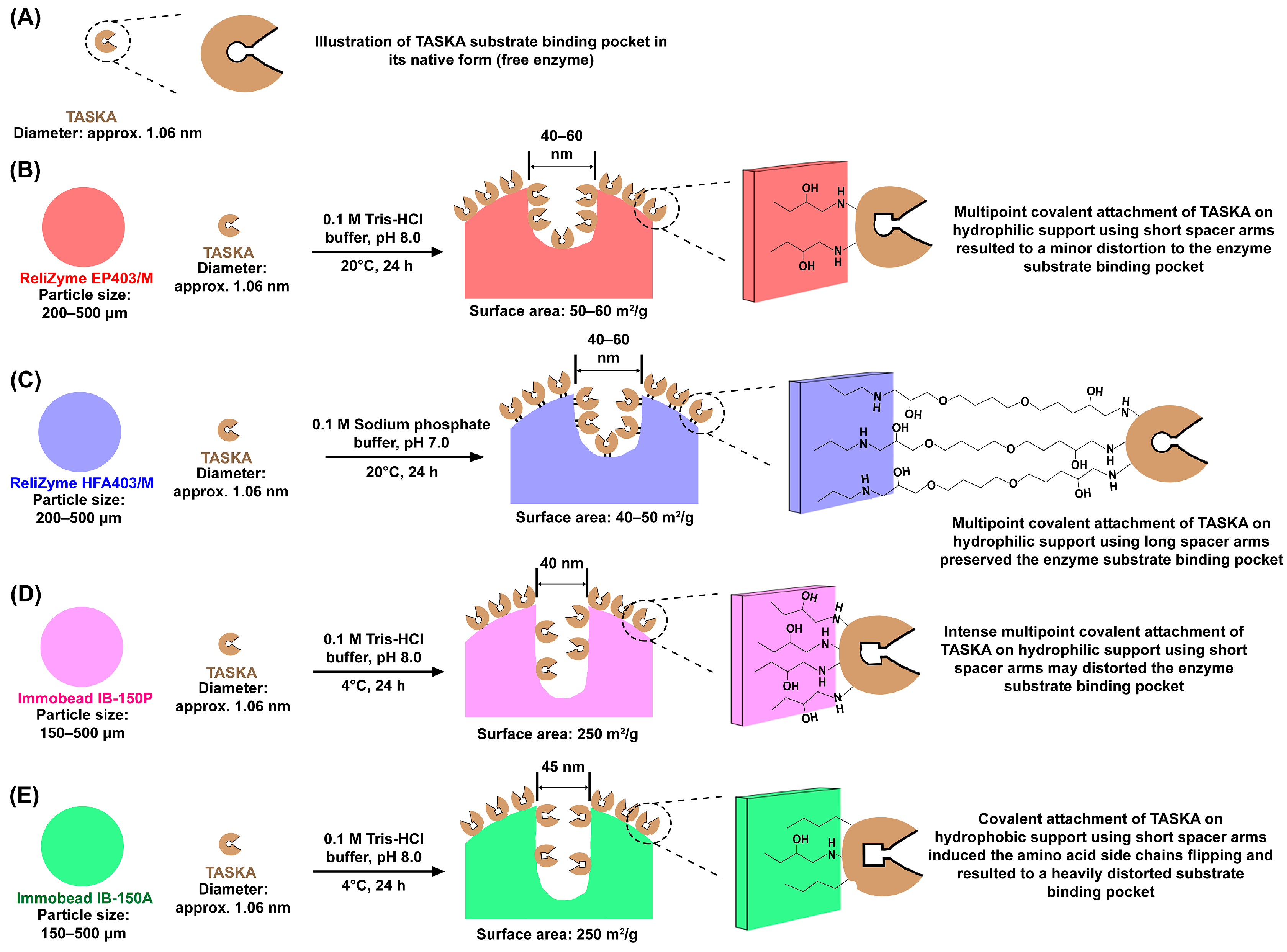
| Support | Matrix | Surface Properties | Functional Group | Content of Functional Groups (μmol/g) | Particle Size (μm) | Surface Area (m2/g) | Density of Functional Groups per Surface Area (μmol/m2) | Average Pore Diameter (nm) | Total Pore Volume (mL/g) | Water Retention (%) |
|---|---|---|---|---|---|---|---|---|---|---|
| ReliZyme EP403/M | Polymethacrylate | Hydrophilic | Epoxide | >30 | 200–500 | 50–60 | >0.5–0.6 | 40–60 | 0.9–1.0 | 65–75 |
| ReliZyme HFA403/M | Polymethacrylate | Hydrophilic | Amino-epoxide | >40 | 200–500 | 40–50 | >0.8–1.0 | 40–60 | 1.2–1.3 | 60–70 |
| Immobead IB-150P | Polyacrylic | Hydrophilic | Epoxide | 1000 | 150–500 | 250 | 4.0 | 40 | 3.0 | 75 |
| Immobead IB-150A | Polyacrylic | Hydrophobic b | Epoxide | 50 | 150–500 | 250 | 0.2 | 45 | 3.0 | 75 |
| Support | Optimum Immobilization Condition a | Immobilization Performance | ||||||
|---|---|---|---|---|---|---|---|---|
| Buffer Type (100 mM) | pH | Temperature (°C) | Offered Protein Concentration per gram Dry Support (mg/g Dry Support) | Offered Enzyme Activity per gram Dry Support (U/g Dry Support) b | Protein Loading Efficiency (%) c | Activity Retention (%) d | Activity Recovery (%) e | |
| ReliZyme EP403/M | Tris-HCl | 8.0 | 20 | 2.8 | 353 | 54 | 100 | 46 |
| ReliZyme HFA403/M | Sodium phosphate | 7.0 | 20 | 2.8 | 353 | 59 | 100 | 93 |
| Immobead IB-150P | Tris-HCl | 8.0 | 4 | 2.8 | 353 | 43 | 100 | 37 |
| Immobead IB-150A | Tris-HCl | 8.0 | 4 | 2.8 | 353 | 46 | 51 | 21 |
© 2016 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kahar, U.M.; Sani, M.H.; Chan, K.-G.; Goh, K.M. Immobilization of α-Amylase from Anoxybacillus sp. SK3-4 on ReliZyme and Immobead Supports. Molecules 2016, 21, 1196. https://doi.org/10.3390/molecules21091196
Kahar UM, Sani MH, Chan K-G, Goh KM. Immobilization of α-Amylase from Anoxybacillus sp. SK3-4 on ReliZyme and Immobead Supports. Molecules. 2016; 21(9):1196. https://doi.org/10.3390/molecules21091196
Chicago/Turabian StyleKahar, Ummirul Mukminin, Mohd Helmi Sani, Kok-Gan Chan, and Kian Mau Goh. 2016. "Immobilization of α-Amylase from Anoxybacillus sp. SK3-4 on ReliZyme and Immobead Supports" Molecules 21, no. 9: 1196. https://doi.org/10.3390/molecules21091196
APA StyleKahar, U. M., Sani, M. H., Chan, K.-G., & Goh, K. M. (2016). Immobilization of α-Amylase from Anoxybacillus sp. SK3-4 on ReliZyme and Immobead Supports. Molecules, 21(9), 1196. https://doi.org/10.3390/molecules21091196








