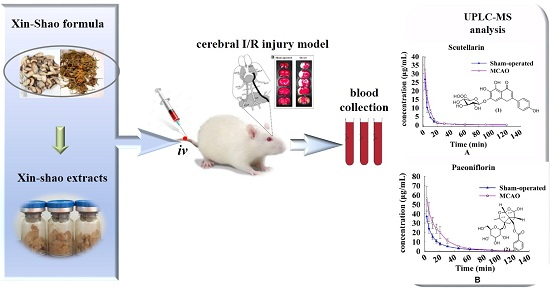
Pharmacokinetic Comparison of Scutellarin and Paeoniflorin in Sham-Operated and Middle Cerebral Artery Occlusion Ischemia and Reperfusion Injury Rats after Intravenous Administration of Xin-Shao Formula
Abstract
:1. Introduction
2. Results
2.1. Neurobehavioral Abnormality and Cerebral Infarcted Volume
2.2. Method Validation
2.2.1. Specificity
2.2.2. Linearity, LLOQ, Precision, and Accuracy
2.2.3. Recovery and Matrix Effects
2.2.4. Stability
2.3. Pharmacokinetic Analysis
3. Discussion
4. Materials and Methods
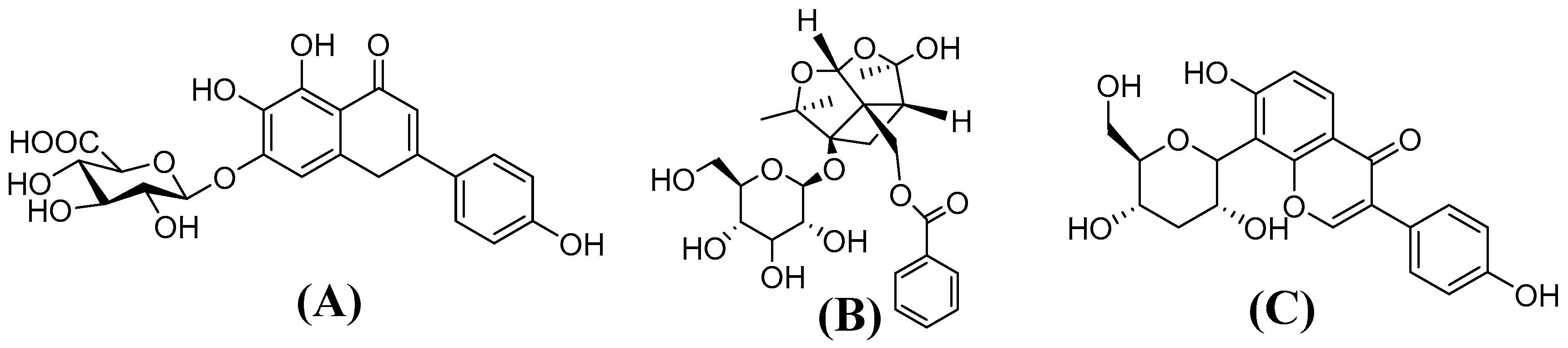
4.1. Chemicals and Reagents
4.2. Preparation of Xin-Shao formula Extracts
4.3. Establishment and Evaluation of MCAO Rat Model
4.3.1. Establishment of MCAO Model
4.3.2. Evaluation of Neurobehavioral Dysfunction
4.3.3. Measurement of Cerebral Infarction Volume
4.4. Apparatus and Analytical Conditions
4.5. Preparation of Calibration Standards and Quality Control (QC) Samples
4.6. Method Validation
4.6.1. Specificity
4.6.2. Linearity, LLOQ, Precision, and Accuracy
4.6.3. Recovery and Matrix Effect
4.6.4. Stability
4.7. Pharmacokinetic Study
4.7.1. Drug Administration and Plasma Sample Collection
4.7.2. Plasma Sample Preparation
4.8. Pharmacokinetic Studies and Statistical Analysis
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
Abbreviations
| AUC(0−t) | area under the plasma drug concentration-time curve from 0 h to the terminal time point |
| Cmax | maximum plasma drug concentration |
| CLz | clearance |
| CCA | common carotid artery |
| CYP450 | cytochrome P450 |
| ESI | electrospray ionization |
| ECA | external carotid artery |
| I/R | ischemia and reperfusion |
| IS | internal standard |
| ICA | internal carotid artery |
| LLOQ | lower limit of quantification |
| MCAO | middle cerebral artery occlusion I/R injury |
| MRT(0−t) | mean residence time |
| n-BuOH | n-butanol |
| QC | quality control |
| RSD | relative standard deviation |
| SIR | selected ion recording |
| SD | standard deviation |
| TMC | traditional Chinese medicine |
| TTC | 2,3,5-triphenyltetrazolium chloride |
| t1/2z | terminal elimination half-life |
| UPLC-MS/MS | ultra-performance liquid chromatography tandem mass spectrometry |
| UGTs | UDP-glucuronosyltransferases |
| Vz | apparent volume of distribution |
References
- Donnan, G.A.; Fisher, M.; Macleod, M.; Davis, S.M. Stroke. Lancet 2008, 371, 1612–1623. [Google Scholar] [CrossRef]
- Zeng, M.F.; Pan, L.M.; Zhu, H.X.; Zhang, Q.C.; Guo, L.W. Comparative pharmacokinetics of baicalin in plasma after oral administration of Huang-Lian-Jie-Du-Tang or pure baicalin in MCAO and sham-operated rats. Fitoterapia 2010, 81, 490–496. [Google Scholar] [CrossRef] [PubMed]
- Fujimura, M.K.; Tominaga, T.J.; Chan, P.H. Neuroprotective effect on antioxidant in ischemic brain injury: Involvement of neuronal apoptosis. Neurocrit. Care 2005, 2, 59–66. [Google Scholar] [CrossRef]
- Chen, C.; Venketasubramanian, N.; Gan, R.N.; Lambert, C.; Picard, D.; Chan, B.P.; Chan, E.; Bousser, M.G.; Xuemin, S. Danqi Piantang Jiaonang (DJ), a traditional Chinese medicine, in poststroke recovery. Stroke 2009, 3, 859–863. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.L.; Huang, Y.; Zheng, L.; Wang, A.M.; Long, Y.J.; He, X.; Lan, Y.Y. Study on the compound compatibility of Xinshao freeze-dried powder injection. Chin. J. Exp. Tradit. Med. Form. 2007, 13, 38–40. [Google Scholar]
- Wang, H.J.; Wang, Y.L.; Lan, Y.Y.; Wang, A.M.; Huang, Y.; Fang, T.H.; Xu, L. Effect of lyophilized Xinshao injection on brain circulation in anesthetized dogs. Lishizhen Med. Mater. Med. Res. 2008, 19, 2898–2899. [Google Scholar]
- Wang, H.J.; Wang, Y.L.; Fang, T.H.; Xu, L. Experimental research of effect of xinshao injection on nervous, respiratory and cardiovascular systems. J. Pharm. Res. 2008, 17, 4–5. [Google Scholar]
- Liu, L.N.; Lan, Y.Y.; Zheng, L.; Long, Y.J.; Huang, Y. Interaction of compatibility of Erigeron breviscapi and radix paeoniae rubra on thrombus formation and coagulation time. Chin. J. Exp. Tradit. Med. Form. 2012, 18, 157–160. [Google Scholar]
- Huang, Y.; Wang, Y.L.; Lan, Y.Y.; Wang, A.M.; Fang, T.H.; Xu, L. Effects of Xinshao injection on cerebral ischemia-reperfusion injury and regional cerebral blood flow in rats. Chin. J. New Drugs 2008, 17, 119–123. [Google Scholar]
- Liu, Z.Y.; Dong, L.; Dong, Y.X.; Li, L.; Lan, Y.Y.; Wang, A.M.; Wang, Y.L. Protective effects of the formula of herba erigerontis and radix paeoniae rubra on the injury induced by H2O2 of PC12 Cells. Chin. J. Exp. Tradit. Med. Form. 2014, 20, 152–156. [Google Scholar]
- Zheng, L.; Mu, J.L.; Tang, Li.; Liu, Y.; Wang, A.M.; Lan, Y.Y. Simultaneous determination of two components of Xin Shao extract in rat plasma by UPLC-MS and their pharmacokinetics and absolute bioavailability. Chin. J. New Drug 2014, 23, 819–823. [Google Scholar]
- Zhu, H.X.; Qian, Z.L.; He, F.; Liu, W.Z.; Pan, L.M.; Zhang, Q.C.; Tang, Y.P. Novel pharmacokinetic studies of the Chinese formula Huang-Lian-Jie-Du-Tang in MCAO rats. Phytomedicine 2013, 20, 767–774. [Google Scholar] [CrossRef] [PubMed]
- He, X.; Xing, D.; Ding, Y.; Li, Y.; Du, L. Effects of cerebral ischemia-reperfusion on pharmacokinetic fate of paeoniflorin after intravenous administration of paeoniae radix extract in rats. J. Ethnopharmacol. 2014, 94, 339–344. [Google Scholar] [CrossRef] [PubMed]
- Tang, H.; Tang, Y.P.; Li, N.G.; Shi, Q.P.; Guo, J.M.; Shang, E.X.; Duan, J.A. Neuroprotective effects of scutellarin and scutellarein on repeatedly cerebral ischemia-reperfusion in rats. Pharmacol. Biochem. Behav. 2014, 118, 51–59. [Google Scholar] [CrossRef] [PubMed]
- Gan, P.; Zhong, M.; Huang, X.; Sun, M.; Wang, Y.; Xiao, Y.; Zeng, C.; Yuan, Q.; Liu, Z.; Zhou, H. Pharmacokinetic comparisons of albiflorin and paeoniflorin after oral administration of Shaoyao-Gancao-Tang and single herb paeony decoction to rats. Planta. Med. 2012, 78, 237–243. [Google Scholar] [CrossRef] [PubMed]
- Zheng, L.; Mu, J.L.; Huang, Y.; Dong, L.; He, F.; Lan, Y.Y. Simultaneous determination of seven ingredients of Xin Shao freeze-dried power injection by UPLC-MS/MS. Chin. J. New Drug 2014, 23, 105–109. [Google Scholar]
- Zhang, Y.Q.; Li, H.; Huang, M.Q.; Huang, M.; Chu, K.D.; Xu, W.; Zhang, S.N.; Que, J.H.; Chen, L.D. Paeoniflorin, a monoterpene glycoside, protects the brain from cerebral ischemic injury via inhibition of apoptosis. Am. J. Chin. Med. 2015, 43, 543–557. [Google Scholar] [CrossRef] [PubMed]
- Guo, R.B.; Wang, G.F.; Zhao, A.P.; Gu, J.; Sun, X.L.; Hu, G. Paeoniflorin protects against ischemia-induced brain damages in rats via inhibiting MAPKs/NF-κB-mediated inflammatory responses. PLoS ONE 2012, 7, e49701. [Google Scholar] [CrossRef] [PubMed]
- Yu, J.B.; Zhang, Jun.; Wang, W.Q.; Huang, L.Q.; Feng, X.F. Comparative study on content of four constituent between wild and cutivated radix paeoniae rubra. Chin. J. Exp. Tradit. Med. Form. 2011, 17, 107–111. [Google Scholar]
- Liu, J.; Chen, L.; Fan, C.R.; Li, H.; Huang, M.Q.; Xiang, Q.; Xu, W.; Xu, W.; Chu, K.D.; Lin, Y. Qualitative and quantitative analysis of major constituents of paeoniae radix alba and paeoniae radix rubra by HPLC-DAD-Q-TOF-MS/MS. Chin. J. Chin. Mater. Med. 2015, 40, 1762–1770. [Google Scholar]
- Niu, W.M.; Liu, Z.B.; Yang, X.H.; Wang, Y.; Shi, H.L. Effect on CATmRNA expression in cerebral ischemia reperfusion rat with hepatic injury treated by protecting liver to nourish brain therapy. Liaoning J. Tradit. Chin. Med. 2013, 40, 342–344. [Google Scholar]
- Zhao, W.; Li, F.J.; Wang, S. Protective effects of chrysophanol on liver injury induced by cerebral ischemia-reperfusion in mice. Acta Neuropharmacol. 2014, 4, 1–9. [Google Scholar]
- Zhong, W.H.; Qian, K.J.; Xiong, J.B.; Ma, K.; Wang, A.Z.; Zou, Y. Curcumin alleviates lipopolysaccharide induced sepsis and liver failure by suppression of oxidative stress-related inflammation via PI3K/AKT and NF-κB related signaling. Biomed. Pharmacother. 2016, 83, 302–313. [Google Scholar] [CrossRef] [PubMed]
- Gao, C.Y.; Chen, X.Y.; Zhong, D.F. Absorption and disposition of scutellarin in rats: A pharmacokinetic explanation for the high exposure of its isomeric metabolite. Drug Metab. Dispos. 2011, 39, 2034–2044. [Google Scholar] [CrossRef] [PubMed]
- Bock, K.W. Roles of human UDP-glucuronosyltransferases in clearance and homeostasis of endogenous substrates, and functional implications. Biochem. Pharmacol. 2015, 96, 77–82. [Google Scholar] [CrossRef] [PubMed]
- Gao, C.Y.; Zhang, H.J.; Guo, Z.T.; You, T.G.; Chen, X.Y.; Zhong, D.F. Mechanistic studies on the absorption and disposition of scutellarin in humans: Selective OATP2B1-mediated Hepatic uptake is a likely key determinant for its unique pharmacokinetic characteristics. Drug Metab. Dispos. 2012, 40, 2009–2020. [Google Scholar] [CrossRef] [PubMed]
- Richardson, T.A.; Sherman, M.; Kalman, D.; Morgan, E.T. Expression of UDP-glucuronosyltransferase isoform mRNAs during inflammation and infection in mouse liver and kidney. Drug Metab. Dispos. 2006, 34, 351–353. [Google Scholar] [CrossRef] [PubMed]
- Petrovic-Djergovic, D.; Goonewardena, S.N.; Pinsky, D.J. Inflammatory disequilibrium in stroke. Circ. Res. 2016, 119, 142–158. [Google Scholar] [CrossRef] [PubMed]
- Yang, X.F.; He, W.; Lu, W.H.; Zeng, F.D. Effects of scutellarin on liver function after brain ischemia/reperfusion in rats. Acta Pharmacol. Sin. 2003, 24, 1118–1124. [Google Scholar] [PubMed]
- Gong, W.H.; Zheng, W.X.; Wang, J.; Chen, S.H.; Pang, B.; Hu, X.M.; Cao, X.L. Coexistence of hyperlipidemia and acute cerebral ischemia/reperfusion induces severe liver damage in a rat model. World J. Gastroenterol. 2012, 18, 4934–4943. [Google Scholar] [CrossRef] [PubMed]
- Bing, Y.T.; Zhu, S.Y.; Jiang, K.; Dong, G.C.; Li, J.; Yang, Z.Q.; Yang, J.; Yue, J. Reduction of thyroid hormones triggers down-regulation of hepatic CYP2B through nuclear receptors CAR and TR in a rat model of acute stroke. Biochem. Pharmacol. 2014, 87, 636–649. [Google Scholar] [CrossRef] [PubMed]
- Tan, Y.; Shen, G.L.; Zhuang, X.M.; Yuan, M.; Li, H.; Gao, Y. Metabolic characteristics of paeoniflorin in vitro. J. Int. Pharm. Res. 2013, 40, 625–633. [Google Scholar]
- Lee, J.H.; Lee, A.; Oh, J.H.; Lee, Y.J. Comparative pharmacokinetic study of paclitaxel and docetaxel instreptozotocin-induced diabetic rats. Biopharm. Drug Dispos. 2012, 33, 474–486. [Google Scholar] [CrossRef] [PubMed]
- Guide for the Care and Use of Laboratory Animals; Institute of Laboratory Animal Resources, National Research Council: Washington, DC, USA, 1996.
- Longa, E.Z.; Weinstein, P.R.; Carlson, S.; Cummins, R. Reversible middle cerebral artery occlusion without craniectomy in rats. Stroke 1989, 20, 84–91. [Google Scholar] [CrossRef] [PubMed]
- Joshi, C.N.; Jain, S.K.; Murthy, P.S. An optimized triphenyltetrazolium chloride method for identification of cerebral infarcts. Brain Res. Protoc. 2004, 13, 11–17. [Google Scholar] [CrossRef] [PubMed]
- Ding, W.T.; Zhou, L.Q.; Liu, W.; Guan, L.; Li, X.Y.; Liu, H.M.; Yan, F.M.; Xu, J.W.; Zeng, W.Y.; Qiu, M. Opposite effects of the gap junction blocker octanol on focal cerebral ischemia occluded for different durations. Mol. Med. Rep. 2014, 9, 2485–2490. [Google Scholar] [CrossRef] [PubMed]
- Sample Availability: Samples of the compounds scutellarin and paeoniflorin are available from the authors.

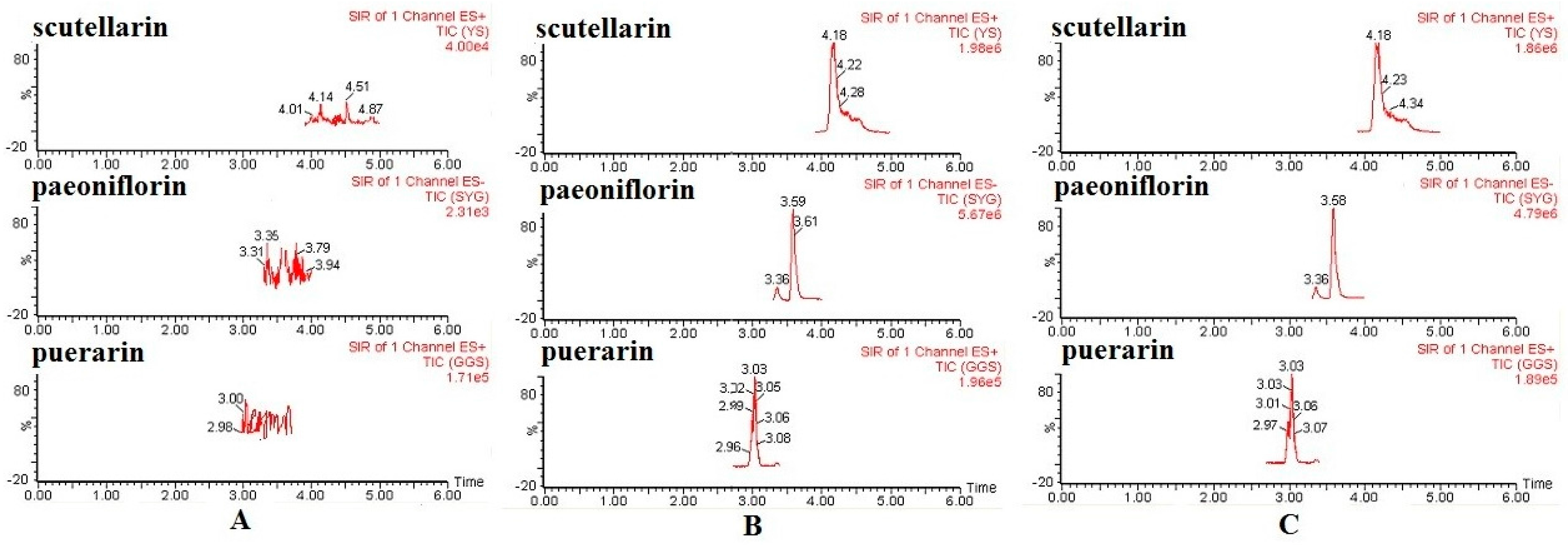

| Components | Concentration Spiked (μg/mL) | Intra-Day | Inter-Day | ||||
|---|---|---|---|---|---|---|---|
| Calculate Concentration (μg/mL) | Precision (RSD, %) | Accuracy (%) | Calculate Concentration (μg/mL) | Precision (RSD, %) | Accuracy (%) | ||
| Scutellarin | 0.1160 | 0.1123 ± 0.0075 | 6.7 | 96.84 | 0.1093 ± 0.0097 | 8.9 | 94.25 |
| 0.9277 | 0.9030 ± 0.037 | 4.1 | 97.34 | 0.94 ± 0.056 | 6.0 | 101.3 | |
| 14.84 | 14.10 ± 0.62 | 4.4 | 95.01 | 14.27 ± 0.91 | 6.4 | 96.14 | |
| Paeoniflorin | 0.1180 | 0.1140 ± 0.0066 | 5.8 | 96.61 | 0.1127 ± 0.0064 | 5.7 | 95.48 |
| 0.9440 | 0.9133 ± 0.040 | 4.4 | 96.75 | 0.888 ± 0.047 | 5.2 | 94.07 | |
| 15.11 | 15.33 ± 0.99 | 6.4 | 101.5 | 14.63 ± 1.1 | 7.6 | 96.85 | |
| Components | Concentration Spiked (μg/mL) | Extraction Recovery (%) | Matrix Effect (%) |
|---|---|---|---|
| Scutellarin | 0.1160 | 84.77 ± 6.6 | 88.79 ± 2.3 |
| 0.9277 | 89.43 ± 2.4 | 91.23 ± 4.0 | |
| 14.84 | 90.52 ± 3.2 | 91.64 ± 3.4 | |
| Paeoniflorin | 0.1180 | 86.72 ± 4.4 | 85.59 ± 2.5 |
| 0.9440 | 89.95 ± 3.6 | 84.39 ± 2.9 | |
| 15.11 | 94.04 ± 1.6 | 87.05 ± 2.4 |
| Components | Concentration Spiked (μg/mL) | 6 h at 24 °C | Three Freeze-Thaw Cycles | Long-Term Stability (−20 °C, 2 weeks) | |||
|---|---|---|---|---|---|---|---|
| Precision (RSD, %) | Accuracy (%) | Precision (RSD, %) | Accuracy (%) | Precision (RSD, %) | Accuracy (%) | ||
| Scutellarin | 0.1160 | 5.0 | 101.3 | 3.9 | 96.55 | 5.2 | 93.39 |
| 0.9277 | 3.3 | 95.47 | 6.3 | 97.16 | 3.2 | 95.47 | |
| 14.84 | 4.3 | 98.54 | 5.4 | 96.59 | 3.7 | 96.91 | |
| Paeoniflorin | 0.1180 | 5.1 | 100.3 | 7 | 103.1 | 6.5 | 94.07 |
| 0.9440 | 4.6 | 95.94 | 4.4 | 97.35 | 5.3 | 99.93 | |
| 15.11 | 3.8 | 95.70 | 8.8 | 96.00 | 4.2 | 97.37 | |
| Parameters | Scutellarin | Paeoniflorin | |||
|---|---|---|---|---|---|
| Sham-Operated Group | MCAO Group | Sham-Operated Group | MCAO Group | ||
| AUC(0–t) | mg·min·L−1 | 3.88 ± 1.89 | 4.431 ± 0.716 | 10.35 ± 2.41 | 18.35 ± 3.71 ** |
| MRT(0–t) | min | 0.26 ± 0.09 | 0.42 ± 0.15 * | 0.38 ± 0.12 | 0.35 ± 0.053 |
| t1/2z | min | 0.81 ± 0.48 | 1.66 ± 0.68 * | 0.39 ± 0.0953 | 0.33 ± 0.091 |
| CLz | L·min·kg−1 | 1.57 ± 0.60 | 0.97 ± 0.26 * | 0.78 ± 0.16 | 0.45 ± 0.11 ** |
| Vz | L·kg−1 | 1.53 ± 0.66 | 2.90 ± 1.70 | 0.43 ± 0.07 | 0.21 ± 0.08 ** |
| Cmax | mg·L−1 | 26.72 ± 12.18 | 32.34 ± 6.407 | 36.40 ± 5.71 | 56.28 ± 12.62 ** |
| Molecular | Polarity | Molecular Mass | Parent (m/z) | Dwell Time (s) | Cone Voltage (v) |
|---|---|---|---|---|---|
| Scutellarin | ESI+ | 462.04 | 463.04 | 0.2 | 22 |
| Paeoniflorin | ESI− | 526.12 | 525.12 | 0.1 | 26 |
| Puerarin (IS) | ESI+ | 416.14 | 417.14 | 0.1 | 40 |
© 2016 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Li, Y.; Lu, Y.; Hu, J.; Gong, Z.; Yang, W.; Wang, A.; Zheng, J.; Liu, T.; Chen, T.; Hu, J.; et al. Pharmacokinetic Comparison of Scutellarin and Paeoniflorin in Sham-Operated and Middle Cerebral Artery Occlusion Ischemia and Reperfusion Injury Rats after Intravenous Administration of Xin-Shao Formula. Molecules 2016, 21, 1191. https://doi.org/10.3390/molecules21091191
Li Y, Lu Y, Hu J, Gong Z, Yang W, Wang A, Zheng J, Liu T, Chen T, Hu J, et al. Pharmacokinetic Comparison of Scutellarin and Paeoniflorin in Sham-Operated and Middle Cerebral Artery Occlusion Ischemia and Reperfusion Injury Rats after Intravenous Administration of Xin-Shao Formula. Molecules. 2016; 21(9):1191. https://doi.org/10.3390/molecules21091191
Chicago/Turabian StyleLi, Yueting, Yuan Lu, Jianchun Hu, Zipeng Gong, Wu Yang, Aimin Wang, Jiang Zheng, Ting Liu, Tingting Chen, Jie Hu, and et al. 2016. "Pharmacokinetic Comparison of Scutellarin and Paeoniflorin in Sham-Operated and Middle Cerebral Artery Occlusion Ischemia and Reperfusion Injury Rats after Intravenous Administration of Xin-Shao Formula" Molecules 21, no. 9: 1191. https://doi.org/10.3390/molecules21091191
APA StyleLi, Y., Lu, Y., Hu, J., Gong, Z., Yang, W., Wang, A., Zheng, J., Liu, T., Chen, T., Hu, J., Mi, L., Li, Y., Lan, Y., & Wang, Y. (2016). Pharmacokinetic Comparison of Scutellarin and Paeoniflorin in Sham-Operated and Middle Cerebral Artery Occlusion Ischemia and Reperfusion Injury Rats after Intravenous Administration of Xin-Shao Formula. Molecules, 21(9), 1191. https://doi.org/10.3390/molecules21091191