Abstract
Three new indole alkaloids, named naucleamide G (1), and nauclealomide B and C (5 and 6), were isolated from the n-BuOH-soluble fraction of an EtOH extract of the leaves of Nauclea officinalis, together with three known alkaloids, paratunamide C (2), paratunamide D (3) and paratunamide A (4). The structures with absolute configurations of the new compounds were identified on the basis of 1D and 2D NMR, HRESIMS, acid hydrolysis and quantum chemical circular dichroism (CD) calculation. According to the structures of isolated indole alkaloids, their plausible biosynthetic pathway was deduced.
1. Introduction
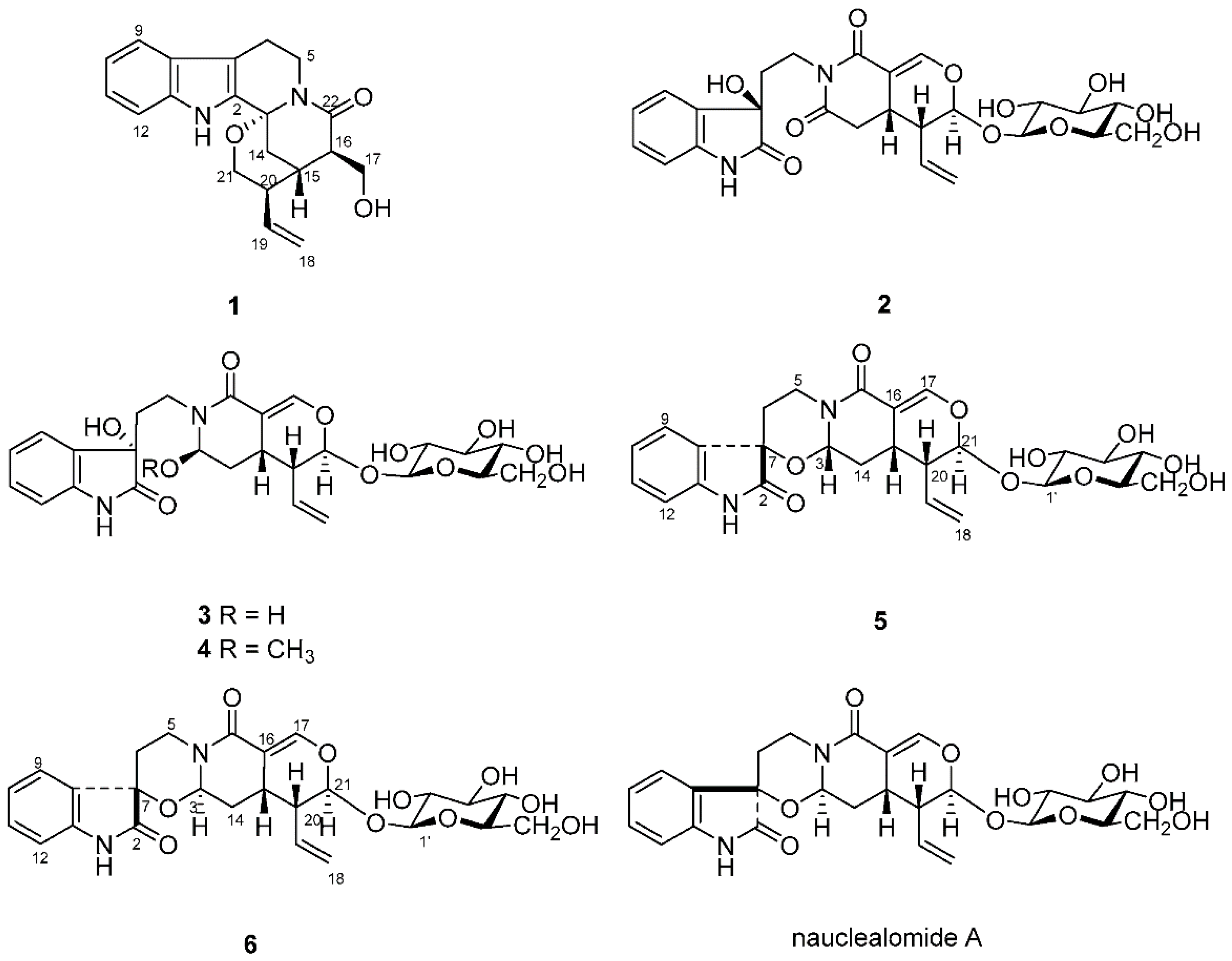
Nauclea officinalis Pierre ex Pitard belongs to the genus Nauclea in family Rubiaceae, which is mainly distributed in Hainan, Guangxi, Guangdong and other provinces in the south of China. As a traditional Chinese medicine, the bark and twigs of N. officinalis is widely used for the treatment of colds, fever, acute tonsillitis, sore throat, conjunctivitis, enteritis, dysentery, eczema and other ailments [1]. Previous phytochemical investigation had revealed that monoterpene indole alkaloids, which present a polycyclic skeleton with a tetrahydro-β-carboline ring, are the main components of this plant [2,3,4,5,6,7,8,9,10,11,12,13]. The complex ring system and diversity of stereostructure of these monoterpene indole alkaloids have attracted great interest for phytochemical studies. As a part of our ongoing program to search for structurally unique alkaloids from the medicinal plants in southern China [5,13], we carried out the continuing investigation on the leaves of N. officinalis. In our present study, three new indole alkaloids, naucleamide G (1), and nauclealomide B and C (5 and 6), together with three known ones, paratunamide C (2), paratunamide D (3) and paratunamide A (4) (Figure 1), were isolated, among which oxindole alkaloids (2–6) were rarely reported from the Nauclea genus. In this paper, we describe the structure elucidation of these new alkaloids by means of NMR, HRESIMS, circular dichroism spectroscopy and computational analysis.
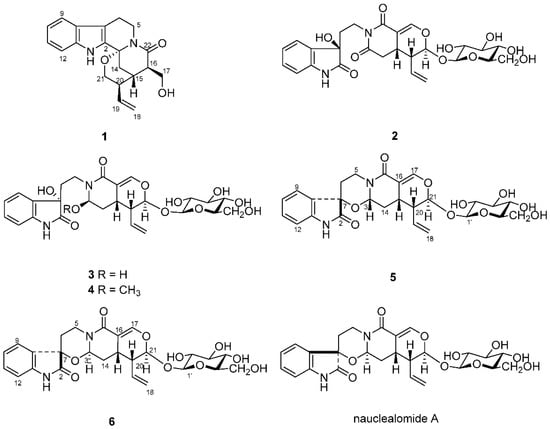
Figure 1.
Structures of compounds 1–6 isolated from N. officinalis, as well as structure of nauclealomide A mentioned in the text.
2. Results and Discussion
Compound 1 showed a molecular formula of C20H22N2O3 based on the HRESIMS (m/z 339.1703 [M + H]+; calculated for C20H23N2O3: m/z 339.1709). The UV spectrum of 1 showed absorption maxima at 211 and 290 nm. The IR spectrum exhibited absorptions at 3373 and 1636 cm−1, which suggested the presence of amino and α, β-unsaturated carbonyl functional groups. The 1H-NMR spectrum of 1 displayed four aromatic protons (δH 7.46 (1H, d, J = 8.0 Hz), 7.37 (1H, d, J = 8.0 Hz), 7.12 (1H, t, J = 8.0 Hz) and 7.00 (1H, t, J = 8.0 Hz)), three terminal vinyl protons (δH 6.39 (1H, ddd, J = 17.3, 10.4, 7.3 Hz), 5.28 (1H, d, J = 17.3 Hz) and 5.23 (1H, d, J = 10.4 Hz)) and one amino proton (δH 11.19 (1H, s)). The 13C-NMR and DEPT spectra of 1 displayed 20 signals, including six methylenes, eight methines, one carbonyl, and five quaternary carbons, suggesting the presence of a monoterpene indole alkaloid.
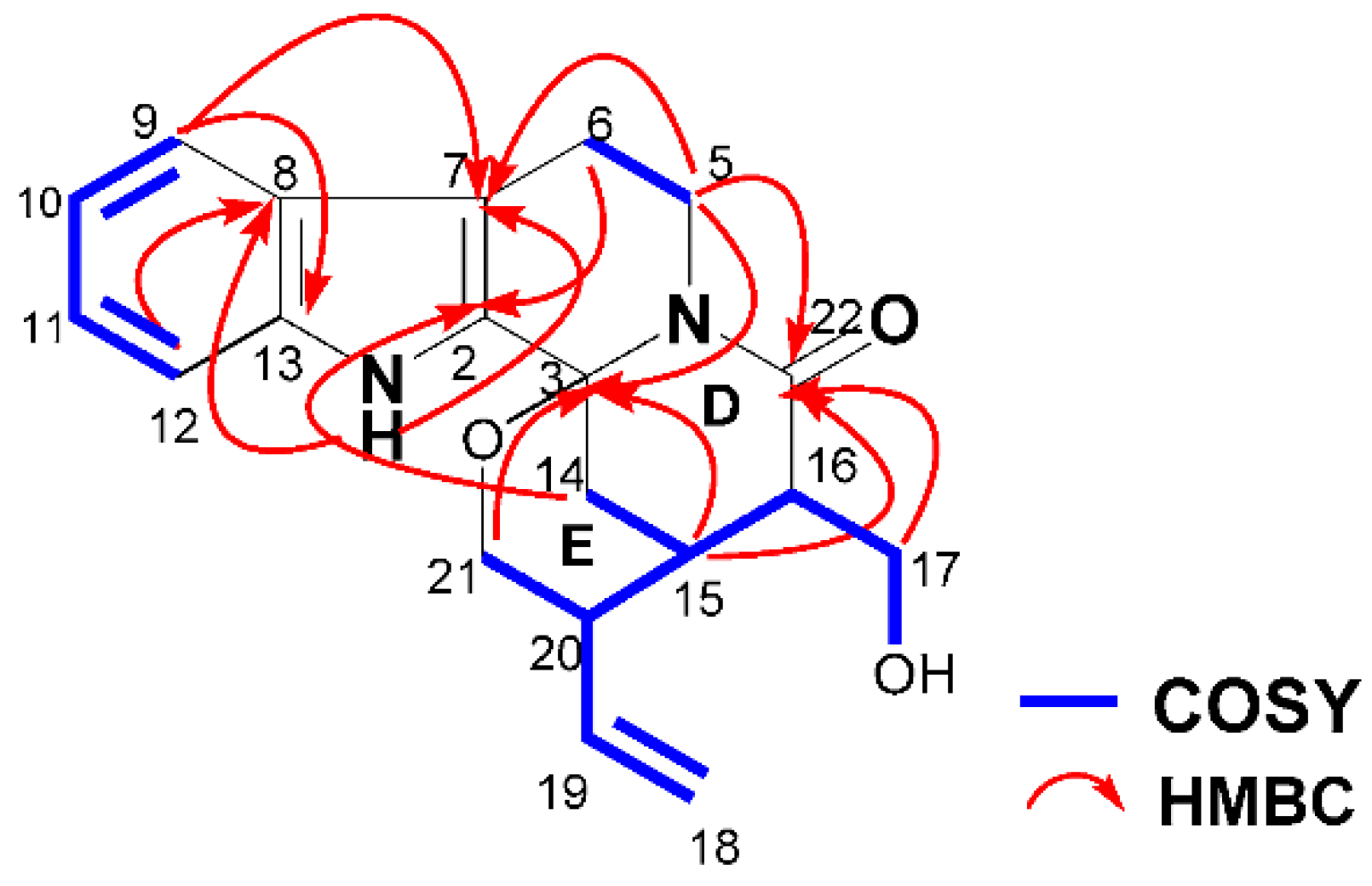
With the aid of Two Dimensional 1H Correlation Spectroscopy (1H-1H COSY), 1H-Detected Heteronuclear Single Quantum Correlation (HSQC), 1H-Detected Heteronuclear Multiple Bond Correlation (HMBC), and Rotating-frame Overhauser Enhancement Spectroscopy (ROESY) experiments, all of the 1H and 13C-NMR signals of 1 were assigned as shown in Table 1. The 1H-1H COSY spectrum of 1 revealed the presence of three spin coupling systems (δH 7.46 (H-9)↔7.00 (H-10)↔7.12 (H-11)↔7.37 (H-12), δH 4.80 (Ha-5)↔2.61 (Hb-6) and δH 5.28 (Ha-18)/5.23 (Hb-18)↔6.39 (H-19)↔2.51 (H-20)↔2.33 (H-15)↔2.79 (H-16)↔4.08 (Ha-17)/3.65 (Hb-17)↔4.69 (H-OH), 2.33 (H-15)↔1.72 (Hb-14), 2.51 (H-20)↔3.59 (Hb-21)). In the HMBC spectrum, the correlations between H-1 (δH 11.19) and C-7 (δC 108.7)/C-8 (δC 125.3)/C-2 (δC 133.8)/C-13 (δC 136.2), between H-9 (δH 7.46) and C-7 (δC 108.7)/C-13 (δC 136.2), between H-12 (δH 7.37) and C-8 (δC 125.3), between Ha-5 (δH 4.80) and C-3 (δC 80.9)/C-7 (δC 108.7), as well as between Ha-6 (δH 2.74)/Hb-6 (δH 2.61) and C-2 (δC 133.8) revealed the presence of a tetrahydro-β-carboline ring. In addition, the correlations between Ha-5 (δH 4.80) and C-22 (δC 170.6), between Ha-14 (δH 2.89) and C-2 (δC 133.8), between Ha-21 (δH 3.66)/Hb-21 (δH 3.59)/Ha-14 (δH 2.89)/Hb-14 (δH 1.72)/H-15 (δH 2.33) and C-3 (δC 80.9), as well as between H-15 (δH 2.33)/Ha-17 (δH 4.08)/Hb-17 (δH 3.65) and C-22 (δC 170.6) revealed the presence of ring D, which was fused to the tetrahydro-β-carboline ring and ring E (Figure 2).

Table 1.
1H (400 MHz) and 13C (100 MHz) NMR data of 1 (dimethylsulfoxide-d6 (DMSO-d6), δ in ppm, J in Hz).
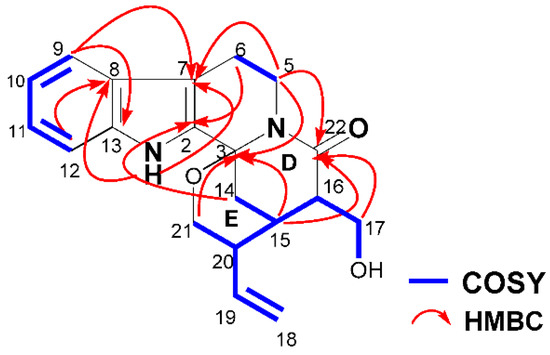
Figure 2.
1H-1H COSY and key HMBC correlations of 1.
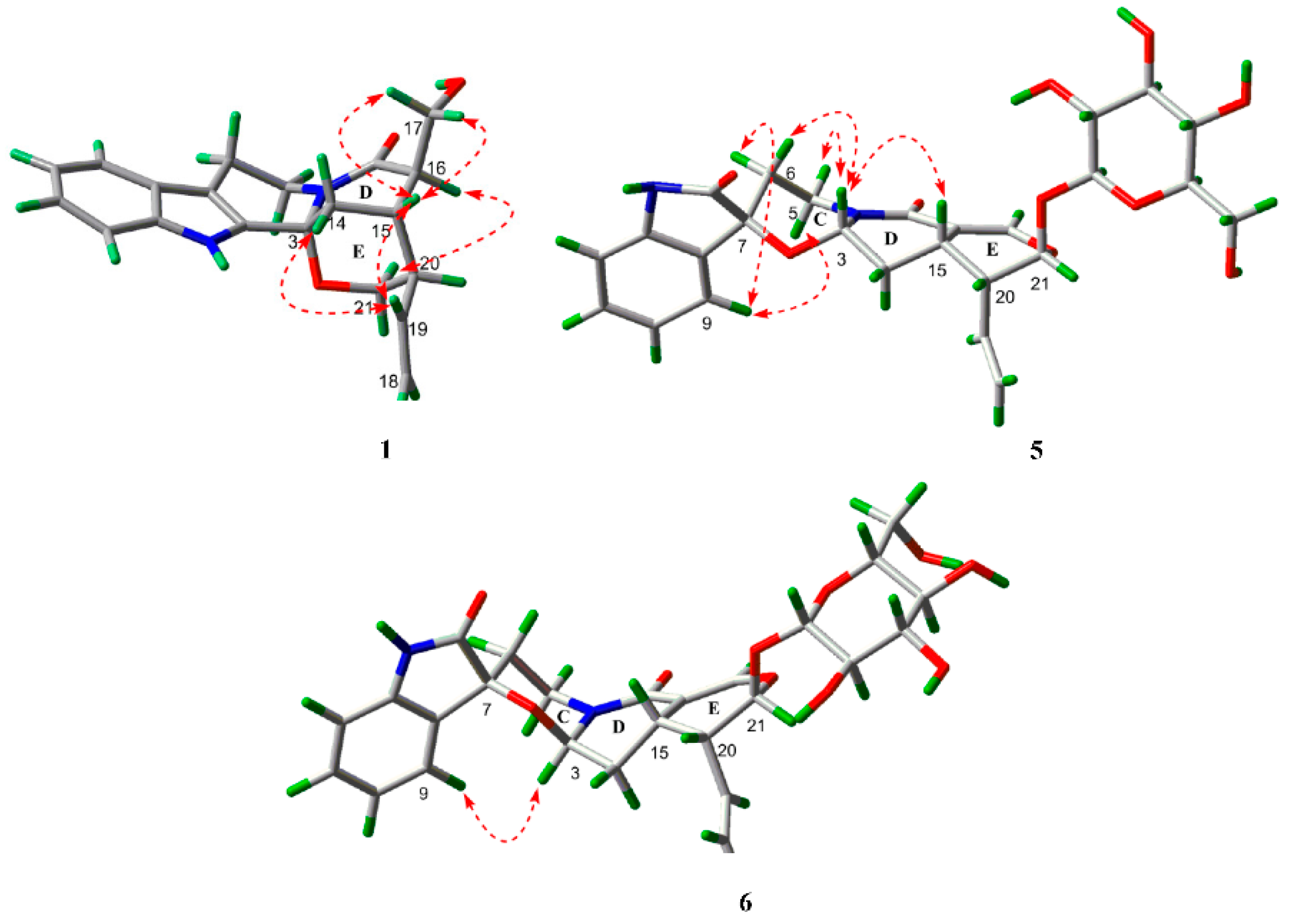
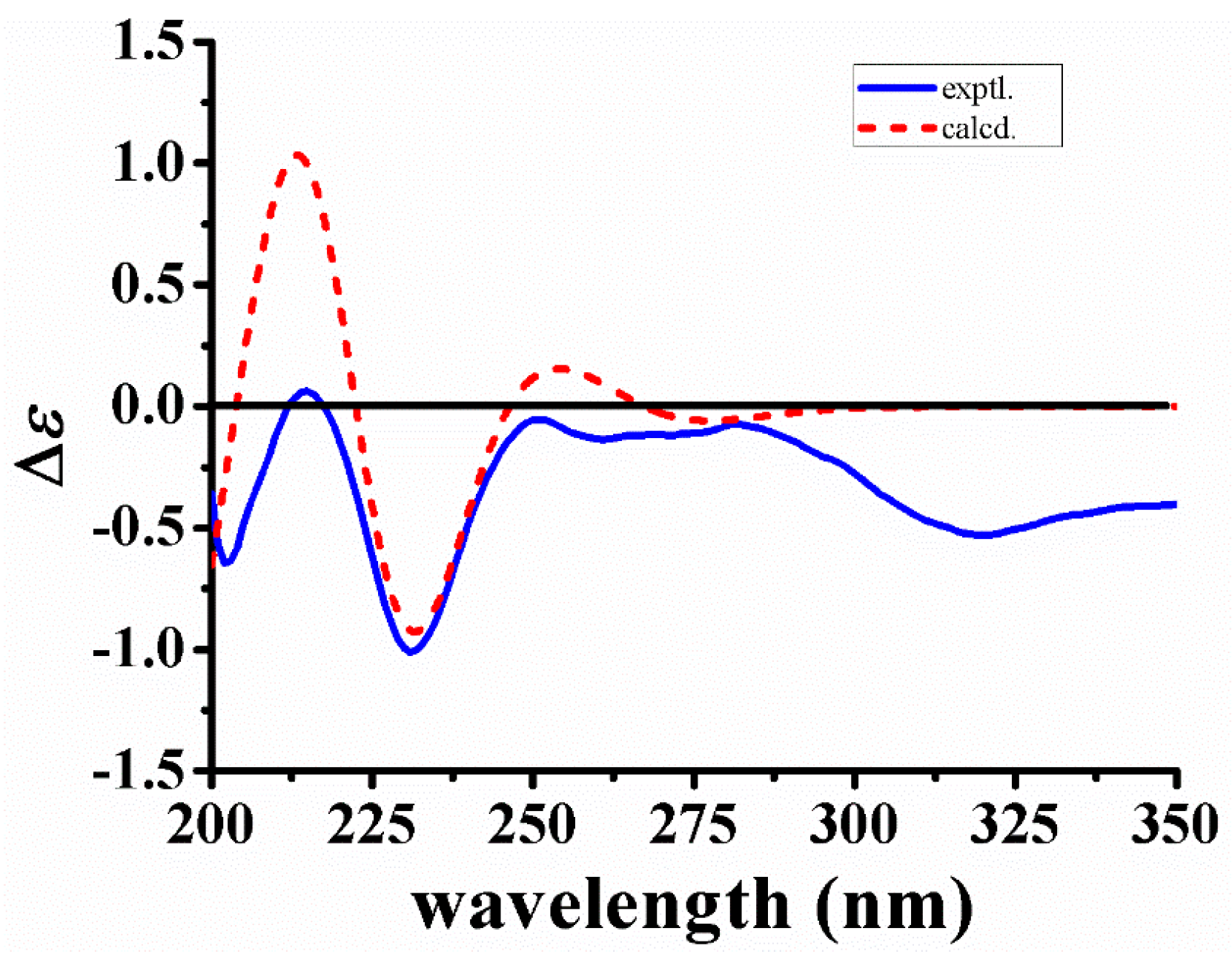
The relative configuration of 1 was deduced from coupling constants and ROESY data. The coupling constants of 3J14a,15 = 3.3 Hz, 3J14b,15 = 2.0 Hz and 3J15,16 < 1.0 Hz, and the ROESY correlations between Hb-21 and H-16, as well as between H-15 and Ha-17/Hb-17 indicated both chair conformations of rings D and E, a cis-ring junction between rings D and E, and different orientations of H-15 and H-16 [14]. In addition, the ROESY correlations between H-19 and Ha-14/H-15 suggested different orientations of H-15 and H-20. On the basis of the above results, the relative configuration of 1 was elucidated as shown in Figure 3. The absolute configuration of 1 was confirmed by the quantum chemical CD calculation method at the (B3LYP/6-31G(d)) level using the GAUSSIAN 09 program, and the calculated CD curve of 3R,15S,16R,20R-1 was similar to the measured spectrum (Figure 4). Therefore, the structure of 1 was established and named naucleamide G.
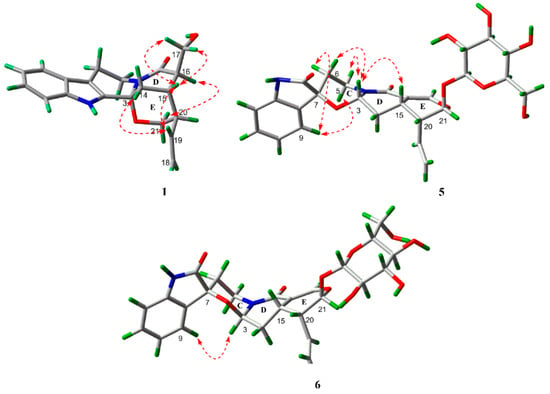
Figure 3.
Selected ROESY correlations of 1, 5 and 6.
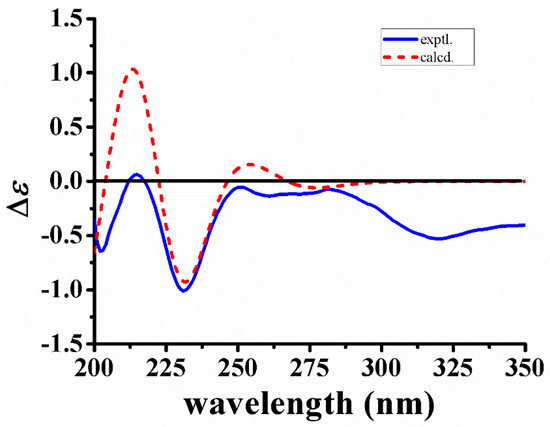
Figure 4.
Calculated and experimental CD spectra of 1.
Compounds 5 and 6 showed the same molecular formula C26H30N2O10 as nauclealomide A by their HRESIMS (m/z 553.1782 [M + Na]+ and 553.1783 [M + Na]+). Moreover, the 1H and 13C-NMR data of 5 and 6 were similar to those of nauclealomide A (Table 2). Thus, the planar structures of 5, 6 were identical to that of nauclealomide A (Figure 1) [13]. The coupling constants of 3J20,21 (J = 1.7 Hz) and 3J20,15 (J = 5.8 Hz) suggested the presence of β/β/α orientation for H-15, H-20 and H-21 in 5 and 6 [15,16,17].

Table 2.
1H (400 MHz) and 13C (100 MHz) NMR data of nauclealomide A, 5 and 6 (CD3OD, δ in ppm, J in Hz).
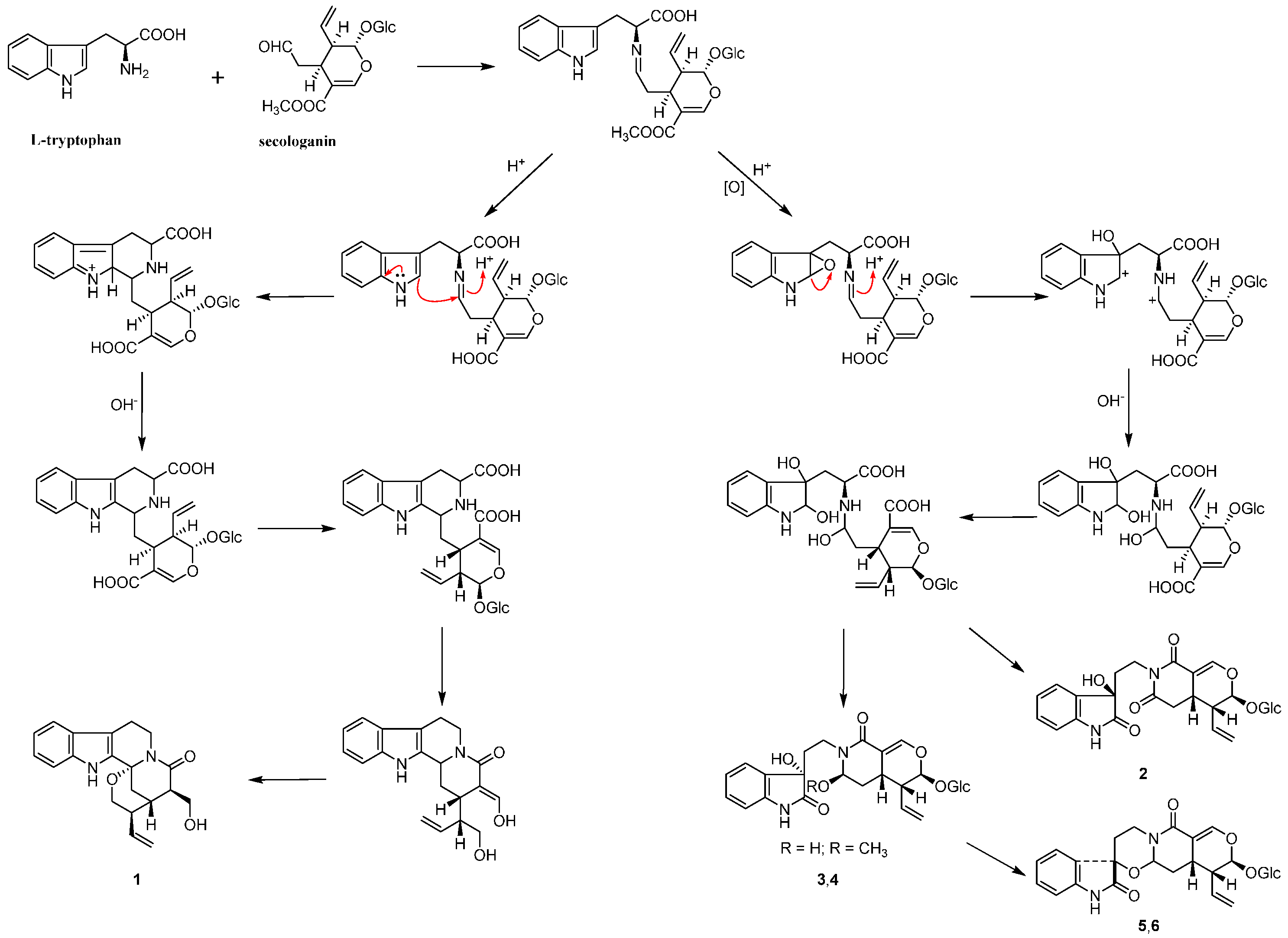
In the 1H-NMR spectrum of 5, the coupling constants of 3J3,14a (J = 9.5 Hz) and 3J3,14b (J = 4.6 Hz) indicated the presence of β configuration of H-3, which was confirmed by the ROESY correlation between H-3 and H-15. In the ROESY spectrum, the correlations between H-3 and Hb-5/Hb-6, as well as between H-9 and Ha-5/Ha-6 indicated the presence of α/β/α/β configuration of Ha-5, Hb-5, Ha-6 and Hb-6 (Figure 3). The absolute configuration of C-15 in these indole alkaloids was determined as S biosynthetically based on secologanin (Scheme 1). Thus, the absolute configuration of 5 was elucidated as 3S, 7S, 15S, 20R, 21S.
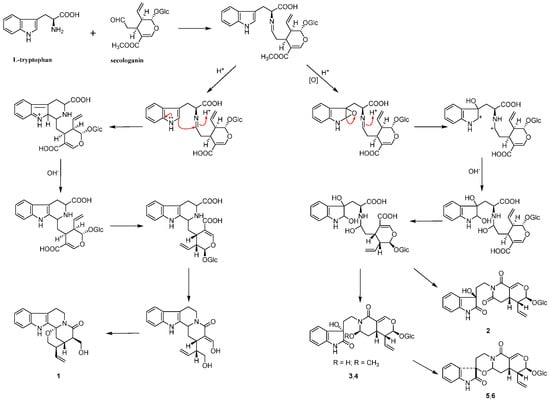
Scheme 1.
The plausible biosynthetic pathway for 1–6.
In the 1H-NMR spectrum of 6, the coupling constants of 3J3,14a (J = 3.6 Hz) and 3J3,14b (J = 2.1 Hz) indicated the presence of α configuration of H-3. In the ROESY spectrum, the correlation between H-3 and H-9 was observed, which suggested that the carbonyl group at C-2 was above the C/D/E plane (Figure 3). Considering the biogenetic pathway for these indole alkaloids, the absolute configuration of C-15 was determined as S. Thus, the absolute configuration of 6 was elucidated as 3R, 7S, 15S, 20R, 21S.
The three known indole alkaloids were identified as paratunamide C (2) [18], paratunamide D (3) [18], and paratunamide A (4) [18] by comparison of their spectroscopic data with those reported in the literature.
Based on the structural features of compounds 1–6, which were all derived from l-tryptophan and secologanin, the plausible biosynthetic pathway for them was deduced (Scheme 1).
3. Materials and Methods
3.1. General Procedures
Optical rotations were measured on a JASCO P-1020 polarimeter (JASCO Corporation, Tokyo, Japan). UV spectra were recorded on a JASCO V-550 UV/VIS spectrophotometer (JASCO Corporation). A JASCO FT/IR-480 plus FT-IR spectrometer (JASCO Corporation) was used for scanning the IR spectra with KBr pellets. 1H, 13C, and 2D NMR spectra were recorded on a Bruker AV-400 spectrometer (Bruker Corporation, Rheinstetten, Germany). HR-ESI-MS data were conducted on an Agilent 6210 LC/MSD TOF mass spectrometer. CD spectra were obtained using a JASCO J-810 circular dichroism spectrometer. For column chromatography, silica gel (300–400 mesh; Qingdao Marine Chemical Group Corporation, Qingdao, China) and Sephadex LH-20 (Pharmacia, Pittsburgh, PA, USA) were used. TLC analyses were carried out using precoated silica gel GF254 plates (Yantai Chemical Industry Research Institute, Yantai, China). Analytic high-performance liquid chromatography (HPLC) was performed on a SHIMADZU chromatography (SHIMADZU Corporation, Kyoto, Japan) equipped with an LC-20AD pump and an SPD-M20A diode-array detector (DAD) with an Inertsil ODS-3 column (4.6 mm × 250 mm, 5 μm). Preparative HPLC was carried out on a SHIMADZU instrument equipped with an LC-20AP pump and an SPD-20A (SHIMADZU Corporation) detector with a YMC-Pack ODS-A column (20 mm × 250 mm, 5 μm).
3.2. Plant Material
The leaves of N. officinalis were collected in Sanya City, Hainan Province, China, in July 2008, and authenticated by Professor Wei-ping Chen (Hainan Branch Institute of Medicinal Plants, Chinese Academy of Medical Science, Haikou, China). A voucher specimen (No. 20090223) was deposited in the Institute of Traditional Chinese Medicine and Natural Products, Jinan University, Guangzhou, China.
3.3. Extraction and Isolation
Air-dried and powdered leaves of N. officinalis (4.8 kg) were extracted with 95% EtOH (50 L × 3, 3 days each) at room temperature. The EtOH solution was evaporated under reduced pressure to afford a residue (250.0 g), which was suspended in water and partitioned successively with light petroleum, EtOAc and n-BuOH. The n-BuOH fraction (60.0 g) was subjected to silica gel CC, eluting with CHCl3-MeOH (100:0→0:100) to give nine fractions (Fr. 1–9). Fr. 1 (5.0 g) was separated by a silica gel column with CHCl3-MeOH (95:5) as the eluent to give fifteen subfractions (Fr. 1-1–Fr. 1-15). Fr. 1–2 (90.0 mg) was subsequently purified by a Sephadex LH-20 column eluted with MeOH to yield compounds 1 (5.0 mg). Fr. 7 (1.0 g) was further purified on a Sephadex LH-20 column using MeOH as eluent, then compounds 2 (22.0 mg), 3 (32.0 mg), 4 (17.0 mg), 5 (12.0 mg) and 6 (8.0 mg) were finally obtained by preparative HPLC using MeOH-H2O (40:60, 8 mL/min) as the mobile phase.
3.4. Compound Characterization
Naucleamide G (1): orange amorphous powder; −28.8 (c 0.18, MeOH); UV (MeOH) λmax (log ε): 211 (4.11), 290 (3.52) nm; IR (KBr): νmax 3373, 1692, 1636, 1409, 1174 cm−1; HR-ESI-MS m/z: 339.1703 [M + H]+ (calcd for C20H23N2O3, 339.1709); 1H-NMR (DMSO-d6, 400 MHz) and 13C-NMR (DMSO-d6, 100 MHz), see Table 1 and Supplementary Materials.
Nauclealomide B (5): yellowish amorphous solid; −269 (c 0.25, MeOH); UV (MeOH) λmax(log ε): 211 (4.31), 243 (4.12) nm; IR (KBr) νmax: 3417, 1713, 1657, 1473, 1067 cm−1; HR-ESI-MS m/z: 553.1782 [M + Na]+ (calcd for C26H30N2O10Na, 553.1793); 1H-NMR (CD3OD, 400 MHz) and 13C-NMR (CD3OD, 100 MHz), see Table 2.
Nauclealomide C (6): yellowish amorphous solid; −117 (c 0.17, MeOH); UV (MeOH) λmax (log ε): 212 (4.33), 242 (4.10) nm; IR (KBr) νmax: 3404, 1733, 1669, 1475, 1061 cm−1; HR-ESI-MS m/z: 553.1783 [M + Na]+ (calcd for C26H30N2O10Na, 553.1793); 1H-NMR (CD3OD, 400 MHz) and 13C-NMR (CD3OD, 100 MHz), see Table 2.
3.5. Acid Hydrolysis and Sugar Analysis
Each solution of compounds 5 and 6 (2 mg each) in 2 mol/L HCl (5 mL) was hydrolyzed at 80 °C for 2 h. The solution was then evaporated under reduced pressure. The residue was dissolved in 1 mL of pyridine containing 2 mg of l-cysteine methyl ester hydrochloride, after which the reaction mixture was heated at 60 °C for 1 h. O-tolyl isothiocyanate (5 μL) was then added to the mixture and heated at 60 °C for an additional 1 h. The reaction mixture was subsequently analyzed by HPLC and detected at 250 nm. Analytical HPLC was performed on an Inertsil ODS-3 column (4.6 × 250 mm, 5 μm) at 20 °C using CH3CN-0.05% CH3COOH in H2O (25:75, 1.0 mL/min) as the mobile phase. Peaks were detected with an SPD-M20A DAD. D-Glucose (tR 16.4 min) was identified as the sugar moiety of indole alkaloids 5 and 6 by comparison with authentic samples of d-glucose (tR 16.4 min) and l-glucose (tR 14.9 min) [19].
4. Conclusions
Six indole alkaloids, including three new ones 1, 5 and 6, named naucleamide G, and nauclealomide B and C, were isolated from the n-BuOH-soluble fraction of an EtOH extract of the leaves of N. officinalis. The structures and absolute configurations of the new indole alkaloids were elucidated by means of NMR, HRESIMS, acid hydrolysis and quantum chemical CD calculation. Compounds 2–4 were isolated from the Nauclea genus for the first time and oxindole alkaloids (2–6) were rare in this genus. Thus, the findings complemented previous reports of the occurrence of indole alkaloids in the Nauclea genus and might be a useful chemotaxonomic marker of this genus. In addition, a plausible biosynthetic pathway for all the alkaloids was proposed. Compound 1 with a tetrahydro-β-carboline ring originated from condensation of l-tryptophan with secologanin to give strictosidine; nevertheless, oxindole alkaloids (2–6) might be generated through another biosynthetic route.
Supplementary Materials
The supplementary materials are available online at www.mdpi.com/1420-3049/21/8/968/s1, HRESIMS, UV, IR, and NMR spectra of indole alkaloids 1, 5 and 6.
Acknowledgments
This work was supported financially by the Postdoctoral Science Foundation of China (No. 2015M582420), the National Natural Science Foundation of China (No. 31500285), the Natural Science Foundation of Guangdong Province (No. 2015A030310529) and the Shenzhen Strategic Emerging Industry Development Project Fund (No. KQCX20140522111508785, CXZZ20150601110000604 and ZDSYS201506031617582).
Author Contributions
Zhen-Dan He initiated and coordinated the project. Long Fan, Cheng-Hui Liao, Ying-Chun Jiang, Qiang-Rong Kang and Kai Zheng performed the extraction, isolation, and structural identification of the compounds. In addition, Long Fan wrote this paper.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Editorial Committee of Chinese Herbs. Chinese Herbs; Shanghai Science and Technology Press: Shanghai, China, 1999; Volume 6, p. 456. [Google Scholar]
- Sun, J.Y.; Lou, H.X.; Xu, H.; Dai, S.J.; Liu, K. Two new indole alkaloids from Nauclea officinalis. Chin. Chem. Lett. 2007, 18, 1084–1086. [Google Scholar] [CrossRef]
- Xuan, W.D.; Chen, H.S.; Du, J.L.; Liang, S.; Li, T.Z.; Cai, D.G. Two new indole alkaloids from Nauclea officinalis. J. Asian Nat. Prod. Res. 2006, 8, 719–722. [Google Scholar] [CrossRef] [PubMed]
- Xuan, W.D.; Bian, J.; Chen, H.S. Alkaloidal constituents of Nauclea officinalis. Chin. Tradit. Herb. Drugs 2007, 38, 170–173. [Google Scholar]
- Fan, L.; Fan, C.L.; Wang, Y.; Zhang, X.Q.; Zhang, Q.W.; Zhang, J.Q.; Ye, W.C. Alkaloids from the leaves of Nauclea officinalis. Acta Pharm. Sin. 2010, 45, 747–751. [Google Scholar]
- Lin, M.; Liu, X.; Yu, D.Q.; Dou, S.Q.; Zhang, Y.J.; Li, Q.M. The structure determination of a new alkaloid-nauclefiline in Nauclea officinalis. Acta Pharm. Sin. 1985, 20, 902–905. [Google Scholar]
- Liew, S.Y.; Mukhtar, M.R.; Hadi, A.H.; Awang, K.; Mustafa, M.R.; Zaima, K.; Morita, H.; Litaudon, M. Naucline, a new indole alkaloid from the bark of Nauclea officinalis. Molecules 2012, 17, 4028–4036. [Google Scholar] [CrossRef] [PubMed]
- Sun, J.Y.; Lou, H.X.; Dai, S.J.; Xu, H.; Zhao, F.; Liu, K. Indole alkaloids from Nauclea officinalis with weak antimalarial activity. Phytochemistry 2008, 69, 1405–1410. [Google Scholar] [CrossRef] [PubMed]
- Lin, M.; Liu, X.; Yu, D.Q. Alkaloids of Nauclea officinalis. Planta Med. 1984, 50, 459–461. [Google Scholar]
- Lin, M.; Li, S.Z.; Liu, X.; Yu, D.Q. Studies on the structures of two new alkaloidal glucosides of Nauclea officinalis Pierre ex pitard. Acta Pharm. Sin. 1989, 24, 32–36. [Google Scholar]
- Xuan, W.D.; Chen, H.S.; Bian, J. A new indole alkaloid glycoside from stems of Nauclea officinalis. Acta Pharm. Sin. 2006, 41, 1064–1067. [Google Scholar]
- Xuan, W.D.; Chen, H.S.; Yuan, Z.X.; Zhu, P. Chemical constituents of Nauclea officinalis. Chin. J. Nat. Med. 2005, 3, 181–183. [Google Scholar]
- Fan, L.; Huang, X.J.; Fan, C.L.; Li, G.Q.; Wu, Z.L.; Li, S.G.; He, Z.D.; Wang, Y.; Ye, W.C. Two new oxindole alkaloid glycosides from the leaves of Nauclea officinalis. Nat. Prod. Commun. 2015, 10, 2087–2090. [Google Scholar] [PubMed]
- Shigemori, H.; Kagata, T.; Ishiyama, H.; Morah, F.; Ohsaki, A.; Kobayashi, J. Naucleamides A–E, new monoterpene indole alkaloids from Nauclea latifolia. Chem. Pharm. Bull. 2003, 51, 58–61. [Google Scholar] [CrossRef] [PubMed]
- Ma, J.; Hecht, S.M. Javaniside, a novel DNA cleavage agent from Alangium javanicum having an unusual oxindole skeleton. Chem. Commun. 2004, 40, 1190–1191. [Google Scholar] [CrossRef] [PubMed]
- Pham, V.C.; Ma, J.; Thomas, S.J.; Xu, Z.D.; Hecht, S.M. Alkaloids from Alangium javanicum and Alangium grisolleoides that mediate Cu2+-dependent DNA strand scission. J. Nat. Prod. 2005, 68, 1147–1152. [Google Scholar] [CrossRef] [PubMed]
- Wu, M.; Wu, P.; Xie, H.H.; Wu, G.J.; Wei, X.Y. Monoterpenoid indole alkaloids mediating DNA strand scission from Turpinia arguta. Planta Med. 2011, 77, 284–286. [Google Scholar] [CrossRef] [PubMed]
- Kagata, T.; Saito, S.; Shigemori, H.; Ohsaki, A.; Ishiyama, H.; Kubota, T.; Kobayashi, J. Paratunamides A–D, oxindole alkaloids from Cinnamodendron axillare. J. Nat. Prod. 2006, 69, 1517–1521. [Google Scholar] [CrossRef] [PubMed]
- Tanaka, T.; Nakashima, T.; Ueda, T.; Tomii, K.; Kouno, I. Facile discrimination of aldose enantiomers by reversed-phase HPLC. Chem. Pharm. Bull. 2007, 55, 899–901. [Google Scholar] [CrossRef] [PubMed]
- Sample Availability: Samples of the compounds are available from the authors.
© 2016 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license ( http://creativecommons.org/licenses/by/4.0/).