Ultrasound Mediated One-Pot, Three Component Synthesis, Docking and ADME Prediction of Novel 5-Amino-2-(4-chlorophenyl)-7-Substituted Phenyl-8,8a-dihydro-7H-(1,3,4)thiadiazolo(3,2-α)pyrimidine-6-carbonitrile Derivatives as Anticancer Agents
Abstract
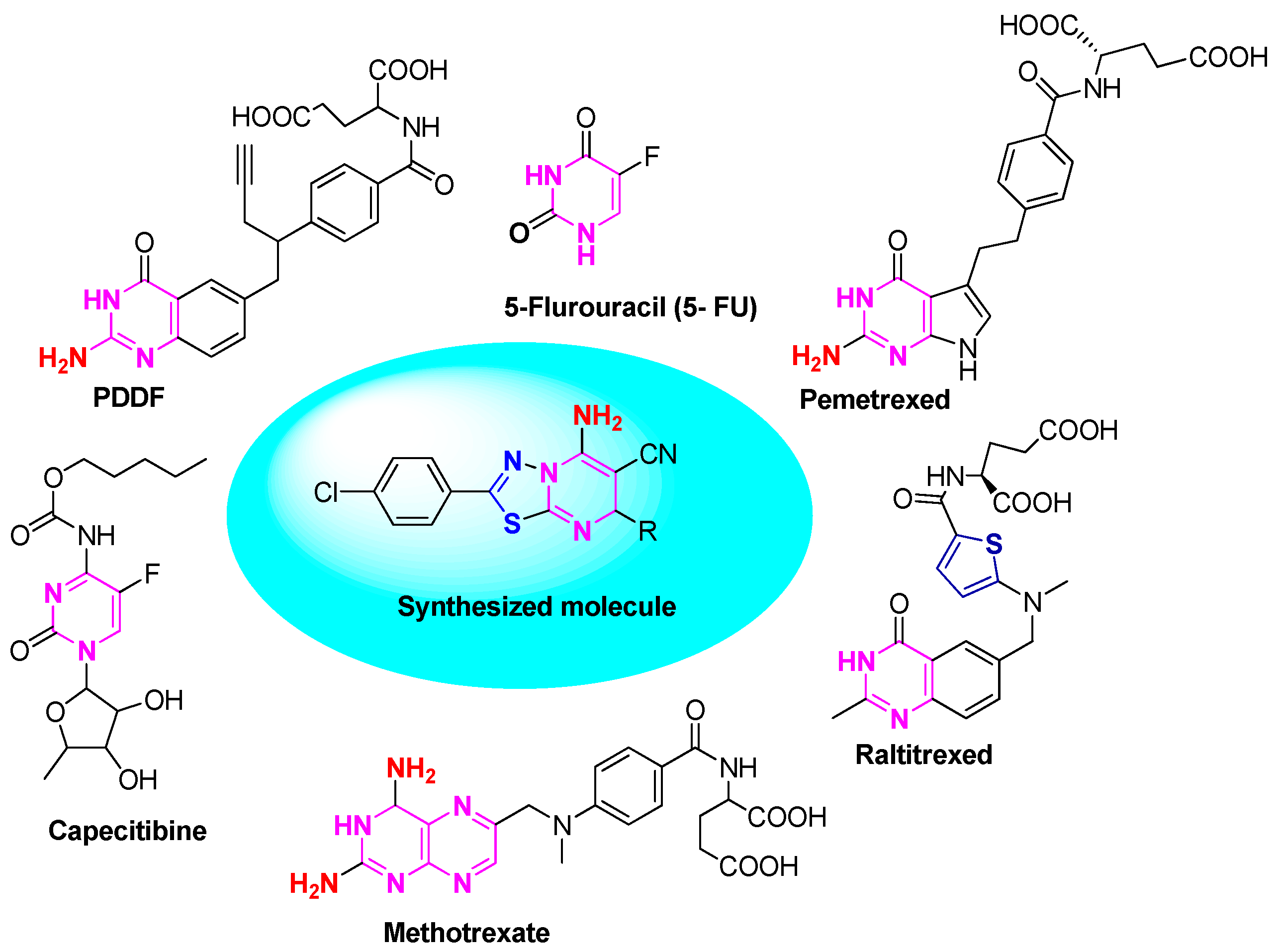
:1. Introduction
2. Result and Discussion
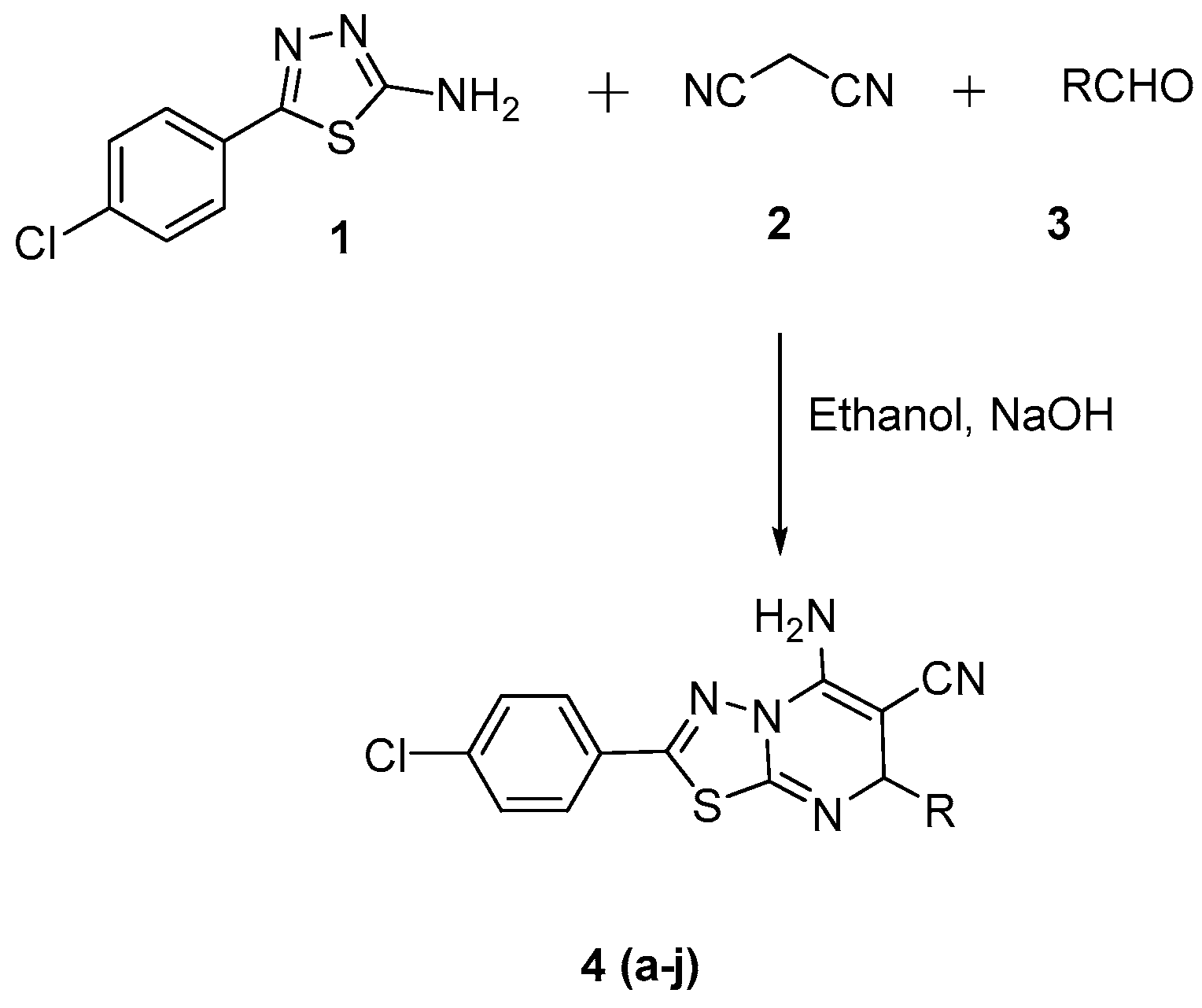
2.1. Chemistry
2.2. In Vitro Anticancer Activity
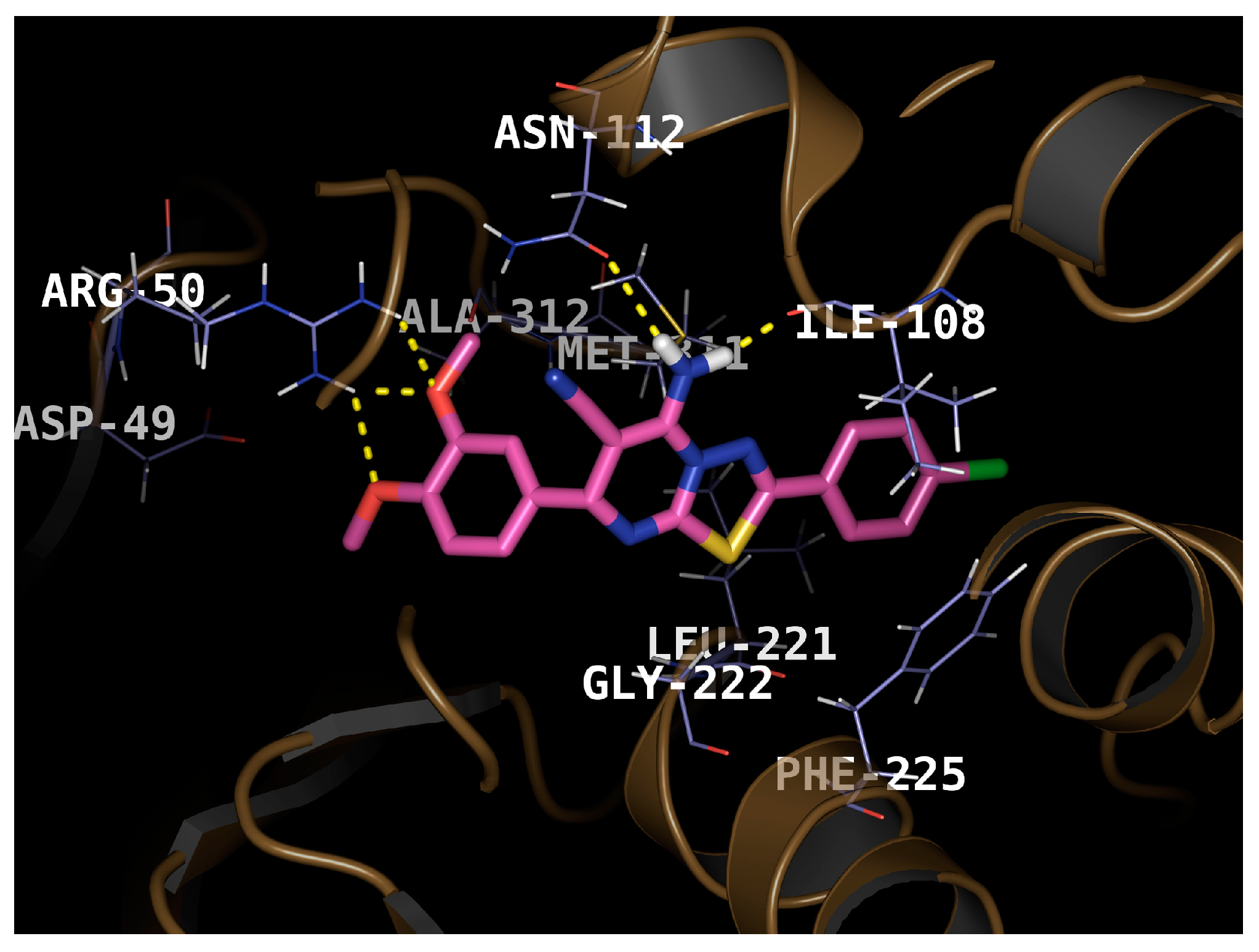
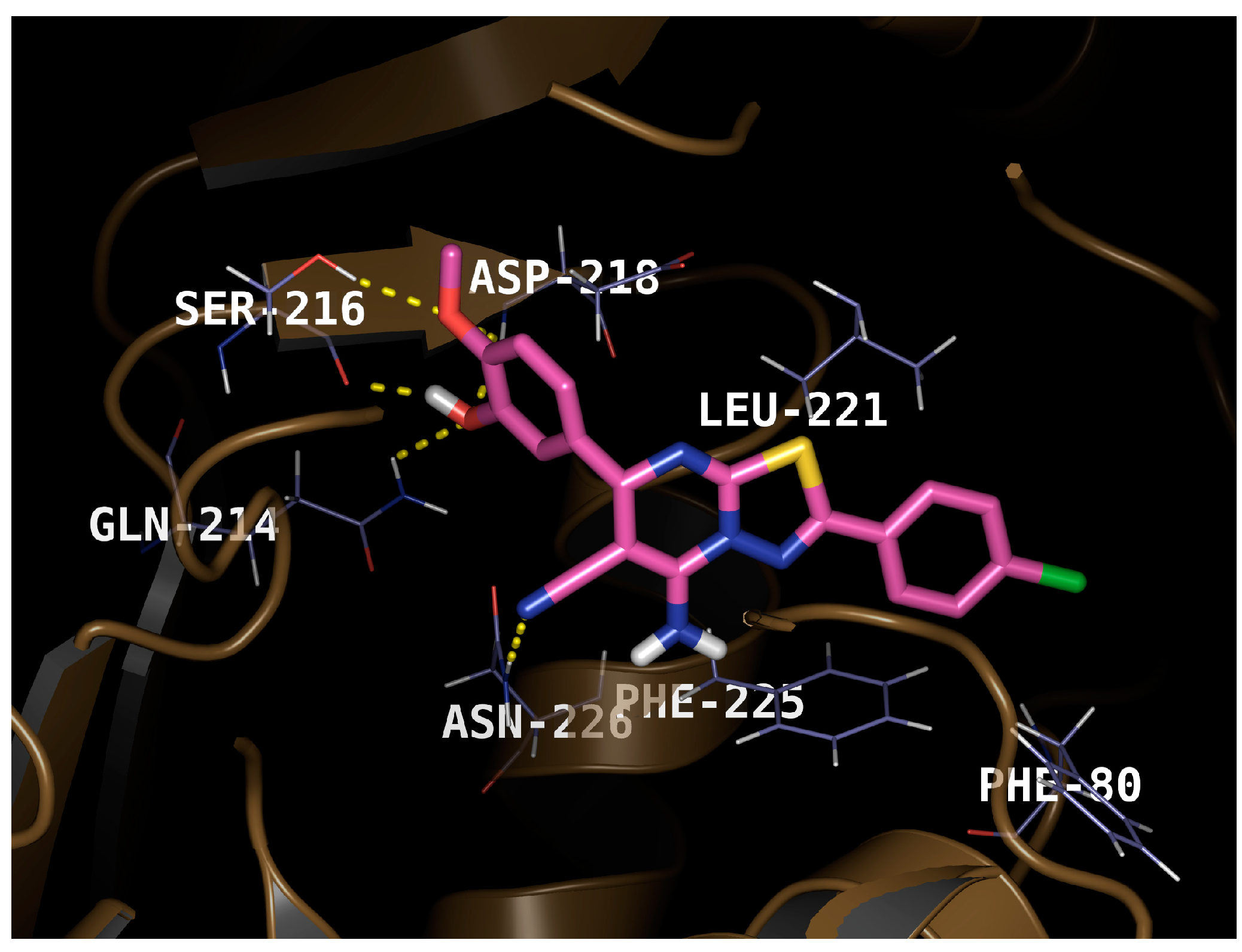
2.3. Molecular Docking
2.4. In Silico ADME Prediction
3. Materials and Methods
3.1. General
3.2. General Procedure for the Synthesis of 5-Amino-2-(4-chlorophenyl)-7-Substituted Phenyl-8,8a-dihydro-7H-(1,3,4)thiadiazolo(3,2-α)pyrimidine-6-carbonitrile Derivatives
3.3. Anticancer Screening
3.4. Molecular Docking
3.5. In Silico ADME Prediction
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Patrick, G.L. An Introduction to Medicinal Chemistry, 4th ed.; Oxford University Press Inc.: New York, NY, USA, 2009; p. 519. [Google Scholar]
- Parrino, B.; Carbone, A.; Ciancimino, C.; Spanò, V.; Montalbano, A.; Barraja, P.; Cirrincione, G.; Diana, P.; Sissi, C.; Palumbo, M.; et al. Water-soluble isoindolo(2,1-a)quinoxalin-6-imines: In vitro antiproliferative activity and molecular mechanism(s) of action. E. J. Med. Chem. 2015, 94, 149–162. [Google Scholar] [CrossRef] [PubMed]
- Spanò, V.; Parrino, B.; Carbone, A.; Montalbano, A.; Salvador, A.; Brun, P.; Vedaldi, D.; Diana, P.; Cirrincione, G.; Barraja, P. Pyrazolo(3,4-h)quinolines promising photosensitizing agents in the treatment of cancer. E. J. Med. Chem. 2015, 102, 334–351. [Google Scholar] [CrossRef] [PubMed]
- Carbone, A.; Parrino, B.; Vita, G.D.; Attanzio, A.; Spanò, V.; Montalbano, A.; Barraja, P.; Tesoriere, L.; Livrea, M.A.; Diana, P.; et al. Synthesis and Antiproliferative Activity of Thiazolyl-bis-pyrrolo(2,3-b)pyridines and Indolyl-thiazolyl-pyrrolo(2,3-c)pyridines, Nortopsentin Analogues. Mar. Drugs 2015, 13, 460–492. [Google Scholar] [CrossRef] [PubMed]
- Chan, D.C.M.; Anderson, A.C. Towards species-specific antifolates. Curr. Med. Chem. 2006, 13, 377. [Google Scholar] [CrossRef] [PubMed]
- Hawser, S.; Lociuro, S.; Islam, K. Dihydrofolate reductase inhibitors as antibacterial agents. Biochem. Pharmacol. 2006, 71, 941–948. [Google Scholar] [CrossRef] [PubMed]
- Blakley, R.L.; Benkovic, S.J. Dihydrofolate Reductase. In Folate and Pterines Chemistry and Biochemistry; Wiley: New York, NY, USA, 1984; p. 191. [Google Scholar]
- MacKenzie, R.E.; Blakley, R.L.; Benkovic, S.J. Biogenesis and interconversion of substituted tetrahydrofolates. In Folates and Pterines Chemistry and Biochemistry; Wiley: New York, NY, USA, 1984; p. 255. [Google Scholar]
- Al-Omary, F.A.; Hassan, G.S.; El-Messery, S.M.; Nagi, M.N.; Habib el, S.E.; El-Subbagh, H.I. Synthesis, biological evaluation and molecular modeling study of some new 2-heteroarylthio-quinazolin-4-ones. Eur. J. Med. Chem. 2013, 63, 33–45. [Google Scholar] [CrossRef] [PubMed]
- Hassan, G.S.; El-Messery, S.M.; Al-Omary, F.A.; Al-Rashood, S.T.; Shabayek, M.I.; Abulfadl, Y.S.; Habib, S.E.; El-Hallouty, S.M.; Fayad, W.; Mohamed, K.M.; et al. 5-(2-aminothiazol-4-yl)-4-phenyl-4H-1,2,4-triazole-3-thiols as a new class of DHFR inhibitors: Synthesis, biological evaluation and molecular modeling study. Eur. J. Med. Chem. 2013, 66, 135–145. [Google Scholar] [CrossRef] [PubMed]
- El-Subbagh, H.I.; Hassan, G.S.; El-Messery, S.M.; Al-Rashood, S.T.; Al-Omary, F.A.; Abulfadl, Y.S.; Shabayek, M.I. Benzodiazepine analogs as a new class of DHFR inhibitors: Synthesis, antitumor testing and molecular modeling study. Eur. J. Med. Chem. 2014, 74, 234–245. [Google Scholar] [CrossRef] [PubMed]
- Sagar, R.; Moon-Ju, K.; Park, S.B. An improved synthesis of pyrimidine- and pyrazole-based acyclo-C-nucleosides as carbohybrids. Tetrahedron Lett. 2008, 49, 5080–5083. [Google Scholar] [CrossRef]
- Rai, U.S.; Isloor, A.M.; Shetty, P.; Vijesh, A.M.; Prabhu, N.; Isloor, S.; Thiageeswaran, M.; Hoong-Kun, F. Novel chromeno(2,3-b)pyrimidine derivatives as potential anti-microbial agents. Eur. J. Med. Chem. 2010, 45, 2695–2699. [Google Scholar]
- Wu, J.; Yu, L.; Yang, F.; Li, J.; Wang, P.; Zhou, W.; Qin, L.; Li, Y.; Luo, J.; Yi, Z.; et al. Optimization of 2-(3-(arylalkyl amino carbonyl) phenyl)-3-(2-methoxyphenyl)-4-thiazolidinone derivatives as potent antitumor growth and metastasis agents. Eur. J. Med. Chem. 2014, 74, 340–357. [Google Scholar] [CrossRef] [PubMed]
- Barraja, P.; Caracausi, L.; Diana, P.; Montalbano, A.; Carbone, A.; Salvador, A.; Brun, P.; Castagliuolo, I.; Tisi, S.; Dall’Acqua, F.; et al. Pyrrolo(3,2-h)quinazolines as Photochemotherapeutic Agents. Chem. Med. Chem. 2011, 6, 1238–1248. [Google Scholar] [CrossRef] [PubMed]
- Meunier, B. Hybrid molecules with a dual mode of action: Dream or reality? Acc. Chem. Res. 2008, 41, 69–77. [Google Scholar] [CrossRef] [PubMed]
- Havrylyuk, D.; Zimenkovsky, B.; Vasylenko, O.; Gzella, A.; Lesyk, R. Synthesis of new 4-thiazolidinone-, pyrazoline-, and isatin-based conjugates with promising antitumor activity. J. Med. Chem. 2012, 55, 8630–8641. [Google Scholar] [CrossRef] [PubMed]
- Taher, A.T.; Georgey, H.H.; El-Subbagh, H.I. Novel 1,3,4-heterodiazole analogues: Synthesis and in vitro antitumor activity. Eur. J. Med. Chem. 2012, 47, 445–451. [Google Scholar] [CrossRef] [PubMed]
- Coburn, R.A.; Glennon, R.A. Mesoionic purinone analogues synthesis and in vitro antibacterial properties of mesoionic thiazolo(3,2-a)pyrimidine-5,7-diones and mesoionic 1,3,4-thiadiazolo(3,2-a)pyrimidine-5,7-diones. J. Pharm. Sci. 1973, 62, 1785–1789. [Google Scholar] [CrossRef] [PubMed]
- Mahran, M.A.; El-Sherbeny, M.A.; El-Obaid, A.M.A.; Badria, F.A. Heterocyclic systems containing pyrimidine nucleus as potential antimicrobial and antitumor agents. Alexandria J. Pharm. Sci. 1998, 12, 39–44. [Google Scholar]
- Coburn, R.A.; Glennon, R.A.; Chmielewicz, Z.F. Mesoionic purinone analogues in vitro antibacterial activity of mesoionic 1,3,4-thiadiazolo(3,2-a)pyrimidine-5,7-diones. J. Med. Chem. 1974, 17, 1025–1027. [Google Scholar] [CrossRef] [PubMed]
- Zhao, B.; Xu, Y.; Deng, Q.G.; Liu, Z.; Wang, L.Y.; Gao, Y. One-pot, three component synthesis of novel 5H-(1,3,4)thiadiazolo(3,2-a)pyrimidine-6-carboxylate derivatives by microwave irradiation. Tetrahedron Lett. 2014, 55, 4521–4524. [Google Scholar] [CrossRef]
- Anastas, P.T.; Warner, J.C. Green Chemistry, Theory and Practice; Oxford University Press: Oxford, UK, 1998. [Google Scholar]
- Alan, R.K.; Kenneth, C.C.; Thomas, H.M.; Curtis, W.C.; Amir, B.I. Synthesis and physicochemical properties of thiadiazolo(3,2-a)pyrimidinesulfonamides and thiadiazolo(3,2-a)triazinesulfonamides as candidates for topically effective carbonic anhydrase inhibitors. J. Med. Chem. 1987, 30, 2059–2062. [Google Scholar]
- Salimov, T.M.; Kukaniev, M.A.; Sattorov, I.T.; Osimov, D.M. Synthesis and antimicrobial activity of 2-bromo-7-methyl-5-oxo-5H-1,3,4- thiadiazolo(3,2-a)pyrimidine. Pharm. Chem. J. 2005, 39, 311–312. [Google Scholar] [CrossRef]
- Vichai, V.; Kirtikara, K. Sulforhodamine B colorimetric assay for cytotoxicity screening. Nat. Protoc. 2006, 1, 1112–1116. [Google Scholar] [CrossRef] [PubMed]
- Friesner, R.A.; Murphy, R.B.; Repasky, M.P.; Frye, L.L.; Greenwood, J.R.; Halgren, T.A.; Sanschagrin, P.C.; Mainz, D.T. Extra precision glide: Docking and scoring incorporating a model of hydrophobic enclosure for protein ligand complexes. J. Med. Chem. 2006, 49, 6177–6196. [Google Scholar] [CrossRef] [PubMed]
- Sarkate, A.P.; Shinde, D.B. Synthesis and docking studies of 2-(nitrooxy)-ethyl-2-(substituted-2,5-diphenyl-oxazole)-acetate as anti-inflammatory, analgesic and nitric oxide releasing agents. Int. J. Pharm. Pharm. Sci. 2015, 7, 128–135. [Google Scholar]
- Sarkate, A.P.; Shinde, D.B. Synthesis and Docking studies of ethyl 4-(4-((2-(nitrooxy)ethoxy)carbonyl)phenyl)-2-substituted-6-substitutedphenyl-1,2,3,4-tetrahydropyrimidine-5-carboxylate as anti-inflammatory, analgesic and nitric oxide releasing agents. Res. J. Pharm. Bio. Chem. Sci. 2015, 6, 391–404. [Google Scholar]
- Sarkate, A.P.; Lokwani, D.K.; Bahekar, S.S.; Shinde, D.B. Synthesis and docking studies of 2-(nitrooxy) ethyl-4-(2-(substitutedphenyl)-4-(substitutedphenyl)-1H-imidazol-1-yl)benzoate as anti-inflammatory, analgesic and nitric oxide releasing agents. Int. J. Pharm. Pharm. Sci. 2015, 7, 197–205. [Google Scholar]
- Lokwani, D.K.; Azad, R.; Sarkate, A.P.; Shinde, D.B. Structure Based Library Design (SBLD) for New 1, 4-dihydropyrimidine Scaffold as simultaneous COX-1/COX-2 and 5-LOX Inhibitors. Biorg. Med. Chem. 2015, 23, 4533–4543. [Google Scholar] [CrossRef] [PubMed]
- Ertl, P.; Rohde, B.; Selzer, P. Fast calculation of molecular polar surface area as a sum of fragment based contributions and its application to the prediction of drug transport properties. J. Med. Chem. 2000, 43, 3714–3717. [Google Scholar] [CrossRef] [PubMed]
- Sample Availability: Samples of the compounds are not available from the authors.
| Entry | Catalyst | Amount (% mol) | Solvent | Method A Conventional | Method B Ultrasound | ||
|---|---|---|---|---|---|---|---|
| Time (h) | Yield (%) | Time (h) | Yield (%) | ||||
| 1 | No catalyst | - | EtOH | 9 | - | 2 | - |
| 2 | NaOH | 30 | EtOH | 9 | 70 | 2 | 89 |
| 3 | NaOH | 20 | EtOH | 9 | 70 | 2 | 89 |
| 4 | NaOH | 20 | H2O | 9 | - | 2 | - |
| 5 | NaOH | 20 | MeOH | 11 | 55 | 2.30 | 60 |
| 6 | NaOH | 20 | CH3CN | 11 | 40 | 2.30 | 50 |
| 7 | NaOH | 20 | DMF | 12 | 40 | 2.30 | 55 |
| 8 | NaOH | 10 | EtOH | 13 | 60 | 2.45 | 65 |
| 9 | NaOH | 5 | EtOH | 15 | 50 | 3.30 | 60 |
| Compound | R | Conventional | Ultrasound | ||
|---|---|---|---|---|---|
| Time (h) | Yield (%) | Time (h) | Yield (%) | ||
| 4a | 4-chlorophenyl | 7 | 70 | 1 | 89 |
| 4b | 2-chlorophenyl | 7.30 | 68 | 1.30 | 85 |
| 4c | 3-chlorophenyl | 7.30 | 65 | 1.30 | 85 |
| 4d | 4-flurophenyl | 7 | 62 | 1.30 | 80 |
| 4e | 4-methoxyphenyl | 8 | 58 | 2 | 75 |
| 4f | 3,4,5-methoxyphenyl | 9 | 58 | 2 | 78 |
| 4g | 3,4-methoxyphenyl | 9 | 55 | 2 | 75 |
| 4h | Phenyl | 7 | 60 | 1 | 80 |
| 4i | 3-hydroxy-4 methoxyphenyl | 9 | 60 | 2 | 82 |
| 4j | furan-2-yl | 8 | 45 | 1.30 | 65 |
| Compound | GI50 μM | |||
|---|---|---|---|---|
| MCF-7 | K-562 | HeLa | PC-3 | |
| 4a | 88.5 | 47.9 | 56.2 | 38.9 |
| 4b | 43.8 | 57.1 | 54.3 | 37.9 |
| 4c | 55.0 | 60.1 | 55.7 | 38.4 |
| 4d | 80.6 | >100 | 58.1 | 30.2 |
| 4e | 38.9 | 54.2 | 43.8 | 26.7 |
| 4f | 38.3 | 58.1 | 48.6 | 25.4 |
| 4g | 34.8 | 54.3 | 47.9 | 25.3 |
| 4h | 38.9 | 58.3 | 38.7 | 34.7 |
| 4i | 32.7 | 55.3 | 34.3 | 28.9 |
| 4j | 82.5 | >100 | 60.9 | 55.3 |
| 5-FU | 32.18 | 47.03 | 43.71 | 12.00 |
| Compound | G-Score | Compound | G-Score |
|---|---|---|---|
| 4a | −4.65 | 4g | −5.59 |
| 4b | −5.59 | 4h | −5.38 |
| 4c | −5.24 | 4i | −7.17 |
| 4d | −5.14 | 4j | −4.98 |
| 4e | −4.74 | 5-FU | −4.75 |
| 4f | −5.14 |
| Compound | MW a | Percentage of Absorption (% ABS) b | logP o/w c (–2.0 to 6.5) | n-ON d (<10) | n-OHNH e (<5) | Polar Surface Area (PSA) f (7–200.0) | Number of Violations of Rule of 5 (<5) | Toxicity |
|---|---|---|---|---|---|---|---|---|
| 4a | 400 | 100 | 4.57 | 4 | 2 | 74.81 | 0 | N |
| 4b | 400 | 100 | 4.42 | 4 | 2 | 74.87 | 0 | N |
| 4c | 400 | 100 | 4.57 | 4 | 2 | 74.81 | 0 | N |
| 4d | 383.8 | 100 | 4.30 | 4 | 2 | 74.81 | 0 | N |
| 4e | 395.8 | 100 | 4.19 | 4.75 | 2 | 83.10 | 0 | N |
| 4f | 455.9 | 100 | 4.47 | 6.25 | 2 | 95.95 | 0 | N |
| 4g | 425.8 | 100 | 4.36 | 5.5 | 2 | 88.02 | 0 | N |
| 4h | 365.8 | 100 | 4.01 | 4 | 2 | 74.81 | 0 | N |
| 4i | 411.8 | 87.50 | 3.46 | 5.5 | 3 | 104.7 | 0 | N |
| 4j | 355.8 | 94.19 | 3.12 | 4.5 | 2 | 83.51 | 0 | N |
| 5-FU | 130.0 | 66.30 | −0.89 | 3.5 | 2 | 87.56 | 0 | N |
© 2016 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tiwari, S.V.; Seijas, J.A.; Vazquez-Tato, M.P.; Sarkate, A.P.; Lokwani, D.K.; Nikalje, A.P.G. Ultrasound Mediated One-Pot, Three Component Synthesis, Docking and ADME Prediction of Novel 5-Amino-2-(4-chlorophenyl)-7-Substituted Phenyl-8,8a-dihydro-7H-(1,3,4)thiadiazolo(3,2-α)pyrimidine-6-carbonitrile Derivatives as Anticancer Agents. Molecules 2016, 21, 894. https://doi.org/10.3390/molecules21080894
Tiwari SV, Seijas JA, Vazquez-Tato MP, Sarkate AP, Lokwani DK, Nikalje APG. Ultrasound Mediated One-Pot, Three Component Synthesis, Docking and ADME Prediction of Novel 5-Amino-2-(4-chlorophenyl)-7-Substituted Phenyl-8,8a-dihydro-7H-(1,3,4)thiadiazolo(3,2-α)pyrimidine-6-carbonitrile Derivatives as Anticancer Agents. Molecules. 2016; 21(8):894. https://doi.org/10.3390/molecules21080894
Chicago/Turabian StyleTiwari, Shailee V., Julio A. Seijas, M. Pilar Vazquez-Tato, Aniket P. Sarkate, Deepak K. Lokwani, and Anna Pratima G. Nikalje. 2016. "Ultrasound Mediated One-Pot, Three Component Synthesis, Docking and ADME Prediction of Novel 5-Amino-2-(4-chlorophenyl)-7-Substituted Phenyl-8,8a-dihydro-7H-(1,3,4)thiadiazolo(3,2-α)pyrimidine-6-carbonitrile Derivatives as Anticancer Agents" Molecules 21, no. 8: 894. https://doi.org/10.3390/molecules21080894
APA StyleTiwari, S. V., Seijas, J. A., Vazquez-Tato, M. P., Sarkate, A. P., Lokwani, D. K., & Nikalje, A. P. G. (2016). Ultrasound Mediated One-Pot, Three Component Synthesis, Docking and ADME Prediction of Novel 5-Amino-2-(4-chlorophenyl)-7-Substituted Phenyl-8,8a-dihydro-7H-(1,3,4)thiadiazolo(3,2-α)pyrimidine-6-carbonitrile Derivatives as Anticancer Agents. Molecules, 21(8), 894. https://doi.org/10.3390/molecules21080894









