Four New Dicaffeoylquinic Acid Derivatives from Glasswort (Salicornia herbacea L.) and Their Antioxidative Activity
Abstract
:1. Introduction
2. Results and Discussion
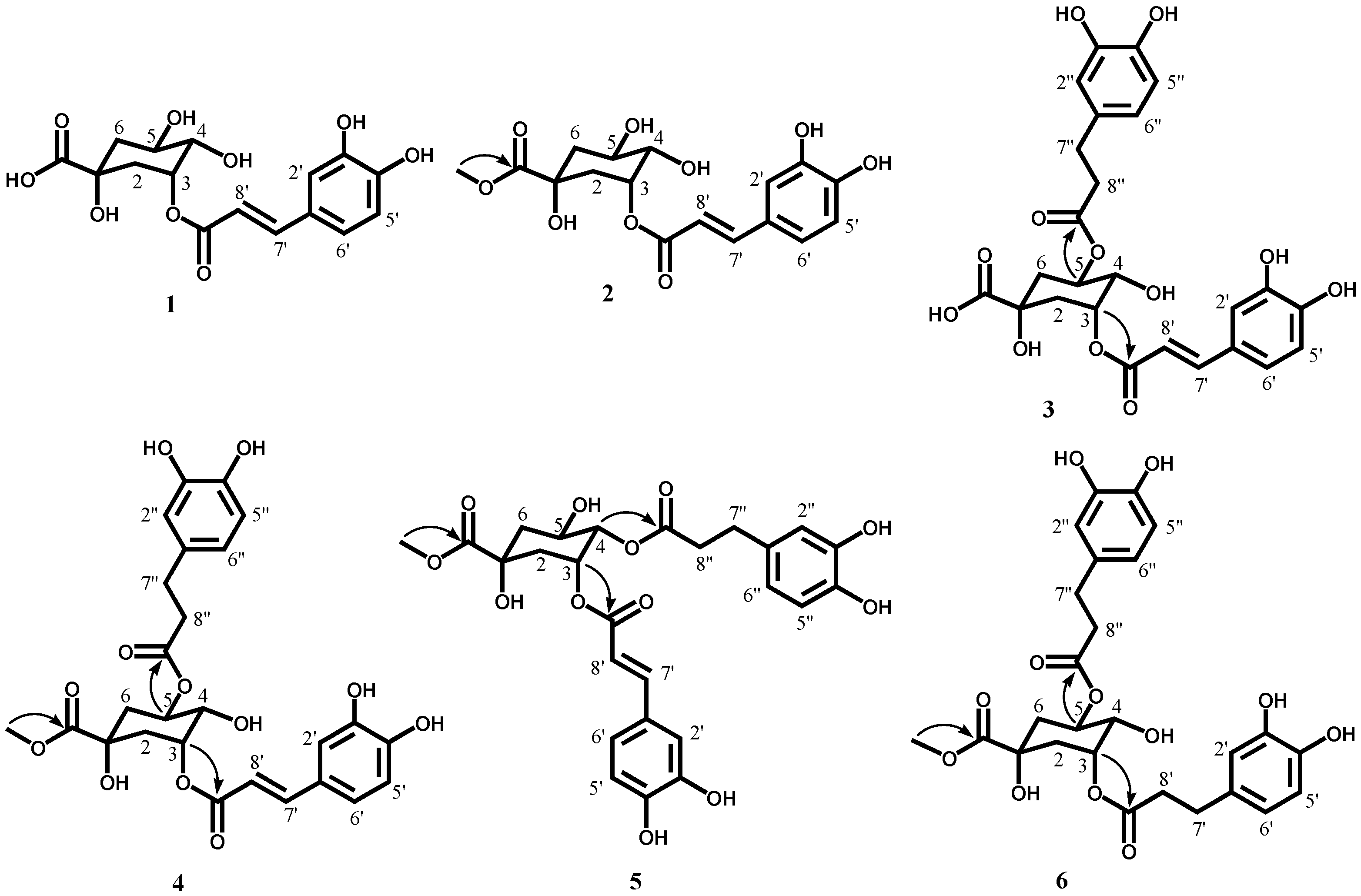
2.1. Isolation and Structural Determination of Antioxidative Compounds
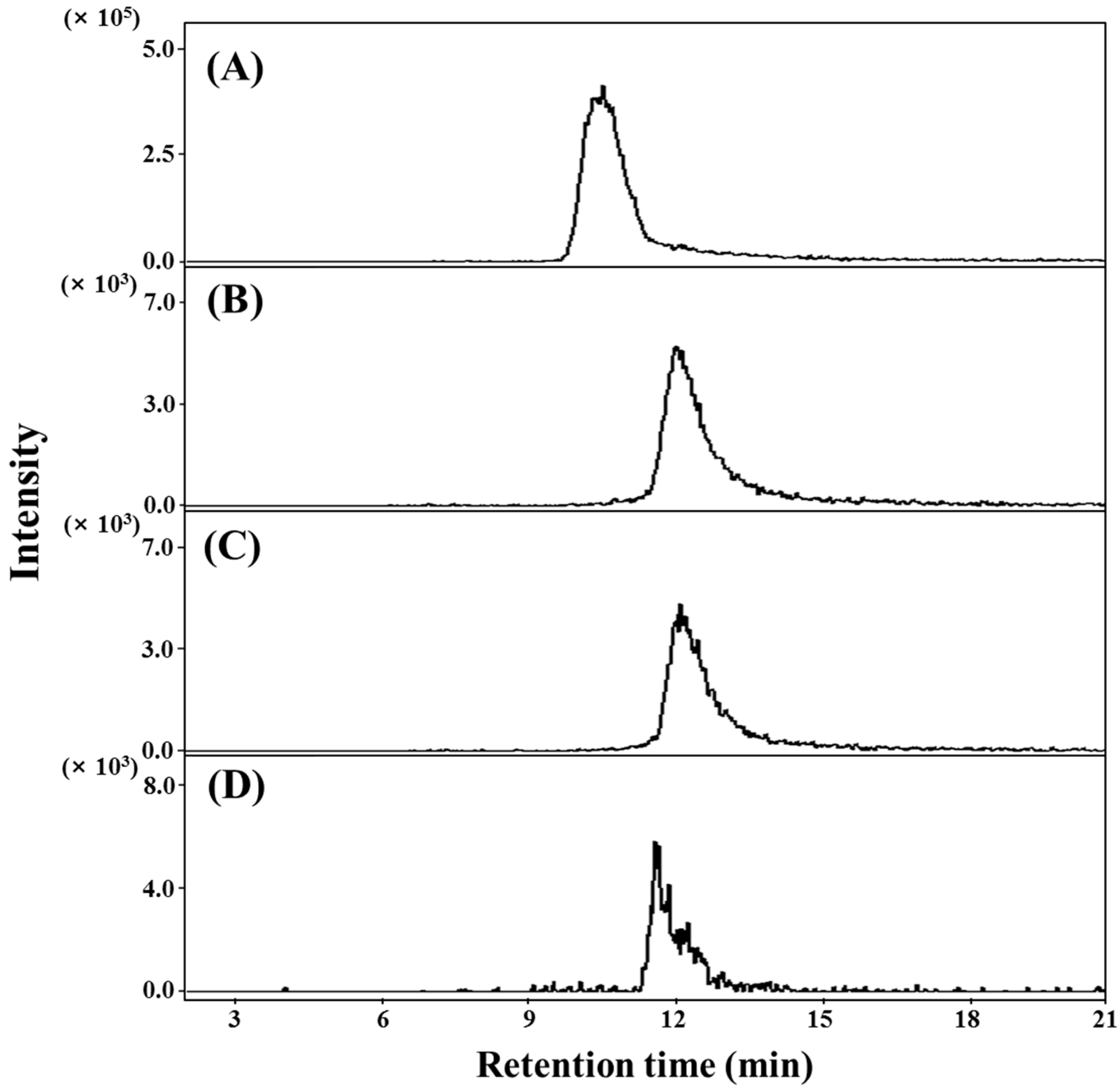
2.2. Qualification and Quantitation of 3–6 in the Aerial Parts of S. herbacae
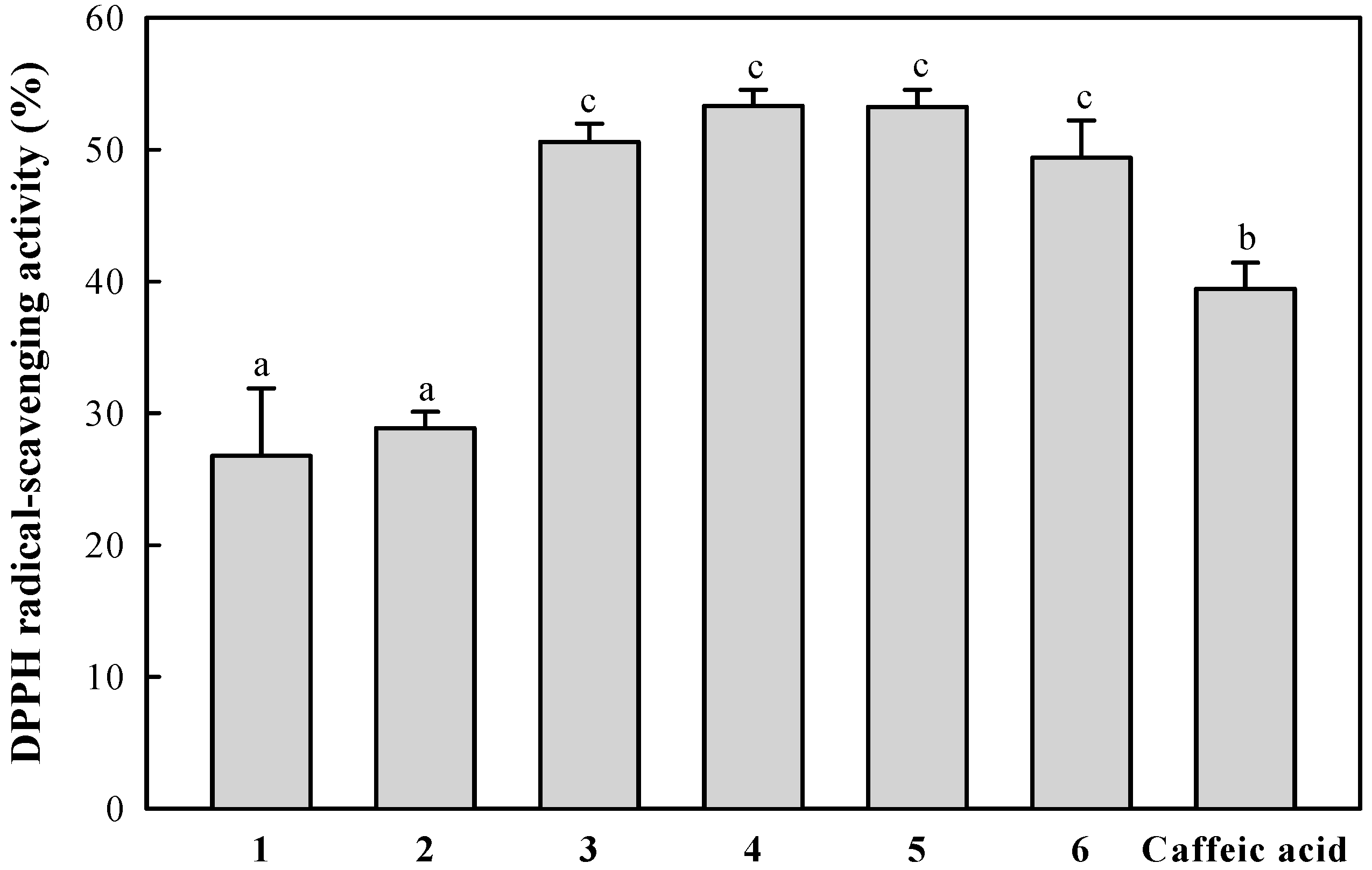
2.3. DPPH Radical-Scavenging Activity of the Isolated Compounds
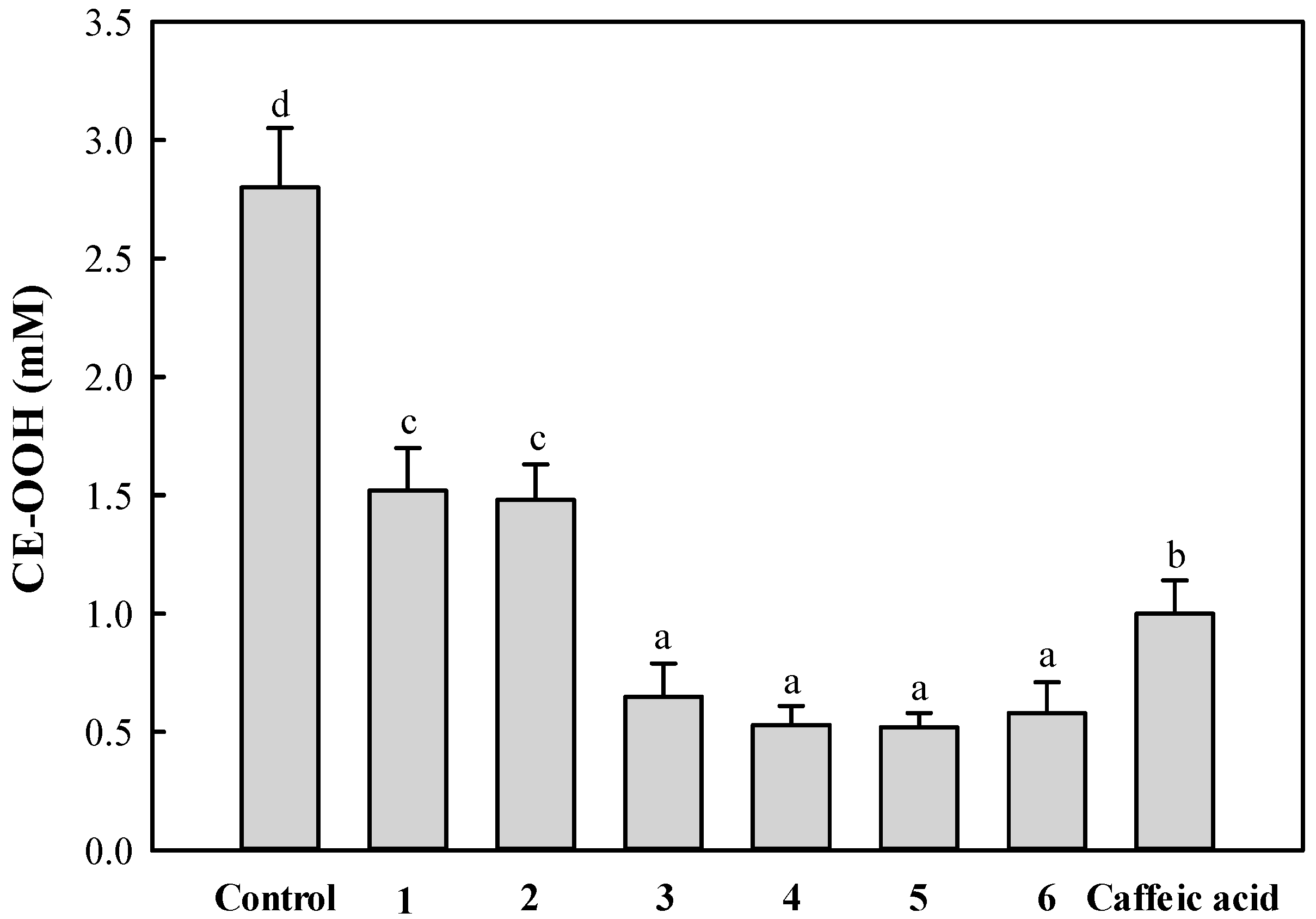
2.4. Inhibitory Effect of the Isolated Compounds on Copper Ion-Induced Rat Plasma Oxidation
3. Experimental Section
3.1. General Experimental Procedures
3.2. Materials and Chemicals
3.3. Extraction and Partition
3.4. Isolation of the EtOAc Fraction
3.5. HPLC ESI MS/MS Analysis of Four Dicaffeoylquinic Acid Derivatives Identified in S. herbacea
3.6. Assay of DPPH Radical-Scavenging
3.7. Determination of the Inhibitory Effect of the Isolated Compounds against Copper Ion-Induced Rat Plasma Oxidation
3.8. Statistical Analysis
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Seifried, H.E.; Anderson, D.E.; Fisher, E.I.; Milner, J.A. A review of the interaction among dietary antioxidants and reactive oxygen species. J. Nutr. Biochem. 2007, 11, 567–579. [Google Scholar] [CrossRef] [PubMed]
- Singh, R.B.; Choudhury, J.; Meester, F.D.; Wilson, D.W. Development of the Mediterranean soup for enteral nutrition and for prevention of cardiovascular diseases. Open Nutraceuticals J. 2012, 5, 90–98. [Google Scholar] [CrossRef]
- Goodman, M.; Bostick, R.M.; Kucuk, O.; Jones, D.P. Clinical trials of antioxidants as cancer prevention agents: Past, present, and future. Free Radic. Biol. Med. 2011, 51, 1068–1084. [Google Scholar] [CrossRef] [PubMed]
- Polimeni, A.; Marotta, F.; Celep, G.S.; Cabeca, A. Novel concepts on functional foods and nutrigenomics in healthy aging and chronic diseases: A review of fermented papaya preparation research progress. Funct. Foods Health Dis. 2012, 2, 120–136. [Google Scholar]
- Glenn, E.P.; Brown, J.J. Salt tolerance and crop potential of halophytes. Crit. Rev. Plant Sci. 1999, 18, 227–255. [Google Scholar] [CrossRef]
- Ksouri, R.; Ksouri, W.M.; Jallali, I.; Debez, A.; Magne, C.; Hiroko, I.; Abdelly, C. Medicinal halophytes: Potent source of health promoting biomolecules with medical, nutraceutical and food applications. Crit. Rev. Biotechnol. 2012, 32, 289–326. [Google Scholar] [CrossRef] [PubMed]
- Parida, A.K.; Das, A.B. Salt tolerance and salinity effects on plants. Ecotoxicol. Environ. Saf. 2005, 60, 324–349. [Google Scholar] [CrossRef] [PubMed]
- Rhee, M.H.; Park, H.J.; Cho, J.Y. Salicornia herbacea: Botanical, chemical and pharmacological review of halophyte marsh plant. J. Med. Plants Res. 2009, 3, 548–555. [Google Scholar]
- Park, I.B.; Park, J.W.; Lee, Y.J.; Shin, G.W.; Kim, H.S.; Jo, Y.C. Quality characteristic of glasswort (Salicornia herbacea L.) fermented by Bacillus substilis. J. Food Sci. Nutr. 2009, 38, 902–908. [Google Scholar]
- Ha, B.J.; Lee, S.H.; Kim, H.J.; Lee, J.Y. The role of Salicornia herbacea in ovariectomy-induced oxidative stress. Biol. Pharm. Bull. 2006, 29, 1305–1309. [Google Scholar] [CrossRef] [PubMed]
- Jung, B.M.; Park, J.A.; Bae, S.J. Growth inhibitory and quinone reductase induction activities of Salicornia herbacea L. fractions on human cancer cell lines in vitro. J. Food Sci. Nutr. 2008, 37, 148–153. [Google Scholar] [CrossRef]
- Kim, K.R.; Choi, J.H.; Woo, M.H.; Kim, Y.H.; Choi, S.W. Effect of enzymatic hydrolysate of hamcho (Salicornia herbacea) on blood glucose and serum lipid composition in streptozotocin-induced diabetic. J. Food Sci. Nutr. 2008, 37, 170–176. [Google Scholar]
- Im, S.A.; Kim, K.J.; Lee, C.K. Immunomodulatory activity of polysaccharides isolation from Salicornia herbacea. Int. Immunopharm. 2006, 6, 1451–1458. [Google Scholar] [CrossRef] [PubMed]
- Lee, Y.S.; Lee, S.H.; Shin, K.H.; Kim, B.K.; Lee, S. Constituents of the halophyte Salicornia herbacea. Arch. Pharm. Res. 2004, 27, 1034–1036. [Google Scholar] [CrossRef] [PubMed]
- Chung, Y.C.; Choi, J.H.; Oh, K.N.; Chun, H.K. Tungtungmadic acid isolated from Salicornia herbacea suppresses the progress of carbon tetrachloride-induced hepatic fibrosis in mice. Off. J. Korean Soc. Toxicol. 2006, 22, 267–273. [Google Scholar]
- Kong, C.S.; Kim, Y.A.; Kim, M.M.; Park, J.S.; Kim, S.K.; Lee, B.J.; Nam, T.J.; Seo, Y.W. Antioxidant activity and inhibition of MMP-9 by isorhamnetin and quercetin 3-O-β-d-glucopyranosides isolated from Salicornia herbacea in HT1080 cells. Food Sci. Biotechnol. 2008, 17, 983–989. [Google Scholar]
- Kim, U.A.; Kong, C.S.; Lee, J.I.; Kim, H.J.; Park, H.Y.; Lee, H.S.; Lee, C.H.; Seo, Y.W. Evaluation of novel antioxidant triterpenoid saponins from the halophyte Salicornia herbacea. Bioorg. Med. Chem. Lett. 2012, 22, 4318–4322. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Zhang, M.; Zhao, Y.; Wang, H.; Liu, T.; Xin, Z. Pentadecyl ferulate, a potent antioxidant and antiproliferative agent from the halophyte Salicornia herbacea. Food Chem. 2013, 141, 2066–2074. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.Y.; Cho, J.Y.; Ma, Y.K.; Park, K.Y.; Lee, S.H.; Ham, K.S.; Lee, H.J.; Park, K.H.; Moon, J.H. Dicaffeoylquinic acid derivatives and flavonoid glucosides from glasswort (Salicornia herbacea L.) and their antioxidative activity. Food Chem. 2011, 125, 55–62. [Google Scholar] [CrossRef]
- Lee, Y.G.; Cho, J.Y.; Kim, C.M.; Lee, S.H.; Kim, W.S.; Jeon, T.I.; Park, K.H.; Moon, J.H. Coumaroyl quinic acid derivatives and flavonoids from immature fruits of pear (Pyrus pyrifolia Nakai). Food Sci. Biotechnol. 2013, 22, 803–810. [Google Scholar] [CrossRef]
- Yamamoto, Y.; Niki, E. Presence of cholesteryl ester hydroperoxides in human blood plasma. Biochem. Biophys. Res. Commun. 1989, 165, 988–993. [Google Scholar] [CrossRef]
- Leitinger, N. Cholesteryl ester oxidation products in atherosclerosis. Mol. Asp. Med. 2003, 24, 239–250. [Google Scholar] [CrossRef]
- Upston, J.M.; Niu, X.; Brown, A.J.; Mashima, R.; Wang, H.; Senthilmohan, R.; Kettle, A.J.; Dean, R.T.; Stocker, R. Disease stage-dependent accumulation of lipid and protein oxidation products in human atherosclerosis. Am. J. Pathol. 2002, 160, 701–710. [Google Scholar] [CrossRef]
- Maruta, Y.; Kawabata, J.; Niki, R. Antioxidative caffeoylquinic acid derivatives in the roots of burdock (Arctium lappa L.). J. Agric. Food Chem. 1995, 43, 2592–2595. [Google Scholar] [CrossRef]
- Mishima, S.; Inoh, Y.; Narita, Y.; Ohta, S.; Sakamoto, T.; Araki, Y.; Suzuki, K.; Akao, Y.; Nozawa, Y. Identification of caffeoylquinic acid derivatives from Brazilian propolis as constituents involved in induction of granulocytic differentiation of HL-60 cells. Bioorg. Med. Chem. 2005, 13, 5814–5818. [Google Scholar] [CrossRef] [PubMed]
- Lee, C.S.; Lee, S.A.; Kim, Y.J.; Seo, S.J.; Lee, M.W. 3,4,5-Tricaffeoyquinic acid inhibits tumor necrosis factor-α-stimulated production of inflammatory mediators in keratinocytes via suppression of AKT- and NF-КB pathway. Int. Immunopharm. 2011, 11, 1715–1723. [Google Scholar] [CrossRef] [PubMed]
- Rice-Evans, C.A.; Miller, N.J.; Paganga, G. Structure-antioxidant activity relationships of flavonoids and phenolic acids. Free Radic. Biol. Med. 1996, 20, 933–956. [Google Scholar] [CrossRef]
- Burda, S.; Oleszek, W. Antioxidant and antiradical activities of flavonoids. J. Agric. Food Chem. 2001, 49, 2774–2779. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.Y.; Cho, J.Y.; Lee, K.D.; Kim, S.J.; Choi, K.C.; Ham, K.S.; Park, K.H.; Moon, J.H. Change of phenylpropanoic acid and flavonol contents in different growth stage of glasswort (Salicornia herbacea L.). Food Sci. Biotechnol. 2014, 23, 685–691. [Google Scholar] [CrossRef]
- Takao, T.; Kitatani, F.; Watanabe, N.; Yagi, A.; Sakata, K. A simple screening method for antioxidants and isolation of several antioxidants produced by marine bacteria from fish and shellfish. Biosci. Biotechnol. Biochem. 1994, 58, 1780–1783. [Google Scholar] [CrossRef]
- Yamaguchi, T.; Hitoshi, T.; Matoba, T.; Terao, J. HPLC method for evaluation of the free radical-scavenging activity of foods by using 1,1-diphenyl-2-picrylhydrazyl. Biosci. Biotechnol. Biochem. 1996, 62, 1201–1204. [Google Scholar] [CrossRef] [PubMed]
- Arai, H.; Terao, J.; Abdalla, D.S.P.; Suzuki, T.; Takama, K. Coulometric detection in high-performance liquid chromatographic analysis of cholesteryl ester hydroperoxides. Free Radic. Biol. Med. 1996, 20, 365–371. [Google Scholar] [CrossRef]
- Sample Availability: Samples of the compounds 1–6 are available from the authors.
| Position | δH (int., multi., J in Hz) | |||
|---|---|---|---|---|
| 3 | 4 | 5 | 6 | |
| 2ax | 2.27 (1H, dd, 14.0, 4.0) | 2.26 (1H, dd, 14.0, 4.0) | 2.24 (1H, dd, 12.0, 2.5) | 2.19 (1H, dd, 14.0, 3.5) |
| 2eq | 2.14 (1H, dd, 14.0, 7.5) | 2.13 (1H, dd, 14.0, 7.5) | 2.08 (1H, dd, 12.0, 7.5) | 2.05 (1H, dd, 14.0, 7.5) |
| 3 | 5.37 (1H, m) | 5.32 (1H, m) | 5.36 (1H, m) | 5.18 (1H, m) a |
| 4 | 3.88 (1H, dd, 7.5, 3.0) | 3.86 (1H, dd, 7.5, 3.0) | 5.02 (1H, dd, 7.5, 3.5) | 3.79 (1H, dd, 7.2, 3.0) |
| 5 | 5.29 (1H, m) | 5.25 (1H, m) | 4.26 (1H, m) | 5.18 (1H, m) a |
| 6ax | 2.11 (1H, br. d, 5.5) | 2.10 (2H, br. d, 5.5) | 2.15 (1H, dt, 9.0, 2.0) | 2.06 (2H, br. d, 5.0) |
| 6eq | 2.11 (1H, br. d, 5.5) | 2.09 (1H, dt, 12.0, 2.0) | ||
| -OCH3 | - | 3.72 (3H, s) | 3.69 (3H, s) | 3.72 (3H, s) |
| 2′ | 7.06 (1H, d, 1.8) | 7.06 (1H, d, 2.0) | 7.03 (1H, d, 2.0) | 6.66 (1H, d, 2.4) |
| 5′ | 6.77 (1H, d, 7.8) | 6.77 (1H, d, 8.0) | 6.78 (1H, d, 8.0) | 6.67 (1H, d, 8.4) |
| 6′ | 6.96 (1H, dd, 8.0, 1.8) | 6.96 (1H, dd, 8.0, 2.0) | 6.95 (1H, dd, 8.0, 2.0) | 6.55 (1H, dd, 8.4, 2.4) |
| 7′ | 7.61 (1H, d, 15.5) | 7.61 (1H, d, 16.0) | 7.50 (1H, d, 15.5) | 2.79 (2H, m) |
| 8′ | 6.34 (1H, d, 15.5) | 6.32 (1H, d, 16.0) | 6.16 (1H, d, 15.5) | 2.60 (2H, m) |
| 2″ | 6.66 (1H, br. s) | 6.64 (1H, d, 2.0) | 6.62 (1H, d, 2.0) | 6.63 (1H, d, 2.1) |
| 5″ | 6.67 (1H, d, 8.0) | 6.66 (1H, d, 8.0) | 6.63 (1H, d, 8.0) | 6.65 (1H, d, 8.4) |
| 6″ | 6.54 (1H, dd, 8.0, 2.0) | 6.53 (1H, dd, 8.0, 2.0) | 6.48 (1H, dd, 8.0, 2.0) | 6.52 (1H, dd, 8.4, 2.1) |
| 7″ | 2.79 (2H, m) | 2.77 (2H, m) | 2.76 (2H, m) | 2.76 (2H, m) |
| 8″ | 2.60 (2H, m) | 2.60 (2H, m) | 2.61 (2H, m) | 2.56 (2H, m) |
| Position | 3 | 4 | 5 | 6 |
|---|---|---|---|---|
| 1 | 75.6 | 75.6 | 75.0 | 75.0 |
| 2 | 37.8 | 37.5 | 38.4 | 37.4 |
| 3 | 72.7 | 74.9 | 75.4 | 72.2 a |
| 4 | 70.8 | 72.6 | 74.8 | 70.1 a |
| 5 | 72.3 | 72.2 | 69.2 | 72.2 |
| 6 | 36.2 | 35.9 | 37.4 | 35.8 |
| 7 | 175.9 | 175.9 | 175.2 | 175.6 |
| -OCH3 | - | 53.1 | 53.2 | 3.1 |
| 1′ | 128.1 | 128.0 | 127.6 | 133.6 |
| 2′ | 115.3 | 115.2 | 115.3 | 116.6 |
| 3′ | 146.9 | 147.0 | 147.0 | 146.3 |
| 4′ | 147.2 | 147.3 | 147.9 | 144.9 |
| 5′ | 116.6 | 116.6 | 116.7 | 116.5 |
| 6′ | 123.1 | 123.2 | 123.2 | 120.9 |
| 7′ | 149.6 | 149.7 | 150.0 | 37.9 |
| 8′ | 115.7 | 115.6 | 114.6 | 31.4 |
| 9′ | 169.0 | 169.0 | 167.8 | 174.6 |
| 1″ | 133.7 | 133.5 | 133.5 | 133.3 |
| 2″ | 116.6 | 116.6 | 116.5 | 116.6 |
| 3″ | 146.3 | 146.4 | 146.4 | 146.3 |
| 4″ | 144.8 | 144.8 | 144.8 | 144.9 |
| 5″ | 116.5 | 116.5 | 116.5 | 116.5 |
| 6″ | 120.7 | 120.7 | 120.6 | 120.9 |
| 7″ | 37.5 | 37.5 | 37.4 | 37.5 |
| 8″ | 31.4 | 31.5 | 31.7 | 31.6 |
| 9″ | 174.2 | 174.0 | 174.2 | 173.9 |
© 2016 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cho, J.-Y.; Kim, J.Y.; Lee, Y.G.; Lee, H.J.; Shim, H.J.; Lee, J.H.; Kim, S.-J.; Ham, K.-S.; Moon, J.-H. Four New Dicaffeoylquinic Acid Derivatives from Glasswort (Salicornia herbacea L.) and Their Antioxidative Activity. Molecules 2016, 21, 1097. https://doi.org/10.3390/molecules21081097
Cho J-Y, Kim JY, Lee YG, Lee HJ, Shim HJ, Lee JH, Kim S-J, Ham K-S, Moon J-H. Four New Dicaffeoylquinic Acid Derivatives from Glasswort (Salicornia herbacea L.) and Their Antioxidative Activity. Molecules. 2016; 21(8):1097. https://doi.org/10.3390/molecules21081097
Chicago/Turabian StyleCho, Jeong-Yong, Jin Young Kim, Yu Geon Lee, Hyoung Jae Lee, Hyun Jeong Shim, Ji Hye Lee, Seon-Jae Kim, Kyung-Sik Ham, and Jae-Hak Moon. 2016. "Four New Dicaffeoylquinic Acid Derivatives from Glasswort (Salicornia herbacea L.) and Their Antioxidative Activity" Molecules 21, no. 8: 1097. https://doi.org/10.3390/molecules21081097
APA StyleCho, J.-Y., Kim, J. Y., Lee, Y. G., Lee, H. J., Shim, H. J., Lee, J. H., Kim, S.-J., Ham, K.-S., & Moon, J.-H. (2016). Four New Dicaffeoylquinic Acid Derivatives from Glasswort (Salicornia herbacea L.) and Their Antioxidative Activity. Molecules, 21(8), 1097. https://doi.org/10.3390/molecules21081097





