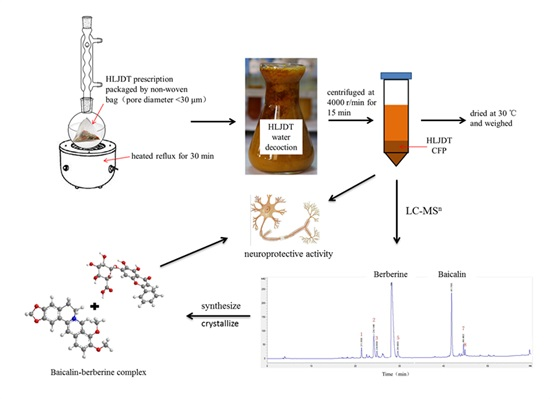
Compositions, Formation Mechanism, and Neuroprotective Effect of Compound Precipitation from the Traditional Chinese Prescription Huang-Lian-Jie-Du-Tang
Abstract
:1. Introduction
2. Results
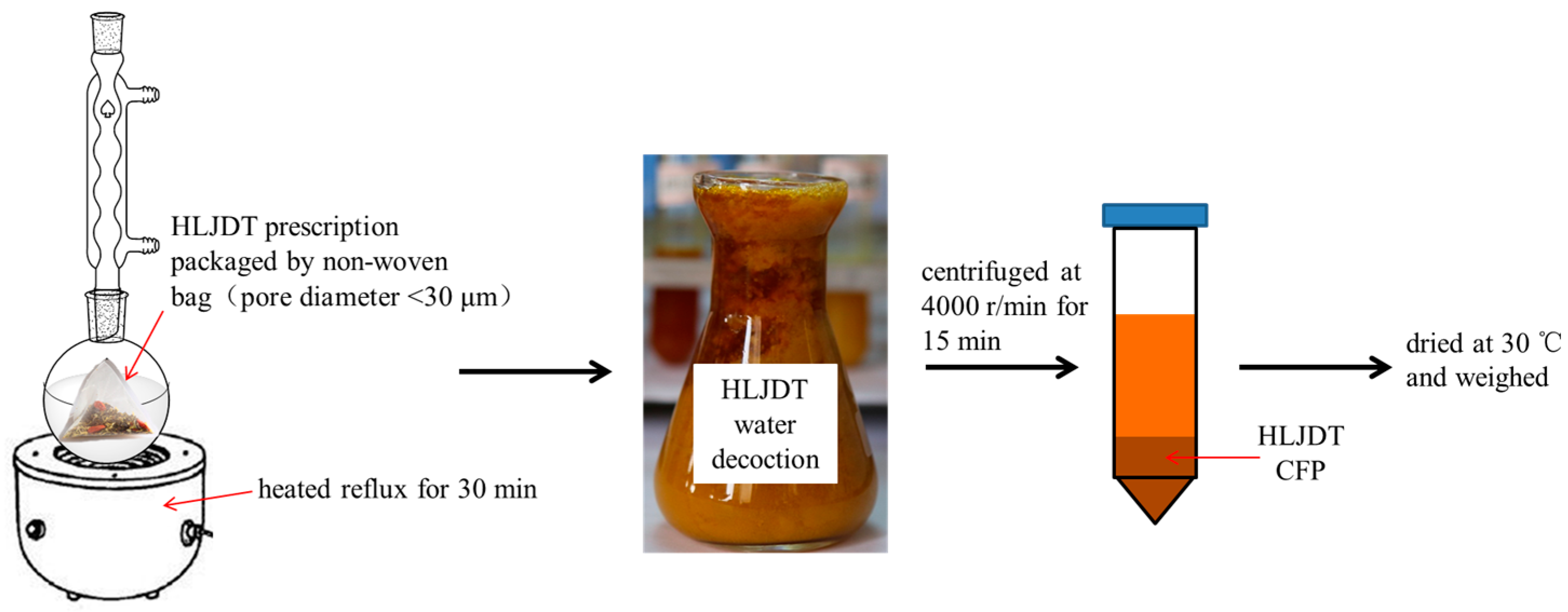
2.1. Preparation of HLJDT CFP
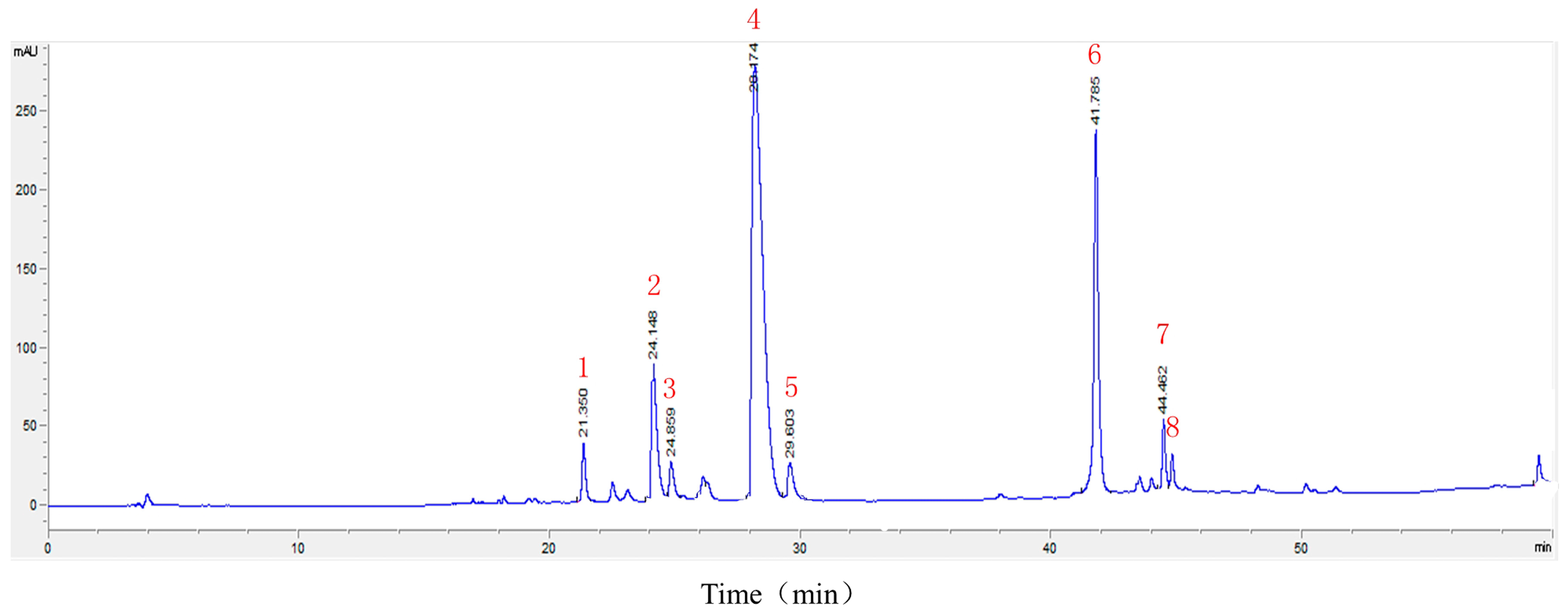
2.2. LC-MSn Analysis of the Constituents of HLJDT CFP
2.3. Synthesis of Simulated Precipitation
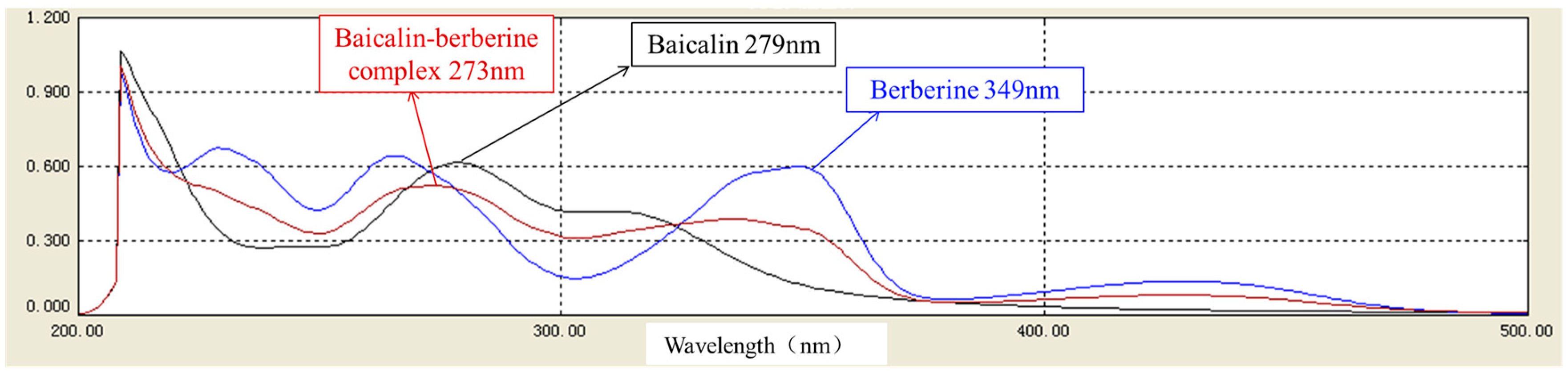
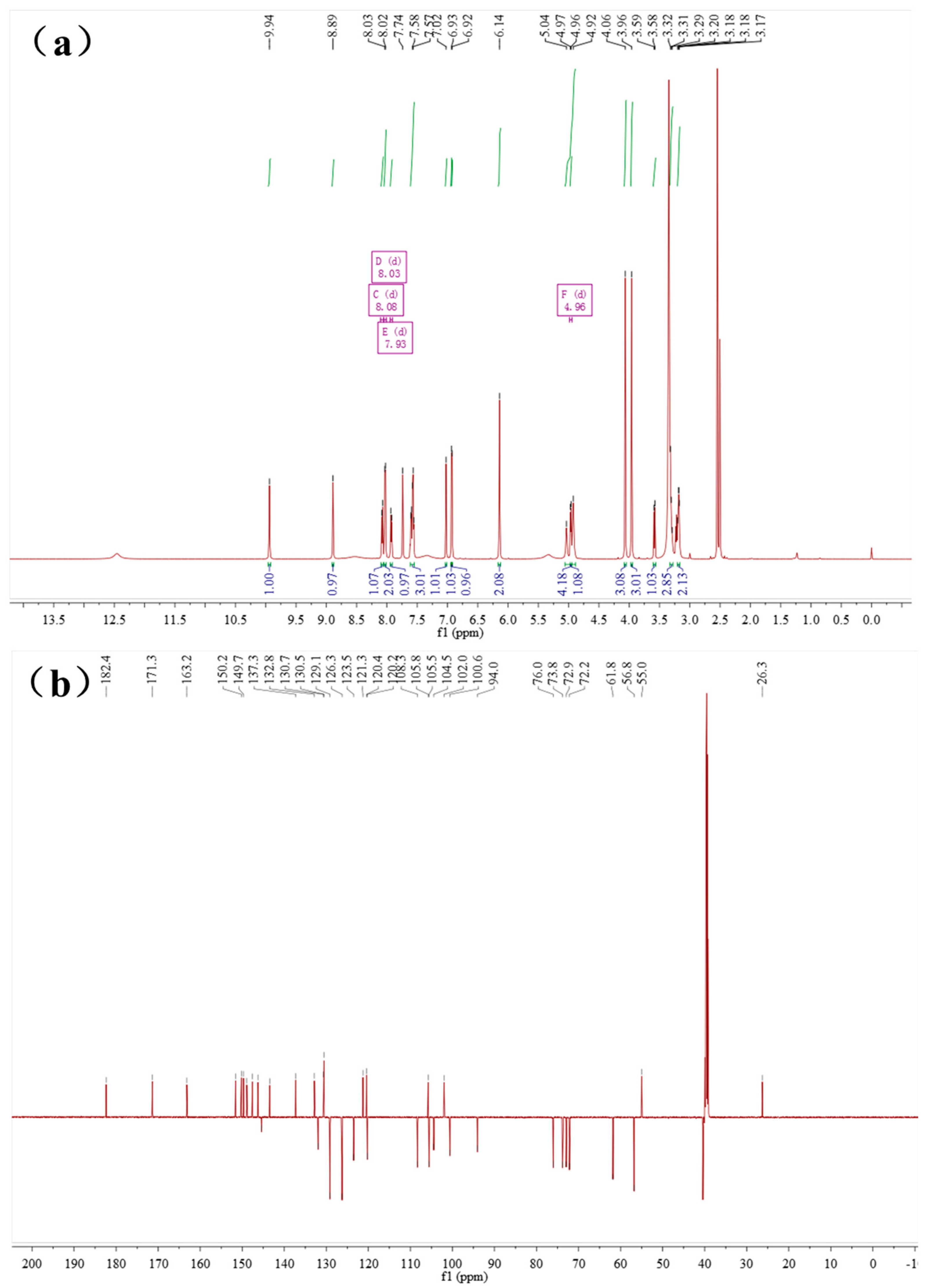
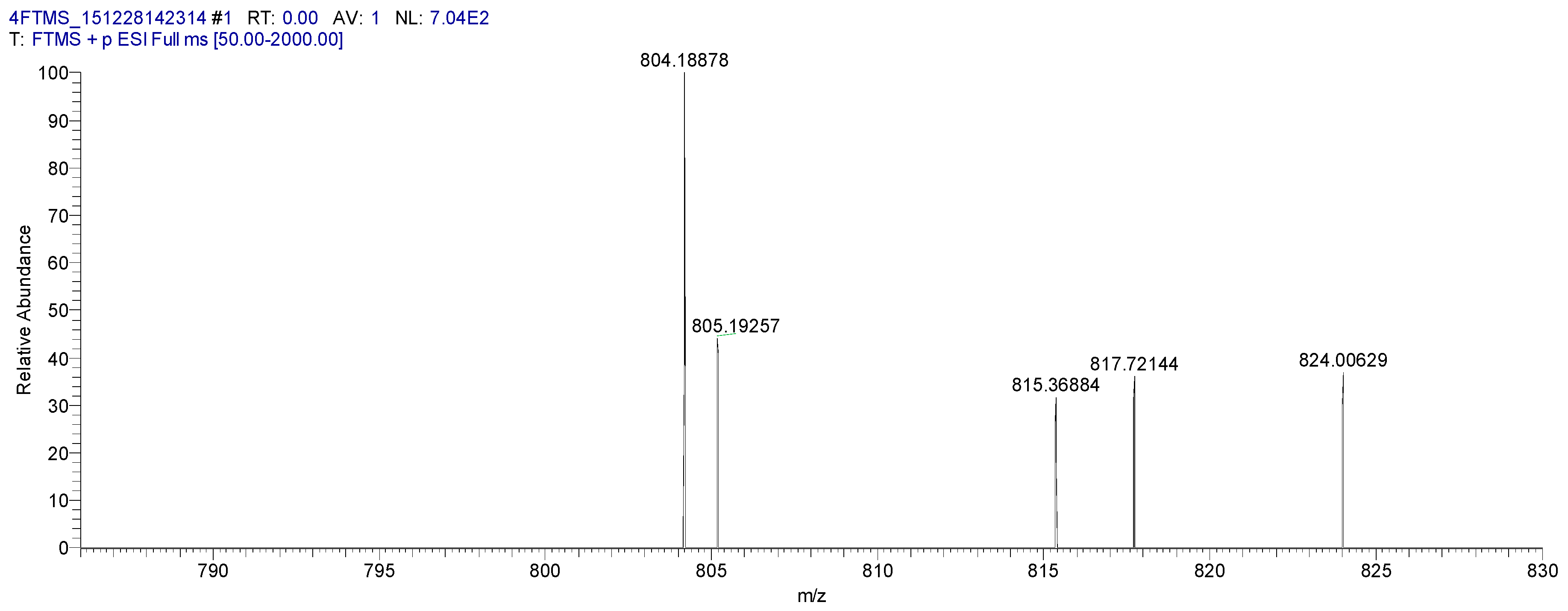
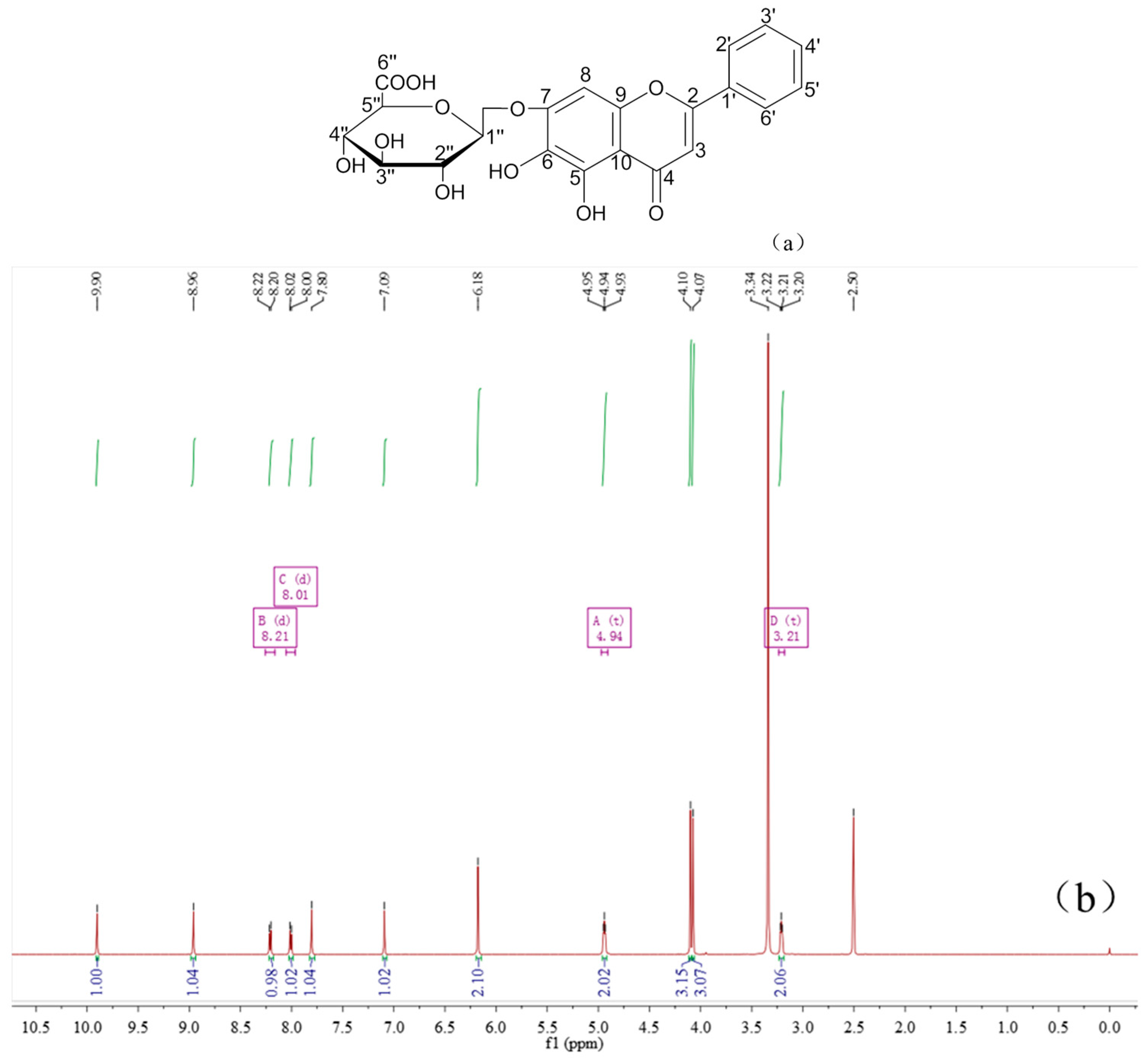
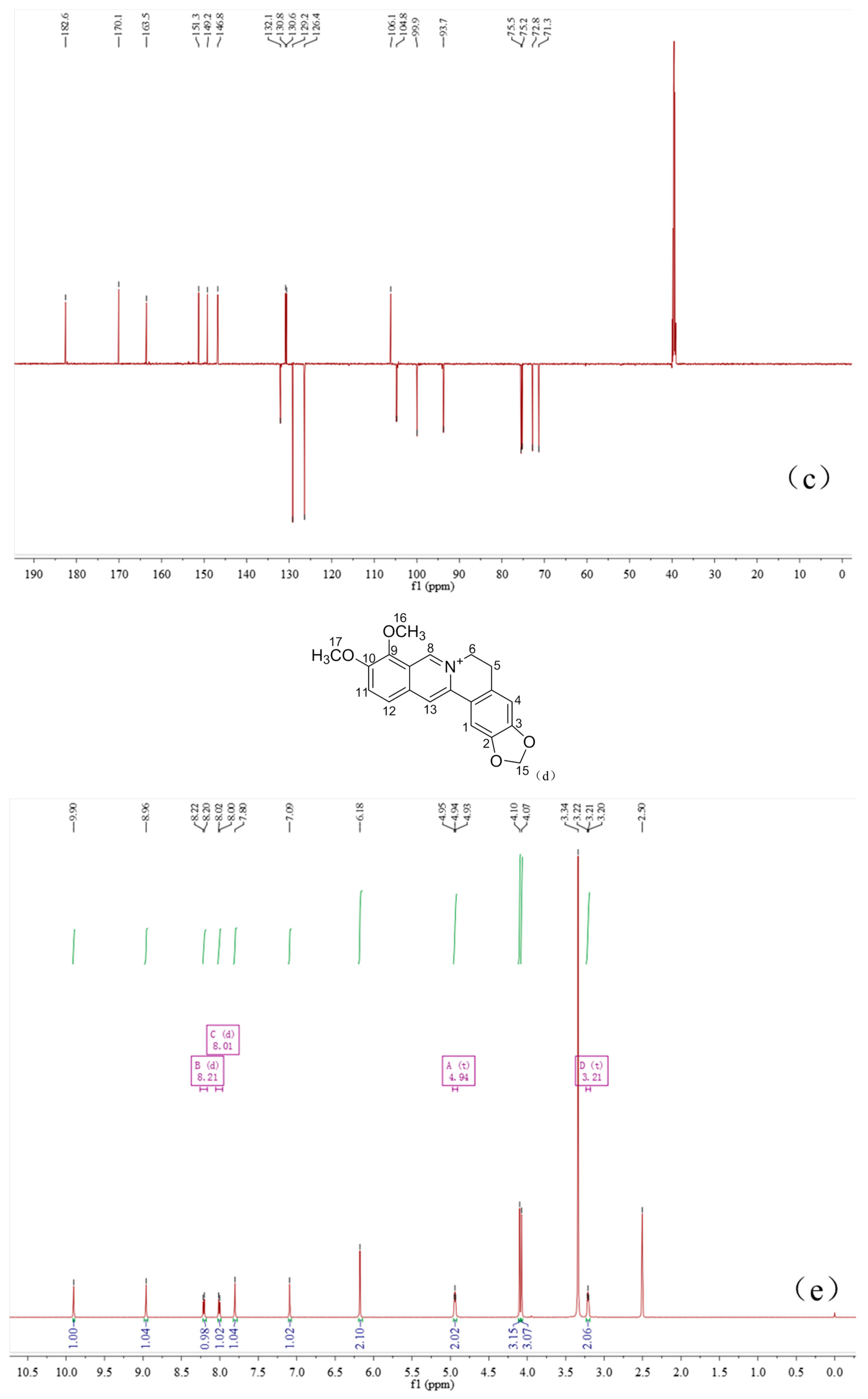
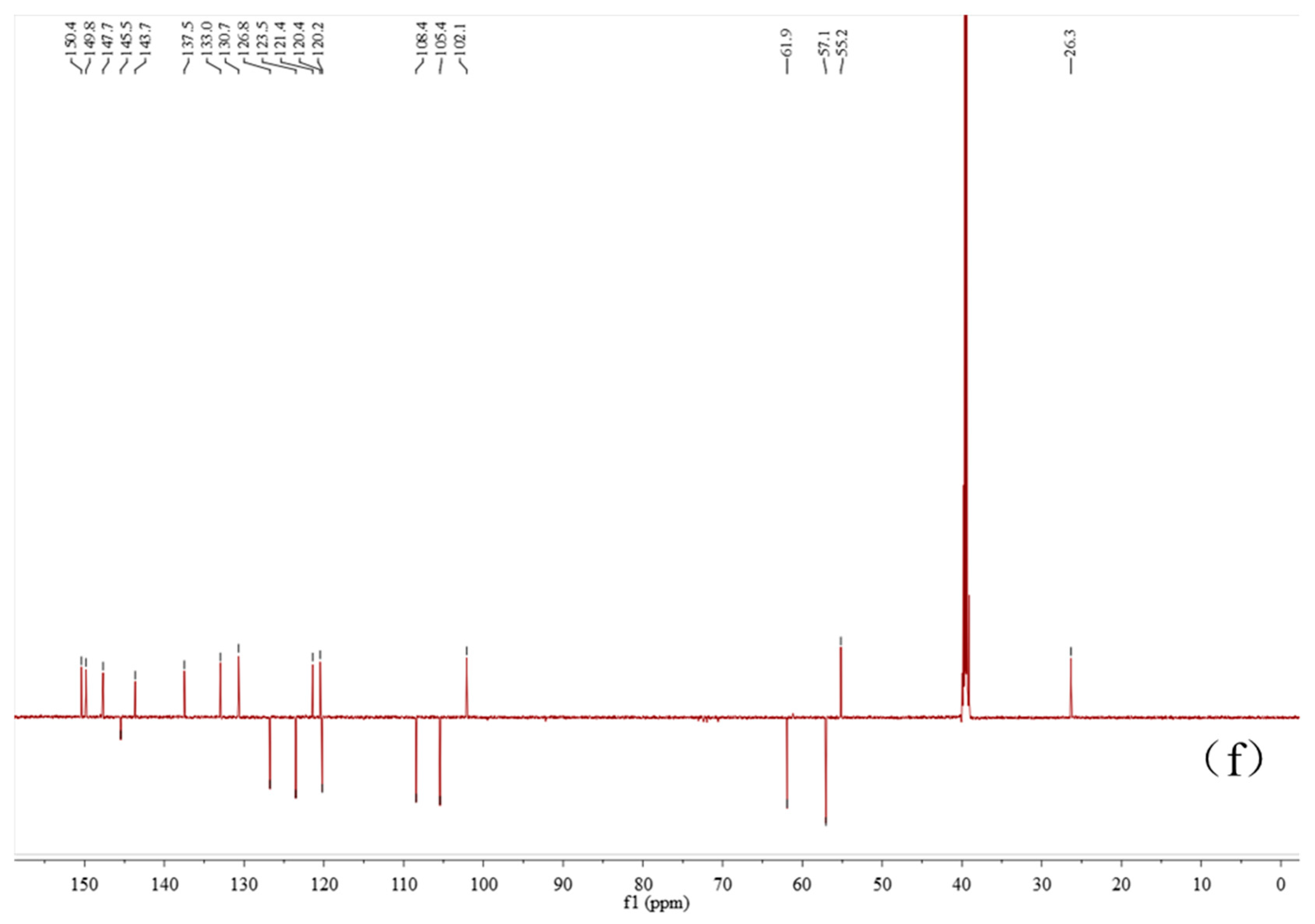
2.4. Characterization of the Baicalin–Berberine Complex
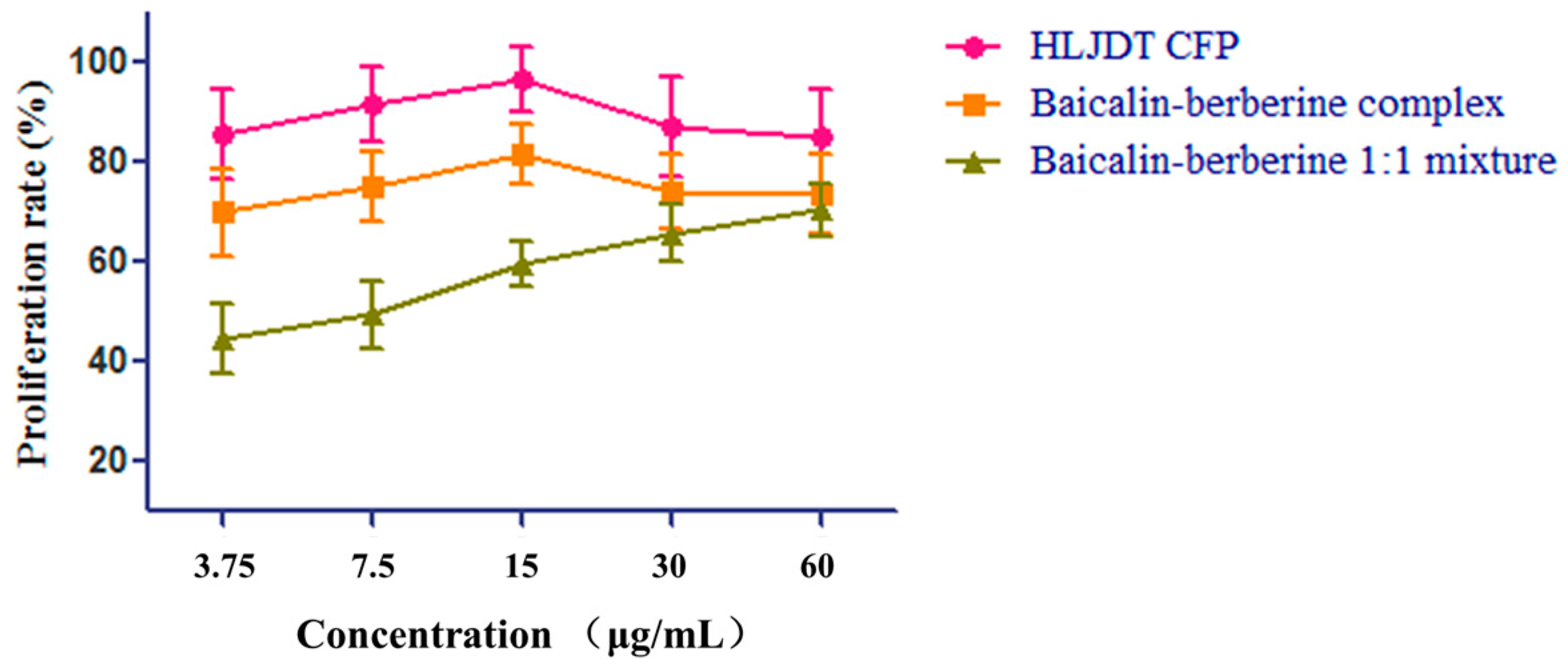
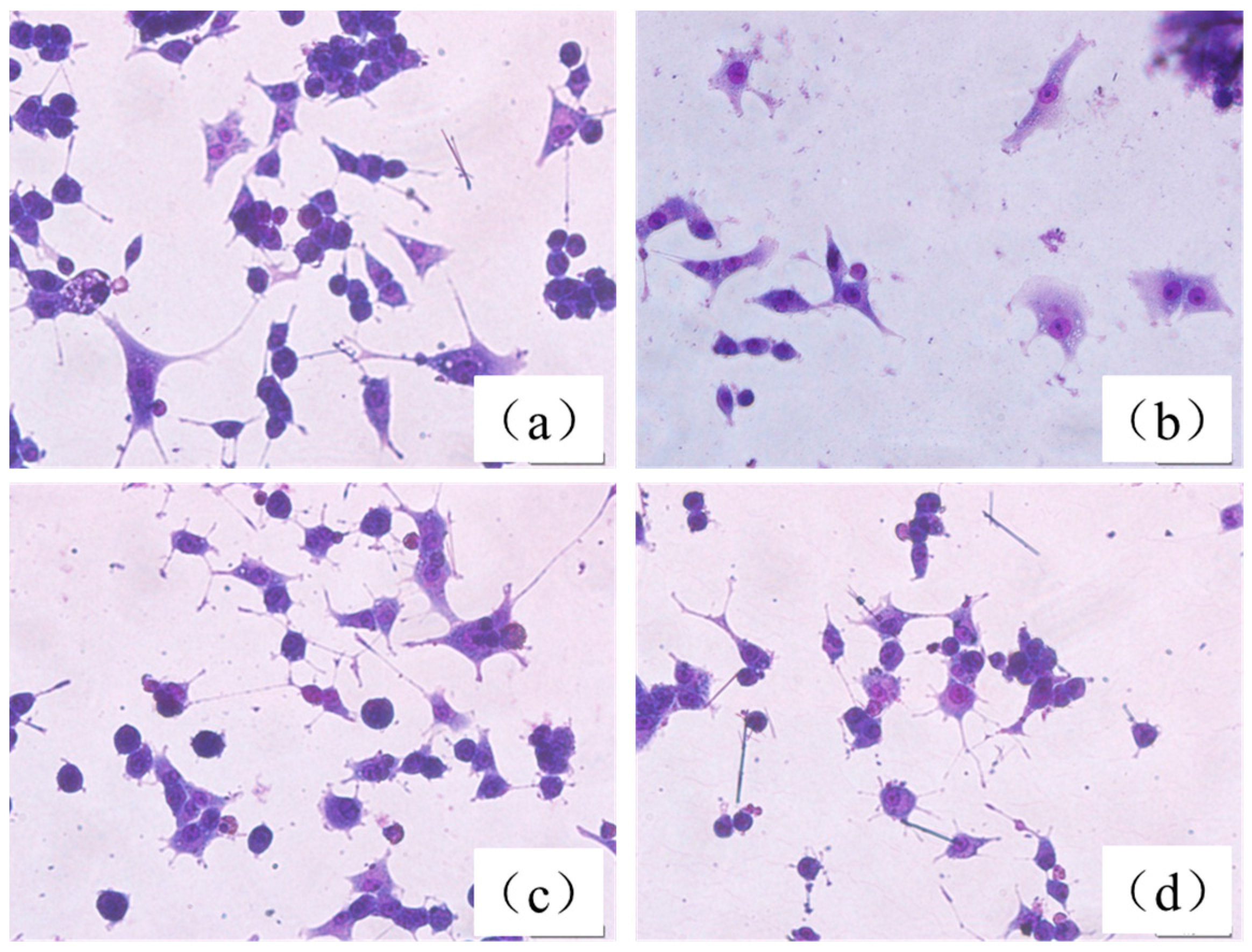
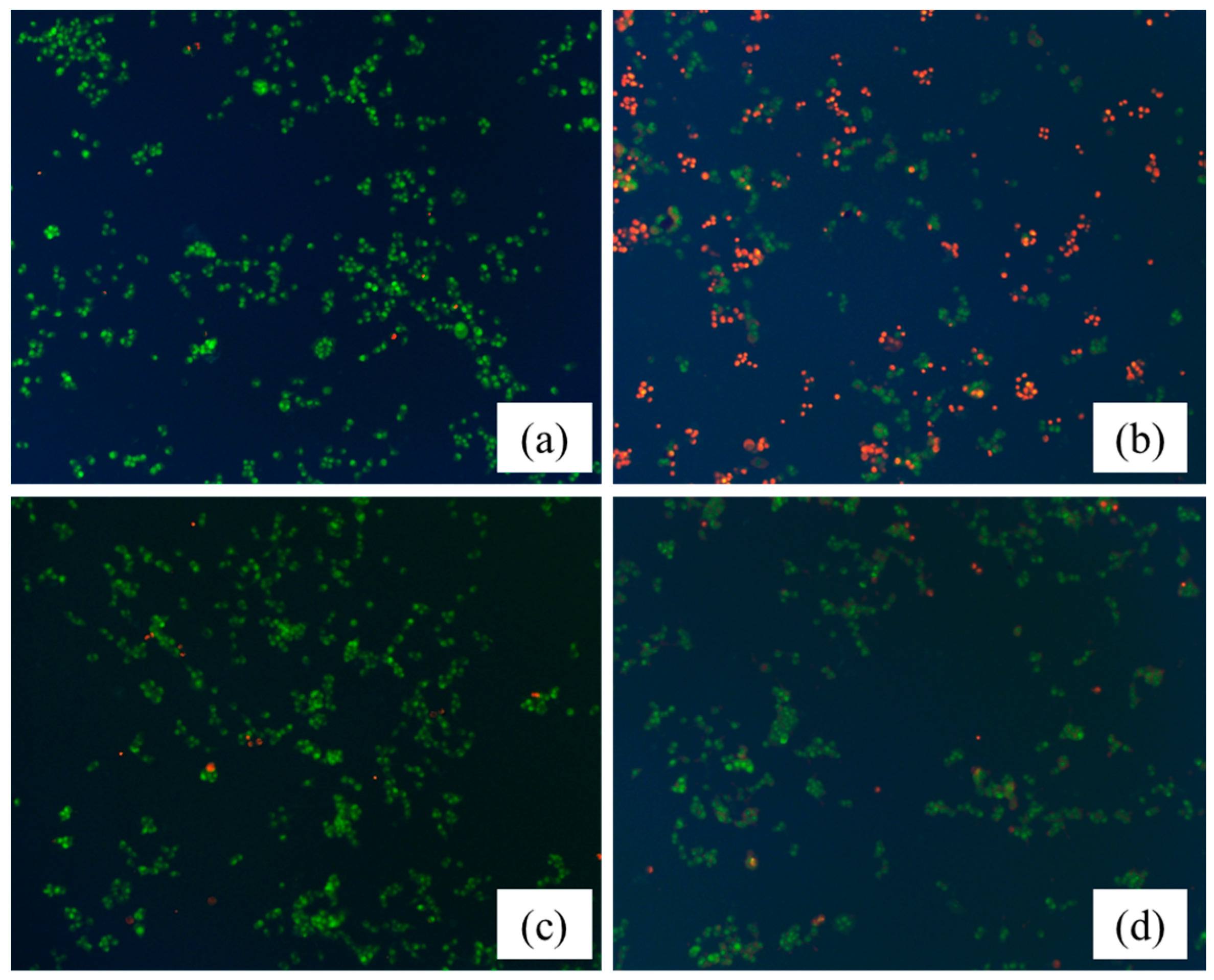
2.5. Protective Effect of the HLJDT CFP and Baicalin–Berberine Complex on Injured PC12 Cells
3. Discussion
4. Materials and Methods
4.1. Preparation of HLJDT CFP
4.2. LC-MSn Analysis of the Constituents of HLJDT CFP
4.3. Synthesis of Simulated Precipitation
4.4. Characterization of the Baicalin–Berberine Complex
4.5. Protective Effect of HLJDT CFP and Baicalin–Berberine Complex on Injured PC12 Cells
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Pan, L.; Fu, J.; Zhu, H.; Guo, L. Preliminary study to investigate dynamic extract process of Huang-Lian-Jie-Du-Tang and the mechanism of subsidence produce. China J. Chin. Mater. Med. 2010, 35, 40–43. [Google Scholar]
- Pei, X.; Pei, M.; Duan, X. Compatability chemistry of acid-alkaline pair medicine of Ephedra sinica and Glycyrrhiza uralencis in maxing shigan decoction. China J. Chin. Mater. Med. 2009, 34, 2466–2468. [Google Scholar]
- Pei, M.; Duan, X.; Pei, X. Compatability chemistry of acid-alkaline pair medicine of fuzi and gancao in sini decoction. China J. Chin. Mater. Med. 2009, 34, 2047–2050. [Google Scholar]
- Pei, M.; Duan, X.; Pei, X. Compatibility chemistry of acid-alkaline pair medicine of dahuang and huangbai in dahuang xiaoshi decoction. China J. Chin. Mater. Med. 2009, 34, 2312–2315. [Google Scholar]
- Duan, X.; Pei, M.; Pei, X. Compatability chemistry of acid-alkaline pair medicine of dahuang and fuzi in dahuang fuzi decoction. China J. Chin. Mater. Med. 2009, 34, 2167–2171. [Google Scholar]
- Dou, S.S.; Liu, L.; Jiang, P.; Zhang, W.D.; Liu, R.H. LC-DAD and LC-ESI-MS chromatographic fingerprinting and quantitative analysis for evaluation of the quality of Huang-Lian-Jie-Du-Tang. Chromatographia 2009, 69, 659–664. [Google Scholar] [CrossRef]
- Ye, Y.; Huang, C.; Jiang, L.; Shen, X.; Zhu, S.; Rao, Y.; Wang, J.; Zhang, Q. Huang-Lian-Jie-Du-Tang extract protects against chronic brain injury after focal cerebral ischemia via hypoxia-inducible-factor-1alpha-regulated vascular endothelial growth factor signaling in mice. Biol. Pharm. Bull. 2012, 35, 355–361. [Google Scholar] [CrossRef] [PubMed]
- Pan, L.; Zhou, J.; Zhu, H.; Wang, W.; Zhang, M.; Tian, X.; Lu, J.; Zeng, M. Study on integrated pharmacokinetics of gardenia acid and geniposide: Time-antioxidant efficacy after oral administration of Huanglian-Zhizi couplet medicine from Huang-Lian-Jie-Du-Tang in mcao rats. Am. J. Chin. Med. 2014, 42, 393–407. [Google Scholar] [CrossRef] [PubMed]
- Wang, P.R.; Wang, J.S.; Zhang, C.; Song, X.F.; Tian, N.; Kong, L.Y. Huang-Lian-Jie-Du-Decotion induced protective autophagy against the injury of cerebral ischemia/reperfusion via mapk-mtor signaling pathway. J. Ethnopharmacol. 2013, 149, 270–280. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Xian, Y.; Lai, Z.; Loo, S.; Chan, W.Y.; Lin, Z.X. Anti-inflammatory and anti-allergic effects and underlying mechanisms of Huang-Lian-Jie-Du extract: Implication for atopic dermatitis treatment. J. Ethnopharmacol. 2016, 185, 41–52. [Google Scholar] [CrossRef] [PubMed]
- Li, C.B.; Li, X.X.; Chen, Y.G.; Gao, H.Q.; Bu, P.L.; Zhang, Y.; Ji, X.P. Huang-Lian-Jie-Du-Tang protects rats from cardiac damages induced by metabolic disorder by improving inflammation-mediated insulin resistance. PLoS ONE 2013, 8, e67530. [Google Scholar] [CrossRef] [PubMed]
- Wei, Y.; Shan, L.; Qiao, L.; Liu, R.; Hu, Z.; Zhang, W. Protective effects of Huang-Lian-Jie-Du-Tang against polymicrobial sepsis induced by cecal ligation and puncture in rats. Evid. Base Complement. Altern. Med. 2013, 2013, 909624. [Google Scholar] [CrossRef] [PubMed]
- Dai, Y.; Miki, K.; Fukuoka, T.; Tokunaga, A.; Tachibana, T.; Kondo, E.; Noguchi, K. Suppression of neuropeptides’ mRNA expression by herbal medicines in a rat model of peripheral inflammation. Life Sci. 2000, 66, 19–29. [Google Scholar] [CrossRef]
- Tanaka, K.; Nara, K.; Nishimura, T.; Serizawa, K.; Miyazaki, T.; Urita, Y.; Miura, O. Fever of unknown origin successfully treated by oren-gedoku-to (Huang-Lian-Jie-Du-Tang). Int. J. Gen. Med. 2013, 6, 829–832. [Google Scholar] [CrossRef] [PubMed]
- Staswick, P.E.; Hermodson, M.A.; Nielsen, N.C. Identification of the acidic and basic subunit complexes of glycinin. J. Biol. Chem. 1981, 256, 8752–8755. [Google Scholar] [PubMed]
- Govorun, D.M.; Kondratyuk, I.V.; Zheltovsky, N.V. Acidic-basic properties of molecular xanthine and its complex formation ability. Biopolym. Cell 1994, 10, 61–64. [Google Scholar] [CrossRef]
- Zheltovsky, N.V.; Samiylenko, S.P.; Kolomiets, I.N.; Kondratyuk, I.V.; Stepanyugin, A.V. The investigation of interactions of hypoxanthine, xanthine and their methyl and glycosyl derivatives with amino acid carboxylic group by spectroscopic methods. Biopolym. Cell 1993, 9, 17–22. [Google Scholar] [CrossRef]
- Li, B.; Yan, W.; Zhang, C.; Zhang, Y.; Liang, M.; Chu, F.; Gong, Y.; Xu, B.; Wang, P.; Lei, H. New synthesis method for sultone derivatives: Synthesis, crystal structure and biological evaluation of S-CA. Molecules 2015, 20, 4307–4318. [Google Scholar] [CrossRef] [PubMed]
- Ma, X.; Yan, J.; Xu, K.; Guo, L.; Li, H. Binding mechanism of trans-n-caffeoyltyramine and human serum albumin: Investigation by multi-spectroscopy and docking simulation. Bioorg. Chem. 2016, 66, 102–110. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.F.; Xia, Y.M.; Fang, Y.; Liu, L.L.; Zou, L. Interaction between bovine serum albumin and berberine chloride extracted from Chinese Herbs of coptis chinensis franch. Chem. J. Chin. Univ. 2004, 25, 2099–2103. [Google Scholar]
- Wang, P.L.; Zhang, H.G.; Chu, F.H.; Xu, X.; Lin, J.X.; Chen, C.X.; Li, G.L.; Cheng, Y.T.; Wang, L.; Li, Q.; et al. Synthesis and protective effect of new ligustrazine-benzoic acid derivatives against CoCl2-induced neurotoxicity in differentiated PC12 cells. Molecules 2013, 18, 13027–13042. [Google Scholar] [CrossRef] [PubMed]
- Xu, B.; Gong, Y.; Xu, X.; Zhang, C.Z.; Zhang, Y.Z.; Chu, F.H.; Liu, H.B.; Wang, P.L.; Lei, H.M. Synthesis and protective effect of new ligustrazine derivatives against CoCl2-induced neurotoxicity in differentiated PC12 cells. Part 2. Medchemcomm 2015, 6, 806–809. [Google Scholar] [CrossRef]
- Zhang, Q.C.; Qian, Z.L.; Pan, L.M.; Li, H.; Zhu, H.X. The traditional Chinese medicine Huang-Lian-Jie-Du-Tang inhibits hypoxia-induced neuronal apoptosis. Afr. J. Pharm. Pharmacol. 2011, 5, 2558–2565. [Google Scholar]
- Durairajan, S.S.; Huang, Y.Y.; Yuen, P.Y.; Chen, L.L.; Kwok, K.Y.; Liu, L.F.; Song, J.X.; Han, Q.B.; Xue, L.; Chung, S.K.; et al. Effects of Huang-Lian-Jie-Du-Tang and its modified formula on the modulation of amyloid-beta precursor protein processing in Alzheimer’s disease models. PLoS ONE 2014, 9, e92954. [Google Scholar]
- Lapchak, P.A.; Maher, P.; Schubert, D.; Zivin, J.A. Baicalein, an antioxidant 12/15-lipoxygenase inhibitor improves clinical rating scores following multiple infarct embolic strokes. Neuroscience 2007, 150, 585–591. [Google Scholar] [CrossRef] [PubMed]
- Zhang, C.; Yan, W.; Li, B.; Xu, B.; Gong, Y.; Chu, F.; Zhang, Y.; Yao, Q.; Wang, P.; Lei, H. A new ligustrazine derivative-selective cytotoxicity by suppression of NF-κB/p65 and COX-2 expression on human hepatoma cells. Part 3. Int. J. Mol. Sci. 2015, 16, 16401–16413. [Google Scholar] [CrossRef] [PubMed]
- Chu, F.H.; Xu, X.; Li, G.L.; Gu, S.; Xu, K.; Gong, Y.; Xu, B.; Wang, M.N.; Zhang, H.Z.; Zhang, Y.Z.; et al. Amino acid derivatives of ligustrazine-oleanolic acid as new cytotoxic agents. Molecules 2014, 19, 18215–18231. [Google Scholar] [CrossRef] [PubMed]
- Sample Availability: Samples of the compounds HLJDT CFP and Baicalin–berberine complex are available from the authors.

| Batch | Coptidis Rhizoma | Scutellariae Radix | Phellodendri Cortex | Gardeniae Fructus | Total Weight | Precipitation Weight | Precipitation Rate |
|---|---|---|---|---|---|---|---|
| 1 | 9.02 g | 6.00 g | 6.01 g | 9.02 g | 30.05 g | 0.73 g | 2.44% |
| 2 | 9.01 g | 5.99 g | 6.01 g | 9.01 g | 30.02 g | 0.85 g | 2.84% |
| 3 | 9.00 g | 6.00 g | 5.99 g | 9.00 g | 29.99 g | 0.78 g | 2.61% |
| average | 30.02 ± 0.03 g | 0.79 ± 0.06 g | 2.63% ± 0.20% | ||||
| Peak | Compounds | tR (min) | MS (m/z) | MSn (m/z) |
|---|---|---|---|---|
| 1 | Geniposide | 21.36 | 386.9 [M − H]− | 224.8 [M − H − C6H10O5]− 123.0 [M − H − C6H10O5 − C4H6O3]− |
| 2 | Coptisine | 24.15 | 320.0 [M]+ | 292.0 [M − C2H4]+ |
| 3 | Epiberberine | 24.86 | 336.1 [M]+ | 320.1 [M − CH4]+ 292.1 [M − CH4 − C2H4]+ |
| 4 | Berberine | 28.17 | 336.1 [M]+ | 320.1 [M − CH4]+ 292.1 [M − CH4 − C2H4]+ |
| 5 | Palmatine | 29.60 | 352.1 [M]+ | 336.1 [M − CH4]+ 308.1 [M − CH4 − C2H4]+ |
| 6 | Baicalin | 41.76 | 447.1 [M + H]+ | 271.0 [M + H − glua 1]+ |
| 7 | Oroxylin A A-7-O-glucuronide | 44.46 | 458.9 [M − H]− | 282.8 [M − H − glua]− |
| 8 | Wogonoside | 45.10 | 458.9 [M − H]− | 282.8 [M − H − glua]− |
| Baicalin (δ ppm) | Baicalin–Berberine Complex (δ ppm) | Difference (ppm) |
|---|---|---|
| 2’’, 3’’, 4’’-OH, 5.52–5.31 | 5.04–4.92 | 0.39–0.48 |
| 1’’-H, 5.26 | 4.96 | 0.30 |
| 5’’-H, 4.08 | 3.58 | 0.50 |
| 2’’, 3’’, 4’’-H, 3.47–3.42 | 3.32–3.29 | 0.13–0.15 |
| 6”-C, 170.1 | 171.3 | 1.2 |
| 1”-C, 99.9 | 100.6 | 0.7 |
| 5”-C, 75.5 | 76.0 | 0.5 |
| 3”-C, 75.2 | 73.8 | 1.4 |
| 4”-C, 71.3 | 72.2 | 0.9 |
| Samples | Proliferation Rate (%) | EC50 (μg/mL) | ||||
|---|---|---|---|---|---|---|
| 60 μg/mL | 30 μg/mL | 15 μg/mL | 7.5 μg/mL | 3.75 μg/mL | ||
| HLJDT CFP | 85.08 ± 9.72 | 86.98 ± 10.03 | 96.79 ± 6.52 | 91.53 ± 7.58 | 85.58 ± 9.03 | 3.35 ± 1.11 |
| Baicalin–Berberine complex | 73.62 ± 7.96 | 74.17 ± 7.46 | 81.48 ± 6.08 | 75.10 ± 6.94 | 70.05 ± 8.72 | 5.79 ± 1.67 |
| Baicalin–Berberine 1:1 mixture | 70.45 ± 5.36 | 65.83 ± 5.85 | 59.43 ± 4.43 | 49.38 ± 6.57 | 44.57 ± 6.84 | 11.1 ± 2.49 |
© 2016 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhang, C.; Zhao, R.; Yan, W.; Wang, H.; Jia, M.; Zhu, N.; Zhu, Y.; Zhang, Y.; Wang, P.; Lei, H. Compositions, Formation Mechanism, and Neuroprotective Effect of Compound Precipitation from the Traditional Chinese Prescription Huang-Lian-Jie-Du-Tang. Molecules 2016, 21, 1094. https://doi.org/10.3390/molecules21081094
Zhang C, Zhao R, Yan W, Wang H, Jia M, Zhu N, Zhu Y, Zhang Y, Wang P, Lei H. Compositions, Formation Mechanism, and Neuroprotective Effect of Compound Precipitation from the Traditional Chinese Prescription Huang-Lian-Jie-Du-Tang. Molecules. 2016; 21(8):1094. https://doi.org/10.3390/molecules21081094
Chicago/Turabian StyleZhang, Chenze, Rui Zhao, Wenqiang Yan, Hui Wang, Menglu Jia, Nailiang Zhu, Yindi Zhu, Yuzhong Zhang, Penglong Wang, and Haimin Lei. 2016. "Compositions, Formation Mechanism, and Neuroprotective Effect of Compound Precipitation from the Traditional Chinese Prescription Huang-Lian-Jie-Du-Tang" Molecules 21, no. 8: 1094. https://doi.org/10.3390/molecules21081094
APA StyleZhang, C., Zhao, R., Yan, W., Wang, H., Jia, M., Zhu, N., Zhu, Y., Zhang, Y., Wang, P., & Lei, H. (2016). Compositions, Formation Mechanism, and Neuroprotective Effect of Compound Precipitation from the Traditional Chinese Prescription Huang-Lian-Jie-Du-Tang. Molecules, 21(8), 1094. https://doi.org/10.3390/molecules21081094