Abstract
Naturally occurring homoisoflavonoids containing either 5,7-dihydroxy-6-methoxy or 7-hydroxy-5,6-dimethoxy groups such as the antiangiogenic homoisoflavanone, cremastranone, were synthesized via three or four linear steps from the known 4-chromenone. This facile synthesis includes chemoselective 1,4-reduction of 4-chromenone and selective deprotection of 3-benzylidene-4-chromanone a containing C7-benzyloxy group.
1. Introduction
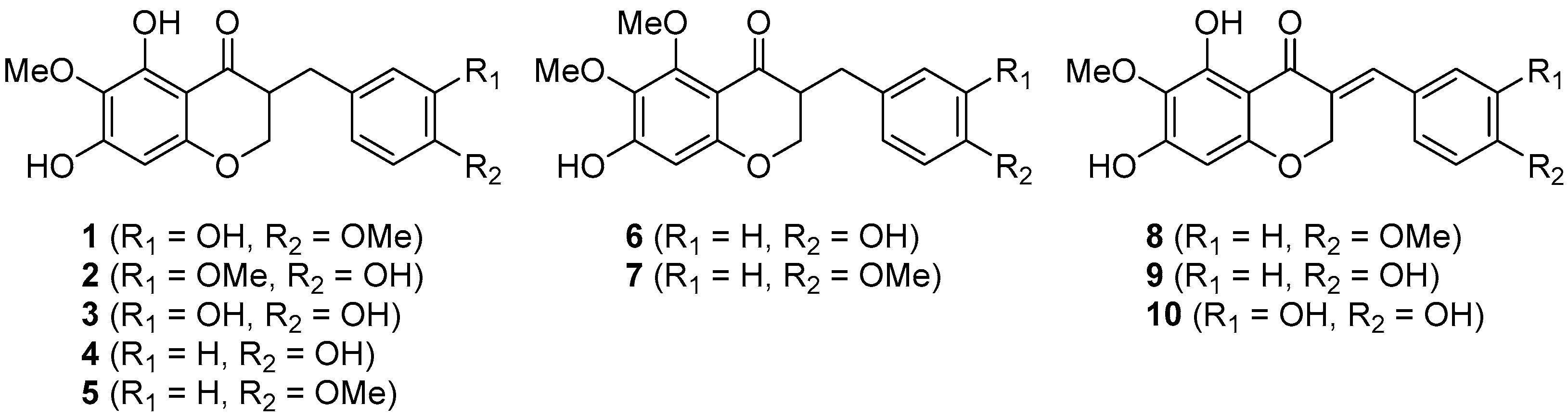
Recently, the antiangiogenic homoisoflavonoid cremastranone (1), isolated from the plants Muscari armeniacum, Chionodoxa luciliae, Scilla natalensis, Merwilla plumbea, and Cremastra appendiculata was synthesized for the first time by us [1,2,3,4,5,6]. Its naturally occurring congeners (2–10), which contain either 5,7-dihydroxy-6-methoxy or 7-hydroxy-5,6-dimethoxy groups, have been reported already as shown in Figure 1 and most of them lack common names other than eucomnalin (9; autumnalin) and 3,9-dihydroeucomnalin (4; 3,9-dihydroautumnalin). Two review articles deal with the natural origins and structures of most homoisoflavonoids [7,8], and thereafter various studies of the homoisoflavonoids have been published [9,10,11]. Nevertheless, there have not been synthetic efforts towards such homoisoflavonoids since the synthesis of 4 and 9 was reported in 1971 [12]. A chemical synthesis of 5,6,7-trisubstituted homoisoflavonoids has the potential to provide a general and expedient entry into a plethora of analogues potentially with interesting biological activities [13,14,15]. Herein we report the first synthesis of cremastranone’s derivatives in three or four steps from the known 4-chromenone, involving a chemoselective 1,4-reduction and manipulation of protecting groups.
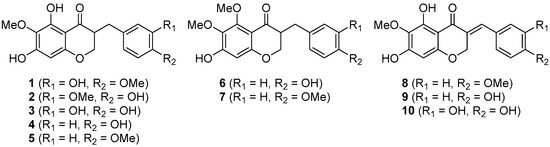
Figure 1.
Naturally occurring homoisoflavonoids containing either 5,7-dihydroxy-6-methoxy (i.e., 1–5 and 8–10) or 7-hydroxy-5,6-dimethoxy (6 and 7).
2. Results and Discussion
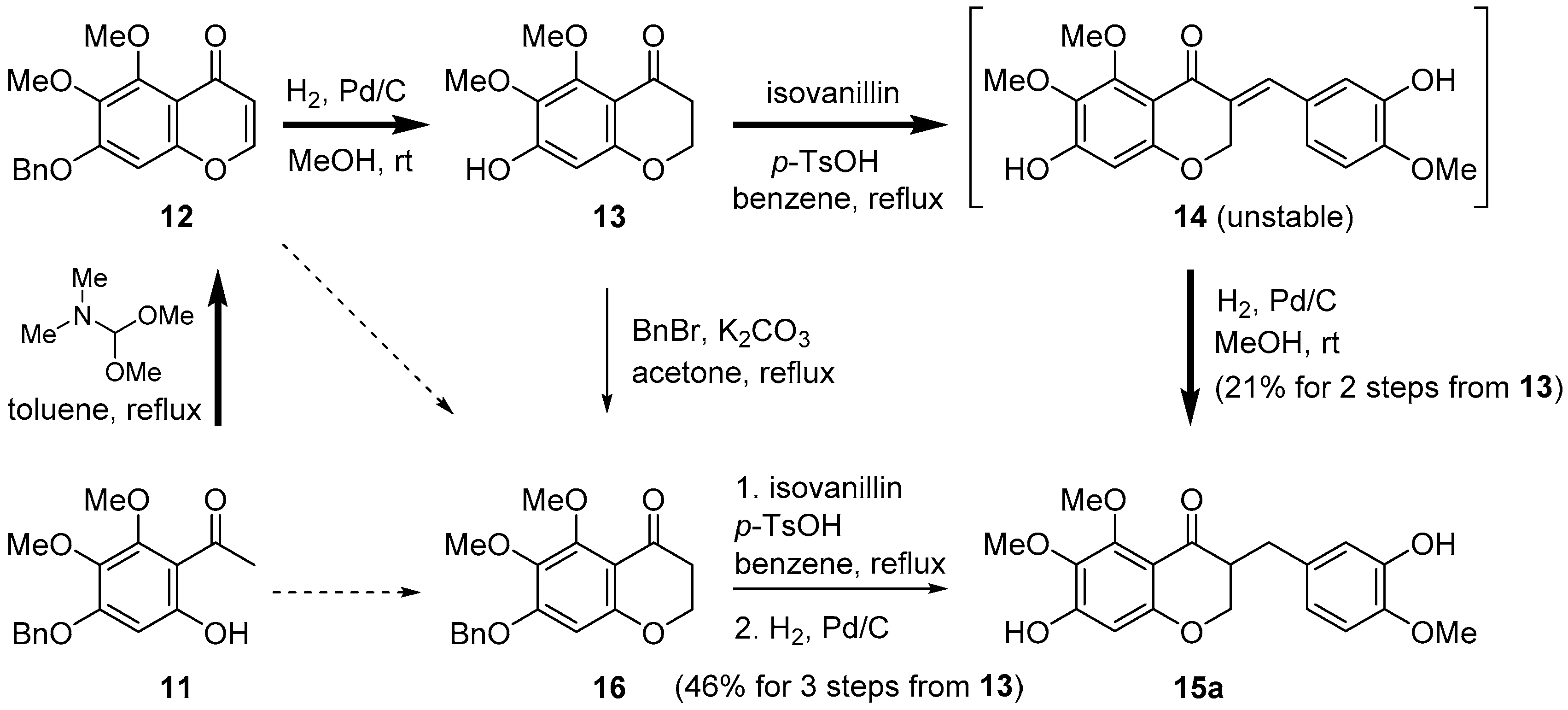
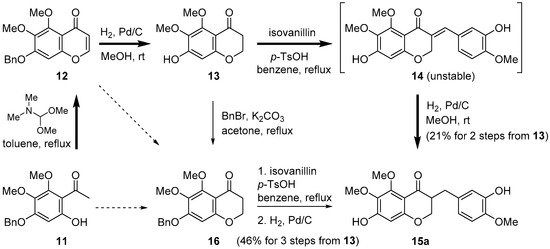
In our initial approach to cremastranone (1), the acetophenone 11 as starting material was treated with N,N-dimethylformamide dimethyl acetal, followed by catalytic hydrogenation to afford the 4-chromanone 13 in a good yield (Scheme 1) [14]. However, aldol condensation of 13 with isovanillin gave the unstable benzylidene products (i.e., 14a) in a low yield; the free OH group on C7 is thought to hamper the general aldol condensation as well as the stability of the desired products. To overcome this drawback, the benzylation of the phenol group on C7 was adopted prior to aldol condensation. The aldol condensation of 16 with isovanillin afforded the desired compound 15a in good yield.
Scheme 1.
Our previous approach using 5,6,7-trisubstituted 3-benzyl-4-chromanone for the reported synthesis of cremastranone (bold arrow) [14] and the alternative pathway (normal arrow).
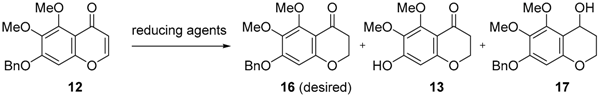
To improve chemical yield and decrease reaction steps, we focused on the direct conversion of either the acetophenone 11 or the chromenone 12 into the desired chromanone 16. However, it was reported that the formation of a 4-chromanone from a 2-hydroxyacetophenone (i.e., 11) is challenging [16]. The direct conversion of 12 to 16 was attempted under some reduction conditions (Table 1) [17]. Even careful catalytic hydrogenation is not likely to control a partial reduction of only the double bond. Among metal hydride reagents, the reduction using 1.5 equivalent of NaBH4 did not proceed over 5 h, forming only a small amount of 4-chromanol 17 (<10%). Treatment with excess NaBH4 afforded only 4-chromanol 17 in 50% yield (80% based on the recovered 12). Diisobutylaluminum hydride (DIBAL) reduction at −60 °C led to the nonselective generation of 16 (20%) and 17 (20%) in low yields. To our delight, the reduction by LiAlH4 underwent only 1,4-addition of hydride without over-reduction quantitatively and had the advantage of being reproducible on a gram scale.

Table 1.
Reduction conditions of 7-benzyloxy-5,6-dimethoxy-4-chromenone (12).
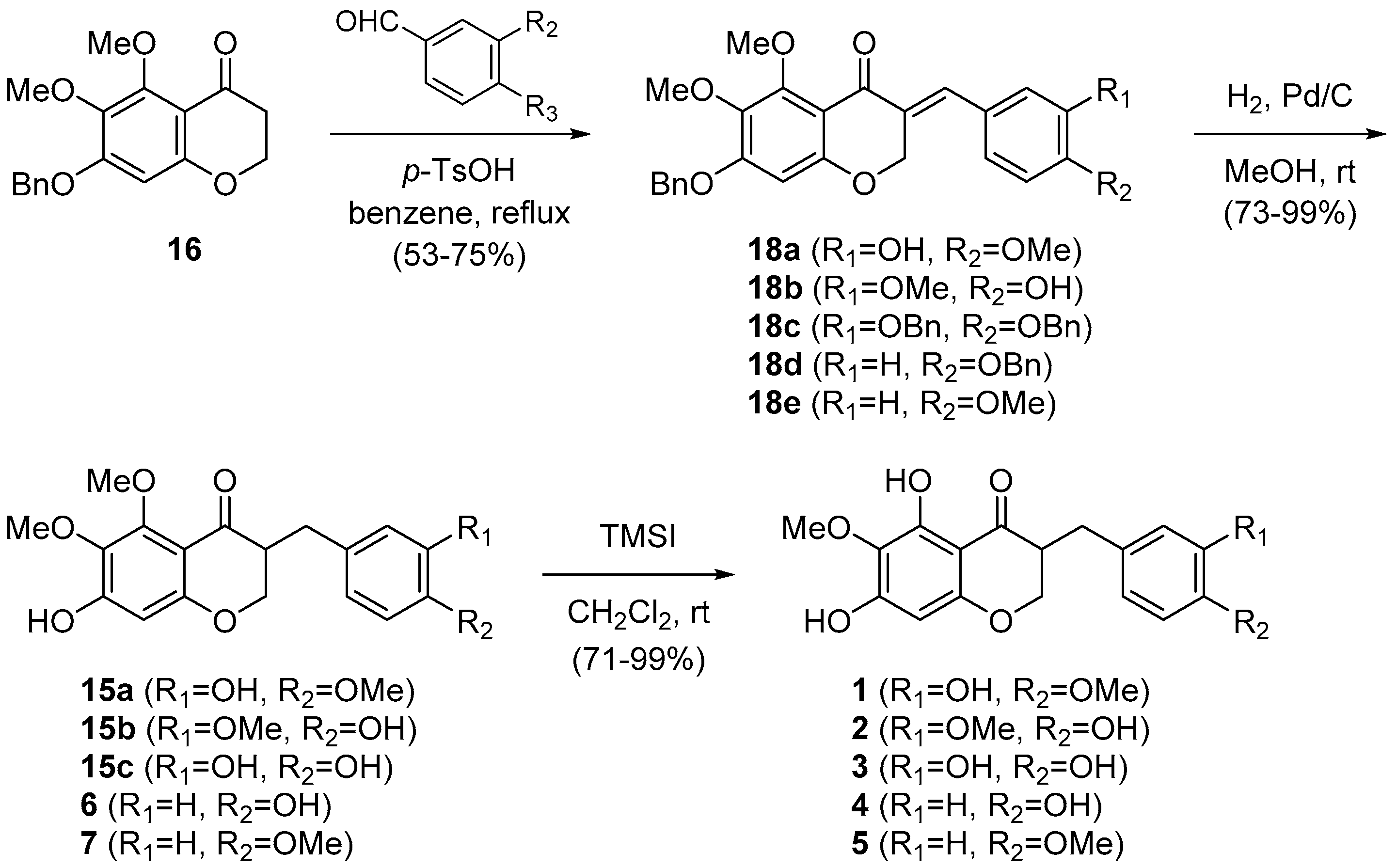
With 16 in hand, we turned to the synthesis of natural 3-benzyl-4-chromanones containing 5,7-dihydroxy-6-methoxy and 7-hydroxy-5,6-dimethoxy groups, similar to cremastranone (Scheme 2). To accomplish this, the 4-chromanone 16 was coupled with a 3,4-disubstituted benzaldehyde (i.e., vanillin or 3,4-dibenzyloxybenzaldehyde) or a 4-substituted benzaldehyde (i.e., 4-methoxybenzaldehyde or 4-benzyloxybenzaldehyde) under acidic conditions to afford 3-benzylidene-4-chromanones (18a–18e). Catalytic hydrogenation of 18b and 18c, followed by trimethylsilyl iodide (TMSI)-promoted C5-demethylation of 15b and 15c provided the desired 2 and 3, respectively. In a similar manner, using catalytic hydrogenation 18d and 18e were transformed to 6 and 7, which were treated with TMSI to provide 4 and 5, respectively. The spectroscopic properties (1H- and 13C-NMR, mass spectrometry) of synthetic 2, 3, 4 [2], 5 [5], 6 [18], and 7 [19] were compatible with those of all the natural products (see Supplementary Materials).
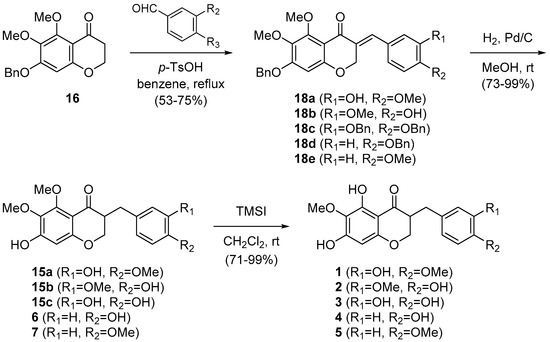
Scheme 2.
Synthesis of natural products, 5,6,7-trisubstituted 3-benzyl-4-chromanones.
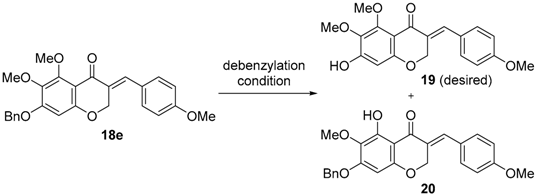
On the other hand, for the synthesis of three 3-benzylidene-4-chromanones (8–10) containing a 5,7-dihydroxy-6-methoxy group, the C7-benzyl group of the synthesized benzylidene intermediates (18c–18e) should be removed while keeping the C3–C9 double bond intact. With 18e in hand, we next attempted the debenzylation on C7 as shown in Table 2. Whereas the general condition of catalytic hydrogenation under H2 and Pd/C afforded the saturated 3-benzyl-4-chromanone such as the synthesis of 7, a small amount (3 mol %) of Pd/C catalyst and a short reaction time (5 min) led to the desired product 19 in moderate yield. TMSI and HBr selectively provided 19, which was demethylated only on the C5 position. But no product formation was observed with the treatment of TiCl and lithium naphthalenide. Unfortunately, the two and three benzyl groups of 18c and 18d, respectively, could not be cleaved simultaneously before the saturation of the C3–C9 double bond, using various conditions of catalytic hydrogenation.

Table 2.
C7-debenzylation conditions of 5,6,7-trisubstituted 3-benzylidene-4-chromanone (18e).
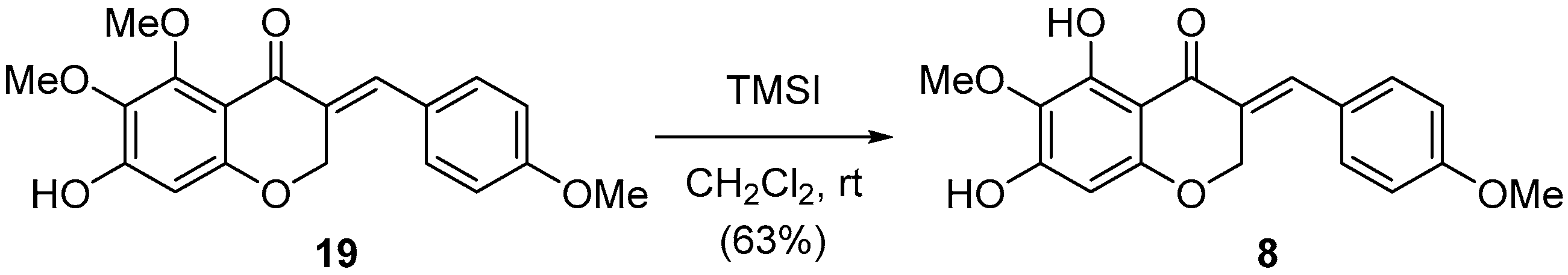
Finally, the debenzylated compound 19 was treated with TMSI to provide the desired 8 in good yield as shown in Scheme 3. The spectroscopic properties (1H- and 13C-NMR, mass spectrometry) of synthetic 8 were compatible with those of the natural product 8 [20]. Having failed to access 9 and 10 via the debenzylation strategy, further studies on other protecting groups on C7 such as PMB (4-methoxybenzyl), MOM (methoxymethyl) and Ms (methanesulfonyl) instead of benzyl ether are under investigation.
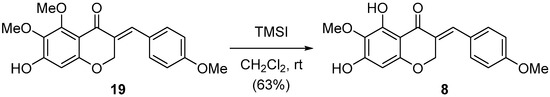
Scheme 3.
Synthesis of natural 3-benzylidene-4-chromanone 8.
3. Materials and Methods
3.1. General Information
All starting materials and reagents were obtained from commercial suppliers and were used without further purification. Air- and moisture-sensitive reactions were performed under an argon atmosphere. Flash column chromatography was performed using silica gel 60 (230–400 mesh, Merck KGaA, Darmstadt, Germany) with the indicated solvents. Thin-layer chromatography was performed using 0.25 mm silica gel plates (Merck KGaA). 1H- and 13C-NMR spectra were recorded on a Bruker 600 MHz spectrometer as solutions in deuteriochloroform (CDCl3) or methanol-d4 (Cambridge Isotope Laboratory, Andover, MA, USA). 1H-NMR data were reported in the order of chemical shift, multiplicity (s, singlet; d, doublet; t, triplet; m, multiplet; and/or multiple resonances), number of protons, and coupling constant (J) in hertz (Hz). High-resolution mass spectra (HRMS) were recorded on a JEOL JMS-700 (EI) (JEOL Ltd., Tokyo, Japan) and an Agilent 6530 Q-TOF LC/MS/MS system (ESI) (Agilent Technologies, Inc., Santa Clara, CA, USA).
3.2. Synthesis
7-(Benzyloxy)-5,6-dimethoxychroman-4-one (16). To a solution of the acetophenone (11) (100 mg, 0.33 mmol) in toluene (2.0 mL) was added N,N-dimethylformamide dimethyl acetal (52 μL, 0.39 mmol). After stirring for 18 h at 80 °C, the mixture was cooled to 0 °C and c-HCl (0.2 mL) was added. After stirring for 1 h at 50 °C. The reaction mixture was diluted with ethyl acetate and the organic phase was washed with water and brine, and dried over anhydrous MgSO4. The solvent was removed under reduced pressure and purified by flash column chromatography on silica gel (ethyl acetate:n-hexane = 1:2) to afford 7-(benzyloxy)-5,6-dimethoxy-4H-chromen-4-one (12) (101 mg, 97%). 1H-NMR (CDCl3, 600 MHz) δ 7.63 (d, 1H, J = 6.0 Hz), 7.46–7.40 (m, 4H), 7.37 (t, 1H, J = 6.6 Hz), 6.71 (s, 1H), 6.17 (d, 1H, J = 6.0 Hz), 5.20 (s, 2H), 3.97 (s, 3H), 3.92 (s, 3H); 13C-NMR (150 MHz, CDCl3) δ 176.2, 156.8, 154.6, 152.9, 140.7, 135.5, 128.8, 128.4, 127.2, 114.2, 113.8, 97.6, 70.9, 62.1, 61.5, 30.9. To a stirred solution of 4-chromenone (12) (10 mg, 0.03 mmol) in dry THF and Et2O (1:1) at −60 °C a solution of LiAlH4 in dry THF was added under N2 atmosphere. After stirring for 5 min, the reaction mixture was diluted with ethyl acetate and the organic phase was washed with water and brine, and dried over anhydrous MgSO4. The solvent was removed under reduced pressure and purified by flash column chromatography on silica gel (ethyl acetate:n-hexane = 1:2) to afford the 7-(benzyloxy)-5,6-dimethoxychroman-4-one (16) (10 mg, 99%). 1H-NMR (CDCl3, 600 MHz) δ 7.43–7.37 (m, 4H), 7.35–7.33 (m, 1H), 6.29 (s, 1H), 5.13 (s, 1H), 4.42 (t, 2H, J = 6.0 Hz), 3.92 (s, 3H), 3.82 (s, 3H), 2.71 (t, 2H, J = 6.0 Hz); 13C-NMR (150 MHz, CDCl3) δ 189.1, 159.8, 158.4, 154.4, 137.7, 135.8, 128.7, 128.2, 127.2, 109.8, 97.4, 70.6, 66.8, 61.6, 61.3, 38.7.
(E)-7-(Benzyloxy)-3-(3-hydroxy-4-methoxybenzylidene)-5,6-dimethoxychroman-4-one (18a). To a solution of the 4-chromanone (16) (80 mg, 0.26 mmol) in benzene (5 mL) was added 3-hydroxy-4-methoxybenzaldehyde (58 mg, 0.38 mmol) and p-toluenesulfonic acid (7 mg, 0.03 mmol) at 0 °C. The reaction mixture was refluxed for 12 h. After cooling to room temperature, the reaction mixture was concentrated under reduced pressure. The residue was purified by flash column chromatography on silica gel (ethyl acetate:n-hexane = 1:2) to afford the resulting 3-benzylidene-4-chromanone (18a) (60 mg, 53%). 1H-NMR (600 MHz, CDCl3) δ 7.73 (s, 1H), 7.42–7.38 (m, 4H), 7.35–7.33 (m, 1H), 6.90 (d, 1H, J = 8.4 Hz), 6.86–6.83 (m, 2H), 6.30 (s, 1H), 5.22 (d, 2H, J = 1.8 Hz), 5.13 (s, 2H), 3.99 (s, 3H), 3.94 (s, 3H), 3.85 (s, 3H); 13C-NMR (150 MHz, CDCl3) δ 179.6, 159.2, 158.2, 154.9, 147.6, 145.5, 138.1, 136.3, 135.8, 130.1, 128.7, 128.2, 128.0, 127.2, 123.2, 115.8, 110.8, 110.6, 97.5, 70.6, 67.6, 61.7, 61.3, 56.0; HRMS (EI): mass calculated for C26H24O7 [M+], 448.1522; found, 448.1521.
(E)-7-(Benzyloxy)-3-(4-hydroxy-3-methoxybenzylidene)-5,6-dimethoxychroman-4-one (18b). To a solution of the 4-chromanone (16) (80 mg, 0.26 mmol) in benzene (5 mL) was added 4-hydroxy-3-methoxybenzaldehyde (58 mg, 0.38 mmol) and p-toluenesulfonic acid (7 mg, 0.03 mmol) at 0 °C. The reaction mixture was refluxed for 12 h. After cooling to room temperature, the reaction mixture was concentrated under reduced pressure. The residue was purified by flash column chromatography on silica gel (ethyl acetate:n-hexane = 1:2) to afford the resulting 3-benzylidene-4-chromanone (18b) (65 mg, 57%). 1H-NMR (600 MHz, CDCl3) δ 7.76 (s, 1H), 7.43–7.38 (m, 4H), 7.35–7.32 (m, 1H), 6.97 (d, 1H, J = 8.4 Hz), 6.82 (d, 1H, J = 1.8 Hz), 6.79 (dd, 1H, J = 7.8 and 1.8 Hz), 6.30 (s, 1H), 5.22 (d, 2H, J = 1.2 Hz), 5.13 (s, 2H), 3.99 (s, 3H), 3.91 (s, 3H), 3.85 (s, 3H); 13C-NMR (150 MHz, CDCl3) δ 179.5, 159.1, 158.2, 154.9, 146.9, 146.5, 138.1, 136.5, 135.8, 129.8, 128.7, 128.2, 127.2, 127.1, 123.7, 114.6, 112.7, 110.8, 97.5, 70.6, 67.6, 61.7, 61.3, 56.0; HRMS (EI): mass calculated for C26H24O7 [M+], 448.1522; found, 448.1521.
(E)-7-(Benzyloxy)-3-(3,4-bis(benzyloxy)benzylidene)-5,6-dimethoxychroman-4-one (18c). To a solution of the 4-chromanone (16) (80 mg, 0.25 mmol) in benzene (5 mL) was added 3,4-bis(benzyloxy)benzaldehyde (120 mg, 0.38 mmol) and p-toluenesulfonic acid (7 mg, 0.03 mmol) at 0 °C. The reaction mixture was refluxed for 12 h. After cooling to room temperature, the reaction mixture was concentrated under reduced pressure. The residue was purified by flash column chromatography on silica gel (ethyl acetate:n-hexane = 1:2) to afford the resulting 3-benzylidene-4-chromanone (18c) (55 mg, 36%). 1H-NMR (600 MHz, CDCl3) δ 7.68 (s, 1H), 7.46–7.31 (m, 15H), 6.96 (d, 2H, J = 9.0 Hz), 6.82 (dd, 2H, J = 4.8 and 3.0 Hz), 6.29 (s, 1H), 5.22 (s, 2H), 5.19 (s, 2H), 5.13 (s, 2H), 5.04 (d, 2H, J = 1.8 Hz), 3.98 (s, 3H), 3.85 (s, 3H); 13C-NMR (150 MHz, CDCl3) δ 179.5, 159.1, 158.2, 154.9, 150.0, 148.4, 138.1, 136.8, 136.7, 136.1, 135.8, 130.1, 128.7, 128.6, 128.6, 128.4, 128.2, 128.0, 127.9, 127.5, 127.2, 127.2, 124.1, 116.9, 114.2, 110.8, 97.5, 71.4, 71.0, 70.6, 67.5, 61.7, 61.3; HRMS (EI): mass calculated for C39H34O7 [M+], 614.2305; found, 614.2308.
(E)-7-(Benzyloxy)-3-(4-(benzyloxy)benzylidene)-5,6-dimethoxychroman-4-one (18d). To a solution of the 4-chromanone (16) (121 mg, 0.39 mmol) in benzene (5 mL) was added 4-(benzyloxy)benzaldehyde (122 mg, 0.58 mmol) and p-toluenesulfonic acid (7 mg, 0.03 mmol) at 0 °C. The reaction mixture was refluxed for 12 h. After cooling to room temperature, the reaction mixture was concentrated under reduced pressure. The residue was purified by flash column chromatography on silica gel (ethyl acetate:n-hexane = 1:2) to afford the resulting 3-benzylidene-4-chromanone (18d) (104 mg, 54%). 1H-NMR (600 MHz, CDCl3) δ 7.78 (s, 1H), 7.44–7.38 (m, 8H), 7.35–7.33 (m, 2H), 7.24 (d, 2H, J = 9.0 Hz), 7.24 (dd, 2H, J = 6.6 and 1.8 Hz), 6.30 (s, 1H), 5.21 (d, 2H, J = 1.8 Hz), 5.13 (s, 2H), 5.11 (s, 2H), 4.00 (s, 3H), 3.85 (s, 3H); 13C-NMR (150 MHz, CDCl3) δ 179.6, 159.6, 159.1, 158.2, 154.9, 138.1, 136.4, 136.1, 135.8, 131.7, 129.8, 128.7, 128.7, 128.2, 128.1, 127.5, 127.4, 127.2, 115.0, 110.8, 97.5, 70.6, 70.1, 67.6, 61.7, 61.3; HRMS (EI): mass calculated for C32H28O6 [M+], 508.1886; found, 508.1885.
(E)-7-(Benzyloxy)-5,6-dimethoxy-3-(4-methoxybenzylidene)chroman-4-one (18e). To a solution of the 4-chromanone (16) (82 mg, 0.26 mmol) in benzene (5 mL) was added 4-methoxybenzaldehyde (50 μL, 0.39 mmol) and p-toluenesulfonic acid (7 mg, 0.03 mmol) at 0 °C. The reaction mixture was refluxed for 12 h. After cooling to room temperature, the reaction mixture was concentrated under reduced pressure. The residue was purified by flash column chromatography on silica gel (ethyl acetate:n-hexane = 1:2) to afford the resulting 3-benzylidene-4-chromanone (18e) (78 mg, 75%). 1H-NMR (600 MHz, CDCl3) δ 7.78 (s, 1H), 7.44–7.38 (m, 8H), 7.35–7.33 (m, 2H), 7.24 (d, 2H, J = 9.0 Hz), 7.24 (dd, 2H, J = 6.6 and 1.8 Hz), 6.30 (s, 1H), 5.21 (d, 2H, J = 1.8 Hz), 5.13 (s, 2H), 5.11 (s, 2H), 4.00 (s, 3H), 3.85 (s, 3H); 13C-NMR (150 MHz, CDCl3) δ 179.6, 159.6, 159.1, 158.2, 154.9, 138.1, 136.4, 136.1, 135.8, 131.7, 129.8, 128.7, 128.7, 128.2, 128.1, 127.5, 127.4, 127.2, 115.0, 110.8, 97.5, 70.6, 70.1, 67.6, 61.7, 61.3; HRMS (EI): mass calculated for C26H24O6 [M+], 432.1573; found, 432.1573.
7-Hydroxy-3-(3-hydroxy-4-methoxybenzyl)-5,6-dimethoxychroman-4-one (15a). A solution of the 3-benzylidene-4-chromanone (18a) (35 mg, 0.07 mmol) and 10% Pd/C (10 mg) in MeOH was placed under an atmosphere of hydrogen. After stirring for 1 h, the reaction mixture was diluted with ethyl acetate, filtered through a Celite pad, and concentrated under reduced pressure. The residue was purified by flash column chromatography on silica gel (ethyl acetate:n-hexane = 1:1) to afford the 3-benzyl-4-chromanone (15a) (22 mg, 87%). 1H-NMR (600 MHz, CD3OD) δ 6.82 (d, 1H, J = 14.4 Hz), 6.67 (d, 1H, J = 1.8 Hz), 6.63 (dd, 1H, J = 8.4 and 2.4 Hz), 6.16 (s, 1H), 4.21 (dd, 1H, J = 11.4 and 4.2 Hz), 4.04 (dd, 1H, J = 11.4 and 7.2 Hz), 3.82 (s, 3H), 3.79 (s, 3H), 3.75 (s, 3H), 3.00 (dd, 1H, J = 13.2 and 4.2 Hz), 2.66–2.60 (m, 1H), 2.58 (dd, 1H, J = 13.8 and 10.8 Hz); 13C-NMR (150 MHz, CD3OD) δ 192.4, 160.0, 158.5, 154.4, 146.3, 146.2, 136.4, 131.2, 119.9, 115.6, 111.5, 107.3, 99.1, 68.6, 60.4, 60.1, 55.0, 48.2, 32.0; HRMS (ESI): mass calcd for C19H20O7 [M + H+], 361.1281; found 361.1270.
7-Hydroxy-3-(4-hydroxy-3-methoxybenzyl)-5,6-dimethoxychroman-4-one (15b). A solution of the 3-benzylidene-4-chromanone (18b) (21 mg, 0.05 mmol) and 10% Pd/C (5 mg) in MeOH was placed under an atmosphere of hydrogen. After stirring for 1 h, the reaction mixture was diluted with ethyl acetate, filtered through a Celite pad and concentrated under reduced pressure. The residue was purified by flash column chromatography on silica gel (ethyl acetate:n-hexane = 1:1) to afford the 3-benzyl-4-chromanone (15b) (15 mg, 73%). 1H-NMR (600 MHz, CDCl3) δ 6.84 (d, 1H, J = 8.4 Hz), 6.73–6.69 (m, 2H), 6.31 (s, 1H), 4.26 (dd, 1H, J = 11.4 and 4.2 Hz), 4.10 (dd, 1H, J = 11.4 and 7.2 Hz), 3.91 (s, 3H), 3.91 (s, 3H), 3.87 (s, 3H), 3.16 (dd, 1H, J = 13.8 and 4.2 Hz), 2.73 (m, 1H), 2.65–2.61 (m, 1H); 13C-NMR (150 MHz, CDCl3) δ 191.6, 159.8, 155.5, 153.5, 146.5, 144.2, 135.2, 130.2, 121.9, 114.3, 111.4, 108.7, 98.8, 68.8, 61.5, 61.4, 55.9, 48.6, 32.6; HRMS (EI): mass calculated for C19H20O7 [M+], 360.1209; found, 360.1208.
3-(3,4-Dihydroxybenzyl)-7-hydroxy-5,6-dimethoxychroman-4-one (15c). A solution of the 3-benzylidene-4-chromanone (18c) (20 mg, 33 μmol) and 10% Pd/C (3.5 mg) in MeOH was placed under an atmosphere of hydrogen. After stirring for 1 h, the reaction mixture was diluted with ethyl acetate, filtered through a Celite pad and concentrated under reduced pressure. The residue was purified by flash column chromatography on silica gel (ethyl acetate:n-hexane = 1:1) to afford the 3-benzyl-4-chromanone (15c) (11 mg, 99%). 1H-NMR (600 MHz, CDCl3) δ 6.79 (d, 1H, J = 8.4 Hz), 6.77 (d, 1H, J = 2.4 Hz), 6.63 (dd, 1H, J = 8.4 and 1.8 Hz), 6.30 (s, 1H), 4.26 (dd, 1H, J = 11.4 and 4.2 Hz), 4.09 (dd, 1H, J = 8.4 and 4.2 Hz), 3.90 (s, 3H), 3.89 (s, 3H), 3.08 (dd, 1H, J = 13.8 and 4.2 Hz), 2.70–2.67 (m, 1H), 2.62 (dd, 1H, J = 13.8 and 10.8 Hz); 13C-NMR (150 MHz, CDCl3) δ 192.2, 159.9, 155.9, 153.5, 143.8, 142.5, 135.2, 131.0, 121.6, 116.0, 115.3, 108.5, 98.9, 68.8, 61.5, 61.4, 48.5, 32.4; HRMS (EI): mass calculated for C18H18O7 [M+], 346.1053; found, 346.1056.
7-Hydroxy-3-(4-hydroxybenzyl)-5,6-dimethoxychroman-4-one (6). A solution of the 3-benzylidene-4-chromanone (18d) (23 mg, 0.05 mmol) and 10% Pd/C (5 mg) in MeOH was placed under an atmosphere of hydrogen. After stirring for 1 h, the reaction mixture was diluted with ethyl acetate, filtered through a Celite pad and concentrated under reduced pressure. The residue was purified by flash column chromatography on silica gel (ethyl acetate:n-hexane = 1:1) to afford the 3-benzyl-4-chromanone (6) (14 mg, 94%). 1H-NMR (600 MHz, CDCl3) δ 7.09 (d, 2H, J = 8.4 Hz), 6.78 (d, 2H, J = 8.4 Hz), 6.30 (s, 1H), 4.62 (dd, 1H, J = 11.4 and 4.2 Hz), 4.08 (dd, 1H, J = 11.4 and 7.2 Hz), 3.91 (s, 3H), 3.90 (s, 3H), 3.15 (dd, 1H, J = 13.8 and 4.2 Hz), 2.73–2.69 (m, 1H), 2.66 (dd, 1H, J = 13.8 and 10.2 Hz); 13C-NMR (150 MHz, CDCl3) δ 191.7, 159.8, 155.6, 154.3, 153.5, 135.2, 130.3, 130.3, 115.4, 108.6, 98.8, 68.8, 61.5, 61.4, 48.5, 32.0; HRMS (EI): mass calculated for C18H18O6 [M+], 330.1103; found, 330.1102.
7-Hydroxy-5,6-dimethoxy-3-(4-methoxybenzyl)chroman-4-one (7). A solution of the 3-benzylidene-4-chromanone (18e) (23 mg, 0.05 mmol) and 10% Pd/C (5 mg) in MeOH was placed under an atmosphere of hydrogen. After stirring for 1 h, the reaction mixture was diluted with ethyl acetate, filtered through a Celite pad and concentrated under reduced pressure. The residue was purified by flash column chromatography on silica gel (ethyl acetate:n-hexane = 1:2) to afford the 3-benzyl-4-chromanone (7) (13 mg, 80%). 1H-NMR (600 MHz, CD3COCD3) δ 7.07–7.05 (m, 2H), 6.76–6.73 (m, 2H), 6.09 (dd, 1H, J = 4.8 and 3.0 Hz), 4.14 (d, 1H, J = 4.2 Hz), 3.94 (d, 1H, J = 8.4 Hz), 3.71 (s, 3H), 3.71–3.62 (m, 6H), 3.00–3.98 (m, 1H), 2.62–2.61 (m, 1H), 2.53–2.49 (m, 1H); 13C-NMR (150 MHz, CDCl3) δ 190.0, 159.6, 158.5, 157.1, 154.7, 136.3, 160.8, 130.8, 130.1, 113.9, 108.5, 99.1, 69.1, 60.8, 60.6, 54.6, 48.3, 31.5; HRMS (EI): mass calculated for C19H20O6 [M+], 344.1260; found, 344.1257.
7-Hydroxy-3-(3-hydroxy-4-methoxybenzyl)-5,6-dimethoxychroman-4-one (2). To a solution of the 7-hydroxy-5,6-dimethoxy-4-chromanone (15b) (10 mg, 0.03 mmol) in CH2Cl2 (5 mL) was added TMSI (5 μL, 0.09 mmol) at 0 °C for 1 h. The mixture was concentrated in vacuo. The residue was purified by flash column chromatography on silica gel (ethyl acetate:n-hexane = 1:2) to afford the 5,7-dihydroxy-6-methoxy-4-chromanone (2) (7.1 mg, 71%). 1H-NMR (600 MHz, CD3OD) δ 6.81 (d, 1H, J = 1.8 Hz), 6.73 (d, 1H, J = 7.8 Hz), 6.67 (dd, 1H, J = 8.4 and 1.8 Hz), 5.90 (s, 1H), 4.25 (dd, 1H, J = 11.4 and 4.2 Hz), 4.09 (dd, 1H, J = 11.4 and 7.2 Hz), 3.83 (s, 3H), 3.77 (s, 3H), 3.12 (dd, 1H, J = 13.8 and 4.8 Hz), 2.86–2.81 (m, 1H), 2.68 (d, 1H, J = 10.2 Hz); 13C-NMR (150 MHz, CD3OD) δ 199.3, 160.1, 159.3, 156.0, 148.2, 145.5, 130.0, 129.6, 121.9, 115.4, 112.8, 102.1, 95.0, 69.5, 60.1, 55.5, 47.3, 32.7; HRMS (EI): mass calculated for C18H18O7 [M+], 346.1053; found, 346.1054.
3-(3,4-Dihydroxybenzyl)-5,7-dihydroxy-6-methoxychroman-4-one (3). To a solution of the 7-hydroxy-5,6-dimethoxy-4-chromanone (15c) (20 mg, 0.03 mmol) in CH2Cl2 (5 mL) was added TMSI (5 μL, 0.09 mmol) at 0 °C for 1 h. The mixture was concentrated in vacuo. The residue was purified by flash column chromatography on silica gel (ethyl acetate:n-hexane = 1:2) to afford the 5,7-dihydroxy-6-methoxy-4-chromanone (3) (12 mg, 99%). 1H-NMR (600 MHz, CD3OD) δ 6.71 (d, 1H, J = 8.4 Hz), 6.67 (d, 1H, J = 2.4 Hz), 6.55 (dd, 1H, J = 7.8 and 1.8 Hz), 5.91 (s, 1H), 4.24 (dd, 1H, J = 11.4 and 4.2 Hz), 4.08 (dd, 1H, J = 11.4 and 7.2 Hz), 3.77 (s, 3H), 3.05 (dd, 1H, J = 13.8 and 4.2 Hz), 2.80–2.75 (m, 1H), 2.60 (dd, 1H, J = 13.8 and 10.2 Hz); 13C-NMR (150 MHz, CD3OD) δ 200.0, 160.5, 159.9, 156.7, 146.2, 144.9, 130.6, 130.2, 121.2, 116.9, 116.2, 102.8, 95.5, 70.0, 60.7, 47.9, 33.0; HRMS (EI): mass calculated for C17H16O7 [M+], 332.0896; found, 332.0898.
5,7-Dihydroxy-3-(4-hydroxybenzyl)-6-methoxychroman-4-one (4). To a solution of the 7-hydroxy-5,6-dimethoxy-4-chromanone (6) (10 mg, 0.03 mmol) in CH2Cl2 (5 mL) was added TMSI (5 μL, 0.09 mmol) at 0 °C for 1 h. The mixture was concentrated in vacuo. The residue was purified by flash column chromatography on silica gel (ethyl acetate:n-hexane = 1:2) to afford the resulting 5,7-dihydroxy-6-methoxy-4-chromanone (4) (9.8 mg, 86%). 1H-NMR (600 MHz, CD3OD) δ 7.06 (d, 2H, J = 8.4 Hz), 6.73 (d, 2H, J = 8.4 Hz), 5.90 (s, 1H), 4.23 (dd, 1H, J = 11.4 and 4.2 Hz), 4.07 (dd, 1H, J = 10.8 and 7.2 Hz), 3.77 (s, 3H), 3.10 (dd, 1H, J = 13.8 and 4.8 Hz), 2.82–2.77 (m, 1H), 2.67 (dd, 1H, J = 13.8 and 10.2 Hz); 13C-NMR (150 MHz, CD3OD) δ 199.8, 160.4, 159.7, 156.8, 156.5, 130.7, 130.0, 129.7, 116.0, 102.5, 95.4, 69.8, 60.5, 47.8, 32.6; HRMS (EI): mass calculated for C17H16O6 [M+], 316.0947; found, 316.0945.
5,7-Dihydroxy-6-methoxy-3-(4-methoxybenzyl)chroman-4-one (5). To a solution of the 7-hydroxy-5,6-dimethoxy-4-chromanone (7) (19 mg, 0.06 mmol) in CH2Cl2 (5 mL) was added TMSI (10 μL, 0.18 mmol) at 0 °C for 1 h. The mixture was concentrated in vacuo. The residue was purified by flash column chromatography on silica gel (ethyl acetate:n-hexane = 1:2) to afford the resulting 5,7-dihydroxy-6-methoxy-4-chromanone (5) (15 mg, 80%). 1H-NMR (600 MHz, CD3OD) δ 7.15 (d, 2H, J = 8.4 Hz), 6.86 (d, 2H, J = 8.4 Hz), 5.90 (s, 1H), 4.23 (dd, 1H, J = 11.4 and 4.2 Hz), 4.06 (dd, 1H, J = 11.4 and 7.8 Hz), 3.77 (s, 3H), 3.76 (s, 3H), 3.13 (dd, 1H, J = 13.8 and 4.8 Hz), 2.84–2.79 (m, 1H), 2.70 (dd, 1H, J = 13.8 and 10.2 Hz); 13C-NMR (150 MHz, CD3OD) δ 200.1, 160.7, 160.1, 160.0, 156.9, 131.4, 131.2, 130.4, 115.1, 103.0, 95.8, 70.3, 61.0, 55.7, 48.1, 32.9; HRMS (EI): mass calculated for C18H18O6 [M+], 330.1103; found, 330.1102.
(E)-7-Hydroxy-5,6-dimethoxy-3-(4-methoxybenzylidene)chroman-4-one (19). A solution of the 3-(4-methoxybenzylidene)-4-chromanone (18e) (37 mg, 0.09 mmol) and 3% Pd/C (4.8 mg) in MeOH was placed under an atmosphere of hydrogen. After stirring for 5 min, the reaction mixture was diluted with ethyl acetate, filtered through a Celite pad and concentrated under reduced pressure. The residue was purified by flash column chromatography on silica gel (ethyl acetate:n-hexane = 1:3) to afford the debenzylated 3-(4-methoxybenzylidene)-4-chromanone (19) (11 mg, 35%). 1H-NMR (600 MHz, CDCl3) δ 7.78 (s, 1H), 7.26 (d, 2H, J = 7.8 Hz), 6.96 (d, 2H, J = 9.0 Hz), 6.31 (s, 1H), 5.22 (d, 2H, J = 1.8 Hz), 3.97 (s, 3H), 3.94 (s, 3H), 3.85 (s, 3H); 13C-NMR (100 MHz, CDCl3) δ 179.7, 160.4, 159.4, 155.3, 153.8, 136.2, 135.4, 131.7, 129.6, 127.1, 114.1, 110.4, 98.9, 67.4, 61.5, 61.4, 55.3; HRMS (EI): mass calculated for C19H18O6 [M+], 342.1103; found, 342.1106.
(E)-5,7-Dihydroxy-6-methoxy-3-(4-methoxybenzylidene)chroman-4-one (8). To a solution of the 3-(4-methoxybenzylidene)-4-chromanone (19) (15 mg, 0.04 mmol) in CH2Cl2 (5 mL) was added TMSI (13 μL, 0.09 mmol) at 0 °C for 1 h. The mixture was concentrated in vacuo. The residue was purified by flash column chromatography on silica gel (ethyl acetate:n-hexane = 1:3) to afford the resulting 5,7-dihydroxy-6-methoxy-4-chromanone (8) (9.1 mg, 63%). 1H-NMR (600 MHz, CDCl3) δ 7.80 (s, 1H), 7.27 (d, 2H, J = 7.2 Hz), 6.97 (d, 2H, J = 8.4 Hz), 6.03 (s, 1H), 5.28 (d, 2H, J = 1.8 Hz), 3.95 (s, 3H), 3.86 (s, 3H); 13C-NMR (100 MHz, CDCl3) δ 185.9, 160.8, 157.8, 157.2, 155.2, 137.2, 132.0, 128.4, 127.3, 126.7, 114.2, 103.3, 94.0, 67.4, 60.9, 55.4; HRMS (EI): mass calculated for C18H16O6 [M+], 328.0947; found, 328.0945.
4. Conclusions
In summary, we have successfully demonstrated the first total synthesis of naturally occurring homoisoflavonoids containing either a 7-hydroxy-5,6-dimethoxy or 5,7-dihydroxy-6-methoxy group on the A ring in four or five steps from the commercially available 11. The key features of this synthetic route involve the following: (1) LiAlH4-mediated selective 1,4-reduction of 4-chromenone; (2) the aldol condensation of arylaldehydes, followed by catalytic hydrogenation; (3) TMSI-mediated C5-demethylation; (4) the selective debenzylation of 3-benzylidene-4-chromanone. These studies provide a timely contribution to the development of a practical synthetic approach to a variety of homoisoflavonoid analogues. Extension of this approach in the synthesis of various natural products and analogues and establishment of structure-activity relationship (SAR) profiles for 5,6,7-trisubstituted homoisoflavonoids compared to antiangiogenic cremastranone are currently underway.
Supplementary Materials
Supplementary materials can be accessed at: http://www.mdpi.com/1420-3049/21/8/1058/s1.
Acknowledgments
This work was supported by grants from the Basic Science Research Program through the National Research Foundation of Korea (NRF) funded by the Ministry of Education (NRF-2013R1A1A2007151) and the Korea Health Technology R & D Project through the Korea Health Industry Development Institute (KHIDI), funded by the Ministry of Health & Welfare (HI14C1135) to S.-Y.S.; grants from the Bright Focus Foundation, and NIH/NEI R01EY025641 to T.W.C., and an unrestricted grant from Research to Prevent Blindness, Inc.
Author Contributions
H.L. and Y.Y. performed the experiments and analyzed all data. I.R. and T.W.C. conceived and suggested the research work. S.-Y.S. conceived and directed the investigations and composed the manuscript with revisions provided by the other authors. All authors read and approved the manuscript.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Lee, B.; Basavarajappa, H.D.; Sulaiman, R.S.; Fei, X.; Seo, S.-Y.; Corson, T.W. First synthesis of the antiangiogenic homoisoflavanone, cremastranone. Org. Biomol. Chem. 2014, 12, 7673–7677. [Google Scholar] [CrossRef] [PubMed]
- Adinolfi, M.; Corsaro, M.M.; Lanzetta, R.; Laonigro, G.; Mangoni, L.; Parrilli, M. Ten homoisoflavanones from two Muscari species. Phytochemistry 1987, 26, 285–290. [Google Scholar] [CrossRef]
- Corsaro, M.M.; Lanzetta, R.; Mancino, A.; Parrilli, M. Homoisoflavanones from Chionodoxa luciliae. Phytochemistry 1992, 31, 1395–1397. [Google Scholar] [CrossRef]
- Crouch, N.R.; Bangani, V.; Mulholland, D.A. Homoisoflavanones from three South African: Scilla species. Phytochemistry 1999, 51, 943–946. [Google Scholar] [CrossRef]
- Du Toit, K.; Elgorashi, E.E.; Malan, S.F.; Drewes, S.E.; van Staden, J.; Crouch, N.R.; Mulholland, D.A. Anti-inflammatory activity and QSAR studies of compounds isolated from Hyacinthaceae species and Tachiadenus longiflorus Griseb. (Gentianaceae). Bioorg. Med. Chem. 2005, 13, 2561–2568. [Google Scholar] [CrossRef] [PubMed]
- Shim, J.S.; Kim, J.H.; Lee, J.; Kim, S.N.; Kwon, H.J. Anti-angiogenic activity of a homoisoflavanone from Cremastra appendiculata. Planta Med. 2004, 70, 171–173. [Google Scholar] [PubMed]
- Du Toit, K.; Drewes, S.E.; Bodenstein, J. The chemical structures, plant origins, ethnobotany and biological activities of homoisoflavanones. Nat. Prod. Res. 2010, 24, 457–490. [Google Scholar] [CrossRef] [PubMed]
- Mulholland, D.A.; Schwikkard, S.L.; Crouch, N.R. The chemistry and biological activity of the Hyacinthaceae. Nat. Prod. Rep. 2013, 30, 1165–1210. [Google Scholar] [CrossRef] [PubMed]
- Famuyiwa, S.O.; Ntumy, A.N.; Andrae-Marobela, K.; Yeboah, S.O. A new homoisoflavonoid and the bioactivities of some selected homoisoflavonoids from the inter-bulb surfaces of Scilla nervosa subsp. rigidifolia. S. Afr. J. Bot. 2013, 88, 17–22. [Google Scholar] [CrossRef]
- Nishida, Y.; Sugahara, S.; Wada, K.; Toyohisa, D.; Tanaka, T.; Ono, M.; Yasuda, S. Inhibitory effects of the ethyl acetate extract from bulbs of Scilla scilloides on lipoxygenase and hyaluronidase activities. Pharm. Biol. 2014, 52, 1351–1357. [Google Scholar] [CrossRef] [PubMed]
- Hafez Ghoran, S.; Saeidnia, S.; Babaei, E.; Kiuchi, F.; Hussain, H. Scillapersicene: A new homoisoflavonoid with cytotoxic activity from the bulbs of Scilla persica HAUSSKN. Nat. Prod. Res. 2016, 30, 1309–1314. [Google Scholar] [CrossRef] [PubMed]
- Farkas, L.; Gottsegen, Á.; Nógrádi, M.; Strelisky, J. Synthesis of homoisoflavanones—II: Constituents of Eucomis autumn alis and E. Punctata. Tetrahedron 1971, 27, 5049–5054. [Google Scholar] [CrossRef]
- Basavarajappa, H.D.; Lee, B.; Fei, X.; Lim, D.; Callaghan, B.; Mund, J.A.; Case, J.; Rajashekhar, G.; Seo, S.-Y.; Corson, T.W. Synthesis and mechanistic studies of a novel homoisoflavanone inhibitor of endothelial cell growth. PLoS ONE 2014, 9, e95694. [Google Scholar]
- Basavarajappa, H.D.; Lee, B.; Lee, H.; Sulaiman, R.S.; An, H.; Magaña, C.; Shadmand, M.; Vayl, A.; Rajashekhar, G.; Kim, E.-Y.; et al. Synthesis and Biological Evaluation of Novel Homoisoflavonoids for Retinal Neovascularization. J. Med. Chem. 2015, 58, 5015–5027. [Google Scholar] [CrossRef] [PubMed]
- Sulaiman, R.S.; Merrigan, S.; Quigley, J.; Qi, X.; Lee, B.; Boulton, M.E.; Kennedy, B.; Seo, S.-Y.; Corson, T.W. A novel small molecule ameliorates ocular neovascularisation and synergises with anti-VEGF therapy. Sci. Rep. 2016, 6, 25509. [Google Scholar] [CrossRef] [PubMed]
- Cox, B.; Waigh, R.D. A Modified Synthesis of 4-Chromanones. Synthesis 1989, 9, 709–710. [Google Scholar] [CrossRef]
- Salakka, A.K.; Jokela, T.H.; Wähälä, K. Multiple hydride reduction pathways in isoflavonoids. Beilstein J. Org. Chem. 2006, 2, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Koorbanally, N.A.; Crouch, N.R.; Harilal, A.; Pillay, B.; Mulholland, D.A. Coincident isolation of a novel homoisoflavonoid from Resnova humifusa and Eucomis montana (Hyacinthoideae: Hyacinthaceae). Biochem. Syst. Ecol. 2006, 34, 114–118. [Google Scholar] [CrossRef]
- Famuyiwa, S.O.; Sichilongo, K.F.; Yeboah, S.O.; Abegaz, B.M. Homoisoflavonoids from the inter-bulb surfaces of Scilla nervosa subsp. rigidifolia. Phytochem. Lett. 2012, 5, 591–595. [Google Scholar] [CrossRef]
- Silayo, A.; Ngadjui, B.T.; Abegaz, B.M. Homoisoflavonoids and stilbenes from the bulbs of Scilla nervosa subsp. rigidifolia. Phytochemistry 1999, 52, 947–955. [Google Scholar] [CrossRef]
- Sample Availability: Samples of the compounds 1–8 are available from the authors.
© 2016 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license ( http://creativecommons.org/licenses/by/4.0/).