Construction of an Immobilized Thermophilic Esterase on Epoxy Support for Poly(ε-caprolactone) Synthesis
Abstract
:1. Introduction
2. Results and Discussion
2.1. Immobilization of Thermophilic Esterase on Epoxy-Sepabeads EC-EP
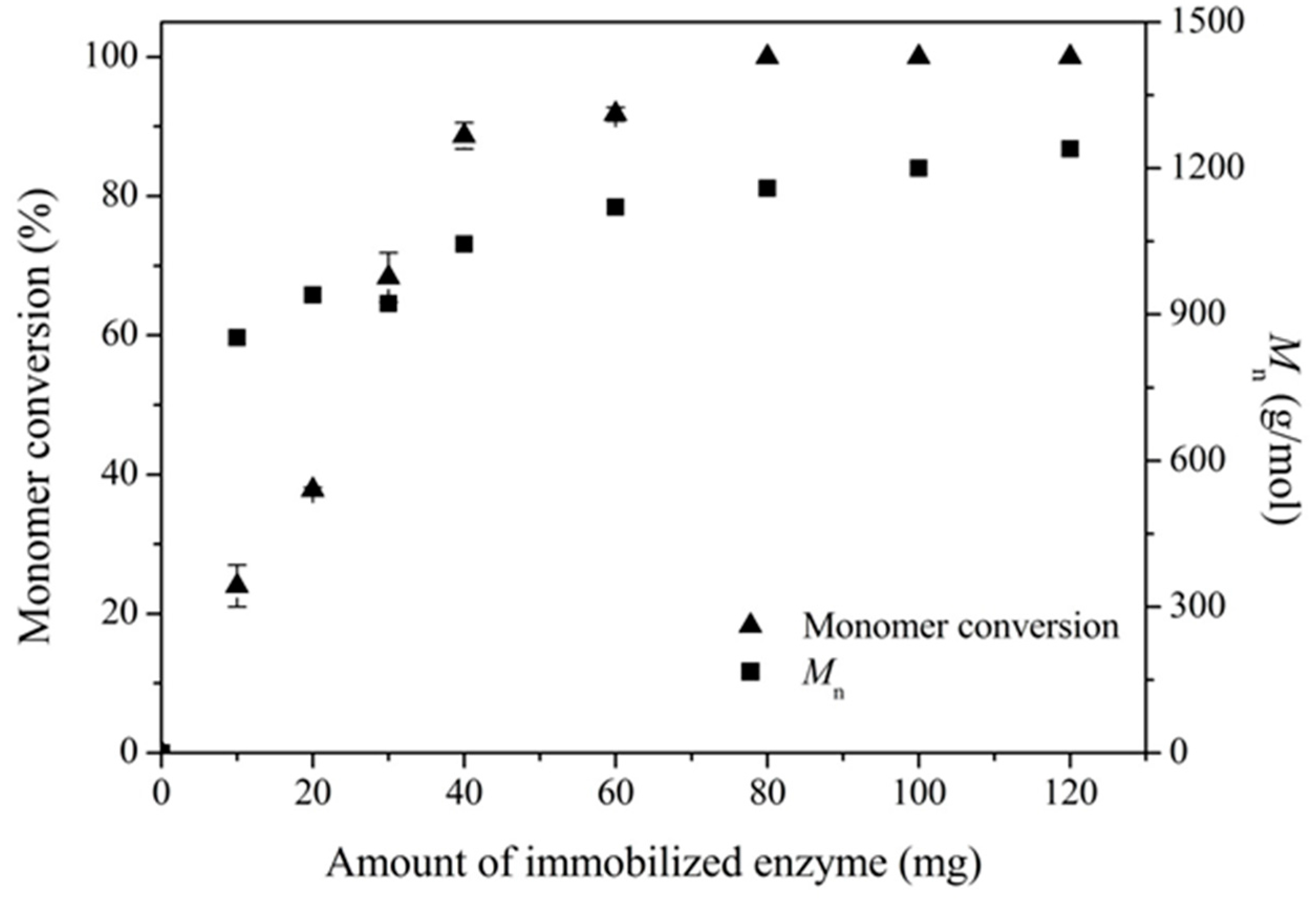
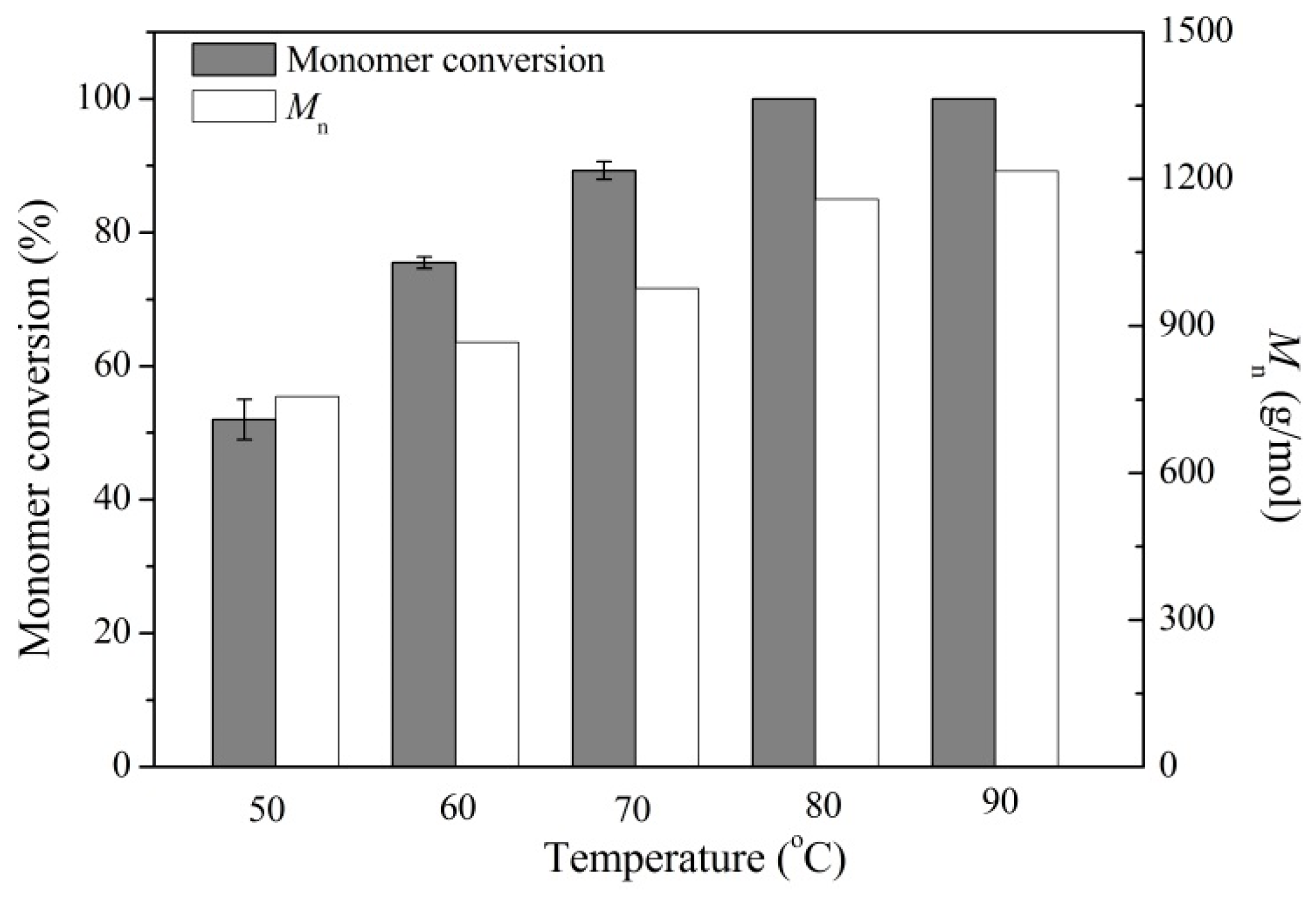
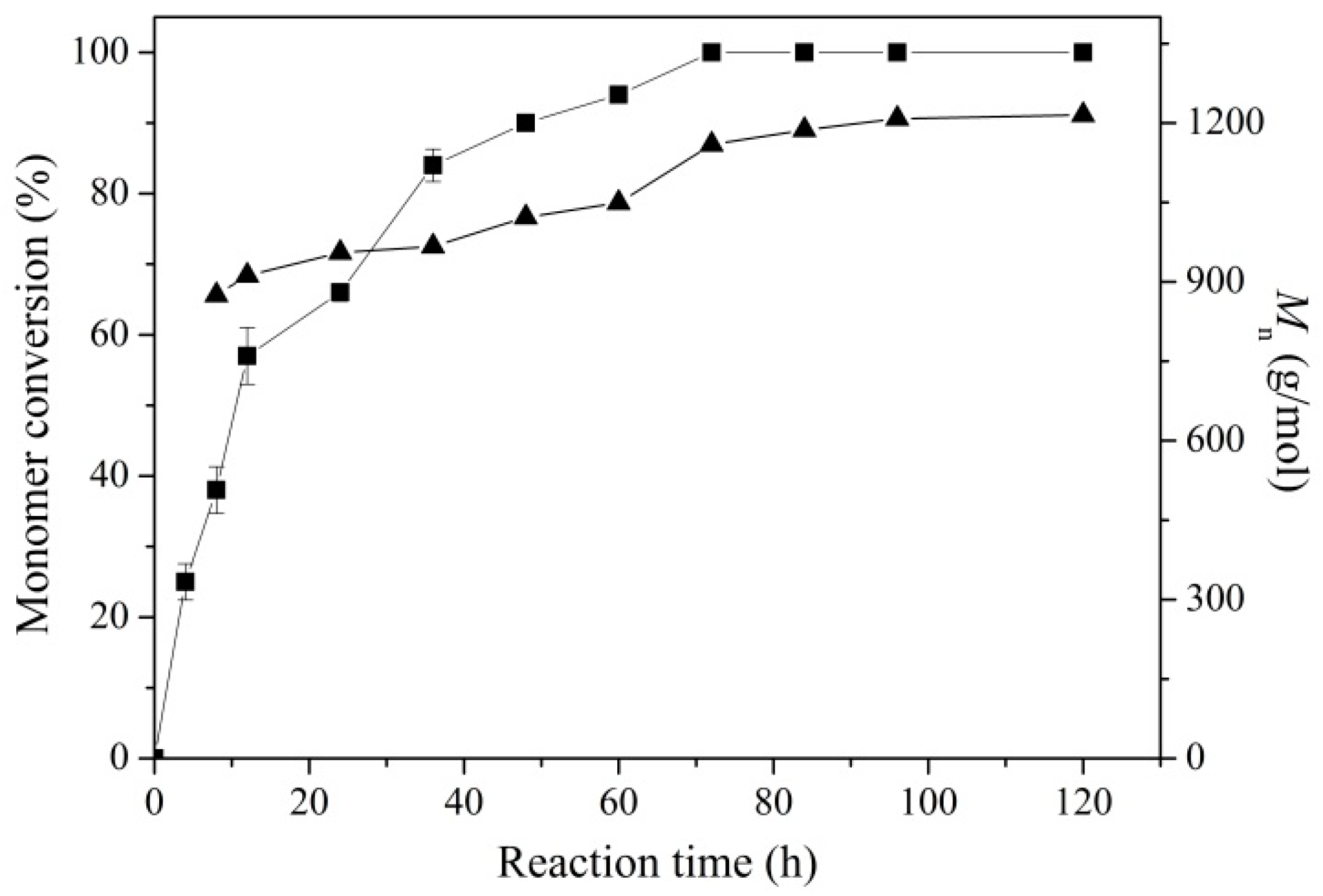
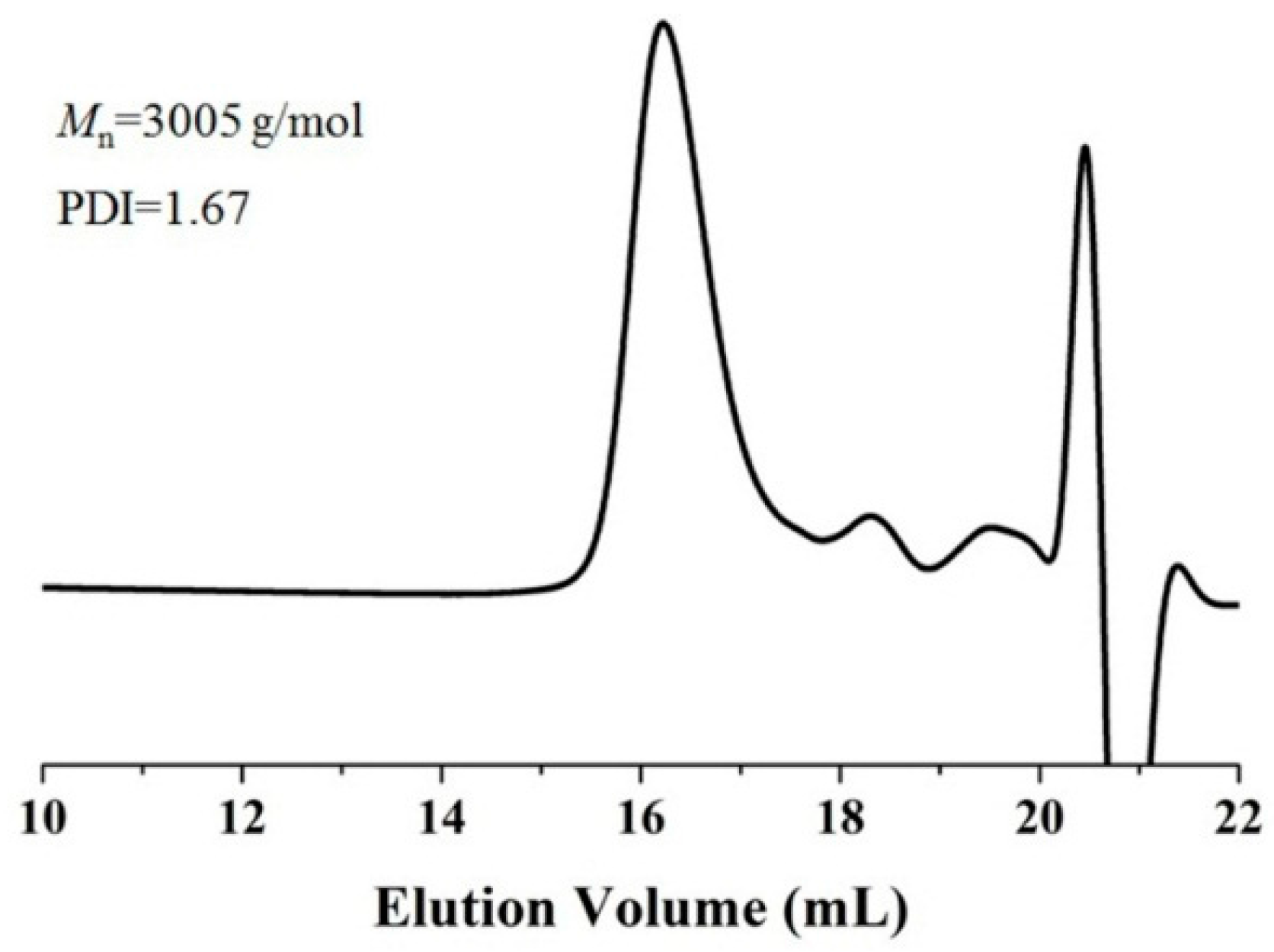
2.2. Enzymatic Synthesis of PCL Using Immobilized Enzyme EC-EP-AFEST
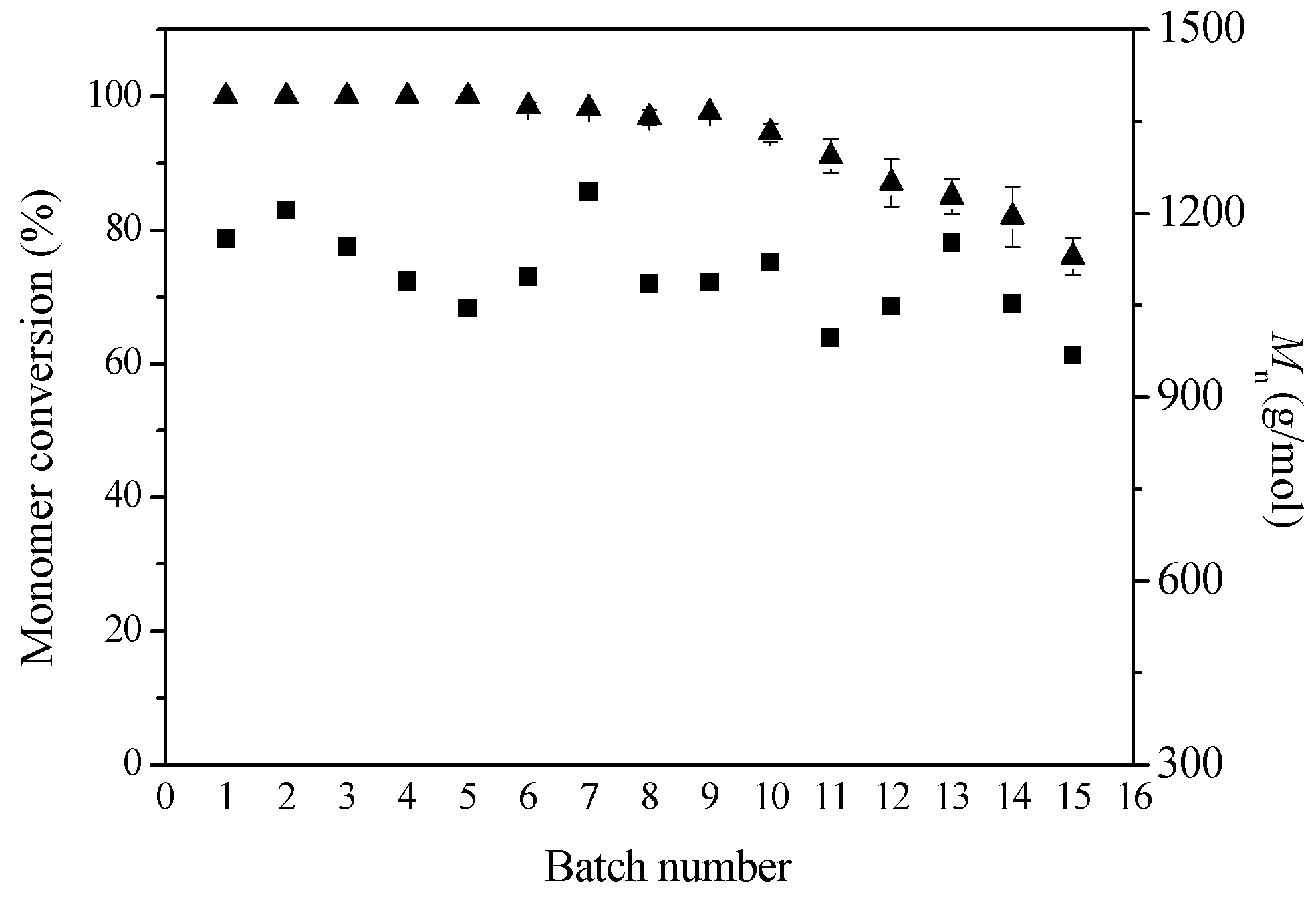
2.3. Operational Stability Analysis
2.4. Gram-Scale Synthesis of PCL Using Immobilized Enzyme EC-EP-AFEST
3. Materials and Methods
3.1. Materials
3.2. Immobilization of Thermophilic Esterase on Epoxy-Sepabeads EC-EP
3.3. Enzymatic PCL Synthesis
3.4. Determination of Monomer Conversion
3.5. Determination of Mn and PDI
3.6. Operational Stability Assay
3.7. Gram-Scale Synthesis of PCL
4. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
Abbreviations
| PCL | Poly(ε-caprolactone) |
| Mn | Number-average molecular weight |
| PDI | Polydispersity index |
| SEM | Scanning electron microscope |
| AFEST | Archaeoglobus fulgidus esterase |
| GC | Gas chromatography |
| CDCl3 | Chloroform-d |
| GPC | Gel permeation chromatography |
References
- Ikada, Y.; Tsuji, H. Biodegradable polyesters for medical and ecological applications. Macromol. Rapid Commun. 2000, 21, 117–132. [Google Scholar] [CrossRef]
- Albertsson, A.C.; Srivastava, R.K. Recent developments in enzyme-catalyzed ring-opening polymerization. Adv. Drug Deliv. Rev. 2008, 60, 1077–1093. [Google Scholar] [CrossRef] [PubMed]
- Kobayashi, S. Recent developments in lipase-catalyzed synthesis of polyesters. Macromol. Rapid Commun. 2009, 30, 237–260. [Google Scholar] [CrossRef] [PubMed]
- Kobayashi, S.; Makino, A. Enzymatic polymer synthesis: An opportunity for green polymer chemistry. Chem. Rev. 2009, 109, 5288–5353. [Google Scholar] [CrossRef] [PubMed]
- Yang, Y.; Yu, Y.; Zhang, Y.; Liu, C.; Shi, W.; Li, Q. Lipase/esterase-catalyzed ring-opening polymerization: A green polyester synthesis technique. Process Biochem. 2011, 46, 1900–1908. [Google Scholar] [CrossRef]
- Shoda, S.; Uyama, H.; Kadokawa, J.; Kimura, S.; Kobayashi, S. Enzymes as green catalysts for precision macromolecular synthesis. Chem. Rev. 2016, 116, 2317–2413. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Shi, H.; Wu, D.; Xing, Z.; Zhang, A.; Yang, Y.; Li, Q. Recent developments in lipase-catalyzed synthesis of polymeric materials. Process Biochem. 2014, 49, 797–806. [Google Scholar] [CrossRef]
- Yang, Y.; Zhang, J.; Wu, D.; Xing, Z.; Zhou, Y.; Shi, W.; Li, Q. Chemoenzymatic synthesis of polymeric materials using lipases as catalysts: A review. Biotechnol. Adv. 2014, 32, 642–651. [Google Scholar] [CrossRef] [PubMed]
- Xiang, S.; Zhang, Q.; Zhang, G.; Jiang, W.; Wang, Y.; Zhou, H.; Li, Q.; Tang, J. Facile synthesis of block copolymers by tandem ROMP and eROP from esters precursors. Biomacromolecules 2014, 15, 3112–3118. [Google Scholar] [CrossRef] [PubMed]
- Jiang, W.; An, N.; Zhang, Q.; Xiang, S.; Bai, Z.; Han, H.; Li, X.; Li, Q.; Tang, J. One-pot combination of eROP and ROMP for the synthesis of block copolymers. Macromol. Chem. Phys. 2015, 216, 2107–2114. [Google Scholar] [CrossRef]
- Henderson, L.A.; Svirkin, Y.Y.; Gross, R.A.; Kaplan, D.L.; Swift, G. Enzyme-catalyzed polymerizations of ε-caprolactone: Effects of initiators on product structure, propagation kinetics, and mechanism. Macromolecules 1996, 29, 7759–7766. [Google Scholar] [CrossRef]
- Namekawa, S.; Suda, S.; Uyama, H.; Kobayashi, S. Lipase-catalyzed ring-opening polymerization of lactones to polyesters and its mechanistic aspects. Int. J. Biol. Macromol. 1999, 25, 145–151. [Google Scholar] [CrossRef]
- Schmidt, S.; Scherkus, C.; Muschiol, J.; Menyes, U.; Winkler, T.; Hummel, W.; Groger, H.; Leise, A.; Herz, H.G.; Bornscheuer, U.T. An enzyme cascade synthesis of ε-caprolactone and its oligomers. Angew. Chem. Int. Ed. 2015, 54, 2784–2787. [Google Scholar] [CrossRef] [PubMed]
- Kumar, A.; Gross, R.A. Candida antarctica lipase B catalyzed polycaprolactone synthesis: Effects of organic media and temperature. Biomacromolecules 2000, 1, 133–138. [Google Scholar] [CrossRef] [PubMed]
- Hunsen, M.; Azim, A.; Mang, H.; Wallner, S.R.; Ronkvist, A.; Xie, W.; Gross, R.A. A cutinase with polyester synthesis activity. Macromolecules 2007, 40, 148–150. [Google Scholar] [CrossRef]
- Levisson, M.; van der Oost, J.; Kengen, S.W.M. Carboxylic ester hydrolases from hyperthermophiles. Extremophiles 2009, 13, 567–581. [Google Scholar] [CrossRef] [PubMed]
- Elleuche, S.; Schroder, C.; Sahm, K.; Antranikian, G. Extremozymes—Biocatalysts with unique properties from extremophilic microorganisms. Curr. Opin. Biotechnol. 2014, 29, 116–123. [Google Scholar] [CrossRef] [PubMed]
- Sarmiento, F.; Peralta, R.; Blamey, J.M. Cold and hot extremozymes: Industrial relevance and current trends. Front. Bioeng. Biotechnol. 2015, 3, 148. [Google Scholar] [CrossRef] [PubMed]
- Ma, J.; Li, Q.; Song, B.; Liu, D.; Zheng, B.; Zhang, Z.; Feng, Y. Ring-opening polymerization of ε-caprolactone catalyzed by a novel thermophilic esterase from the archaeon Archaeoglobus fulgidus. J. Mol. Catal. B Enzym. 2009, 56, 151–157. [Google Scholar] [CrossRef]
- DiCosimo, R.; McAuliffe, J.; Poulose, A.J.; Bohlmann, G. Industrial use of immobilized enzymes. Chem. Soc. Rev. 2013, 42, 6437–6474. [Google Scholar] [CrossRef] [PubMed]
- Liese, A.; Hilterhaus, L. Evaluation of immobilized enzymes for industrial applications. Chem. Soc. Rev. 2013, 42, 6236–6249. [Google Scholar] [CrossRef] [PubMed]
- Rodrigues, R.C.; Ortiz, C.; Berenguer-Murcia, A.; Torres, R.; Fernandez-Lafuente, R. Modifying enzyme activity and selectivity by immobilization. Chem. Soc. Rev. 2013, 42, 6290–6307. [Google Scholar] [CrossRef] [PubMed]
- Sheldon, R.A.; van Pelt, S. Enzyme immobilization in biocatalysis: Why, what and how. Chem. Soc. Rev. 2013, 42, 6223–6235. [Google Scholar] [CrossRef] [PubMed]
- Hickey, A.M.; Marle, L.; McCreedy, T.; Watts, P.; Greenway, G.M.; Littlechild, J.A. Immobilization of thermophilic enzymes in miniaturized flow reactors. Biochem. Soc. Trans. 2007, 35, 1621–1623. [Google Scholar] [CrossRef] [PubMed]
- Cowan, D.A.; Fernandez-Lafuente, R. Enhancing the functional properties of thermophilic enzymes by chemical modification and immobilization. Enzym. Microb. Technol. 2011, 49, 326–346. [Google Scholar] [CrossRef] [PubMed]
- Hilterhaus, L.; Minow, B.; Muller, J.; Berheide, M.; Quitmann, H.; Katzer, M.; Thum, O.; Antranikian, G.; Zeng, A.P.; Liese, A. Practical application of different enzymes immobilized on sepabeads. Bioprocess Biosyst. Eng. 2008, 31, 163–171. [Google Scholar] [CrossRef] [PubMed]
- Brady, D.; Jordaan, J. Advances in enzyme immobilization. Biotechnol. Lett. 2009, 31, 1639–1650. [Google Scholar] [CrossRef] [PubMed]
- Li, G.; Li, Q. Thermophilic esterase from the archaeon Archaeoglobus fulgidus physically immobilized on hydrophobic macroporous resin: A novel biocatalyst for polyester synthesis. Biotechnol. Bioprocess Eng. 2011, 16, 1201–1207. [Google Scholar] [CrossRef]
- Franssen, M.C.R.; Steunenberg, P.; Scott, E.L.; Zuilholf, H.; Sanders, J.P.M. Immobilised enzymes in biorenewables production. Chem. Soc. Rev. 2013, 42, 6491–6533. [Google Scholar] [CrossRef] [PubMed]
- Cerdobbel, A.; Desmet, T.; De Winter, K.; Maertens, J.; Soetaert, W. Increasing the thermostability of sucrose phosphorylase by multipoint covalent immobilization. J. Biotechnol. 2010, 150, 125–130. [Google Scholar] [CrossRef] [PubMed]
- Damnjanovic, J.J.; Zuza, M.G.; Savanovic, J.K.; Bezbradica, D.I.; Mijin, D.Z.; Boskovic-Vragolovic, N.; Knezevic-Jugovic, Z.D. Covalently immobilized lipase catalyzing high-yielding optimized geranyl butyrate synthesis in a batch and fluidized bed reactor. J. Mol. Catal. B Enzym. 2012, 75, 50–59. [Google Scholar] [CrossRef]
- Mateo, C.; Abian, O.; Fernandez-Lorente, G.; Pedroche, J.; Fernandez-Lafuente, R.; Guisan, J.M.; Tam, A.; Daminati, M. Epoxy sepabeads: A novel epoxy support for stabilization of industrial enzymes via very intense multipoint covalent attachment. Biotechnol. Prog. 2002, 18, 629–634. [Google Scholar] [CrossRef] [PubMed]
- Mateo, C.; Torres, R.; Fernandez-Lorente, G.; Ortiz, C.; Fuentes, M.; Hidalgo, A.; Lopez-Gallego, F.; Abian, O.; Palomo, J.M.; Betancor, L.; et al. Epoxy-amino groups: A new tool for improved immobilization of proteins by the epoxy method. Biomacromolecules 2003, 4, 772–777. [Google Scholar] [CrossRef] [PubMed]
- Grazu, V.; Abian, O.; Mateo, C.; Batista-Viera, F.; Fernandez-Lafuente, R.; Guisan, J.M. Novel bifunctional epoxy/thiol-reactive support to immobilize thiol containing proteins by the epoxy chemistry. Biomacromolecules 2003, 4, 1495–1501. [Google Scholar] [CrossRef] [PubMed]
- Bayraktar, H.; Serilmez, M.; Karkaş, T.; Celem, E.B.; Onal, S. Immobilization and stabilization of α-galactosidase on Sepabeads EC-EA and EC-HA. Int. J. Biol. Macromol. 2011, 49, 855–860. [Google Scholar] [CrossRef] [PubMed]
- Barbosa, O.; Torres, R.; Ortiz, C.; Berenguer-Murcia, A.; Rodrigues, R.C.; Fernandez-Lafuente, R. Heterofunctional supports in enzyme immobilization: From traditional immobilization protocols to opportunities in tuning enzyme properties. Biomacromolecules 2013, 14, 2433–2462. [Google Scholar] [CrossRef] [PubMed]
- Manoel, E.A.; Dos Santos, J.C.; Freire, D.M.; Rueda, N.; Fernandez-Lafuente, R. Immobilization of lipases on hydrophobic supports involves the open form of the enzyme. Enzym. Microb. Technol. 2015, 71, 53–57. [Google Scholar] [CrossRef] [PubMed]
- Torres, R.; Mateo, C.; Fernández-Lorente, G.; Ortiz, C.; Fuentes, M.; Palomo, J.M.; Guisan, J.M.; Fernández-Lafuente, R. A novel heterofunctional epoxy-amino sepabeads for a new enzyme immobilization protocol: Immobilization-stabilization of β-galactosidase from Aspergillus oryzae. Biotechnol. Prog. 2003, 19, 1056–1060. [Google Scholar] [CrossRef] [PubMed]
- Mateo, C.; Grazu, V.; Palomo, J.M.; Lopez-Gallego, F.; Fernandez-Lafuente, R.; Guisan, J.M. Immobilization of enzymes on heterofunctional epoxy supports. Nat. Protoc. 2007, 2, 1022–1033. [Google Scholar] [CrossRef] [PubMed]
- Mateo, C.; Grazu, V.; Guisan, J.M. Immobilization of enzymes on monofunctional and heterofunctional epoxy-activated supports. Methods Mol. Biol. 2013, 1051, 43–57. [Google Scholar] [PubMed]
- Garmroodi, M.; Mohammadi, M.; Ramazani, A.; Ashjari, M.; Mohammadi, J.; Sabour, B.; Yousefi, M. Covalent binding of hyper-activated Rhizomucor miehei lipase (RML) on hetero-functionalized siliceous supports. Int. J. Biol. Macromol. 2016, 86, 208–215. [Google Scholar] [CrossRef] [PubMed]
- Manco, G.; Giosuè, E.; D’Auria, S.; Herman, P.; Carrea, G.; Rossi, M. Cloning, overexpression, and properties of a new thermophilic and thermostable esterase with sequence similarity to hormone-sensitive lipase subfamily from the archaeon Archaeoglobus fulgidus. Arch. Biochem. Biophys. 2000, 373, 182–192. [Google Scholar] [CrossRef] [PubMed]
- Klibanov, A.M. Improving enzymes by using them in organic solvents. Nature 2001, 409, 241–246. [Google Scholar] [CrossRef] [PubMed]
- Liu, W.; Wang, F.; Tan, T.; Chen, B. Lipase-catalyzed synthesis and characterization of polymers by cyclodextrin as support architecture. Carbohydr. Polym. 2013, 92, 633–640. [Google Scholar] [CrossRef] [PubMed]
- Yang, Y.; Ge, Y.; Zhao, H.; Shi, W.; Li, Q. Lipase-catalyzed synthesis of poly(ε-caprolactone) and characterization of its solid-state properties. Biocatal. Biotransform. 2011, 29, 337–343. [Google Scholar] [CrossRef]
- Cao, H.; Han, H.; Li, G.; Yang, J.; Zhang, L.; Yang, Y.; Fang, X.; Li, Q. Biocatalytic synthesis of poly(δ-valerolactone) using a thermophilic esterase from Archaeoglobus fulgidus as catalyst. Int. J. Mol. Sci. 2012, 13, 12232–12241. [Google Scholar] [CrossRef] [PubMed]
- Ren, X.; Yu, D.; Yu, L.; Gao, G.; Han, S.; Feng, Y. A new study of cell disruption to release recombinant thermostable enzyme from Escherichia coli by thermolysis. J. Biotechnol. 2007, 129, 668–673. [Google Scholar] [CrossRef] [PubMed]
- Sample Availability: Samples of the recombinanat E. coli harboring thermophilic esterase gene AF1716 and immobilized enzyme on epoxy support Sepabeads EC-EP are available from the authors.

| Solvent | Log P | Monomer Conversion (%) | Mn (g/mol) | PDI |
|---|---|---|---|---|
| Dioxane | −1.10 | 26 | n.d. 1 | n.d. |
| Acetone | −0.23 | 29 | n.d. | n.d. |
| Tetrahydrofuran | 0.49 | 45 | 850 | n.d. |
| Chloroform | 2.00 | 60 | 970 | 1.19 |
| Toluene | 2.50 | 100 | 1160 | 1.21 |
| Cyclohexane | 3.09 | 95 | 940 | 1.22 |
| n-Hexane | 3.50 | 96 | 1110 | 1.22 |
| Solvent-free | — | 82 | 1050 | 1.23 |
© 2016 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ren, H.; Xing, Z.; Yang, J.; Jiang, W.; Zhang, G.; Tang, J.; Li, Q. Construction of an Immobilized Thermophilic Esterase on Epoxy Support for Poly(ε-caprolactone) Synthesis. Molecules 2016, 21, 796. https://doi.org/10.3390/molecules21060796
Ren H, Xing Z, Yang J, Jiang W, Zhang G, Tang J, Li Q. Construction of an Immobilized Thermophilic Esterase on Epoxy Support for Poly(ε-caprolactone) Synthesis. Molecules. 2016; 21(6):796. https://doi.org/10.3390/molecules21060796
Chicago/Turabian StyleRen, Hui, Zhen Xing, Jiebing Yang, Wei Jiang, Gang Zhang, Jun Tang, and Quanshun Li. 2016. "Construction of an Immobilized Thermophilic Esterase on Epoxy Support for Poly(ε-caprolactone) Synthesis" Molecules 21, no. 6: 796. https://doi.org/10.3390/molecules21060796
APA StyleRen, H., Xing, Z., Yang, J., Jiang, W., Zhang, G., Tang, J., & Li, Q. (2016). Construction of an Immobilized Thermophilic Esterase on Epoxy Support for Poly(ε-caprolactone) Synthesis. Molecules, 21(6), 796. https://doi.org/10.3390/molecules21060796





