Optimization of Preparation Conditions for Lysozyme Nanoliposomes Using Response Surface Methodology and Evaluation of Their Stability
Abstract
1. Introduction
2. Results and Discussion
2.1. Fitting the Model
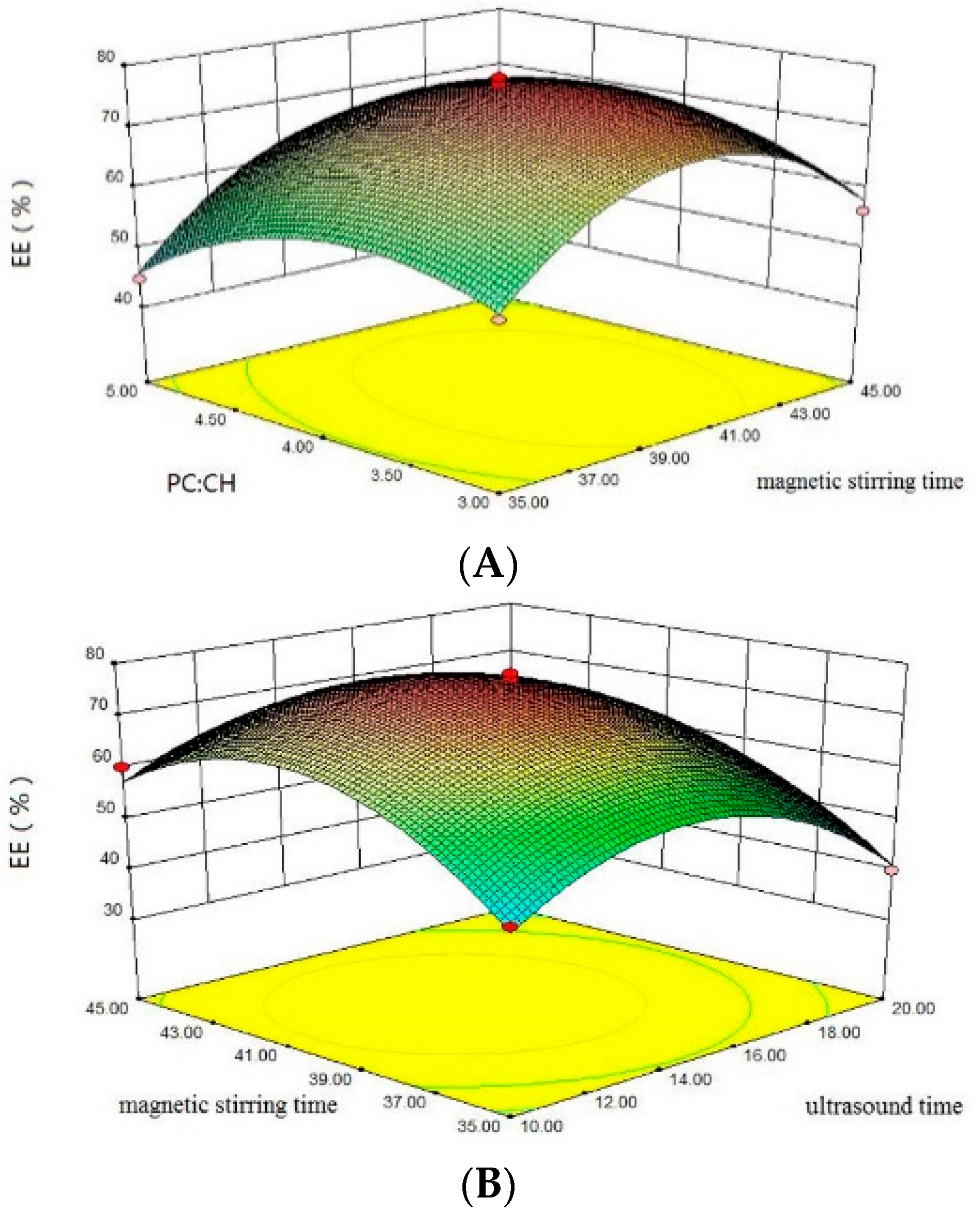
2.2. Encapsulation Efficiency
2.3. Optimization
2.4. Stability of Lysozyme Liposome
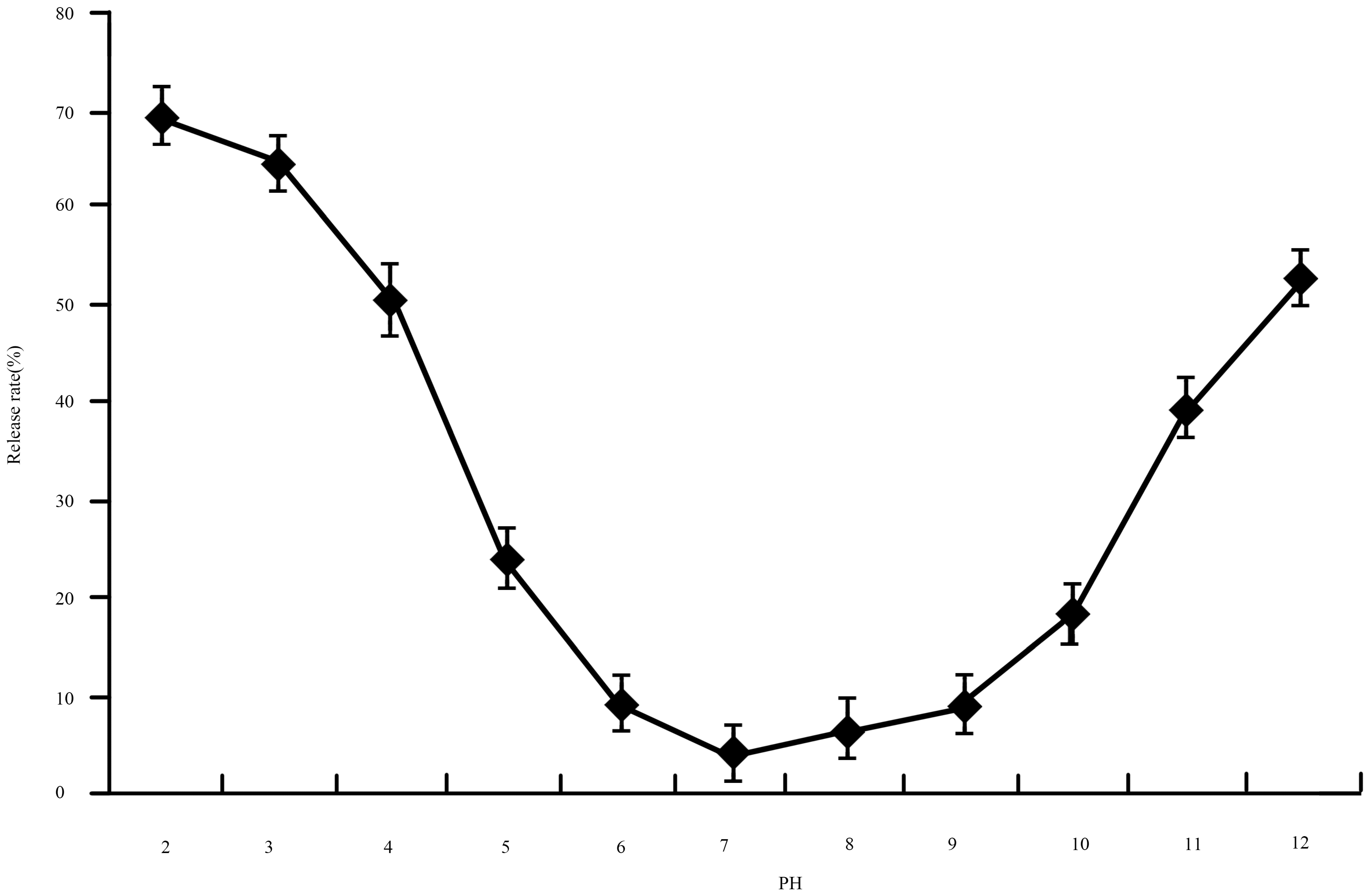
2.4.1. Stability Test to pH
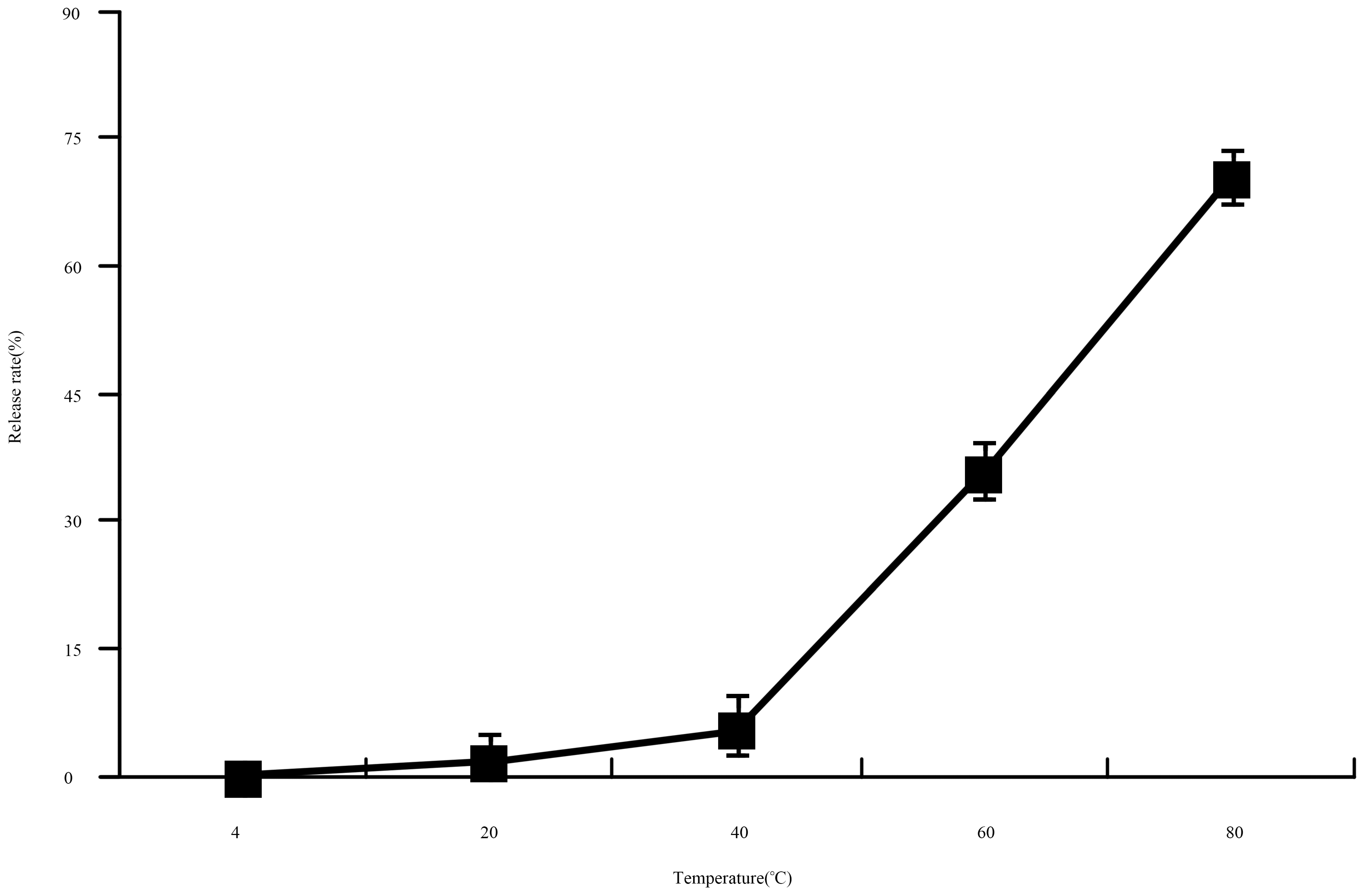
2.4.2. Thermostability Test
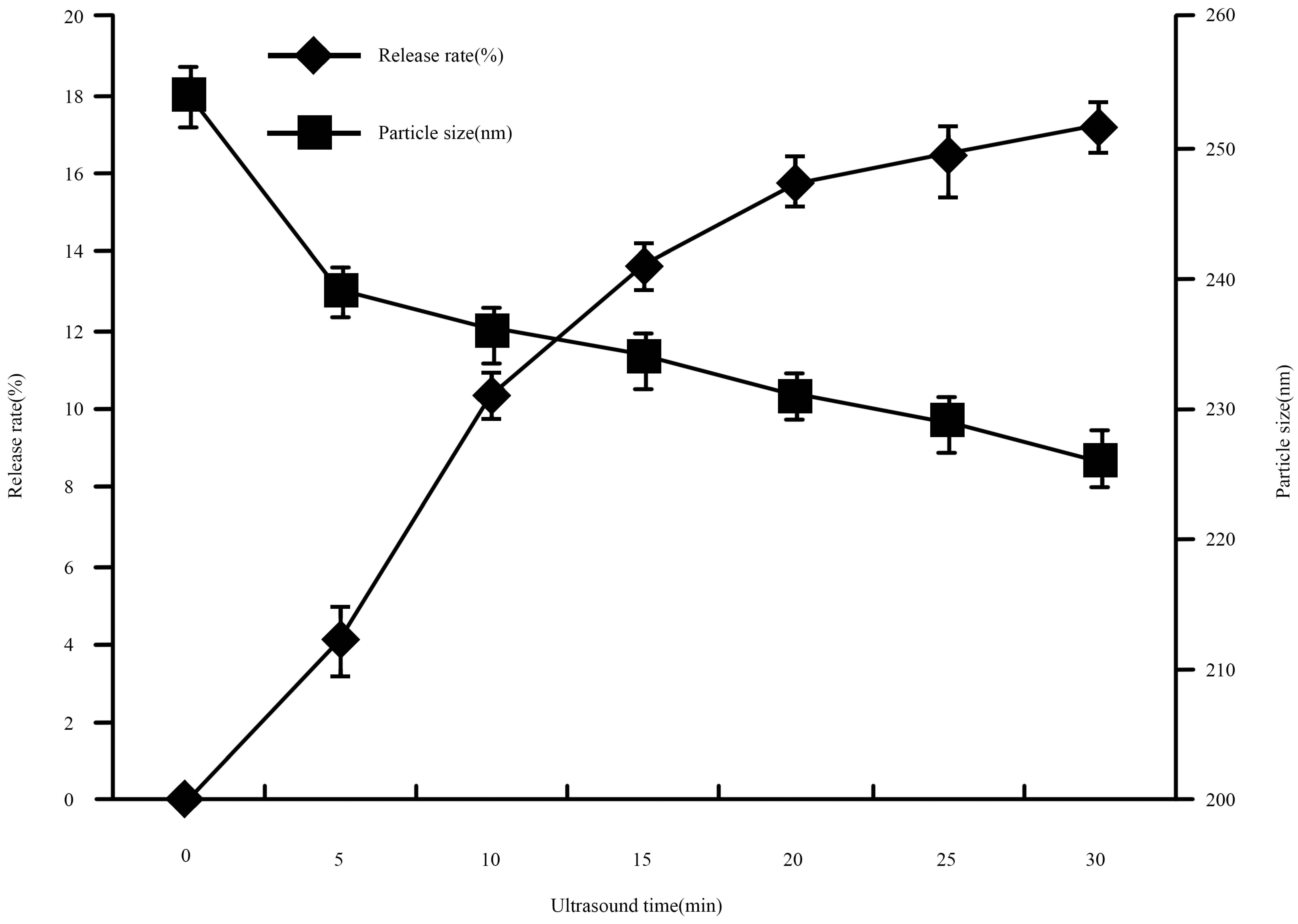
2.4.3. Effect of Sonication
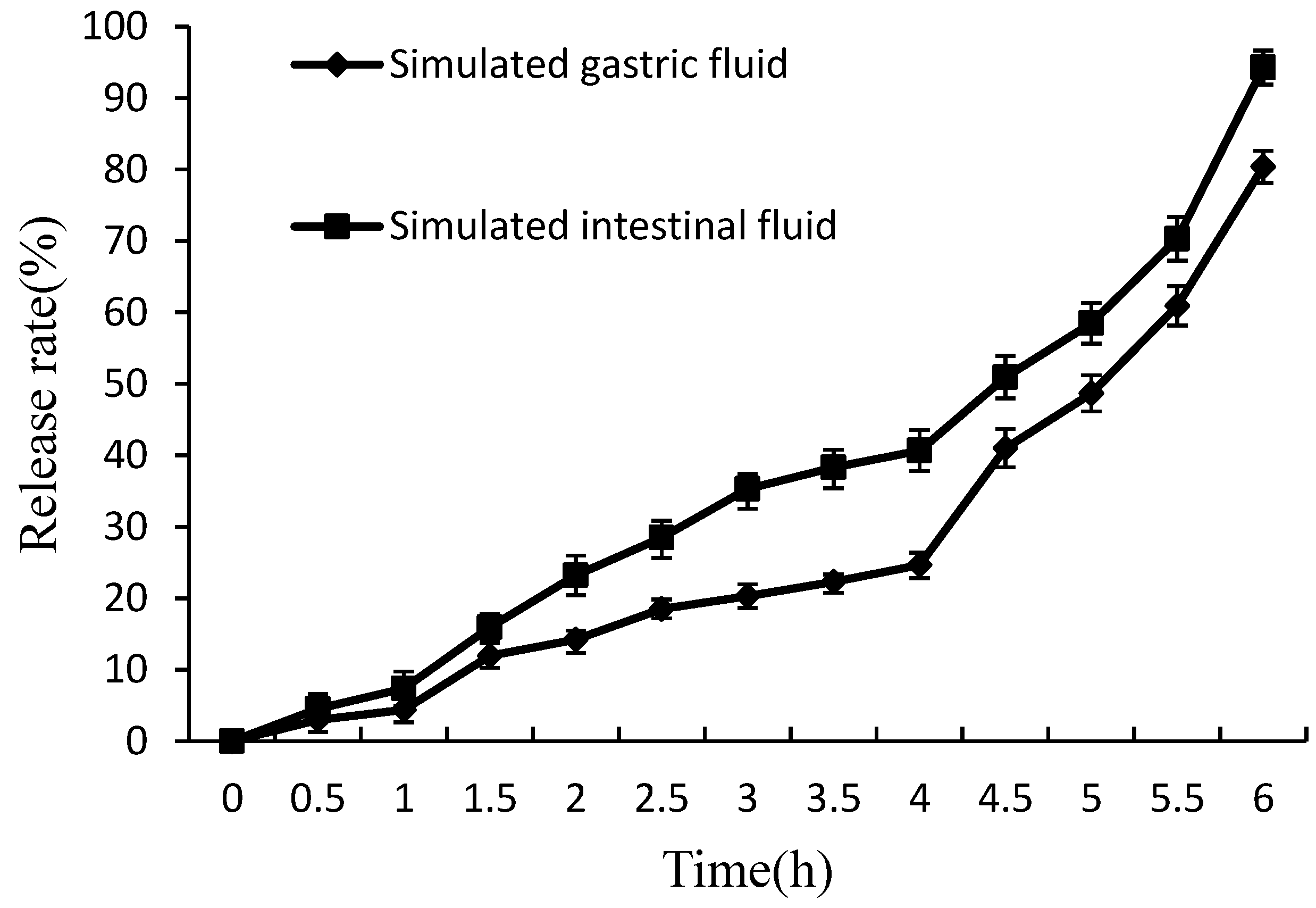
2.4.4. In Vitro Release of Lysozyme from Nanoliposomes
3. Material and Methods
3.1. Materials
3.2. Methods
3.2.1. Preparation of Lysozyme Nanoliposomes
3.2.2. Encapsulation Efficiency Determination
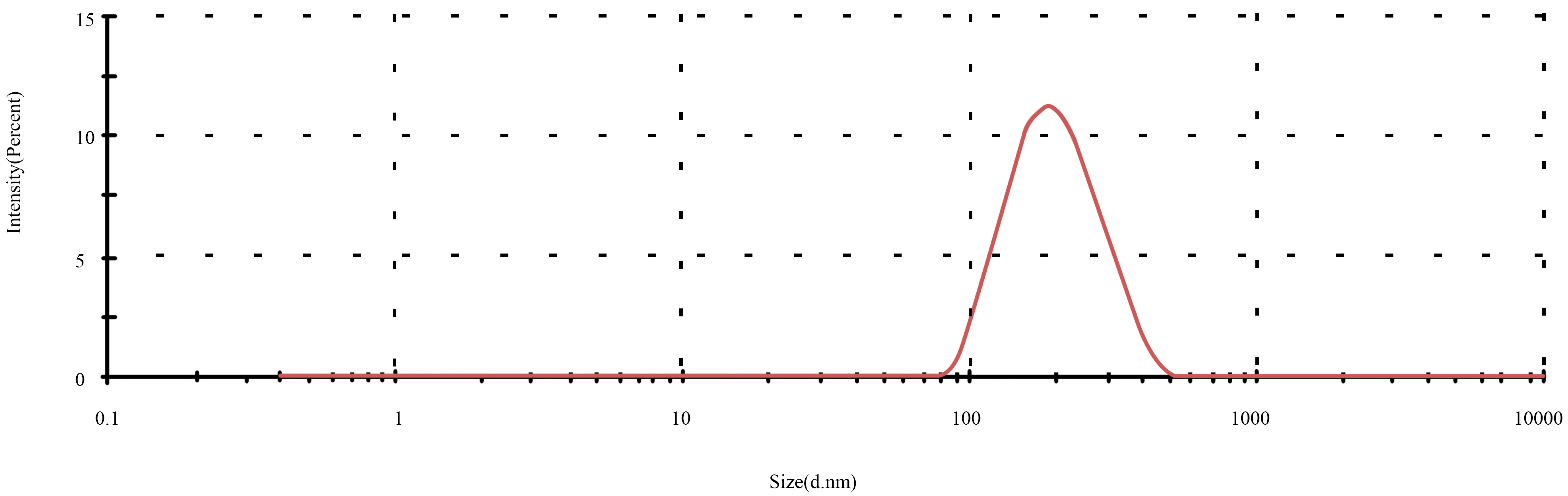
3.2.3. Particle Size
3.2.4. Experimental Design and Optimization
3.2.5. Stability of Lysozyme liposome
Stability Test to pH
Thermostability Assay
Effect of Sonication
In Vitro Release of Lysozyme from Nanoliposomes
3.2.6. Statistical Analysis
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
Abbreviations
| SGF | Simulated gastrointestinal fluid |
| SIF | Simulated intestinal fluid |
| RSM | Response surface methodology |
| PC | Phosphatidylcholine |
| CH | Cholesterol |
| EE | Encapsulation efficiency |
| ANOVA | Analysis of variance |
| SEM | Scanning electron microscope |
References
- Proctor, V.A.; Cunningham, F.; Fung, D.Y. The chemistry of lysozyme and its use as a food preservative and a pharmaceutical. Crit. Rev. Food Sci. Nutr. 1988, 26, 359–395. [Google Scholar] [CrossRef] [PubMed]
- Liburdi, K.; Benucci, I.; Esti, M. Lysozyme in Wine: An Overview of Current and Future Applications. Compr. Rev. Food Sci. F. 2014, 13, 1062–1073. [Google Scholar] [CrossRef]
- Chassy, B.M.; Giuffrida, A. Method for the lysis of Gram-positive, asporogenous bacteria with lysozyme. Appl. Environ. Microbiol. 1980, 39, 153–158. [Google Scholar] [PubMed]
- Holzapfel, W.; Geisen, R.; Schillinger, U. Biological preservation of foods with reference to protective cultures, bacteriocins and food-grade enzymes. Int. J. Food Microbiol. 1995, 24, 343–362. [Google Scholar] [CrossRef]
- Were, L.M.; Bruce, B.; Davidson, P.M.; Weiss, J. Encapsulation of nisin and lysozyme in liposomes enhances efficacy against Listeria monocytogenes. J. Food Prot. 2004, 67, 922–927. [Google Scholar] [PubMed]
- Fitzgerald, P.; Hadgraft, J.; Kreuter, J.; Wilson, C. A γ-scintigraphic evaluation of microparticulate ophthalmic delivery systems: Liposomes and nanoparticles. Int. J. Pharm. 1987, 40, 81–84. [Google Scholar] [CrossRef]
- Alexander, M.; Lopez, A.A.; Fang, Y.; Corredig, M. Incorporation of phytosterols in soy phospholipids nanoliposomes: Encapsulation efficiency and stability. LWT-Food Sci. Technol. 2012, 47, 427–436. [Google Scholar] [CrossRef]
- Charcosset, C. Preparation of emulsions and particles by membrane emulsification for the food processing industry. J. Food Eng. 2009, 92, 241–249. [Google Scholar] [CrossRef]
- Taylor, T.M.; Weiss, J.; Davidson, P.M.; Bruce, B.D. Liposomal nanocapsules in food science and agriculture. Crit. Rev. Food Sci. Nutr. 2005, 45, 587–605. [Google Scholar] [CrossRef] [PubMed]
- Mozafari, M.R.; Khosravi-Darani, K.; Borazan, G.G.; Cui, J.; Pardakhty, A.; Yurdugul, S. Encapsulation of food ingredients using nanoliposome technology. Int. J. Food Prop. 2008, 11, 833–844. [Google Scholar] [CrossRef]
- Reza Mozafari, M.; Johnson, C.; Hatziantoniou, S.; Demetzos, C. Nanoliposomes and their applications in food nanotechnology. J. Liposome Res. 2008, 18, 309–327. [Google Scholar] [CrossRef] [PubMed]
- Laridi, R.; Kheadr, E.; Benech, R.-O.; Vuillemard, J.; Lacroix, C.; Fliss, I. Liposome encapsulated nisin Z: Optimization, stability and release during milk fermentation. Int. Dairy J. 2003, 13, 325–336. [Google Scholar] [CrossRef]
- Socaciu, C.; Bojarski, P.; Aberle, L.; Diehl, H.A. Different ways to insert carotenoids into liposomes affect structure and dynamics of the bilayer differently. Biophys. Chem. 2002, 99, 1–15. [Google Scholar] [CrossRef]
- Yoo, M.; Lee, S.; Kim, S.; Shin, D. Optimizing conditions for E-and Z-ajoene formation from garlic juice using response surface methodology. Food Sci. Nutr. 2014, 2, 605–611. [Google Scholar] [CrossRef] [PubMed]
- Myer, R.; Montgomery, D.C. Response Surface Methodology: Process And Product Optimization Using Designed Experiment; John Wiley and Sons: New York, NY, USA, 2002; pp. 343–350. [Google Scholar]
- Baş, D.; Boyacı, İ.H. Modeling and optimization I: Usability of response surface methodology. J. Food Eng. 2007, 78, 836–845. [Google Scholar] [CrossRef]
- Liyana-Pathirana, C.; Shahidi, F. Optimization of extraction of phenolic compounds from wheat using response surface methodology. Food Chem. 2005, 93, 47–56. [Google Scholar] [CrossRef]
- Wang, S.; Chen, F.; Wu, J.; Wang, Z.; Liao, X.; Hu, X. Optimization of pectin extraction assisted by microwave from apple pomace using response surface methodology. J. Food Eng. 2007, 78, 693–700. [Google Scholar] [CrossRef]
- Pinho, C.; Melo, A.; Mansilha, C.; Ferreira, I.M. Optimization of conditions for anthocyanin hydrolysis from red wine using response surface methodology (RSM). J. Agric. Food Chem. 2010, 59, 50–55. [Google Scholar] [CrossRef] [PubMed]
- Luo, X.; Guan, R.; Chen, X.; Tao, M.; Ma, J.; Zhao, J. Optimization on condition of epigallocatechin-3-gallate (EGCG) nanoliposomes by response surface methodology and cellular uptake studies in Caco-2 cells. Nanoscale Res. Lett. 2014, 9, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Honary, S.; Ebrahimi, P.; Hadianamrei, R. Optimization of particle size and encapsulation efficiency of vancomycin nanoparticles by response surface methodology. Pharm. Dev. Technol. 2014, 19, 987–998. [Google Scholar] [CrossRef] [PubMed]
- Rui, N.; Zheng, S.M.Y.J.-S.; Hai-ying, Y.W. Formulation optimization of antioxidant peptide ultraflexible nano-liposome preparation. Prog. Fish. Sci. 2011, 2, 89–95. [Google Scholar]
- Ramachandra, C.; Rao, P.S. Processing of Aloe vera leaf gel: A review. Am. J. Agric. Biol. Sci. 2008, 3, 502–510. [Google Scholar] [CrossRef]
- Chen, D.; Yu, H.; Mu, H.; Wei, J.; Song, Z.; Shi, H.; Liang, R.; Sun, K.; Liu, W. Novel chitosan derivative for temperature and ultrasound dual-sensitive liposomal microbubble gel. Carbohydr. Polym. 2013, 94, 17–23. [Google Scholar] [CrossRef] [PubMed]
- Tan, C.; Xue, J.; Lou, X.; Abbas, S.; Guan, Y.; Feng, B.; Zhang, X.; Xia, S. Liposomes as delivery systems for carotenoids: Comparative studies of loading ability, storage stability and In vitro release. Food Funct. 2014, 5, 1232–1240. [Google Scholar] [CrossRef] [PubMed]
- Acosta, E. Bioavailability of nanoparticles in nutrient and nutraceutical delivery. Curr. Opin. Colloid Interface Sci. 2009, 14, 3–15. [Google Scholar] [CrossRef]
- Monteiro, N.; Martins, A.; Reis, R.L.; Neves, N.M. Liposomes in tissue engineering and regenerative medicine. J. R. Soc. Interface 2014, 11. [Google Scholar] [CrossRef]
- Lin, L.; Cui, H.; Zhou, H.; Zhang, X.; Bortolini, C.; Chen, M.; Liu, L.; Dong, M. Nanoliposomes containing Eucalyptus citriodora as antibiotic with specific antimicrobial activity. Chem. Commun. 2015, 51, 2653–2655. [Google Scholar] [CrossRef] [PubMed]
- Song, S.; Cheong, L.Z.; Falkeborg, M.; Liu, L.; Dong, M.; Jensen, H.M.; Bertelsen, K.; Thorsen, M.; Tan, T.; Xu, X. Facile synthesis of phosphatidyl saccharides for preparation of anionic nanoliposomes with enhanced stability. PLoS ONE 2013, 8, e73891. [Google Scholar] [CrossRef] [PubMed]
- Jahadi, M.; Khosravi-Darani, K.; Ehsani, M.R.; Mozafari, M.R.; Saboury, A.A.; Pourhosseini, P.S. The encapsulation of flavourzyme in nanoliposome by heating method. J. Food Sci. Technol. 2013, 52, 2063–2072. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Jiang, L.; Sui, X.; Wang, S. Optimization of the aqueous enzymatic extraction of pine kernel oil by response surface methodology. Procedia Eng. 2011, 15, 4641–4652. [Google Scholar] [CrossRef]
- Zhang, Z.-S.; Li, D.; Wang, L.-J.; Ozkan, N.; Chen, X.D.; Mao, Z.-H.; Yang, H.-Z. Optimization of ethanol-water extraction of lignans from flaxseed. Sep. Purif. Technol. 2007, 57, 17–24. [Google Scholar] [CrossRef]
- Zhao, L.; Xiong, H.; Peng, H.; Wang, Q.; Han, D.; Bai, C.; Liu, Y.; Shi, S.; Deng, B. PEG-coated lyophilized proliposomes: Preparation, characterizations and In vitro release evaluation of vitamin E. Eur. Food Res. Technol. 2011, 232, 647–654. [Google Scholar] [CrossRef]
- Sample Availability: Not Available.

| Source | DF | EE% | ||
|---|---|---|---|---|
| Coefficient | Sum of Squares | p Value | ||
| Model | 14 | 76.56 | 3098.91 | <0.0001 |
| Linear | ||||
| X1 | 1 | −2.58 | 80.08 | 0.0069 |
| X2 | 1 | −0.34 | 1.40 | 0.6823 |
| X3 | 1 | −4.73 | 268.85 | <0.0001 |
| X4 | 1 | 3.46 | 143.52 | 0.0008 |
| Quadratic | ||||
| X12 | −8.38 | 455.51 | <0.0001 | |
| X22 | −9.84 | 628.38 | <0.0001 | |
| X32 | −14.63 | 1388.35 | <0.0001 | |
| X42 | −14.47 | 1357.68 | <0.0001 | |
| Interaction | ||||
| X1X2 | −0.55 | 1.21 | 0.7035 | |
| X1X3 | −0.25 | 0.25 | 0.8624 | |
| X1X4 | 1.75 | 12.25 | 0.2367 | |
| X2X3 | 1.73 | 11.90 | 0.2432 | |
| X2X4 | −1.35 | 7.29 | 0.3565 | |
| X3X4 | −1.63 | 10.56 | 0.2703 | |
| Residual | 14 | 112.24 | ||
| Lack of fit | 10 | 92.76 | 0.2797 | |
| Pure error | 4 | 19.47 | ||
| Total | 28 | 3211.15 | ||
| R2 | 0.9650 | |||
| Adj-R2 | 0.9301 | |||
| CV | 4.97 | |||
| Independent Variables | Symbols | Code Levels | ||
|---|---|---|---|---|
| −1 | 0 | 1 | ||
| PC/CH (w/w) | X1 | 3 | 4 | 5 |
| Lysozyme concentration (w/v) | X2 | 1 | 2 | 3 |
| Ultrasound time (min) | X3 | 10 | 15 | 20 |
| Magnetic stirring time (min) | X4 | 35 | 40 | 45 |
© 2016 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wu, Z.; Guan, R.; Lyu, F.; Liu, M.; Gao, J.; Cao, G. Optimization of Preparation Conditions for Lysozyme Nanoliposomes Using Response Surface Methodology and Evaluation of Their Stability. Molecules 2016, 21, 741. https://doi.org/10.3390/molecules21060741
Wu Z, Guan R, Lyu F, Liu M, Gao J, Cao G. Optimization of Preparation Conditions for Lysozyme Nanoliposomes Using Response Surface Methodology and Evaluation of Their Stability. Molecules. 2016; 21(6):741. https://doi.org/10.3390/molecules21060741
Chicago/Turabian StyleWu, Zhipan, Rongfa Guan, Fei Lyu, Mingqi Liu, Jianguo Gao, and Guozou Cao. 2016. "Optimization of Preparation Conditions for Lysozyme Nanoliposomes Using Response Surface Methodology and Evaluation of Their Stability" Molecules 21, no. 6: 741. https://doi.org/10.3390/molecules21060741
APA StyleWu, Z., Guan, R., Lyu, F., Liu, M., Gao, J., & Cao, G. (2016). Optimization of Preparation Conditions for Lysozyme Nanoliposomes Using Response Surface Methodology and Evaluation of Their Stability. Molecules, 21(6), 741. https://doi.org/10.3390/molecules21060741





