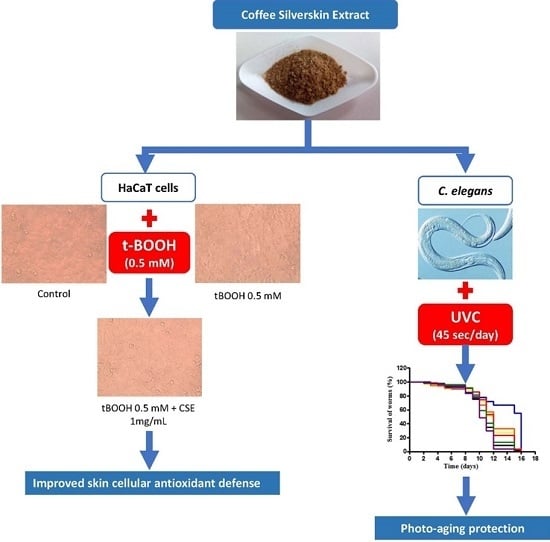
Coffee Silverskin Extract Protects against Accelerated Aging Caused by Oxidative Agents
Abstract
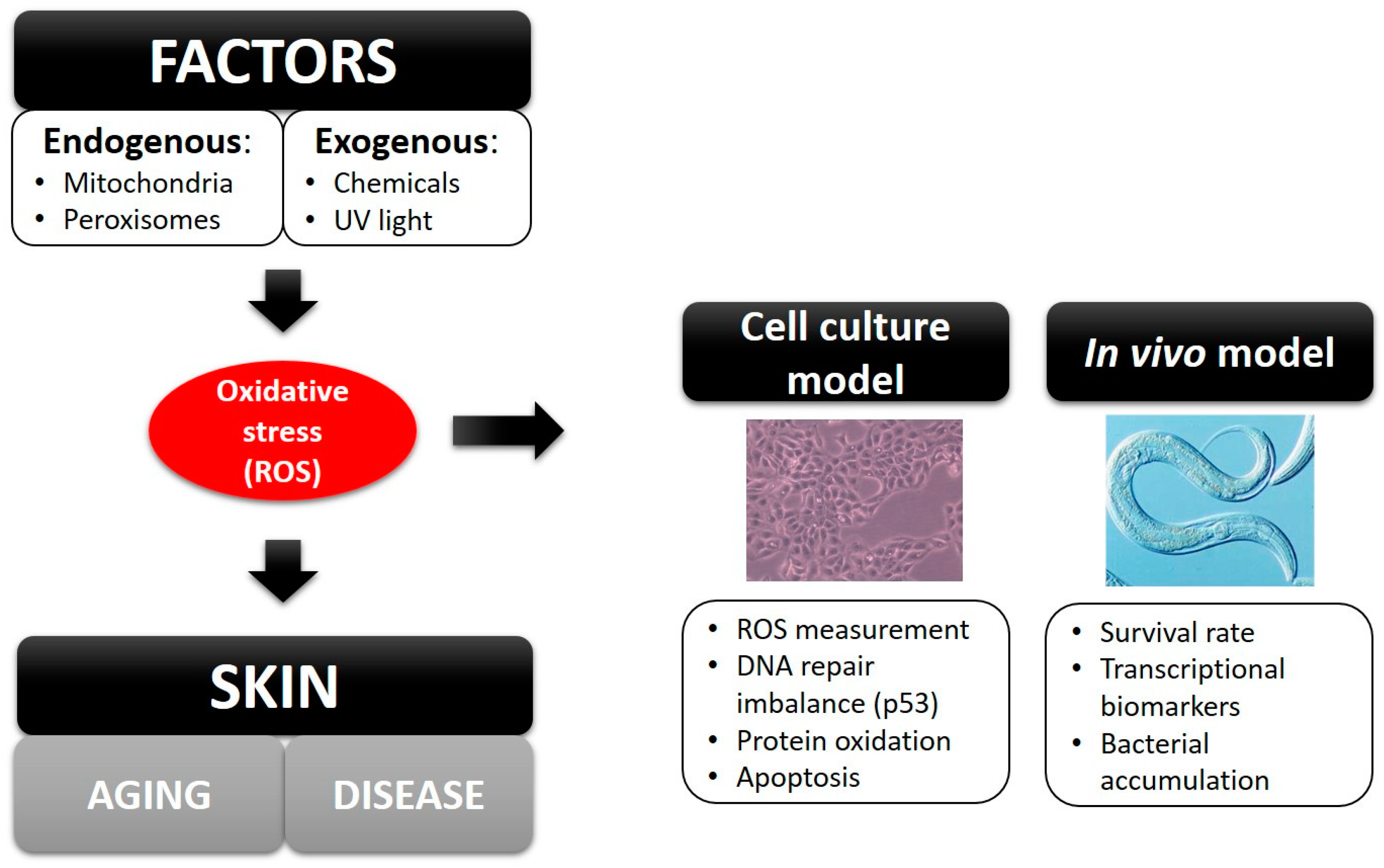
:1. Introduction
2. Results
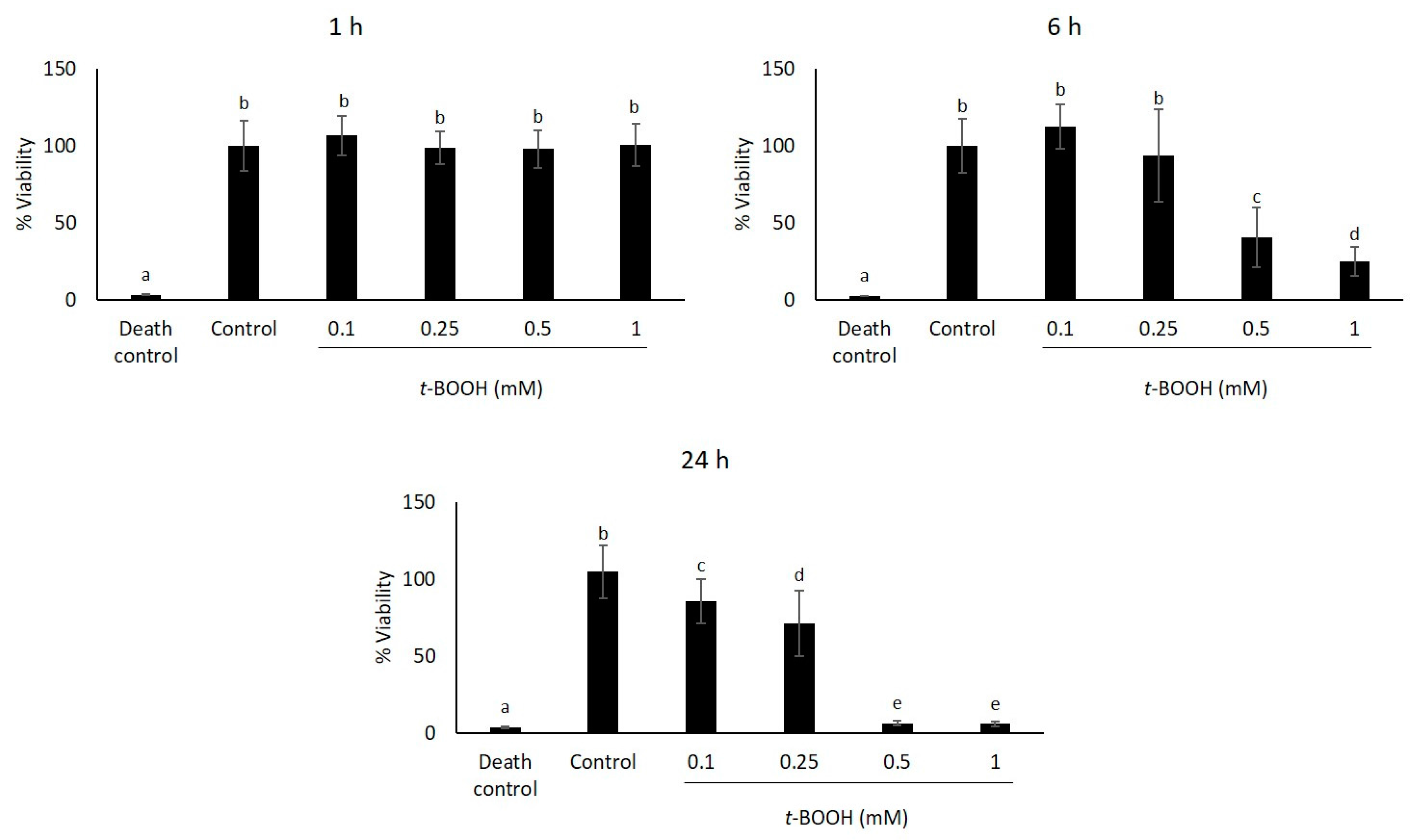
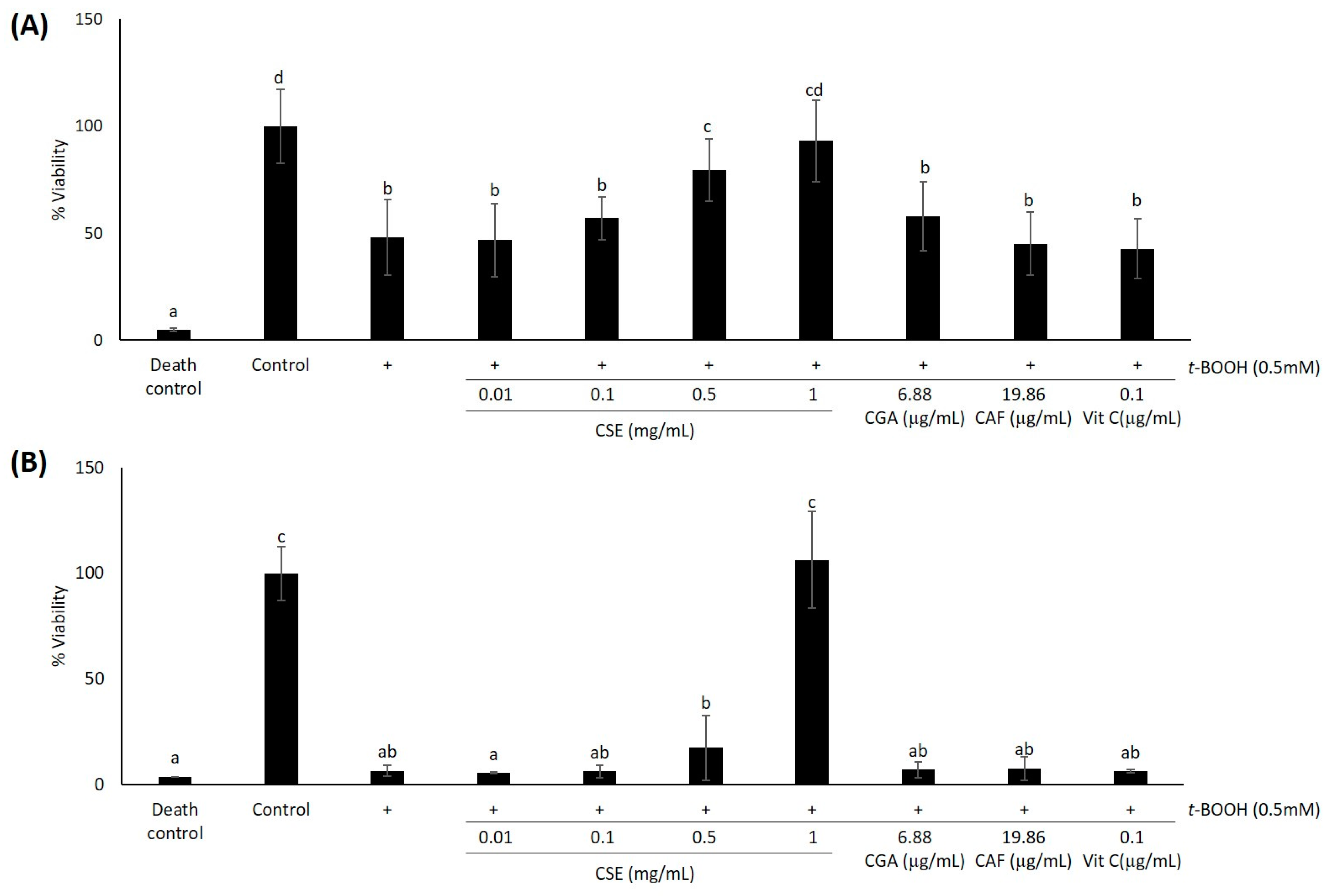
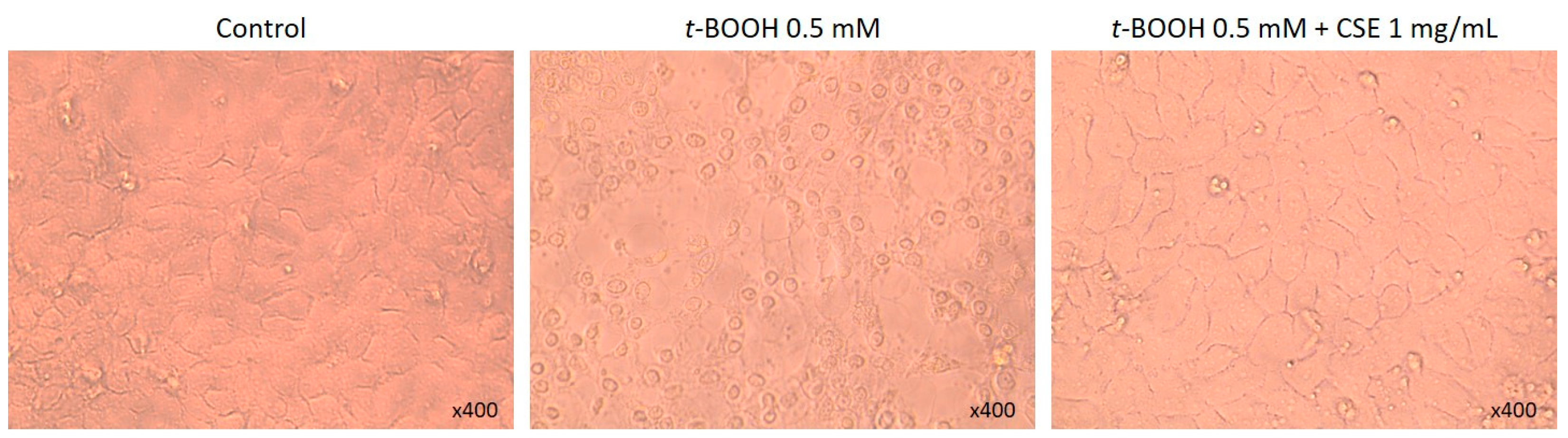
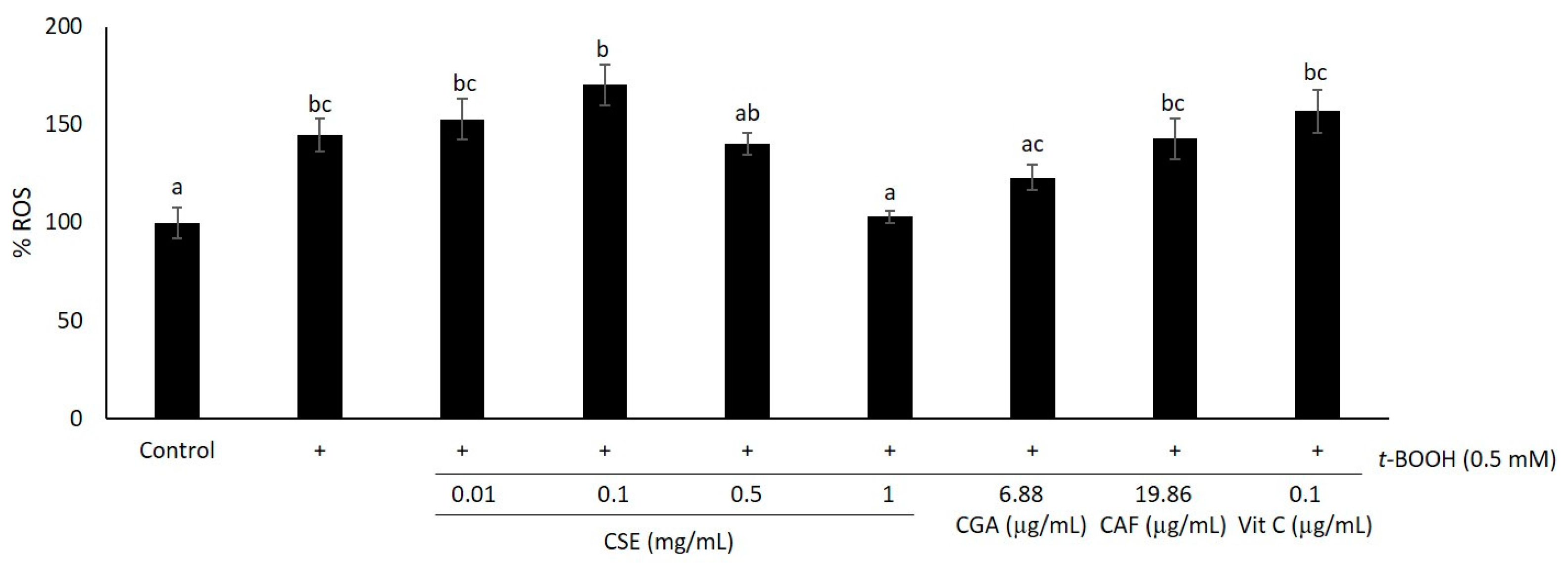
2.1. Study of Coffee Silverskin in HaCaT Cells
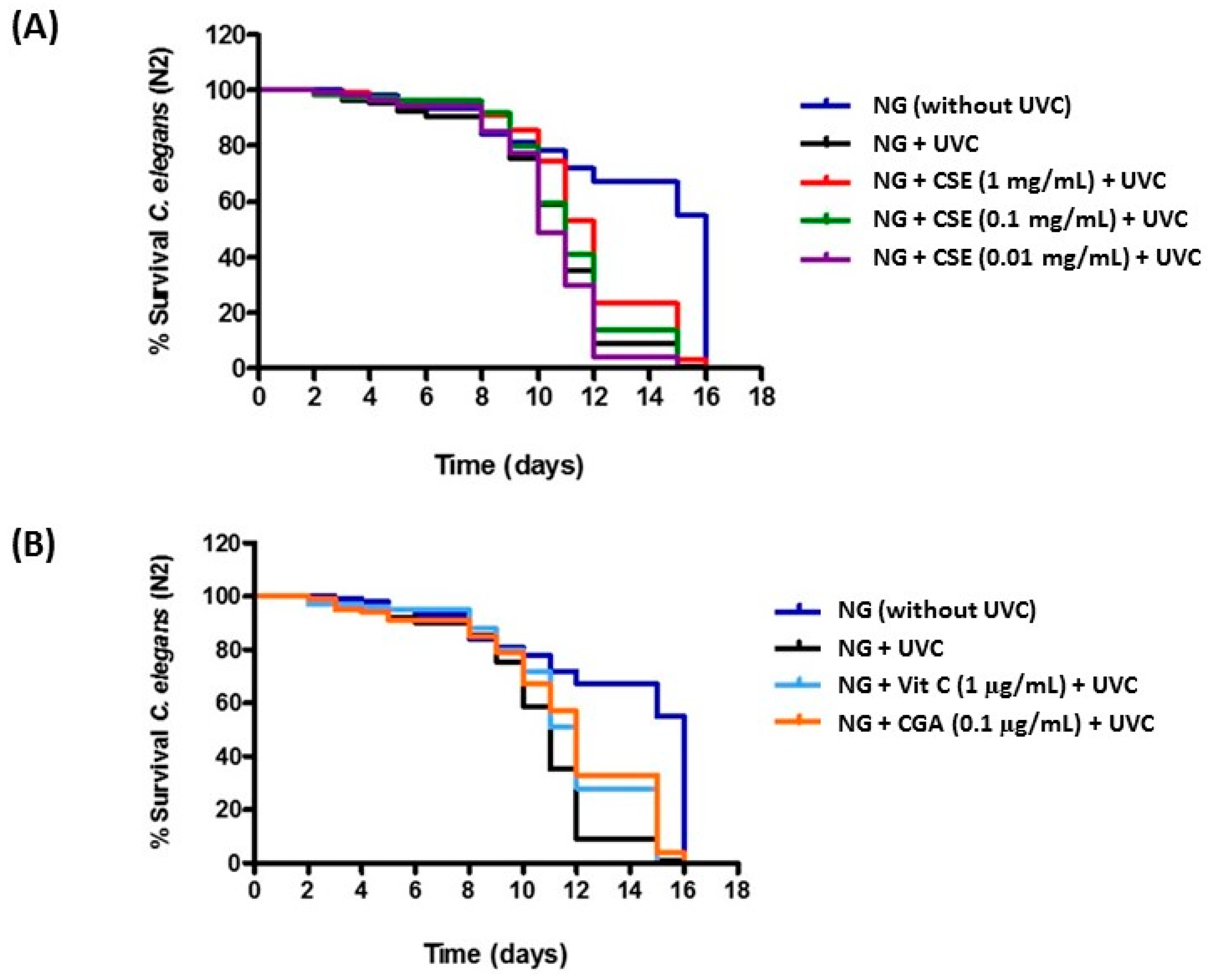
2.2. Study of Coffee Silverskin in C. elegans
3. Discussion
4. Materials and Methods
4.1. Materials
4.2. Preparation of Soluble Extracts from Coffee Silverskin
4.3. CSE Overall Antioxidant Capacity Assay
4.4. Cell Culture and Treatments
4.5. Cell Viability Assays
4.6. ROS Scavenging Assay
4.7. C. elegans Lifespan Assays
4.8. Statistical Analyses
5. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Hancock, J.T.; Desikan, R.; Neill, S.J. Role of reactive oxygen species in cell signalling pathways. Biochem. Soc. Trans. 2001, 29, 345–350. [Google Scholar] [CrossRef] [PubMed]
- Klaunig, J.E.; Kamendulis, L.M.; Hocevar, B. A Oxidative stress and oxidative damage in carcinogenesis. Toxicol. Pathol. 2010, 38, 96–109. [Google Scholar] [CrossRef] [PubMed]
- Kruk, J.; Duchnik, E. Oxidative Stress and Skin Diseases: Possible Role of Physical Activity. Asian Pac. J. Cancer Prev. 2014, 15, 561–568. [Google Scholar] [CrossRef] [PubMed]
- Dalle-Donne, I.; Rossi, R.; Colombo, R.; Giustarini, D.; Milzani, A. Biomarkers of oxidative damage in human disease. Clin. Chem. 2006, 52, 601–623. [Google Scholar] [CrossRef] [PubMed]
- Garigan, D.; Hsu, A.L.; Fraser, A.G.; Kamath, R.S.; Abringet, J.; Kenyon, C. Genetic analysis of tissue aging in Caenorhabditis elegans: A role for heat-shock factor and bacterial proliferation. Genetics 2002, 161, 1101–1112. [Google Scholar] [PubMed]
- Bisht, S.; Sisodia, S. Coffea arabica: A wonder gift to medical science. J. Nat. Pharm. 2010, 1, 58–65. [Google Scholar] [CrossRef]
- Fava, F.; Totaro, G.; Diels, L.; Reis, M.; Duarte, J.; Carioca, O.B.; Poggi-Varaldo, H.M.; Ferreira, B.S. Biowaste biorefinery in Europe: Opportunities and research & development needs. New Biotechnol. 2015, 32, 100–108. [Google Scholar]
- Park, K.; Lee, J.H. Protective effects of resveratrol on UVB-irradiated HaCaT cells through attenuation of the caspase pathway. Oncol. Rep. 2008, 19, 413–417. [Google Scholar] [CrossRef] [PubMed]
- Vitale, N.; Kisslinger, A.; Paladino, S.; Procaccini, C.; Matarese, G.; Pierantoni, G.M.; Mancini, F.P.; Tramontano, D. Resveratrol couples apoptosis with autophagy in UVB-irradiated HaCaT cells. PLoS ONE 2013, 8, e80728. [Google Scholar] [CrossRef] [PubMed]
- Pérez-Sánchez, A.; Barrajón-Catalán, E.; Caturla, N.; Castillo, J.; Benavente-García, O.; Alcaraz, M.; Micol, V. Protective effects of citrus and rosemary extracts on UV-induced damage in skin cell model and human volunteers. J. Photochem. Photobiol. B Biol. 2014, 136, 12–18. [Google Scholar] [CrossRef] [PubMed]
- Almeida, I.F.; Pinto, A.S.; Monteiro, C.; Monteiro, H.; Belo, L.; Fernandes, J.; Bento, A.R.; Duarte, T.L.; Garrido, J.; Bahia, M.F.; et al. Protective effect of C. sativa leaf extract against UV mediated-DNA damage in a human keratinocyte cell line. J. Photochem. Photobiol. B Biol. 2015, 144, 28–34. [Google Scholar] [CrossRef] [PubMed]
- Chiang, H.M.; Lin, T.J.; Chiu, C.Y.; Chang, C.W.; Hsu, K.C.; Fan, P.C.; Wen, K.C. Coffea arabica extract and its constituents prevent photoaging by suppressing MMPs expression and MAP kinase pathway. Food Chem. Toxicol. 2011, 49, 309–318. [Google Scholar] [CrossRef] [PubMed]
- Herman, A.; Herman, A.P. Caffeine’s mechanisms of action and its cosmetic use. Skin Pharmacol. Physiol. 2012, 26, 8–14. [Google Scholar] [CrossRef] [PubMed]
- Fukushima, Y.; Takahashi, Y.; Hori, Y.; Kishimoto, Y.; Shiga, K.; Tanaka, Y.; Masunaga, E.; Tani, M.; Yokoyama, M.; Kondo, K. Skin photoprotection and consumption of coffee and polyphenols in healthy middle-aged Japanese females. Int. J. Dermatol. 2015, 54, 410–418. [Google Scholar] [CrossRef] [PubMed]
- Del Castillo, B.M.D.; Ibañez, E.M.E.; Amigo, B.M.; Herrero, C.M.; PLaza, del M.M.; Ullate, A.M. Application of Products of Coffee Silverskin in Anti-agenic Cosmetic and Functional Food. Patent EP2730171 A1, 14 May 2014. [Google Scholar]
- Rodrigues, F.; Palmeira-de-Oliveira, A.; das Neves, J.; Sarmento, B.; Amaral, M.H.; Oliveira, M.B.P.P. Coffee silverskin: A possible valuable cosmetic ingredient. Pharm. Biol. 2015, 53, 386–394. [Google Scholar] [CrossRef] [PubMed]
- Del Castillo, M.D.; Fernandez-Gomez, B.; Martinez-Saez, N.; Iriondo-DeHond, A.; Mesa, M.D. Coffee by-products. In Coffee: Chemistry, Quality and Health Implications; Farah, A., Ed.; RSC Publishing Inc: Cambridge, UK, In press.
- Brohem, C.A.; Da Silva Cardeal, L.B.; Tiago, M.; Soengas, M.S.; De Moraes Barros, S.B.; Maria-Engler, S.S. Artificial skin in perspective: Concepts and applications. Pigment Cell Melanoma Res. 2011, 24, 35–50. [Google Scholar] [CrossRef] [PubMed]
- Mazzoleni, G.; di Lorenzo, D.; Steimberg, N. Modelling tissues in 3D: The next future of pharmaco-toxicology and food research? Genes Nutr. 2009, 4, 13–22. [Google Scholar] [CrossRef] [PubMed]
- Kenyon, C. The plasticity of aging: Insights from long-lived mutants. Cell 2005, 120, 449–460. [Google Scholar] [CrossRef] [PubMed]
- Baumann, L. Skin ageing and its treatment. J. Pathol. 2007, 211, 241–251. [Google Scholar] [CrossRef] [PubMed]
- Fernandez-Gomez, B.; Nitride, C.; Ullate, M.; Mamone, G.; Ferranti, P.; del Castillo, M.D. Use of phytochemomics for validating the potential of coffee silverskin extract as natural source of inhibitors of the glycoxidation reaction. Food Funct. 2016. underprocessing. [Google Scholar]
- Kucera, O.; Endlicher, R.; Rousar, T.; Lotková, H.; Garnol, T.; Drahota, Z.; Cervinková, Z. The effect of tert -butyl hydroperoxide-induced oxidative stress on lean and steatotic rat hepatocytes in vitro. Oxid. Med. Cell. Longev. 2014, 2014, 752506. [Google Scholar] [CrossRef] [PubMed]
- Boyd, W.A.; Crocker, T.L.; Rodriguez, A.M.; Leung, M.C.K.; Wade, D.; Freedman, J.H.; Van Houten, B.; Meyer, J.N. Nucleotide excision repair genes are expressed at low levels and are not detectably inducible in Caenorhabditis elegans somatic tissues, but their function is required for normal adult life after UVC exposure. Mutat. Res. 2010, 683, 57–67. [Google Scholar] [CrossRef] [PubMed]
- Mesías, M.; Navarro, M.; Martínez-Saez, N.; Ullate, M.; del Castillo, M.D.; Morales, F.J. Antiglycative and carbonyl trapping properties of the water soluble fraction of coffee silverskin. Food Res. Int. 2014, 62, 1120–1126. [Google Scholar] [CrossRef]
- Fernandez-Gomez, B.; Ramos, S.; Goya, L.; Mesa, M.D.; del Castillo, M.D.; Martín, M.Á. Coffee silverskin extract improves glucose-stimulated insulin secretion and protects against streptozotocin-induced damage in pancreatic INS-1E β cells. Food Res. Int. 2016, in press. [Google Scholar] [CrossRef]
- Narita, Y.; Inouye, K. High antioxidant activity of coffee silverskin extracts obtained by the treatment of coffee silverskin with subcritical water. Food Chem. 2012, 135, 943–949. [Google Scholar] [CrossRef] [PubMed]
- Borrelli, R.C.; Esposito, F.; Napolitano, A.; Ritieni, A.; Fogliano, V. Characterization of a new potential functional ingredient: Coffee silverskin. J. Agric. Food Chem. 2004, 52, 1338–1343. [Google Scholar] [CrossRef] [PubMed]
- Rodrigues, F.; Gaspar, C.; Palmeira-de-Oliveira, A.; Sarmento, B.; Helena Amaral, M.; P P Oliveira, M.B. Application of Coffee Silverskin in cosmetic formulations: Physical/antioxidant stability studies and cytotoxicity effects. Drug Dev. Ind. Pharm. 2015, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Bae, S.; Lee, E.-J.; Lee, J.H.; Park, I.-C.; Lee, S.-J.; Hahn, H.J.; Ahn, H.J.; An, S.; An, I.-S.; Cha, H.J. Oridonin protects HaCaT keratinocytes against hydrogen peroxide-induced oxidative stress by altering microRNA expression. Int. J. Mol. Med. 2013, 185–193. [Google Scholar] [CrossRef]
- Birch-Machin, M.A.; Swalwell, H. How mitochondria record the effects of UV exposure and oxidative stress using human skin as a model tissue. Mutagenesis 2010, 25, 101–107. [Google Scholar] [CrossRef] [PubMed]
- Garcia-Cohen, E.C.; Marin, J.; Diez-Picazo, L.D.; Baena, A.B.; Salaices, M.; Rodriguez-Martinez, M.A. Oxidative stress induced by tert-butyl hydroperoxide causes vasoconstriction in the aorta from hypertensive and aged rats: Role of cyclooxygenase-2 isoform. J. Pharmacol. Exp. Ther. 2000, 293, 75–81. [Google Scholar] [PubMed]
- Alía, M.; Ramos, S.; Mateos, R.; Bravo, L.; Goya, L. Response of the antioxidant defense system to tert-butyl hydroperoxide and hydrogen peroxide in a human hepatoma cell line (HepG2). J. Biochem. Mol. Toxicol. 2005, 19, 119–128. [Google Scholar] [CrossRef] [PubMed]
- Koo, S.W.; Hirakawa, S.; Fujii, S.; Kawasumi, M.; Nghiem, P. Protection from photodamage by topical application of caffeine after ultraviolet irradiation. Br. J. Dermatol. 2007, 156, 957–964. [Google Scholar] [CrossRef] [PubMed]
- Fernández-García, E. Skin protection against UV light by dietary antioxidants. Food Funct. 2014, 5, 1994–2003. [Google Scholar] [CrossRef] [PubMed]
- Pandel, R.; Poljšak, B.; Godic, A.; Dahmane, R. Skin Photoaging and the Role of Antioxidants in Its Prevention. ISRN Dermatol. 2013, 2013. [Google Scholar] [CrossRef] [PubMed]
- Aan, G.J.; Zainudin, M.S.A.; Karim, N.A.; Ngah, W.Z.W. Effect of the tocotrienol-rich fraction on the lifespan and oxidative biomarkers in Caenorhabditis elegans under oxidative stress. Clinics (Sao Paulo). 2013, 68, 599–604. [Google Scholar] [CrossRef]
- Dostal, V.; Roberts, C.M.; Link, C.D. Genetic mechanisms of coffee extract protection in a Caenorhabditis elegans model of β-amyloid peptide toxicity. Genetics 2010, 186, 857–866. [Google Scholar] [CrossRef] [PubMed]
- Fitzenberger, E.; Deusing, D.J.; Wittkop, A.; Kler, A.; Kriesl, E.; Bonnländer, B.; Wenzel, U. Effects of Plant Extracts on the Reversal of Glucose-Induced Impairment of Stress-Resistance in Caenorhabditis elegans. Plant Foods Hum. Nutr. 2014, 69, 78–84. [Google Scholar] [CrossRef] [PubMed]
- Wilson, M.A.; Shukitt-Hale, B.; Kalt, W.; Ingram, D.K.; Joseph, J.A.; Wolkow, C.A. Blueberry polyphenols increase lifespan and thermotolerance in Caenorhabditis elegans. Aging Cell 2006, 5, 59–68. [Google Scholar] [CrossRef] [PubMed]
- Vayndorf, E.M.; Lee, S.S.; Liu, R.H. Whole apple extracts increase lifespan, healthspan and resistance to stress in Caenorhabditis elegans. J. Funct. Foods 2013, 5, 1235–1243. [Google Scholar] [CrossRef] [PubMed]
- Wen, H.; Gao, X.; Qin, J. Probing the anti-aging role of polydatin in Caenorhabditis elegans on a chip. Integr. Biol. (Camb.) 2013, 6, 35–43. [Google Scholar] [CrossRef] [PubMed]
- Jadoon, S.; Karim, S.; Asad, M.H.H.B.; Akram, M.R.; Kalsoom Khan, A.; Malik, A.; Chen, C.; Murtaza, G. Anti-aging potential of phytoextract loaded-pharmaceutical creams for human skin cell longetivity. Oxid. Med. Cell. Longev. 2015, 2015. [Google Scholar] [CrossRef] [PubMed]
- Choplet, M.; Annevelink, B. Biorefinery Euroview. Current Situation and Potential of the Biorefinery concept in the EU: Strategic Framework and Guidelines for Its Development. Avaliable online: http://www.biorefinery.nl/fileadmin/biopol/user/documents/PublicDeliverables/BIOPOL_D_4_2_PUBLIC_with_EUROVIEW_-_Final_100709_opt.pdf (accessed on 15 December 2015).
- Fukushima, Y.; Ohie, T.; Yonekawa, Y.; Yonemoto, K.; Aizawa, H.; Mori, Y.; Watanabe, M.; Takeuchi, M.; Hasegawa, M.; Taguchi, C.; et al. Coffee and green tea as a large source of antioxidant polyphenols in the Japanese population. J. Agric. Food Chem. 2009, 57, 1253–1259. [Google Scholar] [CrossRef] [PubMed]
- Onakpoya, I.; Terry, R.; Ernst, E. The use of green coffee extract as a weight loss supplement: A systematic review and meta-analysis of randomised clinical trials. Gastroenterol. Res. Pract. 2011, 2011. [Google Scholar] [CrossRef] [PubMed]
- Wagemaker, T.A.L.; Carvalho, C.R.L.; Maia, N.B.; Baggio, S.R.; Guerreiro Filho, O. Sun protection factor, content and composition of lipid fraction of green coffee beans. Ind. Crops Prod. 2011, 33, 469–473. [Google Scholar] [CrossRef]
- Rodrigues, F.; Pereira, C.; Pimentel, F.B.; Alves, R.C.; Ferreira, M.; Sarmento, B.; Amaral, M.H.; Oliveira, M.B.P.P. Are coffee silverskin extracts safe for topical use? An in vitro and in vivo approach. Ind. Crops Prod. 2015, 63, 167–174. [Google Scholar] [CrossRef]
- Re, R.; Pellegrini, N.; Proteggente, A.; Pannala, A.; Yang, M.; Rice-Evans, C. Antioxidant activity applying an improved ABTS radical cation decolorization assay. Free Radic. Biol. Med. 1999, 26, 1231–1237. [Google Scholar] [CrossRef]
- Oki, T.; Nagai, S.; Yoshinaga, M.; Nishiba, Y.; Suda, I. Contribution of β-Carotene to Radical Scavenging Capacity Varies among Orange-fleshed Sweet Potato Cultivars. Food Sci. Technol. Res 2006, 12, 156–160. [Google Scholar] [CrossRef]
- Bakondi, E.; Gönczi, M.; Szabó, É.; Bai, P.; Pacher, P.; Gergely, P.; Kovács, L.; Hunyadi, J.; Szabó, C.; Csernoch, L.; et al. Role of intracellular calcium mobilization and cell-density-dependent signaling in oxidative-stress-induced cytotoxicity in HaCaT keratinocytes. J. Investig. Dermatol. 2003, 121, 88–95. [Google Scholar] [CrossRef] [PubMed]
- Gomes, A.; Fernandes, E.; Lima, J.L.F.C. Fluorescence probes used for detection of reactive oxygen species. J. Biochem. Biophys. Methods 2005, 65, 45–80. [Google Scholar] [CrossRef] [PubMed]
- Sample Availability: Samples of CSE are available from the authors for official collaboration.
| Strain | Treatment | Life Expectancy (50%) (Days) | p-Value |
|---|---|---|---|
| N2 | NG | 11 | |
| NG + Vit C (0.1 µg/mL) | 12 | 0.0021 ** | |
| NG + CGA (0.1 µg/mL) | 12 | 0.0003 *** | |
| NG + CSE (1 mg/mL) | 12 | 0.0013 ** | |
| NG + CSE (0.1 mg/mL) | 11 | 0.3364 (NS) | |
| NG + CSE (0.01 mg/mL) | 10 | 0.2133 (NS) |
© 2016 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Iriondo-DeHond, A.; Martorell, P.; Genovés, S.; Ramón, D.; Stamatakis, K.; Fresno, M.; Molina, A.; Del Castillo, M.D. Coffee Silverskin Extract Protects against Accelerated Aging Caused by Oxidative Agents. Molecules 2016, 21, 721. https://doi.org/10.3390/molecules21060721
Iriondo-DeHond A, Martorell P, Genovés S, Ramón D, Stamatakis K, Fresno M, Molina A, Del Castillo MD. Coffee Silverskin Extract Protects against Accelerated Aging Caused by Oxidative Agents. Molecules. 2016; 21(6):721. https://doi.org/10.3390/molecules21060721
Chicago/Turabian StyleIriondo-DeHond, Amaia, Patricia Martorell, Salvador Genovés, Daniel Ramón, Konstantinos Stamatakis, Manuel Fresno, Antonio Molina, and Maria Dolores Del Castillo. 2016. "Coffee Silverskin Extract Protects against Accelerated Aging Caused by Oxidative Agents" Molecules 21, no. 6: 721. https://doi.org/10.3390/molecules21060721
APA StyleIriondo-DeHond, A., Martorell, P., Genovés, S., Ramón, D., Stamatakis, K., Fresno, M., Molina, A., & Del Castillo, M. D. (2016). Coffee Silverskin Extract Protects against Accelerated Aging Caused by Oxidative Agents. Molecules, 21(6), 721. https://doi.org/10.3390/molecules21060721