Liquid Chromatography with Tandem Mass Spectrometry: A Sensitive Method for the Determination of Dehydrodiisoeugenol in Rat Cerebral Nuclei
Abstract
:1. Introduction
2. Results and Discussion
2.1. Chromatographic Conditions
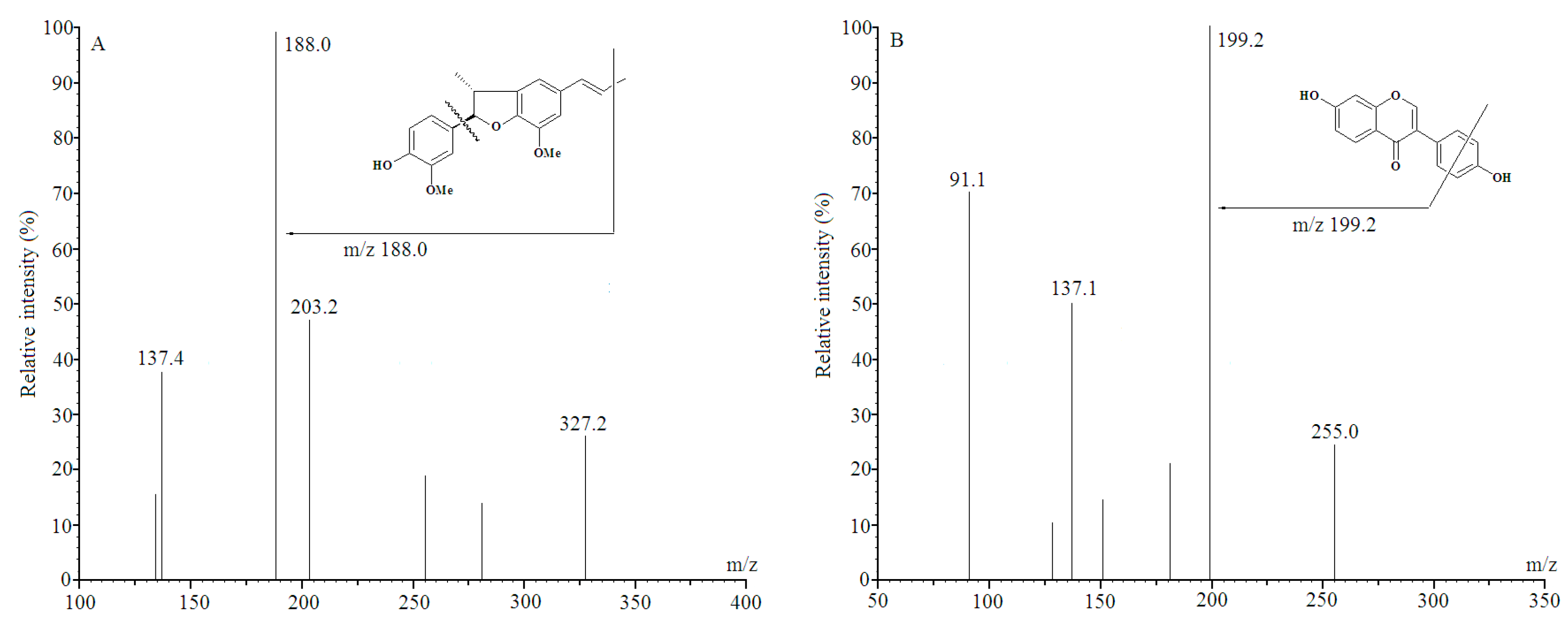
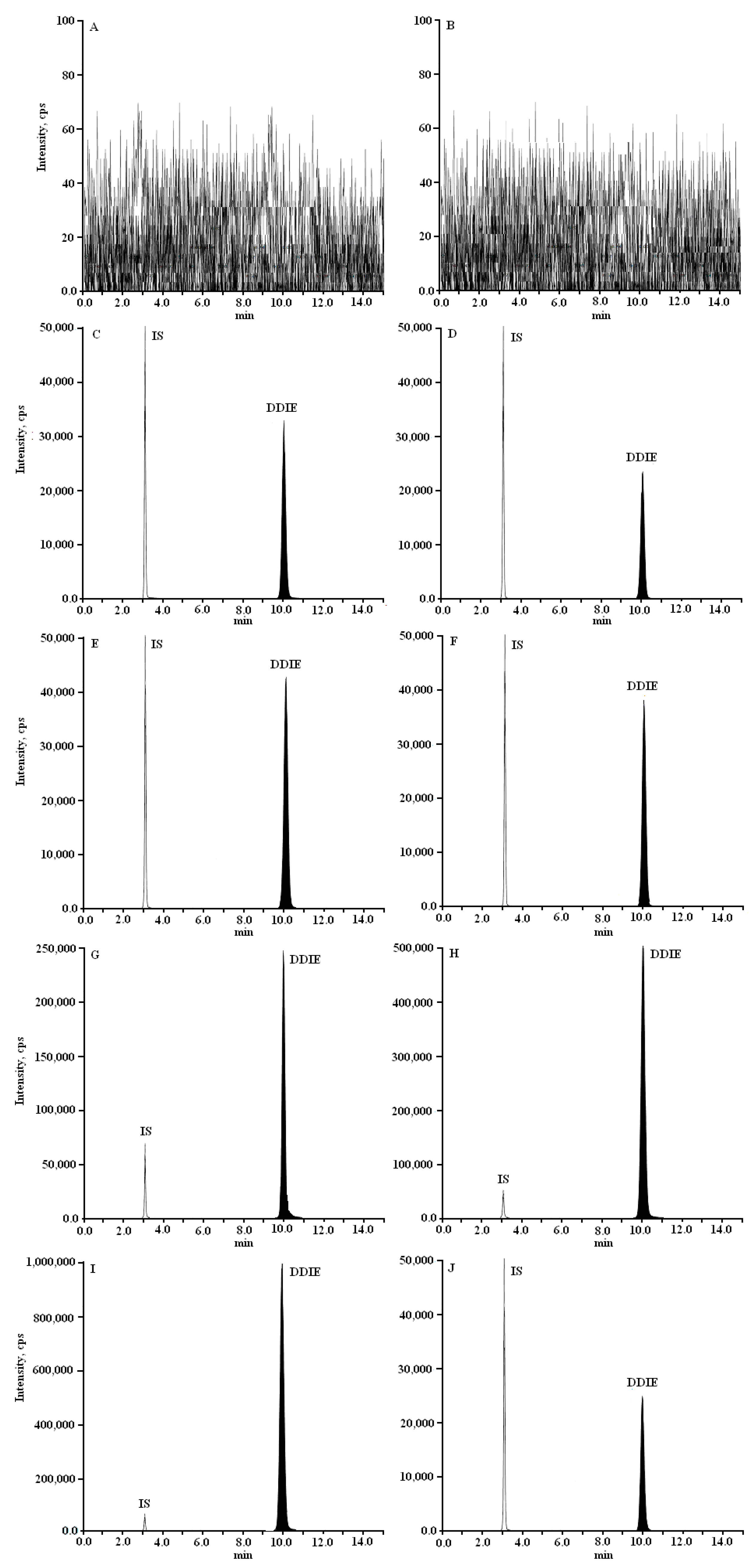
2.2. Specificity
2.3. Linearity of Calibration Curves and Lower Limits of Detection and Quantification
2.4. Precision and Accuracy
2.5. Absolute Recovery and Stability
2.6. Matrix Effects
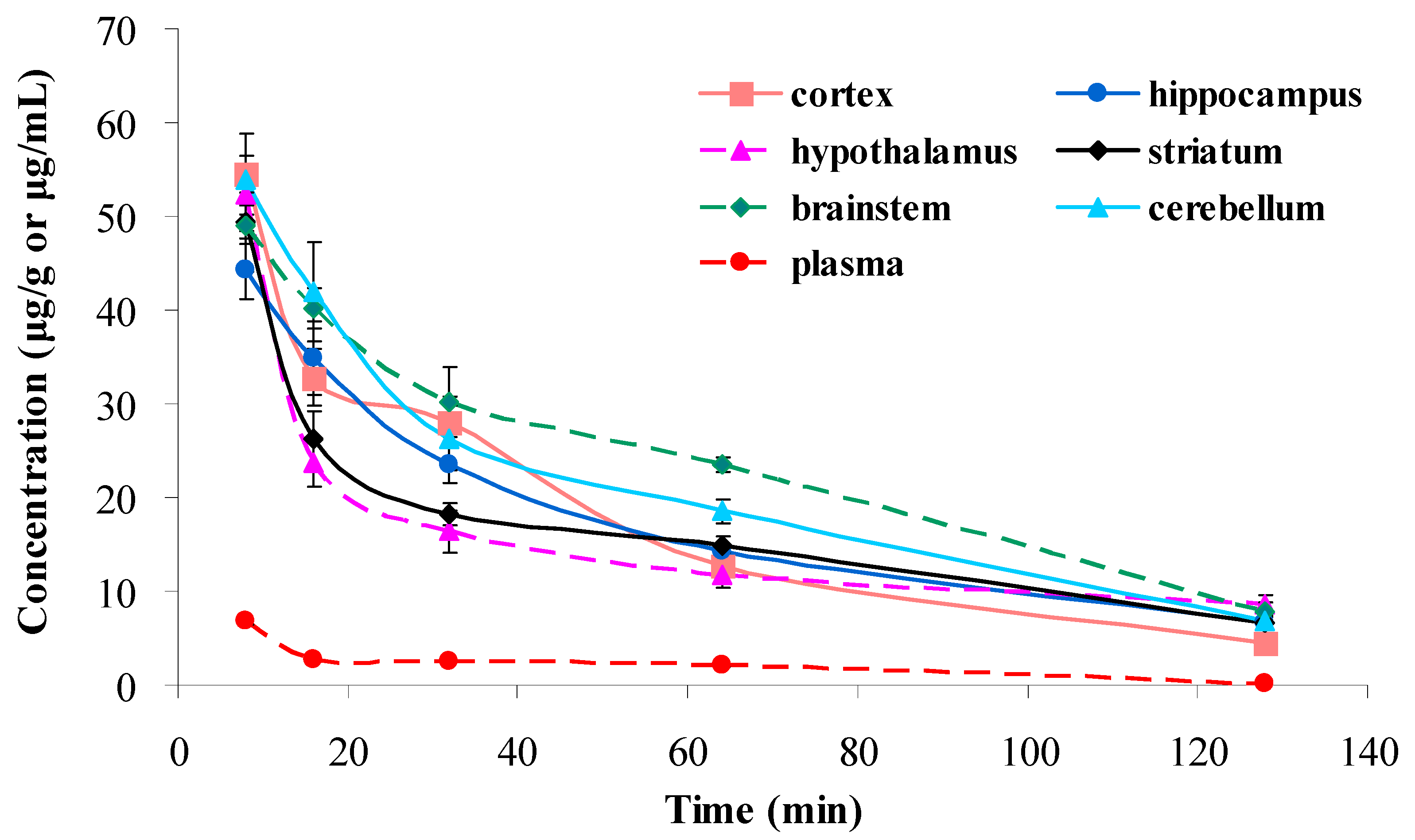
2.7. Application to Cerebral Nuclei Distribution Study
3. Experimental Section
3.1. General Information
3.2. Instrumentation and Conditions
3.3. Animals
3.4. Preparation of Standard and Quality Control Samples
3.5. Sample Preparation
3.6. Method Validation
3.7. Distribution Study
3.8. Statistical Analysis
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Chinese Pharmacopoeia Commission. Pharmacopoeia of the People’s Republic of China; China Medical Science Press: Beijing, China, 2015; Volume I, p. 136. [Google Scholar]
- Sonavane, G.S.; Sarveiya, V.P.; Kasture, V.S.; Kasture, S.B. Anxiogenic activity of Myristica fragrans seeds. Pharmacol. Biochem. Behav. 2002, 71, 239–244. [Google Scholar] [CrossRef]
- Parle, M.; Dhingra, D.; Kulkarni, S.K. Improvement of mouse memory by Myristica fragrans seeds. J. Med. Food 2004, 7, 157–161. [Google Scholar] [CrossRef] [PubMed]
- Dhingra, D.; Sharma, A. Antidepressant-like activity of n-hexane extract of nutmeg (Myristica fragrans) seeds in mice. J. Med. Food 2006, 9, 84–89. [Google Scholar] [CrossRef] [PubMed]
- Li, F.; Yang, X.W.; Krausz, K.W.; Nichols, R.G.; Xu, W.; Patterson, A.D.; Gonzalez, F.J. Modulation of colon cancer by nutmeg. J. Proteome Res. 2015, 14, 1937–1946. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Yang, X.W. Quantitative determination of neolignanoids in the seeds of Myristica fragrans. Mod. Chin. Med. 2008, 10, 10–13. [Google Scholar]
- Yang, X.W.; Huang, X.; Ahmat, M.M.T. New neolignan from seed of Myristica fragrans. China J. Chin. Mater. Med. 2008, 33, 397–402. [Google Scholar]
- Hattori, M.; Hada, S.; Watahiki, A.; Ihara, H.; Shu, Y.Z.; Kakiuchi, N.; Mizuno, T.; Namba, T. Studies on dental caries prevention by traditional medicines. X. Antibacterial action of phenolic components from mace against Streptococcus mutans. Chem. Pharm. Bull. 1986, 34, 3885–3893. [Google Scholar] [CrossRef] [PubMed]
- Murakami, Y.; Shoji, M.; Hirata, A.; Tanaka, S.; Yokoe, I.; Fujisawa, S. Dehydrodiisoeugenol, an isoeugenol dimer, inhibits lipopolysaccharide-stimulated nuclear factor kappa B activation and cyclooxygenase-2 expression in macrophages. Arch. Biochem. Biophys. 2005, 434, 326–332. [Google Scholar] [CrossRef] [PubMed]
- Aponte, J.C.; Jin, Z.; Vaisberg, A.J.; Castillo, D.; Malaga, E.; Lewis, W.H.; Sauvain, M.; Gilman, R.H.; Hammond, G.B. Cytotoxic and anti-infective phenolic compounds isolated from Mikania decora and Cremastosperma microcarpum. Planta. Med. 2011, 77, 1597–1599. [Google Scholar] [CrossRef] [PubMed]
- Hattori, M.; Yang, X.W.; Miyashiro, H.; Namba, T. Inhibitory effects of monomeric and dimeric phenylpropanoids from mace on lipid peroxidation in vivo and in vitro. Phytother. Res. 1993, 7, 395–401. [Google Scholar] [CrossRef]
- Ogata, M. Biological activity of eugenol. Aroma Res. 2004, 5, 259–262. [Google Scholar]
- Bortolomeazzi, R.; Verardo, G.; Liessi, A.; Callea, A. Formation of dehydrodiisoeugenol and dehydrodieugenol from the reaction of isoeugenol and eugenol with DPPH radical and their role in the radical scavenging activity. Food Chem. 2010, 118, 256–265. [Google Scholar] [CrossRef]
- Gross, S.S.; Wolin, M.S. Nitric oxide: Pathophysiological mechanisms. Ann. Rev. Physiol. 1995, 57, 737–769. [Google Scholar] [CrossRef] [PubMed]
- Dawson, V.L.; Dawson, T.M. Nitric oxide in neuronal degeneration. Proc. Soc. Exp. Biol. Med. 1996, 211, 33–40. [Google Scholar] [CrossRef] [PubMed]
- Cao, G.Y.; Yang, X.W.; Xu, W.; Li, F. New inhibitors of nitric oxide production from the seeds of Myristica fragrans. Food Chem. Toxicol. 2013, 62, 167–171. [Google Scholar] [CrossRef] [PubMed]
- Li, F.; Yang, X.W. Determination of dehydrodiisoeugenol in rat tissues using HPLC method. Biomed. Chromatogr. 2008, 22, 1206–1212. [Google Scholar] [CrossRef] [PubMed]
- Wu, N.; Xu, W.; Cao, G.Y.; Yang, Y.F.; Yang, X.B.; Yang, X.W. The blood-brain barrier permeability of lignans and malabaricones from the seeds of Myristica fragrans in the MDCK-pHaMDR cell monolayer model. Molecules 2016, 21. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.B.; Zhu, L.Q.; Yang, X.W. Cerebral nuclei distribution study of dehydrodiisoeugenol as an anxiogenic agent determined by RP-HPLC. Fitoterapia 2013, 84, 47–53. [Google Scholar] [CrossRef] [PubMed]
- Jemal, M. High-throughput quantitative bioanalysis by LC/MS/MS. Biomed. Chromatogr. 2000, 14, 422–429. [Google Scholar] [CrossRef]
- Li, F.; Yang, X.W. Simultaneous determination of diastereomers (+)-licarin A and isolicarin A from Myristica fragrans in rat plasma by HPLC and its application to their pharmacokinetics. Planta Med. 2008, 74, 880–884. [Google Scholar] [CrossRef] [PubMed]
- Li, F.; Yang, X.W. Analysis of anti-inflammatory dehydrodiisoeugenol and metabolites excreted in rat feces and urine using HPLC-UV. Biomed. Chromatogr. 2012, 26, 703–707. [Google Scholar] [CrossRef] [PubMed]
- Sample Availability: Samples of the compounds are not available from the authors.
| Sample | Calibration Curve | r2 | Linear Range (μg/mL) |
|---|---|---|---|
| Plasma | y = 1.5501x + 0.0719 | 0.9940 | 0.05–2.0 |
| Hippocampus | y = 1.2178x + 0.0420 | 0.9948 | 0.05–1.0 |
| Striatum | y = 2.1454x − 0.1719 | 0.9956 | 0.10–2.0 |
| Cortex | y = 1.7553x + 0.1834 | 0.9908 | 2.00–32 |
| Cerebellum | y = 1.5364x + 0.0358 | 0.9972 | 1.00–20 |
| Brainstem | y = 1.5864x + 0.3528 | 0.9944 | 0.50–12 |
| Hypothalamus | y = 1.6004x + 0.0800 | 0.9948 | 0.05–1.5 |
| Samples | Spiked (μg/mL) | Intra-Day | Inter-Day | Recovery | Stability | Matrix Effect | |||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Measured (μg/mL) | RSD (%) | Accuracy (%) | Measured (μg/mL) | RSD (%) | Accuracy (%) | Mean (%) | RSD (%) | Accuracy (%) | RSD (%) | Mean (%) | RSD (%) | ||
| Plasma | 0.25 | 0.24 | 7.31 | 96.81 | 0.25 | 3.14 | 99.10 | 84.31 | 1.91 | 94.77 | 5.58 | 93.02 | 7.22 |
| 0.50 | 0.55 | 6.86 | 110.5 | 0.57 | 2.46 | 113.3 | 81.41 | 1.60 | 108.7 | 5.47 | 95.02 | 7.76 | |
| 1.50 | 1.55 | 5.08 | 103.6 | 1.57 | 1.42 | 104.4 | 86.01 | 3.60 | 103.8 | 5.06 | 95.10 | 2.62 | |
| Hippocampus | 0.10 | 0.09 | 14.5 | 94.76 | 0.11 | 9.37 | 105.4 | 84.09 | 3.70 | 94.40 | 13.9 | 90.26 | 4.90 |
| 0.20 | 021 | 7.54 | 104.6 | 0.22 | 2.02 | 111.3 | 79.83 | 4.80 | 110.4 | 3.41 | 94.58 | 11.9 | |
| 0.80 | 0.80 | 3.77 | 100.4 | 0.79 | 5.19 | 98.20 | 80.35 | 1.17 | 97.73 | 2.64 | 96.10 | 10.7 | |
| Striatum | 0.25 | 0.27 | 3.40 | 107.1 | 0.27 | 4.61 | 107.4 | 82.33 | 13.6 | 107.8 | 2.46 | 93.09 | 9.44 |
| 0.50 | 0.44 | 4.72 | 88.84 | 0.45 | 5.44 | 89.98 | 88.72 | 9.99 | 85.85 | 2.37 | 90.49 | 7.28 | |
| 1.50 | 1.38 | 1.48 | 92.33 | 1.43 | 2.48 | 95.66 | 87.82 | 7.63 | 93.91 | 4.02 | 90.33 | 5.18 | |
| Cortex | 4.00 | 3.98 | 2.22 | 99.42 | 3.99 | 2.61 | 99.67 | 83.97 | 7.98 | 99.61 | 2.04 | 91.78 | 2.53 |
| 8.00 | 7.49 | 7.12 | 93.64 | 7.79 | 3.25 | 97.31 | 87.52 | 4.42 | 94.36 | 2.11 | 97.44 | 1.41 | |
| 24.0 | 23.1 | 9.50 | 96.06 | 22.0 | 4.77 | 91.74 | 83.29 | 8.47 | 92.20 | 4.55 | 95.16 | 5.20 | |
| Cerebellum | 2.50 | 2.61 | 7.45 | 104.4 | 2.69 | 2.64 | 107.7 | 84.35 | 7.62 | 104.5 | 7.55 | 90.42 | 5.33 |
| 5.00 | 5.43 | 4.87 | 108.7 | 5.26 | 5.53 | 105.2 | 81.37 | 9.14 | 108.1 | 5.80 | 91.74 | 8.46 | |
| 15.0 | 15.2 | 5.20 | 101.2 | 15.4 | 3.93 | 102.7 | 87.37 | 1.42 | 99.84 | 3.29 | 92.22 | 4.31 | |
| Brainstem | 1.00 | 0.97 | 6.77 | 96.95 | 0.94 | 9.29 | 93.94 | 82.67 | 8.57 | 89.61 | 1.51 | 93.59 | 2.79 |
| 2.00 | 1.85 | 6.74 | 92.50 | 1.81 | 4.83 | 90.71 | 85.41 | 2.94 | 89.48 | 5.36 | 86.58 | 3.59 | |
| 8.00 | 8.33 | 2.47 | 104.2 | 8.13 | 4.13 | 101.6 | 81.75 | 5.45 | 100.4 | 3.15 | 94.39 | 10.3 | |
| Hypothalamus | 0.10 | 0.11 | 2.52 | 112.9 | 0.11 | 2.62 | 114.1 | 78.42 | 3.10 | 112.6 | 2.14 | 94.20 | 6.87 |
| 0.30 | 0.30 | 13.0 | 101.0 | 0.33 | 2.10 | 109.9 | 89.20 | 6.17 | 99.80 | 11.7 | 90.28 | 5.09 | |
| 1.20 | 1.25 | 5.28 | 103.9 | 1.27 | 1.69 | 105.5 | 82.33 | 3.50 | 103.2 | 4.70 | 93.58 | 4.64 | |
| Samples | t1/2 (min) | AUC0→t (μg·min/g) | AUC0→∞ (μg·min/g) | CL (μg·kg/min) | MRT (min) | Cmax (μg/g) | V1 (L/kg) |
|---|---|---|---|---|---|---|---|
| Plasma | 54.326 ± 0.877 | 309.679 ± 13.254 a | 479.692 ± 26.388 a | 0.083 ± 0.005 b | 42.709 ±1.701 | 6.827 ± 0.278 c | 0.008 ± 0.005 |
| Hippocampus | 62.212 ± 3.833 | 2631.272 ± 230.935 | 2463.297 ± 300.575 | 0.012 ± 0.001 | 63.460 ±0.751 | 44.406 ± 3.236 | 0.669 ± 0.025 |
| Striatum | 69.315 ± 0.010 | 2498.242 ± 226.990 | 3542.121 ± 327.205 | 0.011 ± 0.001 | 73.439 ± 0.330 | 49.506 ± 5.411 | 0.213 ± 0.022 |
| Cortex | 38.227 ± 2.283 | 2742.581 ± 299.720 | 3400.941 ± 429.470 | 0.012 ± 0.002 | 46.490 ± 3.270 | 54.539 ± 4.363 | 0.001 ± 0.001 |
| Cerebellum | 56.059 ± 1.272 | 3144.154 ± 250.522 | 4083.262 ± 251.445 | 0.010 ± 0.001 | 57.935 ± 841.000 | 53.940 ± 1.391 | 0.504 ± 0.064 |
| Brainstem | 55.923 ± 2.433 | 3372.296 ± 223.017 | 4447.434 ± 297.052 | 0.009 ± 0.001 | 65.185 ± 2.772 | 49.082 ± 2.120 | 0.372 ± 0.312 |
| Hypothalamus | 69.315 ± 0.001 | 2418.876 ± 241.893 | 3887.033 ± 222.825 | 0.011 ± 0.001 | 116.471 ± 27.269 | 52.423 ± 3.986 | 0.199 ± 0.008 |
© 2016 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons by Attribution (CC-BY) license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhang, Y.-B.; Yang, X.-B.; Yang, X.-W.; Xu, W.; Li, F.; Gonzezal, F.J. Liquid Chromatography with Tandem Mass Spectrometry: A Sensitive Method for the Determination of Dehydrodiisoeugenol in Rat Cerebral Nuclei. Molecules 2016, 21, 321. https://doi.org/10.3390/molecules21030321
Zhang Y-B, Yang X-B, Yang X-W, Xu W, Li F, Gonzezal FJ. Liquid Chromatography with Tandem Mass Spectrometry: A Sensitive Method for the Determination of Dehydrodiisoeugenol in Rat Cerebral Nuclei. Molecules. 2016; 21(3):321. https://doi.org/10.3390/molecules21030321
Chicago/Turabian StyleZhang, You-Bo, Xin-Bao Yang, Xiu-Wei Yang, Wei Xu, Fei Li, and Frank J. Gonzezal. 2016. "Liquid Chromatography with Tandem Mass Spectrometry: A Sensitive Method for the Determination of Dehydrodiisoeugenol in Rat Cerebral Nuclei" Molecules 21, no. 3: 321. https://doi.org/10.3390/molecules21030321
APA StyleZhang, Y.-B., Yang, X.-B., Yang, X.-W., Xu, W., Li, F., & Gonzezal, F. J. (2016). Liquid Chromatography with Tandem Mass Spectrometry: A Sensitive Method for the Determination of Dehydrodiisoeugenol in Rat Cerebral Nuclei. Molecules, 21(3), 321. https://doi.org/10.3390/molecules21030321






