Rhubarb Anthraquinones Protect Rats against Mercuric Chloride (HgCl2)-Induced Acute Renal Failure
Abstract
:1. Introduction
2. Results
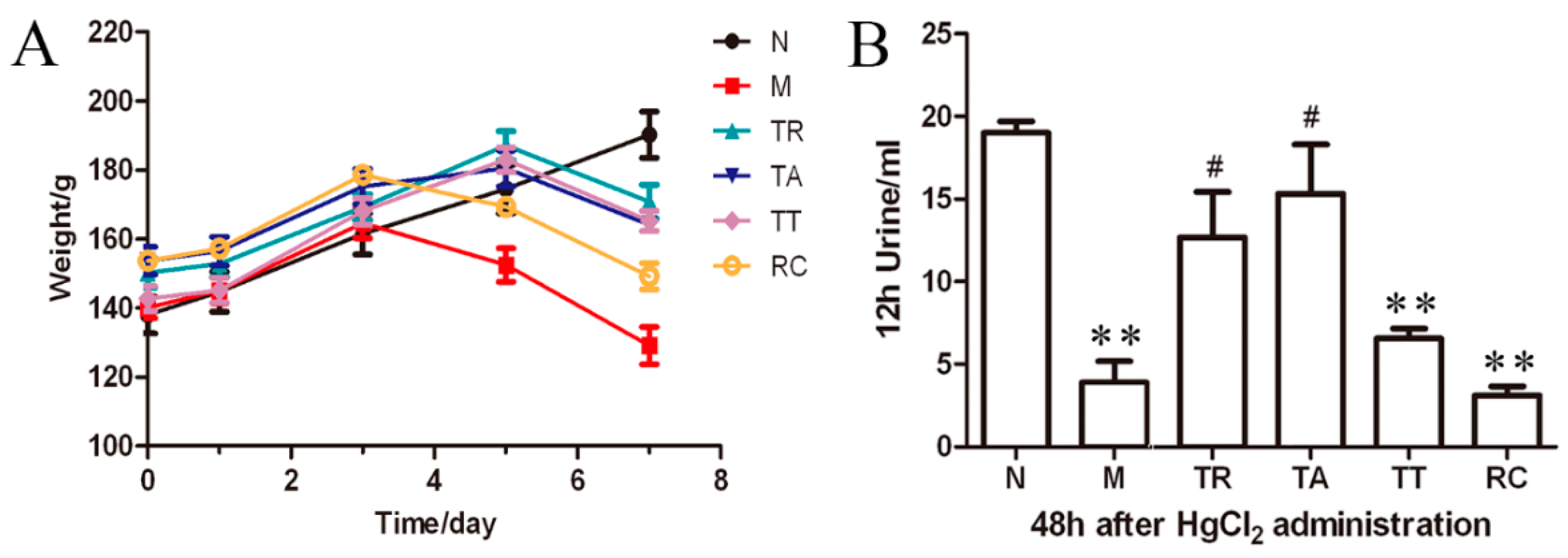
2.1. Common Changes and Survival
2.2. Histopathology
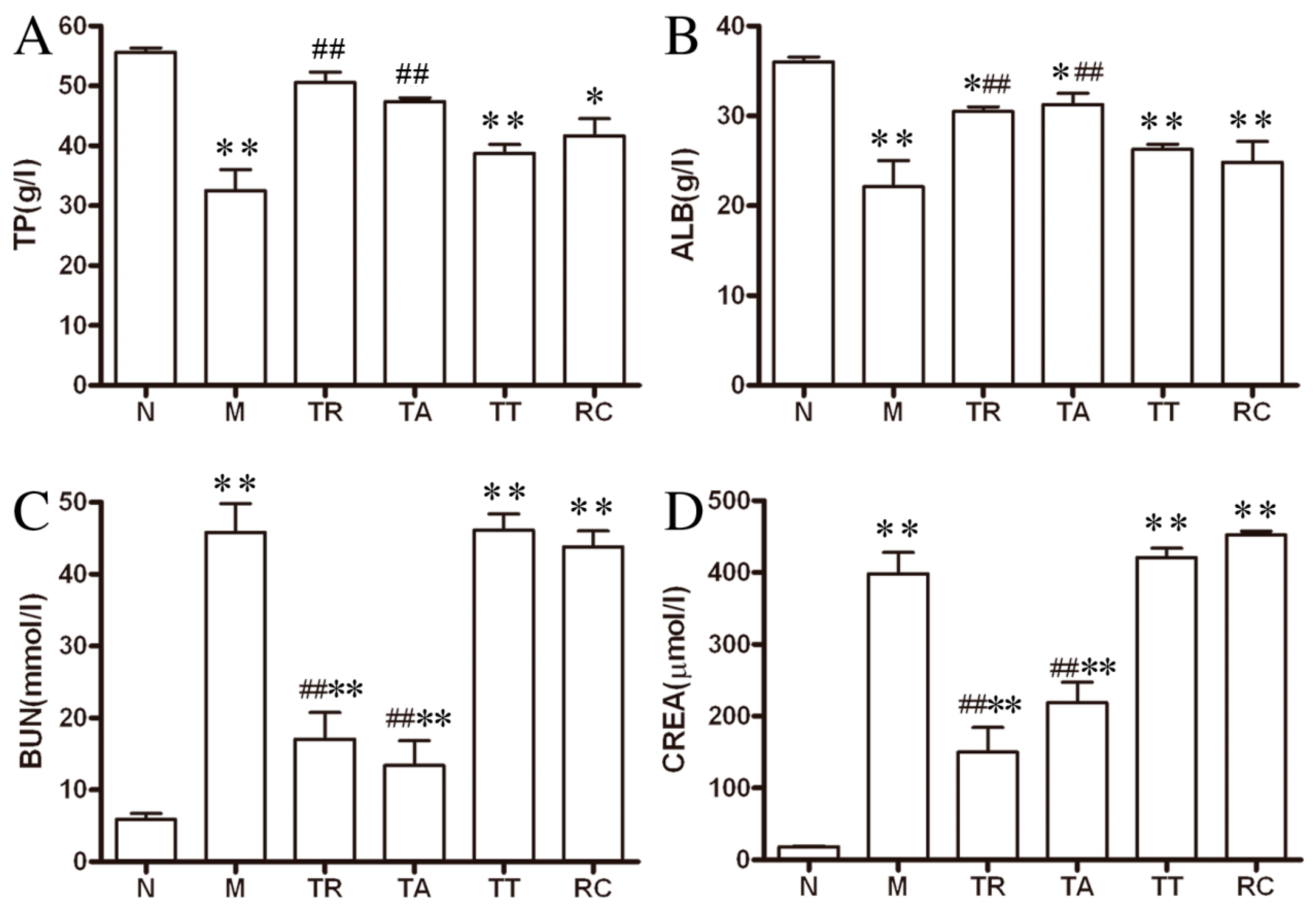
2.3. Biochemical Changes
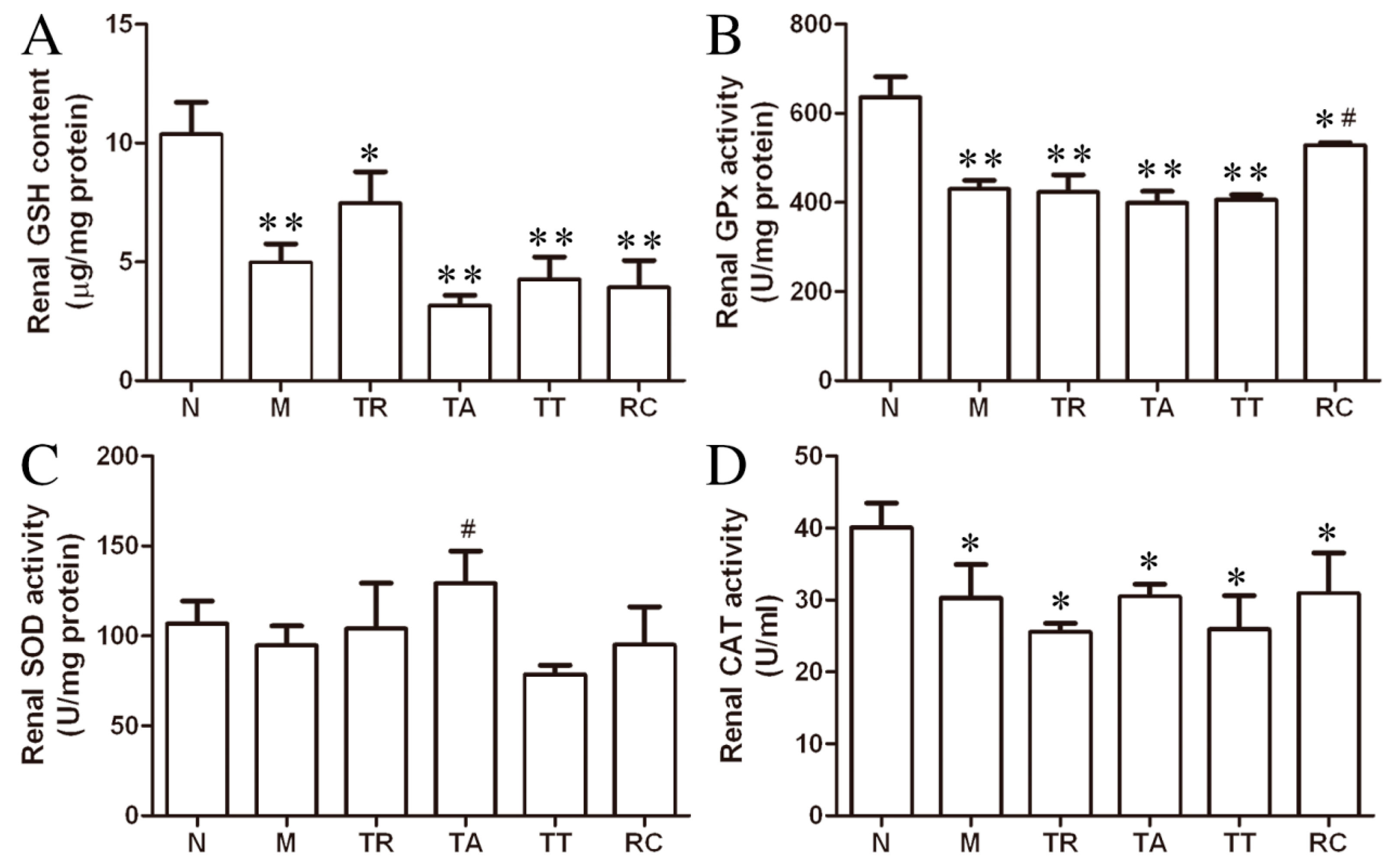
2.4. Effect on Oxidative Stress
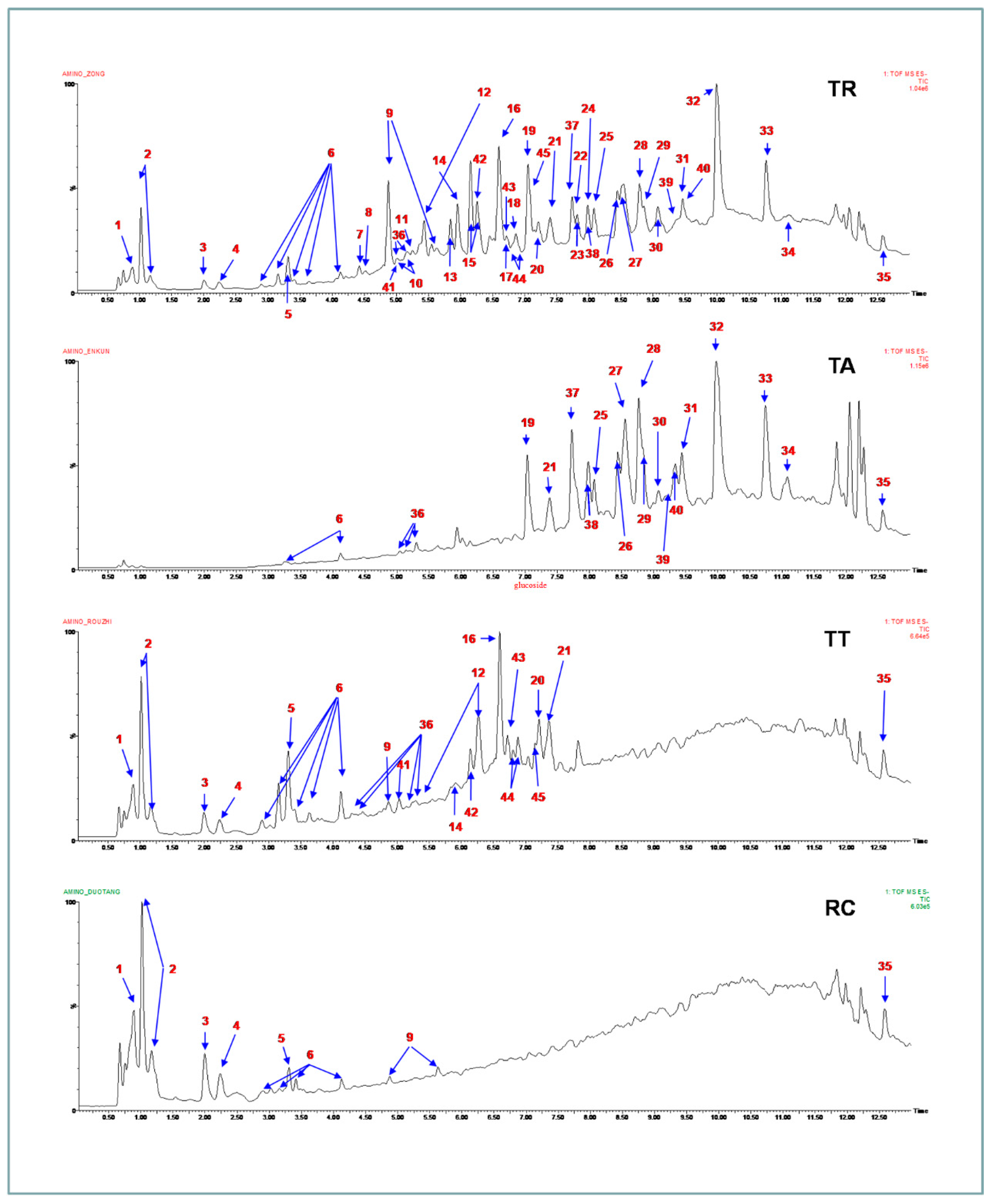
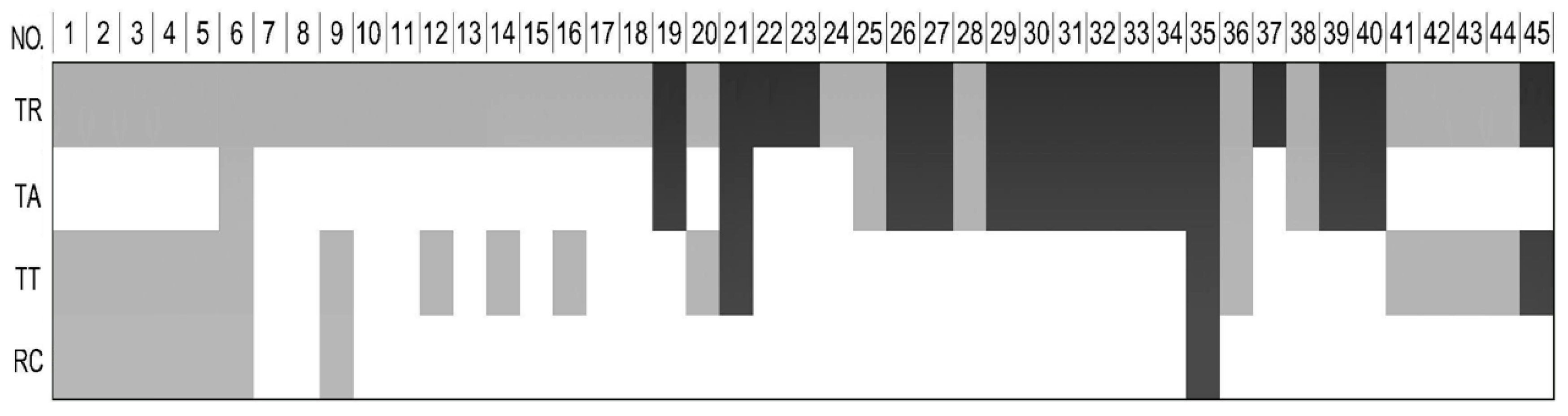
2.5. LC–QTOF-MS Identification for the Components in Each Extract
3. Discussion
4. Experimental Section
4.1. Plant Material and Reagents
4.2. Preparation and Analysis of Rhubarb Extracts
4.3. Animals and Study Design
4.4. Histopathological Examination and Biochemical Analysis
4.5. Measurement of Tissue GSH Level, GPx, T-SOD, and CAT Activities
4.6. Statistics
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Chan, T.Y. Inorganic mercury poisoning associated with skin-lightening cosmetic products. Clin. Toxicol. 2011, 49, 886–891. [Google Scholar] [CrossRef] [PubMed]
- Clarkson, T.W.; Magos, L.; Myers, G.J. The toxicology of mercury-current exposures and clinical manifestations. N. Engl. J. Med. 2003, 349, 1731–1737. [Google Scholar] [CrossRef] [PubMed]
- Harada, M. Minamata disease: Methylmercury poisoning in Japan caused by environmental pollution. Crit. Rev. Toxicol. 1995, 25, 1–24. [Google Scholar] [CrossRef] [PubMed]
- Bernhoft, R.A. Mercury toxicity and treatment: A review of the literature. J. Environ. Pub. Health 2012. [Google Scholar] [CrossRef] [PubMed]
- Zalups, R.K. Molecular Interactions with Mercury in the Kidney. Pharmacol. Rev. 2000, 52, 113–144. [Google Scholar] [PubMed]
- Rafati-Rahimzadeh, M.; Rafati-Rahimzadeh, M.; Kazemi, S.; Moghadamnia, A.A. Current approaches of the management of mercury poisoning: Need of the hour. DARU-J. Pharm. Sci. 2014, 22, 46. [Google Scholar] [CrossRef] [PubMed]
- Cao, Y.; Skaug, M.A.; Andersen, O.; Aaseth, J. Chelation therapy in intoxications with mercury, lead and copper. J. Trace Elem. Med. Biol. 2015, 31, 188–192. [Google Scholar] [CrossRef] [PubMed]
- Andersen, O. Chemical and biological considerations in the treatment of metal intoxications by chelating agents. Mini-Rev. Med. Chem. 2004, 4, 11–21. [Google Scholar] [CrossRef] [PubMed]
- Flora, S.J.S.; Kumar, P. Biochemical and immunotoxicological evaluation of metal chelating drugs in rats. Drug Invest. 1993, 5, 269–273. [Google Scholar] [CrossRef]
- Domingo, J.L. Developmental toxicity of metal chelating agents. Reprod. Toxicol. 1998, 12, 499–510. [Google Scholar] [CrossRef]
- Pharmacopoeia of the People’s Republic of China, 1st ed.; China Chemical Industry Press: Beijing, China, 2010; pp. 167–165.
- Zheng, J.H.; Guo, D.A. Modern research on Rhubarb; Peking University Medical Press: Beijing, China, 2007; pp. 453–454. [Google Scholar]
- Dong, M.X.; Jia, Y.; Zhang, Y.B.; Li, C.C.; Geng, Y.T.; Zhou, L.; Li, X.Y.; Liu, J.C.; Niu, Y.C. Emodin protects rat liver from CCl4-induced fibrogenesis via inhibition of hepatic stellate cells activation. World J. Gastroenterol. 2009, 15, 4753–4762. [Google Scholar] [CrossRef] [PubMed]
- Choi, S.B.; Ko, B.S.; Park, S.K.; Jang, J.S.; Park, S. Insulin sensitizing and alpha-glucoamylase inhibitory action of sennosides, rhein and rhaponticin in Rhein Rhizoma. Life Sci. 2006, 789, 934–942. [Google Scholar] [CrossRef] [PubMed]
- Jha, V. Herbal medicines and chronic kidney disease. Nephrology 2010, 15, 10–17. [Google Scholar] [CrossRef] [PubMed]
- Zhong, Y.; Deng, Y.; Chen, Y.; Chuang, P.Y.; He, J.C. Therapeutic use of traditional Chinese herbal medications for chronic kidney diseases. Kidney Int. 2013, 84, 1108–1118. [Google Scholar] [CrossRef] [PubMed]
- Li, X.M.; Wang, H.Y. Chinese herbal medicine in the treatment of chronic kidney disease. Adv. Chronic Kidney D 2005, 12, 276–281. [Google Scholar] [CrossRef]
- Zhang, G.; El Nahas, A.M. The effect of rhubarb extract on experimental renal fibrosis. Nephrol. Dial. Transpl. 1996, 11, 186–190. [Google Scholar] [CrossRef]
- Sanada, H. Study on the clinical effect of rhubarb on nitrogen-metabolism abnormality due to chronic renal failure and its mechanism. Nihon Jinzo Gakkai Shi 1996, 38, 379–387. [Google Scholar] [PubMed]
- Ye, M.; Guo, D.A.; Han, J.; Chen, H.B.; Zheng, J.H. Analysis of phenolic compounds in Rhubarbs using liquid chromatography coupled with electrospray ionization mass spectrometry. J. Am. Soc. Mass Spectr. 2007, 18, 82–91. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.B.; Zhao, Y.L.; Xiao, X.; Li, H.; Zhao, H.; Zhang, P.; Jin, C. Assessment of the renal protection and hepatotoxicity of rhubarb extract in rats. J. Ethnopharmacol. 2009, 124, 18–25. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.B.; Qin, Y.; Kong, W.J.; Wang, Z.W.; Zeng, L.N.; Fang, F.; Cheng, J.; Zhao, Y.L.; Xiao, X.H. Identification of the antidiarrhoeal components in official rhubarb using liquid chromatography–tandem mass spectrometry. Food Chem. 2011, 129, 1737–1743. [Google Scholar] [CrossRef]
- Song, R.; Xu, L.; Xu, F.; Dong, H.; Tian, Y.; Zhang, Z. Metabolic analysis of rhubarb extract by rat intestinal bacteria using liquid chromatography–tandem mass spectrometry. Biomed. Chromatogr. 2011, 25, 417–426. [Google Scholar] [CrossRef] [PubMed]
- Sakulpanich, A.; Gritsanapan, W. Determination of anthraquinone glycoside content in Cassia fistula leaf extracts for alternative source of laxative drug. Int. J. Biomed. Pharm. Sci. 2009, 3, 42–45. [Google Scholar]
- Zhou, X.M.; Chen, Q.H. Biochemical study of Chinese rhubarb. XXII. Inhibitory effect of anthraquinone derivatives on Na+-K+-ATPase of the rabbit renal medulla and their diuretic action. Acta Pharm. Sin. 1988, 23, 17–20. [Google Scholar]
- Yang, H.; Zhou, Z.G.; Xu, J.; Li, F.Y.; Yi, T.; Huang, C.H. A highly selective ratiometric chemosensor for Hg2+ based on the anthraquinone derivative with urea groups. Tetrahedron 2007, 63, 6732–6736. [Google Scholar] [CrossRef]
- Huang, Q.; Lu, G.D.; Shen, H.M.; Maxey, C.M.; Chung, C.N.O. Anti-cancer properties of anthraquinones from rhubarb. Med. Res. Rev. 2007, 27, 609–630. [Google Scholar] [CrossRef] [PubMed]
- Agrawal, S.; Flora, G.; Bhatnagar, P.; Flora, S.J.S. Comparative oxidative stress, metallothionein induction and organ toxicity following chronic exposure to arsenic, lead and mercury in rats. Cell. Mol. Biol. 2014, 60, 13–21. [Google Scholar] [PubMed]
- Ozbek, E. Induction of oxidative stress in kidney. Int. J. Nephrol. 2012, 2012, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Zeng, L.N.; Ma, Z.J.; Zhao, Y.L.; Zhang, L.D.; Li, R.S.; Wang, J.B.; Zhang, P.; Yan, D.; Li, Q.; Jiang, B.Q.; et al. The protective and toxic effects of rhubarb tannins and anthraquinones in treating hexavalent chromium-injured rats: The Yin/Yang actions of rhubarb. J. Hazard. Mater. 2013, 246, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Su, X.Y.; Wang, Z.Y.; Liu, J.R. In vitro and in vivo antioxidant activity of Pinus koraiensis seed extract containing phenolic compounds. Food Chem. 2009, 117, 681–686. [Google Scholar]
- Ercal, N.; Gurer-Orhan, H.; Aykin-Burns, N. Toxic metals and oxidative stress part I: Mechanisms involved in metal-induced oxidative damage. Curr. Top. Med. Chem. 2001, 1, 529–539. [Google Scholar] [CrossRef] [PubMed]
- European Pharmacopoeia, 5th ed.; European directorate for the quality of medicines Press: Strasbourg, France, 2004; pp. 2363–2364.
- Han, J.; Ye, M.; Xu, M.; Qiao, X.; Chen, H.B.; Wang, B.R.; Zheng, J.H.; Guo, D.A. Comparison of phenolic compounds of rhubarbs in the section deserticola with Rheum palmatum by HPLC-DAD-ESI-MSn. Plant. Med. 2008, 74, 873–879. [Google Scholar] [CrossRef] [PubMed]
- Sample Availability: Samples of the compounds TR, TA, TT, and RC are available from the authors.
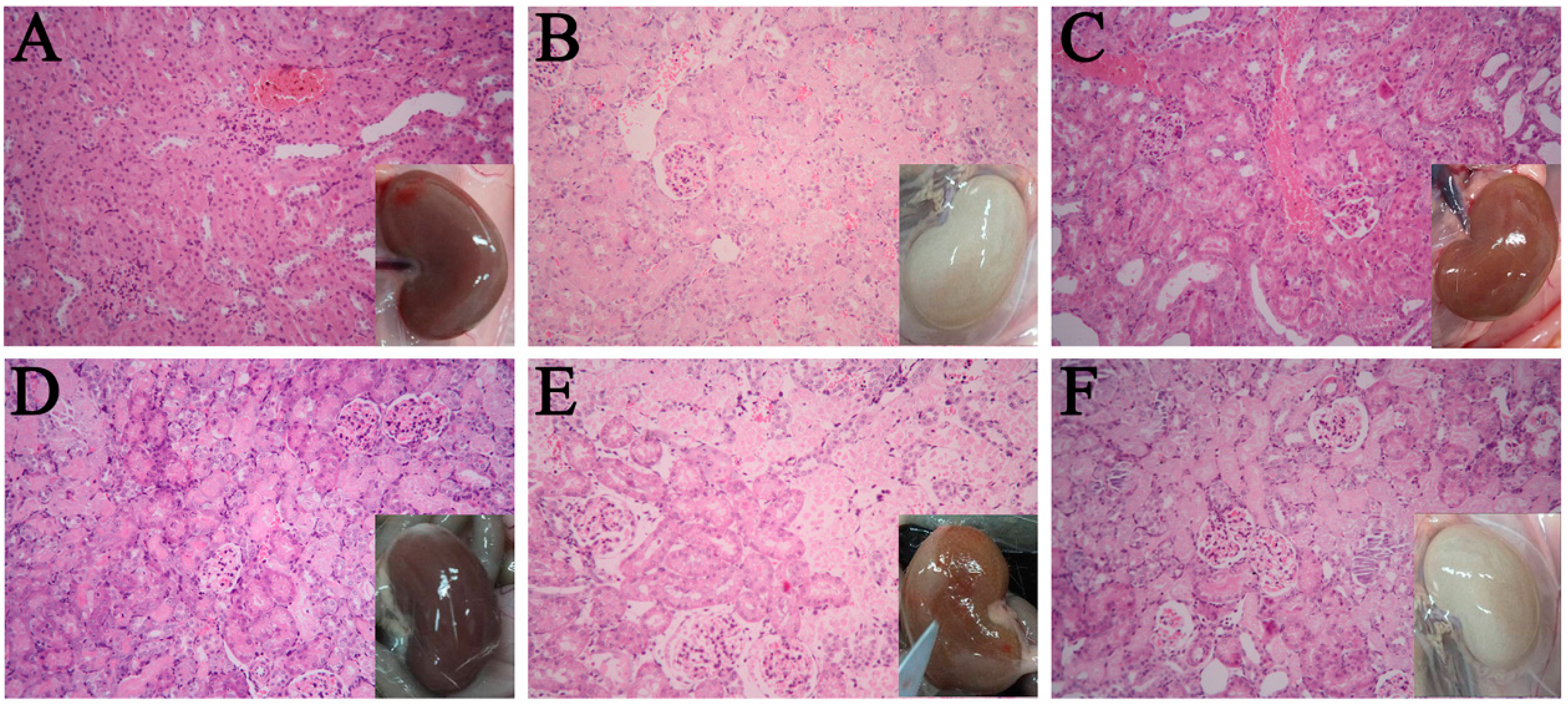
| Groups | Survival a | Renal Tubule Epithelial Cells b | |||
|---|---|---|---|---|---|
| Swelling | Granular Degeneration | Necrosis | Interstitial Vascular Congestion | ||
| N | 8 | - | - | - | - |
| M | 2 | +++ | ++++ | ++++ | ++++ |
| TR | 7 | ++ | + | ++ | ++ |
| TA | 7 | + | ++ | + | + |
| TT | 4 | ++++ | +++ | +++ | +++ |
| RC | 3 | +++ | +++ | +++ | ++++ |
| Peak Number | RT (min) | [M − H]− | Identification | Source |
|---|---|---|---|---|
| 1 | 0.89 | 195.0505 | galactonic acid | TR, TT, RC |
| 2 | 1.02, 1.16 | 683.2243 | galabiose | TR, TT, RC |
| 3 | 2.00 | 191.0189 | citric acid | TR, TT, RC |
| 4 | 2.23 | 128.0351 | pyroglutamic acid | TR, TT, RC |
| 5 | 3.31 | 169.0136 | gallic acid | TR, TT, RC |
| 6 | 2.89, 3.16, 3.42, 3.63, 4.13 | 331.0665 | galloyl glucose | TR, TA, TT, RC |
| 7 | 4.42 | 577.1337 | procyanidin b5 | TR |
| 8 | 4.51 | 451.1238 | catechin glucoside | TR |
| 9 | 4.88, 5.54 | 289.0716 | catechin | TR, TT, RC |
| 10 | 5.01, 5.16 | 729.1448 | epicatechin-epicatechin-gallate | TR |
| 11 | 5.25 | 635.0876 | tri-O-galloyl glucose | TR |
| 12 | 5.33 | 881.1569 | di-O-galloyl procyanidin | TR, TT |
| 13 | 5.84 | 435.1291 | catechin O-rhamnopyranoside | TR |
| 14 | 5.95 | 441.0816 | catechin-O-gallate | TR, TT |
| 15 | 6.15, 6.26 | 477.1388 | coumaroyl-O-galloyl glucose | TR |
| 16 | 6.6 | 541.1349 | resveratrol-(O-galloyl)-glucose | TR, TT |
| 17 | 6.72 | 629.1499 | coumaroyl-di-O-galloyl glucose | TR |
| 18 | 6.86 | 571.1446 | rhapontigenin-(O-galloyl)-glucose | TR |
| 19 | 7.05 | 445.0756 | rhein-O-glucoside | TR, TA |
| 20 | 7.22 | 393.1177 | hydroxymusizin-O-glucopyranoside | TR, TT |
| 21 | 7.4 | 861.191 | sennoside A/B | TR, TA, TT |
| 22 | 7.74 | 487.0867 | rhein-O-acetyl glucose | TR |
| 23 | 7.82 | 947.1873 | sennoside | TR |
| 24 | 7.98 | 623.176 | dicoumaroyl-O-galloyl glucose | TR |
| 25 | 8.07 | 407.1344 | torachrysone-O-glucoside | TR, TA |
| 26 | 8.44 | 415.1017 | chrysophanol-O-glucoside | TR, TA |
| 27 | 8.54 | 431.0966 | emodin-O-glucoside | TR, TA |
| 28 | 8.79 | 607.1818 | dicinnamoyl-O-galloyl glucose | TR, TA |
| 29 | 8.86 | 457.1127 | chrysophanol-O-(O-acetyl)-glucoside | TR, TA |
| 30 | 9.07 | 445.1121 | rhein-O-glucoside | TR, TA |
| 31 | 9.47 | 269.0452 | aloe-emodin | TR, TA |
| 32 | 9.99 | 283.0239 | rhein | TR, TA |
| 33 | 10.76 | 269.0448 | emodin | TR, TA |
| 34 | 11.11 | 253.0501 | chrysophanol | TR, TA |
| 35 | 12.6 | 283.2631 | physcion | TR, TA, TT, RC |
| 36 | 4.26, 4.34, 5.05, 5.14, 5.30 | 483.0777 | di-O-galloyl glucose | TR, TA, TT |
| 37 | 7.72 | 487.0856 | rhein-O-(O-acetyl)-glucoside | TR, TA |
| 38 | 7.99 | 623.1789 | dicoumaroyl-O-galloyl glucose | TR, TA |
| 39 | 9.19 | 473.1071 | aloe-emodin-O-(O-acetyl)-glucoside | TR, TA |
| 40 | 9.33 | 487.1239 | rhein-O-(O-acetyl)-glucoside | TR, TA |
| 41 | 5.03 | 49.1072 | eriodictyol-glucose | TR, TT |
| 42 | 6.14 | 7.0456 | ethyl gallate | TR, TT |
| 43 | 6.72 | 629.1501 | coumaroyl-di-O-galloyl glucose | TR, TT |
| 44 | 6.80, 6.87 | 571.1438 | rhapontigenin-O-galloyl glucose | TR, TT |
| 45 | 7.04 | 847.2088 | sennoside C/D | TR, TT |
© 2016 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons by Attribution (CC-BY) license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gao, D.; Zeng, L.-N.; Zhang, P.; Ma, Z.-J.; Li, R.-S.; Zhao, Y.-L.; Zhang, Y.-M.; Guo, Y.-M.; Niu, M.; Bai, Z.-F.; et al. Rhubarb Anthraquinones Protect Rats against Mercuric Chloride (HgCl2)-Induced Acute Renal Failure. Molecules 2016, 21, 298. https://doi.org/10.3390/molecules21030298
Gao D, Zeng L-N, Zhang P, Ma Z-J, Li R-S, Zhao Y-L, Zhang Y-M, Guo Y-M, Niu M, Bai Z-F, et al. Rhubarb Anthraquinones Protect Rats against Mercuric Chloride (HgCl2)-Induced Acute Renal Failure. Molecules. 2016; 21(3):298. https://doi.org/10.3390/molecules21030298
Chicago/Turabian StyleGao, Dan, Ling-Na Zeng, Pin Zhang, Zhi-Jie Ma, Rui-Sheng Li, Yan-Ling Zhao, Ya-Ming Zhang, Yu-Min Guo, Ming Niu, Zhao-Fang Bai, and et al. 2016. "Rhubarb Anthraquinones Protect Rats against Mercuric Chloride (HgCl2)-Induced Acute Renal Failure" Molecules 21, no. 3: 298. https://doi.org/10.3390/molecules21030298
APA StyleGao, D., Zeng, L.-N., Zhang, P., Ma, Z.-J., Li, R.-S., Zhao, Y.-L., Zhang, Y.-M., Guo, Y.-M., Niu, M., Bai, Z.-F., Xiao, X.-H., Gao, W.-W., & Wang, J.-B. (2016). Rhubarb Anthraquinones Protect Rats against Mercuric Chloride (HgCl2)-Induced Acute Renal Failure. Molecules, 21(3), 298. https://doi.org/10.3390/molecules21030298






