Organic Nanomaterials and Their Applications in the Treatment of Oral Diseases
Abstract
:1. Introduction
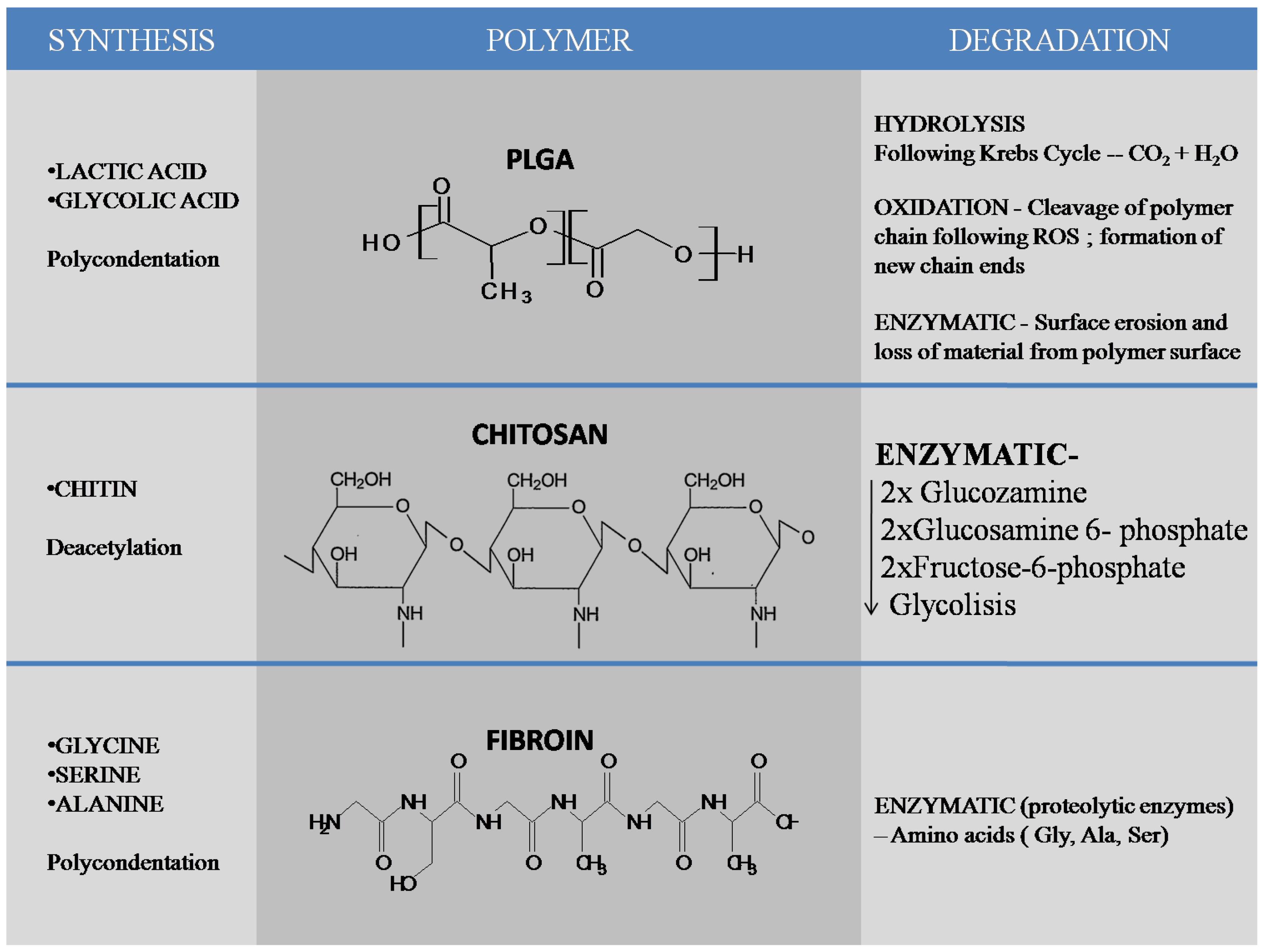
2. Main Organic Nanomaterials Used in the Dental Field
2.1. Natural Organic Nano-Delivery Systems
2.2. Chitosan Nanoparticles (CNPs)
| Chitosan Nanosystems | Application | Year | Study |
|---|---|---|---|
| Bone morphogenetic protein-2 | Bone regeneration | 2015 | [26] |
| Bone morphogenetic protein 7 | Bone regeneration | 2015 | [27] |
| Protein growth factors | Bone regeneration | 2014 | [28] |
| Dexamethasone | Dentin pulp regeneration | 2015 | [29] |
| Cetylpyridinium chloride and naf | Dental toothpastes | 2015 | [30] |
| Chlorhexidinedihydrochloride | Dental toothpastes | 2015 | [30] |
2.3. Composite Chitosan Nanoparticles
| Chitosan Composites | Dental Field | Year | Study |
|---|---|---|---|
| Chitosan-gold nanoparticles | implantology | 2015 | [70] |
| chitosan-poly(ε-caprolactone) nanofibers | bone regeneration | 2015 | [76] |
| chitosan, tripolyphosphate and chondroitin sulfatenanoparticles | bone regeneration | 2012 | [75] |
| composite chitosan/hydroxyapatite nanofibers | bone regeneration | 2008 | [80] |
| chitin hydrogel/nanohydroxyapatitenanocomposite scaffold | bone regeneration | 2011 | [82] |
| chitosan/polyvinyl alcohol/nanohydroxyapatitenanoscaffolds | bone regeneration | 2008 | [83] |
| hydroxyapatite/collagen/chitosan nanocomposite fibers | bone regeneration | 2010 | [84] |
| poly-3-hydroxybutyrate-co-3-hydroxyvalerate/chitosan/hydroxyapatite nanofibrous scaffold | bone regeneration | 2015 | [85] |
| chitosan/polyethylene oxide nanofibers | cartilage tissue engineering | 2005 | [88] |
| chitosan/polyvinyl alcohol nanofibers | oral candidiasis | 2015 | [89] |
| poly(ε-caprolactone)-poly(ethylene glycol) copolymernanofibrous mats incorporated into chitosan | regeneration of periodontium | 2015 | [90] |
| chitosan/polyethylene oxide nanofibers | wound healing | 2014 | [91] |
| poly(ε-caprolactone)/chitosan nanofibers | wound healing | 2014 | [92] |
| chitosan/polyvinyl alcohol nanofibers | wound healing | 2015 | [93] |
| chitosan/collagen nanofibrous membranes | wound healing | 2006 | [94] |
| chitosan hydrogel/nanofibrin composite | wound healing | 2012 | [95] |
| chitosan/sericin nanofibers | 2014 | [96] | |
| chitosan-Eudragit nanofibrous sheets | 2015 | [97] | |
| chitosan/arginine nanofibrous membrane | 2015 | [98] | |
| chitosan/gelatin/shape memory polyurethane nanofibers | 2015 | [99] | |
| tannic acid/chitosan/pullulan composite nanofibers | 2015 | [100] | |
| chitosan-rose bengal nanoparticles | 2014 | [101] | |
2.4. Silk Fibroin Nanoparticles
| Nanoparticle Type | Dental Field | Type of Study | Study |
|---|---|---|---|
| silk fibroin nanofiber membranes | bone tissue engineering | in vivo | [120] |
| silk fibroin nanofibrouselectrospun scaffolds | bone tissue engineering | in vitro | [119] |
| silk fibroin/chitosan nanofibers | bone tissue engineering | in vitro | [122] |
| silk fibroin/chitosan/nanohydroxyapatite nanofibrous membrane scaffolds | bone tissue engineering | in vivo | [123] |
| silk fibroin/chitosan/nanohydroxyapatite nanofibrous membrane | bone tissue engineering | in vivo | [124] |
| silk fibroin/chitin blend fibers | wound healing accelerator | in vitro | [125] |
| silk fibroin and chitosan nanofibrous membranes | wound healing accelerator | in vitro | [127] |
| silk fibroin and chitosan nanofibers | vascular tissue regeneration | in vitro | [128] |
3. Synthetic Organic Nano-Delivery Systems
3.1. PLGA Nanoparticles
| Active Substance | Dental Field | Year | Study |
|---|---|---|---|
| photosensitizer methylene blue | endodontics | 2010 | [135] |
| antibiotic minocycline | periodontics | 2012 | [134] |
| parathyroid hormone | bone regeneration | 2015 | [139] |
| recombinant human bone morphogenetic protein-7 | bone regeneration | 2007 | [145] |
| nafcillin | osteomyelitis treatment | 2008 | [146] |
| simvastatin | osteoporosis treatment | 2015 | [147] |
| lovastatin | healing of fractures | 2007 | [148] |
| LL37 (a human host defense peptide) | wound healing | 2014 | [137] |
| curcumin | wound healing | 2013 | [136] |
| vascular endothelial growth factor | wound healing | 2015 | [138] |
| dexamethasone | gingival fibroblast differentiation | 2015 | [149] |
| amphotericin B | fungal infections treatment | 2015 | [150] |
| chondrogenesis related proteins | chondrogenesis | 2014 | [142] |
| Genes SOX 5, SOX 6, SOX 9 | chondrogenesis | 2011 | [140] |
3.2. Composite PLGA Nanoparticles
| PLGA Composite Nanosytems | Dental Field | Year | Study |
|---|---|---|---|
| PLGA nanoparticles covered with chitosan | dental toothpastes | 2015 | [30] |
| PLGA/chitosan nanofibers | wound regeneration | 2014 | [170] |
| Calcium phosphate/collagen/PLGA nanofibers | bone regeneration | 2011 | [174] |
| PLGA/poly(3-hydroxybutyrate-co-3-hydroxyvalerate) nanoparticles | bone regeneration | 2010 | [175] |
| bis(poly(lactic-co-glycolic acid)-phenylalanine-polyethylene glycol-quaternary ammonium grafteddiethyltriamin nanoparticles | bone regeneration | 2014 | [176] |
| PLGA-HA copolymer nanoparticles | bone regeneration | 2014 | [177] |
| PLGA/polycaprolactonenanoparticles | bone regeneration | 2015 | [178] |
| heparin-fibrin-poly(lactide-co-caprolactone) nanoparticles | chondrogenic differentiation | 2009 | [179] |
| PLGA nanoparticlescovered with hyaluronic acid | osteogenic differentiation | 2015 | [180] |
3.3. Dendrimers, Lipid Nanoparticles and Liposome Applications in the Dental Field
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Boraschi, D.; Italiani, P. From antigen delivery system to adjuvanticy: The board application of nanoparticles in vaccinology. Vaccines 2015, 3, 930–939. [Google Scholar] [CrossRef] [PubMed]
- Brannon-Peppas, L.; Blanchette, J.O. Nanoparticle and targeted systems for cancer therapy. Adv. Drug Deliv. Rev. 2012, 64, 206–212. [Google Scholar] [CrossRef]
- Parveen, S.; Misra, R.; Sahoo, S.K. Nanoparticles: A boon to drug delivery, therapeutics, diagnostics and imaging. Nanomed. Nanotechnol. Biol. Med. 2012, 8, 147–166. [Google Scholar] [CrossRef] [PubMed]
- Kong, L.X.; Peng, Z.; Li, S.D.; Bartold, P.M. Nanotechnology and its role in the management of periodontal diseases. Periodontology 2006, 40, 184–196. [Google Scholar] [CrossRef] [PubMed]
- Narang, R.S.; Narang, J.K. Nanomedicines for dental applications-scope and future perspective. Int. J. Pharm. Investig. 2015, 5, 121–123. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.; Leeuwenburgh, S.C.; Li, Y.; Jansen, J.A. The use of micro-and nanospheres as functional components for bone tissue regeneration. Tissue Eng. B Rev. 2011, 18, 24–39. [Google Scholar] [CrossRef] [PubMed]
- Ortega-Oller, I.; Padial-Molina, M.; Galindo-Moreno, P.; O’Valle, F.; Jódar-Reyes, A.B.; Peula-García, J.M. Bone regeneration from PLGA micro-nanoparticles. BioMed Res. Int. 2015, 2015. [Google Scholar] [CrossRef] [PubMed]
- Virlan, M.J.R.; Miricescu, D.; Totan, A.; Greabu, M.; Tanase, C.; Sabliov, C.M.; Caruntu, C.; Calenic, B. Current uses of poly(lactic-co-glycolic acid) in the dental field: A comprehensive review. J. Chem. 2015, 2015. [Google Scholar] [CrossRef]
- Abdulkareem, E.H.; Memarzadeh, K.; Allaker, R.; Huang, J.; Pratten, J.; Spratt, D. Anti-biofilm activity of zinc oxide and hydroxyapatite nanoparticles as dental implant coating materials. J. Dent. 2015, 43, 1462–1469. [Google Scholar] [CrossRef] [PubMed]
- Lappas, C.M. The immunomodulatory effects of titanium dioxide and silver nanoparticles. Food Chem. Toxicol. 2015, 85, 78–83. [Google Scholar] [CrossRef] [PubMed]
- Shi, H.; Magaye, R.; Castranova, V.; Zhao, J. Titanium dioxide nanoparticles: A review of current toxicological data. Part Fibre Toxicol. 2013, 10, 15. [Google Scholar] [CrossRef] [PubMed]
- Tautzenberger, A.; Kovtun, A.; Ignatius, A. Nanoparticles and their potential for application in bone. Int. J. Nanomed. 2012, 7, 4545–4557. [Google Scholar] [CrossRef] [PubMed]
- Danhier, F.; Ansorena, E.; Silva, J.M.; Coco, R.; Le Breton, A.; Préat, V. PLGA-based nanoparticles: An overview of biomedical applications. J. Control. Release 2012, 161, 505–522. [Google Scholar] [CrossRef]
- Ige, O.O.; Umoru, L.E.; Aribo, S. Natural products: A minefield of biomaterials. ISRN Mater. Sci. 2012, 2012. [Google Scholar] [CrossRef]
- Peng, H.; Yin, Z.; Liu, H.; Chen, X.; Feng, B.; Yuan, H.; Su, B.; Ouyang, H.; Zhang, Y. Electrospun biomimetic scaffold of hydroxyapatite/chitosan supports enhanced osteogenic differentiation of mMSCs. Nanotechnology 2012, 23, 485102. [Google Scholar] [CrossRef] [PubMed]
- Laurencin, C.T.; Jiang, T.; Kumbar, S.G.; Nair, L.S. Biologically active chitosan systems for tissue engineering and regenerative medicine. Curr. Top. Med. Chem. 2008, 8, 354–364. [Google Scholar] [CrossRef]
- Galler, K.; D’souza, R.; Hartgerink, J.; Schmalz, G. Scaffolds for dental pulp tissue engineering. Adv. Dent. Res. 2011, 23, 333–339. [Google Scholar] [CrossRef] [PubMed]
- Bansal, V.; Sharma, P.K.; Sharma, N.; Pal, O.P.; Malviya, R. Applications of chitosan and chitosan derivatives in drug delivery. Adv. Biol. Res. 2011, 5, 28–37. [Google Scholar]
- Bernkop-Schnürch, A.; Dünnhaupt, S. Chitosan-based drug delivery systems. Eur. J. Pharm. Biopharm. 2012, 81, 463–469. [Google Scholar] [CrossRef] [PubMed]
- Laurencin, C.T.; Ashe, K.M.; Henry, N.; Kan, H.M.; Lo, K.W.-H. Delivery of small molecules for bone regenerative engineering: Preclinical studies and potential clinical applications. Drug Discov. Today 2014, 19, 794–800. [Google Scholar] [CrossRef] [PubMed]
- Sonia, T.; Sharma, C.P. Chitosan and its derivatives for drug delivery perspective. In Chitosan for Biomaterials I; Springer: Heidelberg, Germany, 2011; pp. 23–53. [Google Scholar]
- Chen, J.; Pan, P.; Zhang, Y.; Zhong, S.; Zhang, Q. Preparation of chitosan/nano hydroxyapatite organic-inorganic hybrid microspheres for bone repair. Colloids Surf. B Biointerfaces 2015, 134, 401–407. [Google Scholar] [CrossRef] [PubMed]
- Meinel, L.; Betz, O.; Fajardo, R.; Hofmann, S.; Nazarian, A.; Cory, E.; Hilbe, M.; McCool, J.; Langer, R.; Vunjak-Novakovic, G. Silk based biomaterials to heal critical sized femur defects. Bone 2006, 39, 922–931. [Google Scholar] [CrossRef] [PubMed]
- Vepari, C.; Kaplan, D.L. Silk as a biomaterial. Prog. Polym. Sci. 2007, 32, 991–1007. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Wenk, E.; Matsumoto, A.; Meinel, L.; Li, C.; Kaplan, D.L. Silk microspheres for encapsulation and controlled release. J. Control. Release 2007, 117, 360–370. [Google Scholar] [CrossRef] [PubMed]
- Poth, N.; Seiffart, V.; Gross, G.; Menzel, H.; Dempwolf, W. Biodegradable chitosan nanoparticle coatings on titanium for the delivery of BMP-2. Biomolecules 2015, 5, 3–19. [Google Scholar] [CrossRef] [PubMed]
- Eap, S.; Keller, L.; Schiavi, J.; Huck, O.; Jacomine, L.; Fioretti, F.; Gauthier, C.; Sebastian, V.; Schwinté, P.; Benkirane-Jessel, N. A living thick nanofibrous implant bifunctionalized with active growth factor and stem cells for bone regeneration. Int. J. Nanomed. 2015, 10, 1061–1075. [Google Scholar]
- Eap, S.; Ferrand, A.; Schiavi, J.; Keller, L.; Kokten, T.; Fioretti, F.; Mainard, D.; Ladam, G.; Benkirane-Jessel, N. Collagen implants equipped with “fish scale”-like nanoreservoirs of growth factors for bone regeneration. Nanomedicine 2014, 9, 1253–1261. [Google Scholar] [CrossRef] [PubMed]
- Shrestha, S.; Diogenes, A.; Kishen, A. Temporal-controlled dexamethasone releasing chitosan nanoparticle system enhances odontogenic differentiation of stem cells from apical papilla. J. Endod. 2015, 41, 1253–1258. [Google Scholar] [CrossRef] [PubMed]
- Chronopoulou, L.; Nocca, G.; Castagnola, M.; Paludetti, G.; Ortaggi, G.; Sciubba, F.; Bevilacqua, M.; Lupi, A.; Gambarini, G.; Palocci, C. Chitosan based nanoparticles functionalized with peptidomimetic derivatives for oral drug delivery. New Biotechnol. 2016, 33, 23–31. [Google Scholar] [CrossRef] [PubMed]
- Ferrand, A.; Eap, S.; Richert, L.; Lemoine, S.; Kalaskar, D.; Demoustier-Champagne, S.; Atmani, H.; Mély, Y.; Fioretti, F.; Schlatter, G. Osteogenetic properties of electrospun nanofibrous PCL scaffolds equipped with chitosan-based nanoreservoirs of growth factors. Macromol. Biosci. 2014, 14, 45–55. [Google Scholar] [CrossRef] [PubMed]
- Cao, L.; Werkmeister, J.A.; Wang, J.; Glattauer, V.; McLean, K.M.; Liu, C. Bone regeneration using photocrosslinked hydrogel incorporating rhBMP-2 loaded 2-N, 6-O-sulfated chitosan nanoparticles. Biomaterials 2014, 35, 2730–2742. [Google Scholar] [CrossRef] [PubMed]
- Zhou, H.; Qian, J.; Wang, J.; Yao, W.; Liu, C.; Chen, J.; Cao, X. Enhanced bioactivity of bone morphogenetic protein-2 with low dose of 2-N, 6-O-sulfated chitosan in vitro and in vivo. Biomaterials 2009, 30, 1715–1724. [Google Scholar] [CrossRef]
- Ho, M.-H.; Liao, M.-H.; Lin, Y.-L.; Lai, C.-H.; Lin, P.-I.; Chen, R.-M. Improving effects of chitosan nanofiber scaffolds on osteoblast proliferation and maturation. Int. J. Nanomed. 2014, 9, 4293–4304. [Google Scholar]
- Datta, P.; Ghosh, P.; Ghosh, K.; Maity, P.; Samanta, S.K.; Ghosh, S.K.; Mohapatra, P.K.D.; Chatterjee, J.; Dhara, S. In vitro ALP and osteocalcin gene expression analysis and in vivo biocompatibility of N-methylene phosphonic chitosan nanofibers for bone regeneration. J. Biomed. Nanotechnol. 2013, 9, 870–879. [Google Scholar] [CrossRef] [PubMed]
- Romero, R.; Chubb, L.; Travers, J.K.; Gonzales, T.R.; Ehrhart, N.P.; Kipper, M.J. Coating cortical bone allografts with periosteum-mimetic scaffolds made of chitosan, trimethyl chitosan, and heparin. Carbohydr. Polym. 2015, 122, 144–151. [Google Scholar] [CrossRef] [PubMed]
- Shrestha, S.; Diogenes, A.; Kishen, A. Temporal-controlled release of bovine serum albumin from chitosan nanoparticles: Effect on the regulation of alkaline phosphatase activity in stem cells from apical papilla. J. Endod. 2014, 40, 1349–1354. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.; Li, Y.; Sun, X.; Kishen, A.; Deng, X.; Yang, X.; Wang, H.; Cong, C.; Wang, Y.; Wu, M. Biomimetic remineralization of demineralized enamel with nano-complexes of phosphorylated chitosan and amorphous calcium phosphate. J. Mater. Sci. Mater. Med. 2014, 25, 2619–2628. [Google Scholar] [CrossRef] [PubMed]
- DenBesten, P.; Li, W. Chronic fluoride toxicity: Dental fluorosis. Monogr Oral Sci. 2011, 22, 81–96. [Google Scholar] [PubMed]
- Shrestha, A.; Zhilong, S.; Gee, N.K.; Kishen, A. Nanoparticulates for antibiofilm treatment and effect of aging on its antibacterial activity. J. Endod. 2010, 36, 1030–1035. [Google Scholar] [CrossRef] [PubMed]
- Shrestha, A.; Kishen, A. The effect of tissue inhibitors on the antibacterial activity of chitosan nanoparticles and photodynamic therapy. J. Endod. 2012, 38, 1275–1278. [Google Scholar] [CrossRef] [PubMed]
- Rabea, E.I.; Badawy, M.E.-T.; Stevens, C.V.; Smagghe, G.; Steurbaut, W. Chitosan as antimicrobial agent: Applications and mode of action. Biomacromolecules 2003, 4, 1457–1465. [Google Scholar] [CrossRef] [PubMed]
- Upadya, M.; Shrestha, A.; Kishen, A. Role of efflux pump inhibitors on the antibiofilm efficacy of calcium hydroxide, chitosan nanoparticles, and light-activated disinfection. J. Endod. 2011, 37, 1422–1426. [Google Scholar] [CrossRef] [PubMed]
- Kishen, A.; Shi, Z.; Shrestha, A.; Neoh, K.G. An investigation on the antibacterial and antibiofilm efficacy of cationic nanoparticulates for root canal disinfection. J. Endod. 2008, 34, 1515–1520. [Google Scholar] [CrossRef] [PubMed]
- Del Carpio-Perochena, A.; Kishen, A.; Shrestha, A.; Bramante, C.M. Antibacterial properties associated with chitosan nanoparticle treatment on root dentin and 2 types of endodontic sealers. J. Endod. 2015, 41, 1353–1358. [Google Scholar] [CrossRef] [PubMed]
- Del Carpio-Perochena, A.; Bramante, C.M.; Duarte, M.A.H.; de Moura, M.R.; Aouada, F.A.; Kishen, A. Chelating and antibacterial properties of chitosan nanoparticles on dentin. Restor. Dent. Endod. 2015, 40, 195–201. [Google Scholar] [CrossRef] [PubMed]
- DaSilva, L.; Finer, Y.; Friedman, S.; Basrani, B.; Kishen, A. Biofilm formation within the interface of bovine root dentin treated with conjugated chitosan and sealer containing chitosan nanoparticles. J. Endod. 2013, 39, 249–253. [Google Scholar] [CrossRef] [PubMed]
- Hamilton, M.F.; Otte, A.D.; Gregory, R.L.; Pinal, R.; Ferreira-Zandoná, A.; Bottino, M.C. Physicomechanical and antibacterial properties of experimental resin-based dental sealants modified with nylon-6 and chitosan nanofibers. J. Biomed. Mater. Res. B Appl. Biomater. 2014, 103, 1560–1568. [Google Scholar] [CrossRef] [PubMed]
- Liu, H.; Chen, B.; Mao, Z.; Gao, C. Chitosan nanoparticles for loading of toothpaste actives and adhesion on tooth analogs. J. Appl. Polym. Sci. 2007, 106, 4248–4256. [Google Scholar] [CrossRef]
- De Paz, L.E.C.; Resin, A.; Howard, K.A.; Sutherland, D.S.; Wejse, P.L. Antimicrobial effect of chitosan nanoparticles on streptococcus mutans biofilms. Appl. Environ. Microbiol. 2011, 77, 3892–3895. [Google Scholar] [CrossRef] [PubMed]
- Loesche, W.J. Role of streptococcus mutans in human dental decay. Microbiol. Rev. 1986, 50, 353–380. [Google Scholar] [PubMed]
- Moye, Z.D.; Zeng, L.; Burne, R.A. Fueling the caries process: Carbohydrate metabolism and gene regulation by streptococcus mutans. J. Oral Microbiol. 2014, 6. [Google Scholar] [CrossRef] [PubMed]
- Razavi, S.; Zarkesh-Esfahani, H.; Morshed, M.; Vaezifar, S.; Karbasi, S.; Golozar, M.A. Nanobiocomposite of poly(lactide-co-glycolide)/chitosan electrospun scaffold can promote proliferation and transdifferentiation of schwann-like cells from human adipose-derived stem cells. J. Biomed. Mater. Res. A 2015, 103, 2628–2634. [Google Scholar] [CrossRef] [PubMed]
- Lin, H.-Y.; Chen, S.-H.; Chang, S.-H.; Huang, S.-T. Tri-layered chitosan scaffold as a potential skin substitute. J. Biomater. Sci. Polym. Ed. 2015, 26, 855–867. [Google Scholar] [CrossRef] [PubMed]
- Kossovich, L.; Salkovskiy, Y.; Kirillova, I. Electrospun chitosan nanofiber materials as burn dressing. In Proceedings of the 6th World Congress of Biomechanics (WCB 2010), Singapore, 1–6 August 2010; Springer: Heidelberg, Germany; pp. 1212–1214.
- Noh, H.K.; Lee, S.W.; Kim, J.-M.; Oh, J.-E.; Kim, K.-H.; Chung, C.-P.; Choi, S.-C.; Park, W.H.; Min, B.-M. Electrospinning of chitin nanofibers: Degradation behavior and cellular response to normal human keratinocytes and fibroblasts. Biomaterials 2006, 27, 3934–3944. [Google Scholar] [CrossRef] [PubMed]
- Mansur, A.A.; de Carvalho, S.M.; Mansur, H.S. Bioengineered quantum dot/chitosan-tripeptide nanoconjugates for targeting the receptors of cancer cells. Int. J. Biol. Macromol. 2015, 82, 780–789. [Google Scholar] [CrossRef] [PubMed]
- Jayakumar, R.; Menon, D.; Manzoor, K.; Nair, S.; Tamura, H. Biomedical applications of chitin and chitosan based nanomaterials—A short review. Carbohydr. Polym. 2010, 82, 227–232. [Google Scholar] [CrossRef]
- Ramasamy, T.; Tran, T.H.; Cho, H.J.; Kim, J.H.; Kim, Y.I.; Jeon, J.Y.; Choi, H.-G.; Yong, C.S.; Kim, J.O. Chitosan-based polyelectrolyte complexes as potential nanoparticulate carriers: Physicochemical and biological characterization. Pharm. Res. 2014, 31, 1302–1314. [Google Scholar] [CrossRef] [PubMed]
- David, K.I.; Jaidev, L.R.; Sethuraman, S.; Krishnan, U.M. Dual drug loaded chitosan nanoparticles-sugar-coated arsenal against pancreatic cancer. Colloids Surf. B Biointerfaces 2015, 135, 689–698. [Google Scholar] [CrossRef] [PubMed]
- Nogueira, D.R.; Tavano, L.; Mitjans, M.; Pérez, L.; Infante, M.R.; Vinardell, M.P. In vitro antitumor activity of methotrexate via pH-sensitive chitosan nanoparticles. Biomaterials 2013, 34, 2758–2772. [Google Scholar] [CrossRef] [PubMed]
- Machiels, J.-P.H.; Haddad, R.I.; Fayette, J.; Licitra, L.F.; Tahara, M.; Vermorken, J.B.; Clement, P.M.; Gauler, T.; Cupissol, D.; Grau, J.J. Afatinib versus methotrexate as second-line treatment in patients with recurrent or metastatic squamous-cell carcinoma of the head and neck progressing on or after platinum-based therapy (lux-head & neck 1): An open-label, randomised phase 3 trial. Lancet Oncol. 2015, 16, 583–594. [Google Scholar] [PubMed]
- Rao, W.; Wang, H.; Han, J.; Zhao, S.; Dumbleton, J.; Agarwal, P.; Zhang, W.; Zhao, G.; Yu, J.; Zynger, D.L. Chitosan-decorated doxorubicin-encapsulated nanoparticle targets and eliminates tumor reinitiating cancer stem-like cells. ACS Nano 2015, 9, 5725–5740. [Google Scholar] [CrossRef] [PubMed]
- Alshaer, W.; Hillaireau, H.; Vergnaud, J.; Ismail, S.; Fattal, E. Functionalizing liposomes with anti-CD44 aptamer for selective targeting of cancer cells. Bioconj. Chem. 2015, 26, 1307–1313. [Google Scholar] [CrossRef] [PubMed]
- Biddle, A.; Liang, X.; Gammon, L.; Fazil, B.; Harper, L.J.; Emich, H.; Costea, D.E.; Mackenzie, I.C. Cancer stem cells in squamous cell carcinoma switch between two distinct phenotypes that are preferentially migratory or proliferative. Cancer Res. 2011, 71, 5317–5326. [Google Scholar] [CrossRef] [PubMed]
- Harper, L.J.; Costea, D.E.; Gammon, L.; Fazil, B.; Biddle, A.; Mackenzie, I.C. Normal and malignant epithelial cells with stem-like properties have an extended G2 cell cycle phase that is associated with apoptotic resistance. BMC Cancer 2010, 10, 166. [Google Scholar] [CrossRef] [PubMed]
- Jeevitha, D.; Amarnath, K. Chitosan/pla nanoparticles as a novel carrier for the delivery of anthraquinone: Synthesis, characterization and in vitro cytotoxicity evaluation. Colloids Surf. B Biointerfaces 2013, 101, 126–134. [Google Scholar] [CrossRef] [PubMed]
- Bhattarai, G.; Lee, Y.; Lee, M.; Yi, H. Gene delivery of c-myb increases bone formation surrounding oral implants. J. Dent. Res. 2013, 92, 840–845. [Google Scholar] [CrossRef] [PubMed]
- Bhattarai, G.; Lee, Y.H.; Lee, N.H.; Park, I.S.; Lee, M.H.; Yi, H.K. PPARγ delivered by Ch-GNPs onto titanium surfaces inhibits implant-induced inflammation and induces bone mineralization of MC-3T3E1 osteoblast-like cells. Clin. Oral Implant. Res. 2013, 24, 1101–1109. [Google Scholar] [CrossRef] [PubMed]
- Bhattarai, G.; Lee, Y.H.; Yi, H.K. Peroxisome proliferator activated receptor gamma loaded dental implant improves osteogenesis of rat mandible. J. Biomed. Mater. Res. B Appl. Biomater. 2015, 103, 587–595. [Google Scholar] [CrossRef] [PubMed]
- Hassumi, M.Y.; Silva-Filho, V.J.; Campos-Júnior, J.C.; Vieira, S.M.; Cunha, F.Q.; Alves, P.M.; Alves, J.B.; Kawai, T.; Gonçalves, R.B.; Napimoga, M.H. Ppar-γ agonist rosiglitazone prevents inflammatory periodontal bone loss by inhibiting osteoclastogenesis. Int. Immunopharmacol. 2009, 9, 1150–1158. [Google Scholar] [CrossRef] [PubMed]
- Di Paola, R.; Mazzon, E.; Maiere, D.; Zito, D.; Britti, D.; De Majo, M.; Genovese, T.; Cuzzocrea, S. Rosiglitazone reduces the evolution of experimental periodontitis in the rat. J. Dent. Res. 2006, 85, 156–161. [Google Scholar] [CrossRef] [PubMed]
- Lee, Y.-H.; Kang, Y.-M.; Heo, M.-J.; Kim, G.-E.; Bhattarai, G.; Lee, N.-H.; Yu, M.-K.; Yi, H.-K. The survival role of peroxisome proliferator-activated receptor gamma induces odontoblast differentiation against oxidative stress in human dental pulp cells. J. Endod. 2013, 39, 236–241. [Google Scholar] [CrossRef] [PubMed]
- Yu, M.-K.; Lee, J.-C.; Kim, J.-H.; Lee, Y.-H.; Jeon, J.-G.; Jhee, E.-C.; Yi, H.-K. Anti-inflammatory effect of peroxisome proliferator activated receptor gamma on human dental pulp cells. J. Endod. 2009, 35, 524–528. [Google Scholar] [CrossRef] [PubMed]
- Kim, T.-G.; Lee, Y.-H.; Lee, N.-H.; Bhattarai, G.; Lee, I.-K.; Yun, B.-S.; Yi, H.-K. The antioxidant property of pachymic acid improves bone disturbance against AH plus–induced inflammation in MC-3T3E1 cells. J. Endod. 2013, 39, 461–466. [Google Scholar] [CrossRef] [PubMed]
- Jing, X.; Mi, H.-Y.; Peng, J.; Peng, X.-F.; Turng, L.-S. Electrospun aligned poly(propylene carbonate) microfibers with chitosan nanofibers as tissue engineering scaffolds. Carbohydr. Polym. 2015, 117, 941–949. [Google Scholar] [CrossRef] [PubMed]
- Cheng, Y.; Ramos, D.; Lee, P.; Liang, D.; Yu, X.; Kumbar, S.G. Collagen functionalized bioactive nanofiber matrices for osteogenic differentiation of mesenchymal stem cells: Bone tissue engineering. J. Biomed. Nanotechnol. 2014, 10, 287–298. [Google Scholar] [CrossRef] [PubMed]
- Jing, X.; Mi, H.-Y.; Wang, X.-C.; Peng, X.-F.; Turng, L.-S. Shish-kebab-structured poly(ε-caprolactone) nanofibers hierarchically decorated with chitosan-poly(ε-caprolactone) copolymers for bone tissue engineering. ACS Appl. Mater. Interfaces 2015, 7, 6955–6965. [Google Scholar] [CrossRef] [PubMed]
- Hou, Y.; Hu, J.; Park, H.; Lee, M. Chitosan-based nanoparticles as a sustained protein release carrier for tissue engineering applications. J. Biomed. Mater. Res. A 2012, 100, 939–947. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Venugopal, J.R.; El-Turki, A.; Ramakrishna, S.; Su, B.; Lim, C.T. Electrospun biomimetic nanocomposite nanofibers of hydroxyapatite/chitosan for bone tissue engineering. Biomaterials 2008, 29, 4314–4322. [Google Scholar] [CrossRef] [PubMed]
- Liu, H.; Peng, H.; Wu, Y.; Zhang, C.; Cai, Y.; Xu, G.; Li, Q.; Chen, X.; Ji, J.; Zhang, Y. The promotion of bone regeneration by nanofibrous hydroxyapatite/chitosan scaffolds by effects on integrin-bmp/smad signaling pathway in bmscs. Biomaterials 2013, 34, 4404–4417. [Google Scholar] [CrossRef] [PubMed]
- Kumar, P.S.; Srinivasan, S.; Lakshmanan, V.-K.; Tamura, H.; Nair, S.; Jayakumar, R. Synthesis, characterization and cytocompatibility studies of α-chitin hydrogel/nano hydroxyapatite composite scaffolds. Int. J. Biol. Macromol. 2011, 49, 20–31. [Google Scholar] [CrossRef] [PubMed]
- Yang, D.; Jin, Y.; Zhou, Y.; Ma, G.; Chen, X.; Lu, F.; Nie, J. In situ mineralization of hydroxyapatite on electrospun chitosan-based nanofibrous scaffolds. Macromol. Biosci. 2008, 8, 239–246. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Reddy, V.J.; Wong, S.Y.; Li, X.; Su, B.; Ramakrishna, S.; Lim, C.T. Enhanced biomineralization in osteoblasts on a novel electrospun biocomposite nanofibrous substrate of hydroxyapatite/collagen/chitosan. Tissue Eng. A 2010, 16, 1949–1960. [Google Scholar] [CrossRef] [PubMed]
- Zhang, S.; Prabhakaran, M.P.; Qin, X.; Ramakrishna, S. Biocomposite scaffolds for bone regeneration: Role of chitosan and hydroxyapatite within poly-3-hydroxybutyrate-co-3-hydroxyvalerate on mechanical properties and in vitro evaluation. J. Mech. Behav. Biomed. Mater. 2015, 51, 88–98. [Google Scholar] [CrossRef] [PubMed]
- Zhong, Z.; Qin, J.; Ma, J. Cellulose acetate/hydroxyapatite/chitosan coatings for improved corrosion resistance and bioactivity. Mater. Sci. Eng. C 2015, 49, 251–255. [Google Scholar] [CrossRef] [PubMed]
- Lin, C.-C.; Fu, S.-J.; Lin, Y.-C.; Yang, I.-K.; Gu, Y. Chitosan-coated electrospun PLA fibers for rapid mineralization of calcium phosphate. Int. J. Biol. Macromol. 2014, 68, 39–47. [Google Scholar] [CrossRef] [PubMed]
- Subramanian, A.; Vu, D.; Larsen, G.F.; Lin, H.-Y. Preparation and evaluation of the electrospun chitosan/peo fibers for potential applications in cartilage tissue engineering. J. Biomater. Sci. Polym. Ed. 2005, 16, 861–873. [Google Scholar] [CrossRef] [PubMed]
- Tonglairoum, P.; Ngawhirunpat, T.; Rojanarata, T.; Panomsuk, S.; Kaomongkolgit, R.; Opanasopit, P. Fabrication of mucoadhesive chitosan coated polyvinylpyrrolidone/cyclodextrin/clotrimazole sandwich patches for oral candidiasis. Carbohydr. Polym. 2015, 132, 173–179. [Google Scholar] [CrossRef] [PubMed]
- Jiang, W.; Li, L.; Zhang, D.; Huang, S.; Jing, Z.; Wu, Y.; Zhao, Z.; Zhao, L.; Zhou, S. Incorporation of aligned pcl–peg nanofibers into porous chitosan scaffolds improved the orientation of collagen fibers in regenerated periodontium. Acta Biomater. 2015, 25, 240–252. [Google Scholar] [CrossRef] [PubMed]
- Bertoncelj, V.; Pelipenko, J.; Kristl, J.; Jeras, M.; Cukjati, M.; Kocbek, P. Development and bioevaluation of nanofibers with blood-derived growth factors for dermal wound healing. Eur. J. Pharm. Biopharm. 2014, 88, 64–74. [Google Scholar] [CrossRef] [PubMed]
- Lou, T.; Leung, M.; Wang, X.; Chang, J.Y.F.; Tsao, C.T.; Sham, J.G.C.; Edmondson, D.; Zhang, M. Bi-layer scaffold of chitosan/PCL-nanofibrous mat and PLLA-microporous disc for skin tissue engineering. J. Biomed. Nanotechnol. 2014, 10, 1105–1113. [Google Scholar] [CrossRef] [PubMed]
- Sarhan, W.A.; Azzazy, H.M. High concentration honey chitosan electrospun nanofibers: Biocompatibility and antibacterial effects. Carbohydr. Polym. 2015, 122, 135–143. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.-P.; Chang, G.-Y.; Chen, J.-K. Electrospun collagen/chitosan nanofibrous membrane as wound dressing. Colloids Surf. A Physicochem. Eng. Asp. 2008, 313, 183–188. [Google Scholar] [CrossRef]
- Kumar, P.S.; Raj, N.M.; Praveen, G.; Chennazhi, K.P.; Nair, S.V.; Jayakumar, R. In vitro and in vivo evaluation of microporous chitosan hydrogel/nanofibrin composite bandage for skin tissue regeneration. Tissue Eng. A 2012, 19, 380–392. [Google Scholar] [CrossRef] [PubMed]
- Zhao, R.; Li, X.; Sun, B.; Zhang, Y.; Zhang, D.; Tang, Z.; Chen, X.; Wang, C. Electrospun chitosan/sericin composite nanofibers with antibacterial property as potential wound dressings. Int. J. Biol. Macromol. 2014, 68, 92–97. [Google Scholar] [CrossRef] [PubMed]
- Choi, J.S.; Han, S.H.; Hyun, C.; Yoo, H.S. Buccal adhesive nanofibers containing human growth hormone for oral mucositis. J. Biomed. Mater. Res. B Appl. Biomater. 2015. [Google Scholar] [CrossRef] [PubMed]
- Antunes, B.; Moreira, A.; Gaspar, V.; Correia, I. Chitosan/arginine-chitosan polymer blends for assembly of nanofibrous membranes for wound regeneration. Carbohydr. Polym. 2015, 130, 104–112. [Google Scholar] [CrossRef] [PubMed]
- Tan, L.; Hu, J.; Huang, H.; Han, J.; Hu, H. Study of multi-functional electrospun composite nanofibrous mats for smart wound healing. Int. J. Biol. Macromol. 2015, 79, 469–476. [Google Scholar] [CrossRef] [PubMed]
- Xu, F.; Weng, B.; Gilkerson, R.; Materon, L.A.; Lozano, K. Development of tannic acid/chitosan/pullulan composite nanofibers from aqueous solution for potential applications as wound dressing. Carbohydr. Polym. 2015, 115, 16–24. [Google Scholar] [CrossRef] [PubMed]
- Shrestha, A.; Hamblin, M.R.; Kishen, A. Photoactivated rose bengal functionalized chitosan nanoparticles produce antibacterial/biofilm activity and stabilize dentin-collagen. Nanomed. Nanotechnol. Biol. Med. 2014, 10, 491–501. [Google Scholar] [CrossRef] [PubMed]
- Tonglairoum, P.; Ngawhirunpat, T.; Rojanarata, T.; Kaomongkolgit, R.; Opanasopit, P. Fabrication of a novel scaffold of clotrimazole-microemulsion-containing nanofibers using an electrospinning process for oral candidiasis applications. Colloids Surf. B Biointerfaces 2015, 126, 18–25. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Cheng, F.; Gao, J.; Wang, L. Antibacterial wound dressing from chitosan/polyethylene oxide nanofibers mats embedded with silver nanoparticles. J. Biomater. Appl. 2015, 29, 1086–1095. [Google Scholar] [CrossRef] [PubMed]
- Croisier, F.; Sibret, P.; Dupont-Gillain, C.C.; Genet, M.J.; Detrembleur, C.; Jérôme, C. Chitosan-coated electrospun nanofibers with antibacterial activity. J. Mater. Chem. B 2015, 3, 3508–3517. [Google Scholar] [CrossRef]
- Gholipour-Kanani, A.; Bahrami, S.H.; Joghataie, M.T.; Samadikuchaksaraei, A.; Ahmadi-Taftie, H.; Rabbani, S.; Kororian, A.; Erfani, E. Tissue engineered poly(caprolactone)-chitosan-poly(vinyl alcohol) nanofibrous scaffolds for burn and cutting wound healing. IET Nanobiotechnol. 2014, 8, 123–131. [Google Scholar] [CrossRef] [PubMed]
- Koosha, M.; Mirzadeh, H. Electrospinning, mechanical properties, and cell behavior study of chitosan/PVA nanofibers. J. Biomed. Mater. Res. A 2015, 103, 3081–30963. [Google Scholar] [CrossRef] [PubMed]
- Zhou, Y.; Yang, D.; Chen, X.; Xu, Q.; Lu, F.; Nie, J. Electrospun water-soluble carboxyethyl chitosan/poly(vinyl alcohol) nanofibrous membrane as potential wound dressing for skin regeneration. Biomacromolecules 2007, 9, 349–354. [Google Scholar] [CrossRef] [PubMed]
- Chen, Z.; Cao, S.; Wang, H.; Li, Y.; Kishen, A.; Deng, X.; Yang, X.; Wang, Y.; Cong, C.; Wang, H. Biomimetic remineralization of demineralized dentine using scaffold of CMC/ACP nanocomplexes in an in vitro tooth model of deep caries. PLoS ONE 2015, 10, e0116553. [Google Scholar] [CrossRef] [PubMed]
- Targino, A.G.R.; Flores, M.A.P.; dos Santos Junior, V.E.; Bezerra, F.D.G.B.; de Freire, L.H.; Galembeck, A.; Rosenblatt, A. An innovative approach to treating dental decay in children. A new anti-caries agent. J. Mater. Sci. Mater. Med. 2014, 25, 2041–2047. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Liu, Y.; Liao, N.; Cui, F.; Park, M.; Kim, H.-Y. Fabrication and durable antibacterial properties of electrospun chitosan nanofibers with silver nanoparticles. Int. J. Biol. Macromol. 2015, 79, 638–643. [Google Scholar] [CrossRef] [PubMed]
- Rhim, J.-W.; Hong, S.-I.; Park, H.-M.; Ng, P.K. Preparation and characterization of chitosan-based nanocomposite films with antimicrobial activity. J. Agric. Food Chem. 2006, 54, 5814–5822. [Google Scholar] [CrossRef] [PubMed]
- Sanpui, P.; Murugadoss, A.; Prasad, P.D.; Ghosh, S.S.; Chattopadhyay, A. The antibacterial properties of a novel chitosan-Ag-nanoparticle composite. Int. J. Food Microbiol. 2008, 124, 142–146. [Google Scholar] [CrossRef] [PubMed]
- Cao, X.; Cheng, C.; Ma, Y.; Zhao, C. Preparation of silver nanoparticles with antimicrobial activities and the researches of their biocompatibilities. J. Mater. Sci. Mater. Med. 2010, 21, 2861–2868. [Google Scholar] [CrossRef] [PubMed]
- Shrestha, A.; Kishen, A. Antibiofilm efficacy of photosensitizer-functionalized bioactive nanoparticles on multispecies biofilm. J. Endod. 2014, 40, 1604–1610. [Google Scholar] [CrossRef] [PubMed]
- Shrestha, A.; Kishen, A. Polycationic chitosan-conjugated photosensitizer for antibacterial photodynamic therapy. Photochem. Photobiol. 2012, 88, 577–583. [Google Scholar] [CrossRef] [PubMed]
- Saatchi, M.; Shokraneh, A.; Navaei, H.; Maracy, M.R.; Shojaei, H. Antibacterial effect of calcium hydroxide combined with chlorhexidine on Enterococcus faecalis: A systematic review and meta-analysis. J. Appl. Oral Sci. 2014, 22, 356–365. [Google Scholar] [CrossRef] [PubMed]
- Greabu, M.; Virlan, M.J.R.; Calenic, B.; Zaharia, C. Silk fibroin and potential uses in regenerative dentistry—A systematic review. Stomatol. Educ. J. 2015, 1, 108–115. [Google Scholar]
- Wenk, E.; Merkle, H.P.; Meinel, L. Silk fibroin as a vehicle for drug delivery applications. J. Control. Release 2011, 150, 128–141. [Google Scholar] [CrossRef] [PubMed]
- Li, C.; Vepari, C.; Jin, H.-J.; Kim, H.J.; Kaplan, D.L. Electrospun silk-BMP-2 scaffolds for bone tissue engineering. Biomaterials 2006, 27, 3115–3124. [Google Scholar] [CrossRef] [PubMed]
- Kim, K.-H.; Jeong, L.; Park, H.-N.; Shin, S.-Y.; Park, W.-H.; Lee, S.-C.; Kim, T.-I.; Park, Y.-J.; Seol, Y.-J.; Lee, Y.-M. Biological efficacy of silk fibroin nanofiber membranes for guided bone regeneration. J. Biotechnol. 2005, 120, 327–339. [Google Scholar] [CrossRef] [PubMed]
- Liu, L.; Zhang, X.; Liu, X.; Liu, J.; Lu, G.; Kaplan, D.L.; Zhu, H.; Lu, Q. Biomineralization of stable and monodisperse vaterite microspheres using silk nanoparticles. ACS Appl. Mater. Interfaces 2015, 7, 1735–1745. [Google Scholar] [CrossRef] [PubMed]
- Lai, G.-J.; Shalumon, K.; Chen, S.-H.; Chen, J.-P. Composite chitosan/silk fibroin nanofibers for modulation of osteogenic differentiation and proliferation of human mesenchymal stem cells. Carbohydr. Polym. 2014, 111, 288–297. [Google Scholar] [CrossRef] [PubMed]
- Lai, G.-J.; Shalumon, K.; Chen, J.-P. Response of human mesenchymal stem cells to intrafibrillar nanohydroxyapatite content and extrafibrillar nanohydroxyapatite in biomimetic chitosan/silk fibroin/nanohydroxyapatite nanofibrous membrane scaffolds. Int. J. Nanomed. 2015, 10, 567–584. [Google Scholar]
- Shalumon, K.; Lai, G.-J.; Chen, C.-H.; Chen, J.-P. Modulation of bone-specific tissue regeneration by incorporating bone morphogenetic protein and controlling the shell thickness of silk fibroin/chitosan/nanohydroxyapatite core-shell nanofibrous membranes. ACS Appl. Mater. Interfaces 2015, 7, 21170–21181. [Google Scholar] [CrossRef] [PubMed]
- Yoo, C.R.; Yeo, I.-S.; Park, K.E.; Park, J.H.; Lee, S.J.; Park, W.H.; Min, B.-M. Effect of chitin/silk fibroin nanofibrous bicomponent structures on interaction with human epidermal keratinocytes. Int. J. Biol. Macromol. 2008, 42, 324–334. [Google Scholar] [CrossRef] [PubMed]
- Park, K.E.; Jung, S.Y.; Lee, S.J.; Min, B.-M.; Park, W.H. Biomimetic nanofibrous scaffolds: Preparation and characterization of chitin/silk fibroin blend nanofibers. Int. J. Biol. Macromol. 2006, 38, 165–173. [Google Scholar] [CrossRef] [PubMed]
- Cai, Z.-X.; Mo, X.-M.; Zhang, K.-H.; Fan, L.-P.; Yin, A.-L.; He, C.-L.; Wang, H.-S. Fabrication of chitosan/silk fibroin composite nanofibers for wound-dressing applications. Int. J. Mol. Sci. 2010, 11, 3529–3539. [Google Scholar] [CrossRef] [PubMed]
- Dunne, L.W.; Iyyanki, T.; Hubenak, J.; Mathur, A.B. Characterization of dielectrophoresis-aligned nanofibrous silk fibroin-chitosan scaffold and its interactions with endothelial cells for tissue engineering applications. Acta Biomater. 2014, 10, 3630–3640. [Google Scholar] [CrossRef] [PubMed]
- Numata, K.; Mieszawska-Czajkowska, A.J.; Kvenvold, L.A.; Kaplan, D.L. Silk-based nanocomplexes with tumor-homing peptides for tumor-specific gene delivery. Macromol. Biosci. 2012, 12, 75–82. [Google Scholar] [CrossRef] [PubMed]
- Qu, J.; Liu, Y.; Yu, Y.; Li, J.; Luo, J.; Li, M. Silk fibroin nanoparticles prepared by electrospray as controlled release carriers of cisplatin. Mater. Sci. Eng. C 2014, 44, 166–174. [Google Scholar] [CrossRef] [PubMed]
- Marin, E.; Briceño, M.I.; Caballero-George, C. Critical evaluation of biodegradable polymers used in nanodrugs. Int. J. Nanomed. 2013, 8, 3071–3090. [Google Scholar]
- Lü, J.-M.; Wang, X.; Marin-Muller, C.; Wang, H.; Lin, P.H.; Yao, Q.; Chen, C. Current advances in research and clinical applications of PLGA-based nanotechnology. Expert Rev. Mol. Diagn. 2009, 9, 325–341. [Google Scholar] [CrossRef] [PubMed]
- Liu, H.; Slamovich, E.B.; Webster, T.J. Less harmful acidic degradation of poly(lactic-co-glycolic acid) bone tissue engineering scaffolds through titania nanoparticle addition. Int. J. Nanomed. 2006, 1, 541–545. [Google Scholar] [CrossRef]
- Kashi, T.S.J.; Eskandarion, S.; Esfandyari-Manesh, M.; Marashi, S.M.A.; Samadi, N.; Fatemi, S.M.; Atyabi, F.; Eshraghi, S.; Dinarvand, R. Improved drug loading and antibacterial activity of minocycline-loaded PLGA nanoparticles prepared by solid/oil/water ion pairing method. Int. J. Nanomed. 2012, 7, 221–234. [Google Scholar]
- Pagonis, T.C.; Chen, J.; Fontana, C.R.; Devalapally, H.; Ruggiero, K.; Song, X.; Foschi, F.; Dunham, J.; Skobe, Z.; Yamazaki, H. Nanoparticle-based endodontic antimicrobial photodynamic therapy. J. Endod. 2010, 36, 322–328. [Google Scholar] [CrossRef] [PubMed]
- Chereddy, K.K.; Coco, R.; Memvanga, P.B.; Ucakar, B.; des Rieux, A.; Vandermeulen, G.; Préat, V. Combined effect of PLGA and curcumin on wound healing activity. J. Control. Release 2013, 171, 208–215. [Google Scholar] [CrossRef] [PubMed]
- Chereddy, K.K.; Her, C.-H.; Comune, M.; Moia, C.; Lopes, A.; Porporato, P.E.; Vanacker, J.; Lam, M.C.; Steinstraesser, L.; Sonveaux, P. PLGA nanoparticles loaded with host defense peptide LL37 promote wound healing. J. Control. Release 2014, 194, 138–147. [Google Scholar] [CrossRef] [PubMed]
- Chereddy, K.K.; Lopes, A.; Koussoroplis, S.; Payen, V.; Moia, C.; Zhu, H.; Sonveaux, P.; Carmeliet, P.; des Rieux, A.; Vandermeulen, G. Combined effects of PLGA and vascular endothelial growth factor promote the healing of non-diabetic and diabetic wounds. Nanomed. Nanotechnol. Biol. Med. 2015, 11, 1975–1984. [Google Scholar] [CrossRef] [PubMed]
- Gentile, P.; Nandagiri, V.K.; Pabari, R.; Daly, J.; Tonda-Turo, C.; Ciardelli, G.; Ramtoola, Z. Influence of parathyroid hormone-loaded PLGA nanoparticles in porous scaffolds for bone regeneration. Int. J. Mol. Sci. 2015, 16, 20492–20510. [Google Scholar] [CrossRef] [PubMed]
- Park, J.S.; Yang, H.N.; Woo, D.G.; Jeon, S.Y.; Do, H.-J.; Lim, H.-Y.; Kim, J.-H.; Park, K.-H. Chondrogenesis of human mesenchymal stem cells mediated by the combination of sox trio SOX5, 6, and 9 genes complexed with PEI-modified PLGA nanoparticles. Biomaterials 2011, 32, 3679–3688. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.-H.; Park, J.S.; Yang, H.N.; Woo, D.G.; Jeon, S.Y.; Do, H.-J.; Lim, H.-Y.; Kim, J.M.; Park, K.-H. The use of biodegradable PLGA nanoparticles to mediate SOX9 gene delivery in human mesenchymal stem cells (hMSCs) and induce chondrogenesis. Biomaterials 2011, 32, 268–278. [Google Scholar] [CrossRef] [PubMed]
- Jeon, S.Y.; Park, J.S.; Yang, H.N.; Woo, D.G.; Park, K.-H. Aggrecan-and comp-loaded poly-(lactic-co-glycolic acid) nanoparticles stimulate chondrogenic differentiation of human mesenchymal stem cells. Stem Cells Dev. 2013, 23, 305–317. [Google Scholar] [CrossRef] [PubMed]
- Jeon, S.Y.; Park, J.S.; Yang, H.N.; Lim, H.J.; Yi, S.W.; Park, H.; Park, K.-H. Co-delivery of Cbfa-1-targeting siRNA and SOX9 protein using PLGA nanoparticles to induce chondrogenesis of human mesenchymal stem cells. Biomaterials 2014, 35, 8236–8248. [Google Scholar] [CrossRef] [PubMed]
- Yang, H.N.; Park, J.S.; Woo, D.G.; Jeon, S.Y.; Do, H.-J.; Lim, H.-Y.; Kim, S.W.; Kim, J.-H.; Park, K.-H. Chondrogenesis of mesenchymal stem cells and dedifferentiated chondrocytes by transfection with SOX trio genes. Biomaterials 2011, 32, 7695–7704. [Google Scholar] [CrossRef] [PubMed]
- Wei, G.; Jin, Q.; Giannobile, W.V.; Ma, P.X. The enhancement of osteogenesis by nano-fibrous scaffolds incorporating rhBMP-7 nanospheres. Biomaterials 2007, 28, 2087–2096. [Google Scholar] [CrossRef] [PubMed]
- Pillai, R.R.; Somayaji, S.N.; Rabinovich, M.; Hudson, M.C.; Gonsalves, K.E. Nafcillin-loaded PLGA nanoparticles for treatment of osteomyelitis. Biomedical Mater. 2008, 3, 034114. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.; Liu, J.; Tao, S.; Chai, G.; Wang, J.; Hu, F.-Q.; Yuan, H. Tetracycline-grafted PLGA nanoparticles as bone-targeting drug delivery system. Int. J. Nanomed. 2015, 10, 5671–5685. [Google Scholar]
- Garrett, I.; Gutierrez, G.; Rossini, G.; Nyman, J.; McCluskey, B.; Flores, A.; Mundy, G. Locally delivered lovastatin nanoparticles enhance fracture healing in rats. J. Orthop. Res. 2007, 25, 1351–1357. [Google Scholar] [CrossRef] [PubMed]
- Chronopoulou, L.; Amalfitano, A.; Palocci, C.; Nocca, G.; Callà, C.; Arcovito, A. Dexamethasone-loaded biopolymeric nanoparticles promote gingival fibroblasts differentiation. Biotechnol. Prog. 2015, 31, 1381–1387. [Google Scholar] [CrossRef] [PubMed]
- Souza, A.; Nascimento, A.; de Vasconcelos, N.; Jerônimo, M.; Siqueira, I.; R-Santos, L.; Cintra, D.; Fuscaldi, L.; Júnior, O.P.; Titze-de-Almeida, R. Activity and in vivo tracking of amphotericin B loaded PLGA nanoparticles. Eur. J. Med. Chem. 2015, 95, 267–276. [Google Scholar] [CrossRef] [PubMed]
- Radcliffe, C.; Potouridou, L.; Qureshi, R.; Habahbeh, N.; Qualtrough, A.; Worthington, H.; Drucker, D. Antimicrobial activity of varying concentrations of sodium hypochlorite on the endodontic microorganisms actinomyces israelii, A. Naeslundii, Candida albicans and Enterococcus faecalis. Int. Endod. J. 2004, 37, 438–446. [Google Scholar] [CrossRef] [PubMed]
- Foschi, F.; Cavrini, F.; Montebugnoli, L.; Stashenko, P.; Sambri, V.; Prati, C. Detection of bacteria in endodontic samples by polymerase chain reaction assays and association with defined clinical signs in italian patients. Oral Microbiol. Immunol. 2005, 20, 289–295. [Google Scholar] [CrossRef] [PubMed]
- Puig-Silla, M.; Dasí, F. Oxidative stress parameters in saliva and its association with periodontal disease and types of bacteria. Dis. Markers 2015, 501, 653537. [Google Scholar] [CrossRef]
- Ardila, C.M.; Alzate, J.; Guzmán, I.C. Relationship between gram negative enteric rods, aggregatibacter actinomycetemcomitans, and clinical parameters in periodontal disease. J. Indian Soc. Periodontol. 2012, 16, 65–69. [Google Scholar] [CrossRef] [PubMed]
- Zambon, J.J.; Christersson, L.A.; Slots, J. Actinobacillus actinomycetemcomitans in human periodontal disease: Prevalence in patient groups and distribution of biotypes and serotypes within families. J. Periodontol. 1983, 54, 707–711. [Google Scholar] [CrossRef] [PubMed]
- Son, J.S.; Kwon, T.-Y.; Kim, K.-H. Osteogenic evaluation of porous calcium phosphate granules with drug delivery system using nanoparticle carriers. J. Nanosci. Nanotechnol. 2015, 15, 130–133. [Google Scholar] [CrossRef] [PubMed]
- Subbiah, R.; Hwang, M.P.; Van, S.Y.; Do, S.H.; Park, H.; Lee, K.; Kim, S.H.; Yun, K.; Park, K. Osteogenic/angiogenic dual growth factor delivery microcapsules for regeneration of vascularized bone tissue. Adv. Healthc. Mater. 2015, 4, 1982–1992. [Google Scholar] [CrossRef] [PubMed]
- Uskoković, V.; Hoover, C.; Vukomanović, M.; Uskoković, D.P.; Desai, T.A. Osteogenic and antimicrobial nanoparticulate calcium phosphate and poly-(d,l-lactide-co-glycolide) powders for the treatment of osteomyelitis. Mater. Sci. Eng. C 2013, 33, 3362–3373. [Google Scholar] [CrossRef] [PubMed]
- Jamshidi-Adegani, F.; Langroudi, L.; Ardeshirylajimi, A.; Dinarvand, P.; Dodel, M.; Doostmohammadi, A.; Rahimian, A.; Zohrabi, P.; Seyedjafari, E.; Soleimani, M. Coating of electrospun poly(lactic-co-glycolic acid) nanofibers with willemite bioceramic: Improvement of bone reconstruction in rat model. Cell Biol. Int. 2014, 38, 1271–1279. [Google Scholar] [CrossRef] [PubMed]
- Ho, M.H.; Chiang, C.P.; Liu, Y.F.; Kuo, M.Y.P.; Lin, S.K.; Lai, J.Y.; Lee, B.S. Highly efficient release of lovastatin from poly(lactic-co-glycolic acid) nanoparticles enhances bone repair in rats. J. Orthop. Res. 2011, 29, 1504–1510. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.Y.; Koak, J.Y.; Heo, S.J.; Kim, S.K.; Lee, S.J.; Nam, S.Y. Osseointegration of anodized titanium implants coated with poly(lactide-co-glycolide)/basic fibroblast growth factor by electrospray. Int. J. Oral Maxillofac. Implant. 2009, 25, 315–320. [Google Scholar]
- Irmak, G.; Demirtaş, T.T.; Çetin-Altındal, D.; Çalış, M.; Gümüşderelioğlu, M. Sustained release of 17β-estradiol stimulates osteogenic differentiation of adipose tissue-derived mesenchymal stem cells on chitosan-hydroxyapatite scaffolds. Cells Tissues Organs 2014, 199, 37–50. [Google Scholar] [CrossRef] [PubMed]
- Manoukian, M.A.; Ott, S.V.; Rajadas, J.; Inayathullah, M. Polymeric nanoparticles to combat squamous cell carcinomas in patients with dystrophic epidermolysis bullosa. Recent Pat. Nanomed. 2014, 4, 15–24. [Google Scholar] [CrossRef] [PubMed]
- Acharya, S.; Sahoo, S.K. PLGA nanoparticles containing various anticancer agents and tumour delivery by epr effect. Adv. Drug Deliv. Rev. 2011, 63, 170–183. [Google Scholar] [CrossRef] [PubMed]
- Özel, T.; White, S.; Nguyen, E.; Moy, A.; Brenes, N.; Choi, B.; Betancourt, T. Enzymatically activated near infrared nanoprobes based on amphiphilic block copolymers for optical detection of cancer. Lasers Surg. Med. 2015, 47, 579–594. [Google Scholar] [CrossRef] [PubMed]
- Dong, Y.; Feng, S.-S. Poly(d,l-lactide-co-glycolide) (PLGA) nanoparticles prepared by high pressure homogenization for paclitaxel chemotherapy. Int. J. Pharm. 2007, 342, 208–214. [Google Scholar] [CrossRef] [PubMed]
- Dong, Y.; Feng, S.-S. Poly(d,l-lactide-co-glycolide)/montmorillonite nanoparticles for oral delivery of anticancer drugs. Biomaterials 2005, 26, 6068–6076. [Google Scholar] [CrossRef] [PubMed]
- Su, W.-P.; Cheng, F.-Y.; Shieh, D.-B.; Yeh, C.-S.; Su, W.-C. PLGA nanoparticles codeliver paclitaxel and Stat3 sirna to overcome cellular resistance in lung cancer cells. Int. J. Nanomed. 2012, 7, 4269–4283. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Chen, F.; Nie, J.; Yang, D. Electrospun poly(lactic acid)/chitosan core-shell structure nanofibers from homogeneous solution. Carbohydr. Polym. 2012, 90, 1445–1451. [Google Scholar] [CrossRef] [PubMed]
- Ajalloueian, F.; Tavanai, H.; Hilborn, J.; Donzel-Gargand, O.; Leifer, K.; Wickham, A.; Arpanaei, A. Emulsion electrospinning as an approach to fabricate PLGA/chitosan nanofibers for biomedical applications. BioMed Res. Int. 2014, 2014. [Google Scholar] [CrossRef] [PubMed]
- De Faria, A.F.; Perreault, F.; Shaulsky, E.; Hoover Arias Chavez, L.; Elimelech, M. Antimicrobial electrospun biopolymer nanofiber mats functionalized with graphene oxide? Silver nanocomposites. ACS Appl. Mater. Interfaces 2015, 7, 12751–12759. [Google Scholar] [CrossRef] [PubMed]
- Gentile, P.; Chiono, V.; Carmagnola, I.; Hatton, P.V. An overview of poly(lactic-co-glycolic) acid (PLGA)-based biomaterials for bone tissue engineering. Int. J. Mol. Sci. 2014, 15, 3640–3659. [Google Scholar] [CrossRef] [PubMed]
- Mehrasa, M.; Asadollahi, M.A.; Ghaedi, K.; Salehi, H.; Arpanaei, A. Electrospun aligned PLGA and PLGA/gelatin nanofibers embedded with silica nanoparticles for tissue engineering. Int. J. Biol. Macromol. 2015, 79, 687–695. [Google Scholar] [CrossRef] [PubMed]
- Hild, N.; Schneider, O.D.; Mohn, D.; Luechinger, N.A.; Koehler, F.M.; Hofmann, S.; Vetsch, J.R.; Thimm, B.W.; Müller, R.; Stark, W.J. Two-layer membranes of calcium phosphate/collagen/PLGA nanofibres: In vitro biomineralisation and osteogenic differentiation of human mesenchymal stem cells. Nanoscale 2011, 3, 401–409. [Google Scholar] [CrossRef] [PubMed]
- Yilgor, P.; Hasirci, N.; Hasirci, V. Sequential BMP-2/BMP-7 delivery from polyester nanocapsules. J. Biomed. Mater. Res. A 2010, 93, 528–536. [Google Scholar] [CrossRef] [PubMed]
- Wang, C.-Z.; Fu, Y.-C.; Jian, S.-C.; Wang, Y.-H.; Liu, P.-L.; Ho, M.-L.; Wang, C.-K. Synthesis and characterization of cationic polymeric nanoparticles as simvastatin carriers for enhancing the osteogenesis of bone marrow mesenchymal stem cells. J. Colloid Interface Sci. 2014, 432, 190–199. [Google Scholar] [CrossRef] [PubMed]
- Choi, G.H.; Lee, H.J.; Lee, S.C. Titanium-adhesive polymer nanoparticles as a surface-releasing system of dual osteogenic growth factors. Macromol. Biosci. 2014, 14, 496–507. [Google Scholar] [CrossRef] [PubMed]
- Hassan, A.H.; Hosny, K.M.; Murshid, Z.A.; Alhadlaq, A.; Alyamani, A.; Naguib, G. Depot injectable biodegradable nanoparticles loaded with recombinant human bone morphogenetic protein-2: Preparation, characterization, and in vivo evaluation. Drug Des. Dev. Ther. 2015, 9, 3599–3606. [Google Scholar] [CrossRef] [PubMed]
- Jung, Y.; Chung, Y.-I.; Kim, S.H.; Tae, G.; Kim, Y.H.; Rhie, J.W.; Kim, S.-H.; Kim, S.H. In situ chondrogenic differentiation of human adipose tissue-derived stem cells in a TGF-β 1 loaded fibrin–poly(lactide-caprolactone) nanoparticulate complex. Biomaterials 2009, 30, 4657–4664. [Google Scholar] [CrossRef] [PubMed]
- Riffault, M.; Six, J.-L.; Netter, P.; Gillet, P.; Grossin, L. PLGA-based nanoparticles: A safe and suitable delivery platform for osteoarticular pathologies. Pharm. Res. 2015, 32, 3886–3898. [Google Scholar] [CrossRef]
- Gajbhiye, V.; Palanirajan, V.K.; Tekade, R.K.; Jain, N.K. Dendrimers as therapeutic agents: A systematic review. J. Pharm. Pharmacol. 2009, 61, 989–1003. [Google Scholar] [CrossRef] [PubMed]
- Bharali, D.J.; Khalil, M.; Gurbuz, M.; Simone, T.M.; Mousa, S.A. Nanoparticles and cancer therapy: A concise review with emphasis on dendrimers. Int. J. Nanomed. 2009, 4, 1–7. [Google Scholar]
- Backlund, C.; Sergesketter, A.; Offenbacher, S.; Schoenfisch, M. Antibacterial efficacy of exogenous nitric oxide on periodontal pathogens. J. Dent. Res. 2014, 93, 1089–1094. [Google Scholar] [CrossRef] [PubMed]
- Bengazi, F.; Lang, N.P.; Canciani, E.; Viganò, P.; Velez, J.U.; Botticelli, D. Osseointegration of implants with dendrimers surface characteristics installed conventionally or with piezosurgery®. A comparative study in the dog. Clin. Oral Implant. Res. 2014, 25, 10–15. [Google Scholar] [CrossRef] [PubMed]
- Galli, C.; Piemontese, M.; Meikle, S.T.; Santin, M.; Macaluso, G.M.; Passeri, G. Biomimetic coating with phosphoserine-tethered poly(epsilon-lysine) dendrons on titanium surfaces enhances wnt and osteoblastic differentiation. Clin. Oral Implant. Res. 2014, 25, e133–e139. [Google Scholar] [CrossRef]
- Eichler, M.; Katzur, V.; Scheideler, L.; Haupt, M.; Geis-Gerstorfer, J.; Schmalz, G.; Ruhl, S.; Müller, R.; Rupp, F. The impact of dendrimer-grafted modifications to model silicon surfaces on protein adsorption and bacterial adhesion. Biomaterials 2011, 32, 9168–9179. [Google Scholar] [CrossRef] [PubMed]
- Wang, T.; Yang, S.; Wang, L.; Feng, H. Use of poly(amidoamine) dendrimer for dentinal tubule occlusion: A preliminary study. PLoS ONE 2015, 10, e0124735. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.; Yang, J.; Liang, K.; Li, J.; He, L.; Yang, X.; Peng, S.; Chen, X.; Ding, C.; Li, J. Effective dentin restorative material based on phosphate-terminated dendrimer as artificial protein. Colloids Surf. B Biointerfaces 2015, 128, 304–314. [Google Scholar] [CrossRef] [PubMed]
- Jia, R.; Lu, Y.; Yang, C.-w.; Luo, X.; Han, Y. Effect of generation 4.0 polyamidoamine dendrimer on the mineralization of demineralized dentinal tubules in vitro. Arch. Oral Biol. 2014, 59, 1085–1093. [Google Scholar] [CrossRef] [PubMed]
- Ekambaram, P.; Sathali, A.A.H.; Priyanka, K. Solid lipid nanoparticles: A review. Sci. Rev. Chem. Commun. 2012, 2, 80–102. [Google Scholar]
- Abed, N.; Couvreur, P. Nanocarriers for antibiotics: A promising solution to treat intracellular bacterial infections. Int. J. Antimicrob. Agents 2014, 43, 485–496. [Google Scholar] [CrossRef] [PubMed]
- Drulis-Kawa, Z.; Dorotkiewicz-Jach, A. Liposomes as delivery systems for antibiotics. Int. J. Pharm. 2010, 387, 187–198. [Google Scholar] [CrossRef] [PubMed]
- Jones, M.N. The surface properties of phospholipid liposome systems and their characterisation. Adv. Colloid Interface Sci. 1995, 54, 93–128. [Google Scholar] [CrossRef]
- Salem, I.I.; Flasher, D.L.; Düzgüneş, N. Liposome-encapsulated antibiotics. Methods Enzymol. 2005, 391, 261–291. [Google Scholar] [PubMed]
- Vyas, S.; Sihorkar, V.; Dubey, P. Preparation, characterization and in vitro antimicrobial activity of metronidazole bearing lectinized liposomes for intra-periodontal pocket delivery. Die Pharm. 2001, 56, 554–560. [Google Scholar]
- Robinson, A.M.; Creeth, J.E.; Jones, M.N. The specificity and affinity of immunoliposome targeting to oral bacteria. Biochim. Biophys. Acta 1998, 1369, 278–286. [Google Scholar] [CrossRef]
- Jones, M.N.; Francis, S.E.; Hutchinson, F.J.; Handley, P.S.; Lyle, I.G. Targeting and delivery of bactericide to adsorbed oral bacteria by use of proteoliposomes. Biochim. Biophys. Acta 1993, 1147, 251–261. [Google Scholar] [CrossRef]
- Mugabe, C.; Halwani, M.; Azghani, A.O.; Lafrenie, R.M.; Omri, A. Mechanism of enhanced activity of liposome-entrapped aminoglycosides against resistant strains of pseudomonas aeruginosa. Antimicrob. Agents Chemother. 2006, 50, 2016–2022. [Google Scholar] [CrossRef] [PubMed]
- Ko, A.; Yee, M.; Skupin-Mrugalska, P.; Düzgünes, N. Photodynamic therapy of porphyromonas gingivalis via liposome-encapsulated sensitizers. J. Calif. Dent. Assoc. 2013, 41, 827–830. [Google Scholar] [PubMed]
- Kawazoe, A.; Inubushi, T.; Miyauchi, M.; Ishikado, A.; Tanaka, E.; Tanne, K.; Takata, T. Orally administered liposomal lactoferrin inhibits inflammation-related bone breakdown without interrupting orthodontic tooth movement. J. Periodontol. 2013, 84, 1454–1462. [Google Scholar] [CrossRef]
- Wu, Z.; Ma, H.M.; Kukita, T.; Nakanishi, Y.; Nakanishi, H. Phosphatidylserine-containing liposomes inhibit the differentiation of osteoclasts and trabecular bone loss. J. Immunol. 2010, 184, 3191–3201. [Google Scholar] [CrossRef] [PubMed]
- Sugano, M.; Negishi, Y.; Endo-Takahashi, Y.; Hamano, N.; Usui, M.; Suzuki, R.; Maruyama, K.; Aramaki, Y.; Yamamoto, M. Gene delivery to periodontal tissue using bubble liposomes and ultrasound. J. Periodontal Res. 2014, 49, 398–404. [Google Scholar] [CrossRef] [PubMed]
- Liu, D.; Yang, P. Minocycline hydrochloride nanoliposomes inhibit the production of TNF-α in LPS-stimulated macrophages. Int. J. Nanomed. 2012, 7, 4769–4775. [Google Scholar] [CrossRef] [PubMed][Green Version]
© 2016 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons by Attribution (CC-BY) license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Virlan, M.J.R.; Miricescu, D.; Radulescu, R.; Sabliov, C.M.; Totan, A.; Calenic, B.; Greabu, M. Organic Nanomaterials and Their Applications in the Treatment of Oral Diseases. Molecules 2016, 21, 207. https://doi.org/10.3390/molecules21020207
Virlan MJR, Miricescu D, Radulescu R, Sabliov CM, Totan A, Calenic B, Greabu M. Organic Nanomaterials and Their Applications in the Treatment of Oral Diseases. Molecules. 2016; 21(2):207. https://doi.org/10.3390/molecules21020207
Chicago/Turabian StyleVirlan, Maria Justina Roxana, Daniela Miricescu, Radu Radulescu, Cristina M. Sabliov, Alexandra Totan, Bogdan Calenic, and Maria Greabu. 2016. "Organic Nanomaterials and Their Applications in the Treatment of Oral Diseases" Molecules 21, no. 2: 207. https://doi.org/10.3390/molecules21020207
APA StyleVirlan, M. J. R., Miricescu, D., Radulescu, R., Sabliov, C. M., Totan, A., Calenic, B., & Greabu, M. (2016). Organic Nanomaterials and Their Applications in the Treatment of Oral Diseases. Molecules, 21(2), 207. https://doi.org/10.3390/molecules21020207






