Comparative Study on the Characteristics of Weissella cibaria CMU and Probiotic Strains for Oral Care
Abstract
:1. Introduction
2. Results
2.1. Resistance against Lysozyme and Hydrogen Peroxide
2.1.1. Lysozyme
2.1.2. Hydrogen Peroxide
2.2. Acidogenic Potential
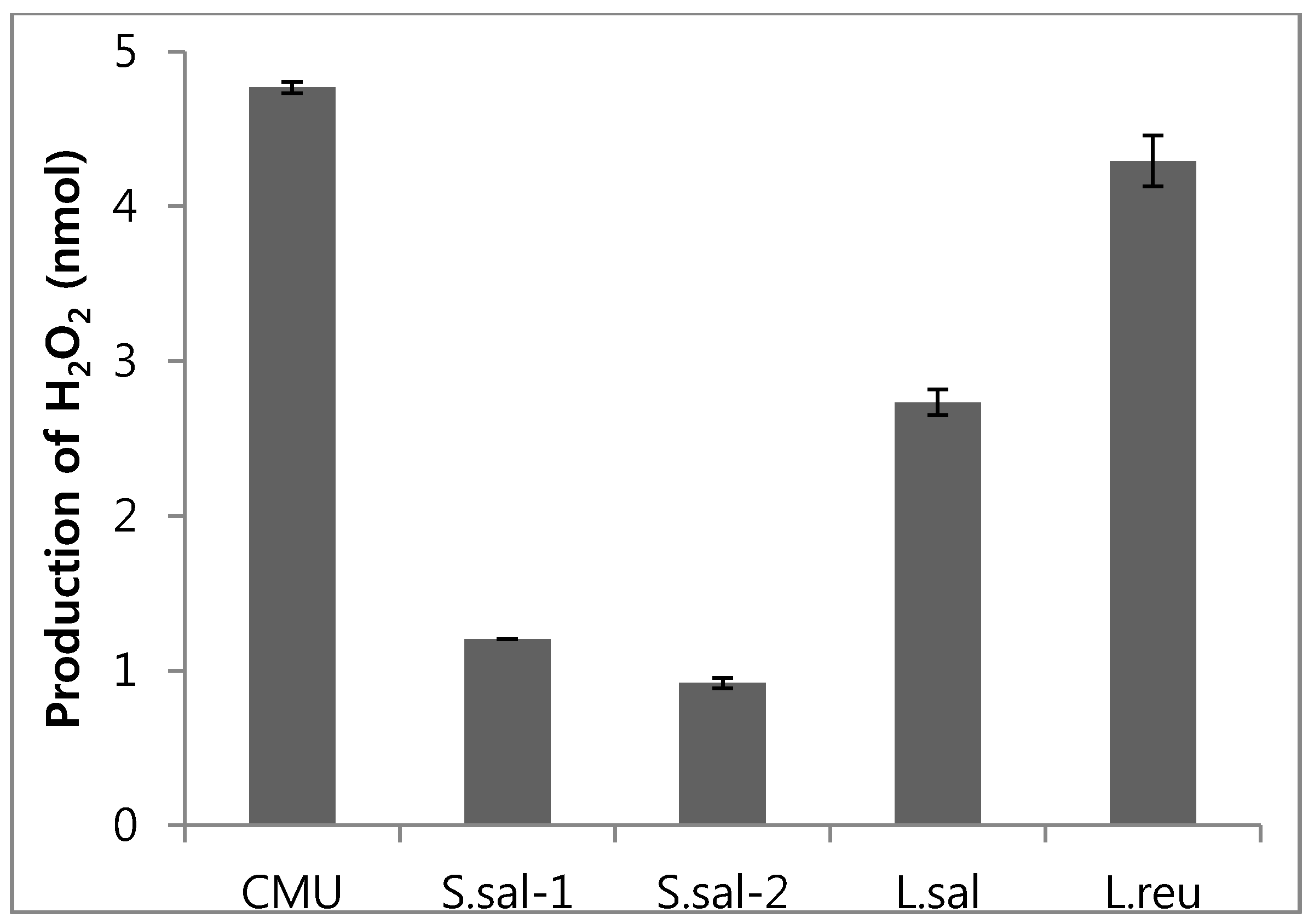
2.3. Hydrogen Peroxide Production Potential
2.4. Inhibition of Biofilm Formation
2.5. Antibacterial Activity
2.6. Coaggregation
2.7. Inhibition of VSC Production
3. Discussion
4. Materials and Methods
4.1. Bacterial Strains and Growth Conditions
4.2. Lysozyme Resistance on Bacterial Growth
4.3. Hydrogen Peroxide Resistance on Bacterial Growth
4.4. Acidogenic Potential
4.5. Hydrogen Peroxide Estimation
4.6. Inhibition of Biofilm Formation
4.7. Antibacterial Activity
4.8. Coaggregation Reaction
4.9. Inhibition of VSC (Volatile Sulfur Compounds) Production
4.10. Statistics
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Aas, J.A.; Paster, B.J.; Stokes, L.N.; Olsen, I.; Dewhirst, F.E. Defining the normal bacterial flora of the oral cavity. J. Clin. Microbiol. 2005, 43, 5721–5732. [Google Scholar] [CrossRef] [PubMed]
- Do, T.; Devine, D.; Marsh, P.D. Oral biofilms: Molecular analysis, challenges, and future prospects in dental diagnostics. Clin. Cosmet. Investig. Dent. 2013, 5, 11–19. [Google Scholar] [PubMed]
- Signoretto, C.; Bianchi, F.; Burlacchini, G.; Sivieri, F.; Spratt, D.; Canepari, P. Drinking habits are associated with changes in the dental plaque microbial community. J. Clin. Microbiol. 2010, 48, 347–356. [Google Scholar] [CrossRef] [PubMed]
- Baehni, P.; Guggenheim, B. Potential of diagnostic microbiology for treatment and prognosis of dental caries and periodontal diseases. Crit. Rev. Oral Biol. Med. 1996, 7, 259–277. [Google Scholar] [CrossRef] [PubMed]
- Loesche, W.; Straffon, L. Longitudinal investigation of the role of Streptococcus mutans in human fissure decay. Infect. Immun. 1979, 26, 498–507. [Google Scholar] [PubMed]
- Bowen, W.; Koo, H. Biology of Streptococcus mutans-derived glucosyltransferases: Role in extracellular matrix formation of cariogenic biofilms. Caries Res. 2011, 45, 69–86. [Google Scholar] [CrossRef] [PubMed]
- Bae, K.H.; Kim, H.D.; Jung, S.H.; Park, D.Y.; Kim, J.B.; Paik, D.I.; Chung, S.C. Validation of the Korean version of the oral health impact profile among the Korean elderly. Community Dent. Oral Epidemiol. 2007, 35, 73–79. [Google Scholar] [CrossRef] [PubMed]
- Williams, R.C.; Barnett, A.; Claffey, N.; Davis, M.; Gadsby, R.; Kellett, M.; Lip, G.Y.; Thackray, S. The potential impact of periodontal disease on general health: A consensus view. Curr. Med. Res. Opin. 2008, 24, 1635–1643. [Google Scholar] [CrossRef] [PubMed]
- Humphrey, L.L.; Fu, R.; Buckley, D.I.; Freeman, M.; Helfand, M. Periodontal disease and coronary heart disease incidence: A systematic review and meta-analysis. J. Gen. Intern. Med. 2008, 23, 2079–2086. [Google Scholar] [CrossRef] [PubMed]
- Haffajee, A.D.; Socransky, S.S. Microbial etiological agents of destructive periodontal diseases. Periodontology 2000 1994, 5, 78–111. [Google Scholar] [CrossRef] [PubMed]
- Nikawa, H.; Makihira, S.; Fukushima, H.; Nishimura, H.; Ozaki, Y.; Ishida, K.; Darmawan, S.; Hamada, T.; Hara, K.; Matsumoto, A. Lactobacillus reuteri in bovine milk fermented decreases the oral carriage of mutans streptococci. Int. J. Food Microbiol. 2004, 95, 219–223. [Google Scholar] [CrossRef] [PubMed]
- Kang, M.S.; Kim, B.G.; Chung, J.; Lee, H.C.; Oh, J.S. Inhibitory effect of Weissella cibaria isolates on the production of volatile sulphur compounds. J. Clin. Periodontol. 2006, 33, 226–232. [Google Scholar] [CrossRef] [PubMed]
- Kang, M.-S.; Chung, J.; Kim, S.-M.; Yang, K.-H.; Oh, J.-S. Effect of Weissella cibaria isolates on the formation of Streptococcus mutans biofilm. Caries Res. 2006, 40, 418–425. [Google Scholar] [CrossRef] [PubMed]
- Burton, J.; Chilcott, C.; Tagg, J. The rationale and potential for the reduction of oral malodour using Streptococcus salivarius probiotics. Oral Dis. 2005, 11, 29–31. [Google Scholar] [CrossRef] [PubMed]
- Ishikawa, H.; Aiba, Y.; Nakanishi, M.; Oh-hashi, Y.; Koga, Y. Suppression of Periodontal Pathogenic Bacteria in the Saliva of Humans by the Administration of Lactobacillus salivarius TI2711. J. Jpn. Soc. Periondontol. 2003, 45, 105–112. [Google Scholar] [CrossRef]
- Kopp-Hoolihan, L. Prophylactic and therapeutic uses of probiotics: A review. J. Am. Diet. Assoc. 2001, 101, 229–241. [Google Scholar] [CrossRef]
- Brown, A.C.; Valiere, A. Probiotics and medical nutrition therapy. Nutr. Clin. Care 2004, 7, 56–68. [Google Scholar] [PubMed]
- Isolauri, E.; Sütas, Y.; Kankaanpää, P.; Arvilommi, H.; Salminen, S. Probiotics: Effects on immunity. Am. J. Clin. Nutr. 2001, 73, 444s–450s. [Google Scholar] [CrossRef] [PubMed]
- Parvez, S.; Malik, K.; Ah Kang, S.; Kim, H.Y. Probiotics and their fermented food products are beneficial for health. J. Appl. Microbiol. 2006, 100, 1171–1185. [Google Scholar] [CrossRef] [PubMed]
- Bjorkroth, K.J.; Schillinger, U.; Geisen, R.; Weiss, N.; Hoste, B.; Holzapfel, W.H.; Korkeala, H.J.; Vandamme, P. Taxonomic study of Weissella confusa and description of Weissella cibaria sp. nov. detected in food and clinical samples. Int. J. Syst. Evolut. Microbiol. 2002, 52, 141–148. [Google Scholar] [CrossRef] [PubMed]
- Lee, K.W.; Park, J.-Y.; Chun, J.-Y.; Han, N.-S.; Kim, J.-H. Importance of Weissella species during kimchi fermentation and future works. Korean J. Microbiol. Biotechnol. 2010, 38, 341–348. [Google Scholar]
- Wu, C.D.; Darout, I.; Skaug, N. Chewing sticks: Timeless natural toothbrushes for oral cleansing. J. Periodontal Res. 2001, 36, 275–284. [Google Scholar] [CrossRef] [PubMed]
- Callewaert, L.; Michiels, C.W. Lysozymes in the animal kingdom. J. Biosci. 2010, 35, 127–160. [Google Scholar] [CrossRef] [PubMed]
- Thomas, E.L. Myeloperoxidase-hydrogen peroxide-chloride antimicrobial system: Effect of exogenous amines on antibacterial action against Escherichia coli. Infect. Immun. 1979, 25, 110–116. [Google Scholar] [PubMed]
- Burton, J.P.; Wescombe, P.A.; Macklaim, J.M.; Chai, M.H.; MacDonald, K.; Hale, J.D.; Tagg, J.; Reid, G.; Gloor, G.B.; Cadieux, P.A. Persistence of the oral probiotic Streptococcus salivarius M18 is dose dependent and megaplasmid transfer can augment their bacteriocin production and adhesion characteristics. PLoS ONE 2013, 8, e65991. [Google Scholar] [CrossRef] [PubMed]
- Tingirikari, J.M.; Kothari, D.; Goyal, A. Superior prebiotic and physicochemical properties of novel dextran from Weissella cibaria JAG8 for potential food applications. Food Funct. 2014, 5, 2324–2330. [Google Scholar] [CrossRef] [PubMed]
- Kõll-Klais, P.; Mändar, R.; Leibur, E.; Marcotte, H.; Hammarström, L.; Mikelsaar, M. Oral lactobacilli in chronic periodontitis and periodontal health: Species composition and antimicrobial activity. Oral Microbiol. Immunol. 2005, 20, 354–361. [Google Scholar] [CrossRef] [PubMed]
- Krasse, P.; Carlsson, B.; Dahl, C.; Paulsson, A.; Nilsson, A.; Sinkiewicz, G. Decreased gum bleeding and reduced gingivitis by the probiotic Lactobacillus reuteri. Swed. Dent. J. 2005, 30, 55–60. [Google Scholar]
- Kolenbrander, P.E. Oral microbial communities: Biofilms, interactions, and genetic systems 1. Annu. Rev. Microbiol. 2000, 54, 413–437. [Google Scholar] [CrossRef] [PubMed]
- Schmidt, N.F.; Missan, S.R.; Tarbet, W.J.; Cooper, A.D. The correlation between organoleptic mouth-odor ratings and levels of volatile sulfur compounds. Oral Surg. Oral Med. Oral Pathol. 1978, 45, 560–567. [Google Scholar] [CrossRef]
- Iacono, V.J.; MacKay, B.J.; DiRienzo, S.; Pollock, J.J. Selective antibacterial properties of lysozyme for oral microorganisms. Infect. Immun. 1980, 29, 623–632. [Google Scholar] [PubMed]
- Turchi, B.; Mancini, S.; Fratini, F.; Pedonese, F.; Nuvoloni, R.; Bertelloni, F.; Ebani, V.V.; Cerri, D. Preliminary evaluation of probiotic potential of Lactobacillus plantarum strains isolated from Italian food products. World J. Microbiol. Biotechnol. 2013, 29, 1913–1922. [Google Scholar] [CrossRef] [PubMed]
- Sun, J.; Hu, X.-L.; Le, G.-W.; Shi, Y.-H. Inhibition of Fe-induced colon oxidative stress by lactobacilli in mice. World J. Microbiol. Biotechnol. 2013, 29, 209–216. [Google Scholar] [CrossRef] [PubMed]
- Bosch, M.; Nart, J.; Audivert, S.; Bonachera, M.A.; Alemany, A.S.; Fuentes, M.C.; Cuné, J. Isolation and characterization of probiotic strains for improving oral health. Arch. Oral Biol. 2012, 57, 539–549. [Google Scholar] [CrossRef] [PubMed]
- Viswanathan, K.; Vadivoo, V.; Raj, G.D. Rapid determination of hydrogen peroxide produced by Lactobacillus using enzyme coupled rhodamine isocyanide/calcium phosphate nanoparticles. Biosens. Bioelectron. 2014, 61, 200–208. [Google Scholar] [CrossRef] [PubMed]
- Djordjevic, D.; Wiedmann, M.; McLandsborough, L. Microtiter plate assay for assessment of Listeria monocytogenes biofilm formation. Appl. Environ. Microbiol. 2002, 68, 2950–2958. [Google Scholar] [CrossRef] [PubMed]
- Kang, M.-S.; Oh, J.-S.; Lee, H.-C.; Lim, H.-S.; Lee, S.-W.; Yang, K.-H.; Choi, N.-K.; Kim, S.-M. Inhibitory effect of Lactobacillus reuteri on periodontopathic and cariogenic bacteria. J. Microbiol. 2011, 49, 193–199. [Google Scholar] [CrossRef] [PubMed]
- Cisar, J.; Kolenbrander, P.; McIntire, F. Specificity of coaggregation reactions between human oral streptococci and strains of Actinomyces viscosus or Actinomyces naeslundii. Infect. Immun. 1979, 24, 742–752. [Google Scholar] [PubMed]
- Sample Availability: Samples of W. cibaria CMU, S. sal-1, S. sal-2, L-sal, and L. reu are not available from the authors.

| Probiotics | PAV * | F Values | ||||
|---|---|---|---|---|---|---|
| Glu | Fru | Suc | Lac | YE | ||
| CMU | 37.29 ± 0.09 a | 40.09 ± 0.05 b | 47.28 ± 0.04 c | 41.71 ± 0.19 d | 56.31 ± 0.09 e | 15,055.652 *** |
| S. sal-1 | 26.08 ± 0.04 a | 26.44 ± 0.07 a | 28.58 ± 0.15 b | 28.74 ± 0.44 b | 51.03 ± 0.06 c | 7563.223 *** |
| S. sal-2 | 22.28 ± 0.03 a | 29.62 ± 0.15 b | 25.03 ± 0.04 c | 29.48 ± 0.01 d | 37.13 ± 0.04 e | 17,975.809 *** |
| L. sal | 31.1 6± 0.08 a | 29.00 ± 0.01 b | 32.78 ± 0.05 c | 32.33 ± 0.15 d | 44.97 ± 0.01 e | 18,270.924 *** |
| L. reu | 35.82± 0.19 a | 39.16 ± 0.01 b | 36.37 ± 0.01 c | 39.87 ± 0.10 d | 56.47 ± 0.15 e | 16,185.140 *** |
| Probiotics | S. mutans:Probiotics | |||
|---|---|---|---|---|
| 1:1 | 2:1 | 4:1 | 8:1 | |
| CMU | 94.7 ± 0.3 | 96.0 ± 0.1 | 96.8 ± 0.5 | 95.4 ± 0.1 |
| S. sal-1 | 59.5 ± 2.8 | 54.6 ± 3.4 | 51.5 ± 2.8 | 24.1 ± 2.0 |
| S. sal-2 | 0 | 0 | 0 | 0 |
| L. sal | 93.9 ± 0.5 | 90.7 ± 2.3 | 84.6 ± 4.4 | 78.8 ± 4.5 |
| L. reu | 85.3 ± 4.0 | 0 | 0 | 0 |
| Probiotics | F. nucleatum | P. gingivalis | ||||||
|---|---|---|---|---|---|---|---|---|
| 1:1 | 2:1 | 4:1 | 8:1 | 1:1 | 2:1 | 4:1 | 8:1 | |
| CMU | 97.9 ± 0.1 a | 96.0 ± 0.5 a | 90.6 ± 0.8 a | 36.1 ± 2.8 a | 96.9 ± 0.4 a | 99.0 ± 0.2 a | 98.8 ± 0.2 a | 99.7 ± 0.3 a |
| S. sal-1 | 41.3 ± 2.8 b | 12.9 ± 3.1 b | 3.9 ± 4.5 b | 0.0 b | 82.9 ± 2.7 b | 55.5 ± 7.4 b | 28.7 ± 5.9 b | 0.0 b |
| S. sal-2 | 47.0 ± 1.2 c | 15.2 ± 8.5 b | 2.0 ± 6.2 b | 0.0 b | 80.0 ± 3.4 b | 36.5 ± 9.7 c | 9.9 ± 0.7 c | 5.2 ± 15.6 c |
| L. sal | 97.4 ± 0.3 a | 94.9 ± 0.4 a | 97.8 ± 0.4 c | 94.3 ± 0.8 c | 96.6 ± 0.5 a | 96.7 ± 0.6 a | 98.2 ± 0.2 a | 98.9 ± 0.1 a |
| L. reu | 97.3 ± 0.1 a | 95.7 ± 1.1 a | 97.0 ± 0.4 c | 51.3 ± 2.5 d | 95.7 ± 0.5 a | 97.4 ± 0.8 a | 98.1 ± 0.4 a | 98.5 ± 0.4 a |
| F values | 1389.452 *** | 358.904 *** | 795.049 *** | 1551.685 *** | 52.484 *** | 84.464 *** | 800.976 *** | 607.859 *** |
| Probiotics | F. nucleatum | P. gingivalis | ||||||
|---|---|---|---|---|---|---|---|---|
| 1:1 | 2:1 | 4:1 | 8:1 | 1:1 | 2:1 | 4:1 | 8:1 | |
| CMU | 90.9 ± 3.4 a | 40.1 ± 8.1 a | 0.0 a | 0.0 a | 96.2 ± 0.3 a | 81.9 ± 5.1 a | 3.8 ± 20.2 a | 0.0 |
| S. sal-1 | 42.8 ± 1.6 b | 22.8 ± 7.5 b | 15.3 ± 3.4 b | 11.1 ± 4.1 b | 35.5 ± 17.8 b | 12.0 ± 19.2 b | 0.0 a | 0.0 |
| S. sal-2 | 40.4 ± 4.0 b | 19.6 ± 4.0 b | 13.2 ± 5.7 b | 13.4 ± 4.2 b | 37.1 ± 6.1 b | 0.0 c | 0.0 a | 0.0 |
| L. sal | 96.3 ± 0.2 c | 96.0 ± 1.9 c | 47.3 ± 12.5 c | 0.0 a | 96.3 ± 0.5 a | 96.3 ± 0.5 d | 91.6 ± 2.5 b | 0.0 |
| L. reu | 94.2 ± 1.3 a,c | 79.8 ± 4.5 d | 0.0 a | 0.0 a | 95.1 ± 0.5 a | 94.7 ± 0.4 a,d | 5.8 ± 8.0 a | 0.0 |
| F values | 380.033 *** | 110.069 *** | 27.888 *** | 20.166 *** | 45.148 *** | 131.017 *** | 173.838 *** | |
| Probiotics | Coaggregation (%) | |||
|---|---|---|---|---|
| S. mutans | S. sobriuns | F. nucleatum | P. gingivalis | |
| CMU | 5.8 ± 0.6 a | 0.0 a | 81.2 ± 0.4 a | 3.7 ± 1.2 a |
| S. sal-1 | 0.0 b | 13.8 ± 1.3 b | 78.9 ± 0.3 b | 0.0 b |
| S. sal-2 | 0.0 b | 2.5 ± 0.2 c | 72.7 ± 0.7 c | 1.5 ± 0.8 c |
| L. sal | 0.0 b | 0.0 a | 0.0 d | 0.0 b |
| L. reu | 0.0 b | 0.0 a | 49.6 ± 0.5 e | 0.0 b |
| F values | 328.737 *** | 309.850 *** | 16,212.931 *** | 20.446 *** |
| Strains | VSC (ppb) by F. nucleatum | VSC (ppb) by P. gingivalis | ||||
|---|---|---|---|---|---|---|
| H2S | CH3SH | Inhibition (%) | H2S | CH3SH | Inhibition (%) | |
| Mono | 25,640 ± 702 | 37,225 ± 782 | 0 | 12,791 ± 432 | 33,553 ± 876 | 0 |
| CMU | 1352 ± 1272 a | 527 ± 152 a | 97.0 | 2095 ± 182 a | 717 ± 171 a | 93.9 |
| S. sal-1 | 3540 ± 835 b | 1532 ± 500 b | 91.9 | 1849 ± 172 a | 1423 ± 78 b | 92.9 |
| S. sal-2 | 6777 ± 838 c | 2030 ± 246 b,c | 86.0 | 1531 ± 82 b | 1475 ± 210 b | 93.5 |
| L. sal | 7081 ± 290 c | 2278 ± 323 c | 85.1 | 1844 ± 78 a | 1335 ± 130 b | 93.1 |
| L. reu | 9470 ± 833 d | 2242 ± 505 c | 81.4 | 3253 ± 204 c | 2290 ± 162 c | 88.0 |
| F values | 40.340 *** | 11.592 *** | 57.013 *** | 38.527 *** | ||
© 2016 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Jang, H.-J.; Kang, M.-S.; Yi, S.-H.; Hong, J.-Y.; Hong, S.-P. Comparative Study on the Characteristics of Weissella cibaria CMU and Probiotic Strains for Oral Care. Molecules 2016, 21, 1752. https://doi.org/10.3390/molecules21121752
Jang H-J, Kang M-S, Yi S-H, Hong J-Y, Hong S-P. Comparative Study on the Characteristics of Weissella cibaria CMU and Probiotic Strains for Oral Care. Molecules. 2016; 21(12):1752. https://doi.org/10.3390/molecules21121752
Chicago/Turabian StyleJang, Hye-Jin, Mi-Sun Kang, Sung-Hun Yi, Ji-Young Hong, and Sang-Pil Hong. 2016. "Comparative Study on the Characteristics of Weissella cibaria CMU and Probiotic Strains for Oral Care" Molecules 21, no. 12: 1752. https://doi.org/10.3390/molecules21121752
APA StyleJang, H.-J., Kang, M.-S., Yi, S.-H., Hong, J.-Y., & Hong, S.-P. (2016). Comparative Study on the Characteristics of Weissella cibaria CMU and Probiotic Strains for Oral Care. Molecules, 21(12), 1752. https://doi.org/10.3390/molecules21121752






