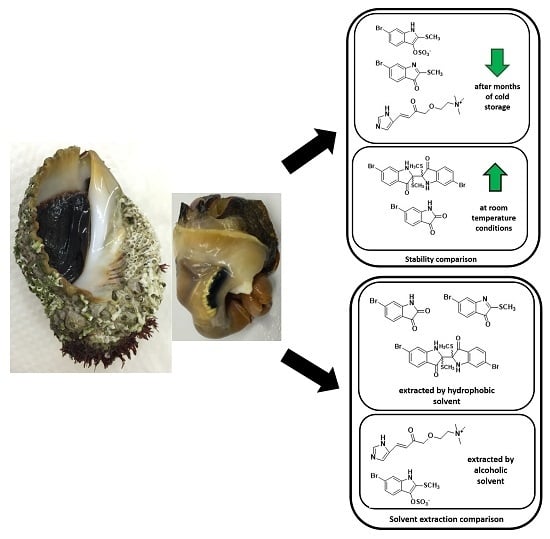
Extraction and Quantification of Bioactive Tyrian Purple Precursors: A Comparative and Validation Study from the Hypobranchial Gland of a Muricid Dicathais orbita
Abstract
:1. Introduction
2. Results and Discussion
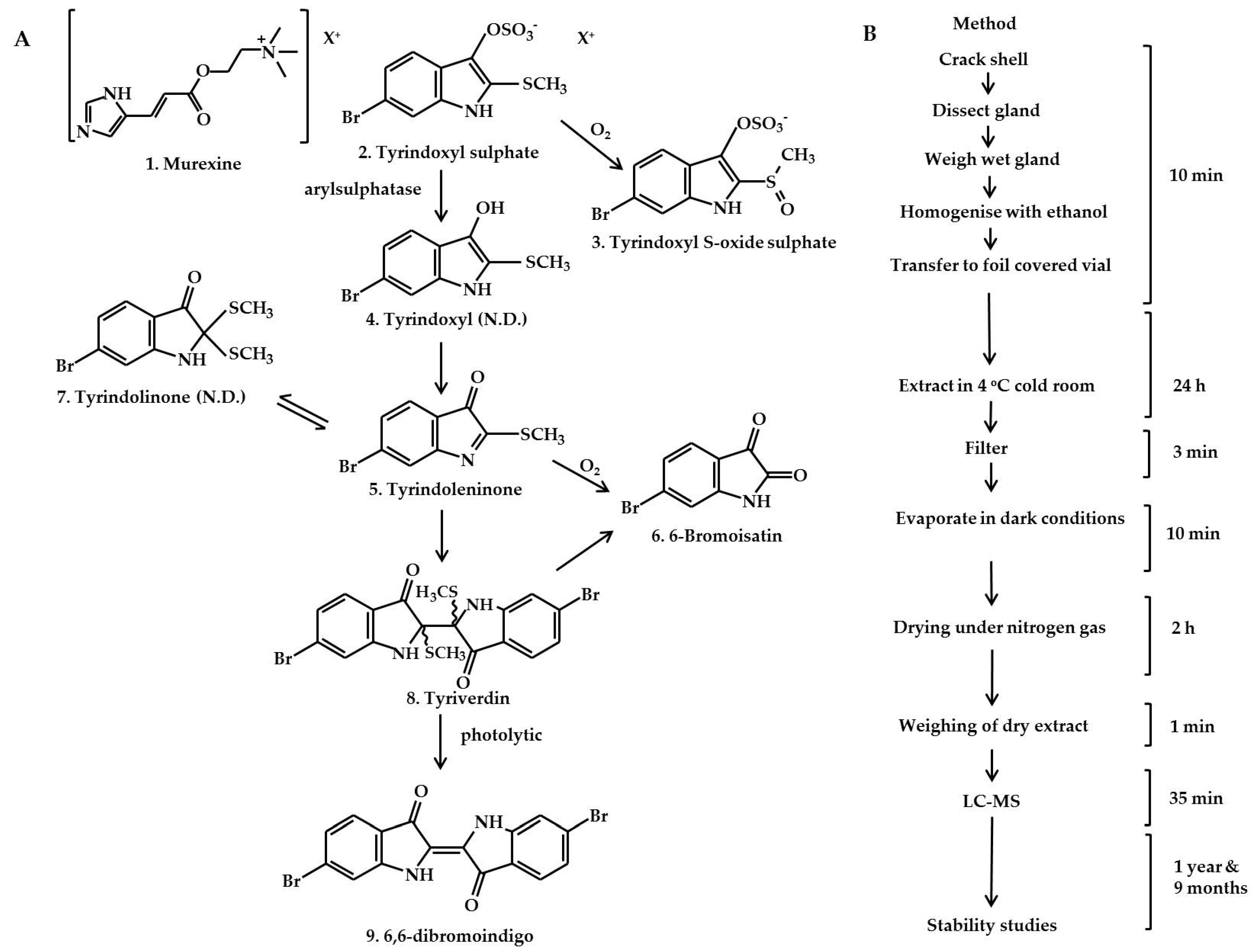
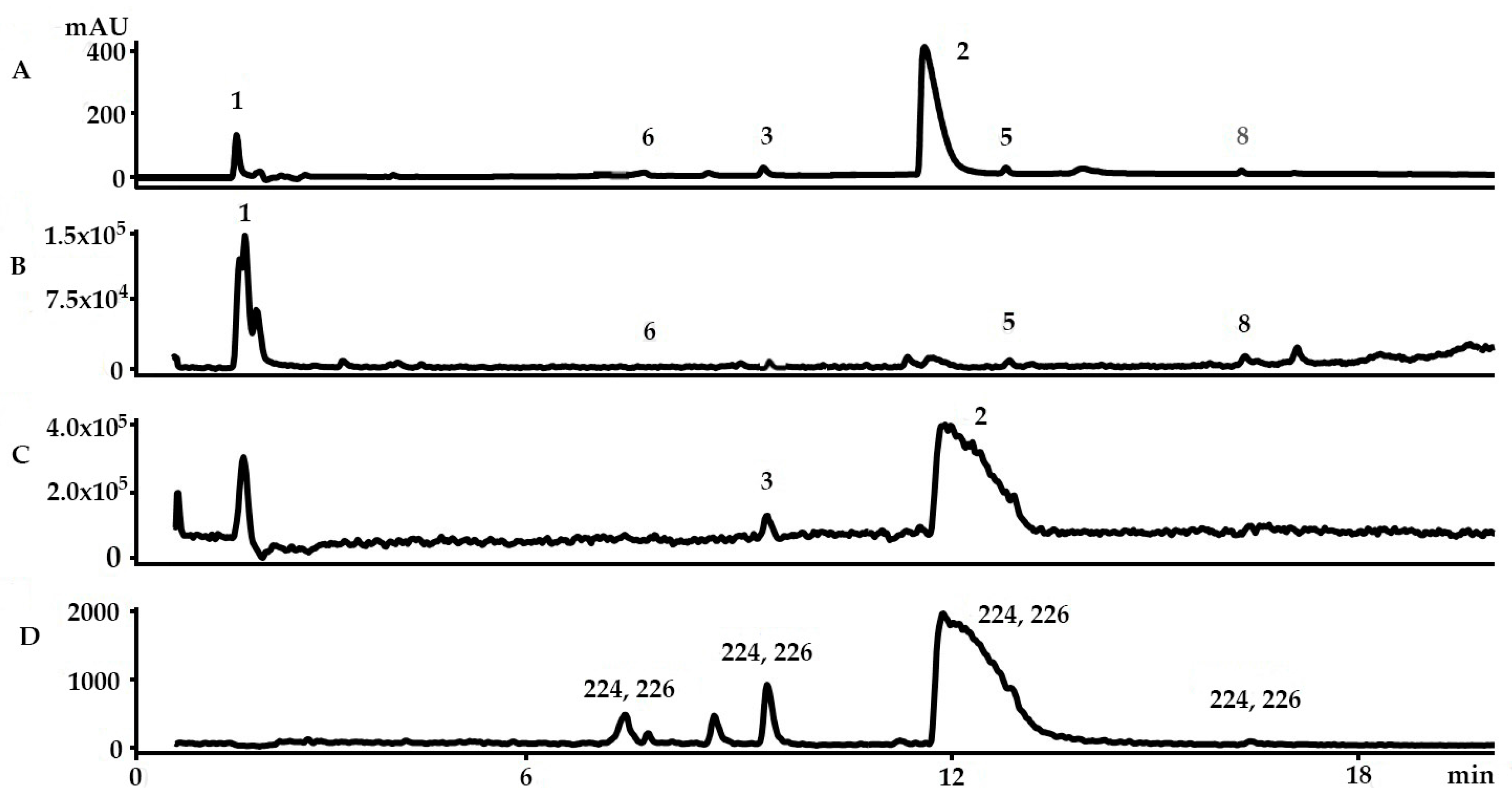
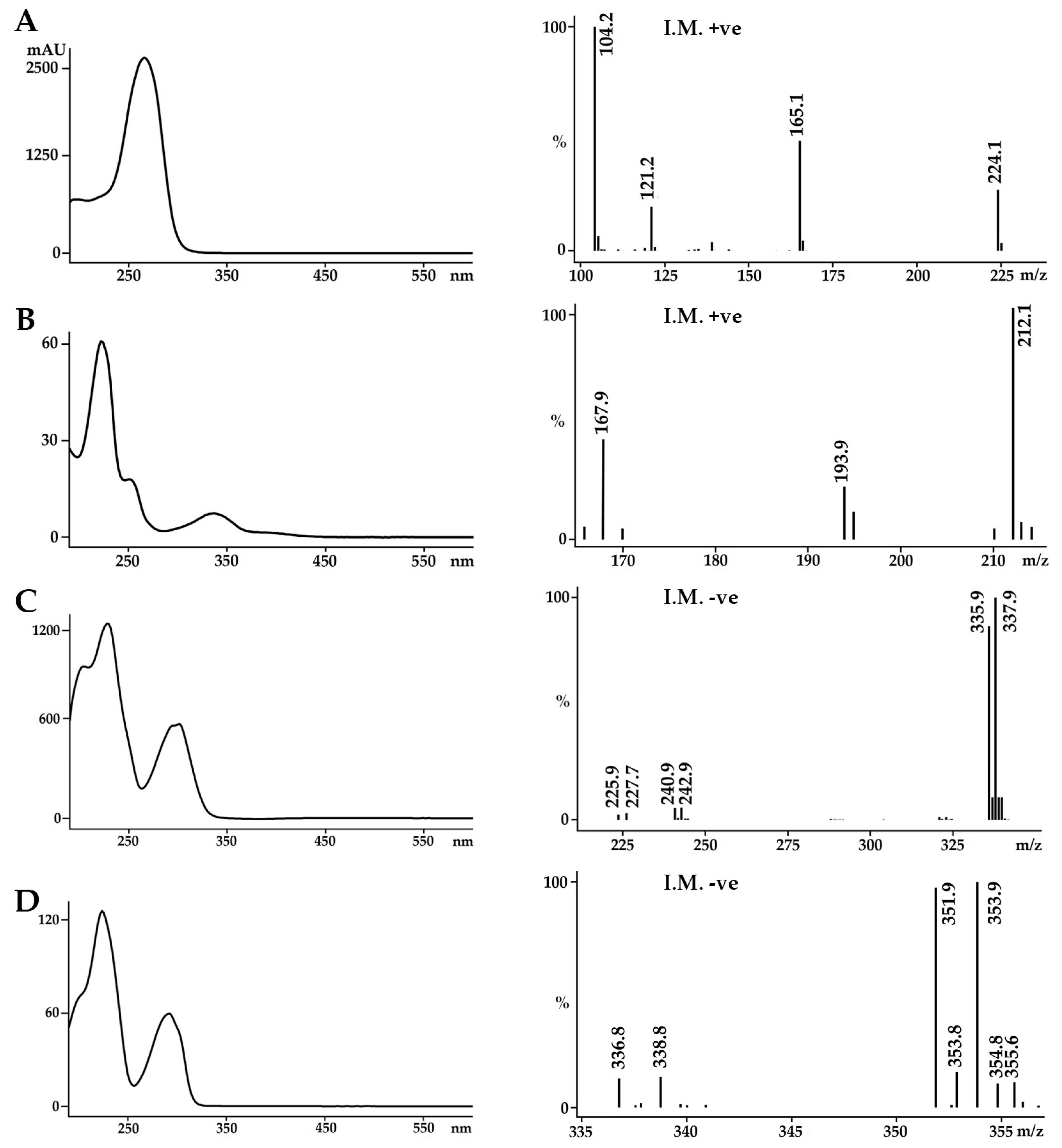
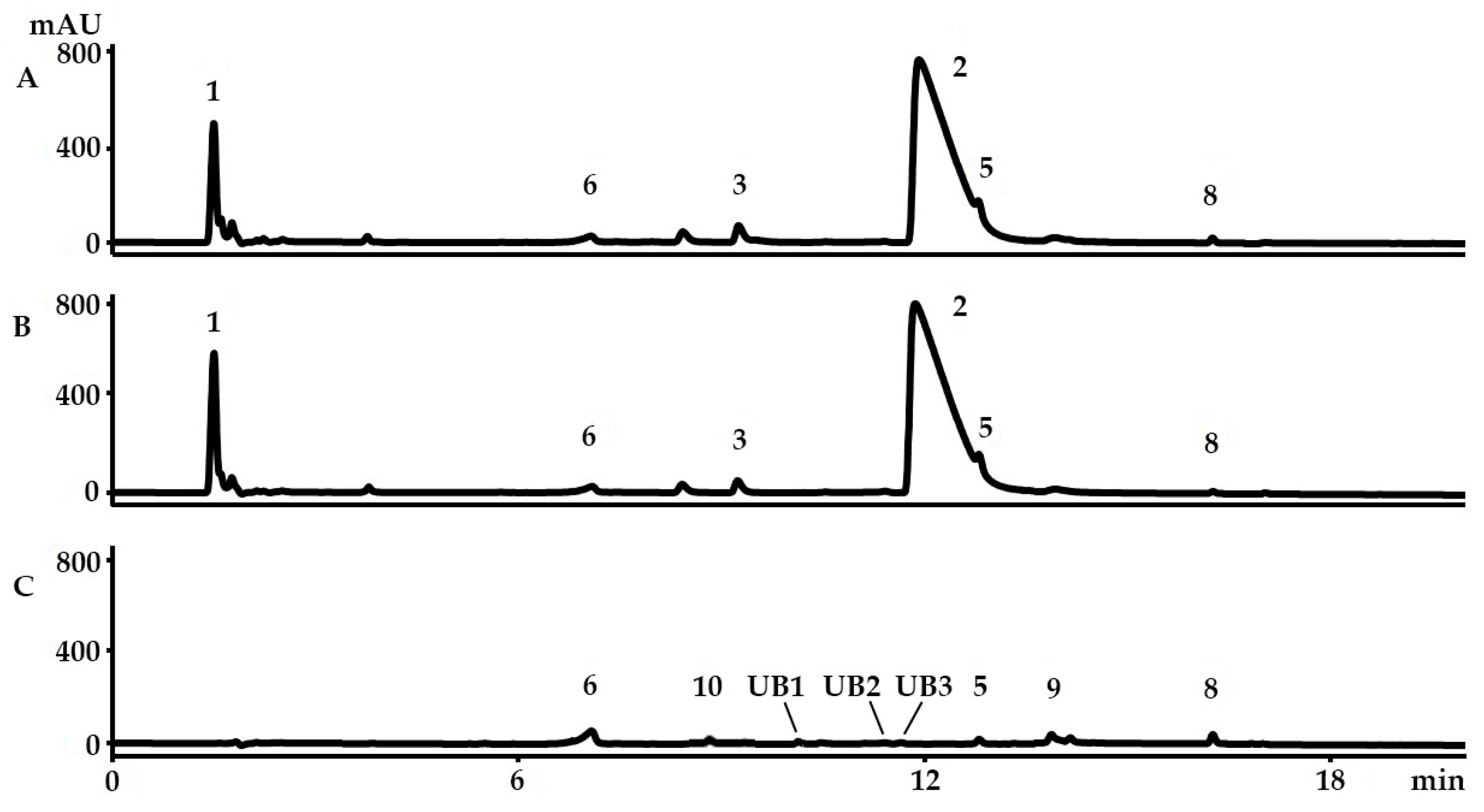
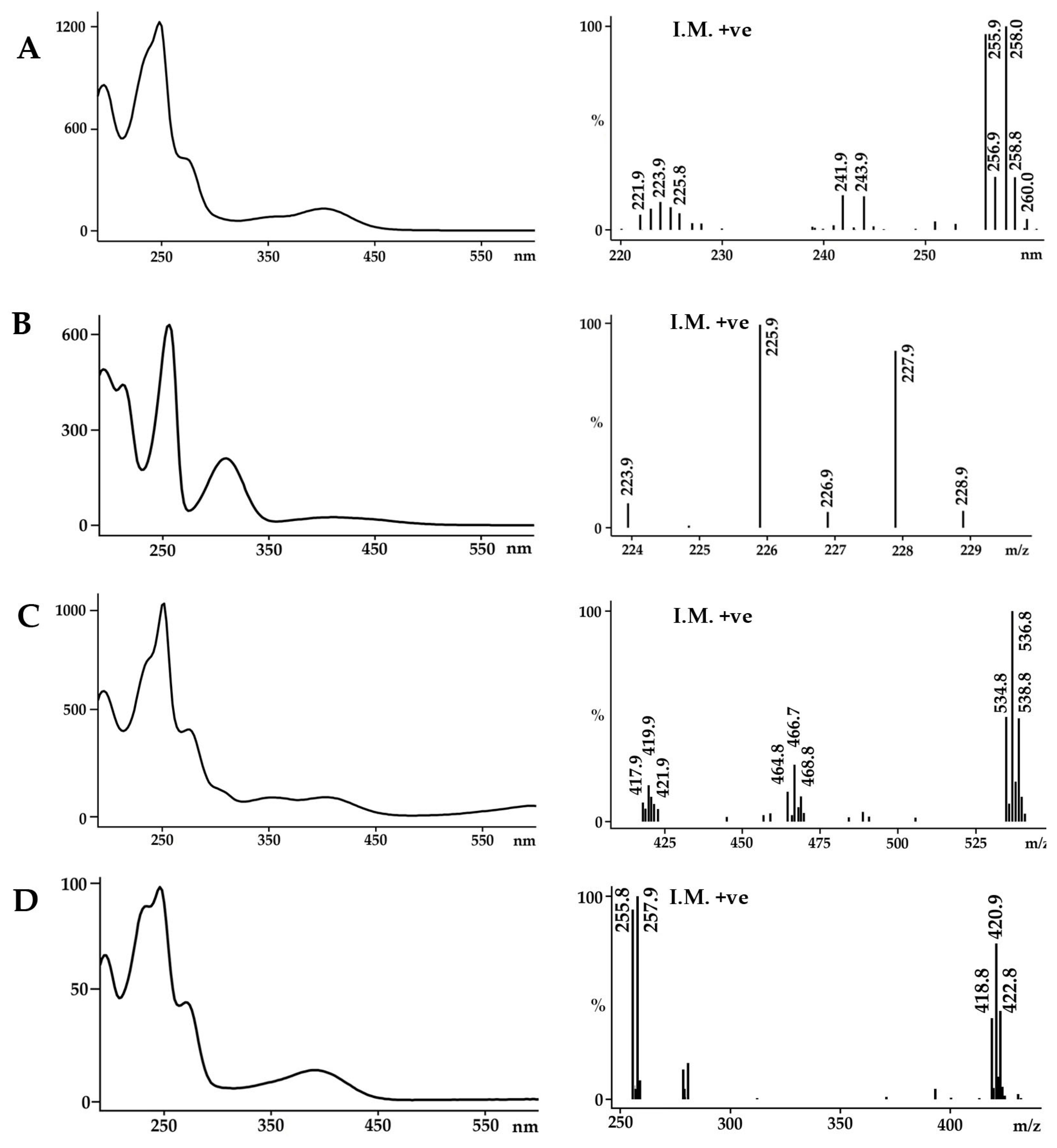
2.1. Identification of the Bioactive Compounds in the Extracts
2.2. Validation Studies
Precision, Linearity, Limit of Detection (LOD), and Limit of Quantification (LOQ)
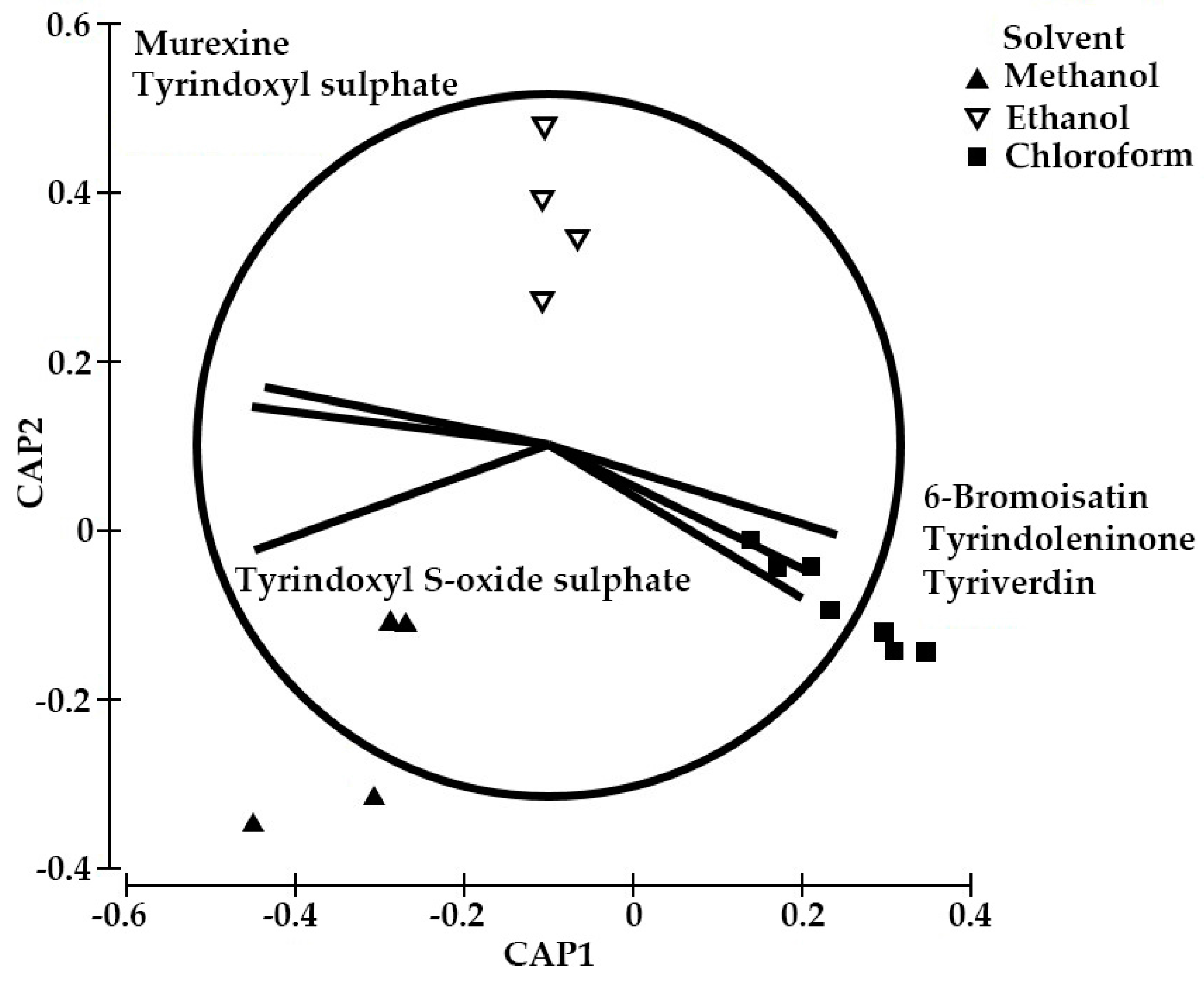
2.3. Yield of the Extracts and Quantification of the Main Precursors from Different Organic Solvents
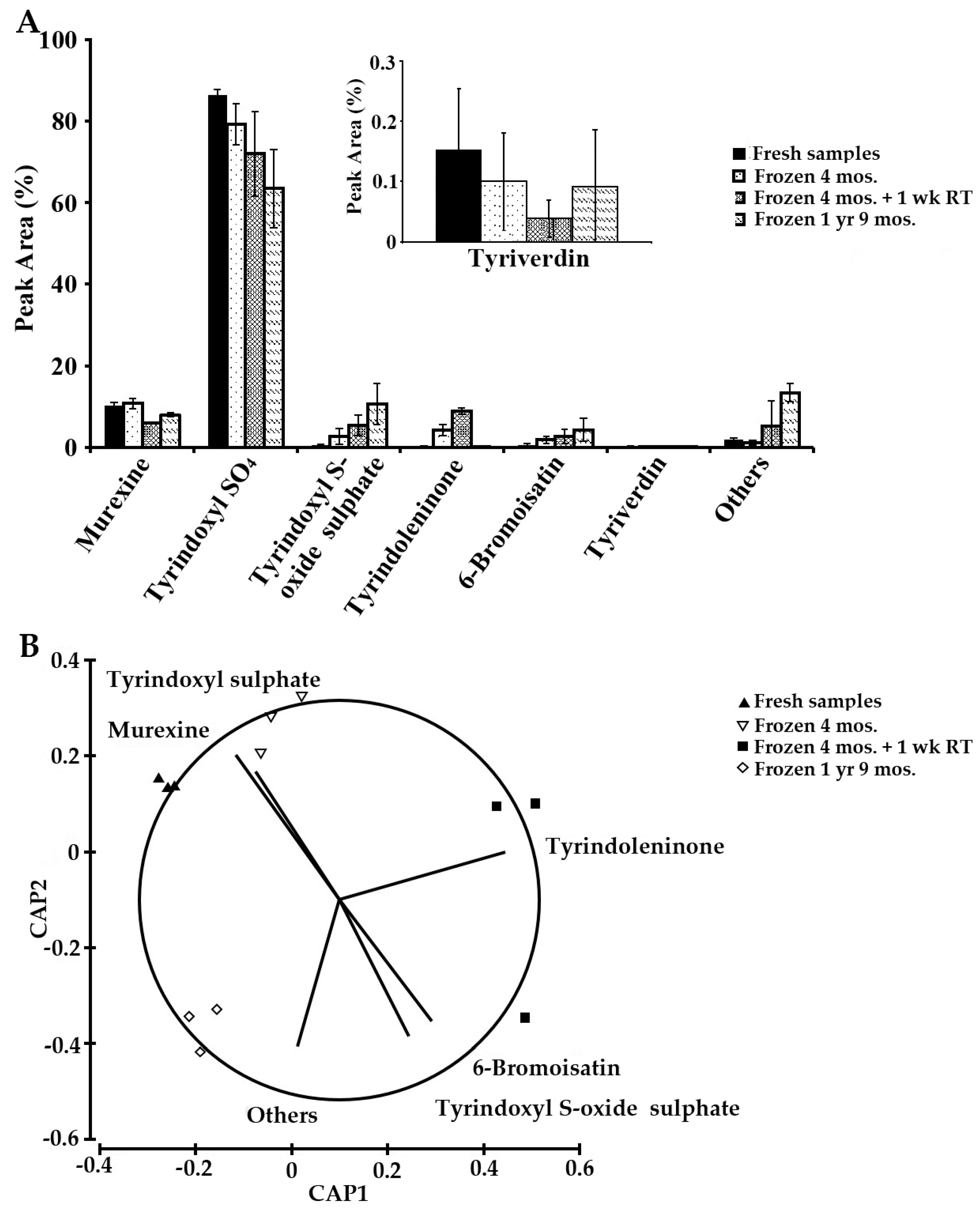
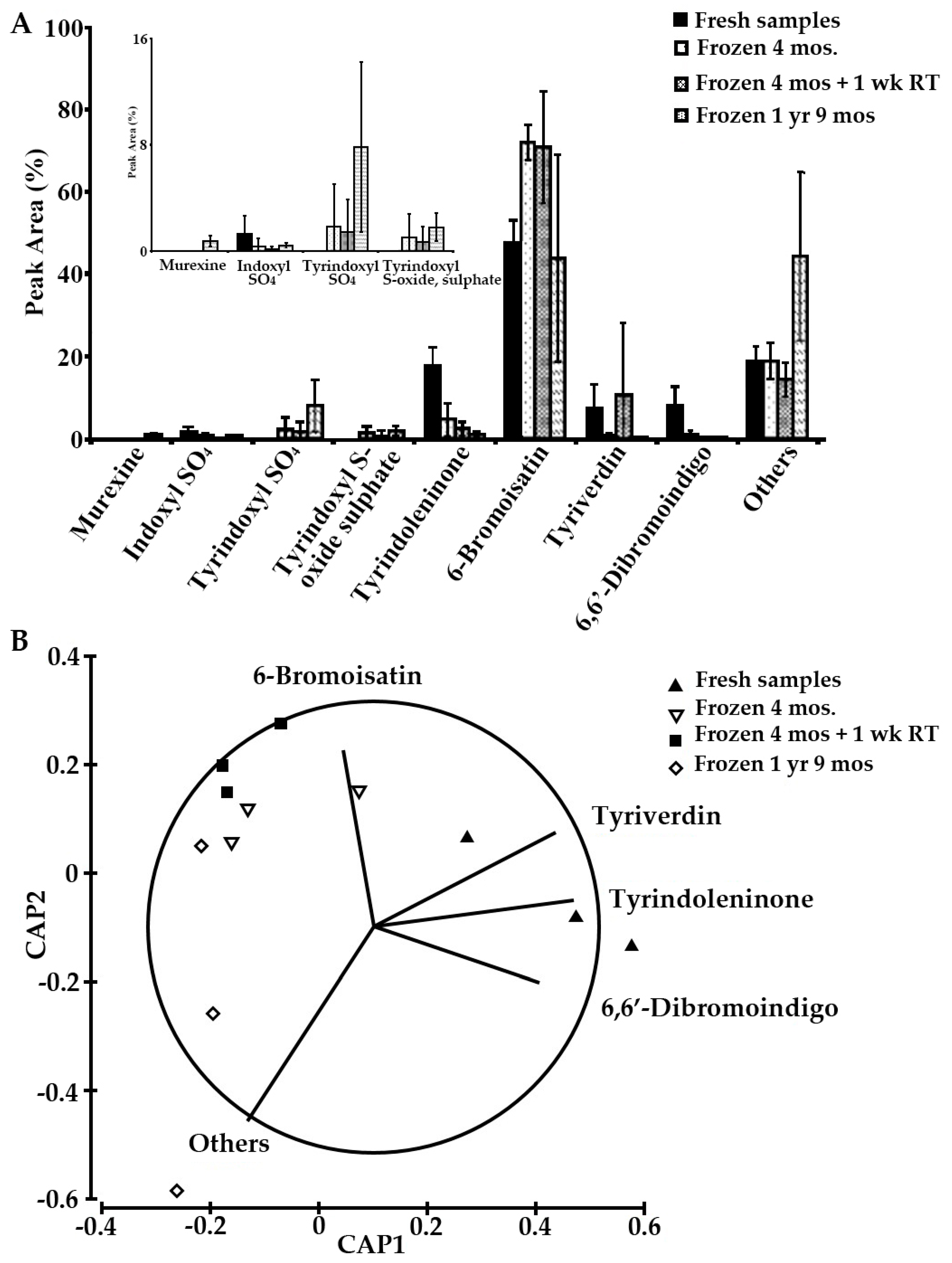
2.4. Sample Stability Test
3. Materials and Methods
3.1. Collection of Samples
3.2. Dissection and Extraction
Extraction Method
3.3. LC-MS Analysis
3.4. Identification of the Secondary Metabolites
3.4.1. Purification of Reference Standards
3.4.2. Confirmation of Reference Compound Identification
3.5. Method Validation: Precision, Linearity Curve, Limit of Detection (LOD), and Limit of Quantitation (LOQ)
3.6. Stability Testing
3.7. Statistical Analysis
4. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Blunt, J.W.; Copp, B.R.; Keyzers, R.A.; Munro, M.H.; Prinsep, M.R. Marine natural products. Nat. Prod. Rep. 2015, 32, 116–211. [Google Scholar] [CrossRef] [PubMed]
- Villa, F.A.; Gerwick, L. Marine natural product drug discovery: Leads for treatment of inflammation, cancer, infections, and neurological disorders. Immunopharm. Immunotoxicol. 2010, 32, 228–237. [Google Scholar] [CrossRef] [PubMed]
- Li, A.C.; Sabo, A.M.; McCormick, T.; Johnston, S.M. Quantitative analysis of squalamine, a self-ionization-suppressing aminosterol sulfate, in human plasma by LC-MS/MS. J. Pharm. Biomed. 2004, 34, 631–641. [Google Scholar] [CrossRef]
- Doggrell, S.A. Lyprinol—Is it a useful anti-inflammatory agent? Evid.-Based Complement. Altern. 2011, 2011, 307121. [Google Scholar] [CrossRef] [PubMed]
- Roy, L.; Harrell, C.C.; Ryan, A.S.; Thorsteinsson, T.; Sancilio, F.D. Development and validation of a single HPLC method for analysis of purines in fish oil supplements. Food Nutr. Sci. 2013, 4, 1255–1259. [Google Scholar] [CrossRef]
- Benkendorff, K.; Rudd, D.; Nongmaithem, B.D.; Liu, L.; Young, F.; Edwards, V.; Avila, C.; Abbott, C.A. Are the traditional medical uses of Muricidae molluscs substantiated by their pharmacological properties and bioactive compounds? Mar. Drugs 2015, 13, 5237–5275. [Google Scholar] [CrossRef] [PubMed]
- Cooksey, C. Tyrian purple: 6,6′-Dibromoindigo and related compounds. Molecules 2001, 6, 736–769. [Google Scholar] [CrossRef]
- Benkendorff, K. Natural products research in the Australian marine invertebrate Dicathais orbita. Mar. Drugs 2013, 11, 1370–1398. [Google Scholar] [CrossRef] [PubMed]
- Putheti, R.P.; Okigbo, R.N.; Patil, S.C.; Advanapu, M.; Leburu, R. Method development and validations: Characterization of critical elements in the development of pharmaceuticals. Int. J. Health Res. 2008, 1, 11–20. [Google Scholar] [CrossRef]
- Betz, J.M.; Brown, P.N.; Roman, M.C. Accuracy, precision, and reliability of chemical measurements in natural products research. Fitoterapia 2011, 82, 44–52. [Google Scholar] [CrossRef] [PubMed]
- Green, J.M. Doing a thorough method validation can be tedious, but the consequences of not doing it right are wasted time, money and resources. Anal. Chem. 1996, 68, 305A–309A. [Google Scholar] [CrossRef]
- Edwards, V.; Benkendorff, K.; Young, F. Marine compounds selectively induce apoptosis in female reproductive cancer cells but not in primary-derived human reproductive granulosa cells. Mar. Drugs 2012, 10, 64–83. [Google Scholar] [CrossRef] [PubMed]
- Esmaeelian, B.; Benkendorff, K.; Johnston, M.R.; Abbott, C.A. Purified brominated indole derivatives from Dicathais orbita induce apoptosis and cell cycle arrest in colorectal cancer cell lines. Mar. Drugs 2013, 11, 3802–3822. [Google Scholar] [CrossRef] [PubMed]
- Esmaeelian, B.; Abbott, C.A.; Le Leu, R.K.; Benkendorff, K. 6-Bromoisatin found in muricid mollusc extracts inhibits colon cancer cell proliferation and induces apoptosis, preventing early stage tumor formation in a colorectal cancer rodent model. Mar. Drugs 2014, 12, 17–35. [Google Scholar] [CrossRef] [PubMed]
- Benkendorff, K.; Bremner, J.B.; Davis, A.R. Tyrian purple precursors in the egg masses of the Australian muricid, Dicathais orbita: A possible role. J. Chem. Ecol. 2000, 26, 1037–1050. [Google Scholar] [CrossRef]
- Benkendorff, K.; Brenmner, J.B.; Davis, A.R. Indole derivatives from the egg masses muricid molluscs. Molecules 2001, 6, 70–78. [Google Scholar] [CrossRef]
- Westley, C.B.; Benkendorff, K.; McIver, C.M.; Le Leu, R.K.; Abbott, C.A. Gastrointestinal and hepatotoxicity assessment of an anticancer extract from muricid molluscs. Evid.-Based Complement. Altern. 2013, 837370. [Google Scholar] [CrossRef] [PubMed]
- Roseghini, M.; Severini, C.; Erspamer, G.F.; Erspamer, V. Choline esters and biogenic amines in the hypobranchial gland of 55 molluscan species of the neogastropod Muricoidea superfamily. Toxicon 1996, 34, 33–55. [Google Scholar] [CrossRef]
- Shiomi, K.; Ishiia, M.; Shimakuraa, K.; Nagashimaa, Y.; Chinob, M. Tigloylcholine: A new choline ester toxin from the hypobranchial gland of two species of muricid gastropods (Thais clavigera and Thais bronni). Toxicon 1998, 36, 795–798. [Google Scholar] [CrossRef]
- Bobzin, S.C.; Yang, S.; Kasten, T.P. Application of liquid chromatography-nuclear magnetic resonance spectroscopy to the identification of natural products. J. Chromatogr. B 2000, 748, 259–267. [Google Scholar] [CrossRef]
- Westley, C.; Benkendorff, K. Sex-specific Tyrian purple genesis: Precursor and pigment distribution in the reproductive system of the marine mollusc, Dicathais orbita. J. Chem. Ecol. 2008, 34, 44–56. [Google Scholar] [CrossRef] [PubMed]
- Westley, C.B.; McIver, C.M.; Abbott, C.A.; Le Leu, R.K.; Benkendorff, K. Enhanced acute apoptotic response to azoxymethane-induced DNA damage in the rodent colonic epithelium by Tyrian purple precursors: A potential colorectal cancer chemopreventative. Cancer Biol. Ther. 2010, 9, 34–42. [Google Scholar] [CrossRef]
- Benkendorff, K.; McIver, C.M.; Abbott, C.A. Bioactivity of the murex homeopathic remedy and of extracts from an Australian muricid mollusc against human cancer cells. Evid.-Based Complement. Altern. 2011, 879585. [Google Scholar]
- Chou, S.; Spoo, W. Toxicological Profile for Chloroform; U.S. Department of Health and Human Services: Atlanta, GA, USA, 1997; pp. 1–291.
- Valianou, L.; Karapanagiotis, I.; Chryssoulakis, Y. Comparison of extraction methods for the analysis of natural dyes in historical textiles by high-performance liquid chromatography. Anal. Bioanal. Chem. 2009, 395, 2175–2189. [Google Scholar] [CrossRef] [PubMed]
- Suleria, H.A.R.; Gobe, G.; Masci, P.; Osborne, S.A. Marine bioactive compounds and health promoting perspectives; innovation pathways for drug discovery. Trends Food Sci. Technol. 2016, 50, 44–55. [Google Scholar] [CrossRef]
- Rudd, D.; Benkendorff, K. Supercritical CO2 extraction of bioactive Tyrian purple precursors from the hypobranchial gland of a marine gastropod. J. Supercrit. Fluids 2014, 94. [Google Scholar] [CrossRef]
- Ronci, M.; Rudd, D.; Guinan, T.; Benkendorff, K.; Voelcker, N.H. Mass spectrometry imaging on porous silicon: Investigating the distribution of bioactives in marine mollusc tissues. Anal. Chem. 2012, 84, 8996–9001. [Google Scholar] [CrossRef] [PubMed]
- Rudd, D.; Benkendorff, K.; Voelcker, N.H. Solvent separating secondary metabolites directly from biosynthetic tissue for surface-assisted laser desorption ionisation mass spectrometry. Mar. Drugs 2015, 13, 1410–1431. [Google Scholar] [CrossRef] [PubMed]
- Duke, C.; Eichholzer, J.; Macleod, J. The synthesis of the isomeric N-methyl derivatives of murexine. Aust. J. Chem. 1981, 34, 1739–1744. [Google Scholar] [CrossRef]
- Naegel, L.C.; Murillo Alvarez, J.I. Biological and chemical properties of the secretion from the hypobranchial gland of the purple snail Plicopurpura pansa (Gould, 1853). J. Shellfish Res. 2005, 24, 421–428. [Google Scholar]
- Fouquet, H.; Bielig, H.J. Biological precursors and genesis of Tyrian-purple. Angew. Chem. Int. Ed. Engl. 1971, 10, 816–817. [Google Scholar] [CrossRef]
- Pitt, J.J. Principles and applications of liquid chromatography-mass spectrometry in clinical biochemistry. Clin. Biochem. Rev. 2009, 30, 19–34. [Google Scholar] [PubMed]
- Baker, J.T.; Duke, C.C. Isolation of choline and choline ester salts of tyrindoxyl sulphate from the marine molluscs Dicathais orbita and Mancinella keineri. Tetrahedron Lett. 1976, 15, 1233–1234. [Google Scholar] [CrossRef]
- Baker, J.T.; Sutherland, M.D. Pigments of marine animals VIII. Precursors of 6,6′-dibromoindigotin (Tyrian purple) from the mollusc Dicathais orbita Gmelin. Tetrahedron Lett. 1968, 1, 43–46. [Google Scholar] [CrossRef]
- Christophersen, C.; Watjen, F.; Buchardt, O.; Anthoni, U. A revised structure of tyriverdin the precursor of Tyrian purple. Tetrahedron 1978, 34, 2779–2781. [Google Scholar] [CrossRef]
- Cooksey, C. Marine indirubins. In Indirubin, the Red Shade of Indigo; Meijer, L., Guyard, N., Skatsounis, L., Eisenbrand, G., Eds.; Progress in Life Series: Roscoff, France, 2006; pp. 23–30. [Google Scholar]
- Andreotti, A.; Bonaduce, I.; Colombini, M.P.; Ribechini, E. Characterisation of natural indigo and shellfish purple by mass spectrometric techniques. Rapid Commun. Mass Spectrom. 2004, 18, 1213–1220. [Google Scholar] [CrossRef] [PubMed]
- Corcoran, O.; Spraul, M. LC-NMR-MS in drug discovery. Drug Discov. Today 2003, 8, 624–631. [Google Scholar] [CrossRef]
- USA-Food and Drug Administration. Guidance for Industry Bioanalytical Method Validation; U.S. Department of Health and Human Services, Food and Drug Administration, Center for Drug Evaluation and Research, Center for Veterinary Medicine: Rockville, MD, USA, 2001.
- Armbruster, D.A.; Pry, T. Limit of blank, limit of detection and limit of quantitation. Clin. Biochem. Rev. 2008, 29, S49–S52. [Google Scholar] [PubMed]
- Alwarthan, A. Chemiluminescent determination of tryptophan in a flow injection system. Anal. Chim. Acta 1995, 317, 223–237. [Google Scholar] [CrossRef]
- Ngangbam, A.K.; Baten, A.; Waters, D.L.; Whalan, S.; Benkendorff, K. Characterization of bacterial communities associated with the Tyrian purple producing gland in a marine gastropod. PLoS ONE 2015, 10, e0140725. [Google Scholar] [CrossRef] [PubMed]
- Ngangbam, A.K.; Waters, D.L.; Whalan, S.; Baten, A.; Benkendorff, K. Indole-producing bacteria from the biosynthetic organs of a muricid mollusc could contribute to Tyrian purple production. J. Shellfish Res. 2015, 34, 443–454. [Google Scholar] [CrossRef]
- Lim, H.K.; Chung, E.J.; Kim, J.C.; Choi, G.J.; Jang, K.S.; Chung, Y.R.; Cho, K.Y.; Lee, S.W. Characterization of a forest soil metagenome clone that confers indirubin and indigo production on Escherichia coli. Appl. Environ. Microbiol. 2005, 17, 7768–7777. [Google Scholar] [CrossRef] [PubMed]
- Ong, E.S. Extraction methods and chemical standardization of botanicals and herbal preparations. J. Chromatogr. B 2004, 812, 22–33. [Google Scholar] [CrossRef]
- Rudd, D.; Ronci, M.; Johnston, M.R.; Guinan, T.; Voelcker, N.H.; Benkendorff, K. Mass spectrometry imaging reveals new biological roles for choline esters and Tyrian purple precursors in muricid molluscs. Sci. Rep. 2015, 5, 13408. [Google Scholar] [CrossRef] [PubMed]
- Sample Availability: Not available.
| Compound | δ (ppm) | Multiplicity | Relative Area | Assignment |
|---|---|---|---|---|
| 1. Murexine | 3.18 | s | 9 | 1 |
 | 3.62–3.68 | m | 2 | 2 |
| 4.53–4.68 | m | 2 | 3 | |
| 6.62 | d | 1 | 4 | |
| 7.68 | d | 1 | 5 | |
| 7.50 | s | 1 | 6 | |
| 7.92 | s | 1 | 7 | |
| 2. Tyrindoxyl sulphate | 2.50 | s | 3 | 1 |
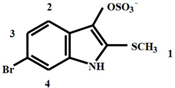 | 7.20 | dd | 1 | 2 |
| 7.55 | d | 1 | 3 | |
| 7.65 | d | 1 | 4 |
| % Yield | logP 1 | Methanol | Ethanol | Chloroform | |||
|---|---|---|---|---|---|---|---|
| 17.53 ± 3.14 %g/g | 12.20 ± 3.17 %g/g | 7.00 ± 3.52 %g/g | |||||
| Dry Extract (mg/mg) | Wet Tissue (mg/mg) | Dry Extract (mg/mg) | Wet Tissue (mg/mg) | Dry Extract (mg/mg) | Wet Tissue (mg/mg) | ||
| (1) Murexine | −3.37 | 0.431 ± 0.175 | 0.087 ± 0.023 | 0.468 ± 0.275 | 0.071 ± 0.024 | n.d. | n.d. |
| (2) Tyrindoxyl sulphate | −0.35 | 0.309 ± 0.124 | 0.062 ± 0.016 | 0.293 ± 0.167 | 0.044 ± 0.014 | n.d. | n.d. |
| (3) Tyrindoxyl S-oxide sulphate 2 | - | 0.005 ± 0.002 | 0.001 ± 0.001 | <LOD | <LOD | n.d. | n.d. |
| (5) Tyrindoleninone 3 | 2.89 | 0.004 ± 0.002 | 0.001 ± 0.001 | 0.003 ± 0.002 | 0.001 ± 0.000 | 0.018 ± 0.004 | 0.001 ± 0.001 |
| (6) 6-Bromoisatin | 1.62 | 0.006 ± 0.002 | 0.001 ± 0.000 | 0.005 ± 0.003 | 0.001 ± 0.000 | 0.044 ± 0.015 | 0.003 ± 0.001 |
| (8) Tyriverdin 3 | 4.67 | 0.004 ± 0.002 | 0.001 ± 0.000 | 0.003 ± 0.001 | >0.001 ± 0.000 | 0.035 ± 0.014 | 0.002 ± 0.000 |
| (9) 6,6′-Dibromoindigo 3 | 4.47 | n.d. | n.d. | n.d. | n.d. | 0.031 ± 0.017 | 0.002 ± 0.001 |
© 2016 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Valles-Regino, R.; Mouatt, P.; Rudd, D.; Yee, L.H.; Benkendorff, K. Extraction and Quantification of Bioactive Tyrian Purple Precursors: A Comparative and Validation Study from the Hypobranchial Gland of a Muricid Dicathais orbita. Molecules 2016, 21, 1672. https://doi.org/10.3390/molecules21121672
Valles-Regino R, Mouatt P, Rudd D, Yee LH, Benkendorff K. Extraction and Quantification of Bioactive Tyrian Purple Precursors: A Comparative and Validation Study from the Hypobranchial Gland of a Muricid Dicathais orbita. Molecules. 2016; 21(12):1672. https://doi.org/10.3390/molecules21121672
Chicago/Turabian StyleValles-Regino, Roselyn, Peter Mouatt, David Rudd, Lachlan H. Yee, and Kirsten Benkendorff. 2016. "Extraction and Quantification of Bioactive Tyrian Purple Precursors: A Comparative and Validation Study from the Hypobranchial Gland of a Muricid Dicathais orbita" Molecules 21, no. 12: 1672. https://doi.org/10.3390/molecules21121672
APA StyleValles-Regino, R., Mouatt, P., Rudd, D., Yee, L. H., & Benkendorff, K. (2016). Extraction and Quantification of Bioactive Tyrian Purple Precursors: A Comparative and Validation Study from the Hypobranchial Gland of a Muricid Dicathais orbita. Molecules, 21(12), 1672. https://doi.org/10.3390/molecules21121672