Molecular Mechanisms and Metabolomics of Natural Polyphenols Interfering with Breast Cancer Metastasis
Abstract
:1. Introduction
2. The Molecular Mechanism of Natural Polyphenols on Breast Cancer Metastasis In Vitro
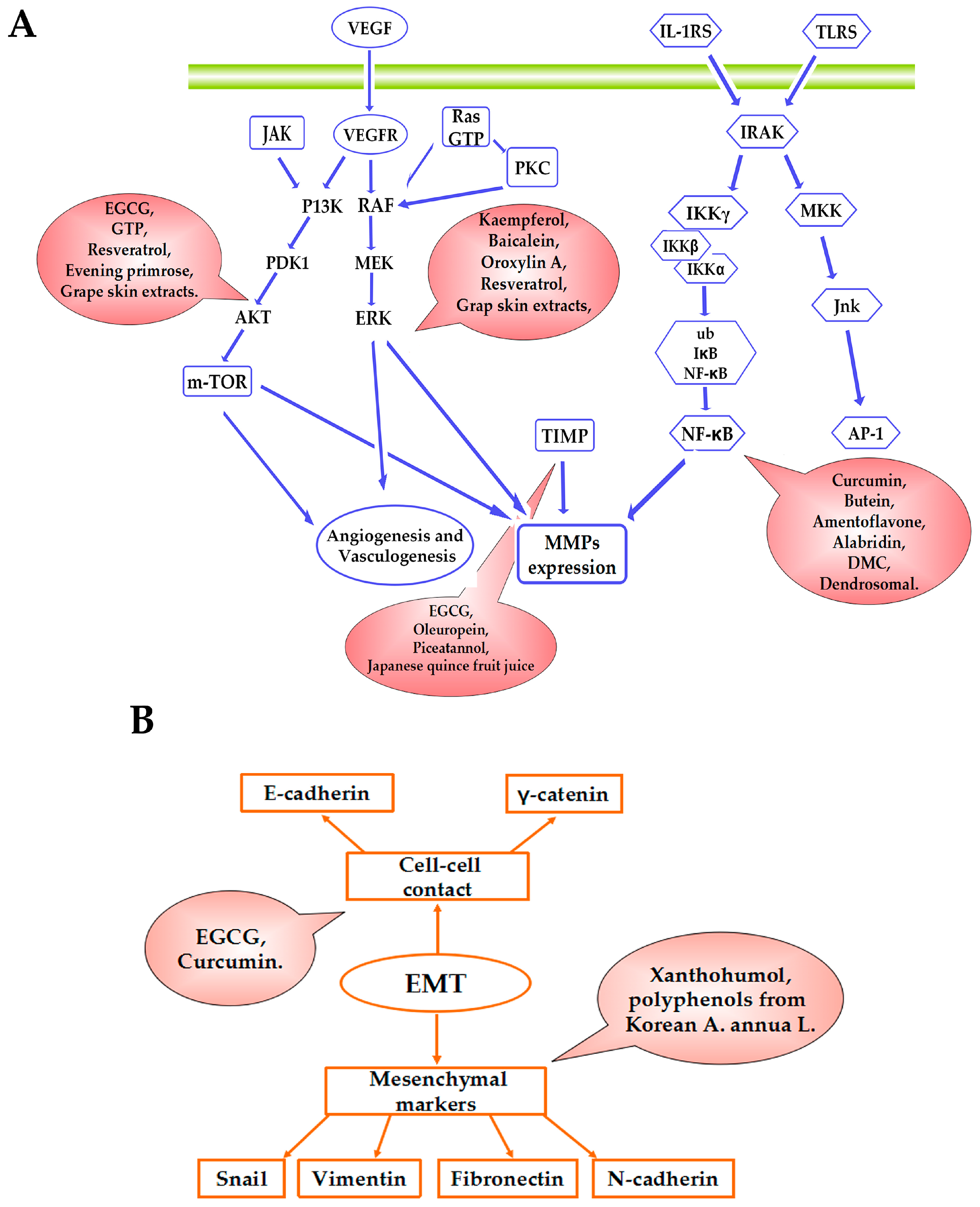
2.1. MMPs
2.1.1. MMPs Involving Tissue Inhibitor of Matrix Metalloproteinase
2.1.2. MMPs Involving Common Signaling Pathways
2.1.3. MMPs Involving NF-κB
2.2. Anti-Angiogenesis
2.3. NF-κB
2.4. EMT
2.5. mTOR
2.6. Others
3. The Molecular Mechanism of Natural Polyphenols on Breast Cancer Metastasis In Vivo
3.1. Natural Polyphenols Monomers
3.2. Combined Natural Polyphenols and Other Antimetastatic Drugs
3.3. Natural Polyphenols Extracts
4. Metabolomics
4.1. Natural Polyphenols
4.2. Polyphenolic Extracts
5. Conclusions and Future Perspectives
Acknowledgments
Conflicts of Interest
References
- Sang, S.; Lambert, J.D.; Ho, C.T.; Yang, C.S. The chemistry and biotransformation of tea constituents. Pharmacol. Res. 2011, 64, 87–99. [Google Scholar] [CrossRef] [PubMed]
- Manach, C.; Scalbert, A.; Morand, C.; Rémésy, C.; Jiménez, L. Polyphenols: Food sources and bioavailability. Am. J. Clin. Nutr. 2004, 79, 727–747. [Google Scholar] [PubMed]
- Lall, R.K.; Syed, D.N.; Adhami, V.M.; Khan, M.I.; Mukhtar, H. Dietary polyphenols in prevention and treatment of prostate cancer. Int. J. Mol. Sci. 2015, 16, 3350–3376. [Google Scholar] [CrossRef] [PubMed]
- Harbowy, M.E.; Balentine, D.A.; Dr., Davies, A.P.; Dr., Cai, Y. Critical reviews in plant sciences. Tea Chem. 1997, 16, 415–480. [Google Scholar]
- Chacko, S.M.; Thambi, P.T.; Kuttan, R.; Nishigaki, I. Beneficial effects of green tea: A literature review. Chin. Med. 2010, 5, 13. [Google Scholar] [CrossRef] [PubMed]
- Carocho, M.; Ferreira, I.C. A review on antioxidants, prooxidants and related controversy: Natural and synthetic compounds, screening and analysis methodologies and future perspectives. Food Chem. Toxicol. 2013, 51, 15–25. [Google Scholar] [CrossRef] [PubMed]
- Link, A.; Balaguer, F.; Goel, A. Cancer chemoprevention by dietary polyphenols: Promising role for epigenetics. Biochem. Pharmacol. 2010, 80, 1771–1792. [Google Scholar] [CrossRef] [PubMed]
- Zhou, Q.; Bennett, L.L.; Zhou, S. Multifaceted ability of naturally occurring polyphenols against metastatic cancer. Clin. Exp. Pharmacol. Physiol. 2016, 43, 394–409. [Google Scholar] [CrossRef] [PubMed]
- Tevaarwerk, A.J.; Gray, R.J.; Schneider, B.P.; Smith, M.L.; Wagner, L.I.; Fetting, J.H.; Davidson, N.; Goldstein, L.J.; Miller, K.D.; Sparano, J.A. Survival in patients with metastatic recurrent breast cancer after adjuvant chemotherapy. Cancer 2013, 119, 1140–1148. [Google Scholar] [CrossRef] [PubMed]
- American Cancer Society. Cancer Facts and Figures 2015; American Cancer Society: Atlanta, GA, USA, 2015. [Google Scholar]
- Massagué, J.; Obenauf, A.C. Metastatic colonization by circulating tumour cells. Nature 2016, 529, 298–306. [Google Scholar] [CrossRef] [PubMed]
- Kodack, D.; Askoxylakis, V.; Ferraro, G.; Dai, F.; Jain, R. Emerging strategies for treating brain metastases from breast cancer. Cancer Cell 2015, 27, 163–175. [Google Scholar] [CrossRef] [PubMed]
- Hess, K.R.; Varadhachary, G.R.; Taylor, S.H.; Wei, W.; Raber, M.N.; Lenzi, R.; Abbruzzese, J.L. Metastatic patterns in adenocarcinoma? Cancer 2006, 106, 1624–1633. [Google Scholar] [CrossRef] [PubMed]
- Bos, P.D.; Zhang, H.F.; Nadal, C.; Shu, W.; Gomis, R.R.; Nguyen, D.X.; Minn, A.J.; Vijver, M.J.V.D.; Gerald, W.L.; Foekens, J.A. Genes that mediate breast cancer metastasis to the brain. Nature 2009, 459, 1005–1009. [Google Scholar] [CrossRef] [PubMed]
- Wishart, D.S. Emerging applications of metabolomics in drug discovery and precision medicine. Nat. Rev. Drug Discov. 2016, 15, 484. [Google Scholar] [CrossRef] [PubMed]
- Delmas, D.; Xiao, J.B. Natural polyphenols properties: Chemopreventive and chemosensitizing activities. Anticancer Agents Med. Chem. 2012, 12, 835. [Google Scholar] [CrossRef] [PubMed]
- Steeg, P.S.; Theodorescu, D. Metastasis: A therapeutic target for cancer. Nat. Clin. Pract. Oncol. 2008, 5, 206–219. [Google Scholar] [CrossRef] [PubMed]
- Jiang, Y.L.; Liu, Z.P. Natural products as anti-invasive and anti-metastatic agents. Curr. Med. Chem. 2011, 18, 808–829. [Google Scholar] [CrossRef] [PubMed]
- Loffek, S.; Schilling, O.; Franzke, C.W. Biological role of matrix metalloproteinases: A critical balance. Eur. Respir. J. 2011, 38, 191–208. [Google Scholar] [CrossRef] [PubMed]
- Zitka, O.; Kukacka, J.; Krizkova, S.; Huska, D.; Adam, V.; Masarik, M.; Prusa, R.; Kizek, R. Matrix metalloproteinases. Curr. Med. Chem. 2010, 17, 3751–3768. [Google Scholar] [CrossRef] [PubMed]
- Gialeli, C.; Theocharis, A.D.; Karamanos, N.K. Roles of matrix metalloproteinases in cancer progression and their pharmacological targeting. FEBS J. 2011, 278, 16–27. [Google Scholar] [CrossRef] [PubMed]
- Ling, H.; Yang, H.; Tan, S.H.; Chui, W.K.; Chew, E.H. 6-Shogaol, an active constituent of ginger, inhibits breast cancer cell invasion by reducing matrix metalloproteinase-9 expression via blockade of nuclear factor-kappa b activation. Br. J. Pharmacol. 2010, 161, 1763–1777. [Google Scholar] [CrossRef] [PubMed]
- Ibaragi, S.; Shimo, T.; Iwamoto, M.; Hassan, N.M.M.; Kodama, S.; Isowa, S.; Sasaki, A. Parathyroid hormone-related peptide regulates matrix metalloproteinase-13 gene expression in bone metastatic breast cancer cells. Anticancer Res. 2012, 30, 5029–5036. [Google Scholar]
- Bahar, M.; Khaghani, S.; Pasalar, P.; Paknejad, M.; Khorramizadeh, M.R.; Mirmiranpour, H.; Nejad, S.G. Exogenous coenzyme q10 modulates MMP-2 activity in MCF-7 cell line as a breast cancer cellular model. Nutr. J. 2010, 9, 2131–2132. [Google Scholar] [CrossRef] [PubMed]
- Mylona, E.; Magkou, C.; Giannopoulou, I.; Agrogiannis, G.; Markaki, S.; Keramopoulos, A.; Nakopoulou, L. Expression of tissue inhibitor of matrix metalloproteinases (TIMP)-3 protein in invasive breast carcinoma: Relation to tumor phenotype and clinical outcome. Breast Cancer Res. 2006, 8, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Deb, G.; Thakur, V.S.; Limaye, A.M.; Gupta, S. Epigenetic induction of tissue inhibitor of matrix metalloproteinase-3 by green tea polyphenols in breast cancer cells. Mol. Carcinog. 2015, 54, 485–499. [Google Scholar] [CrossRef] [PubMed]
- Hassan, Z.K.; Elamin, M.H.; Daghestani, M.H.; Omer, S.A.; Al-Olayan, E.M.; Elobeid, M.A.; Virk, P.; Mohammed, O.B. Oleuropein induces anti-metastatic effects in breast cancer. Asian Pac. J. Cancer Prev. 2012, 13, 4555–4559. [Google Scholar] [CrossRef] [PubMed]
- Song, H.; Jung, J.I.; Cho, H.J.; Her, S.; Kwon, S.H.; Yu, R.; Kang, Y.H.; Lee, K.W.; Park, J.H. Inhibition of tumor progression by oral piceatannol in mouse 4T1 mammary cancer is associated with decreased angiogenesis and macrophage infiltration. J. Nutr. Biochem. 2015, 26, 1368–1378. [Google Scholar] [CrossRef] [PubMed]
- Lewandowska, U.; Szewczyk, K.; Owczarek, K.; Hrabec, Z.; Podsedek, A.; Koziolkiewicz, M.; Hrabec, E. Flavanols from japanese quince (Chaenomeles japonica) fruit inhibit human prostate and breast cancer cell line invasiveness and cause favorable changes in Bax/Bcl-2 mRNA ratio. Nutr. Cancer 2013, 65, 273–285. [Google Scholar] [CrossRef] [PubMed]
- Kushima, Y.; Iida, K.; Nagaoka, Y.; Kawaratani, Y.; Shirahama, T.; Sakaguchi, M.; Baba, K.; Hara, Y.; Uesato, S. Inhibitory effect of (−)-epigallocatechin and (−)-epigallocatechin gallate against heregulin beta 1-induced migration/invasion of the MCF-7 breast carcinoma cell line. Biol. Pharm. Bull. 2009, 32, 899–904. [Google Scholar] [CrossRef] [PubMed]
- Belguise, K.; Guo, S.; Sonenshein, G.E. Activation of FOXO3a by the green tea polyphenol epigallocatechin-3-gallate induces estrogen receptor alpha expression reversing invasive phenotype of breast cancer cells. Cancer Res. 2007, 67, 5763–5770. [Google Scholar] [CrossRef] [PubMed]
- Mineva, N.D.; Paulson, K.E.; Naber, S.P.; Yee, A.S.; Sonenshein, G.E. Epigallocatechin-3-gallate inhibits stem-like inflammatory breast cancer cells. PLoS ONE 2013, 8, e73464. [Google Scholar] [CrossRef] [PubMed]
- Thangapazham, R.L.; Passi, N.; Maheshwari, R.K. Green tea polyphenol and epigallocatechin gallate induce apoptosis and inhibit invasion in human breast cancer cells. Cancer Biol. Ther. 2014, 6, 1938–1943. [Google Scholar] [CrossRef]
- Gunther, S.; Ruhe, C.; Derikito, M.G.; Bose, G.; Sauer, H.; Wartenberg, M. Polyphenols prevent cell shedding from mouse mammary cancer spheroids and inhibit cancer cell invasion in confrontation cultures derived from embryonic stem cells. Cancer Lett. 2007, 250, 25–35. [Google Scholar] [CrossRef] [PubMed]
- Li, C.; Zhao, Y.; Yang, D.; Yu, Y.; Guo, H.; Zhao, Z.; Zhang, B.; Yin, X. Inhibitory effects of kaempferol on the invasion of human breast carcinoma cells by downregulating the expression and activity of matrix metalloproteinase-9. Biochem. Cell. Biol. 2015, 93, 16–27. [Google Scholar] [CrossRef] [PubMed]
- Rivera Rivera, A.; Castillo-Pichardo, L.; Gerena, Y.; Dharmawardhane, S. Anti-breast cancer potential of quercetin via the Akt/AMPK/mammalian target of rapamycin (mTOR) signaling cascade. PLoS ONE 2016, 11, e0157251. [Google Scholar] [CrossRef] [PubMed]
- Sun, Y.; Lu, N.; Ling, Y.; Gao, Y.; Chen, Y.; Wang, L.; Hu, R.; Qi, Q.; Liu, W.; Yang, Y.; et al. Oroxylin a suppresses invasion through down-regulating the expression of matrix metalloproteinase-2/9 in MDA-MB-435 human breast cancer cells. Eur. J. Pharmacol. 2009, 603, 22–28. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Ling, Y.; Chen, Y.; Li, C.L.; Feng, F.; You, Q.D.; Lu, N.; Guo, Q.L. Flavonoid baicalein suppresses adhesion, migration and invasion of MDA-MB-231 human breast cancer cells. Cancer Lett. 2010, 297, 42–48. [Google Scholar] [CrossRef] [PubMed]
- Vantyghem, S.A.; Wilson, S.M.; Postenka, C.O.; Al-Katib, W.; Tuck, A.B.; Chambers, A.F. Dietary genistein reduces metastasis in a postsurgical orthotopic breast cancer model. Cancer Res. 2005, 65, 3396–3403. [Google Scholar] [PubMed]
- Lee, H.S.; Ha, A.W.; Kim, W.K. Effect of resveratrol on the metastasis of 4T1 mouse breast cancer cells in vitro and in vivo. Nutr. Res. Pract. 2012, 6, 294–300. [Google Scholar] [CrossRef] [PubMed]
- Tang, F.Y.; Chiang, E.P.; Sun, Y.C. Resveratrol inhibits heregulin-β1-mediated matrix metalloproteinase-9 expression and cell invasion in human breast cancer cells. J. Nutr. Biochem. 2008, 19, 287–294. [Google Scholar] [CrossRef] [PubMed]
- Tang, F.Y.; Su, Y.C.; Chen, N.C.; Hsieh, H.S.; Chen, K.S. Resveratrol inhibits migration and invasion of human breast-cancer cells. Mol. Nutr. Food Res. 2008, 52, 683–691. [Google Scholar] [CrossRef] [PubMed]
- Lee-Chang, C.; Bodogai, M.; Martin-Montalvo, A.; Wejksza, K.; Sanghvi, M.; Moaddel, R.; de Cabo, R.; Biragyn, A. Inhibition of breast cancer metastasis by resveratrol-mediated inactivation of tumor-evoked regulatory b cells. J. Immunol. 2013, 191, 4141–4151. [Google Scholar] [CrossRef] [PubMed]
- Castillo-Pichardo, L.; Cubano, L.A.; Dharmawardhane, S. Dietary grape polyphenol resveratrol increases mammary tumor growth and metastasis in immunocompromised mice. BMC Complement. Altern. Med. 2013, 13, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Chua, A.W.; Hay, H.S.; Rajendran, P.; Shanmugam, M.K.; Li, F.; Bist, P.; Koay, E.S.; Lim, L.H.; Kumar, A.P.; Sethi, G. Butein downregulates chemokine receptor CXCR4 expression and function through suppression of NF-κb activation in breast and pancreatic tumor cells. Biochem. Pharmacol. 2010, 80, 1553–1562. [Google Scholar] [CrossRef] [PubMed]
- Viola, K.; Kopf, S.; Rarova, L.; Jarukamjorn, K.; Kretschy, N.; Teichmann, M.; Vonach, C.; Atanasov, A.G.; Giessrigl, B.; Huttary, N.; et al. Xanthohumol attenuates tumour cell-mediated breaching of the lymphendothelial barrier and prevents intravasation and metastasis. Arch. Toxicol. 2013, 87, 1301–1312. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.M.; Noh, E.M.; Kwon, K.B.; Kim, J.S.; You, Y.O.; Hwang, J.K.; Hwang, B.M.; Kim, B.S.; Lee, S.H.; Lee, S.J.; et al. Curcumin suppresses the TPA-induced invasion through inhibition of PKCα-dependent MMP-expression in MCF-7 human breast cancer cells. Phytomedicine 2012, 19, 1085–1092. [Google Scholar] [CrossRef] [PubMed]
- Farhangi, B.; Alizadeh, A.M.; Khodayari, H.; Khodayari, S.; Dehghan, M.J.; Khori, V.; Heidarzadeh, A.; Khaniki, M.; Sadeghiezadeh, M.; Najafi, F. Protective effects of dendrosomal curcumin on an animal metastatic breast tumor. Eur. J. Pharmacol. 2015, 758, 188–196. [Google Scholar] [CrossRef] [PubMed]
- Kronski, E.; Fiori, M.E.; Barbieri, O.; Astigiano, S.; Mirisola, V.; Killian, P.H.; Bruno, A.; Pagani, A.; Rovera, F.; Pfeffer, U.; et al. miR181b is induced by the chemopreventive polyphenol curcumin and inhibits breast cancer metastasis via down-regulation of the inflammatory cytokines CXCL1 and -2. Mol. Oncol. 2014, 8, 581–595. [Google Scholar] [CrossRef] [PubMed]
- Bachmeier, B.; Nerlich, A.G.; Iancu, C.M.; Cilli, M.; Schleicher, E.; Vené, R.; Dell'Eva, R.; Jochum, M.; Albini, A.; Pfeffer, U. The chemopreventive polyphenol curcumin prevents hematogenous breast cancer metastases in immunodeficient mice. Cell. Physiol. Biochem. 2007, 19, 137–152. [Google Scholar] [CrossRef] [PubMed]
- Gallardo, M.; Calaf, G.M. Curcumin inhibits invasive capabilities through epithelial mesenchymal transition in breast cancer cell lines. Int. J. Oncol. 2016, 49, 1019–1027. [Google Scholar] [CrossRef] [PubMed]
- Yodkeeree, S.; Ampasavate, C.; Sung, B.; Aggarwal, B.B.; Limtrakul, P. Demethoxycurcumin suppresses migration and invasion of MDA-MB-231 human breast cancer cell line. Eur. J. Pharmacol. 2009, 627, 8–15. [Google Scholar] [CrossRef] [PubMed]
- Sepporta, M.V.; Fuccelli, R.; Rosignoli, P.; Ricci, G.; Servili, M.; Morozzi, G.; Fabiani, R. Oleuropein inhibits tumour growth and metastases dissemination in ovariectomised nude mice with MCF-7 human breast tumour xenografts. J. Funct. Foods 2014, 8, 269–273. [Google Scholar] [CrossRef]
- Zheng, X.; Ke, Y.; Feng, A.; Yuan, P.; Zhou, J.; Yu, Y.; Wang, X.; Feng, W. The mechanism by which amentoflavone improves insulin resistance in HepG2 cells. Molecules 2016, 21. [Google Scholar] [CrossRef] [PubMed]
- Mu, J.; Ning, S.; Wang, X.; Si, L.; Jiang, F.; Li, Y.; Li, Z. The repressive effect of miR-520a on NF-κB/IL-6/STAT-3 signal involved in the glabridin-induced anti-angiogenesis in human breast cancer cells. RSC Adv. 2015, 5, 34257–34264. [Google Scholar] [CrossRef]
- Shibata, M.A.; Akao, Y.; Shibata, E.; Nozawa, Y.; Ito, T.; Mishima, S.; Morimoto, J.; Otsuki, Y. Vaticanol C, a novel resveratrol tetramer, reduces lymph node and lung metastases of mouse mammary carcinoma carrying p53 mutation. Cancer Chemother. Pharmacol. 2007, 60, 681–691. [Google Scholar] [CrossRef] [PubMed]
- Xiong, X.Y.; Hu, X.J.; Li, Y.; Liu, C.M. Inhibitory effects of enterolactone on growth and metastasis in human breast cancer. Nutr. Cancer 2015, 67, 1324–1332. [Google Scholar] [CrossRef] [PubMed]
- Liu, L.; Sun, L.; Wu, Q.; Guo, W.; Li, L.; Chen, Y.; Li, Y.; Gong, C.; Qian, Z.; Wei, Y. Curcumin loaded polymeric micelles inhibit breast tumor growth and spontaneous pulmonary metastasis. Int. J. Pharm. 2013, 443, 175–182. [Google Scholar] [CrossRef] [PubMed]
- Castillo-Pichardo, L.; Dharmawardhane, S.F. Grape polyphenols inhibit Akt/mammalian target of rapamycin signaling and potentiate the effects of gefitinib in breast cancer. Nutr. Cancer 2012, 64, 1058–1069. [Google Scholar] [CrossRef] [PubMed]
- Castillo-Pichardo, L.; Martinez-Montemayor, M.M.; Martinez, J.E.; Wall, K.M.; Cubano, L.A.; Dharmawardhane, S. Inhibition of mammary tumor growth and metastases to bone and liver by dietary grape polyphenols. Clin. Exp. Metastasis 2009, 26, 505–516. [Google Scholar] [CrossRef] [PubMed]
- Luo, K.W.; Ko, C.H.; Yue, G.G.; Lee, J.K.; Li, K.K.; Lee, M.; Li, G.; Fung, K.P.; Leung, P.C.; Lau, C.B. Green tea (Camellia sinensis) extract inhibits both the metastasis and osteolytic components of mammary cancer 4T1 lesions in mice. J. Nutr. Biochem. 2014, 25, 395–403. [Google Scholar] [CrossRef] [PubMed]
- Baliga, M.S.; Meleth, S.; Kadiyar, S.K. Growth inhibitory and antimetastatic effect of green tea polyphenols on metastasis-specific mouse mammary carcinoma 4T1 cells in vitro and in vivo systems. Clin. Cancer Res. 2005, 11, 1918–1927. [Google Scholar] [CrossRef] [PubMed]
- Yang, X.; Belosay, A.; Hartman, J.A.; Song, H.; Zhang, Y.; Wang, W.; Doerge, D.R.; Helferich, W.G. Dietary soy isoflavones increase metastasis to lungs in an experimental model of breast cancer with bone micro-tumors. Clin. Exp. Metastasis 2015, 32, 323–333. [Google Scholar] [CrossRef] [PubMed]
- Chang, C.H.; Ou, T.T.; Yang, M.Y.; Huang, C.C.; Wang, C.J. Nelumbo nucifera gaertn leaves extract inhibits the angiogenesis and metastasis of breast cancer cells by downregulation connective tissue growth factor (CTGF) mediated PI3K/AKT/ERK signaling. J. Ethnopharmacol. 2016, 188, 111–122. [Google Scholar] [CrossRef] [PubMed]
- Manoj, G.S.; Kumar, T.R.S.; Varghese, S.; Murugan, K. Effect of methanolic and water extract of Leucobryum bowringii Mitt. On growth, migration and invasion of MCF 7 human breast cancer cells in vitro. Ind. J. Exp. Biol. 2012, 50, 602–611. [Google Scholar]
- Noratto, G.; Porter, W.; Byrne, D.; Cisneros-Zevallos, L. Polyphenolics from peach (Prunus persica var. Rich lady) inhibit tumor growth and metastasis of MDA-MB-435 breast cancer cells in vivo. J. Nutr. Biochem. 2014, 25, 796–800. [Google Scholar] [CrossRef] [PubMed]
- Mileo, A.M.; di Venere, D.; Linsalata, V.; Fraioli, R.; Miccadei, S. Artichoke polyphenols induce apoptosis and decrease the invasive potential of the human breast cancer cell line MDA-MB231. J. Cell Physiol. 2012, 227, 3301–3309. [Google Scholar] [CrossRef] [PubMed]
- Sun, T.; Chen, Q.Y.; Wu, L.J.; Yao, X.M.; Sun, X.J. Antitumor and antimetastatic activities of grape skin polyphenols in a murine model of breast cancer. Food Chem. Toxicol. 2012, 50, 3462–3467. [Google Scholar] [CrossRef] [PubMed]
- Lewandowska, U.; Szewczyk, K.; Owczarek, K.; Hrabec, Z.; Podsedek, A.; Sosnowska, D.; Hrabec, E. Procyanidins from evening primrose (Oenothera paradoxa) defatted seeds inhibit invasiveness of breast cancer cells and modulate the expression of selected genes involved in angiogenesis, metastasis, and apoptosis. Nutr. Cancer 2013, 65, 1219–1231. [Google Scholar] [CrossRef] [PubMed]
- Yeap, S.K.; Abu, N.; Mohamad, N.E.; Beh, B.K.; Ho, W.Y.; Ebrahimi, S.; Yusof, H.M.; Ky, H.; Tan, S.W.; Alitheen, N.B. Chemopreventive and immunomodulatory effects of Murraya koenigii aqueous extract on 4T1 breast cancer cell-challenged mice. BMC Complement. Altern. Med. 2015, 15, 306. [Google Scholar] [CrossRef] [PubMed]
- Mantena, S.K.; Baliga, M.S.; Katiyar, S.K. Grape seed proanthocyanidins induce apoptosis and inhibit metastasis of highly metastatic breast carcinoma cells. Carcinogenesis 2006, 27, 1682–1691. [Google Scholar] [CrossRef] [PubMed]
- Vuong, T.; Mallet, J.F.; Ouzounova, M.; Rahbar, S.; Hernandez-Vargas, H.; Herceg, Z.; Matar, C. Role of a polyphenol-enriched preparation on chemoprevention of mammary carcinoma through cancer stem cells and inflammatory pathways modulation. J. Transl. Med. 2016, 14, 13. [Google Scholar] [CrossRef] [PubMed]
- Ko, Y.S.; Lee, W.S.; Panchanathan, R.; Joo, Y.N.; Choi, Y.H.; Kim, G.S.; Jung, J.M.; Ryu, C.H.; Shin, S.C.; Kim, H.J. Polyphenols from artemisia annua l inhibit adhesion and emt of highly metastatic breast cancer cells MDA-MB-231. Phytother. Res. 2016, 30, 1180–1188. [Google Scholar] [CrossRef] [PubMed]
- Dihlmann, S.; Kloor, M.; Fallsehr, C.; Doeberitz, M.V.K. Regulation of AKT1 expression by β-catenin/Tcf/Lef signaling in colorectal cancer cells. Carcinogenesis 2005, 26, 1503–1512. [Google Scholar] [CrossRef] [PubMed]
- So, F.V.; Guthrie, N.; Chambers, A.F.; Carroll, K.K. Inhibition of proliferation of estrogen receptor-positive MCF-7 human breast cancer cells by flavonoids in the presence and absence of excess estrogen. Cancer Lett. 1997, 112, 127–133. [Google Scholar] [CrossRef]
- Bork, P. The modular architecture of a new family of growth regulators related to connective tissue growth factor. FEBS Lett. 1993, 327, 125–130. [Google Scholar] [CrossRef]
- Chang, C.C.; Shih, J.Y.; Jeng, Y.M.; Su, J.L.; Lin, B.Z.; Chen, S.T.; Chau, Y.P.; Yang, P.C.; Kuo, M.L. Connective tissue growth factor and its role in lung adenocarcinoma invasion and metastasis. J. Natl. Cancer Inst. 2004, 96, 364–375. [Google Scholar] [CrossRef] [PubMed]
- Lin, B.R. Connective tissue growth factor inhibits metastasis and acts as an independent prognostic marker in colorectal cancer. Gastroenterology 2005, 128, 9–23. [Google Scholar] [CrossRef] [PubMed]
- Lin, M.C.; Kao, S.H.; Chung, P.J.; Chan, K.C.; Yang, M.Y.; Wang, C.J. Improvement for high fat diet-induced hepatic injuries and oxidative stress by flavonoid-enriched extract from nelumbo nucifera leaf. J. Agric. Food Chem. 2009, 57, 5825–5932. [Google Scholar] [CrossRef] [PubMed]
- O’Dea, E.; Hoffmann, A. NF-κb signaling. Wiley Interdiscip. Rev. Syst. Bio. Med. 2009, 1, 107–115. [Google Scholar] [CrossRef] [PubMed]
- Huber, M.A.; Azoitei, N.; Baumann, B.; Grünert, S.; Sommer, A.; Pehamberger, H.; Kraut, N.; Beug, H.; Wirth, T.; Huber, M.A.; et al. NF-κb is essential for epithelial-mesenchymal transition and metastasis in a model of breast cancer progression. J. Clin. Investig. 2004, 114, 569–584. [Google Scholar] [CrossRef] [PubMed]
- Zunling, L.; Yanxia, G.; Hanming, J.; Tingguo, Z.; Changzhu, J.; Young, C.Y.; Huiqing, Y. Differential regulation of MMPS by E2F1, Sp1 and NF-κb controls the small cell lung cancer invasive phenotype. BMC Cancer 2014, 14, 276. [Google Scholar]
- Aggarwal, B.B. Nuclear factor-κB: The enemy within. Cancer Cell 2004, 6, 203–208. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.S.; Guan, J.; Yang, F.Q.; Liu, H.G.; Cheng, X.J.; Li, S.P. Qualitative and quantitative analysis of four species of curcuma rhizomes using twice development thin layer chromatography. J. Pharm. Biomed. Anal. 2008, 48, 1024–1028. [Google Scholar] [CrossRef] [PubMed]
- Fidler, I.J. Angiogenesis and cancer metastasis. Cancer J. 2000, 6 (Suppl. S2), 388–390. [Google Scholar]
- Song, M.Q.; Ramaswamy, S.; Ramachandran, S.; Flowers, L.C.; Horowitz, I.R.; Rock, J.A.; Parthasarathy, S. Angiogenic role for glycodelin in tumorigenesis. Proc. Natl. Acad. Sci. USA 2001, 98, 9265–9270. [Google Scholar] [CrossRef] [PubMed]
- Viale, G.; Regan, M.M.; Mastropasqua, M.G.; Maffini, F.; Maiorano, E.; Colleoni, M.; Price, K.N.; Golouh, R.; Perin, T.; Brown, R.W.; et al. Predictive value of tumor Ki-67 expression in two randomized trials of adjuvant chemoendocrine therapy for node-negative breast cancer. J. Natl. Cancer Inst. 2008, 100, 207–212. [Google Scholar] [CrossRef] [PubMed]
- Park, E.H.; Park, J.Y.; Yoo, H.S.; Yoo, J.E.; Lee, H.L. Assessment of the anti-metastatic properties of sanguiin H-6 in huvecs and MDA-MB-231 human breast cancer cells. Bioorg. Med. Chem. Lett. 2016, 26, 3291–3294. [Google Scholar] [CrossRef] [PubMed]
- Li, F.; Zhang, J.; Arfuso, F.; Chinnathambi, A.; Zayed, M.E.; Alharbi, S.A.; Kumar, A.P.; Ahn, K.S.; Sethi, G. NF-κb in cancer therapy. Arch. Toxicol. 2015, 89, 711–731. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.H.; Chen, W.L.; Liu, Y.C. Amentoflavone induces anti-angiogenic and anti-metastatic effects through suppression of NF-κb activation in MCF-7 cells. Anticancer Res. 2015, 35, 6685–6693. [Google Scholar] [PubMed]
- Pandey, P.; Singh, S.; Tewari, N.; Srinivas, K.V.N.S.; Shukla, A.; Gupta, N.; Vasudev, P.G.; Khan, F.; Pal, A.; Bhakuni, R.S. Hairy root mediated functional derivatization of artemisinin and their bioactivity analysis. J. Mol. Catal. B Enzym. 2015, 113, 95–103. [Google Scholar] [CrossRef]
- Shen, L.; Sundstedt, A.; Ciesielski, M.; Miles, K.M.; Celander, M.; Adelaiye, R.; Orillion, A.; Ciamporcero, E.; Ramakrishnan, S.; Ellis, L. Tasquinimod modulates suppressive myeloid cells and enhances cancer immunotherapies in murine models. Cancer Immunol. Res. 2015, 3, 136–148. [Google Scholar] [CrossRef] [PubMed]
- Sen, R.; Baltimore, D. Inducibility of κ immunoglobulin enhancer-binding protein NF-κb by a posttranslational mechanism. Cell 1987, 47, 921–928. [Google Scholar] [CrossRef]
- Cicek, M.; Oursler, M.J. Breast cancer bone metastasis and current small therapeutics. Cancer Metastasis Rev. 2006, 25, 635–644. [Google Scholar] [CrossRef] [PubMed]
- Karin, M.; Cao, Y.; Greten, F.R.; Li, Z.W. NF-κb in cancer: From innocent bystander to major culprit. Nat. Rev. Cancer 2002, 2, 301–310. [Google Scholar] [CrossRef] [PubMed]
- Ghosh, S.; Karin, M. Missing pieces in the NF-κb puzzle. Cell 2002, 109, S81–96. [Google Scholar] [CrossRef]
- Dhamija, S.; Diederichs, S. From junk to master regulators of invasion: lncRNA functions in migration, EMT and metastasis. Int. J. Cancer 2015, 139, 269–280. [Google Scholar] [CrossRef] [PubMed]
- Couse, J.; Korach, K. Estrogen receptor null mice: What have we learned and where will they lead us? Endocr. Rev. 1999, 20, 358–417. [Google Scholar] [CrossRef] [PubMed]
- Wu, X.; Sun, L.; Xiao, W.; Peng, S.; Li, Z.; Zhang, C.; Yan, W.; Peng, G.; Rong, M. Breast cancer invasion and metastasis by mPRα through the PI3K/Akt signaling pathway. Pathol. Oncol. Res. 2016, 22, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Tashiro, E.; Henmi, S.; Odake, H.; Ino, S.; Imoto, M. Involvement of the MEK/ERK pathway in egf-induced e-cadherin down-regulation. Biochem. Biophys. Res. Commun. 2016, 477, 801–806. [Google Scholar] [CrossRef] [PubMed]
- Gallardo, M.; Calaf, G.M. Curcumin and epithelial-mesenchymal transition in breast cancer cells transformed by low doses of radiation and estrogen. Int. J. Oncol. 2016, 48, 2534–2542. [Google Scholar] [CrossRef] [PubMed]
- Magalhães, P.J.; Carvalho, D.O.; Cruz, J.M.; Guido, L.F.; Barros, A.A. Fundamentals and health benefits of xanthohumol, a natural product derived from hops and beer. Nat. Prod. Commun. 2009, 4, 591–610. [Google Scholar] [PubMed]
- Lindstedt, S.; Hansson, J.; Hlebowicz, J. The effect of negative wound pressure therapy on haemodynamics in a laparostomy wound model. Int. Wound J. 2013, 10, 285–290. [Google Scholar] [CrossRef] [PubMed]
- Zhou, H.; Huang, S. Mtor signaling in cancer cell motility and tumor metastasis. Crit. Rev. Eukaryot. Gene Expr. 2010, 20, 1–16. [Google Scholar] [CrossRef] [PubMed]
- Walker, E.H.; Pacold, M.E.; Perisic, O.; Stephens, L.; Hawkins, P.T.; Wymann, M.P.; Williams, R.L. Structural determinants of phosphoinositide 3-kinase inhibition by wortmannin, LY294002, quercetin, myricetin, and staurosporine. Mol. Cell 2000, 6, 909–919. [Google Scholar] [CrossRef]
- Weixel, K.M.; Marciszyn, A.; Alzamora, R.; Li, H.; Fischer, O.; Edinger, R.S.; Hallows, K.R.; Johnson, J.P. Resveratrol inhibits the epithelial sodium channel via phopshoinositides and AMP-activated protein kinase in kidney collecting duct cells. PLoS ONE 1932, 8, e78019. [Google Scholar] [CrossRef] [PubMed]
- Jagetia, G.C.; Aggarwal, B.B. “Spicing up” Of the immune system by curcumin. J. Clin. Immunol. 2007, 27, 19–35. [Google Scholar] [CrossRef] [PubMed]
- Safavy, A.; Raisch, K.P.; Mantena, S.; Sanford, L.L.; Sham, S.W.; Krishna, N.R.; Bonner, J.A. Design and development of water-soluble curcumin conjugates as potential anticancer agents. J. Med. Chem. 2007, 50, 6284–6288. [Google Scholar] [CrossRef] [PubMed]
- Anand, P.; Kunnumakkara, A.B.; Newman, R.A.; Aggarwal, B.B. Bioavailability of curcumin: Problems and promises. Mol. Pharm. 2007, 4, 807–818. [Google Scholar] [CrossRef] [PubMed]
- Shang, W.; Lu, W.; Han, M.; Qiao, J. The interactions of anticancer agents with tea catechins: Current evidence from preclinical studies. Anticancer Agents Med. Chem. 2014, 14, 1343–1350. [Google Scholar] [CrossRef] [PubMed]
- Somers-Edgar, T.J.; Scandlyn, M.J.; Stuart, E.C.; Nedelec, M.J.L.; Valentine, S.P.; Rosengren, R.J. The combination of epigallocatechin gallate and curcumin suppresses erα-breast cancer cell growth in vitro and in vivo. Int. J. Cancer 2008, 122, 1966–1971. [Google Scholar] [CrossRef] [PubMed]
- Luo, T.; Wang, J.; Yin, Y.; Hua, H.; Jing, J.; Sun, X.; Li, M.; Zhang, Y.; Jiang, Y. (−)-epigallocatechin gallate sensitizes breast cancer cells to paclitaxel in a murine model of breast carcinoma. Breast Cancer Res. 2010, 12, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Kang, H.J.; Lee, S.H.; Price, J.E.; Kim, L.S. Curcumin suppresses the paclitaxel-induced nuclear factor-κb in breast cancer cells and potentiates the growth inhibitory effect of paclitaxel in a breast cancer nude mice model. Breast J. 2009, 15, 223–229. [Google Scholar] [CrossRef] [PubMed]
- Tomás-Barberán, F.A.; Gil, M.I.; Cremin, P.; Waterhouse, A.L.; Hess-Pierce, B.; Kader, A.A. HPLC-DAD-ESIMS analysis of phenolic compounds in nectarines, peaches, and plums. J. Agric. Food Chem. 2001, 49, 4748–4760. [Google Scholar] [CrossRef] [PubMed]
- Puchades-Carrasco, L.; Pineda-Lucena, A. Metabolomics in pharmaceutical research and development. Curr. Opin. Biotechnol. 2015, 35, 73–77. [Google Scholar] [CrossRef] [PubMed]
- Spencer, J.; Abd, E.M.M.; Minihane, A. Metabolism of dietary phytochemicals: A review of the metabolic forms identified in humans. Curr. Top. Nutraceutical. Res. 2006, 4, 187–203. [Google Scholar]
- Felgines, C.; Texier, O.; Besson, C.; Lyan, B. Strawberry pelargonidin glycosides are excreted in urine as intact glycosides and glucuronidated pelargonidin derivatives in rats. Br. J. Nutr. 2007, 98, 1126–1131. [Google Scholar] [CrossRef] [PubMed]
- Denkert, C.; Budczies, J.; Kind, T.; Weichert, W.; Tablack, P.; Sehouli, J.; Niesporek, S.; Konsgen, D.; Dietel, M.; Fiehn, O. Mass spectrometry-based metabolic profiling reveals different metabolite patterns in invasive ovarian carcinomas and ovarian borderline tumors. Cancer Res. 2006, 66, 10795–10804. [Google Scholar] [CrossRef] [PubMed]
- Yi, L.; Dong, N.; Shao, L.; Yi, Z.; Yi, Z. Chemical features of pericarpium citri reticulatae and pericarpium citri reticulatae viride revealed by GC-MS metabolomics analysis. Food Chem. 2014, 186, 192–199. [Google Scholar] [CrossRef] [PubMed]
- Fardet, A.; Llorach, R.; Martin, J.F.; Besson, C.; Lyan, B.; Pujosguillot, E.; Scalbert, A. A liquid chromatography-quadrupole time-of-flight (LC-QTOF)-based metabolomic approach reveals new metabolic effects of catechin in rats fed high-fat diets. J. Proteome Res. 2008, 7, 2388–2398. [Google Scholar] [CrossRef] [PubMed]
- Kusano, M.; Yang, Z.G.; Okazaki, Y.; Nakabayashi, R.; Fukushima, A.; Saito, K. Using metabolomic approaches to explore chemical diversity in rice. Mol. Plant 2015, 8, 58–67. [Google Scholar] [CrossRef] [PubMed]
- Solanky, K.S.; Bailey, N.J.; Holmes, E.; Lindon, J.C.; Davis, A.L.; Mulder, T.P.; Van Duynhoven, J.P.; Nicholson, J.K. NMR-based metabonomic studies on the biochemical effects of epicatechin in the rat. J. Agric. Food Chem. 2003, 51, 4139–4145. [Google Scholar] [CrossRef] [PubMed]
- Van Dorsten, F.A.; Daykin, C.A.; Mulder, T.P.; van Duynhoven, J.P. Metabonomics approach to determine metabolic differences between green tea and black tea consumption. J. Agri. Food Chem. 2006, 54, 6929–6938. [Google Scholar] [CrossRef] [PubMed]
- Hodgson, A.B.; Randell, R.K.; Mahabir-Jagessar-T, K.; Lotito, S.; Mulder, T.; Mela, D.J.; Jeukendrup, A.E.; Jacobs, D.M. Acute effects of green tea extract intake on exogenous and endogenous metabolites in human plasma. J. Agri. Food Chem. 2014, 62, 1198–1208. [Google Scholar] [CrossRef] [PubMed]
- Xie, G.; Zhao, A.; Zhao, L.; Chen, T.; Chen, H.; Qi, X.; Zheng, X.; Ni, Y.; Cheng, Y.; Lan, K. Metabolic fate of tea polyphenols in humans. J. Proteome. Res. 2012, 11, 3449–3457. [Google Scholar] [CrossRef] [PubMed]
- Van Dorsten, F.A.; Grün, C.H.; van Velzen, E.J.J.; Jacobs, D.M.; Draijer, R.; van Duynhoven, J.P.M. The metabolic fate of red wine and grape juice polyphenols in humans assessed by metabolomics. Mol. Nutr. Food Res. 2010, 54, 897–908. [Google Scholar] [CrossRef] [PubMed]
- Dudley, E.; Lemiere, F.; Dongen, W.V.; Langridge, J.I.; El-Sharkawi, S.; Games, D.E.; Esmans, E.L.; Newton, R.P. Analysis of urinary nucleosides. III. Identification of 5′-Deoxycytidine in urine of a patient with head and neck cancer. Rapid Commun. Mass Spectrom. 2003, 17, 1132–1136. [Google Scholar] [CrossRef] [PubMed]
- Schram, K.H. Urinary nucleosides &dagger. Mass Spectrom. 1998, 17, 131–251. [Google Scholar]
- Fenyves, D.; Pomier-Layrargues, G.; Willems, B.; Côté, J. Intrahepatic pressure measurement: Not an accurate reflection of portal vein pressure. Hepatology 1988, 8, 211–216. [Google Scholar] [CrossRef] [PubMed]
- Peduto, A.; Baumgartner, M.R.; Verhoeven, N.M.; Rabier, D.; Spada, M.; Nassogne, M.C.; Poll-The, B.T.T.; Bonetti, G.; Jakobs, C.; Saudubray, J.M. Hyperpipecolic acidaemia: A diagnostic tool for peroxisomal disorders. Mol. Genet. Metab. 2004, 82, 224–230. [Google Scholar] [CrossRef] [PubMed]
- Bollard, M.E.; Holmes, E.; Lindon, J.C.; Mitchell, S.C.; Branstetter, D.; Zhang, W.; Nicholson, J.K. Investigations into biochemical changes due to diurnal variation and estrus cycle in female rats using high-resolution 1 h NMR spectroscopy of urine and pattern recognition. Anal. Biochem. 2001, 295, 194–202. [Google Scholar] [CrossRef] [PubMed]
- Thielecke, F.; Boschmann, M. The potential role of green tea catechins in the prevention of the metabolic syndrome—A review. Chemlnform 2009, 70, 11–24. [Google Scholar] [CrossRef] [PubMed]
- Fujiki, H.; Sueoka, E.; Watanabe, T.; Suganuma, M. Synergistic enhancement of anticancer effects on numerous human cancer cell lines treated with the combination of egcg, other green tea catechins, and anticancer compounds. J. Cancer Res. Clin. Oncol. 2015, 141, 1511–1522. [Google Scholar] [CrossRef] [PubMed]
- Zapf, M.A.C.; Kothari, A.N.; Weber, C.E.; Arffa, M.L.; Wai, P.Y.; Driver, J.; Gupta, G.N.; Kuo, P.C.; Mi, Z.Y. Green tea component epigallocatechin-3-gallate decreases expression of osteopontin via a decrease in mrna half-life in cell lines of metastatic hepatocellular carcinoma. Surgery 2015, 158, 1039–1047. [Google Scholar] [CrossRef] [PubMed]
- Wang, D.; Xiao, R.; Hu, X.; Xu, K.; Hou, Y.; Zhong, Y.; Meng, J.; Fan, B.; Liu, L. Comparative safety evaluation of Chinese Pu-erh green tea extract and Pu-erh black tea extract in wistar rats. J. Agric. Food Chem. 2010, 58, 1350–1358. [Google Scholar] [CrossRef] [PubMed]
- Zhao, X.; Qian, Y.; Zhou, Y.L.; Wang, R.; Wang, Q.; Li, G.J. Pu-erh tea has in vitro anticancer activity in TCA8113 cells and preventive effects on buccal mucosa cancer in U14 cells injected mice in vivo. Nutr. Cancer 2014, 66, 1059–1069. [Google Scholar] [CrossRef] [PubMed]
- Jacobs, D.M.; Fuhrmann, J.C.; van Dorsten, F.A.; Rein, D.; Peters, S.; van Velzen, E.J.; Hollebrands, B.; Draijer, R.; van, D.J.; Garczarek, U. Impact of short-term intake of red wine and grape polyphenol extract on the human metabolome. J. Agric. Food Chem. 2012, 60, 3078–3085. [Google Scholar] [CrossRef] [PubMed]
- Johnson, R.H. Incidence of breast cancer with distant involvement among women in the United States, 1976–2009. JAMA 2013, 309, 1229. [Google Scholar] [CrossRef] [PubMed]
- Lin, N.U.; Carey, L.A.; Liu, M.C.; Younger, J.; Come, S.E.; Ewend, M.; Harris, G.J.; Bullitt, E.; Ad, V.D.A.; Henson, J.W. Phase II trial of lapatinib for brain metastases in patients with human epidermal growth factor receptor 2-positive breast cancer. J. Clin. Oncol. 2008, 26, 1993–1999. [Google Scholar] [CrossRef] [PubMed]
- Ekenel, M.; Hormigo, A.M.; Peak, S.; Deangelis, L.M.; Abrey, L.E. Capecitabine therapy of central nervous system metastases from breast cancer. J. Neurooncol. 2007, 85, 223–227. [Google Scholar] [CrossRef] [PubMed]
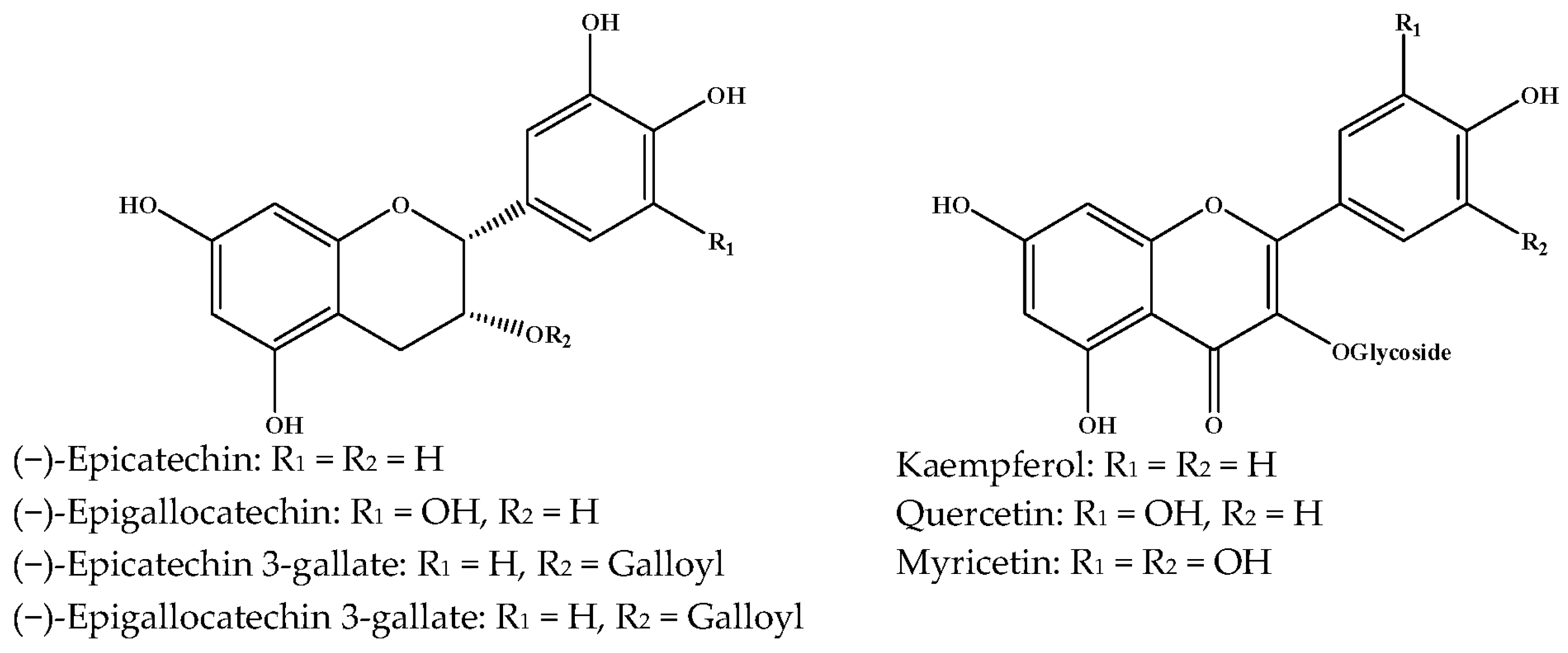

| Natural Polyphenols | Cancer Type or Animal Type | Effective Concentrations or Doses | Result | Ref. |
|---|---|---|---|---|
| (−)-Epigallocatechin Gallate (EGCG) 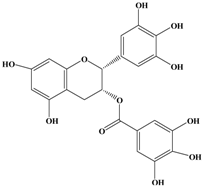 | MCF-7 | In vitro: 30 μM | Suppressing the Heregulin β1-stimulated activation of epidermal growth factor receptor-related protein B2 (ErbB2)/ErbB3/protein kinase B. | [30] |
| Mouse mammary cancer virus-Her-2/neu cell line NF639 | In vitro: 60 μg/mL | Up-regulation of the epithelial genes E-cadherin, γ-catenin, MTA3, and estrogen receptor α (ERα). Down-regulation of proinvasive snail gene. Activation of FOXO3a. | [31] | |
| Inflammatory Breast Cancer lines: SUM-149 and SUM-190 | In vitro: 5–160 μg/mL | Elevation of the levels of cleaved Caspase-3 and PARP. | [32] | |
| MCF-7 MDA-MB-231 | In vitro: 20 μM | Reducing EZH2 and class I HDAC protein levels, inducing TIMP-3 levels,suppressing invasiveness and activity of MMP-2 and MMP-9. | [26] | |
(−)-Epigallocatechin (EGC)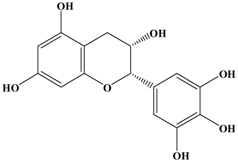 | MDA-MB-231 human breast cancer cell line | In vitro: 50, 80 μg/mL | Inhibition of MMP-9 expression and AKT signaling pathway by inhibiting both at the RNA and protein level. | [33] |
| MCF-7 | In vitro: 30 μM | Disruption of the Heregulin β1-stimulated activation of ErbB2/ErbB3/Akt. | [30] | |
(−)-Epicatechin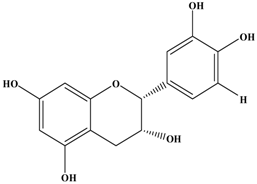 | Murine mammacarcinoma cell line 4T1 | In vitro: 10 μM | Inhibiting cell shedding and invasion by their anti-oxidative capacity and down-regulation of MMP-9 expression. | [34] |
Kaempferol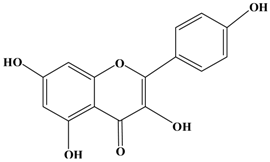 | MDA-MB-231 human breast carcinoma cells | In vitro: 10, 20, 40 μM | Blocking the PKCδ/MAPK/AP-1 cascade and subsequentiy suppressing MMP-9 expression. | [35] |
Quercetin | Metastatic MDA-MB-231 MDA-MB-435 Female severe combined immunodeficiency (SCID) mice | In vitro: 15 μM In vivo: 15 mg/kg 3X per week i.p. | Induction of mTOR activities through Akt inhibition and AMPK activation. | [36] |
Oroxylin A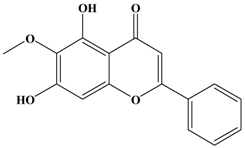 | MDA-MB-435 | In vitro: 1, 10 and 100 μM | Inhibiting the expressions of MMP-2, MMP-9 and ERK1/2 | [37] |
Baicalein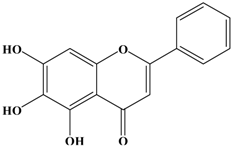 | MDA-MB-231 | In vitro: 2, 10, 50 μM | Down-regulating the expression of MMP-2/9 involved MAPK signaling pathway. | [38] |
Genistein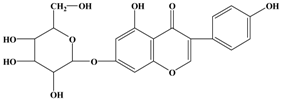 | MDA-MB-435/HAL | In vivo: 750 μg/g | Reducing the percent metastatic burden in the lungs, and affect the outgrowth of seeded tumor cells by dietary intervention following cancer surgery. | [39] |
Resveratrol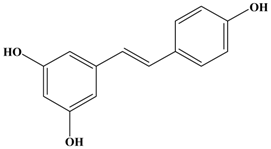 | 4T1 Female BALB/c mice | In vitro: 0–30 μM In vivo: 100 mg/kg/day 200 mg/kg/day | Decreasing the activity and expression of MMP-9. | [40] |
| MCF-7 | In vitro: 2, 5 and 10 μM | Inhibition of HRG-β1-mediated MMP-9 expression via down-regulation of the MAPK/ERK signaling pathway. | [41] | |
| MDA-MB-435 | In vitro: 10 and 20 μM | Suppressing insulin-like growth factor (IGF)-1-mediated cell migration and invasion and MMP-2 expression via inhibition of the PI3K/Akt signaling pathway. | [42] | |
| Female BALB/c mice (4T1) | In vivo: 50 mg/mouse/2day i.p. | Inactivating Stat3, preventing the generation and function of tBregs, including expression of TGF-β. | [43] | |
| Immunocompromised mice Green fluorescent protein (GFP) tagged-MDA-MB-231 (ERα(-), ERβ(+)) or a metastatic variant of GFP-MDA-MB-435 (ER (-)) cells | In vivo: 0.5, 5 and 50 mg/mouse 5 days/week i.g. | Increased expression of the Rac downstream effector PAK1, JNK and Akt. | [44] | |
Piceatannol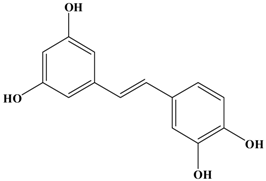 | Female BALB/c mice (4T1) | In vivo: 10 and 20 mg/kg/day i.g. In vitro: 30 μM | Reducing the expression of MMP-9 in both cancer and lung tissues and increasing apoptotic cells and expression of both Bax and cleaved caspase-3 but reducing Bcl-2 expression in cancer tissues. | [28] |
Butein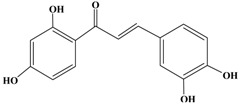 | HER2-over-expressing breast cancer SKBr3 cells | In vitro: 10,25,50 μM | Inhibition of CXCR4 expression correlated with the suppression of CXCL12- induced migration and invasion, and inhibiting of NF-κB activation. | [45] |
Xanthohumol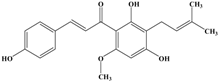 | MCF-7 MDA-MB-231 | In vitro: MCF-7: 5 μmol/L MDA-MB-231: 50 μmol/L | Inhibiting the activity of CYP, SELE and NF-κB, affecting ICAM-1 expression and adherence to LECs, suppressing paxillin, MCL2 and S100A4. | [46] |
Curcumin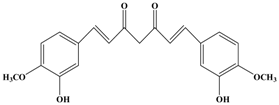 | MCF-7 | In vitro: 30 μM | Suppression of the PKCα, MAPK and NF-κB/AP-1 pathway and TPA-induced MMP-9 expression | [47] |
| 4T1 BALB/c mice | In vitro: 5–20 μM In vivo: 40 mg/kg 80 mg/kg | Suppression of NF-κB expression by down-regulation of VEGF, COX-2, and MMP-9 expressions. | [48] | |
| MDA-MB-231 MDA-MB-231MOCK injecting into the left cardiac ventricle CD-1 Foxn1nu female mice. | In vitro: 25 μM | Up-regulation of miR181b and down-regulation of CXCL1 and 2. | [49] | |
| MDA-MB-231 MDA-MB-231MOCK injecting into the left cardiac ventricle CD-1 Foxn1nu female mice. | In vitro: 25 μM In vivo: feding with standard diet containing 1% curcumin | Reducing the expression of MMPs via down-regulation of NF-κB and AP-1 activity and transcriptional. | [50] | |
| MCF-10F; MDA-Mb-231; Tumor 2 | In vitro: 30 µM for 48 h | Decreasing E-cadherin, N-cadherin, β-catenin, Slug, AXL, Twist1, Vimentin and Fibronectin protein expression involved in EMT. | [51] | |
Demethoxycurcumin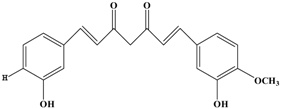 | MDA-MB-231 | In vitro: 1, 7.5, 15 μM | Inhibiting the DNA binding activity of NF-κB, decreasing the levels of ECM degradation-associated proteins including MMP-9, MT1-MMP, uPA , uPAR, ICAM-1 and CXCR4, up-regulating the level of PAI-1. | [52] |
Oleuropein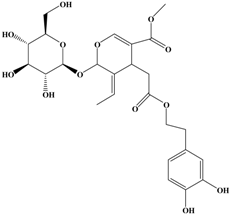 | MDA-cell line | In vitro: 200 μg/mL | Increasing the TIMPs, and then suppressing the MMPs expressions | [27] |
| Ovariectomised nude mice MCF-7 | In vivo: 125 mg/kg diet | Possessing a potent in vivo anti-cancer activity inhibiting both the MCF-7 cells xenograft growth and their invasiveness into the lung. | [53] | |
Amentoflavone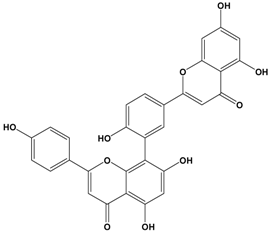 | MCF-7 | In vivo: 50 and 100 μM in 0.1% DMSO | Inhibiting NF-κB activation decreases expression and secretion of angiogenesis- and metastasis-related proteins. Amentoflavone may induce anti-angiogenic and anti-metastatic effects through suppression of NF-κB activation. | [54] |
Glabridin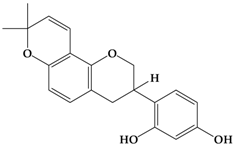 | MDA-Mb-231 | In vitro: 8 μM for 24, 48, 72 h | Attenuating the angiogenic ability by the microRNA-520a (miR-520a)-mediated inhibition of the NF-κB/IL-6/STAT-3 signal pathway. | [55] |
| A resveratrol tetramer | BJMC3879 Female BALB/c mice | In vitro: 8 μM In vivo: 22.9 mg/kg/day 44.7 mg/kg/day i.p. | Modulating the transcription level of mutant p53. Suppressing metastasis to both lymph nodes and lungs. | [56] |
| Enterolactone, an active polyphenol metabolite of Lignan | MDA-MB-231 cells | In vitro: IC50 = 261.9 ± 10.5 μM | Down-regulating phosphorylation of the FAK/paxillin pathway, inhibiting migration and invasion of cells. | [57] |
| Curcumin loaded polymeric micelles | Subcutaneous 4T1 breast cancer model | In vivo: 30 mg/kg/day | Inhibiting cancer growth and spontaneous pulmonary metastasis. | [58] |
| Grape natural polyphenols: resveratrol,quercetin,catechin,(or combination with gefitinib) | Human metastatic breast cancer cell GFP-MDA-MB-231 (ERα(−), ERβ(+)) Immunocompromised mice | In vitro: 5 μM In vivo: 5 mg/kg/day respectively i.g. or 5 mg/kg/2days and 200 mg/kg/2days i.g. | Reducing Akt activity, inducing the activation of AMPK and inhibiting mTOR signaling pathway. | [59] |
| ER(−) GFP-MDA-MB-435 Female athymic nu/nu mice | In vitro: 0.5 or 5 μM In vivo: 5 mg/kg/day together i.g | Up-regulating FOXO1 and NFKBIA (IκBα), activating apoptosis and inhibiting NF-κB activity, reducing metastasis. | [60] | |
| Mixed natural polyphenols resveratrol, baicalein, epicatechin, epigallocatechin polyphenon 60 | 4T1 multicellular cancer spheroids | In vitro: 100 μM | Suppressing invasion by down-regulation of MMP-9 expression and their anti-oxidative capacity. | [34] |
| Natural polyphenols extract from Green tea | MCF-7 MDA-MB-231 | In vitro: 10 μg/mL | Reducing EZH2 and class I HDAC protein levels, inducing TIMP-3 mRNA and protein levels, suppressing invasiveness and activity of MMP-2 and MMP-9. | [26] |
| Mouse mammary carcinoma 4T1 cells Female BALB/c mice | In vitro: 0.06–0.125 mg/mL In vivo: 0.6 g/kg/day i.g. | Increasing the expression of Bax-to-Bcl-2 ratio, activating caspase-8 and caspase-3, decreasing lung and liver metastasis, protecting the bone from breast cancer-induced bone destruction. | [61] | |
| Mouse mammary carcinoma 4T1 cells Female BALB/c mice | In vivo: 0.2% and 0.5% w/v in drinking water and was started 7 days before cancer cells inoculation | Decreasing the protein expression of Bcl-2 concomitantly increase in Bax, cytochrome c release, Apaf-1, and cleavage of caspase 3 and PARP proteins,inhibiting lungs metastasis. | [62] | |
| Natural polyphenols extract from Green tea | MDA-MB-231 human breast cancer cell line | In vitro: 40, 60 μg/mL | Inhibition of MMP-9 expression and AKT signaling pathway by inhibiting both at the RNA and protein level. | [33] |
| Natural polyphenols extract from Soy isoflavones | Female BALB/c mice (4T1) | In vivo: 750 mg/kg/day diet genistein equivalent | Increasing Ki-67 protein expression amd stimulating metastatic cancer formation in lungs. | [63] |
| Natural polyphenols extract from Japanese quince fruit | MDA-MB-231 | In vitro: 25, 50, 75, 100 μM catechin equivalents | Decreasing the MMP-9 activity and stimulating the TIMP-1 expression. | [29] |
| Natural polyphenols extract from Nelumbo nucifera Gaertn leaves | MDA-MB-231 | In vitro: 0.5~2.0 mg/mL | Blocking vascular-like structure formation, suppressing CTGF expression reducing the MMP2 and VEGF expression, and attenuating PI3K-AKT-ERK activation. | [64] |
| Natural polyphenols extract from Leucobryum bowringii Mitt. | MCF-7 | In vitro: 10, 25 and 50 μg/mL | Inhibition of MMP-2 and MMP-9 activities. | [65] |
| Natural polyphenols extract from Peach phenolics | MDA-MB-435 | In vivo: 0.8–1.6 mg chlorogenic acid equivalent /day i.g. | Down-regulating the gene expression of MMPs, and up-regulating hβ2G gene expression in the lungs. | [66] |
| Natural polyphenols extract from Artichoke | MDA-MB-231 | In vitro: 200 μM | Decreasing of proteolytic activity of MMP-2, involved in degrading components of the extracellular matrix. | [67] |
| Natural polyphenols extract from Grape skin | 4T1 cells Balb/c mice implanting 4T1 subcutaneously | In vitro: 0.5 and 1.0 mg/ml in drinking water | Blocking the PI3k/Akt and MAPK pathways. | [68] |
| Natural polyphenols extract from Evening primrose | MDA-MB-231 | In vitro: IC50 = 58 μM (gallic acid equivalents) | Decreasing the activity of MMP-9 through reducing the expression levels of the following proteins: VEGF, c-Fos, c-Jun. | [69] |
| Natural polyphenols extract from Murraya koenigii | MDA-MB-231 Left mammary fat pad subcutaneously 4T1 | In vitro: MDA-MB-231 (IC50 = 2.40 ± 0.26) 4T1 (IC50 = 1.50 ± 0.90) In vivo: 50 mg/kg 200 mg/kg | Decreasing the level of nitric oxide and inflammation-related cytokines and genes, including iNOS, iCAM, NF-κB and c-MYC and reducing lung metastasis. | [70] |
| Natural polyphenols extract from Grape seed proanthocyanidins | 4T1 cells were implanted subcutaneously in Balb/c mice | In vivo: 0.2% and 0.5%, w/w in a diet | Increasing the ratio of Bax:Bcl-2 proteins, cytochrome c release, induction of Apaf-1 and activation of caspase 3, inhibiting the metastasis of cancer cells to the lungs. | [71] |
| Natural polyphenols extract from biotransformation of blueberry juice by Serratia vaccinii | murine 4T1 human MCF7 and MDA-MB-231 BALB/c mouse model | In vitro: 100 μM (gallic acid equivalent) In vivo: 2.9 mL/day | Decreasing lung metastasis by controlling PI3K/AKT, MAPK/ERK. | [72] |
| Natural polyphenols extract from Korean A. annua L. | MDA-MB-231 | In vitro: 1, 10, 30 ug/mL | Inhibiting the cancer cell adhesion to ECs through suppression of vascular cell adhesion molecule-1 and invasion through suppression of EMT, MMP-2 and MMP-9. | [73] |
| Intervention | Subjects (Samples) | Analytical-Technique | Modified Endogenous Metabolites | Biological Hypotheses | Ref. |
|---|---|---|---|---|---|
| Animal study: normolipidemic (5% w/w) or hyperlipidemic (15 and 25%) diets with or without catechin supplementation (0.2% w/w). | Male Wistar rats (urine) | LC-QTOF | ↑ Pipecolinic acid ↑ Nicotinic acid ↑ Dihydroxyquinoline ↑ Deoxycytidine | Possible inhibition of microbiota growth by catechin. Chronic liver dysfunction or peroxisomal disorders and increase in DNA breakdown. | [120] |
| Animal study: a single dose of 22 mg of epicatechin | 220–270 g male Sprague-Dawley (SD) rats (urine) | 1H-NMR | ↓ Taurine ↓ Creatinine ↓ Dimethylamine ↓ 2-Oxoglutarate ↓ Citrate. | Modification in carbohydrate metabolism; Changes in liver and kidney functions. | [122] |
| Human study: Consumption of green tea (6 g/day), black tea (6 g/day) or caffeine (control) for 2 days | 17 nonsmoking male (urine and plasma) | 1H-NMR | ↑ Succinate ↑ Oxaloacetate ↑ 2-oxoglutarate | Stimulation of oxidative energy metabolism | [123] |
| Human study: a single dose (acute) of GTE or placebo (PLA) and following 1 day, 7 days, GTE (2 × 559 mg catechins/day, 120 mg caffeine/day), or PLA supplementation | (age 22 ± 5 year, weight 78 ± 10.6 kg) 39 healthy physically active male (plasma) | HPLC-MRM-MS | ↑ Caffeine ↑ Taurine ↑ 3,4-Dihydroxyphenyle thylene glycol ↓ Hippurate ↑Salicylate ↑ Fatty acids ↑ Serotonin ↑ Triglycerides ↓ Cholesterylesters and ↑ Sphingosines | Influencing the changes in lipid metabolism and vascular function. | [124] |
| Human study: a dose equivalent to 5 cups of commercially prepared tea. | Range 22–32 years healthy men and women (urine) | UPLC-QTOFMS and GC-TOFMS | ↑ ornithine ↑ valine ↑ tyrosine ↑ 2-methylguanosine ↑ 2-aminobutyric acid ↓ urea | Pu-erh tea metabolites | [125] |
| Human study: MIX and the GJX supplements was 800 mg gallic acid equivalents (GAEs) per day for 4 weeks. | (Age: 18–70 years) 33 men and 25 women | 1H-NMR GC-MS | ↑ Nitric oxide ↑ Phenylacetylglutamine ↑ 4-hydroxymandelic acid ↑ Vanillylmandelic acid ↑ Homovanillic acid Urine:↑ Hippuric acid | Promoting vascular endothelial function, indicator of gut microbiota-mediated degradation, benefiting the neurological or cardiovascular health. | [126] |
© 2016 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ci, Y.; Qiao, J.; Han, M. Molecular Mechanisms and Metabolomics of Natural Polyphenols Interfering with Breast Cancer Metastasis. Molecules 2016, 21, 1634. https://doi.org/10.3390/molecules21121634
Ci Y, Qiao J, Han M. Molecular Mechanisms and Metabolomics of Natural Polyphenols Interfering with Breast Cancer Metastasis. Molecules. 2016; 21(12):1634. https://doi.org/10.3390/molecules21121634
Chicago/Turabian StyleCi, Yingqian, Jinping Qiao, and Mei Han. 2016. "Molecular Mechanisms and Metabolomics of Natural Polyphenols Interfering with Breast Cancer Metastasis" Molecules 21, no. 12: 1634. https://doi.org/10.3390/molecules21121634
APA StyleCi, Y., Qiao, J., & Han, M. (2016). Molecular Mechanisms and Metabolomics of Natural Polyphenols Interfering with Breast Cancer Metastasis. Molecules, 21(12), 1634. https://doi.org/10.3390/molecules21121634





