Cytotoxic, Antitumor and Immunomodulatory Effects of the Water-Soluble Polysaccharides from Lotus (Nelumbo nucifera Gaertn.) Seeds
Abstract
:1. Introduction
2. Results
2.1. Extraction of Lotus Seed Polysaccharides
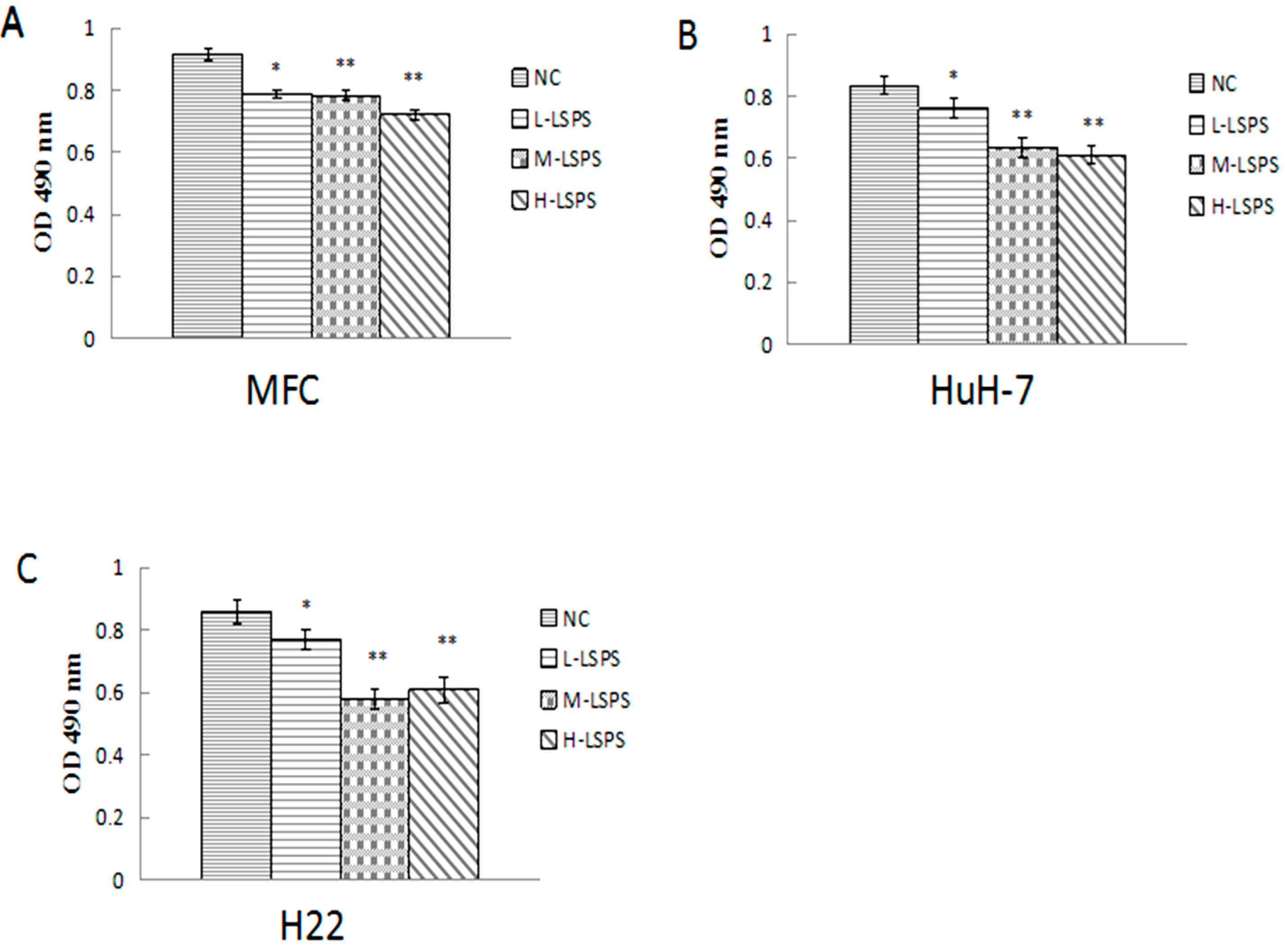
2.2. Cytotoxic Effects on MFC, HuH-7 and H22 Cells
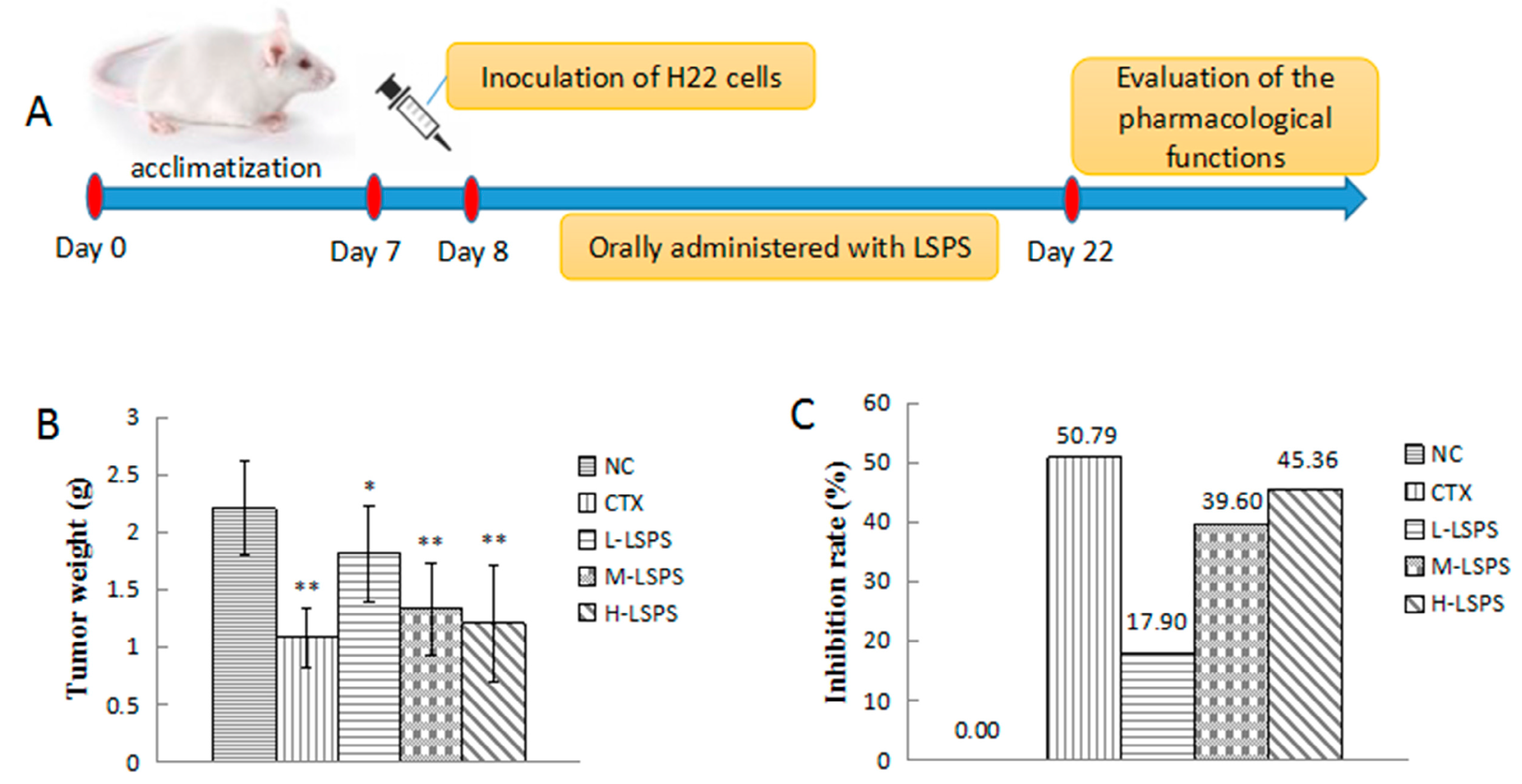
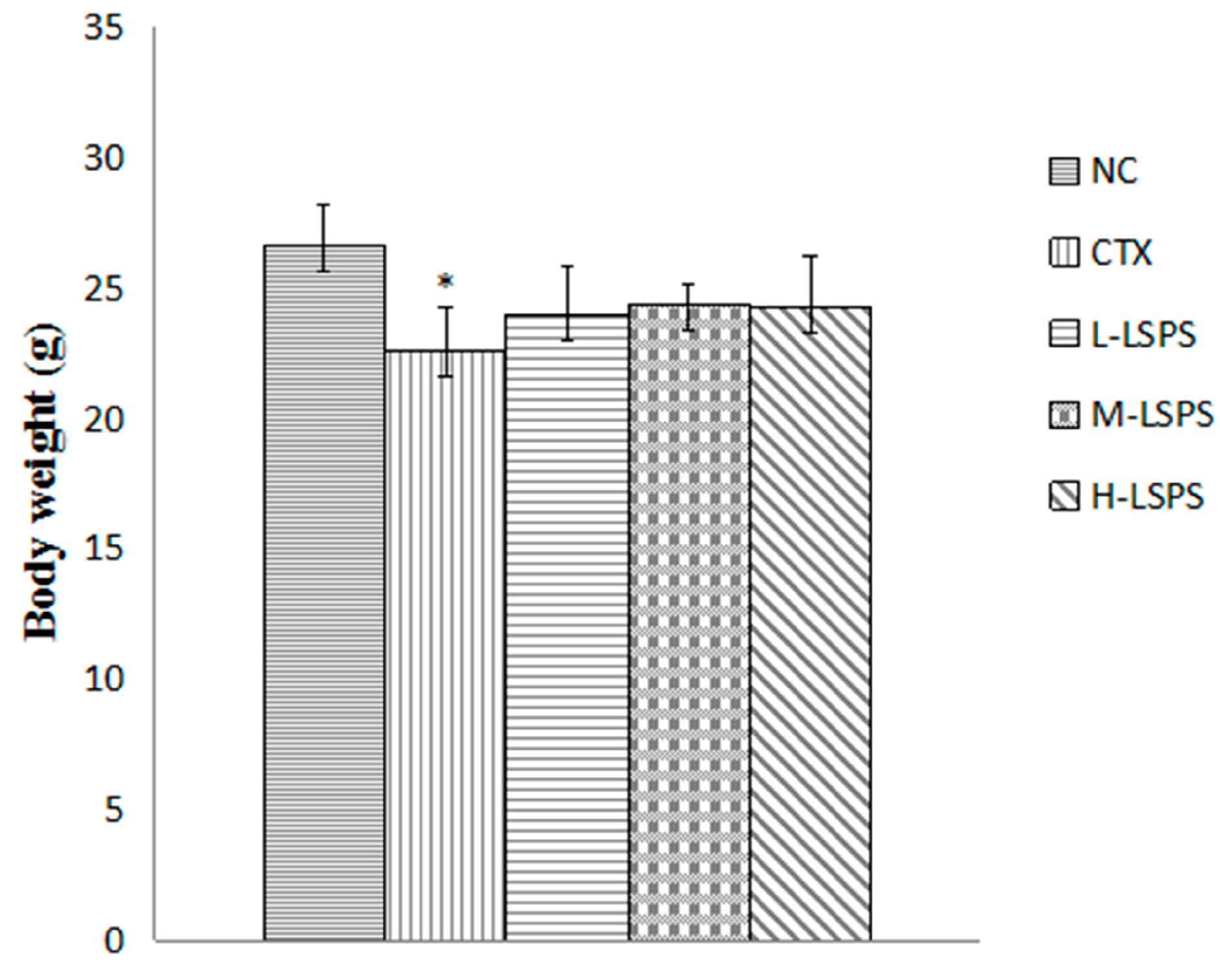
2.3. Effect of LSPS on Tumor Growth and Body Weight
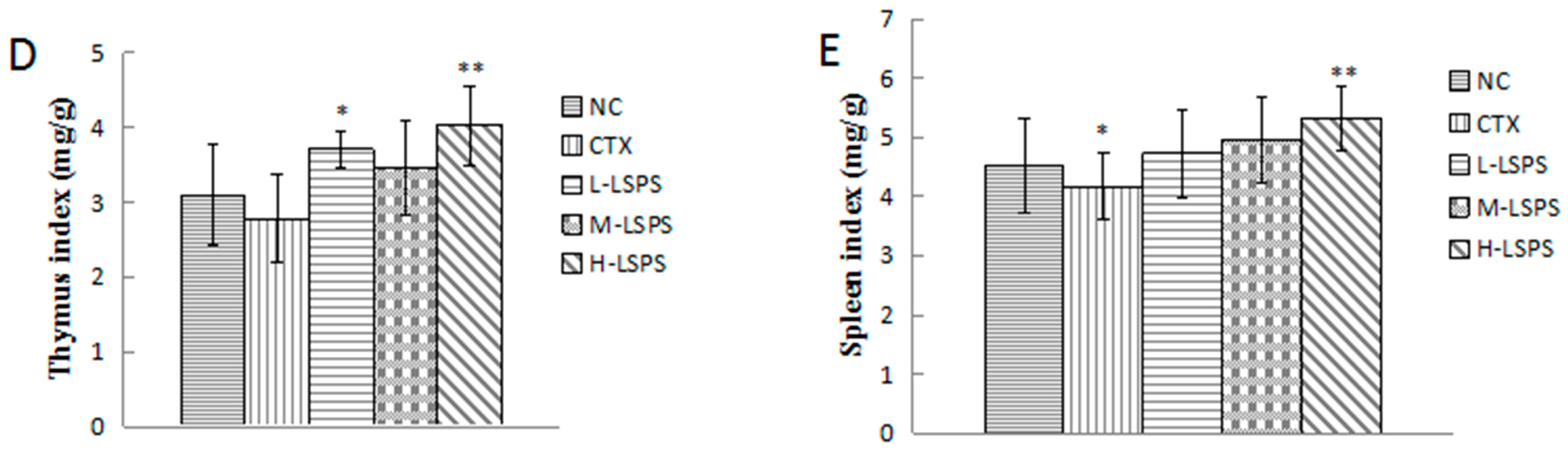
2.4. Effect of LSPS on Immune Organs
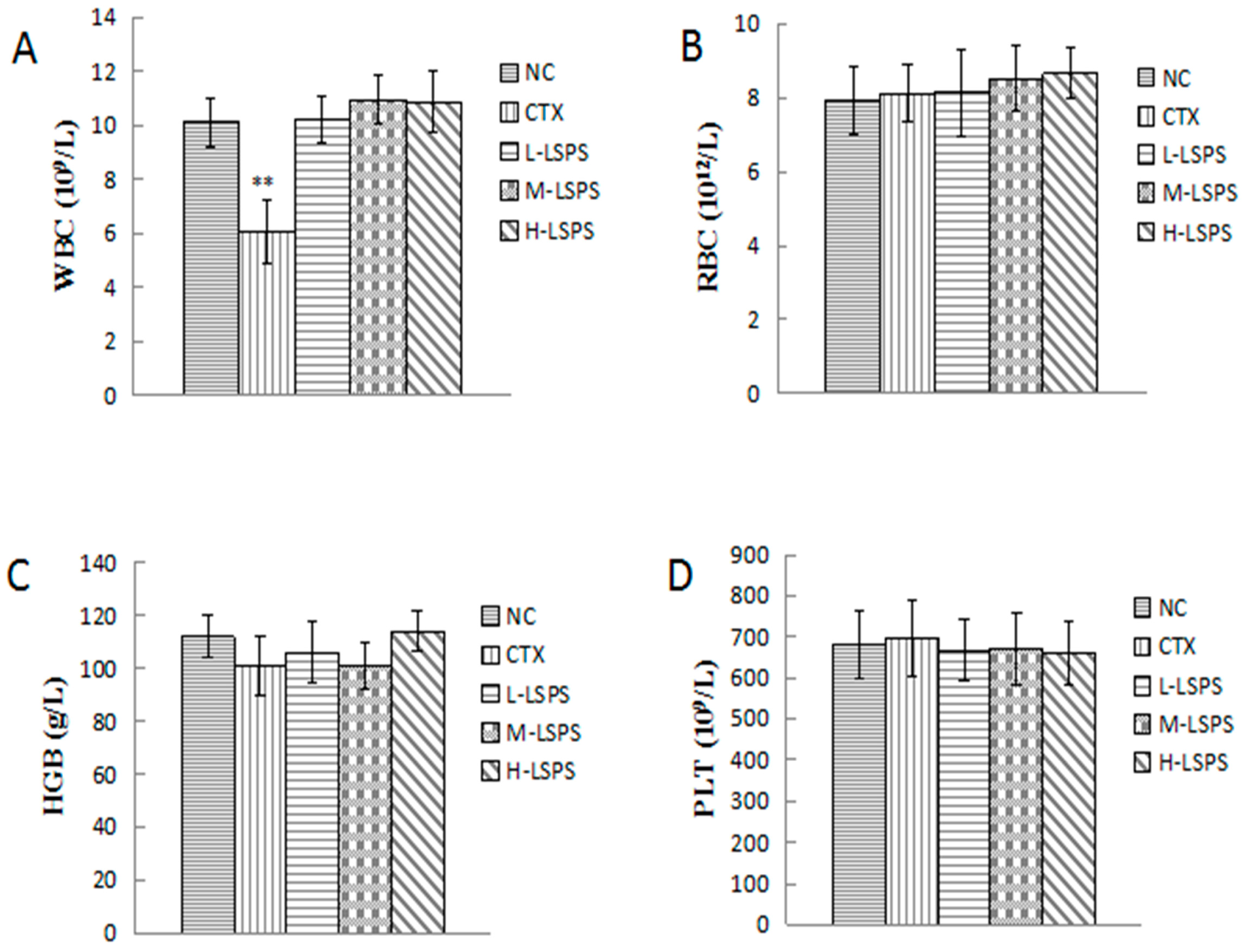
2.5. Effect of LSPS on Blood Physiochemical Parameters
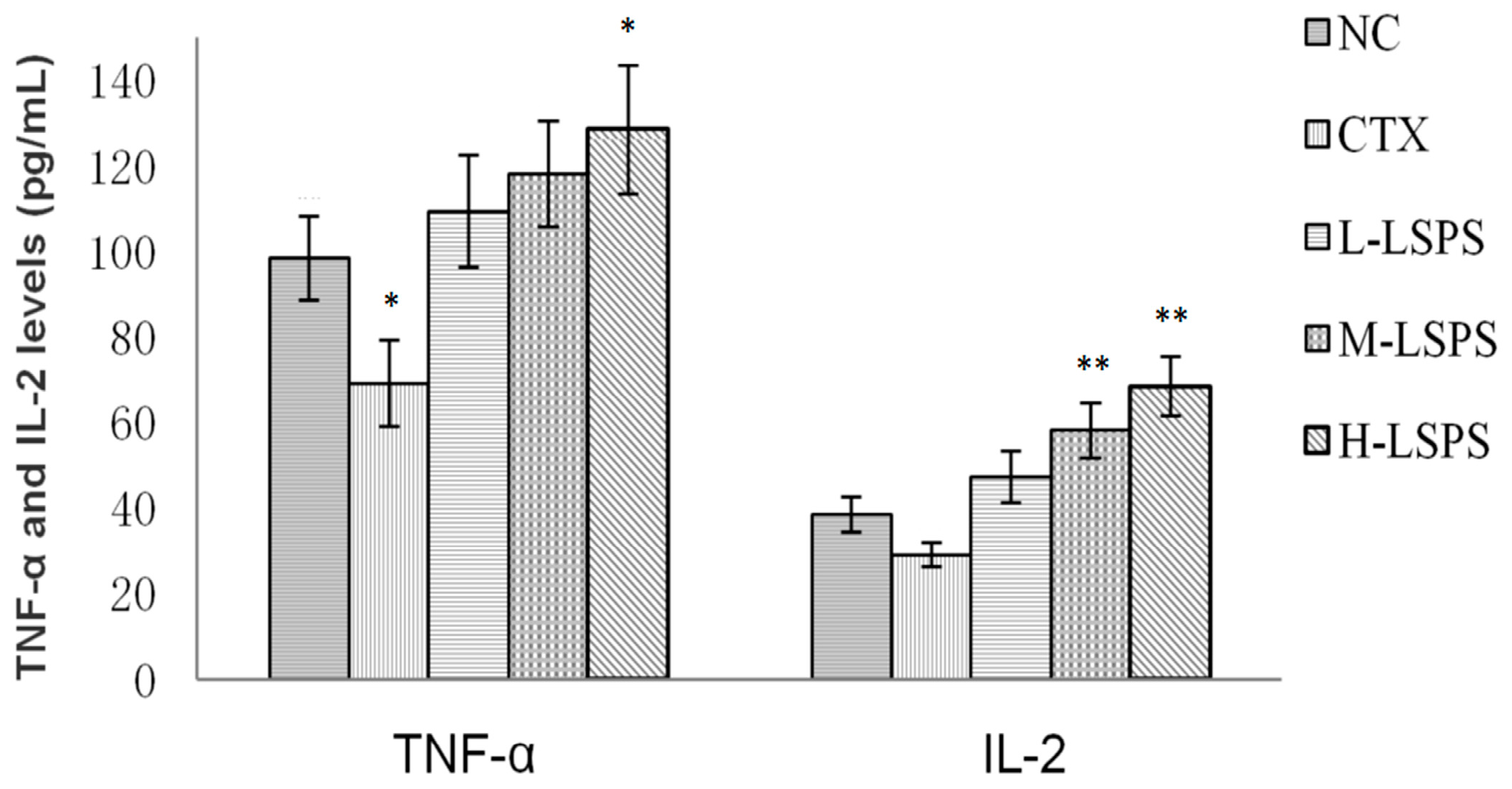
2.6. Effect of LSPS on Serum TNF-α and IL-2 Levels
2.7. Effect of LSPS on SOD Activity and MDA Level
3. Discussion
4. Materials and Methods
4.1. Materials and Reagents
4.2. Extraction of the Hot Water–Soluble Polysaccharides
4.3. Cell Lines and Animals
4.4. In Vitro Cytotoxicity on MFC, HuH-7 and H22 Cell Lines
4.5. In Vivo Antitumor Activity in H22 Mice Hepatocellular Carcinoma Model
4.6. Determination of Blood Physiochemical Parameters
4.7. Determination of Serum TNF-α and IL-2 Levels
4.8. Determination of SOD Activity and MDA Level
4.9. Statistical Analysis
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Lanzotti, V.; Xiao, J.B. Thematic Issue: Natural Products in Cancer Prevention and Therapy, a selection of topics presented in the 2nd Edition PSE Symposium (Naples, Italy, 25th to 28th of June 2013). Anti-Cancer Agents Med. Chem. 2014, 14, 1313–1314. [Google Scholar] [CrossRef]
- Dobson, J. Reducing the side effects of cyclophosphamide chemotherapy in dogs. Vet. Rec. 2014, 174, 248–249. [Google Scholar] [CrossRef] [PubMed]
- Xiao, J.B.; Jiang, H.X. A review on the structure-function relationship aspect of polysaccharides from tea materials. Crit. Rev. Food Sci. Nutr. 2015, 55, 930–938. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Lu, X.; Zeng, S.; Huang, X.; Guo, Z.; Zheng, Y.; Tian, Y.; Zheng, B. Nutritional composition, physiological functions and processing of lotus (Nelumbo nucifera Gaertn.) seeds: A review. Phytochem. Rev. 2015, 14, 321–334. [Google Scholar] [CrossRef]
- Bhat, R.; Sridhar, K.R. Nutritional quality evaluation of electron beam-irradiated lotus (Nelumbo nucifera) seeds. Food Chem. 2008, 107, 174–184. [Google Scholar] [CrossRef]
- Zeng, H.Y.; Cai, L.H.; Cai, X.L.; Wang, Y.J.; Li, Y.Q. Amino acid profiles and quality from lotus seed proteins. J. Sci. Food Agric. 2013, 93, 1070–1075. [Google Scholar] [CrossRef] [PubMed]
- Guo, H.B. Cultivation of lotus (Nelumbo nucifera Gaertn. ssp. nucifera) and its utilization in China. Genet. Resour. Crop Evol. 2008, 56, 323–330. [Google Scholar] [CrossRef]
- Mukherjee, P.K.; Mukherjee, D.; Maji, A.K.; Rai, S.; Heinrich, M. The sacred lotus (Nelumbo nucifera)—Phytochemical and therapeutic profile. J. Pharm. Pharmacol. 2009, 61, 407–422. [Google Scholar] [CrossRef] [PubMed]
- Zhu, M.; Liu, T.; Guo, M. Current Advances in the Metabolomics Study on Lotus Seeds. Front. Plant Sci. 2016, 7, 891. [Google Scholar] [CrossRef] [PubMed]
- Kim, M.-J.; Shin, H.-S. Antioxidative effect of lotus seed and seedpod extracts. Food Sci. Biotechnol. 2012, 21, 1761–1766. [Google Scholar] [CrossRef]
- Kredy, H.M.; Huang, D.; Xie, B.; He, H.; Yang, E.; Tian, B.; Xiao, D. Flavonols of lotus (Nelumbo nucifera, Gaertn.) seed epicarp and their antioxidant potential. Eur. Food Res. Technol. 2010, 231, 387–394. [Google Scholar] [CrossRef]
- Liu, Y.; Ma, S.S.; Ibrahim, S.A.; Li, E.H.; Yang, H.; Huang, W. Identification and antioxidant properties of polyphenols in lotus seed epicarp at different ripening stages. Food Chem. 2015, 185, 159–164. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Zheng, B.; Tian, Y.; Huang, S. Microwave-assisted extraction and anti-oxidation activity of polyphenols from lotus (Nelumbo nucifera Gaertn.) seeds. Food Sci. Biotechnol. 2012, 21, 1577–1584. [Google Scholar] [CrossRef]
- Sung, J.; Sung, J.-S.; Shin, H.-S. Cytoprotective effects of lotus (Nelumbo nucifera Gaertner) seed extracts on oxidative damaged mouse embryonic fibroblast cell. Food Sci. Biotechnol. 2011, 20, 1533–1537. [Google Scholar] [CrossRef]
- Yen, G.-C.; Duh, P.-D.; Su, H.-J. Antioxidant properties of lotus seed and its effect on DNA damage in human lymphocytes. Food Chem. 2005, 89, 379–385. [Google Scholar] [CrossRef]
- Qi, S.; Zhou, D. Lotus seed epicarp extract as potential antioxidant and anti-obesity additive in Chinese cantonese sausage. Meat Sci. 2013, 93, 257–262. [Google Scholar] [CrossRef] [PubMed]
- Liu, C.P.; Tsai, W.J.; Lin, Y.L.; Liao, J.F.; Chen, C.F.; Kuo, Y.C. The extracts from Nelumbo nucifera suppress cell cycle progression, cytokine genes expression, and cell proliferation in human peripheral blood mononuclear cells. Life Sci. 2004, 75, 699–716. [Google Scholar] [CrossRef] [PubMed]
- Poornima, P.; Weng, C.F.; Padma, V.V. Neferine, an alkaloid from lotus seed embryo, inhibits human lung cancer cell growth by MAPK activation and cell cycle arrest. BioFactors 2014, 40, 121–131. [Google Scholar] [CrossRef] [PubMed]
- Poornima, P.; Quency, R.S.; Padma, V.V. Neferine induces reactive oxygen species mediated intrinsic pathway of apoptosis in HepG2 cells. Food Chem. 2013, 136, 659–667. [Google Scholar] [CrossRef] [PubMed]
- Zhang, C.; Huang, K. Characteristic immunostimulation by MAP, a polysaccharide isolated from the mucus of the loach, Misgurnus anguillicaudatus. Carbohydr. Polym. 2005, 59, 75–82. [Google Scholar] [CrossRef]
- Jiang, Y.; Ng, T.B.; Liu, Z.; Wang, C.; Li, N.; Qiao, W.; Liua, F. Immunoregulatory and anti-HIV-1 enzyme activities of antioxidant components from lotus (Nelumbo nucifera Gaertn.) rhizome. Biosci. Rep. 2011, 31, 381–390. [Google Scholar] [CrossRef] [PubMed]
- Liao, C.-H.; Guo, S.-J.; Lin, J.-Y. Characterisation of the chemical composition and in vitro anti-inflammation assessment of a novel lotus (Nelumbo nucifera Gaertn) plumule polysaccharide. Food Chem. 2011, 125, 930–935. [Google Scholar] [CrossRef]
- Liao, C.-H.; Lin, J.-Y. Lotus (Nelumbo nucifera Gaertn) plumule polysaccharide ameliorates pancreatic islets loss and serum lipid profiles in non-obese diabetic mice. Food Chem. Toxicol. 2013, 58, 416–422. [Google Scholar] [CrossRef] [PubMed]
- Liao, C.-H.; Lin, J.-Y. Lotus (Nelumbo nucifera Gaertn) plumule polysaccharide protects the spleen and liver from spontaneous inflammation in non-obese diabetic mice by modulating pro-/anti-inflammatory cytokine gene expression. Food Chem. 2011, 129, 245–252. [Google Scholar] [CrossRef]
- Liao, C.-H.; Lin, J.-Y. Purification, partial characterization and anti-inflammatory characteristics of lotus (Nelumbo nucifera Gaertn) plumule polysaccharides. Food Chem. 2012, 135, 1818–1827. [Google Scholar] [CrossRef] [PubMed]
- Liao, C.-H.; Lin, J.-Y. Purified active lotus plumule (Nelumbo nucifera Gaertn) polysaccharides exert anti-inflammatory activity through decreasing toll-like receptor-2 and -4 expressions using mouse primary splenocytes. J. Ethnopharmacol. 2013, 147, 164–173. [Google Scholar] [CrossRef] [PubMed]
- Tian, Y.T.; Zheng, B.D.; Chen, C.; Zheng, Y.F. Ultrasound-Assisted Extraction, Preliminary Characterization, and Antioxidant Activity of a Novel Water-Soluble Polysaccharide from Lotus (Nelumbo nucifera Gaertn.) Seeds. Sep. Sci. Technol. 2012, 47, 2408–2416. [Google Scholar]
- Li, H.; Gu, L.; Zhong, Y.; Chen, Y.; Zhang, L.; Zhang, A.R.; Sobol, R.W.; Chen, T.; Li, J. Administration of polysaccharide from Panax notoginseng prolonged the survival of H22 tumor-bearing mice. OncoTargets Ther. 2016, 9, 3433–3441. [Google Scholar]
- Chen, X.; Nie, W.; Yu, G.; Li, Y.; Hu, Y.; Lu, J.; Jin, L. Antitumor and immunomodulatory activity of polysaccharides from Sargassum fusiforme. Food Chem. Toxicol. 2012, 50, 695–700. [Google Scholar] [CrossRef] [PubMed]
- Ding, X.; Zhu, F.; Gao, S. Purification, antitumour and immunomodulatory activity of water-extractable and alkali-extractable polysaccharides from Solanum nigrum L. Food Chem. 2012, 131, 677–684. [Google Scholar] [CrossRef]
- Fan, L.; Ding, S.; Ai, L.; Deng, K. Antitumor and immunomodulatory activity of water-soluble polysaccharide from Inonotus obliquus. Carbohydr. Polym. 2012, 90, 870–874. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.Y.; Feng, C.P.; Li, X.; Chang, M.C.; Meng, J.L.; Xu, L.J. Immunomodulatory and antioxidative activity of Cordyceps militaris polysaccharides in mice. Int. J. Biol. Macromol. 2016, 86, 594–598. [Google Scholar] [CrossRef] [PubMed]
- McQueen, K.; Gottumukkala, V.; Davies, J.F.; Riedel, B. Perioperative Implications of the Global Cancer Epidemic. Curr. Anesthesiol. Rep. 2015, 5, 243–249. [Google Scholar] [CrossRef]
- Zhang, Y.; Li, Q.; Wang, J.; Cheng, F.; Huang, X.; Cheng, Y.; Wang, K. Polysaccharide from Lentinus edodes combined with oxaliplatin possesses the synergy and attenuation effect in hepatocellular carcinoma. Cancer Lett. 2016, 377, 117–125. [Google Scholar] [CrossRef] [PubMed]
- Zhao, T.; Mao, G.; Mao, R.; Zou, Y.; Zheng, D.; Feng, W.; Ren, Y.; Wang, W.; Zheng, W.; Song, J.; et al. Antitumor and immunomodulatory activity of a water-soluble low molecular weight polysaccharide from Schisandra chinensis (Turcz.) Baill. Food Chem. Toxicol. 2013, 55, 609–616. [Google Scholar] [CrossRef] [PubMed]
- Matalon, S.T.; Ornoy, A.; Lishner, M. Review of the potential effects of three commonly used antineoplastic and immunosuppressive drugs (cyclophosphamide, azathioprine, doxorubicin on the embryo and placenta). Reprod. Toxicol. 2004, 18, 219–230. [Google Scholar] [CrossRef] [PubMed]
- Thetsrimuang, C.; Khammuang, S.; Chiablaem, K.; Srisomsap, C.; Sarnthima, R. Antioxidant properties and cytotoxicity of crude polysaccharides from Lentinus polychrous Lév. Food Chem. 2011, 128, 634–639. [Google Scholar] [CrossRef]
- Wang, J.; Zhao, Y.; Li, W.; Wang, Z.; Shen, L. Optimization of polysaccharides extraction from Tricholoma mongolicum Imai and their antioxidant and antiproliferative activities. Carbohydr. Polym. 2015, 131, 322–330. [Google Scholar] [CrossRef] [PubMed]
- Wu, Q.; Qu, H.; Jia, J.; Kuang, C.; Wen, Y.; Yan, H.; Gui, Z. Characterization, antioxidant and antitumor activities of polysaccharides from purple sweet potato. Carbohydr. Polym. 2015, 132, 31–40. [Google Scholar] [CrossRef] [PubMed]
- Mao, G.H.; Ren, Y.; Feng, W.W.; Li, Q.; Wu, H.Y.; Jin, D.; Zhao, T.; Xu, C.Q.; Yang, L.Q.; Wu, X.Y. Antitumor and immunomodulatory activity of a water-soluble polysaccharide from Grifola frondosa. Carbohydr. Polym. 2015, 134, 406–412. [Google Scholar] [CrossRef] [PubMed]
- Jiang, Z.; Han, B.; Li, H.; Yang, Y.; Liu, W. Carboxymethyl chitosan represses tumor angiogenesis in vitro and in vivo. Carbohydr. Polym. 2015, 129, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Terlikowski, S.J. Local immunotherapy with rhTNF-alpha mutein induces strong antitumor activity without overt toxicity—A review. Toxicology 2002, 174, 143–152. [Google Scholar] [CrossRef]
- Hou, Y.; Ding, X.; Hou, W.; Song, B.; Wang, T.; Wang, F.; Li, J.; Zeng, Y.; Zhong, J.; Xu, T.; et al. Pharmacological evaluation for anticancer and immune activities of a novel polysaccharide isolated from Boletus speciosus Frost. Mol. Med. Rep. 2014, 9, 1337–1344. [Google Scholar] [CrossRef] [PubMed]
- Jiang, S.; Qiu, L.; Li, Y.; Li, L.; Wang, X.; Liu, Z.; Guo, Y.; Wang, H. Effects of Marsdenia tenacissima polysaccharide on the immune regulation and tumor growth in H22 tumor-bearing mice. Carbohydr. Polym. 2016, 137, 52–58. [Google Scholar] [CrossRef] [PubMed]
- Wang, N.; Yang, J.; Lu, J.; Qiao, Q.; Wu, T.; Du, X.; Bao, G.; He, X. A polysaccharide from Salvia miltiorrhiza Bunge improves immune function in gastric cancer rats. Carbohydr. Polym. 2014, 111, 47–55. [Google Scholar] [CrossRef] [PubMed]
- Sample Availability: Samples of the compounds are available from the authors.

| NC | CTX | L-LSPS | M-LSPS | H-LSPS | |
|---|---|---|---|---|---|
| SOD (U/mg protein) | 140.09 ± 10.16 | 130.69 ± 11.23 | 147.81 ± 9.68 | 160.29 ± 13.89 * | 161.39 ± 13.18 * |
| MDA (nmol/mg protein) | 2.10 ± 0.19 | 2.01 ± 0.24 | 1.92 ± 0.19 | 1.83 ± 0.24 * | 1.67 ± 0.21 ** |
© 2016 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zheng, Y.; Wang, Q.; Zhuang, W.; Lu, X.; Miron, A.; Chai, T.-T.; Zheng, B.; Xiao, J. Cytotoxic, Antitumor and Immunomodulatory Effects of the Water-Soluble Polysaccharides from Lotus (Nelumbo nucifera Gaertn.) Seeds. Molecules 2016, 21, 1465. https://doi.org/10.3390/molecules21111465
Zheng Y, Wang Q, Zhuang W, Lu X, Miron A, Chai T-T, Zheng B, Xiao J. Cytotoxic, Antitumor and Immunomodulatory Effects of the Water-Soluble Polysaccharides from Lotus (Nelumbo nucifera Gaertn.) Seeds. Molecules. 2016; 21(11):1465. https://doi.org/10.3390/molecules21111465
Chicago/Turabian StyleZheng, Yafeng, Qi Wang, Weijing Zhuang, Xu Lu, Anca Miron, Tsun-Thai Chai, Baodong Zheng, and Jianbo Xiao. 2016. "Cytotoxic, Antitumor and Immunomodulatory Effects of the Water-Soluble Polysaccharides from Lotus (Nelumbo nucifera Gaertn.) Seeds" Molecules 21, no. 11: 1465. https://doi.org/10.3390/molecules21111465
APA StyleZheng, Y., Wang, Q., Zhuang, W., Lu, X., Miron, A., Chai, T.-T., Zheng, B., & Xiao, J. (2016). Cytotoxic, Antitumor and Immunomodulatory Effects of the Water-Soluble Polysaccharides from Lotus (Nelumbo nucifera Gaertn.) Seeds. Molecules, 21(11), 1465. https://doi.org/10.3390/molecules21111465










