Isobutylhydroxyamides from Zanthoxylum bungeanum and Their Suppression of NO Production
Abstract
:1. Introduction
2. Results and Discussion
2.1. Compound Characterization
2.2. Biological Activity Assays
3. Experimental Section
3.1. General Procedures
3.2. Plant Materials
3.3. Extraction, Isolation and Purification
3.4. Identification
3.5. Nitric Oxide Production Inhibition Assay
3.6. Cytotoxicity Assays
4. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Bader, M.; Stark, T.D.; Dawid, C.; Lösch, S.; Hofmann, T. All-trans-configuration in Zanthoxylum alkylamides swaps the tingling with a numbing sensation and diminishes salivation. J. Agric. Food Chem. 2014, 62, 2479–2488. [Google Scholar] [CrossRef] [PubMed]
- Yang, X. Aroma constituents and alkylamides of red and green huajiao (Zanthoxylum bungeanum and Zanthoxylum schinifolium). J. Agric. Food. Chem. 2008, 56, 1689–1696. [Google Scholar] [CrossRef] [PubMed]
- Kato, A.; Moriyasu, M.; Ichimaru, M.; Nishiyama, Y. Isolation of alkaloidal constituents of Zanthoxylum usambarense and Zanthoxylum chalybeum using ion-pair HPLC. J. Nat. Prod. 1996, 59, 316–318. [Google Scholar] [CrossRef]
- Huang, S.; Zhao, L.; Zhou, X.L.; Ying, M.; Wang, C.J.; Weng, J. New alkylamides from pericarps of Zanthoxylum bungeanum. Chin. Chem. Lett. 2012, 23, 1247–1250. [Google Scholar] [CrossRef]
- Xiong, Q.; Shi, D.; Yamamoto, H.; Mizuno, M. Alkylamides from pericarps of Zanthoxylum bungeanum. Phytochemistry 1997, 46, 1123–1126. [Google Scholar] [CrossRef]
- Zhao, Z.F.; Zhu, R.X.; Zhong, K.; He, Q.; Luo, A.M.; Gao, H. Characterization and comparison of the pungent components in commercial Zanthoxylum bungeanum oil and Zanthoxylum schinifolium oil. J. Food. Sci. 2013, 78, C1516–C1522. [Google Scholar] [CrossRef] [PubMed]
- Hatano, T.; Inada, K.; Ogawa, T.O.; Ito, H.; Yoshida, T. Aliphatic acid amides of the fruits of Zanthoxylum piperitum. Phytochemistry 2004, 65, 2599–2604. [Google Scholar] [CrossRef] [PubMed]
- Lan, Y.; Wu, Q.; Mao, Y.Q.; Wang, Q.; An, J.; Chen, Y.Y.; Wang, W.P.; Zhao, B.C.; Liu, N.; Zhang, Y.W. Cytotoxicity and enhancement activity of essential oil from Zanthoxylum bungeanum Maxim. as a natural transdermal penetration enhancer. J. Zhejiang Univ. Sci. B 2014, 15, 153–164. [Google Scholar] [CrossRef] [PubMed]
- Yang, L.C.; Li, R.; Tan, J.; Jiang, Z.T. Polyphenolics composition of the leaves of Zanthoxylum bungeanum Maxim. grown in Hebei, China, and their radical scavenging activities. J. Agric. Food Chem. 2013, 61, 1772–1778. [Google Scholar] [CrossRef] [PubMed]
- Navarrete, A.; Hong, E. Anthelmintic properties of α-sanshool from Zanthoxylum liebmannianum. Planta Med. 1996, 62, 250–251. [Google Scholar] [CrossRef] [PubMed]
- Hashimoto, K.; Satoh, K.; Kase, Y.; Ishige, A.; Kubo, M.; Sasaki, H.; Nishikawa, S.; Kurosawa, S.; Yakabi, K.; Nakamura, T. Modulatory effect of aliphatic acid amides from Zanthoxylum piperitum on isolated gastrointestinal tract. Planta Med. 2001, 67, 179–181. [Google Scholar] [CrossRef] [PubMed]
- Jang, K.H.; Chang, Y.H.; Kim, D.D.; Oh, K.B.; Oh, U.; Shin, J. New polyunsaturated fatty acid amides isolated from the seeds of Zanthoxylum piperitum. Arch. Pharm. Res. 2008, 31, 569–572. [Google Scholar] [CrossRef] [PubMed]
- Dossou, K.S.S.; Devkota, K.P.; Morton, C.; Egan, J.M.; Lu, G.; Beutler, J.A.; Moaddel, R. Identification of CB1/CB2 ligands from Zanthoxylum bungeanum. J. Nat. Prod. 2013, 76, 2060–2064. [Google Scholar] [CrossRef] [PubMed]
- Devkota, K.P.; Wilson, J.; Henrich, C.J.; McMahon, J.B.; Reilly, K.M.; Beutler, J.A. Isobutylhydroxyamides from the pericarp of Nepalese Zanthoxylum armatum inhibit NF1-defective tumor cell line growth. J. Nat. Prod. 2013, 76, 59–63. [Google Scholar] [CrossRef] [PubMed]
- Park, Y.D.; Lee, W.S.; An, S.; Jeong, T.S. Human acyl-CoA: Cholesterol acyltransferase inhibitory activities of aliphatic acid amides from Zanthoxylum piperitum DC. Biol. Pharm. Bull. 2007, 30, 205–207. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Yang, S.Y.; Tai, B.H.; Song, S.B.; Li, W.; Yan, X.T.; Sun, Y.N.; Thao, N.P.; Kim, Y.H. NF-κB activation and PPAR transactivational effects of a new aliphatic acid amide from pericarps of Zanthoxylum piperitum. Bull. Korean Chem. Soc. 2014, 35, 2361–2366. [Google Scholar] [CrossRef]
- Tian, J.M.; Wang, Y.; Xu, Y.Z.; Yu, Z.C.; Wei, A.Z.; Zhang, W.M.; Gao, J.M. Characterization of isobutylhydroxyamides with NGF-potentiating activity from Zanthoxylum bungeanum. Bioorg. Med. Chem. Lett. 2016, 26, 338–342. [Google Scholar] [CrossRef] [PubMed]
- Tang, J.J.; Zhang, F.Y.; Wang, D.M.; Tian, J.M.; Dong, S.; Gao, J.M. Semisynthesis and antifeedant activity of new derivatives of a dihydro-β-agarofuran from Parnassia wightiana. Int. J. Mol. Sci. 2013, 14, 19484–19493. [Google Scholar] [CrossRef] [PubMed]
- Liu, H.W.; Yu, X.Z.; Padula, D.; Pescitelli, G.; Lin, Z.W.; Wang, F.; Ding, K.; Lei, M.; Gao, J.M. Lignans from Schisandra sphenathera Rehd. et Wils. and semisynthetic schisantherin A analogues: Absolute configuration, and their estrogenic and anti-proliferative activity. Eur. J. Med. Chem. 2013, 59, 265–273. [Google Scholar] [CrossRef] [PubMed]
- Bai, M.-M.; Shi, W.; Tian, J.-M.; Lei, M.; Kim, J.H.; Sun, Y.N.; Kim, Y.H.; Gao, J.-M. Soluble epoxide hydrolase inhibitory and anti-inflammatory components from the leaves of Eucommia ulmoides Oliver (Duzhong). J. Agric. Food Chem. 2015, 63, 2198–2205. [Google Scholar] [CrossRef] [PubMed]
- Wang, D.-M.; Zhang, C.-C.; Zhang, Q.; Shafiq, N.; Pescitelli, G.; Li, D.-W.; Gao, J.-M. Wightianines A–E, dihydro-β-agarofuran sesquiterpenes from Parnassia wightiana, and their antifungal and insecticidal activities. J. Agric. Food Chem. 2014, 62, 6669–6676. [Google Scholar] [CrossRef] [PubMed]
- Yasuda, I.; Takeya, K.; Itokawa, H. Distribution of unsaturated aliphatic acid amides in Japanese Zanthoxylum species. Phytochemistry 1982, 21, 1295–1298. [Google Scholar] [CrossRef]
- Chen, I.-S.; Chen, T.-L.; Lin, W.-Y.; Tsai, I.-L.; Chen, Y.-C. Isobutylamides from the fruit of Zanthoxylum integrifoliolum. Phytochemistry 1999, 52, 357–360. [Google Scholar] [CrossRef]
- Kashiwada, Y.; Ito, C.; Katagiri, H.; Mase, I.; Komatsu, K.; Namba, T.; Ikeshiro, Y. Amides of the fruit of Zanthoxylum spp. Phytochemistry 1997, 44, 1125–1127. [Google Scholar] [CrossRef]
- Mizutani, K.; Fukunaga, Y.; Tanaka, O.; Takasugi, N.; Saruwatari, Y.; Fuwa, T.; Yamauchi, T.; Wang, J.; Jia, M.R.; Li, F.-Y.; et al. Amides from haujiao, pericarps of Zanthoxylum bungeanum Maxim. Chem. Pharm. Bull. 1988, 36, 2362–2365. [Google Scholar] [CrossRef]
- Kim, Y.H.; Choi, K.H.; Park, J.W.; Kwon, T.K. LY294002 inhibits LPS-induced NO production through a inhibition of NF-κB activation: Independent mechanism of phosphatidylinositol 3-kinase. Immunol. Lett. 2005, 99, 45–50. [Google Scholar] [CrossRef] [PubMed]
- Mosmann, T. Rapid colorimetric assay for cellular growth and survival: Application to proliferation and cytotoxicity assays. J. Immunol. Methods 1983, 65, 55–63. [Google Scholar] [CrossRef]
- Hou, Y.C.; Janczuk, A.; Wang, P.G. Current trends in the development of nitric oxide donors. Curr. Pharm. Des. 1999, 5, 417–441. [Google Scholar]
- Culotta, E.; Koshland, D.E., Jr. NO news is good news. Science 1992, 258, 1862–1864. [Google Scholar] [CrossRef] [PubMed]
- Kaibori, M.; Sakitani, K.; Oda, M.; Kamiyama, Y.; Masu, Y.; Nishizawa, M.; Ito, S.; Okumura, T. Immunosuppressant FK506 inhibits inducible nitric oxide synthase gene expression at a step of NF-κB activation in rat hepatocytes. J. Hepatol. 1999, 30, 1138–1145. [Google Scholar] [CrossRef]
- Sample Availability: Not available.
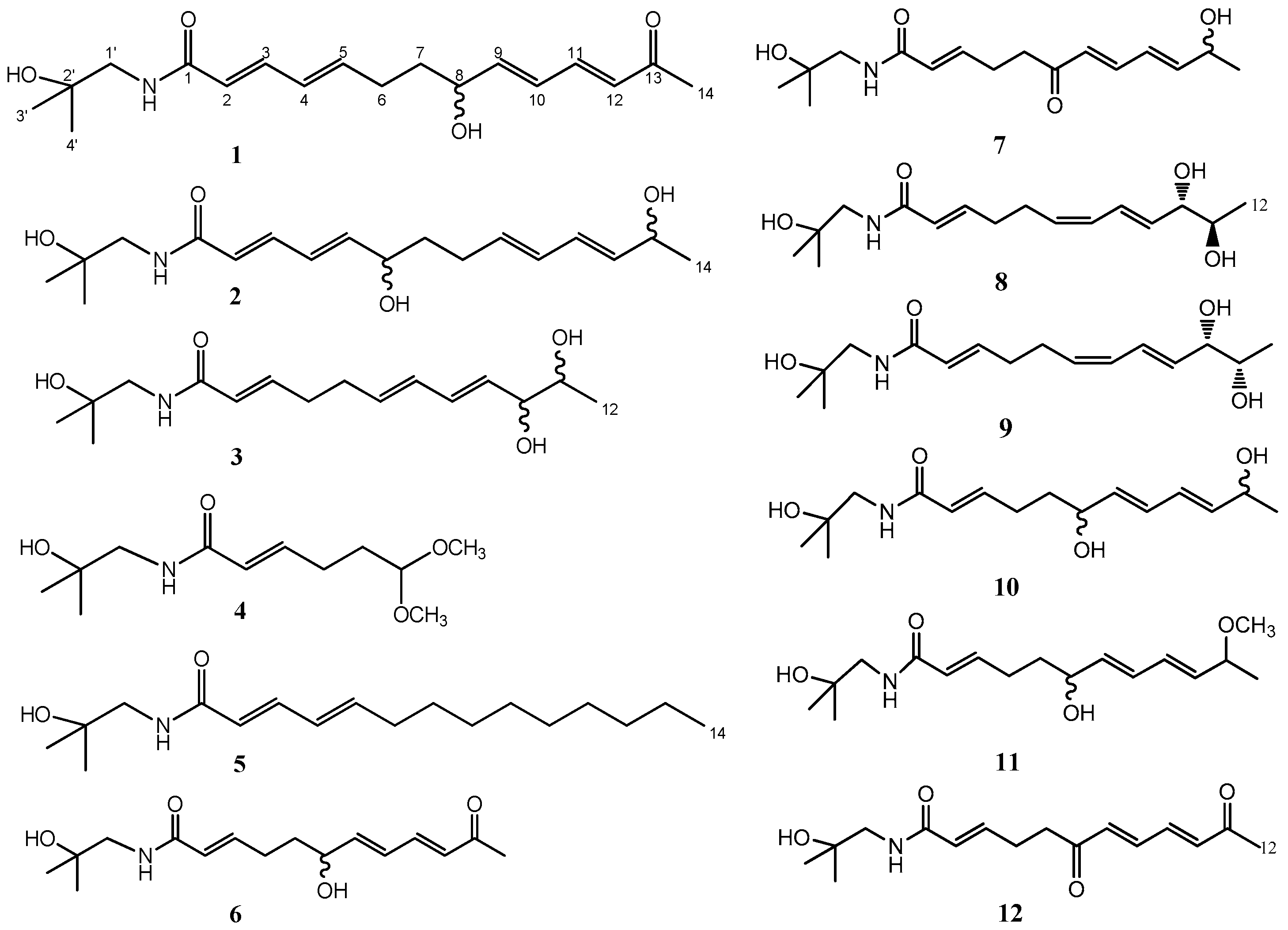
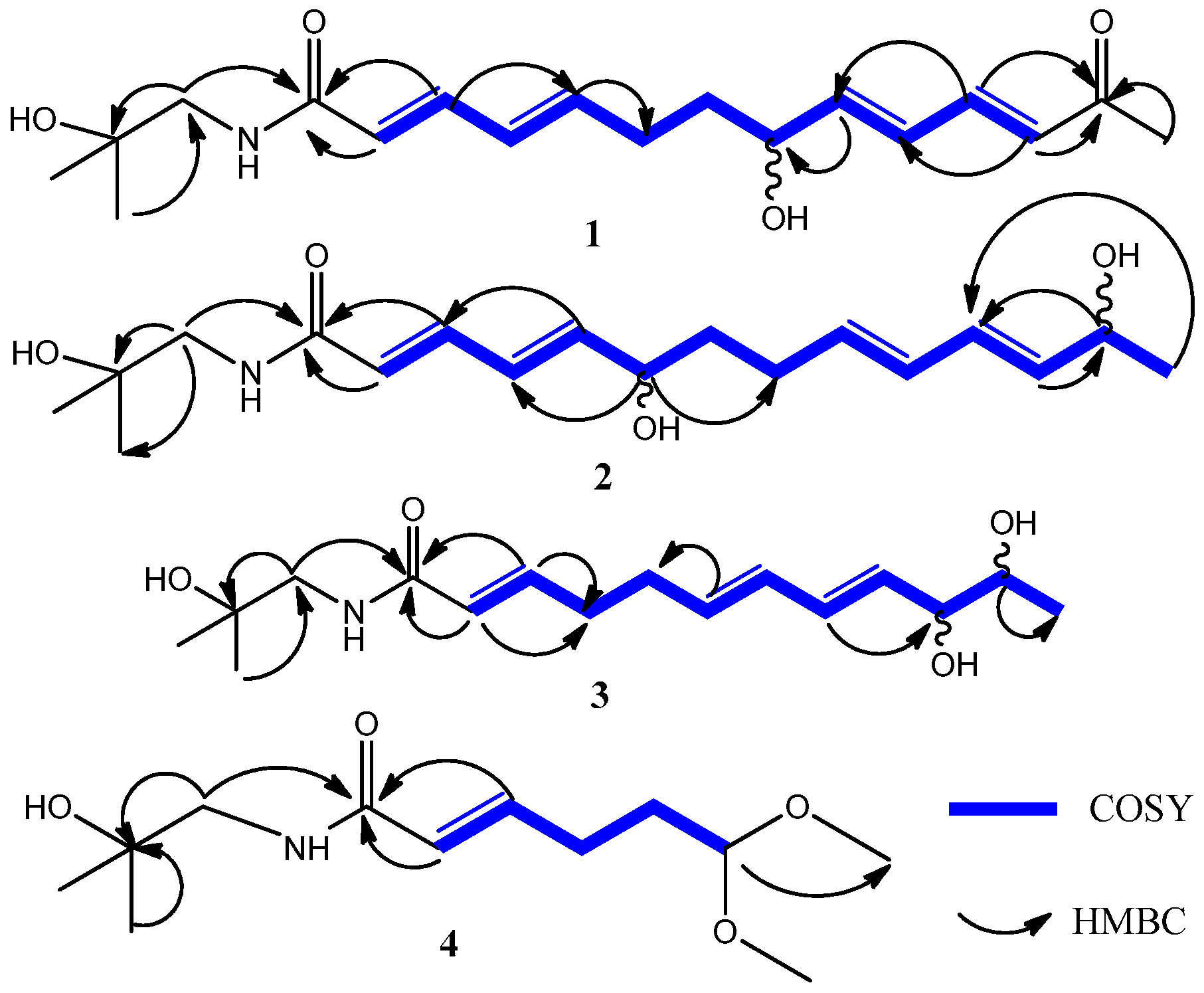
| No. | 1 | 2 | 3 | |||
|---|---|---|---|---|---|---|
| δH (J in Hz) | δC a | δH (J in Hz) | δC a | δH (J in Hz) | δC a | |
| 1 | 169.4 | 169.5 | 169.0 | |||
| 2 | 6.01 d (15.1) | 123.2 | 6.0 m | 123.1 | 5.29 d (15.0) | 125.1 |
| 3 | 7.14 dd (15.7, 10.7) | 142.4 | 7.14 dd (15.1, 10.7) | 142.4 | 6.80 dt (15.6, 6.5) | 145.0 |
| 4 | 6.23 dd (15.7, 10.7) | 130.3 | 6.25 dd (15.1, 10.7) | 130.1 | 2.30 dt (12.5, 6.5) | 32.8 |
| 5 | 6.18 dt (15.7, 10.2) | 143.2 | 5.70 dd (15.1, 7.4) | 143.1 | 2.25 dt (12.5, 6.5) | 32.4 |
| 6 | 2.28 overlap | 29.8 | 6.09 dd (13.1, 7.4) | 72.4 | 5.70 dt (15.3, 7.0) | 133.2 |
| 7 | 1.66 m | 37.0 | 1.63 m | 37.4 | 6.12 dd (15.3, 10.6) | 132.1 |
| 8 | 4.22 q (6.0) | 71.8 | 2.23 td (14.8, 7.4) | 29.9 | 6.23 dd (15.3, 10.6) | 131.9 |
| 9 | 6.28 dd (15.6, 11.4) | 148.2 | 6.15 dt (15.8, 7.4) | 137.0 | 5.68 ddd (22.0, 14.6, 7.0) | 134.0 |
| 10 | 6.44 dd (15.6, 10.8) | 128.9 | 6.22 d (14.3) | 131.1 | 3.90 m | 77.5 |
| 11 | 7.29 dd (15.6, 10.8) | 145.4 | 6.22 d (14.3) | 138.6 | 3.66 m | 71.6 |
| 12 | 6.15 d (15.6) | 131.2 | 5.75 m | 129.8 | 1.13 d (6.4) | 18.6 |
| 13 | 201.5 | 4.27 q (6.4) | 68.8 | |||
| 14 | 2.28 overlap | 27.0 | 1.23 d (6.4) | 23.6 | ||
| 1′ | 3.26 s | 51.2 | 3.26 s | 51.1 | 3.25 s | 51.1 |
| 2′ | 71.7 | 71.6 | 71.7 | |||
| 3′/4′ | 1.18 s | 27.2 | 1.18 s | 27.2 | 1.17 s | 27.2 |
| No. | δH Multi. (J in Hz) a | δC a | δH Multi. (J in Hz) b |
|---|---|---|---|
| 1 | 169.0 | ||
| 2 | 6.02 dt (15.4, 1.5) | 124.9 | 6.03 d (15.5) |
| 3 | 6.80 dt (15.4, 6.9) | 145.1 | 6.61 m |
| 4 | 2.25 td (8.4, 1.5) | 28.1 | 2.13 dd (14.9, 6.6) |
| 5 | 1.74 m | 32.4 | 1.63 dd (14.4, 6.5) |
| 6 | 4.39 t (5.7) | 105.5 | 4.34 t (5.5) |
| 1′ | 3.25 s | 51.1 | 3.07 d (6.0) |
| 2′ | 71.6 | ||
| 3′/4′ | 1.17 s | 27.2 | 1.04 s |
| 2 × OCH3 | 3.33 c | 53.6 | 3.22 s |
| No. | IC50 Values (µM) |
|---|---|
| 1 | 48.7 ± 0.32 |
| 2 | NT |
| 3 | NT |
| 4 | NT |
| 5 | 27.1 ± 1.15 |
| 6 | 49.8 ± 0.38 |
| 7 | NT |
| 8 | 112.9 ± 0.91 |
| 9 | 62.3 ± 1.12 |
| 10 | NT |
| 11 | NT |
| 12 | 39.4 ± 0.63 |
| Dexamethasone | 1.54 ± 0.07 |
© 2016 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wang, Y.; Li, C.-H.; Luo, B.; Sun, Y.N.; Kim, Y.H.; Wei, A.-Z.; Gao, J.-M. Isobutylhydroxyamides from Zanthoxylum bungeanum and Their Suppression of NO Production. Molecules 2016, 21, 1416. https://doi.org/10.3390/molecules21101416
Wang Y, Li C-H, Luo B, Sun YN, Kim YH, Wei A-Z, Gao J-M. Isobutylhydroxyamides from Zanthoxylum bungeanum and Their Suppression of NO Production. Molecules. 2016; 21(10):1416. https://doi.org/10.3390/molecules21101416
Chicago/Turabian StyleWang, Yuan, Chun-Huan Li, Bo Luo, Ya Nan Sun, Young Ho Kim, An-Zhi Wei, and Jin-Ming Gao. 2016. "Isobutylhydroxyamides from Zanthoxylum bungeanum and Their Suppression of NO Production" Molecules 21, no. 10: 1416. https://doi.org/10.3390/molecules21101416
APA StyleWang, Y., Li, C.-H., Luo, B., Sun, Y. N., Kim, Y. H., Wei, A.-Z., & Gao, J.-M. (2016). Isobutylhydroxyamides from Zanthoxylum bungeanum and Their Suppression of NO Production. Molecules, 21(10), 1416. https://doi.org/10.3390/molecules21101416






