Effects of Saponins on Lipid Metabolism: A Review of Potential Health Benefits in the Treatment of Obesity
Abstract
:1. Introduction
2. Obesity and Plant Secondary Metabolites
3. Saponins: Structure and Medicinal Properties
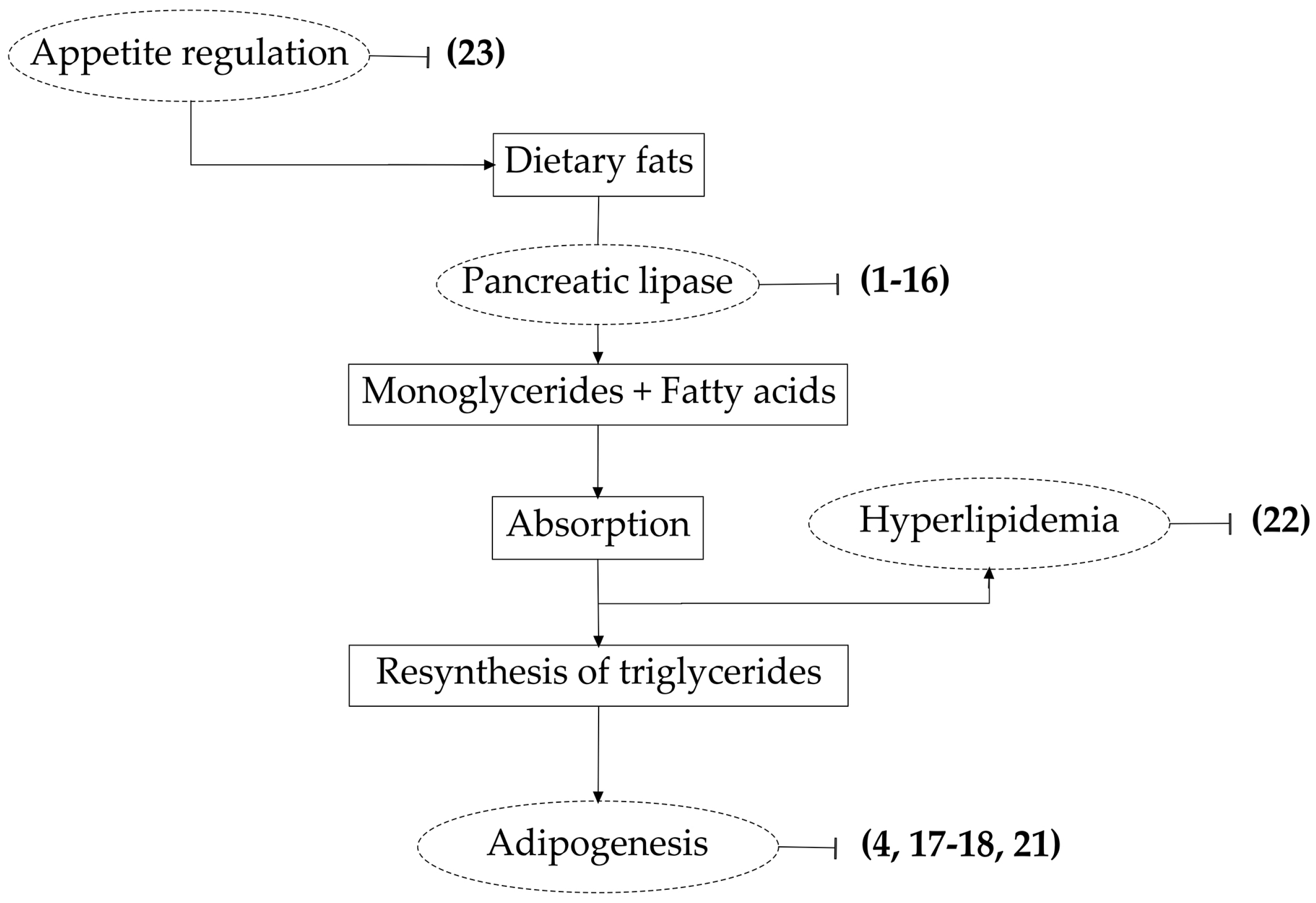
4. Saponins and Pancreatic Lipase Inhibition
4.1. Lipase Inhibition
4.2. Lipase inhibition by Saponin-Containing Plant Extracts
4.3. Saponins Inducing Lipase Inhibition
5. Saponins and Adipogenesis Inhibition
5.1. Adipogenesis
5.2. Saponin Fraction and Pure Compounds Inhibiting Adipogenesis
6. Saponins and Appetite Regulation
6.1. Control of Food Intake and Energy Homeostasis
6.2. Saponins Affecting the Expression of Appetite Peptides
7. Further Effect of Saponins on Lipid Metabolism: Anti-Hyperlipidemic Activity
7.1. Hyperlipidemia
7.2. Saponin-Rich Extracts and Pure Compounds with Antihyperlipidemic Activity
8. Other Saponin Containing-Fractions Affecting Weight Reduction
9. Conclusions
Conflicts of Interest
Abbreviations
| AgRP | agouti-related peptide |
| AMPK | AMP-activated protein kinase |
| C/EBPα | CCAAT/enhancer-binding protein alpha |
| CB1 | cannabinoid type 1 receptors |
| CCK | cholecystokinin |
| CNS | central nervous system |
| EGCG | epigallocatechin gallate |
| EMEA | European Agency for the Evaluation of Medicinal Products |
| FDA | Food and Drug Administration |
| GLP-1 | glucagon-like peptide 1 |
| HDL | high density lipoproteins |
| HeLa | human cervical cancer cells |
| Hep-G2 | hepatocellular carcinoma cell line |
| HL-60 | promyelocytic leukemia cells |
| HT1080 | fibrosarcoma cell line |
| ICD | International Classification of Diseases |
| LDL | low density lipoproteins |
| MAPK | mitogen-activated protein kinase |
| MDA-MB-453 | breast cancer cell line |
| NPY | neuropeptide Y |
| PL | pancreatic lipase |
| PPARγ | peroxisome proliferator-activated receptor γ |
| PYY | peptide YY |
| SREBP-1c | sterol regulatory element-binding protein-1c |
| VLDL | very low density lipoproteins |
| WHO | World Health Organization |
References
- Frühbeck, G.; Toplak, H.; Woodward, E.; Yumuk, V.; Maislos, M.; Oppert, J.-M. Obesity: The gateway to ill health-an EASO position statement on a rising public health, clinical and scientific challenge in Europe. Obes. Facts 2013, 6, 117–120. [Google Scholar] [CrossRef] [PubMed]
- Shook, R.P.; Blair, S.N.; Duperly, J.; Hand, G.A.; Matsudo, S.M.; Slavin, J.L. What is causing the worldwide rise in body weight? US Endocrinol. 2014, 10, 44–52. [Google Scholar] [CrossRef]
- Avena, N.M.; Gold, J.A.; Kroll, C.; Gold, M.S. Further developments in the neurobiology of food and addiction: Update on the state of the science. Nutrition 2012, 28, 341–343. [Google Scholar] [CrossRef] [PubMed]
- Martinez, J.A. Body-weight regulation: Causes of obesity. Proc. Nutr. Soc. 2000, 59, 337–345. [Google Scholar] [CrossRef] [PubMed]
- Spiegelman, B.M.; Flier, J.S. Obesity and the regulation of energy balance. Cell 2001, 104, 531–543. [Google Scholar] [CrossRef]
- Guh, D.P.; Zhang, W.; Bansback, N.; Amarsi, Z.; Birmingham, C.L.; Anis, A.H. The incidence of co-morbidities related to obesity and overweight: A systematic review and meta-analysis. BMC Public Health 2009, 9. [Google Scholar] [CrossRef] [PubMed]
- Derosa, G.; Maffioli, P. Anti-obesity drugs: A review about their effects and their safety. Expert Opin. Drug Saf. 2012, 11, 459–471. [Google Scholar] [CrossRef] [PubMed]
- Al-Suwailem, K.; Al-Tamimi, A.; Al-Omar, M.; Al-Suhibani, M. Safety and mechanism of action of orlistat (tetrahydrolipstatin) as the first local antiobesity drug. J. Appl. Sci. Res. 2006, 2, 205–208. [Google Scholar]
- Kang, J.G.; Park, C.-Y. Anti-obesity drugs: A review about their effects and safety. Diabetes Metab. J. 2012, 36, 13–25. [Google Scholar] [CrossRef] [PubMed]
- Hwang, Y.P.; Choi, J.H.; Kim, H.G.; Lee, H.-S.; Chung, Y.C.; Jeong, H.G. Saponins from Platycodon grandiflorum inhibit hepatic lipogenesis through induction of SIRT1 and activation of AMP-activated protein kinase in high-glucose-induced HepG2 cells. Food Chem. 2013, 140, 115–123. [Google Scholar] [CrossRef] [PubMed]
- Birari, R.B.; Bhutani, K.K. Pancreatic lipase inhibitors from natural sources: Unexplored potential. Drug Discov. Today 2007, 12, 879–889. [Google Scholar] [CrossRef] [PubMed]
- De la Garza, A.L.; Milagro, F.I.; Boque, N.; Campión, J.; Martínez, J.A. Natural inhibitors of pancreatic lipase as new players in obesity treatment. Planta Med. 2011, 77, 773–785. [Google Scholar] [CrossRef] [PubMed]
- Gooda Sahib, N.; Saari, N.; Ismail, A.; Khatib, A.; Mahomoodally, F.; Abdul Hamid, A. Plants’ metabolites as potential antiobesity agents. Sci. World J. 2012, 2012. [Google Scholar] [CrossRef] [PubMed]
- Hasani-Ranjbar, S.; Nayebi, N.; Larijani, B.; Abdollahi, M. A systematic review of the efficacy and safety of herbal medicines used in the treatment of obesity. World J. Gastroenterol. 2009, 15, 3073–3085. [Google Scholar] [CrossRef] [PubMed]
- Wong, C.P.; Kaneda, T.; Morita, H. Plant natural products as an anti-lipid droplets accumulation agent. J. Nat. Med. 2014, 68, 253–266. [Google Scholar] [CrossRef] [PubMed]
- Moro, C.; Basile, G. Obesity and medicinal plants. Fitoterapia 2000, 71, S73–S82. [Google Scholar] [CrossRef]
- Seyedan, A.; Alshawsh, M.A.; Alshagga, M.A.; Koosha, S.; Mohamed, Z. Medicinal plants and their inhibitory activities against pancreatic lipase: A review. Evid. Based Complement. Altern. Med. 2015, 2015. [Google Scholar] [CrossRef] [PubMed]
- Torres-Fuentes, C.; Schellekens, H.; Dinan, T.G.; Cryan, J.F. A natural solution for obesity: Bioactives for the prevention and treatment of weight gain. A review. Nutr. Neurosci. 2015, 18, 49–65. [Google Scholar] [CrossRef] [PubMed]
- Vasudeva, N.; Yadav, N.; Sharma, S.K. Natural products: A safest approach for obesity. Chin. J. Integr. Med. 2012, 18, 473–480. [Google Scholar] [CrossRef] [PubMed]
- Vermaak, I.; Viljoen, A.M.; Hamman, J.H. Natural products in anti-obesity therapy. Nat. Prod. Rep. 2011, 28, 1493–1533. [Google Scholar] [CrossRef] [PubMed]
- Williams, D.J.; Edwards, D.; Hamernig, I.; Jian, L.; James, A.P.; Johnson, S.K.; Tapsell, L.C. Vegetables containing phytochemicals with potential anti-obesity properties: A review. Food Res. Int. 2013, 52, 323–333. [Google Scholar] [CrossRef]
- Yun, J.W. Possible anti-obesity therapeutics from nature—A review. Phytochemistry 2010, 71, 1625–1641. [Google Scholar] [CrossRef] [PubMed]
- Zhang, W.L.; Zhu, L.; Jiang, J.G. Active ingredients from natural botanicals in the treatment of obesity. Obes. Rev. 2014, 15, 957–967. [Google Scholar] [CrossRef] [PubMed]
- Wang, S.; Moustaid-Moussa, N.; Chen, L.; Mo, H.; Shastri, A.; Su, R.; Bapat, P.; Kwun, I.; Shen, C.-L. Novel insights of dietary polyphenols and obesity. J. Nutr. Biochem. 2014, 25, 1–18. [Google Scholar] [CrossRef] [PubMed]
- Meydani, M.; Hasan, S.T. Dietary polyphenols and obesity. Nutrients 2010, 2, 737–751. [Google Scholar] [CrossRef] [PubMed]
- Uchiyama, S.; Taniguchi, Y.; Saka, A.; Yoshida, A.; Yajima, H. Prevention of diet-induced obesity by dietary black tea polyphenols extract in vitro and in vivo. Nutrition 2011, 27, 287–292. [Google Scholar] [CrossRef] [PubMed]
- Lu, C.; Zhu, W.; Shen, C.-L.; Gao, W. Green tea polyphenols reduce body weight in rats by modulating obesity-related genes. PLoS ONE 2012, 7, e38332. [Google Scholar] [CrossRef] [PubMed]
- Balunas, M.J.; Kinghorn, A.D. Drug discovery from medicinal plants. Life Sci. 2005, 78, 431–441. [Google Scholar] [CrossRef] [PubMed]
- Newman, D.J.; Cragg, G.M. Natural products as sources of new drugs over the 30 years from 1981 to 2010. J. Nat. Prod. 2012, 75, 311–335. [Google Scholar] [CrossRef] [PubMed]
- Bongarde, U.; Shinde, V. Review on natural fiber reinforcement polymer composites. Int. J. Eng. Sci. Innov. Technol. 2014, 3, 431–436. [Google Scholar]
- Guarrera, P.M.; Salerno, G.; Caneva, G. Food, flavouring and feed plant traditions in the Tyrrhenian sector of Basilicata, Italy. J. Ethnobiol. Ethnomed. 2006, 2. [Google Scholar] [CrossRef]
- Kaufman, P.B.; Cseke, L.J.; Warber, S.; Duke, J.A.; Brielmann, H.L. Natural Products from Plants; CRC Press: Boca Raton, FL, USA, 1999. [Google Scholar]
- Capuzzo, A.; Maffei, M.E.; Occhipinti, A. Supercritical fluid extraction of plant flavors and fragrances. Molecules 2013, 18, 7194–7238. [Google Scholar] [CrossRef] [PubMed]
- Araniti, F.; Sunseri, F.; Abenavoli, M.R. Phytotoxic activity and phytochemical characterization of Lotus ornithopodioides L., a spontaneous species of Mediterranean area. Phytochem. Lett. 2014, 8, 179–183. [Google Scholar] [CrossRef]
- Araniti, F.; Sorgonà, A.; Lupini, A.; Abenavoli, M. Screening of Mediterranean wild plant species for allelopathic activity and their use as bio-herbicides. Allelopath. J. 2012, 29, 107–124. [Google Scholar]
- Araniti, F.; Lupini, A.; Mercati, F.; Statti, G.A.; Abenavoli, M.R. Calamintha nepeta L. (Savi) as source of phytotoxic compounds: Bio-guided fractionation in identifying biological active molecules. Acta Physiol. Plant. 2013, 35, 1979–1988. [Google Scholar] [CrossRef]
- Lupini, A.; Araniti, F.; Sunseri, F.; Abenavoli, M.R. Coumarin interacts with auxin polar transport to modify root system architecture in Arabidopsis thaliana. Plant Growth Regul. 2014, 74, 23–31. [Google Scholar] [CrossRef]
- Batish, D.R.; Singh, H.P.; Kohli, R.K.; Kaur, S. Eucalyptus essential oil as a natural pesticide. For. Ecol. Manag. 2008, 256, 2166–2174. [Google Scholar] [CrossRef]
- Isman, M.B. Plant essential oils for pest and disease management. Crop Prot. 2000, 19, 603–608. [Google Scholar] [CrossRef]
- Singh, G.; Suresh, S.; Bayineni, V.K.; Kadeppagari, R.K. Lipase inhibitors from plants and their medical applications. Int. J. Pharm. Pharm. Sci. 2015, 7, 1–5. [Google Scholar]
- Ninomiya, K.; Matsuda, H.; Shimoda, H.; Nishida, N.; Kasajima, N.; Yoshino, T.; Morikawa, T.; Yoshikawa, M. Carnosic acid, a new class of lipid absorption inhibitor from sage. Bioorg. Med. Chem. Lett. 2004, 14, 1943–1946. [Google Scholar] [CrossRef] [PubMed]
- Trigueros, L.; Peña, S.; Ugidos, A.; Sayas-Barberá, E.; Pérez-Álvarez, J.; Sendra, E. Food ingredients as anti-obesity agents: A review. Crit. Rev. Food Sci. Nutr. 2013, 53, 929–942. [Google Scholar] [CrossRef] [PubMed]
- Velasquez, M.T.; Bhathena, S.J. Role of dietary soy protein in obesity. Int. J. Med. Sci. 2007, 4, 72–82. [Google Scholar] [CrossRef] [PubMed]
- Francis, G.; Kerem, Z.; Makkar, H.P.; Becker, K. The biological action of saponins in animal systems: A review. Br. J. Nutr. 2002, 88, 587–605. [Google Scholar] [CrossRef] [PubMed]
- Netala, V.R.; Ghosh, S.B.; Bobbu, P.; Anitha, D.; Tartte, V. Triterpenoid saponins: A review on biosynthesis, applications and mechanism of their action. Int. J. Pharm. Pharm. Sci. 2014, 7, 24–28. [Google Scholar]
- Milgate, J.; Roberts, D. The nutritional & biological significance of saponins. Nutr. Res. 1995, 15, 1223–1249. [Google Scholar]
- Baumann, E.; Stoya, G.; Völkner, A.; Richter, W.; Lemke, C.; Linss, W. Hemolysis of human erythrocytes with saponin affects the membrane structure. Acta Histochem. 2000, 102, 21–35. [Google Scholar] [CrossRef] [PubMed]
- Cannon, J.G.; Burton, R.A.; Wood, S.G.; Owen, N.L. Naturally occurring fish poisons from plants. J. Chem. Educ. 2004, 81. [Google Scholar] [CrossRef]
- Wina, E.; Muetzel, S.; Becker, K. The Impact of Saponins or Saponin-Containing Plant Materials on Ruminant Production A Review. J. Agric. Food Chem. 2005, 53, 8093–8105. [Google Scholar] [CrossRef] [PubMed]
- Podolak, I.; Galanty, A.; Sobolewska, D. Saponins as cytotoxic agents: A review. Phytochem. Rev. 2010, 9, 425–474. [Google Scholar] [CrossRef] [PubMed]
- Kerwin, S. Soy saponins and the anticancer effects of soybeans and soy-based foods. Curr. Med. Chem. Anti-Cancer Agents 2004, 4, 263–272. [Google Scholar] [CrossRef] [PubMed]
- Fuchs, H.; Bachran, D.; Panjideh, H.; Schellmann, N.; Weng, A.; Melzig, M.; Sutherland, M.; Bachran, C. Saponins as tool for improved targeted tumor therapies. Curr. Drug Targets 2009, 10, 140–151. [Google Scholar] [CrossRef] [PubMed]
- Park, J.H.; Kwak, J.H.; Khoo, J.H.; Park, S.-H.; Kim, D.U.; Ha, D.M.; Choi, S.U.; Kang, S.C.; Zee, O.P. Cytotoxic effects of triterpenoid saponins from Androsace umbellata against multidrug resistance (MDR) and non-MDR cells. Arch. Pharm. Res. 2010, 33, 1175–1180. [Google Scholar] [CrossRef] [PubMed]
- Rooney, S.; Ryan, M. Effects of alpha-hederin and thymoquinone, constituents of Nigella sativa, on human cancer cell lines. Anticancer Res. 2005, 25, 2199–2204. [Google Scholar] [PubMed]
- Kiem, P.V.; Thu, V.K.; Yen, P.H.; Nhiem, N.X.; Tung, N.H.; Cuong, N.X.; Minh, C.V.; Huong, H.T.; Hyun, J.-H.; Kang, H.-K. New triterpenoid saponins from Glochidion eriocarpum and their cytotoxic activity. Chem. Pharm. Bull. 2009, 57, 102–105. [Google Scholar] [CrossRef] [PubMed]
- Zhou, L.-B.; Chen, T.-H.; Bastow, K.F.; Shibano, M.; Lee, K.-H.; Chen, D.-F. Filiasparosides AD, cytotoxic steroidal saponins from the roots of Asparagus filicinus. J. Nat. Prod. 2007, 70, 1263–1267. [Google Scholar] [CrossRef] [PubMed]
- Elekofehinti, O.O. Saponins: Anti-diabetic principles from medicinal plants—A review. Pathophysiology 2015, 22, 95–103. [Google Scholar] [CrossRef] [PubMed]
- Reis, P.; Holmberg, K.; Watzke, H.; Leser, M.; Miller, R. Lipases at interfaces: A review. Adv. Colloid Interface Sci. 2009, 147, 237–250. [Google Scholar] [CrossRef] [PubMed]
- Han, L.-K.; Xu, B.-J.; Kimura, Y.; Zheng, Y.-N.; Okuda, H. Platycodi radix affects lipid metabolism in mice with high fat diet–induced obesity. J. Nutr. 2000, 130, 2760–2764. [Google Scholar] [PubMed]
- Oishi, Y.; Sakamoto, T.; Udagawa, H.; Taniguchi, H.; Kobayashi-Hattori, K.; Ozawa, Y.; Takita, T. Inhibition of increases in blood glucose and serum neutral fat by Momordica charantia saponin fraction. Biosci. Biotechnol. Biochem. 2007, 71, 735–740. [Google Scholar] [CrossRef] [PubMed]
- Hu, J.-N.; Zhu, X.-M.; Han, L.-K.; Saito, M.; Sun, Y.-S.; Yoshikawa, M.; Kimura, Y.; Zheng, Y.-N. Anti-obesity effects of escins extracted from the seeds of Aesculus turbinata BLUME (Hippocastanaceae). Chem. Pharm. Bull. 2008, 56, 12–16. [Google Scholar] [CrossRef] [PubMed]
- Zheng, Q.; Li, W.; Han, L.; Koike, K. Pancreatic lipase-inhibiting triterpenoid saponins from Gypsophila oldhamiana. Chem. Pharm. Bull. 2007, 55, 646–650. [Google Scholar] [CrossRef] [PubMed]
- Yao, Y.; Zhu, Y.; Gao, Y.; Shi, Z.; Hu, Y.; Ren, G. Suppressive effects of saponin-enriched extracts from quinoa on 3T3-L1 adipocyte differentiation. Food Funct. 2015, 6, 3282–3290. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.H.; Hahm, D.H.; Yang, D.C.; Kim, J.H.; Lee, H.J.; Shim, I. Effect of crude saponin of Korean red ginseng on high-fat diet-induced obesity in the rat. J. Pharmacol. Sci. 2005, 97, 124–131. [Google Scholar] [CrossRef] [PubMed]
- Karu, N.; Reifen, R.; Kerem, Z. Weight gain reduction in mice fed Panax ginseng saponin, a pancreatic lipase inhibitor. J. Agric. Food Chem. 2007, 55, 2824–2828. [Google Scholar] [CrossRef] [PubMed]
- Hamao, M.; Matsuda, H.; Nakamura, S.; Nakashima, S.; Semura, S.; Maekubo, S.; Wakasugi, S.; Yoshikawa, M. Anti-obesity effects of the methanolic extract and chakasaponins from the flower buds of Camellia sinensis in mice. Bioorg. Med. Chem. 2011, 19, 6033–6041. [Google Scholar] [CrossRef] [PubMed]
- Elekofehinti, O.; Kamdem, J.; Kade, I.; Rocha, J.; Adanlawo, I. Hypoglycemic, antiperoxidative and antihyperlipidemic effects of saponins from Solanum anguivi Lam. fruits in alloxan-induced diabetic rats. S. Afr. J. Bot. 2013, 88, 56–61. [Google Scholar] [CrossRef]
- Khan, N.; Akhtar, M.S.; Khan, B.A.; de Andrade Braga, V.; Reich, A. Antiobesity, hypolipidemic, antioxidant and hepatoprotective effects of Achyranthes aspera seed saponins in high cholesterol fed albino rats. Arch. Med. Sci. 2015, 11, 1261–1271. [Google Scholar] [CrossRef] [PubMed]
- Liu, W.; Zheng, Y.; Han, L.; Wang, H.; Saito, M.; Ling, M.; Kimura, Y.; Feng, Y. Saponins (Ginsenosides) from stems and leaves of Panax quinquefolium prevented high-fat diet-induced obesity in mice. Phytomedicine 2008, 15, 1140–1145. [Google Scholar] [CrossRef] [PubMed]
- Reddy, R.M.I.; Latha, P.B.; Vijaya, T.; Rao, D.S. The saponin-rich fraction of a Gymnema sylvestre R. Br. aqueous leaf extract reduces cafeteria and high-fat diet-induced obesity. Z. Naturforsch. C 2012, 67, 39–46. [Google Scholar] [CrossRef] [PubMed]
- Xu, B.J.; Han, L.K.; Zheng, Y.N.; Lee, J.H.; Sung, C.K. In vitro inhibitory effect of triterpenoidal saponins from Platycodi Radix on pancreatic lipase. Arch. Pharm. Res. 2005, 28, 180–185. [Google Scholar] [CrossRef] [PubMed]
- Lee, E.J.; Kang, M.; Kim, Y.S. Platycodin D inhibits lipogenesis through AMPKα-PPARγ2 in 3T3-L1 cells and modulates fat accumulation in obese mice. Planta Med. 2012, 78, 1536–1542. [Google Scholar] [CrossRef] [PubMed]
- Han, L.K.; Nose, R.; Li, W.; Gong, X.J.; Zheng, Y.N.; Yoshikawa, M.; Koike, K.; Nikaido, T.; Okuda, H.; Kimura, Y. Reduction of fat storage in mice fed a high-fat diet long term by treatment with saponins prepared from Kochia scoparia fruit. Phytother. Res. 2006, 20, 877–882. [Google Scholar] [CrossRef] [PubMed]
- Kimura, H.; Ogawa, S.; Jisaka, M.; Kimura, Y.; Katsube, T.; Yokota, K. Identification of novel saponins from edible seeds of Japanese horse chestnut (Aesculus turbinata Blume) after treatment with wooden ashes and their nutraceutical activity. J. Pharm. Biomed. Anal. 2006, 41, 1657–1665. [Google Scholar] [CrossRef] [PubMed]
- Sun, B.-S.; Chen, Y.-P.; Wang, Y.-B.; Tang, S.-W.; Pan, F.-Y.; Li, Z.; Sung, C.-K. Anti-obesity effects of mogrosides extracted from the fruits of Siraitia grosvenorii (Cucurbitaceae). Afr. J. Pharm. Pharmacol. 2012, 6, 1492–1501. [Google Scholar]
- Li, F.; Li, W.; Fu, H.; Zhang, Q.; Koike, K. Pancreatic lipase-inhibiting triterpenoid saponins from fruits of Acanthopanax senticosus. Chem. Pharm. Bull. 2007, 55, 1087–1089. [Google Scholar] [CrossRef] [PubMed]
- Yoshizumi, K.; Hirano, K.; Ando, H.; Hirai, Y.; Ida, Y.; Tsuji, T.; Tanaka, T.; Satouchi, K.; Terao, J. Lupane-type saponins from leaves of Acanthopanax sessiliflorus and their inhibitory activity on pancreatic lipase. J. Agric. Food Chem. 2006, 54, 335–341. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, P.H.; Gauhar, R.; Hwang, S.L.; Dao, T.T.; Park, D.C.; Kim, J.E.; Song, H.; Huh, T.L.; Oh, W.K. New dammarane-type glucosides as potential activators of AMP-activated protein kinase (AMPK) from Gynostemma pentaphyllum. Bioorg. Med. Chem. 2011, 19, 6254–6260. [Google Scholar] [CrossRef] [PubMed]
- Seo, J.B.; Park, S.W.; Choe, S.S.; Jeong, H.W.; Park, J.Y.; Choi, E.-W.; Seen, D.-S.; Jeong, J.-Y.; Lee, T.G. Foenumoside B from Lysimachia foenum-graecum inhibits adipocyte differentiation and obesity induced by high-fat diet. Biochem. Biophys. Res. Commun. 2012, 417, 800–806. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.-J.; Hwang, J.-T.; Kim, M.J.; Yang, H.-J.; Sung, M.J.; Kim, S.-H.; Park, S.; Gu, E.-J.; Park, Y.; Kwon, D.Y. The inhibitory effect of saponin derived from Cheonggukjang on adipocyte differentiation In vitro. Food Sci. Biotechnol. 2014, 23, 1273–1278. [Google Scholar] [CrossRef]
- Poudel, B.; Lim, S.-W.; Ki, H.-H.; Nepali, S.; Lee, Y.-M.; Kim, D.-K. Dioscin inhibits adipogenesis through the AMPK/MAPK pathway in 3T3-L1 cells and modulates fat accumulation in obese mice. Int. J. Mol. Med. 2014, 34, 1401–1408. [Google Scholar] [CrossRef] [PubMed]
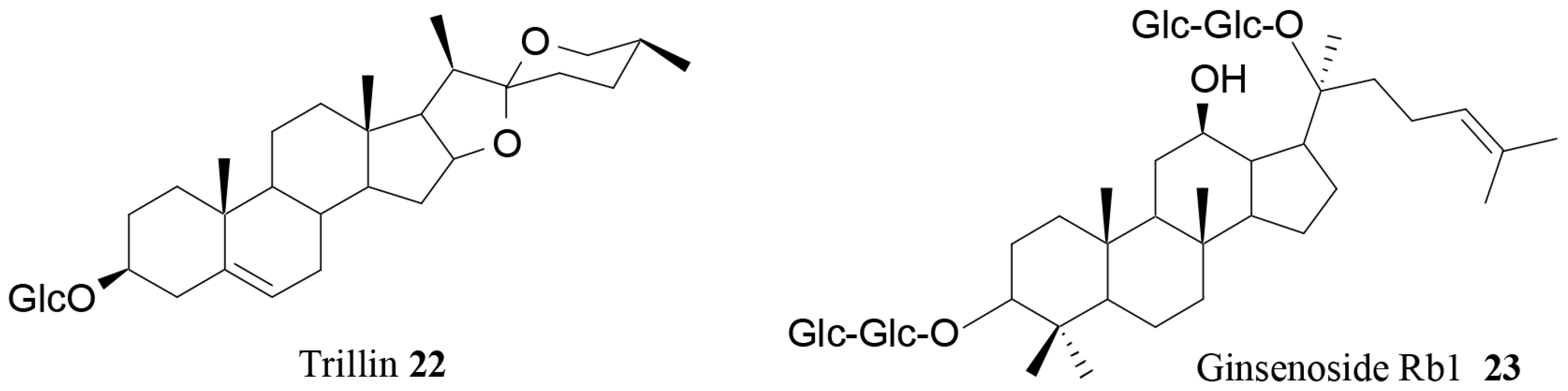
- Wang, T.; Choi, R.C.; Li, J.; Bi, C.W.; Ran, W.; Chen, X.; Dong, T.T.; Bi, K.; Tsim, K.W. Trillin, a steroidal saponin isolated from the rhizomes of Dioscorea nipponica, exerts protective effects against hyperlipidemia and oxidative stress. J. Ethnopharmacol. 2012, 139, 214–220. [Google Scholar] [CrossRef] [PubMed]
- Zhao, H.; Sim, J.; Shim, S.; Ha, Y.; Kang, S.; Kim, Y. Antiobese and hypolipidemic effects of platycodin saponins in diet-induced obese rats: Evidences for lipase inhibition and calorie intake restriction. Int. J. Obes. 2005, 29, 983–990. [Google Scholar] [CrossRef] [PubMed]
- Su, J.; Wang, H.; Ma, C.; Liu, C.; Rahman, M.T.; Gao, C.; Nie, R. Hypolipidemic mechanism of gypenosides via inhibition of pancreatic lipase and reduction in cholesterol micellar solubility. Eur. Food Res. Technol. 2016, 242, 305–312. [Google Scholar] [CrossRef]
- Gauhar, R.; Hwang, S.-L.; Jeong, S.-S.; Kim, J.-E.; Song, H.; Park, D.C.; Song, K.-S.; Kim, T.Y.; Oh, W.K.; Huh, T.-L. Heat-processed Gynostemma pentaphyllum extract improves obesity in ob/ob mice by activating AMP-activated protein kinase. Biotechnol. Lett. 2012, 34, 1607–1616. [Google Scholar] [CrossRef] [PubMed]
- Megalli, S.; Davies, N.M.; Roufogalis, B.D. Anti-hyperlipidemic and hypoglycemic effects of Gynostemma pentaphyllum in the Zucker fatty rat. J. Pharm. Pharm. Sci. 2006, 9, 281–291. [Google Scholar] [PubMed]
- Konturek, P.; Konturek, J.; Cześnikiewicz-Guzik, M.; Brzozowski, T.; Sito, E.; Konturek, S. Neuro-hormonal control of food intake: Basic mechanisms and clinical implications. J. Physiol. Pharm. Off. J. Pol. Physiol. Soc. 2005, 56, 5–25. [Google Scholar]
- Konturek, S.; Konturek, P.; Pawlik, T.; Brzozowski, T. Brain-gut axis and its role in the control of food intake. J. Physiol. Pharmacol. 2004, 55, 137–154. [Google Scholar] [PubMed]
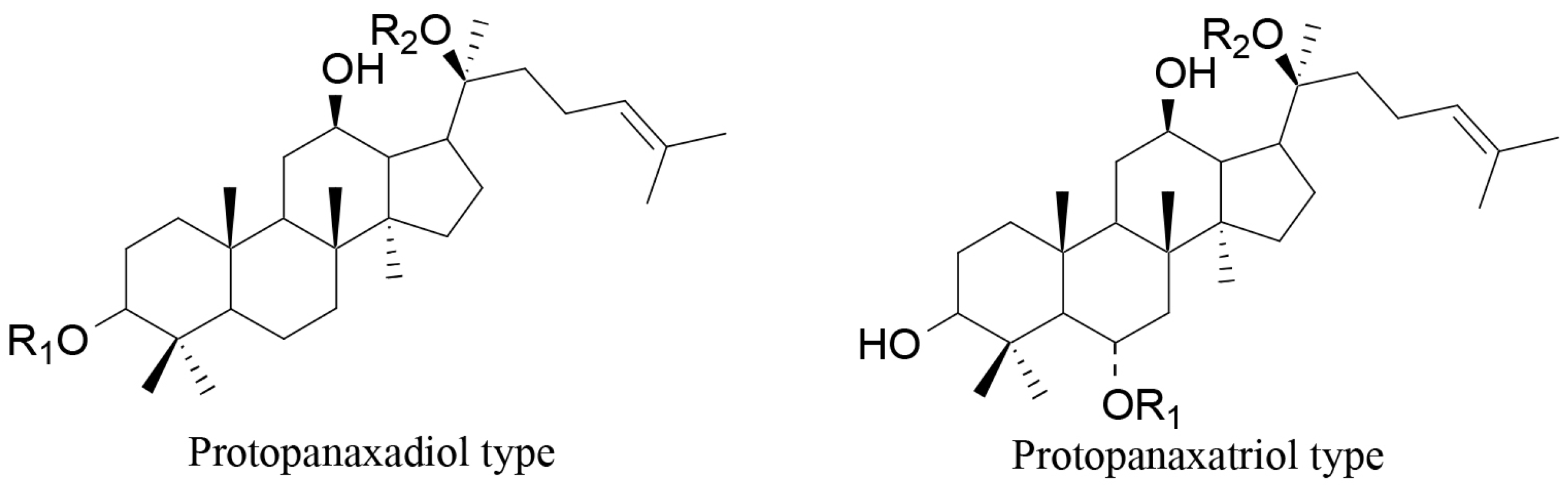
- Kim, J.H.; Kang, S.A.; Han, S.M.; Shim, I. Comparison of the antiobesity effects of the protopanaxadiol- and protopanaxatriol-type saponins of red ginseng. Phytother. Res. 2009, 23, 78–85. [Google Scholar] [CrossRef] [PubMed]
- Morton, G.; Cummings, D.; Baskin, D.; Barsh, G.; Schwartz, M. Central nervous system control of food intake and body weight. Nature 2006, 443, 289–295. [Google Scholar] [CrossRef] [PubMed]
- Wang, Q.; Bing, C.; Al-Barazanji, K.; Mossakowaska, D.E.; Wang, X.-M.; McBay, D.L.; Neville, W.A.; Taddayon, M.; Pickavance, L.; Dryden, S. Interactions between leptin and hypothalamic neuropeptide Y neurons in the control of food intake and energy homeostasis in the rat. Diabetes 1997, 46, 335–341. [Google Scholar] [CrossRef] [PubMed]
- Court, W.E. Ginseng: The Genus Panax; Harwood Academic: Reading, UK, 2000. [Google Scholar]
- Nirosha, K.; Divya, M.; Vamsi, S.; Sadiq, M. A review on hyperlipidemia. Int. J. Novel Trends Pharm. Sci. 2014, 4, 81–92. [Google Scholar]
- Latha, B.P.; Vijaya, T.; Reddy, R.; Ismail, M.; Rao, S.D. Therapeutic efficacy of Achyranthes aspera saponin extract in high fat diet induced hyperlipidaemia in male wistar rats. Afr. J. Biotechnol. 2011, 10, 17038–17042. [Google Scholar]
- De Resende, P.E.; Verza, S.G.; Kaiser, S.; Gomes, L.F.; Kucharski, L.C.; Ortega, G.G. The activity of mate saponins (Ilex paraguariensis) in intra-abdominal and epididymal fat, and glucose oxidation in male Wistar rats. J. Ethnopharmacol. 2012, 144, 735–740. [Google Scholar] [CrossRef] [PubMed]
- Liu, R.; Zhang, J.; Liu, W.; Kimura, Y.; Zheng, Y. Anti-obesity effects of protopanaxdiol types of ginsenosides isolated from the leaves of American ginseng (Panax quinquefolius L.) in mice fed with a high-fat diet. Fitoterapia 2010, 81, 1079–1087. [Google Scholar] [CrossRef] [PubMed]
- Heck, C.I.; De Mejia, E.G. Yerba Mate Tea (Ilex paraguariensis): A comprehensive review on chemistry, health implications, and technological considerations. J. Food Sci. 2007, 72, R138–R151. [Google Scholar] [CrossRef] [PubMed]
- Pang, J.; Choi, Y.; Park, T. Ilex paraguariensis extract ameliorates obesity induced by high-fat diet: Potential role of AMPK in the visceral adipose tissue. Arch. Biochem. Biophys. 2008, 476, 178–185. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.J.; Ko, J.; Storni, C.; Song, H.J.; Cho, Y.G. Effect of green mate in overweight volunteers: A randomized placebo-controlled human study. J. Funct. Foods 2012, 4, 287–293. [Google Scholar] [CrossRef]
- Martins, F.; Noso, T.M.; Porto, V.B.; Curiel, A.; Gambero, A.; Bastos, D.H.; Ribeiro, M.L.; Carvalho, P.D.O. Maté Tea Inhibits In vitro Pancreatic Lipase Activity and Has Hypolipidemic Effect on High-fat Diet-induced Obese Mice. Obesity 2010, 18, 42–47. [Google Scholar] [CrossRef] [PubMed]
- Arçari, D.P.; Bartchewsky, W.; Santos, T.W.; Oliveira, K.A.; Funck, A.; Pedrazzoli, J.; Souza, M.F.; Saad, M.J.; Bastos, D.H.; Gambero, A. Antiobesity Effects of yerba maté Extract (Ilex paraguariensis) in High-fat Diet–induced Obese Mice. Obesity 2009, 17, 2127–2133. [Google Scholar] [CrossRef] [PubMed]
- Lathaa, B.P.; Reddya, I.R.M.; Vijaya, T.; Raoc, S.D.; Ismaild, S.M.; Girisha, B. Effect of saponin rich extract of Achyranthes aspera on high fat diet fed male wistar rats. J. Pharm. Res. 2011, 4, 3190–3193. [Google Scholar]
- Chaudhary, N.; Bhardwaj, J.; Seo, H.J.; Kim, M.Y.; Shin, T.S.; Kim, J.D. Camellia sinensis fruit peel extract inhibits angiogenesis and ameliorates obesity induced by high-fat diet in rats. J. Funct. Foods 2014, 7, 479–486. [Google Scholar] [CrossRef]
- Lin, N.; Cai, D.-L.; Jin, D.; Chen, Y.; Shi, J.-J. Ginseng panaxoside Rb1 reduces body weight in diet-induced obese mice. Cell Biochem. Biophys. 2014, 68, 189–194. [Google Scholar] [CrossRef] [PubMed]
- Han, L.-K.; Zheng, Y.-N.; Yoshikawa, M.; Okuda, H.; Kimura, Y. Anti-obesity effects of chikusetsusaponins isolated from Panax japonicus rhizomes. BMC Complement. Altern. Med. 2005, 5. [Google Scholar] [CrossRef] [PubMed]
| Plant Species | Plant Part | Study | Action | Reference |
|---|---|---|---|---|
| Platycodon grandiflorum (Jacq.) A.DC. | Roots | In vitro In vivo | Lipase inhibition | [10,59] |
| Momordica charantia L. | _ | In vitro In vivo | Lipase inhibition | [60] |
| Aesculus turbinata Blume | Seeds | In vitro In vivo | Lipase inhibition, suppression of body weight increase, hepatic triacylglycerol content and total cholesterol content | [61] |
| Gypsophila oldhamiana (Miq.) | Root | In vitro | Lipase inhibition | [62] |
| Chenopodium quinoa Willd. | Seeds | In vitro | Downregulation of adipogenic transcription factors | [63] |
| Panax ginseng C.A. Meyer | Roots | In vitro In vivo | Lipase inhibition, downregulation of hypothalamic NPY and serum leptin | [64,65] |
| Camellia sinensis L | Flower buds | In vivo | Suppression of mRNA levels of neuropeptide Y | [66] |
| Solanum anguivi Lam. | Fruits | In vivo | Antihyperlipidemic activity | [67] |
| Achyranthes aspera L. | Seeds | In vivo | Antihyperlipidemic activity | [68] |
| Panax quinquefolium L. | Leaves | In vitro In vivo | Lipase inhibition, decrease of adipose tissue weight | [69] |
| Gymnema sylvestre R. Br. | Leaves | In vivo | Decrease of food consumption and body weight | [70] |
| Saponin | Plant Species | Study | Action | Reference |
|---|---|---|---|---|
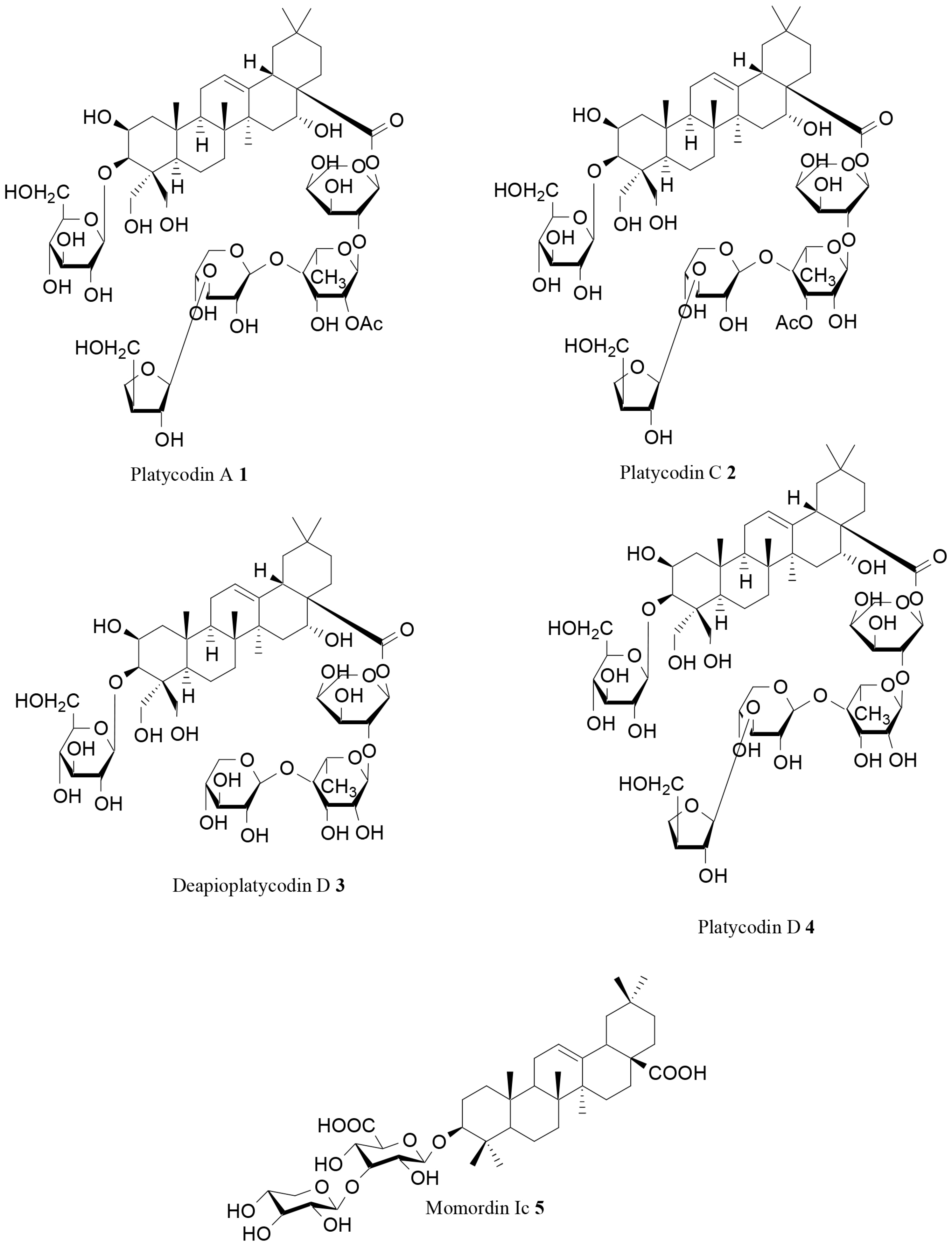
| Platycodin A (1) | Platycodon grandiflorum (Jacq.) A.DC. | In vitro | Lipase inhibition | [71] |
| Platycodin C (2) | Platycodon grandiflorum (Jacq.) A.DC. | In vitro | Lipase inhibition | [71] |
| Deapioplatycodin D (3) | Platycodon grandiflorum (Jacq.) A.DC. | In vitro | Lipase inhibition | [71] |
| Platycodin D (4) | Platycodon grandiflorum (Jacq.) A.DC. | In vitro In vivo | Lipase inhibition, AMPK activation, prevention of abdominal fat accumulation | [71,72] |
| Momordin Ic (5) | Kochia scoparia (L.) Schard | In vitro | Lipase inhibition | [73] |
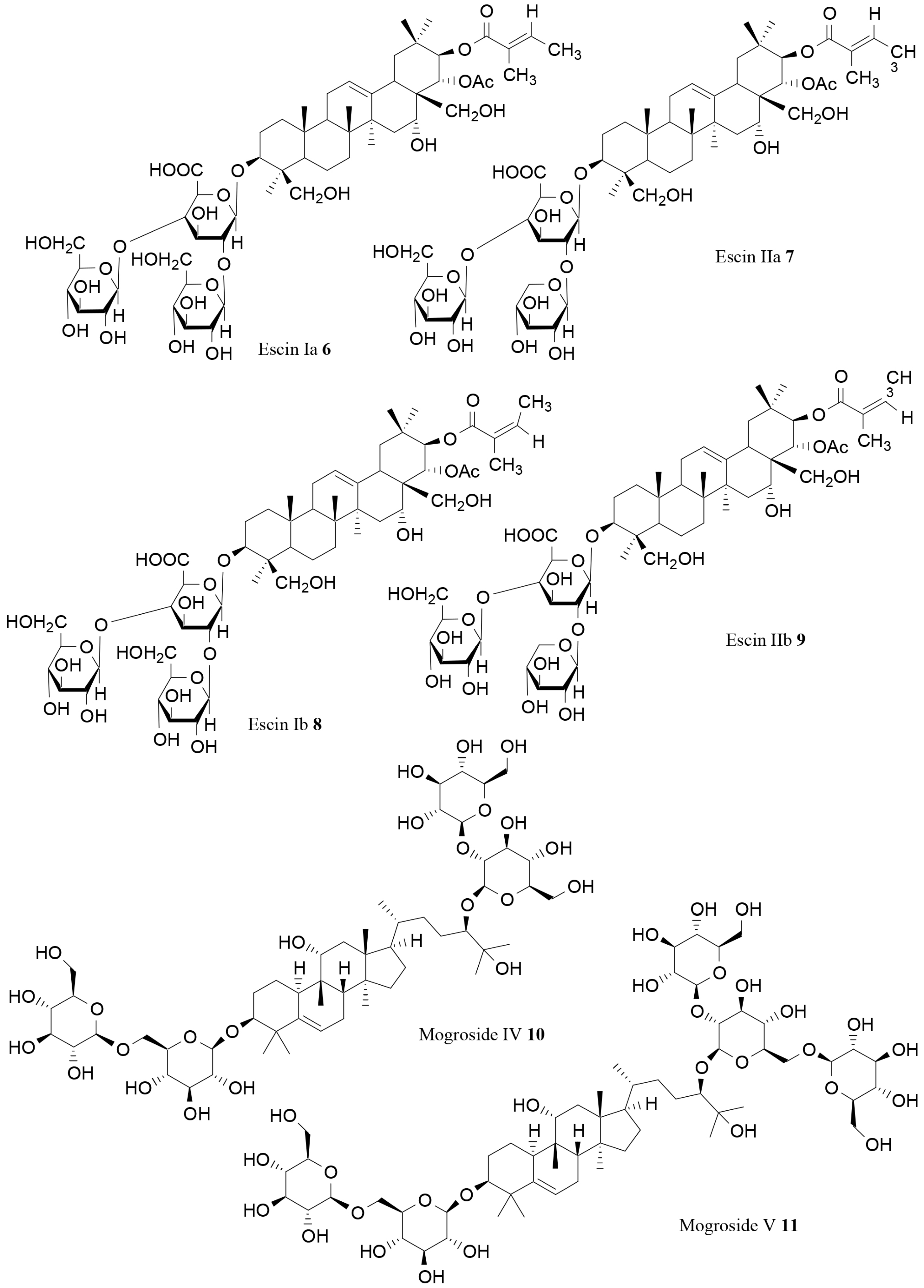
| Escin Ia (6) | Aesculus turbinata Blume | In vitro | Lipase inhibition | [74] |
| Escin IIa (7) | Aesculus turbinata Blume | In vitro | Lipase inhibition | [74] |
| Escin Ib (8) | Aesculus turbinata Blume | In vitro | Lipase inhibition | [74] |
| Escin IIb (9) | Aesculus turbinata Blume | In vitro | Lipase inhibition | [74] |
| Mogroside IV(10) | Siraitia grosvenorii C. Jeffrey | In vitro | Lipase inhibition | [75] |
| Mogroside V (11) | Siraitia grosvenorii C. Jeffrey | In vitro | Lipase inhibition | [75] |
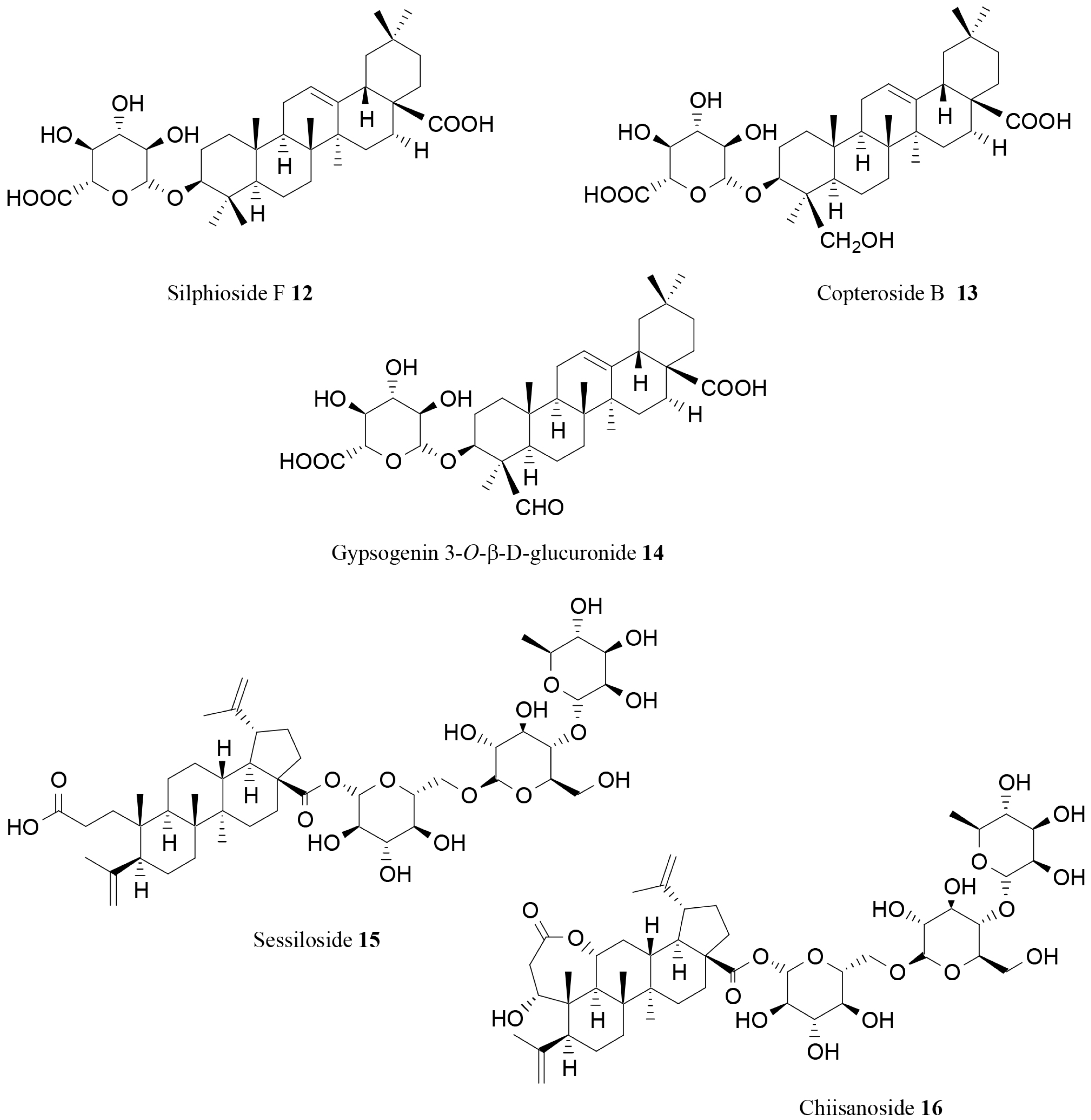
| Silphioside F (12) | Acanthopanax senticosus (Rupr. et Maxim.) Harms | In vitro | Lipase inhibition | [76] |
| Copteroside B (13) | Acanthopanax senticosus (Rupr. et Maxim.) Harms | In vitro | Lipase inhibition | [76] |
| Gypsogenin 3-O-β-d-glucuronide (14) | Acanthopanax senticosus (Rupr. et Maxim.) Harms | In vitro | Lipase inhibition | [76] |
| Sessiloside (15) | Acanthopanax sessiliflorus (Rupr. et Maxim.) Seem | In vitro | Lipase inhibition | [77] |
| Chiisanoside (16) | Acanthopanax sessiliflorus (Rupr. et Maxim.) Seem | In vitro | Lipase inhibition | [77] |
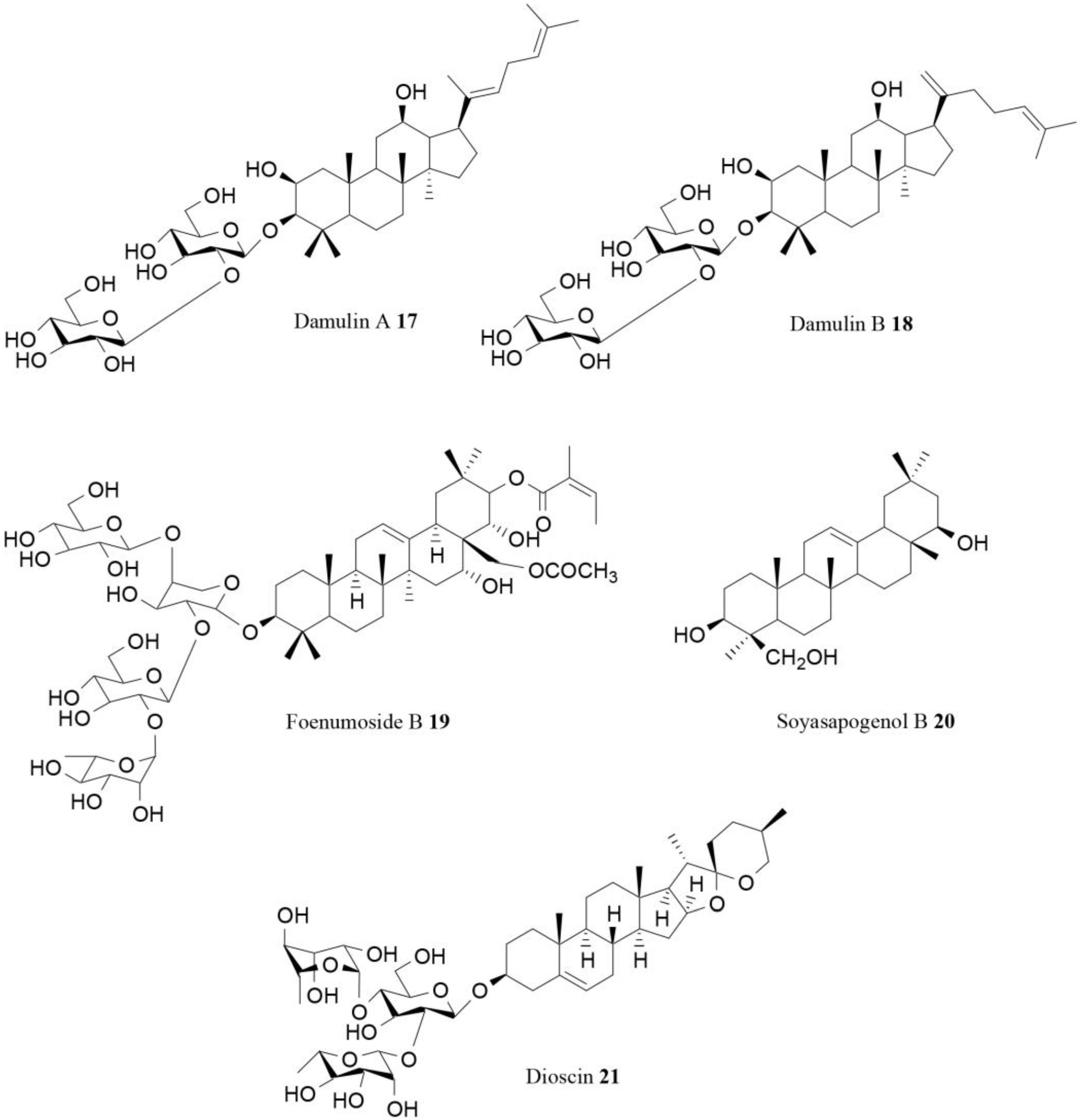
| Damulin A (17) | Gynostemma pentaphyllum Makino | In vitro | AMPK activation | [78] |
| Damulin (18) | Gynostemma pentaphyllum Makino | In vitro | AMPK activation | [78] |
| Foenumoside B (19) | Lysimachia foenum-graecum Hance | In vitro In vivo | AMPK activation, reduction of body weight gain | [79] |
| Soyasapogenol B (20) | Korean fermented soy food named cheonggukjang | In vitro | AMPK activation | [80] |
| Dioscin (21) | Several species | In vitro | Influence on AMPK/MAPK | [81] |
| Trillin (22) | Dioscorea nipponica Makino | In vivo | Antihyperlipidemic activity | [82] |
| Ginsenoside Rb1 (23) | Ginseng | In vivo | Modulation of serum levels of PYY and NPY | [67] |
© 2016 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Marrelli, M.; Conforti, F.; Araniti, F.; Statti, G.A. Effects of Saponins on Lipid Metabolism: A Review of Potential Health Benefits in the Treatment of Obesity. Molecules 2016, 21, 1404. https://doi.org/10.3390/molecules21101404
Marrelli M, Conforti F, Araniti F, Statti GA. Effects of Saponins on Lipid Metabolism: A Review of Potential Health Benefits in the Treatment of Obesity. Molecules. 2016; 21(10):1404. https://doi.org/10.3390/molecules21101404
Chicago/Turabian StyleMarrelli, Mariangela, Filomena Conforti, Fabrizio Araniti, and Giancarlo A. Statti. 2016. "Effects of Saponins on Lipid Metabolism: A Review of Potential Health Benefits in the Treatment of Obesity" Molecules 21, no. 10: 1404. https://doi.org/10.3390/molecules21101404
APA StyleMarrelli, M., Conforti, F., Araniti, F., & Statti, G. A. (2016). Effects of Saponins on Lipid Metabolism: A Review of Potential Health Benefits in the Treatment of Obesity. Molecules, 21(10), 1404. https://doi.org/10.3390/molecules21101404