Structurally Related Monoterpenes p-Cymene, Carvacrol and Thymol Isolated from Essential Oil from Leaves of Lippia sidoides Cham. (Verbenaceae) Protect Mice against Elastase-Induced Emphysema
Abstract
:1. Introduction
2. Results
2.1. Chemical Analysis of Isolated Monoterpenes from Essential Oil from Leaves of L. sidoides
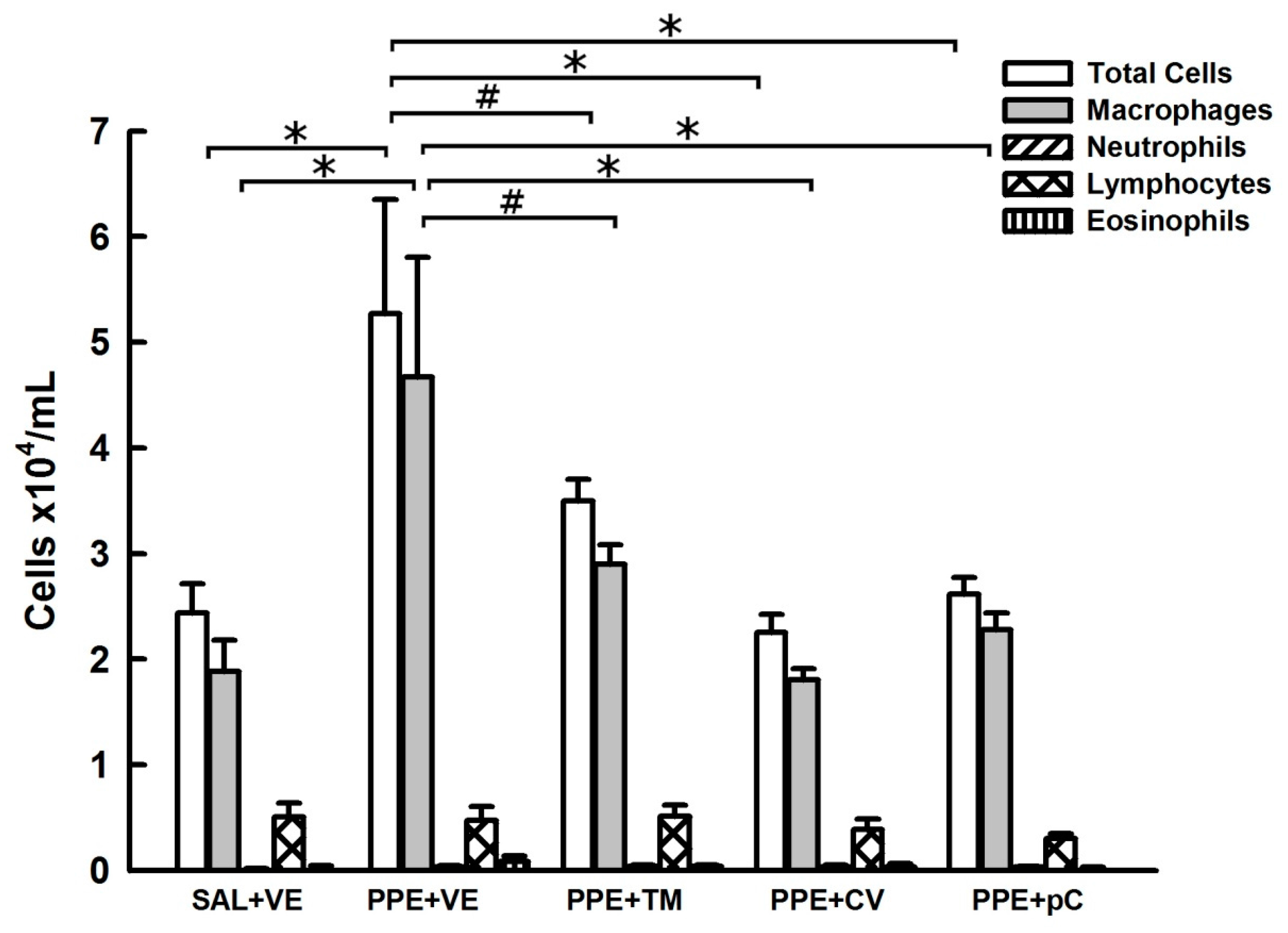
2.2. Monoterpenes Reduced BALF Lung Inflammation in PPE-Induced Emphysema in Mice
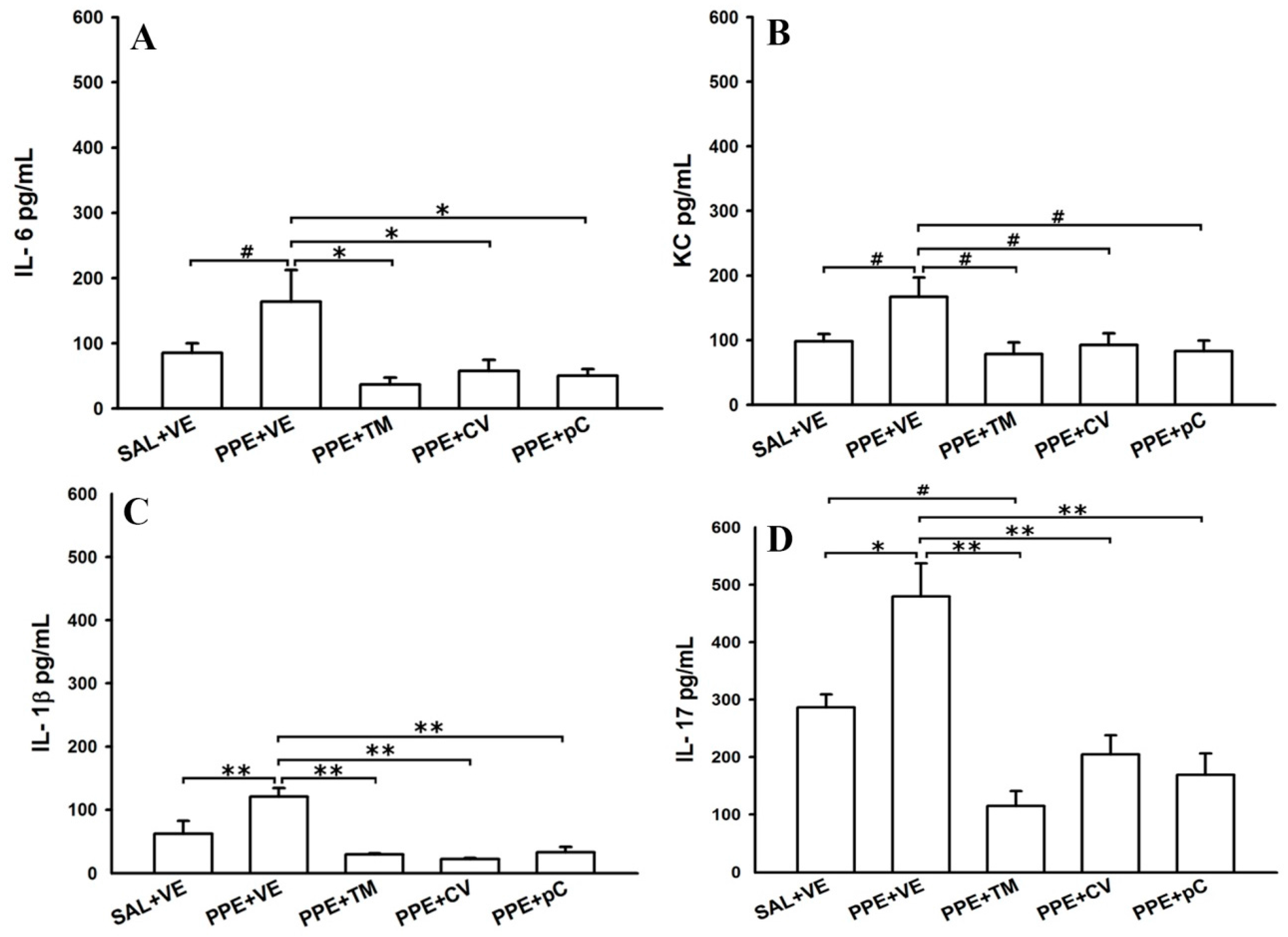
2.3. Monoterpenes Reduced Cytokines in BALF in PPE-Induced Emphysema in Mice
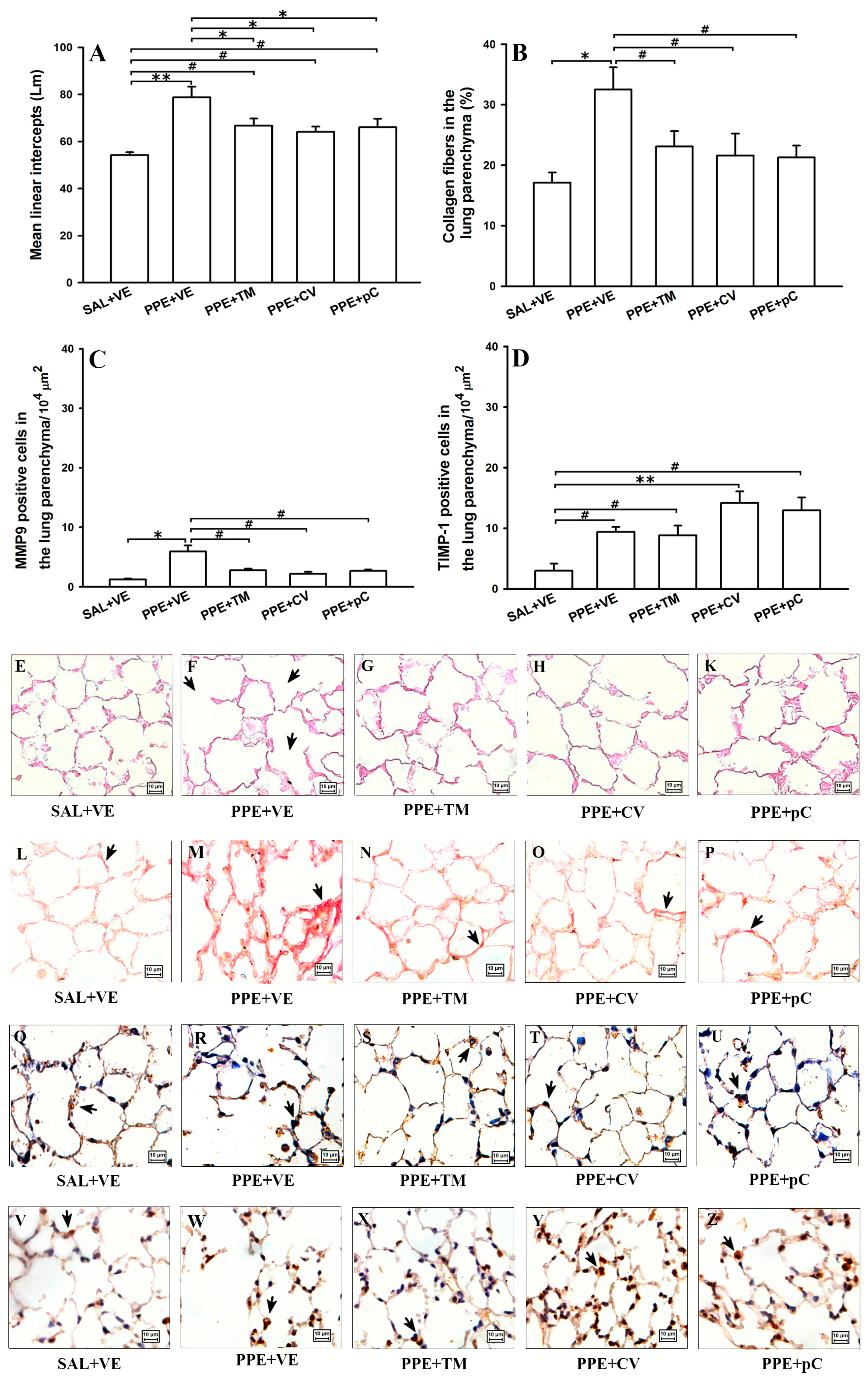
2.4. Monoterpenes Prevented Alveolar Septa Destruction Reduced the Collagen Fibers Deposition Probably by Controlling MMP-9/TIMP-1 in PPE-Induced Emphysema Model
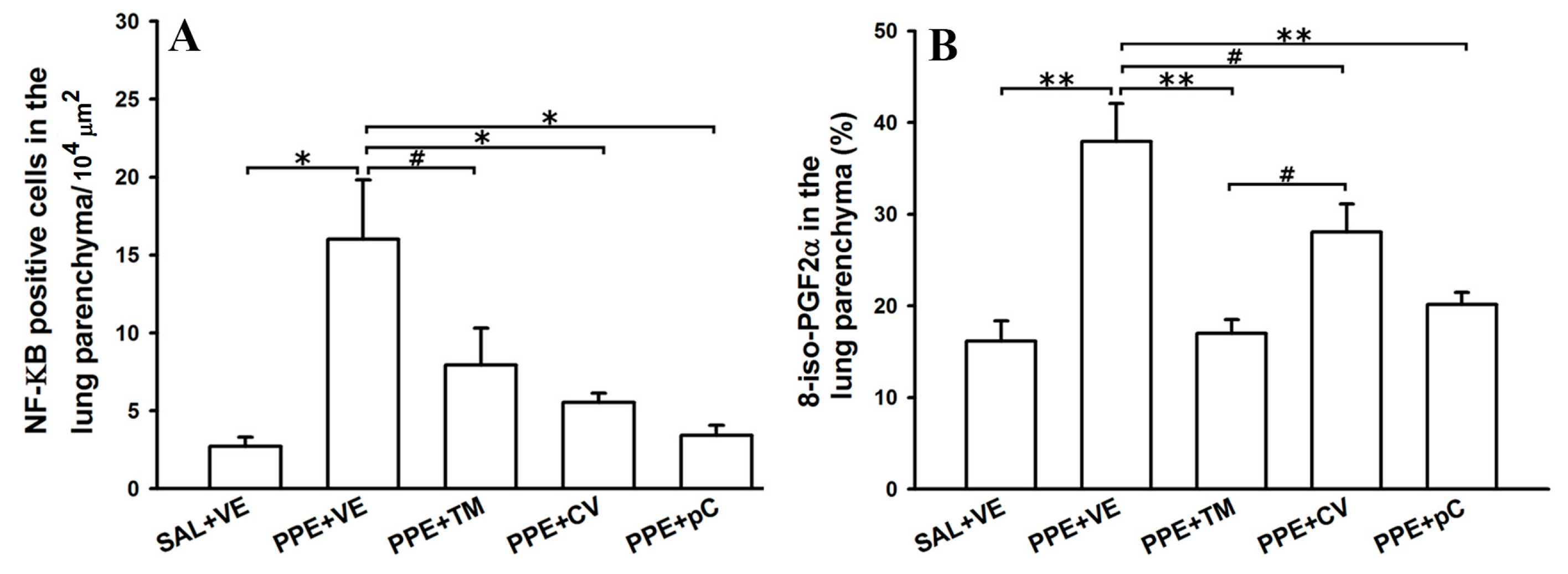
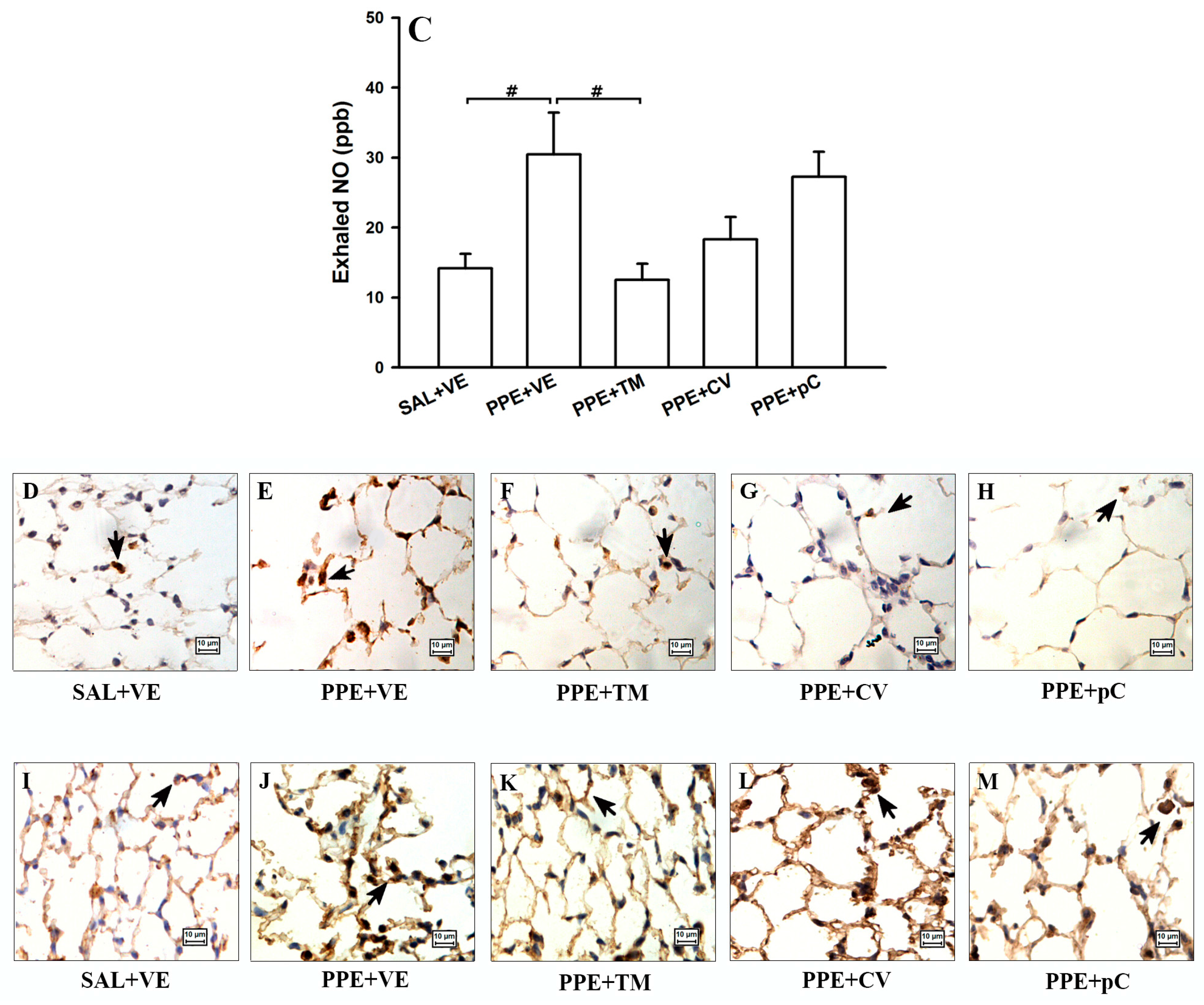
2.5. The Monoterpenes Probably Acts in Reduction of Emphysema and Lung Inflammation in Mice by Reducing Oxidative Stress and in NF-κB Expression in Lung
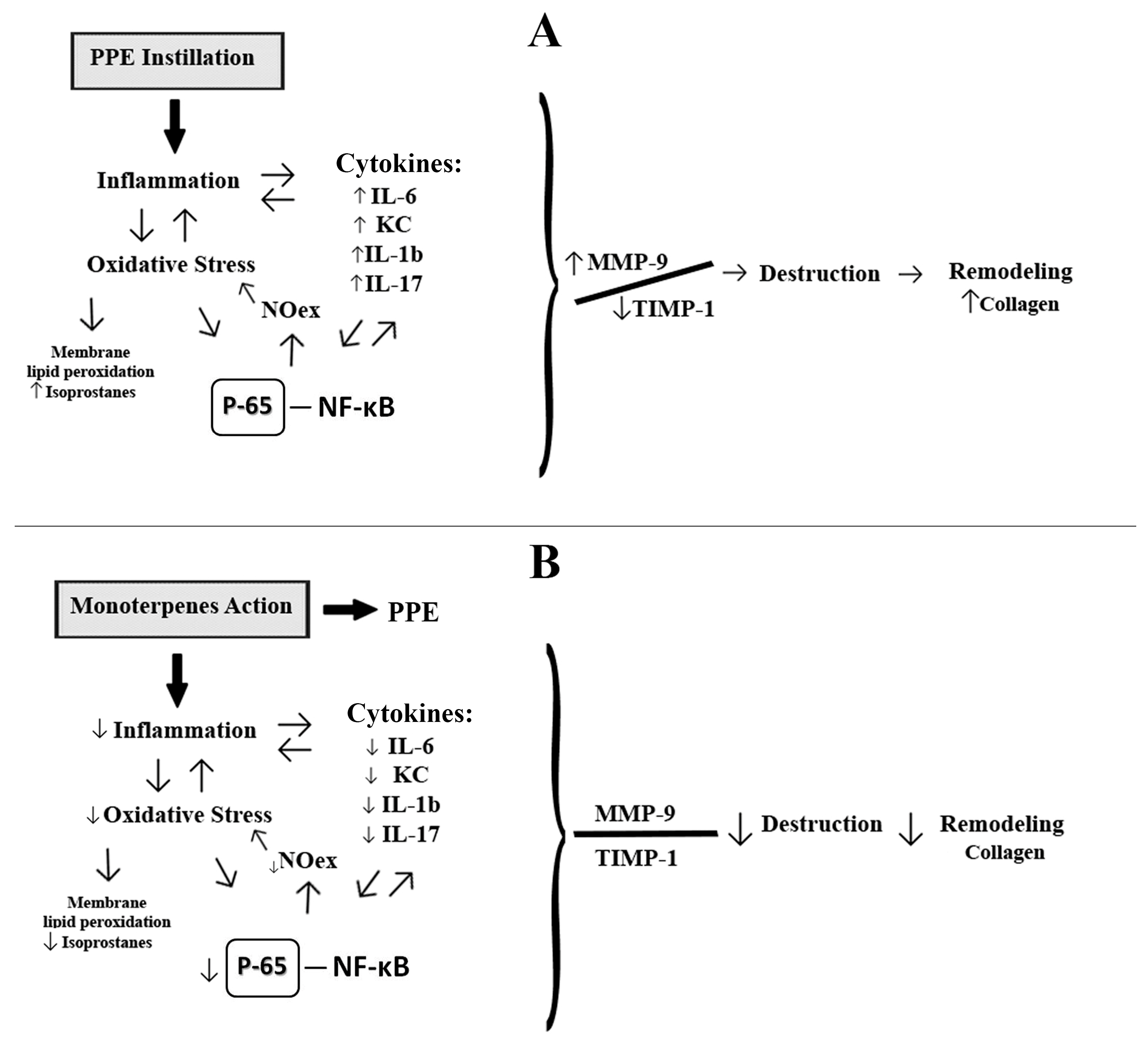
3. Discussion
4. Materials and Methods
4.1. General Experimental Procedures
4.2. Chemical and Reagents
4.3. Plant Material
4.4. Essential Oil Extraction
4.5. Isolation of Monoterpenes
4.5.1. p-Cymene
4.5.2. Carvacrol
4.5.3. Thymol
4.6. Animals
4.7. Experimental Design
4.8. PPE-Induced Emphysema in Mice
4.9. Monoterpenes Treatment
4.10. Exhaled Nitric Oxide and BALF Collection
4.11. Lung Histopathology and Immunohistochemistry
4.12. Statistical Analyses
Acknowledgments
Author Contributions
Conflicts of Interest
References
- GOLD. Global strategies for diagnosis, management and prevention of chronic obstructive pulmonary, NHLBI/WHO Workshop report. Am. J. Respir. Crit. Care. Med. 2001, 163, 1256–1276. [Google Scholar]
- Silva, O.R.; Montes, J.F.; García-Valero, J.; Olloquequi, J. Cellular effectors of the inflammatory response in chronic obstructive pulmonary disease (COPD). Rev. Med. Chil. 2015, 143, 1162–1171. [Google Scholar]
- Lappalainen, U.; Whitsett, J.A.; Wert, S.E.; Tichelaar, J.W.; Bry, K. Interleukin-1beta causes pulmonary inflammation, emphysema, and airway remodeling in the adult murine lung. Am. J. Respir. Cell. Mol. Biol. 2005, 32, 311–318. [Google Scholar] [CrossRef] [PubMed]
- Caramori, G.; Adcock, I.M.; Di Stefano, A.; Chung, K.F. Cytokine inhibition in the treatment of COPD. Int. J. Chron. Obstruct. Pulm. Dis. 2014, 9, 397–412. [Google Scholar]
- Di Stefano, A.; Caramori, G.; Gnemmi, I.; Contoli, M.; Vicari, C.; Capelli, A.; Magno, F.; D’Anna, S.E.; Zanini, A.; Brun, P.; et al. T helper type 17-related cytokine expression is increased in the bronchial mucosa of stable chronic obstructive pulmonary disease patients. Clin. Exp. Immunol. 2009, 157, 316–324. [Google Scholar] [CrossRef] [PubMed]
- Bozinovski, S.; Seow, H.J.; Chan, S.P.; Anthony, D.; McQualter, J.; Hansen, M.; Jenkins, B.J.; Anderson, G.P.; Vlahos, R. Innate cellular sources of interleukin-17A regulate macrophage accumulation in cigarette- smoke-induced lung inflammation in mice. Clin. Sci. (Lond.) 2015, 129, 785–796. [Google Scholar] [CrossRef] [PubMed]
- D’Acquisto, F.; Maione, F.; Pederzoli-Ribeil, M. From IL-15 to IL-33: The never-ending list of new players in inflammation. Is it time to forget the humble aspirin and move ahead? Biochem. Pharmacol. 2010, 79, 525–534. [Google Scholar] [CrossRef] [PubMed]
- Rainsford, K.D. Anti-inflammatory drugs in the 21st century. Subcell. Biochem. 2007, 42, 3–27. [Google Scholar] [PubMed]
- Rauf, A.; Khan, R.; Raza, M.; Khan, H.; Pervez, S.; de Feo, V.; Maione, F.; Mascolo, N. Suppression of inflammatory response by chrysin, a flavone isolated from Potentilla evestita Th. Wolf. In silico predictive study on its mechanistic effect. Fitoterapia 2015, 103, 129–135. [Google Scholar] [CrossRef] [PubMed]
- Panday, A.; Inda, M.E.; Bagam, P.; Sahoo, M.K.; Osorio, D.; Batra, S. Transcription Factor NF-κB: An Update on Intervention Strategies. Arch. Immunol. Ther. Exp. 2016. [Google Scholar] [CrossRef] [PubMed]
- Hang, C.H.; Shi, J.X.; Li, J.S.; Li, W.Q.; Wu, W. Expressions of intestinal NF-κB, TNF-alpha, and IL-6 following traumatic brain injury in rats. J. Surg. Res. 2005, 123, 188–193. [Google Scholar] [CrossRef] [PubMed]
- Stegmaier, J.C.; Kirchhoff, C.; Bogner, V.; Matz, M.; Kanz, K.G.; Mutschler, W.; Biberthaler, P. Dynamics of neutrophilic NF-κB translocation in relation to IL-8 mRNA expression after major trauma. Inflamm. Res. 2008, 57, 547–554. [Google Scholar] [CrossRef] [PubMed]
- Boots, A.W.; Haenen, G.R.; Bast, A. Oxidant metabolism in chronic obstructive pulmonary disease. Eur. Respir. J. Suppl. 2003, 46, 14s–27s. [Google Scholar] [CrossRef] [PubMed]
- Barnes, P.J.; Shapiro, S.D.; Pauwels, R.A. Chronic obstructive pulmonary disease: Molecular and cellular mechanisms. Eur. Respir. J. 2003, 22, 672–688. [Google Scholar] [CrossRef] [PubMed]
- Antczak, A.; Ciebiada, M.; Pietras, T.; Piotrowski, W.J.; Kurmanowska, Z.; Górski, P. Exhaled eicosanoids and biomarkers of oxidative stress in exacerbation of chronic obstructive pulmonary disease. Arch. Med. Sci. 2012, 8, 277–285. [Google Scholar] [CrossRef] [PubMed]
- Inonu, H.; Doruk, S.; Sahin, S.; Erkorkmaz, U.; Celik, D.; Celikel, S.; Seyfikli, Z. Oxidative stress levels in exhaled breath condensate associated with COPD and smoking. Respir. Care. 2012, 57, 413–419. [Google Scholar] [CrossRef] [PubMed]
- Antus, B.; Kardos, Z. Oxidative stress in COPD: Molecular background and clinical monitoring. Curr. Med. Chem. 2015, 22, 627–650. [Google Scholar] [CrossRef] [PubMed]
- Taguchi, L.; Pinheiro, N.M.; Olivo, C.R.; Choqueta-Toledo, A.; Grecco, S.S.; Lopes, F.D.; Caperuto, L.C.; Martins, M.A.; Tiberio, I.F.; Câmara, N.O.; et al. A flavanone from Baccharis retusa (Asteraceae) prevents elastase-induced emphysema in mice by regulating NF-κB, oxidative stress and metalloproteinases. Respir. Res. 2015, 16, 79. [Google Scholar] [CrossRef] [PubMed]
- Prado, C.M.; Leick-Maldonado, E.A.; Yano, L.; Leme, A.S.; Capelozzi, V.L.; Martins, M.A.; Tibério, I.F. Effects of nitric oxide synthases in chronic allergic airway inflammation and remodeling. Am. J. Respir. Cell. Mol. Biol. 2006, 35, 457–465. [Google Scholar] [CrossRef] [PubMed]
- Gemicioglu, B.; Musellim, B.; Dogan, I.; Guven, K. Fractional exhaled nitric oxide (FeNo) in different asthma phenotypes. Allergy Rhinol. 2014, 5, 157–161. [Google Scholar] [CrossRef] [PubMed]
- MacNee, W. Pathogenesis of chronic obstructive pulmonary disease. Proc. Am. Thorac. Soc. 2005, 2, 258–266. [Google Scholar] [CrossRef] [PubMed]
- Grzela, K.; Litwiniuk, M.; Zagorska, W.; Grzela, T. Airway Remodeling in Chronic Obstructive Pulmonary Disease and Asthma: The Role of Matrix Metalloproteinase-9. Arch. Immunol. Ther. Exp. (Warsz.) 2016, 64, 47–55. [Google Scholar] [CrossRef] [PubMed]
- Maione, F.; Russo, R.; Khan, H.; Mascolo, N. Medicinal plants with anti-inflammatory activities. Nat. Prod. Res. 2016, 30, 1343–1352. [Google Scholar] [CrossRef] [PubMed]
- Khan, H. Medicinal Plants in Light of History: Recognized Therapeutic Modality. J. Evid. Based Complement. Altern. Med. 2014, 19, 216–219. [Google Scholar] [CrossRef] [PubMed]
- Bakkali, F.; Averbeck, S.; Averbeck, D.; Idaomar, M. Biological effects of essential oils—A review. Food Chem. Toxicol. 2008, 46, 446–475. [Google Scholar] [CrossRef] [PubMed]
- Riachi, L.G.; De Maria, C.A. Peppermint antioxidants revisited. Food Chem. 2015, 176, 72–81. [Google Scholar] [CrossRef] [PubMed]
- Soares, S.E. Ácidos fenólicos como antioxidantes. Rev. Nutr. 2002, 15, 71–81. [Google Scholar] [CrossRef]
- Burt, S. Essential oils: Their antibacterial properties and potential applications in foods—A review. Int. J. Food Microbiol. 2004, 94, 223–253. [Google Scholar] [CrossRef] [PubMed]
- Ruberto, G.; Baratta, M.T.; Deans, S.G.; Dorman, H.J. Antioxidant and antimicrobial activity of Foeniculum vulgare and Crithmum maritimum essential oils. Planta Med. 2000, 66, 687–693. [Google Scholar] [CrossRef] [PubMed]
- De Araújo, D.A.; Freitas, C.; Cruz, J.S. Essential oils components as a new path to understand ion channel molecular pharmacology. Life Sci. 2011, 89, 540–544. [Google Scholar] [CrossRef] [PubMed]
- Rahman, A.U.; Ahmad, V.U. 13C-NMR of Natural Products: Monoterpenes and sesquiterpenes; Plenum Press: New York, NY, USA, 1992; Volume 1. [Google Scholar]
- Adams, R.P. Identification of Essential oils by Ion Trap Mass Spectroscopy; Academic Press: San Diego, CA, USA, 1995; p. 465. [Google Scholar]
- Wright, J.L.; Cosio, M.; Churg, A. Animal models of chronic obstructive pulmonary disease. Am. J. Physiol. Lung Cell. Mol. Physiol. 2008, 295, L1–L15. [Google Scholar] [CrossRef] [PubMed]
- Lopes, F.D.; Toledo, A.C.; Olivo, C.R.; Prado, C.M.; Leick, E.A.; Medeiros, M.C.; Santos, A.B.; Garippo, A.; Martins, M.A.; Mauad, T. A comparative study of extracellular matrix remodeling in two murine models of emphysema. Histol. Histopathol. 2013, 28, 269–276. [Google Scholar] [PubMed]
- Antunes, M.A.; Rocco, P.R. Elastase-induced pulmonary emphysema: Insights from experimental models. An. Acad. Bras. Cienc. 2011, 83, 1385–1396. [Google Scholar] [CrossRef] [PubMed]
- Ohnishi, S.; Nagaya, N. Tissue regeneration as next-generation therapy for COPD—Potential applications. Int. J. Chron. Obstruct Pulm. Dis. 2008, 3, 509–514. [Google Scholar]
- Muñoz-Barrutia, A.; Ceresa, M.; Artaechevarria, X.; Montuenga, L.M.; Ortiz-de-Solorzano, C. Quantification of lung damage in an elastase-induced mouse model of emphysema. Int. J. Biomed. Imaging 2012, 2012, 734734. [Google Scholar] [CrossRef] [PubMed]
- WORLD HEALTH ORGANIZATION (WHO). Available online: http://www.who.int/respiratory/copd/burden/en/ (accessed on 2 July 2016).
- Cavalcante, A.G.; de Bruin, P.F. The role of oxidative stress in COPD: Current concepts and perspectives. J. Bras. Pneumol. 2009, 35, 1227–1237. [Google Scholar] [PubMed]
- Kurimoto, E.; Miyahara, N.; Kanehiro, A.; Waseda, K.; Taniguchi, A.; Ikeda, G.; Koga, H.; Nishimori, H.; Tanimoto, Y.; Kataoka, M.; et al. IL-17A is essential to the development of elastase-induced pulmonary inflammation and emphysema in mice. Respir. Res. 2013, 14, 5. [Google Scholar] [CrossRef] [PubMed]
- Lourenço, J.D.; Neves, L.P.; Olivo, C.R.; Duran, A.; Almeida, F.M.; Arantes, P.M.; Prado, C.M.; Leick, E.A.; Tanaka, A.S.; Martins, M.A.; et al. A treatment with a protease inhibitor recombinant from the cattle tick (Rhipicephalus Boophilus microplus) ameliorates emphysema in mice. PLoS ONE 2014, 9, e98216. [Google Scholar] [CrossRef] [PubMed]
- Barnes, P.J. Alveolar macrophages as orchestrators of COPD. COPD 2004, 1, 59–70. [Google Scholar] [CrossRef] [PubMed]
- Murugan, V.; Peck, M.J. Signal transduction pathways linking the activation of alveolar macrophages with the recruitment of neutrophils to lungs in chronic obstructive pulmonary disease. Exp. Lung Res. 2009, 35, 439–485. [Google Scholar] [CrossRef] [PubMed]
- Pappas, K.; Papaioannou, A.I.; Kostikas, K.; Tzanakis, N. The role of macrophages in obstructive airways disease: Chronic obstructive pulmonary disease and asthma. Cytokine 2013, 64, 613–625. [Google Scholar] [CrossRef] [PubMed]
- Rufino, R.; Lapa e Silva, J.R. Cellular and biochemical bases of chronic obstructive pulmonary disease. J. Bras. Pneumol. 2006, 32, 241–248. [Google Scholar] [CrossRef] [PubMed]
- Bozic, C.R.; Gerard, N.P.; von Uexkull-Guldenband, C.; Kolakowski, L.F.; Conklyn, M.J.; Breslow, R.; Showell, H.J.; Gerard, C. The murine interleukin 8 type B receptor homologue and its ligands. Expression and biological characterization. J. Biol. Chem. 1994, 269, 29355–29358. [Google Scholar] [PubMed]
- Tanino, M.; Betsuyaku, T.; Takeyabu, K.; Tanino, Y.; Yamaguchi, E.; Miyamoto, K.; Nishimura, M. Increased levels of interleukin-8 in BAL fluid from smokers susceptible to pulmonary emphysema. Thorax 2002, 57, 405–411. [Google Scholar] [CrossRef] [PubMed]
- Soler, N.; Ewig, S.; Torres, A.; Filella, X.; Gonzalez, J.; Zaubet, A. Airway inflammation and bronchial microbial patterns in patients with stable chronic obstructive pulmonary disease. Eur. Respir. J. 1999, 14, 1015–1022. [Google Scholar] [CrossRef] [PubMed]
- Lorè, N.I.; Bragonzi, A.; Cigana, C. The IL-17A/IL-17RA axis in pulmonary defence and immunopathology. Cytokine Growth Factor Rev. 2016, 30, 19–27. [Google Scholar] [CrossRef] [PubMed]
- Krstic, J.; Obradovic, H.; Kukolj, T.; Mojsilovic, S.; Okic-Dordevic, I.; Bugarski, D.; Santibanez, J.F. An Overview of Interleukin-17A and Interleukin-17 Receptor A Structure, Interaction and Signaling. Protein Pept. Lett. 2015, 22, 570–578. [Google Scholar] [CrossRef] [PubMed]
- Maione, F.; Paschalidis, N.; Mascolo, N.; Dufton, N.; Perretti, M.; D’Acquisto, F. Interleukin 17 sustains rather than induces inflammation. Biochem. Pharmacol. 2009, 77, 878–887. [Google Scholar] [CrossRef] [PubMed]
- Willis, C.R.; Siegel, L.; Leith, A.; Mohn, D.; Escobar, S.; Wannberg, S.; Misura, K.; Rickel, E.; Rottman, J.B.; Comeau, M.R.; et al. IL-17RA Signaling in Airway Inflammation and Bronchial Hyperreactivity in Allergic Asthma. Am. J. Respir. Cell. Mol. Biol. 2015, 53, 810–821. [Google Scholar] [CrossRef] [PubMed]
- Jones, C.E.; Chan, K. Interleukin-17 stimulates the expression of interleukin-8, growth-related oncogene-alpha, and granulocyte-colony-stimulating factor by human airway epithelial cells. Am. J. Respir. Cell. Mol. Biol. 2002, 26, 748–753. [Google Scholar] [CrossRef] [PubMed]
- Rahman, M.S.; Yang, J.; Shan, L.Y.; Unruh, H.; Yang, X.; Halayko, A.J.; Gounni, A.S. IL-17R activation of human airway smooth muscle cells induces CXCL-8 production via a transcriptional-dependent mechanism. Clin. Immunol. 2005, 115, 268–276. [Google Scholar] [CrossRef] [PubMed]
- Zhang, L.; Cheng, Z.; Liu, W.; Wu, K. Expression of interleukin (IL)-10, IL-17A and IL-22 in serum and sputum of stable chronic obstructive pulmonary disease patients. COPD 2013, 10, 459–465. [Google Scholar] [CrossRef] [PubMed]
- Xie, G.; Chen, N.; Soromou, L.W.; Liu, F.; Xiong, Y.; Wu, Q.; Li, H.; Feng, H.; Liu, G. p-Cymene protects mice against lipopolysaccharide-induced acute lung injury by inhibiting inflammatory cell activation. Molecules 2012, 17, 8159–8173. [Google Scholar] [CrossRef] [PubMed]
- Boskabady, M.H.; Gholami Mahtaj, L. Lung inflammation changes and oxidative stress induced by cigarette smoke exposure in guinea pigs affected by Zataria multiflora and its constituent, carvacrol. BMC Complement. Altern. Med. 2015, 15, 39. [Google Scholar] [CrossRef] [PubMed]
- Sun, J.; Wu, J.; Xu, C.; Luo, Q.; Li, B.; Dong, J. Paeoniflorin attenuates allergic inflammation in asthmatic mice. Int. Immunopharmacol. 2015, 24, 88–94. [Google Scholar] [CrossRef] [PubMed]
- Zhao, C.; Sun, J.; Fang, C.; Tang, F. 1,8-cineol attenuates LPS-induced acute pulmonary inflammation in mice. Inflammation 2014, 37, 566–572. [Google Scholar] [CrossRef] [PubMed]
- Zhou, E.; Fu, Y.; Wei, Z.; Yu, Y.; Zhang, X.; Yang, Z. Thymol attenuates allergic airway inflammation in ovalbumin (OVA)-induced mouse asthma. Fitoterapia 2014, 96, 131–137. [Google Scholar] [CrossRef] [PubMed]
- Cataldo, D.D.; Bettiol, J.; Noël, A.; Bartsch, P.; Foidart, J.M.; Louis, R. Matrix metalloproteinase-9, but not tissue inhibitor of matrix metalloproteinase-1, increases in the sputum from allergic asthmatic patients after allergen challenge. Chest 2002, 122, 1553–1559. [Google Scholar] [CrossRef] [PubMed]
- Gagliardo, R.; La Grutta, S.; Chanez, P.; Profita, M.; Paternò, A.; Cibella, F.; Bousquet, J.; Viegi, G.; Gjomarkaj, M. Non-invasive markers of airway inflammation and remodeling in childhood asthma. Pediatr. Allergy Immunol. 2009, 20, 780–790. [Google Scholar] [CrossRef] [PubMed]
- Atkinson, J.J.; Senior, R.M. Matrix metalloproteinase-9 in lung remodeling. Am. J. Respir. Cell. Mol. Biol. 2003, 28, 12–24. [Google Scholar] [CrossRef] [PubMed]
- Blackwell, T.S.; Christman, J.W. The role of nuclear factor-kappa B in cytokine gene regulation. Am. J. Respir. Cell. Mol. Biol. 1997, 17, 3–9. [Google Scholar] [CrossRef] [PubMed]
- Schuliga, M. NF-κB Signaling in Chronic Inflammatory Airway Disease. Biomolecules 2015, 5, 1266–1283. [Google Scholar] [CrossRef] [PubMed]
- Edwards, M.R.; Bartlett, N.W.; Clarke, D.; Birrell, M.; Belvisi, M.; Johnston, S.L. Targeting the NF-κB pathway in asthma and chronic obstructive pulmonary disease. Pharmacol. Ther. 2009, 121, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Rajendrasozhan, S.; Yang, S.R.; Edirisinghe, I.; Yao, H.; Adenuga, D.; Rahman, I. Deacetylases and NF-κB in redox regulation of cigarette smoke-induced lung inflammation: Epigenetics in pathogenesis of COPD. Antioxid. Redox Signal. 2008, 10, 799–811. [Google Scholar] [CrossRef] [PubMed]
- Ko, F.W.; Lau, C.Y.; Leung, T.F.; Wong, G.W.; Lam, C.W.; Hui, D.S. Exhaled breath condensate levels of 8-isoprostane, growth related oncogene alpha and monocyte chemoattractant protein-1 in patients with chronic obstructive pulmonary disease. Respir. Med. 2006, 100, 630–638. [Google Scholar] [CrossRef] [PubMed]
- Tufvesson, E.; Ekberg, M.; Bjermer, L. Inflammatory biomarkers in sputum predict COPD exacerbations. Lung 2013, 191, 413–416. [Google Scholar] [CrossRef] [PubMed]
- Anciães, A.M.; Olivo, C.R.; Prado, C.M.; Kagohara, K.H.; Silva Pinto, T.; Moriya, H.T.; Mauad, T.; Arruda Martins, M.; Lopes, F.D. Respiratory mechanics do not always mirror pulmonary histological changes in emphysema. Clinics (Sao Paulo) 2011, 66, 1797–1803. [Google Scholar] [PubMed]
- Toledo, A.C.; Magalhaes, R.M.; Hizume, D.C.; Vieira, R.P.; Biselli, P.J.; Moriya, H.T.; Mauad, T.; Lopes, F.D.; Martins, M.A. Aerobic exercise attenuates pulmonary injury induced by exposure to cigarette smoke. Eur. Respir. J. 2012, 39, 254–264. [Google Scholar] [CrossRef] [PubMed]
- Malerba, M.; Radaeli, A.; Olivini, A.; Damiani, G.; Ragnoli, B.; Montuschi, P.; Ricciardolo, F.L. Exhaled nitric oxide as a biomarker in COPD and related comorbidities. BioMed Res. Int. 2014, 2014, 271918. [Google Scholar] [CrossRef] [PubMed]
- Baratta, M.T.; Dorman, H.J.; Deans, S.G.; Figueiredo, A.C.; Barroso, J.G.; Ruberto, G. Antimicrobial and antioxidant properties of some commercial essential oils. Flavour Fragr. J. 1998, 13, 235–244. [Google Scholar] [CrossRef]
- Walde, A.; Ve, B.; Scheline, R.R.; Monge, P. p-Cymene metabolism in rats and guinea-pigs. Xenobiotica 1983, 13, 503–512. [Google Scholar] [CrossRef] [PubMed]
- Khan, M.S.; Ahmad, I.; Cameotra, S.S. Carum copticum and Thymus vulgaris oils inhibit virulence in Trichophyton rubrum and Aspergillus spp. Braz. J. Microbiol. 2014, 45, 523–531. [Google Scholar] [CrossRef] [PubMed]
- Sivamani, P.; Singaravelu, G.; Thiagarajan, V.; Jayalakshmi, T.; Ramesh Kumar, G. Comparative molecular docking analysis of essential oil constituents as elastase inhibitors. Bioinformation 2012, 8, 457–460. [Google Scholar] [CrossRef] [PubMed]
- Kacem, R.; Meraihi, Z. Effects of essential oil extracted from Nigella sativa (L.) seeds and its main components on human neutrophil elastase activity. Yakugaku Zasshi 2006, 126, 301–305. [Google Scholar] [CrossRef] [PubMed]
- Toledo, A.C.; Sakoda, C.P.; Perini, A.; Pinheiro, N.M.; Magalhães, R.M.; Grecco, S.; Tibério, I.F.; Câmara, N.O.; Martins, M.A.; Lago, J.H.; et al. Flavonone treatment reverses airway inflammation and remodelling in an asthma murine model. Br. J. Pharmacol. 2013, 168, 1736–1749. [Google Scholar] [CrossRef] [PubMed]
- Perea, S.E.; Baladron, I.; Garcia, Y.; Perera, Y.; Lopez, A.; Soriano, J.L.; Batista, N.; Palau, A.; Hernández, I.; Farina, H.; et al. CIGB-300, a synthetic peptide-based drug that targets the CK2 phosphoaceptor domain. Translational and clinical research. Mol. Cell. Biochem. 2011, 356, 45–50. [Google Scholar] [CrossRef] [PubMed]
- Sample Availability: Samples of the compounds are available from the authors.
© 2016 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Games, E.; Guerreiro, M.; Santana, F.R.; Pinheiro, N.M.; De Oliveira, E.A.; Lopes, F.D.T.Q.S.; Olivo, C.R.; Tibério, I.F.L.C.; Martins, M.A.; Lago, J.H.G.; et al. Structurally Related Monoterpenes p-Cymene, Carvacrol and Thymol Isolated from Essential Oil from Leaves of Lippia sidoides Cham. (Verbenaceae) Protect Mice against Elastase-Induced Emphysema. Molecules 2016, 21, 1390. https://doi.org/10.3390/molecules21101390
Games E, Guerreiro M, Santana FR, Pinheiro NM, De Oliveira EA, Lopes FDTQS, Olivo CR, Tibério IFLC, Martins MA, Lago JHG, et al. Structurally Related Monoterpenes p-Cymene, Carvacrol and Thymol Isolated from Essential Oil from Leaves of Lippia sidoides Cham. (Verbenaceae) Protect Mice against Elastase-Induced Emphysema. Molecules. 2016; 21(10):1390. https://doi.org/10.3390/molecules21101390
Chicago/Turabian StyleGames, Ellen, Marina Guerreiro, Fernanda R. Santana, Nathalia M. Pinheiro, Emerson A. De Oliveira, Fernanda D.T.Q.S. Lopes, Clarice R. Olivo, Iolanda F.L.C. Tibério, Mílton A. Martins, João Henrique G. Lago, and et al. 2016. "Structurally Related Monoterpenes p-Cymene, Carvacrol and Thymol Isolated from Essential Oil from Leaves of Lippia sidoides Cham. (Verbenaceae) Protect Mice against Elastase-Induced Emphysema" Molecules 21, no. 10: 1390. https://doi.org/10.3390/molecules21101390
APA StyleGames, E., Guerreiro, M., Santana, F. R., Pinheiro, N. M., De Oliveira, E. A., Lopes, F. D. T. Q. S., Olivo, C. R., Tibério, I. F. L. C., Martins, M. A., Lago, J. H. G., & Prado, C. M. (2016). Structurally Related Monoterpenes p-Cymene, Carvacrol and Thymol Isolated from Essential Oil from Leaves of Lippia sidoides Cham. (Verbenaceae) Protect Mice against Elastase-Induced Emphysema. Molecules, 21(10), 1390. https://doi.org/10.3390/molecules21101390






