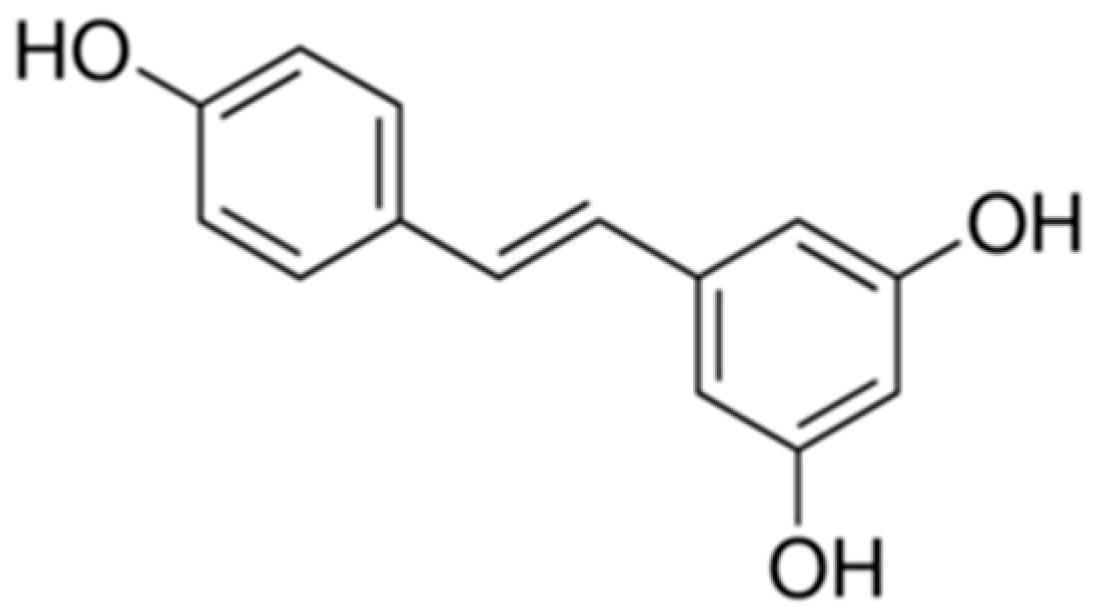
Resveratrol Ameliorates the Depressive-Like Behaviors and Metabolic Abnormalities Induced by Chronic Corticosterone Injection
Abstract
:1. Introduction
2. Results
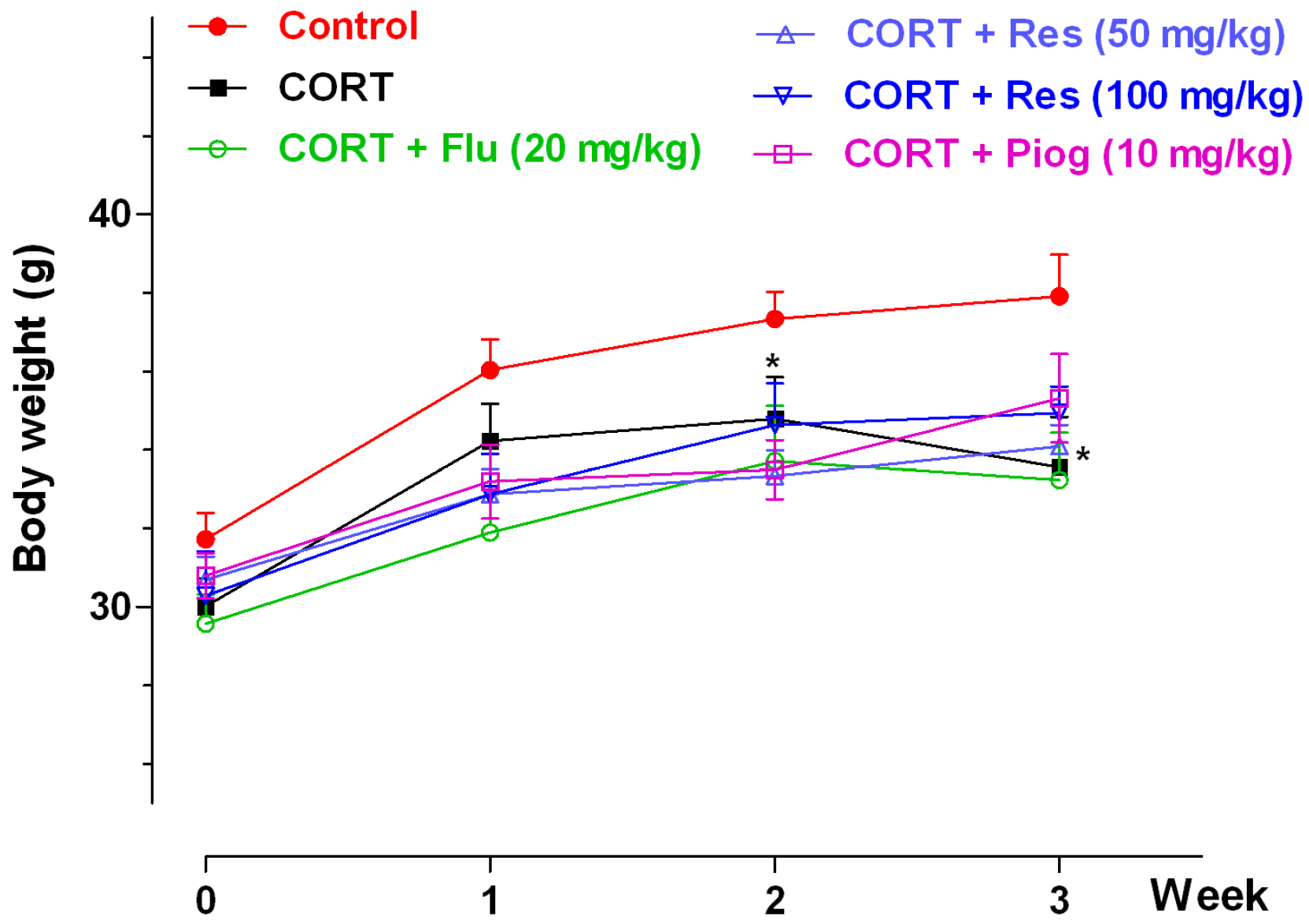
2.1. Effects of Resveratrol on Body Weight
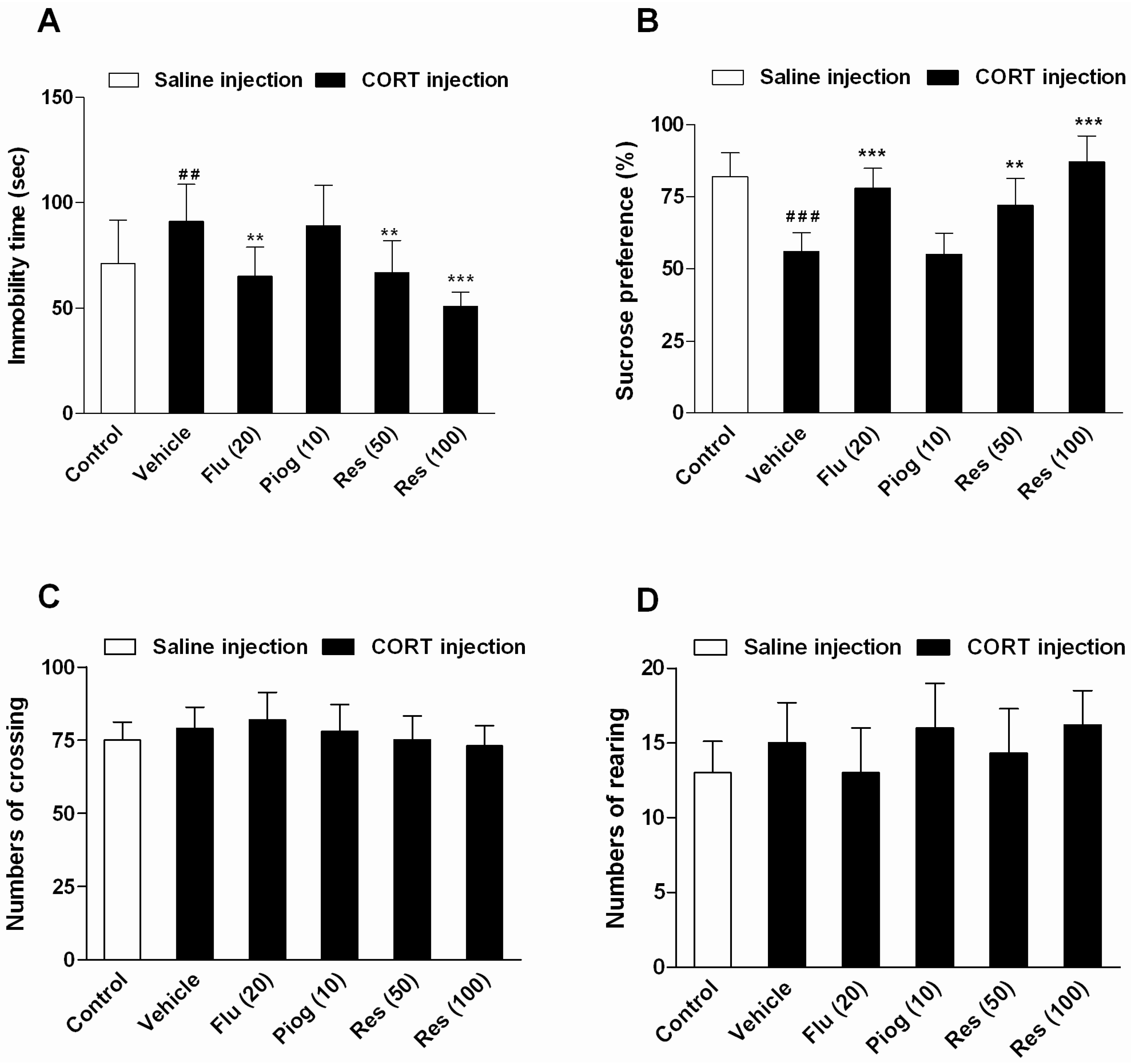
2.2. Effects of Resveratrol on Depressive-Like Behaviors Induced by CORT
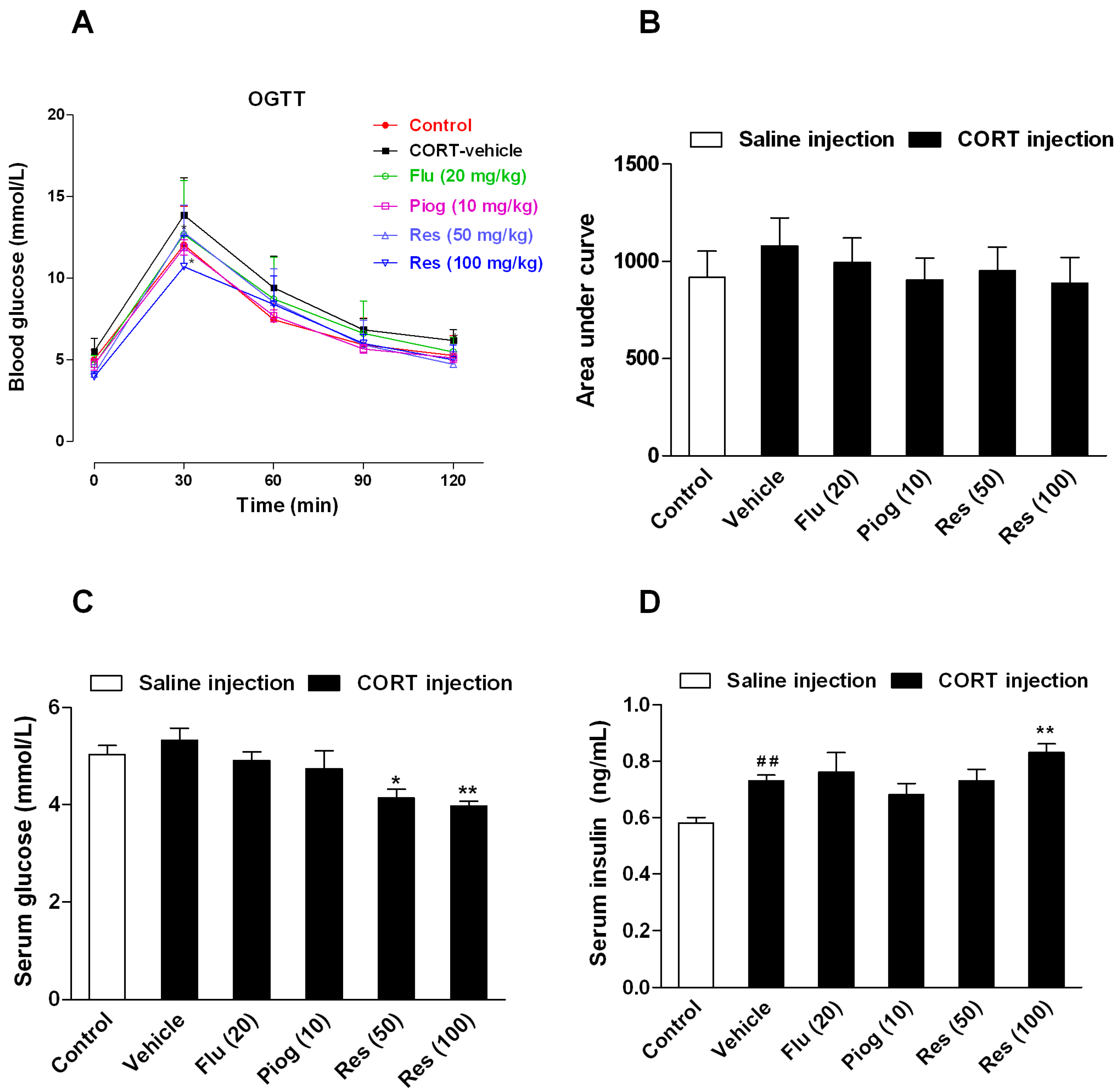
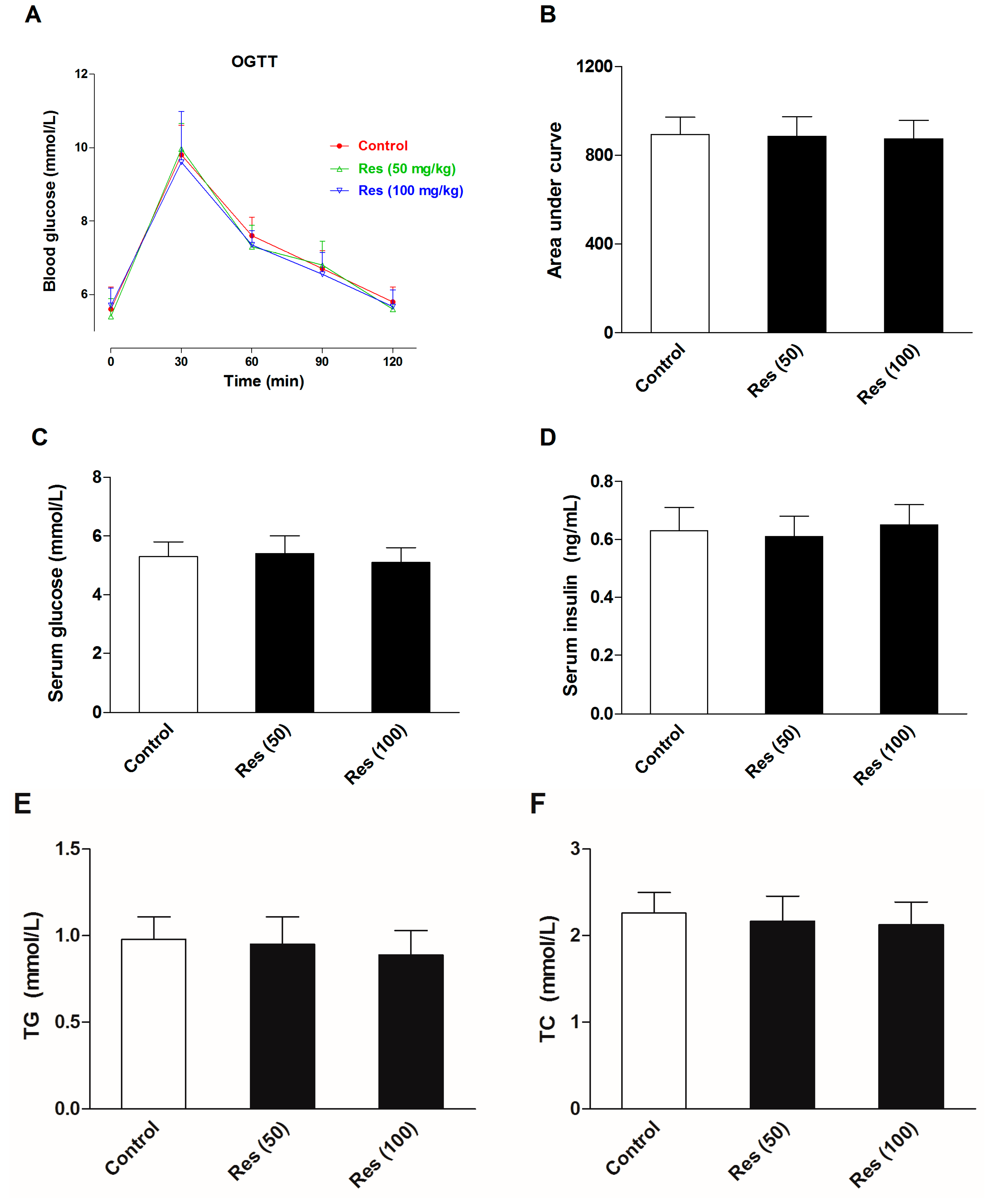
2.3. Effects of Resveratrol on the Insulin Sensitivity in CORT-Treated Mice
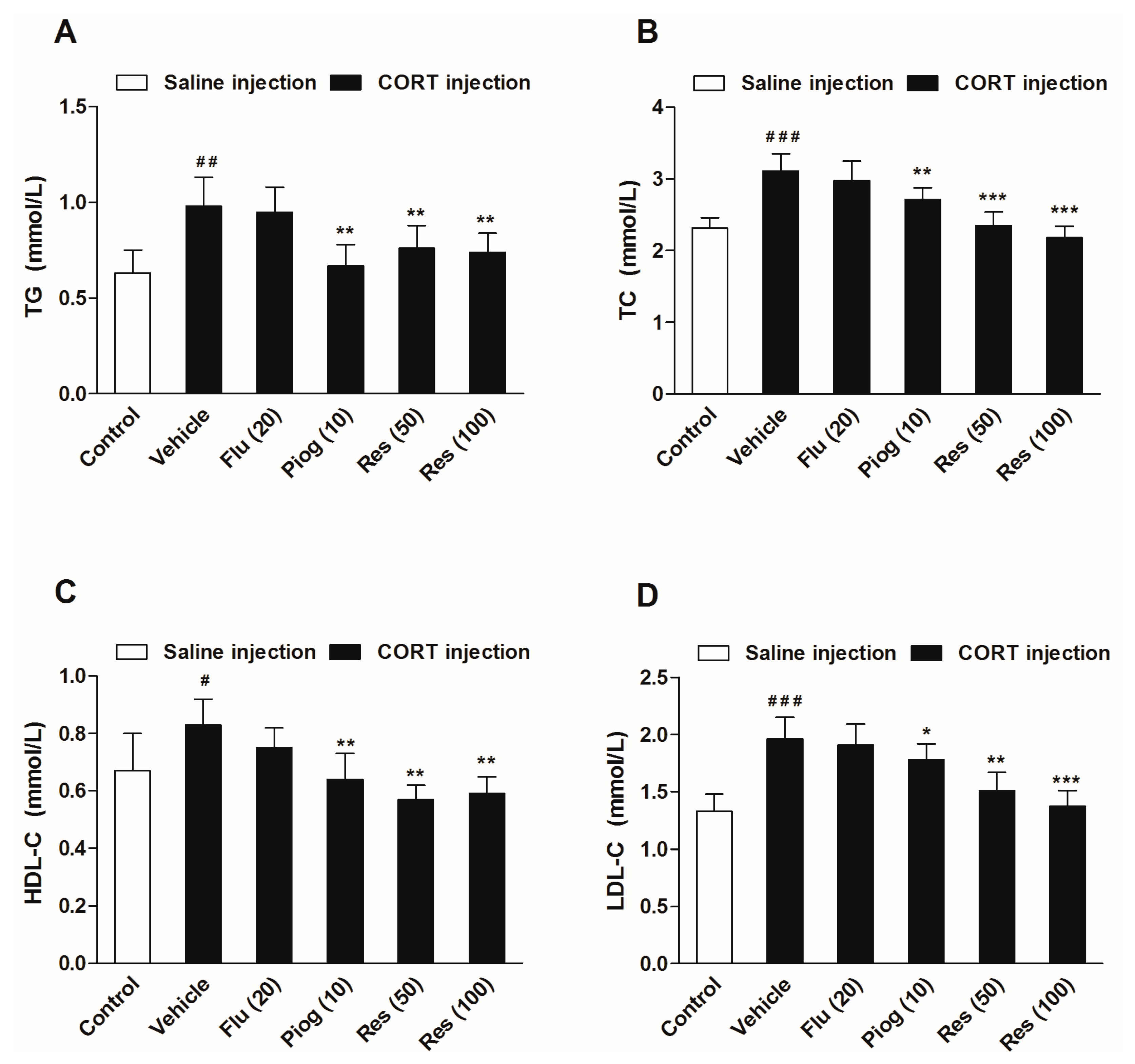
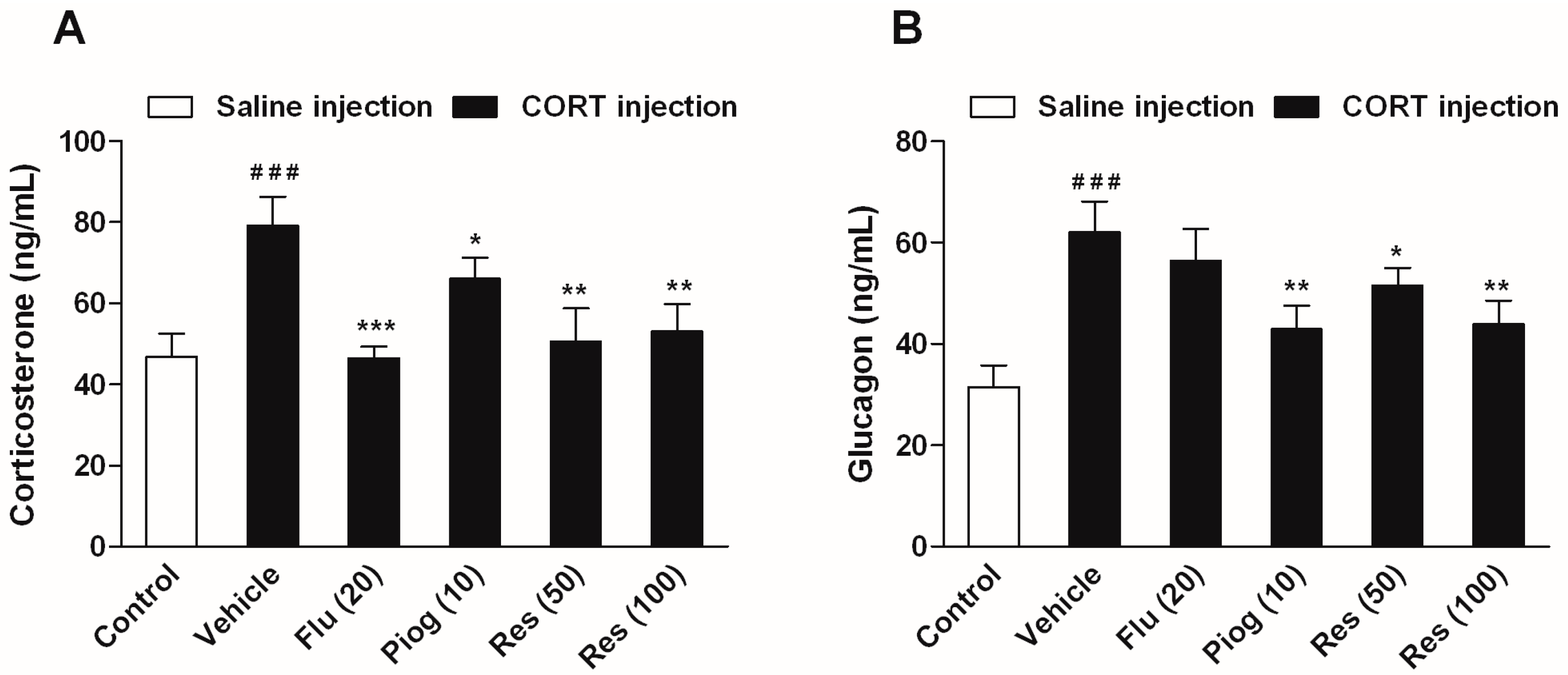
2.4. Effects of Resveratrol on the Serum Level of Lipids and Hormones in CORT-Treated Mice
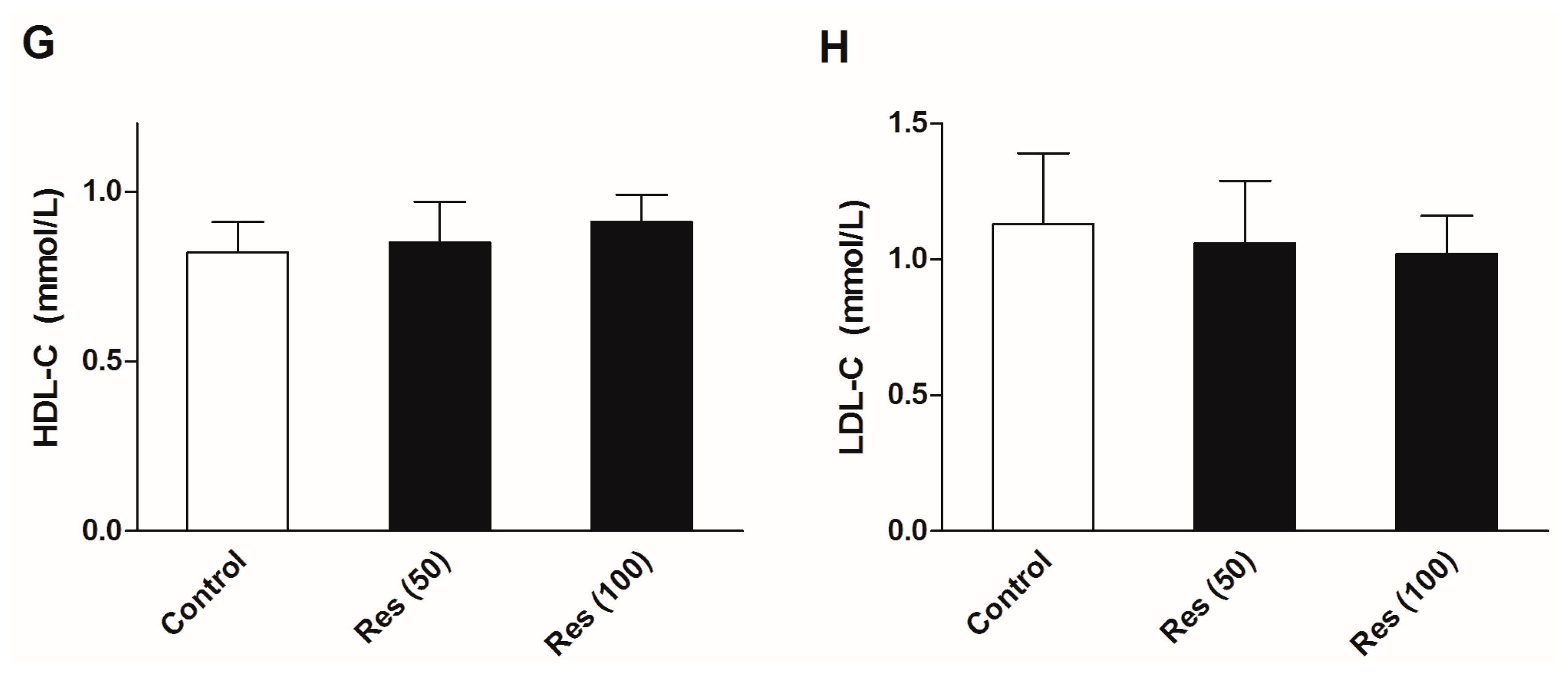
2.5. Effects of Resveratrol on the Serum Biochemical Levels in Normal Mice
3. Discussion
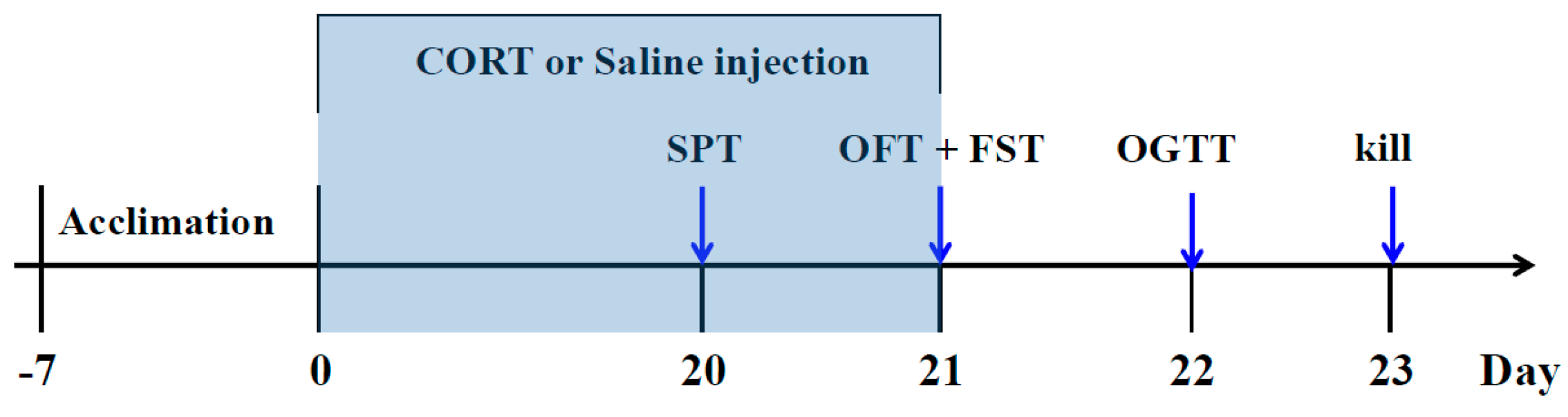
4. Materials and Methods
4.1. Chemicals and Reagents
4.2. Animals
4.3. The CORT-Treated Procedure
4.4. The Depressive-Like Behavior Test
4.5. Oral Glucose Tolerance Test (OGTT)
4.6. Serum Biochemical Assay
4.7. Statistical Analysis
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Boyle, M.P.; Kolber, B.J.; Vogt, S.K.; Wozniak, D.F.; Muglia, L.J. Forebrain glucocorticoid receptors modulate anxiety-associated locomotor activation and adrenal responsiveness. J. Neurosci. 2006, 26, 1971–1978. [Google Scholar] [CrossRef] [PubMed]
- Anagnostis, P.; Athyros, V.G.; Tziomalos, K.; Karagiannis, A.; Mikhailidis, D.P. Clinical review: The pathogenetic role of cortisol in the metabolic syndrome: A hypothesis. J. Clin. Endocrinol. Metab. 2009, 94, 2692–2701. [Google Scholar] [CrossRef] [PubMed]
- Kassi, E.; Pervanidou, P.; Kaltsas, G.; Chrousos, G. Metabolic syndrome: Definitions and controversies. BMC Med. 2011, 9. [Google Scholar] [CrossRef] [PubMed]
- Després, J.P. Body fat distribution and risk of cardiovascular disease: An update. Circulation 2012, 126, 1301–1313. [Google Scholar] [CrossRef] [PubMed]
- Liu, A.; Abbasi, F.; Reaven, G.M. Adiposity indices in the prediction of metabolic abnormalities associated with cardiovascular disease in non-diabetic adults. Nutr. Metab. Cardiovasc. Dis. 2011, 21, 553–560. [Google Scholar] [CrossRef] [PubMed]
- Anacker, C.; Cattaneo, A.; Luoni, A.; Musaelyan, K.; Zunszain, P.A.; Milanesi, E.; Rybka, J.; Berry, A.; Cirulli, F.; Thuret, S.; et al. Glucocorticoid-related molecular signaling pathways regulating hippocampal neurogenesis. Neuropsychopharmacology 2013, 38, 872–873. [Google Scholar] [CrossRef] [PubMed]
- Gould, E.; Cameron, H.A.; Daniels, D.C.; Woolley, C.S.; McEwen, B.S. Adrenal hormones suppress cell division in the adult rat dentate gyrus. J. Neurosci. 1992, 12, 3642–3650. [Google Scholar] [PubMed]
- Stranahan, A.M.; Arumugam, T.V.; Cutler, R.G.; Lee, K.; Egan, J.M.; Mattson, M.P. Diabetes impairs hippocampal function through glucocorticoid-mediated effects on new and mature neurons. Nat. Neurosci. 2008, 11, 309–317. [Google Scholar] [CrossRef] [PubMed]
- Pariante, C.M. Risk factors for development of depression and psychosis. Glucocorticoid receptors and pituitary implications for treatment with antidepressant and glucocorticoids. Ann. N. Y. Acad. Sci. 2009, 1179, 144–152. [Google Scholar] [CrossRef] [PubMed]
- Li, M.; Fu, Q.; Li, Y.; Li, S.; Xue, J.; Ma, S. Emodin opposes chronic unpredictable mild stress induced depressive-like behavior in mice by upregulating the levels of hippocampal glucocorticoid receptor and brain-derived neurotrophic factor. Fitoterapia 2014, 98, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Yi, L.T.; Luo, L.; Wu, Y.J.; Liu, B.B.; Liu, X.L.; Geng, D.; Liu, Q. Circadian variations in behaviors, BDNF and cell proliferation in depressive mice. Metab. Brain Dis. 2015, 30, 1495–1503. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.C.; Wang, L.L.; Pei, Y.Y.; Shen, J.D.; Li, H.B.; Wang, B.Y.; Bai, M. Baicalin decreases SGK1 expression in the hippocampus and reverses depressive-like behaviors induced by corticosterone. Neuroscience 2015, 311, 130–137. [Google Scholar] [CrossRef] [PubMed]
- Wu, T.C.; Chen, H.T.; Chang, H.Y.; Yang, C.Y.; Hsiao, M.C.; Cheng, M.L.; Chen, J.C. Mineralocorticoid receptor antagonist spironolactone prevents chronic corticosterone induced depressive-like behavior. Psychoneuroendocrinology 2013, 38, 871–873. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Y.; Ma, R.; Shen, J.; Su, H.; Xing, D.; Du, L. A mouse model of depression induced by repeated corticosterone injections. Eur. J. Pharmacol. 2008, 581, 113–120. [Google Scholar] [CrossRef] [PubMed]
- Van Donkelaar, E.L.; Vaessen, K.R.; Pawluski, J.L.; Sierksma, A.S.; Blokland, A.; Cañete, R.; Steinbusch, H.W. Long-term corticosterone exposure decreases insulin sensitivity and induces depressive-like behaviour in the C57BL/6NCrl mouse. PLoS ONE 2014, 9, e106960. [Google Scholar] [CrossRef] [PubMed]
- Dunbar, J.A.; Reddy, P.; Davis-Lameloise, N.; Philpot, B.; Laatikainen, T.; Kilkkinen, A.; Bunker, S.J.; Best, J.D.; Vartiainen, E.; Lo, S.K.; et al. Depression: An important comorbidity with metabolic syndrome in a general population. Diabetes Care 2008, 31, 2368–2372. [Google Scholar] [CrossRef] [PubMed]
- Skilton, M.R.; Moulin, P.; Terra, J.L.; Bonnet, F. Associations between anxiety, depression, and the metabolic syndrome. Biol. Psychiatry 2007, 62, 1251–1257. [Google Scholar] [CrossRef] [PubMed]
- Bratek, A.; Koźmin-Burzyńska, A.; Górniak, E.; Krysta, K. Psychiatric disorders associated with Cushing’s syndrome. Psychiatr. Danub. 2015, 27 (Suppl. 1), S339–S343. [Google Scholar] [PubMed]
- Kelly, W.F.; Checkley, S.A.; Bender, D.A.; Mashiter, K. Cushing’s syndrome and depression—A prospective study of 26 patients. Br. J. Psychiatry 1983, 142, 16–19. [Google Scholar] [CrossRef] [PubMed]
- Saud, S.M.; Li, W.; Morris, N.L.; Matter, M.S.; Colburn, N.H.; Kim, Y.S.; Young, M.R. Resveratrol prevents tumorigenesis in mouse model of Kras activated sporadic colorectal cancer by suppressing oncogenic Kras expression. Carcinogenesis 2014, 35, 2778–2786. [Google Scholar] [CrossRef] [PubMed]
- Yao, J.; Wang, J.Y.; Liu, L.; Li, Y.X.; Xun, A.Y.; Zeng, W.S.; Jia, C.H.; Wei, X.X.; Feng, J.L.; Zhao, L.; et al. Anti-oxidant effects of resveratrol on mice with DSS-induced ulcerative colitis. Arch. Med. Res. 2010, 41, 288–294. [Google Scholar] [CrossRef] [PubMed]
- Ge, L.; Liu, L.; Liu, H.; Liu, S.; Xue, H.; Wang, X.; Yuan, L.; Wang, Z.; Liu, D. Resveratrol abrogates lipopolysaccharide-induced depressive-like behavior, neuro-inflammatory response, and CREB/BDNF signaling in mice. Eur. J. Pharmacol. 2015, 768, 49–57. [Google Scholar] [CrossRef] [PubMed]
- Liu, S.; Li, T.; Liu, H.; Wang, X.; Bo, S.; Xie, Y.; Bai, X.; Wu, L.; Wang, Z.; Liu, D. Resveratrol exerts antidepressant properties in the chronic unpredictable mild stress model through the regulation of oxidative stress and mTOR pathway in the rat hippocampus and prefrontal cortex. Behav. Brain. Res. 2016, 302, 191–199. [Google Scholar] [CrossRef] [PubMed]
- Zhao, X.; Yu, C.; Wang, C.; Zhang, J.F.; Zhou, W.H.; Cui, W.G.; Ye, F.; Xu, Y. Chronic resveratrol treatment exerts antihyperalgesic effect and corrects co-morbid depressive like behaviors in mice with mononeuropathy: Involvement of serotonergic system. Neuropharmacology 2014, 85, 131–141. [Google Scholar] [CrossRef] [PubMed]
- Ali, S.H.; Madhana, R.M.; Athira, K.V.; Kasala, E.R.; Bodduluru, L.N.; Pitta, S.; Mahareddy, J.R.; Lahkar, M. Resveratrol ameliorates depressive-like behavior in repeated corticosterone-induced depression in mice. Steroids 2015, 101, 37–42. [Google Scholar] [CrossRef] [PubMed]
- Willner, P.; Towell, A.; Sampson, D.; Sophokleous, S.; Muscat, R. Reduction of sucrose preference by chronic unpredictable mild stress, and its restoration by a tricyclic antidepressant. Psychopharmacology 1987, 93, 358–364. [Google Scholar] [CrossRef] [PubMed]
- Porsolt, R.D.; Bertin, A.; Jalfre, M. Behavioral despair in mice: A primary screening test for antidepressants. Arch. Int. Pharmacodyn. Ther. 1977, 229, 327–336. [Google Scholar] [PubMed]
- Fransson, L.; Dos, S.C.; Wolbert, P.; Sjöholm, A.; Rafacho, A.; Ortsäter, H. Liraglutide counteracts obesity and glucose intolerance in a mouse model of glucocorticoid-induced metabolic syndrome. Diabetol. Metab. Syndr. 2014, 6, 3. [Google Scholar] [CrossRef] [PubMed]
- Gulliford, M.C.; Charlton, J.; Latinovic, R. Risk of diabetes associated with prescribed glucocorticoids in a large population. Diabetes Care 2006, 29, 2728–2729. [Google Scholar] [CrossRef] [PubMed]
- Karatsoreos, I.N.; Bhagat, S.M.; Bowles, N.P.; Weil, Z.M.; Pfaff, D.W.; McEwen, B.S. Endocrine and physiological changes in response to chronic corticosterone: A potential model of the metabolic syndrome in mouse. Endocrinology 2010, 151, 2117–2127. [Google Scholar] [CrossRef] [PubMed]
- Lee, B.; Sur, B.; Kwon, S.; Yeom, M.; Shim, I.; Lee, H.; Hahm, D.H. Chronic administration of catechin decreases depression and anxiety-like behaviors in a rat model using chronic corticosterone injections. Biomol. Ther. 2013, 21, 313–322. [Google Scholar] [CrossRef] [PubMed]
- Yu, J.; Yu, B.; He, J.; Zheng, P.; Mao, X.B.; Han, G.Q.; Chen, D.W. Chronic glucocorticoid exposure-induced epididymal adiposity is associated with mitochondrial dysfunction in white adipose tissue of male C57BL/6J mice. PLoS ONE 2014, 9, e112628. [Google Scholar] [CrossRef] [PubMed]
- Chimin, P.; Farias, T.S.M.; Torres-Leal, F.L.; Bolsoni-Lopes, A.; Campaña, A.B.; Andreotti, S.; Lima, F.B. Chronic glucocorticoid treatment enhances lipogenic activity in visceral adipocytes of male Wistar rats. Acta Physiol. 2014, 211, 409–420. [Google Scholar] [CrossRef] [PubMed]
- Fransson, L.; Franzén, S.; Rosengren, V.; Wolbert, P.; Sjöholm, Å.; Ortsäter, H. β-Cell adaptation in a mouse model of glucocorticoid-induced metabolic syndrome. J. Endocrinol. 2013, 219, 231–241. [Google Scholar] [CrossRef] [PubMed]
- Rouse, M.; Younès, A.; Egan, J.M. Resveratrol and curcumin enhance pancreatic β-cell function by inhibiting phosphodiesterase activity. J. Endocrinol. 2014, 223, 107–117. [Google Scholar] [CrossRef] [PubMed]
- Nøhr, M.K.; Dudele, A.; Poulsen, M.M.; Ebbesen, L.H.; Radko, Y.; Christensen, L.P.; Jessen, N.; Richelsen, B.; Lund, S.; Pedersen, S.P. LPS-enhanced glucose-stimulated insulin secretion is normalized by resveratrol. PLoS ONE 2016, 1, e0146840. [Google Scholar] [CrossRef] [PubMed]
- Liu, Z.; Jiang, C.; Zhang, J.; Liu, B.; Du, Q. Resveratrol inhibits inflammation and ameliorates insulin resistant endothelial dysfunction via regulation of AMP-activated protein kinase and sirtuin 1 activities. J. Diabetes 2016, 8, 324–335. [Google Scholar] [CrossRef] [PubMed]
- Sadi, G.; Pektaş, M.B.; Koca, H.B.; Tosun, M.; Koca, T. Resveratrol improves hepatic insulin signaling and reduces the inflammatory response in streptozotocin-induced diabetes. Gene 2015, 570, 213–220. [Google Scholar] [CrossRef] [PubMed]
- Zheng, X.; Zhu, S.; Chang, S.; Cao, Y.; Dong, J.; Li, J.; Long, R.; Zhou, Y. Protective effects of chronic resveratrol treatment on vascular inflammatory injury in streptozotocin-induced type 2 diabetic rats: Role of NF-κB signaling. Eur. J. Pharmacol. 2013, 720, 147–157. [Google Scholar] [CrossRef] [PubMed]
- Haohao, Z.; Guijun, Q.; Juan, Z.; Wen, K.; Lulu, C. Resveratrol improves high-fat diet induced insulin resistance by rebalancing subsarcolemmal mitochondrial oxidation and antioxidantion. J. Physiol. Biochem. 2015, 71, 121–131. [Google Scholar] [CrossRef] [PubMed]
- Sample Availability: Samples of the compouds are available from the authors.
© 2016 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Li, Y.-C.; Liu, Y.-M.; Shen, J.-D.; Chen, J.-J.; Pei, Y.-Y.; Fang, X.-Y. Resveratrol Ameliorates the Depressive-Like Behaviors and Metabolic Abnormalities Induced by Chronic Corticosterone Injection. Molecules 2016, 21, 1341. https://doi.org/10.3390/molecules21101341
Li Y-C, Liu Y-M, Shen J-D, Chen J-J, Pei Y-Y, Fang X-Y. Resveratrol Ameliorates the Depressive-Like Behaviors and Metabolic Abnormalities Induced by Chronic Corticosterone Injection. Molecules. 2016; 21(10):1341. https://doi.org/10.3390/molecules21101341
Chicago/Turabian StyleLi, Yu-Cheng, Ya-Min Liu, Ji-Duo Shen, Jun-Jie Chen, Yang-Yi Pei, and Xiao-Yan Fang. 2016. "Resveratrol Ameliorates the Depressive-Like Behaviors and Metabolic Abnormalities Induced by Chronic Corticosterone Injection" Molecules 21, no. 10: 1341. https://doi.org/10.3390/molecules21101341
APA StyleLi, Y.-C., Liu, Y.-M., Shen, J.-D., Chen, J.-J., Pei, Y.-Y., & Fang, X.-Y. (2016). Resveratrol Ameliorates the Depressive-Like Behaviors and Metabolic Abnormalities Induced by Chronic Corticosterone Injection. Molecules, 21(10), 1341. https://doi.org/10.3390/molecules21101341





