New Diphenol and Isocoumarins from the Aerial Part of Lawsonia inermis and Their Inhibitory Activities against NO Production
Abstract
:1. Introduction
2. Results and Discussion
2.1. Isolation and Structural Elucidation
2.2. Structure Identification of the Known Isolates
2.3. Inhibitory Activity against Nitric Oxide Production
3. Experimental Section
3.1. General
3.2. Chemicals
3.3. Plant Material
3.4. Extraction and Isolation
3.5. Cell Culture
3.6. Cell Viability
3.7. Measurement of Nitric Oxide/Nitrite
3.8. Statistical Analysis
4. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Chen, H.Y.; Qian, C. Flora of China; Editorial Committee of the Flora of China: Beijing, China, 2007; pp. 274–288. [Google Scholar]
- Lin, Y.X.; Chang, Y.S.; Chen, I.S.; Ou, J.C. The Catalogue of Medicinal Plant Resources in Taiwan; Committee on Chinese Medicine and Pharmacy: Taipei, Taiwan, 2003.
- Ahmed, S.; Rahman, A.; Alam, A.; Saleem, M.; Athar, M.; Sultana, S. Evaluation of the efficacy of Lawsonia alba in the alleviation of carbon tetrachloride-induced oxidative stress. J. Ethnopharmacol. 2000, 69, 157–164. [Google Scholar] [CrossRef]
- Liou, J.R.; Mohamed, E.S.; Du, Y.C.; Tseng, C.N.; Hwang, T.L.; Chuang, Y.L.; Hsu, Y.M.; Hsieh, P.W.; Wu, C.C.; Chen, S.L.; et al. 1,5-Diphenylpent-3-en-1-ynes and methyl naphthalene carboxylates from Lawsonia inermis and their anti-inflammatory activity. Phytochemistry 2013, 88, 67–73. [Google Scholar] [CrossRef] [PubMed]
- Babu, P.D.; Subhasree, R.S. Antimicrobial activities of Lawsonia inermis—A review. Acad. J. Plant Sci. 2009, 2, 231–232. [Google Scholar]
- Neuman, J.R.C.; Behar, J.V. 2-Carbomethoxybenzocyclobutenone. Synthesis of a photochemically sensitive small-ring system by a pyrolytic Wolff rearrangement. J. Am. Chem. Soc. 1967, 89, 4550–4551. [Google Scholar]
- Sevil, Ö.; Metin, B. The chemistry of homophthalic acid: A new synthetic strategy for construction of substituted isocoumarin and indole skeletons. Tetrahedron 2008, 64, 5531–5540. [Google Scholar]
- Schnekenburger, J. Acylierung von Homophthalsäureanhydrid mit kohlensäureäthylesterchlorid. J. Arch. Pharm. 1965, 298, 405–410. [Google Scholar] [CrossRef]
- Kagawa, H.; Shigematsu, A.; Ohta, S.; Harigaya, Y. Preparative monohydroxyflavanone syntheses and a protocol for gas chromatography-mass spectrometry analysis of monohydroxyflavanones. Chem. Pharm. Bull. 2005, 53, 547–554. [Google Scholar] [CrossRef] [PubMed]
- Parveen, S.; Riaz, N.; Saleem, M.; Khan, J.; Ahmad, S.; Ashraf, M.; Ejaz, S.A.; Tareen, R.B.; Jabbar, A. Bioactive phenolics from Launaea intybacea. J. Chem. Soc. Pak. 2012, 34, 1–7. [Google Scholar]
- Liu, M.; Yang, S.; Jin, L.; Hu, D.; Wu, Z.; Yang, S. Chemical constituents of the ethyl acetate extract of Belamcanda chinensis (L.) DC roots and their antitumor activities. Molecules 2012, 17, 6156–6169. [Google Scholar] [CrossRef] [PubMed]
- Qi, S.H.; Wu, D.G.; Ma, Y.B.; Luo, X.D. A novel flavane form Carapa guianensis. Acta Bot. Sin. 2003, 45, 1129–1133. [Google Scholar]
- Geller, D.A.; Billiar, T.R. Molecular biology of nitric oxide synthases. Cancer Metastasis Rev. 1998, 17, 7–23. [Google Scholar] [CrossRef] [PubMed]
- Moncada, S.; Palmer, R.M.; Higgs, E.A. Nitric oxide: Physiology, pathophysiology, and pharmacology. Pharmcol. Rev. 1991, 43, 109–142. [Google Scholar]
- Sample Availability: Samples of the compounds are available from the authors.

 ) and HMBC (
) and HMBC (  ) correlations of 1.
) correlations of 1.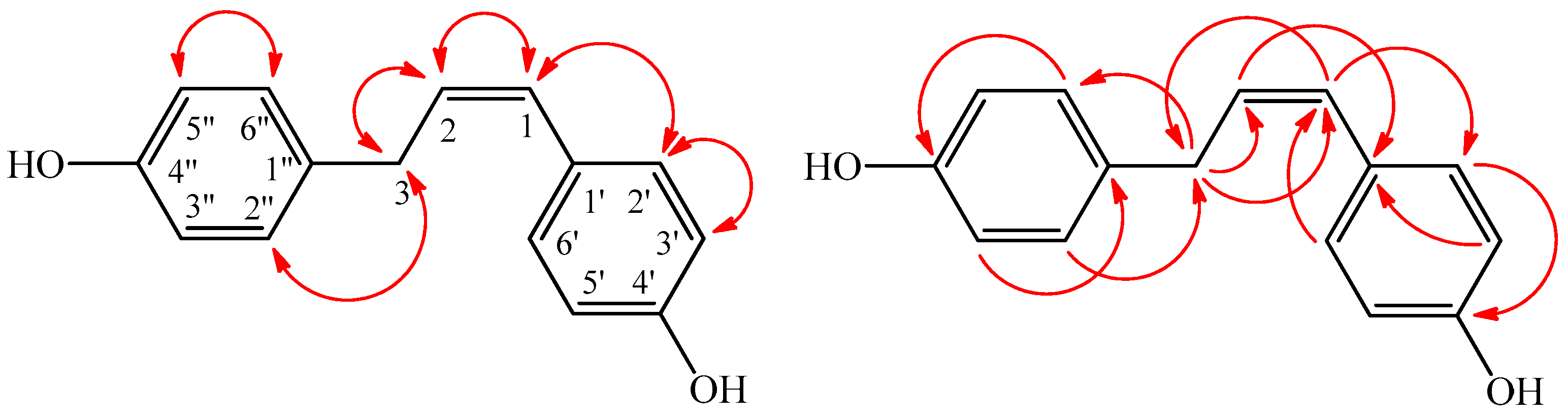
 ) and HMBC (
) and HMBC (  ) correlations of 2.
) correlations of 2.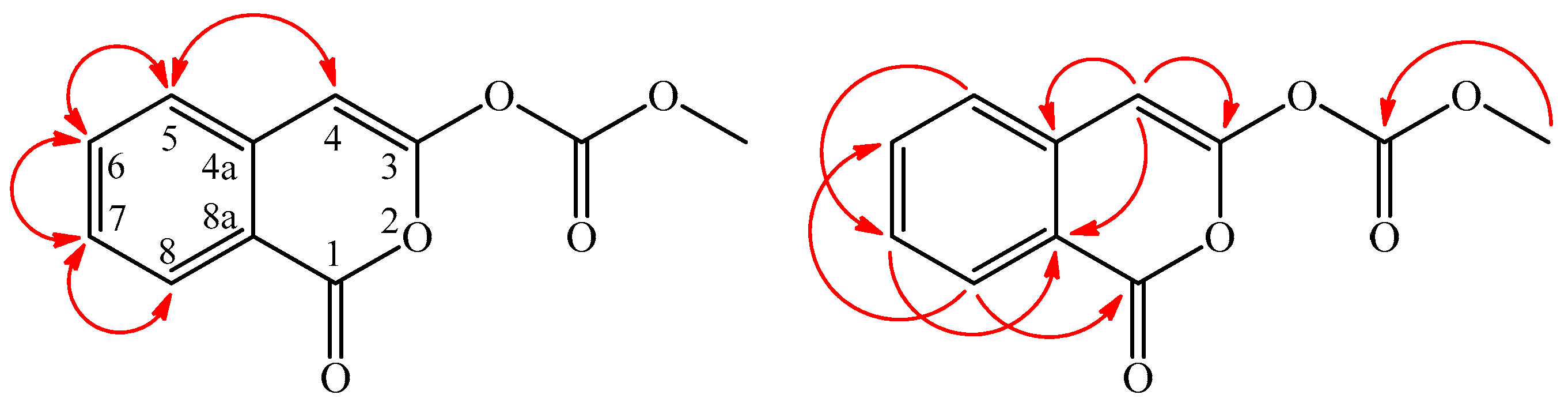
 ) and HMBC (
) and HMBC (  ) correlations of 3.
) correlations of 3.
| Position | δC | δH | NOESY | HMBC a |
|---|---|---|---|---|
| 1 | 110.5 | 5.70 (d, J = 10.5 ) | 2, 2′, 6′ | 2, 3, 2′, 6′ |
| 2 | 142.6 | 6.00 (d, J = 10.5, 7.5) | 1, 3 | 1′, 1″ |
| 3 | 36.8 | 3.57 (d, J = 7.5) | 2, 2″, 6″ | 1, 2, 2″, 6″ |
| 1′ | 129.0 | |||
| 2′, 6′ | 134.1 | 7.25 (d, J = 8.5) | 1, 3′, 5′ | 1, 4′ |
| 3′, 5′ | 116.7 | 6.73 (d, J = 8.5) | 2′, 6′ | 1′ |
| 4′ | 157.7 | |||
| 1″ | 132.2 | |||
| 2″, 6″ | 130.6 | 7.04 (d, J = 8.3) | 3, 3″, 5″ | 3, 4″ |
| 3″, 5″ | 116.4 | 6.68 (d, J = 8.3) | 2″, 6″ | 1″ |
| 4″ | 156.8 |
| Position | δC | δH | NOESY | HMBC a |
|---|---|---|---|---|
| 1 | 165.5 | |||
| 2 | ||||
| 3 | 154.1 | |||
| 4 | 95.6 | 5.88 (s) | 5 | 3, 4a, 8a |
| 4a | 138.9 | |||
| 5 | 126.0 | 7.69 (dd, J = 7.1, 1.5) | 4, 6 | 4, 7, 8a |
| 6 | 135.0 | 7.74 (td, J = 7.1, 1.7) | 5, 7 | 8, 4a |
| 7 | 121.2 | 7.78 (td, J = 7.1, 1.5) | 6, 8 | 5, 8a |
| 8 | 132.3 | 7.99 (dd, J = 7.1, 1.7) | 1, 6, 4a | |
| 8a | 124.7 | |||
| OCOR | 164.1 | |||
| OCOCH3 | 52.0 | 3.85 (s) | OCOCH3 |
| Position | δC | δH | NOESY | HMBC a |
|---|---|---|---|---|
| 1 | 165.5 | |||
| 2 | ||||
| 3 | 154.0 | |||
| 4 | 96.0 | 5.88 (s) | 5 | 3, 4a, 8a |
| 4a | 139.0 | |||
| 5 | 126.0 | 7.69 (dd, J = 7.1, 1.5) | 4, 6 | 4, 7, 8a |
| 6 | 135.0 | 7.77 (td, J = 7.1, 1.7) | 5, 7 | 8, 4a |
| 7 | 121.2 | 7.74 (td, J = 7.1, 1.5) | 6, 8 | 5, 8a |
| 8 | 132.3 | 7.98 (dd, J = 7.1, 1.7) | 1, 6, 4a | |
| 8a | 124.8 | |||
| OCOR | 163.6 | |||
| OCOCH2CH3 | 61.0 | 4.30 (q, J = 7.3) | OCOCH2CH3 | OCOCH2CH3 |
| OCOCH2CH3 | 14.3 | 1.36 (t, J = 7.3) | OCOCH2CH3 | OCOCH2CH3 |
| Compounds | IC50 (μg/mL) a |
|---|---|
| 1 | 5.63 ± 3.64 |
| 2 | >20 |
| 3 | >20 |
| 4 | 15.72 ± 2.52 |
| 5 | 8.67 ± 3.84 |
| 6 | 6.67 ± 3.48 |
| 7 | 6.17 ± 2.86 |
| 8 | 7.61 ± 3.34 |
| 9 | 14.52 ± 3.31 |
| Indomethacin b | 78.56 ± 1.38 |
© 2016 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yang, C.-S.; Huang, H.-C.; Wang, S.-Y.; Sung, P.-J.; Huang, G.-J.; Chen, J.-J.; Kuo, Y.-H. New Diphenol and Isocoumarins from the Aerial Part of Lawsonia inermis and Their Inhibitory Activities against NO Production. Molecules 2016, 21, 1299. https://doi.org/10.3390/molecules21101299
Yang C-S, Huang H-C, Wang S-Y, Sung P-J, Huang G-J, Chen J-J, Kuo Y-H. New Diphenol and Isocoumarins from the Aerial Part of Lawsonia inermis and Their Inhibitory Activities against NO Production. Molecules. 2016; 21(10):1299. https://doi.org/10.3390/molecules21101299
Chicago/Turabian StyleYang, Chang-Syun, Hui-Chi Huang, Sheng-Yang Wang, Ping-Jyun Sung, Guan-Jhong Huang, Jih-Jung Chen, and Yueh-Hsiung Kuo. 2016. "New Diphenol and Isocoumarins from the Aerial Part of Lawsonia inermis and Their Inhibitory Activities against NO Production" Molecules 21, no. 10: 1299. https://doi.org/10.3390/molecules21101299
APA StyleYang, C.-S., Huang, H.-C., Wang, S.-Y., Sung, P.-J., Huang, G.-J., Chen, J.-J., & Kuo, Y.-H. (2016). New Diphenol and Isocoumarins from the Aerial Part of Lawsonia inermis and Their Inhibitory Activities against NO Production. Molecules, 21(10), 1299. https://doi.org/10.3390/molecules21101299









