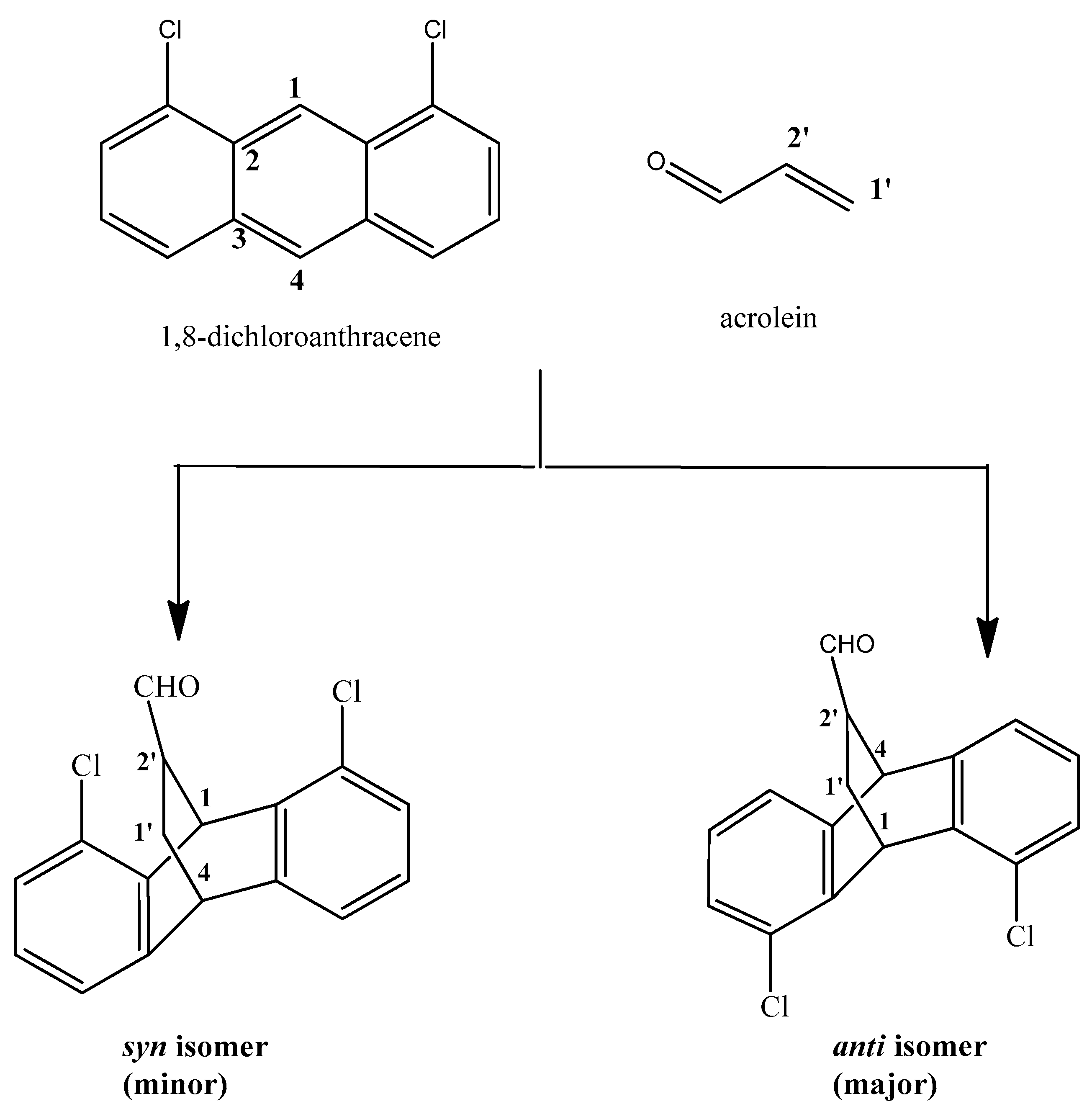
Theoretical Study on Regioselectivity of the Diels-Alder Reaction between 1,8-Dichloroanthracene and Acrolein
Abstract
:1. Introduction
2. Computational Details
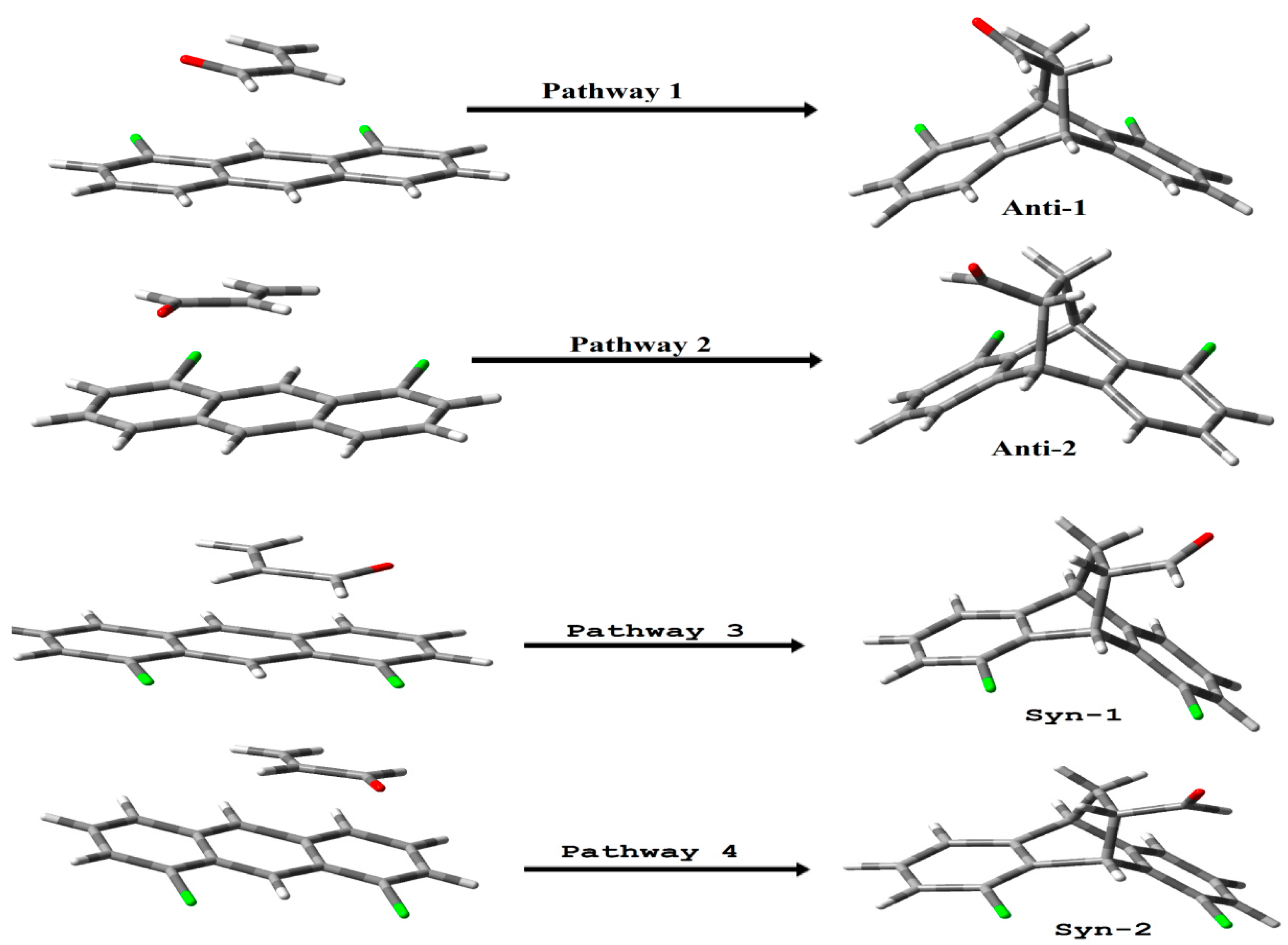
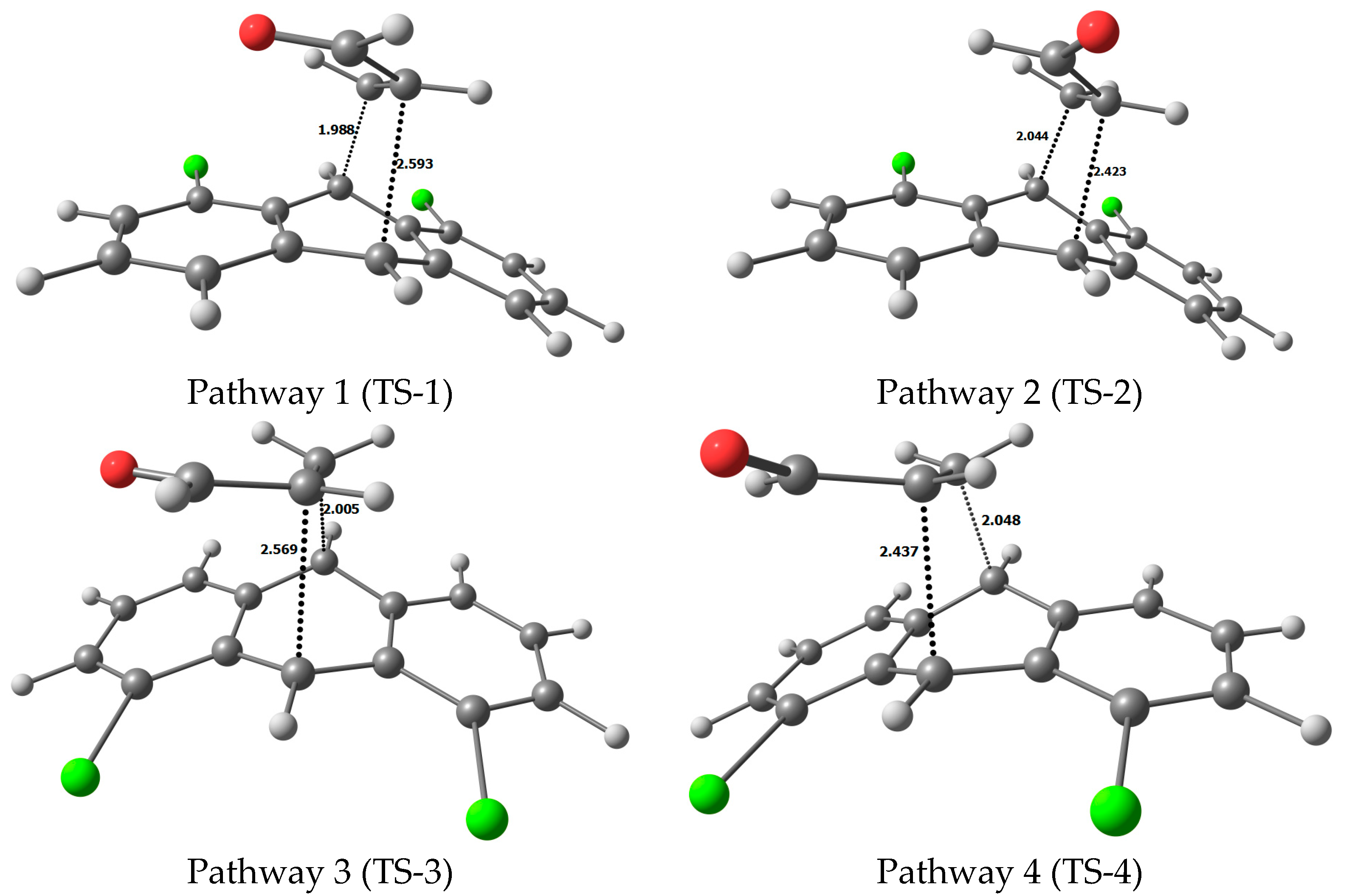
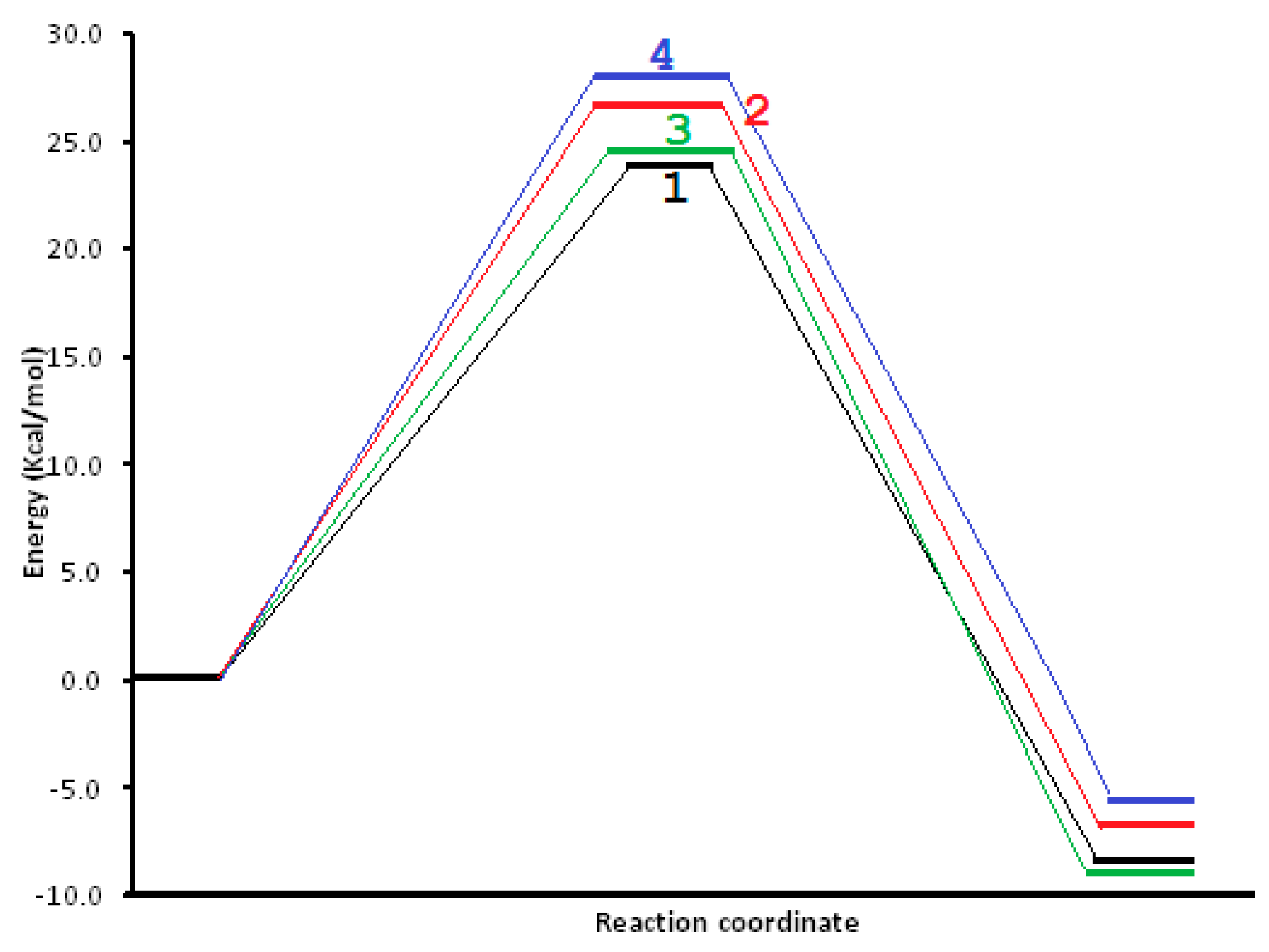
3. Results and Discussion
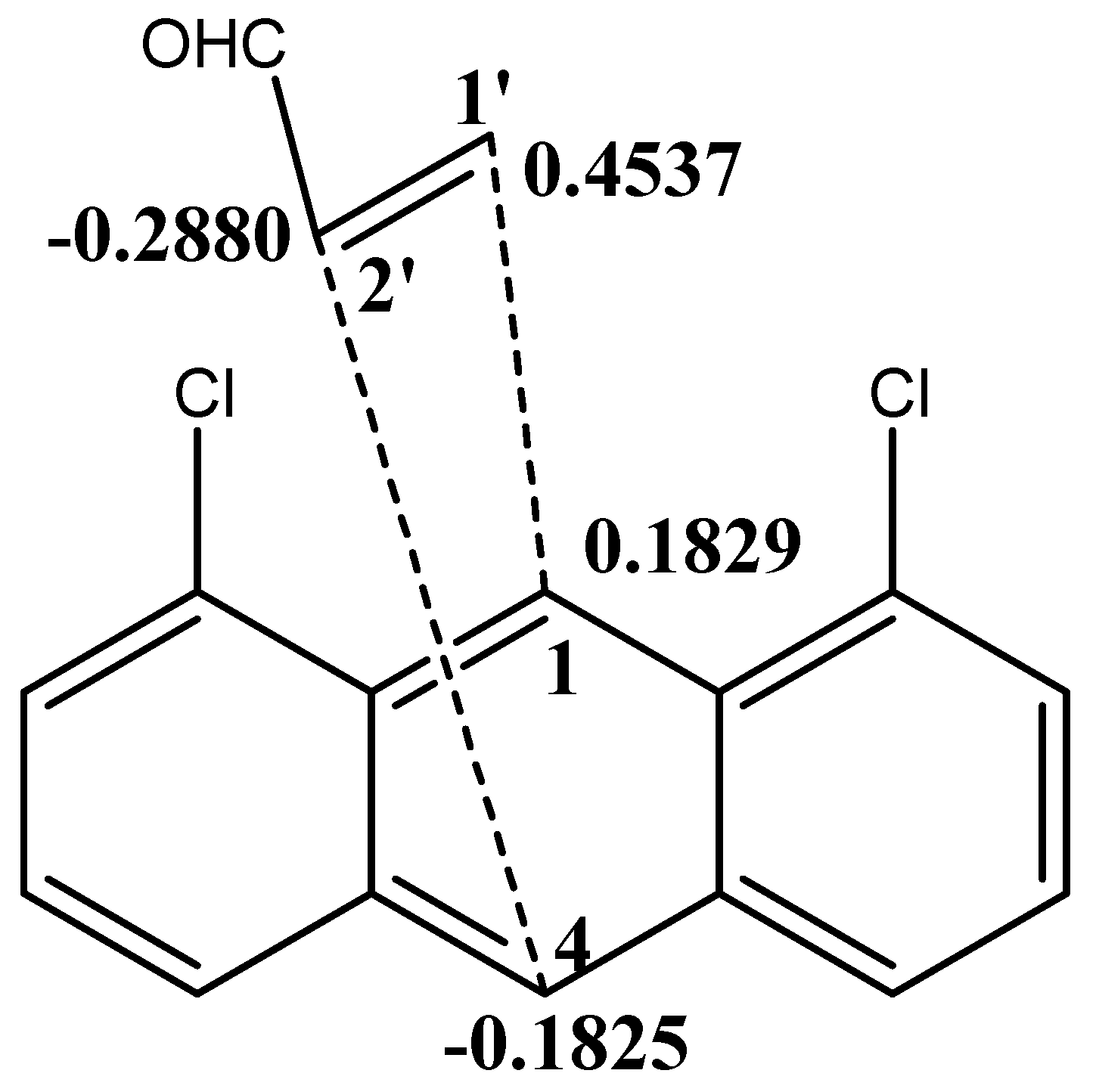
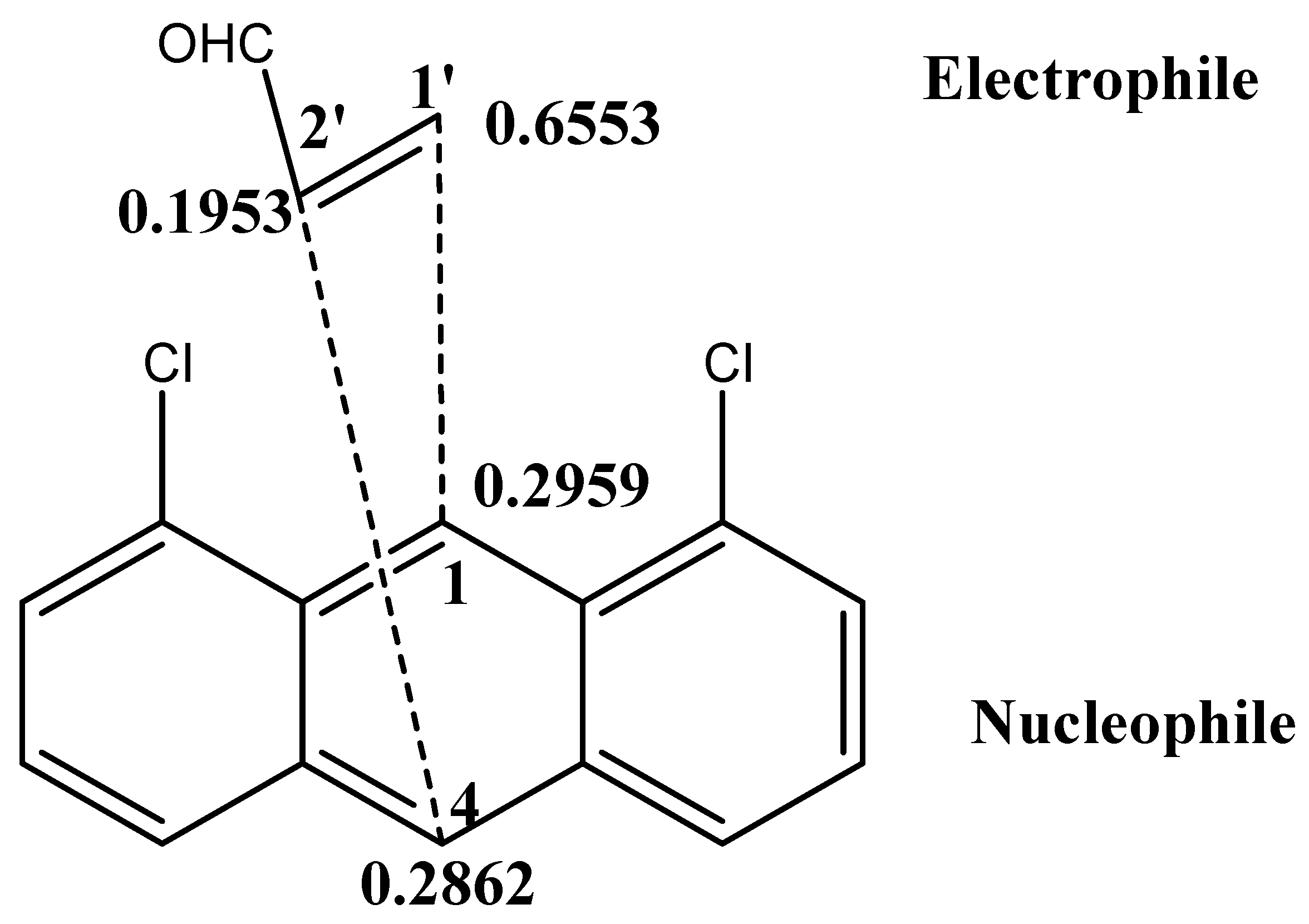
4. Molecular Mechanism
5. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Brocksom, T.J.; Nakamura, J.; Ferreira, M.L.; Brocksom, U. The Diels-Alder reaction: An update. J. Braz. Chem. Soc. 2001, 12, 597–622. [Google Scholar] [CrossRef]
- Mehta, G.; Uma, R. Stereoelectronic control in Diels-Alder reaction of dissymmetric 1,3-dienes. Acc. Chem. Res. 2000, 33, 278–286. [Google Scholar] [CrossRef] [PubMed]
- Kanishchev, O.S.; Sanselme, M.; Bouillon, J.-P. Hetero-Diels-Alder reactions of perfluoroalkyl thioamides with electron-rich 1,3-dienes: Synthesis of new 2-aminosubstituted-3, 6-dihydro-2h-thiopyrans and related compounds. Tetrahedron 2013, 69, 1322–1336. [Google Scholar] [CrossRef]
- Khan, R.; Singh, T.P.; Singh, M.D. Letter highly regioselective Diels-Alder reaction of 9-substituted anthracenes with citraconic anhydride. Synlett 2014, 25, 696–700. [Google Scholar]
- Huang, D.; Tan, Y.; Sun, Y.; Zheng, C.; Wang, Z. Quantum chemical calculation study on terphenyl arylamines hole transport materials. J. Soc. Inf. Display 2015, 23, 182–185. [Google Scholar] [CrossRef]
- Meek, J.S.; Dewey, F.M.; Hanna, M.W. Diels-Alder reactions of naphthacene. J. Org. Chem. 1967, 32, 69–72. [Google Scholar] [CrossRef]
- Dewar, M.J.; Pyron, R.S. Nature of the transition state in some diels-alder reactions. J. Am. Chem. Soc. 1970, 92, 3098–3103. [Google Scholar] [CrossRef]
- Karama, U.; El-Azhary, A.A.; Almansour, A.I.; Al-Kahtani, A.A.; Al-Turki, T.M.; Jaafar, M.H. Computational and spectral investigation of 5,12-dihydro-5,12-ethanonaphthacene-13-carbaldehyde. Molecules 2011, 16, 6741–6746. [Google Scholar] [CrossRef] [PubMed]
- Karama, U.S.; Sultan, M.A.S.; Tahir, K.E.H.E.; Almansour, A.I. Antidepressant Compounds. Patent US9125866 B1, 8 September 2015. [Google Scholar]
- Karama, U.; Sultan, M.A.; Almansour, A.I.; El-Taher, K.E. Synthesis of chlorinated tetracyclic compounds and testing for their potential antidepressant effect in mice. Molecules 2016, 21, 61. [Google Scholar] [CrossRef] [PubMed]
- Becke, A.D. Density-functional exchange-energy approximation with correct asymptotic behavior. Phys. Rev. A 1988, 38, 3098. [Google Scholar] [CrossRef]
- Lee, C.; Yang, W.; Parr, R.G. Development of the colle-salvetti correlation-energy formula into a functional of the electron density. Phys. Rev. B 1988, 37, 785. [Google Scholar] [CrossRef]
- Frisch, M.; Trucks, G.; Schlegel, H.; Scuseria, G.; Robb, M.; Cheeseman, J.; Montgomery, J., Jr.; Vreven, T.; Kudin, K.; Burant, J.; et al. Gaussian 03, Revision c. 02; Gaussian Inc.: Wallingford, CT, USA, 2004.
- Nacereddine, A.K.; Yahia, W.; Bouacha, S.; Djerourou, A. A theoretical investigation of the regio- and stereo-selectivities of the 1,3-dipolar cycloaddition of C-diethoxyphosphoryl-N-methylnitrone with substituted alkenes. Tetrahedron Lett. 2010, 51, 2617–2621. [Google Scholar] [CrossRef]
- Dennington, R.; Keith, T.; Millam, J. GaussView, Version 5; Semichem Inc.: Shawnee Mission, KS, USA, 2009.
- Parr, R.G.; Szentpaly, L.; Liu, S. Electrophilicity index. J. Am. Chem. Soc. 1999, 121, 1922–1924. [Google Scholar] [CrossRef]
- Parr, R.G.; Pearson, R.G. Absolute hardness: Companion parameter to absolute electronegativity. J. Am. Chem. Soc. 1983, 105, 7512–7516. [Google Scholar] [CrossRef]
- Parr, R.G.; Yang, W. Density-Functional Theory of Atoms and Molecules; Oxford University Press: New York, NY, USA, 1989; Volume 16. [Google Scholar]
- Kohn, W.; Sham, L.J. Self-consistent equations including exchange and correlation effects. Phys. Rev. 1965, 140, A1133. [Google Scholar] [CrossRef]
- Domingo, L.R.; Chamorro, E.; Pérez, P. Understanding the reactivity of captodative ethylenes in polar cycloaddition reactions. A theoretical study. J. Org. Chem. 2008, 73, 4615–4624. [Google Scholar] [CrossRef] [PubMed]
- Domingo, L.R.; Pérez, P. The nucleophilicity n index in organic chemistry. Org. Biomol. Chem. 2011, 9, 7168–7175. [Google Scholar] [CrossRef] [PubMed]
- Reed, A.E.; Weinhold, F. Natural bond orbital analysis of near-hartree-fock water dimer. J. Chem. Phys. 1983, 78, 4066–4073. [Google Scholar] [CrossRef]
- Domingo, L.R.; Aurell, M.J.; Pérez, P.; Contreras, R. Quantitative characterization of the local electrophilicity of organic molecules. Understanding the regioselectivity on diels-alder reactions. J. Phys. Chem. A 2002, 106, 6871–6875. [Google Scholar] [CrossRef]
- Domingo, L.R.; Aurell, M.J.; Pérez, P.; Contreras, R. Quantitative characterization of the global electrophilicity power of common diene/dienophile pairs in Diels-Alder reactions. Tetrahedron 2002, 58, 4417–4423. [Google Scholar] [CrossRef]
- Domingo, L.R.; Pérez, P. Global and local reactivity indices for electrophilic/nucleophilic free radicals. Org. Biomol. Chem. 2013, 11, 4350–4358. [Google Scholar] [CrossRef] [PubMed]
- Jaramillo, P.; Domingo, L.R.; Chamorro, E.; Pérez, P. A further exploration of a nucleophilicity index based on the gas-phase ionization potentials. J. Mol. Struct. THEOCHEM 2008, 865, 68–72. [Google Scholar] [CrossRef]
- Pérez, P.; Domingo, L.R.; Aurell, M.J.; Contreras, R. Quantitative characterization of the global electrophilicity pattern of some reagents involved in 1,3-dipolar cycloaddition reactions. Tetrahedron 2003, 59, 3117–3125. [Google Scholar] [CrossRef]
- Houk, K.N. Frontier molecular orbital theory of cycloaddition reactions. Acc. Chem. Res. 1975, 8, 361–369. [Google Scholar] [CrossRef]
- Sample Availability: Not available.
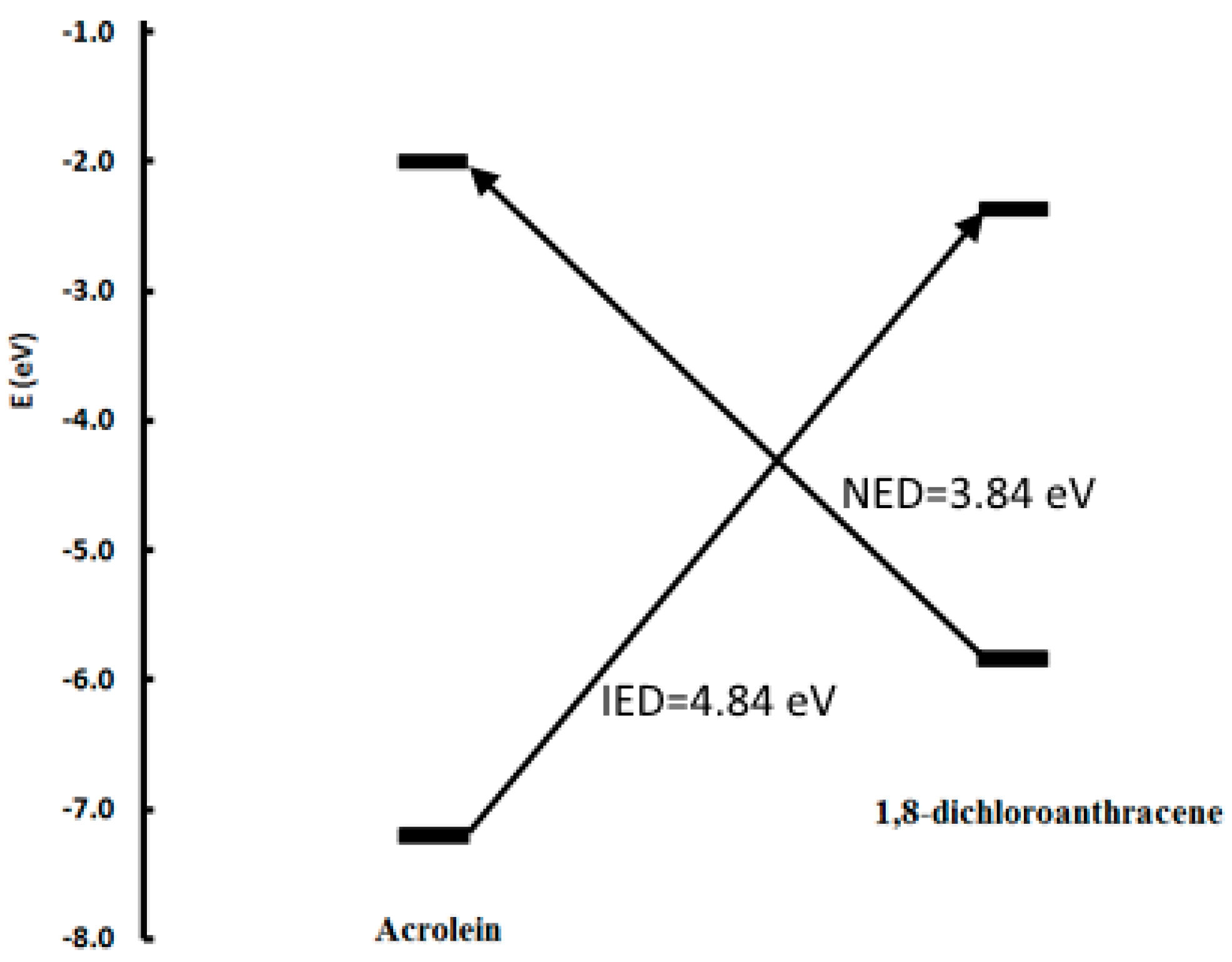
| Reactant | EHOMO | ELUMO | μ | ω | N |
|---|---|---|---|---|---|
| 1,8-dichloroanthracene | −5.8456 | −2.3737 | −4.1096 | 2.4322 | 3.5231 |
| acrolein | −7.2092 | −2.0014 | −4.6053 | 2.0363 | 2.1595 |
| Transition State (TS) | Charge Transfer (e) |
|---|---|
| TS-1 | 0.0337 |
| TS-2 | 0.0333 |
| TS-3 | 0.0448 |
| TS-4 | 0.0407 |
© 2016 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sultan, M.A.; Karama, U.; Almansour, A.I.; Soliman, S.M. Theoretical Study on Regioselectivity of the Diels-Alder Reaction between 1,8-Dichloroanthracene and Acrolein. Molecules 2016, 21, 1277. https://doi.org/10.3390/molecules21101277
Sultan MA, Karama U, Almansour AI, Soliman SM. Theoretical Study on Regioselectivity of the Diels-Alder Reaction between 1,8-Dichloroanthracene and Acrolein. Molecules. 2016; 21(10):1277. https://doi.org/10.3390/molecules21101277
Chicago/Turabian StyleSultan, Mujeeb A., Usama Karama, Abdulrahman I. Almansour, and Saied M. Soliman. 2016. "Theoretical Study on Regioselectivity of the Diels-Alder Reaction between 1,8-Dichloroanthracene and Acrolein" Molecules 21, no. 10: 1277. https://doi.org/10.3390/molecules21101277
APA StyleSultan, M. A., Karama, U., Almansour, A. I., & Soliman, S. M. (2016). Theoretical Study on Regioselectivity of the Diels-Alder Reaction between 1,8-Dichloroanthracene and Acrolein. Molecules, 21(10), 1277. https://doi.org/10.3390/molecules21101277







