Garlic Supplementation Ameliorates UV-Induced Photoaging in Hairless Mice by Regulating Antioxidative Activity and MMPs Expression
Abstract
:1. Introduction
2. Results
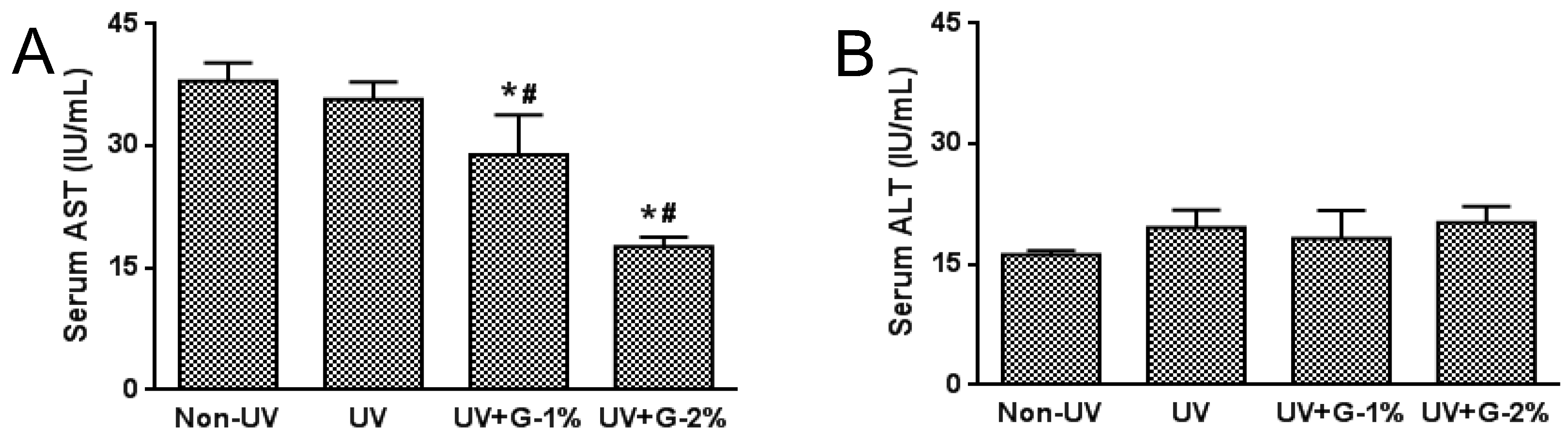
2.1. Body Weight and Serum Biochemical Markers
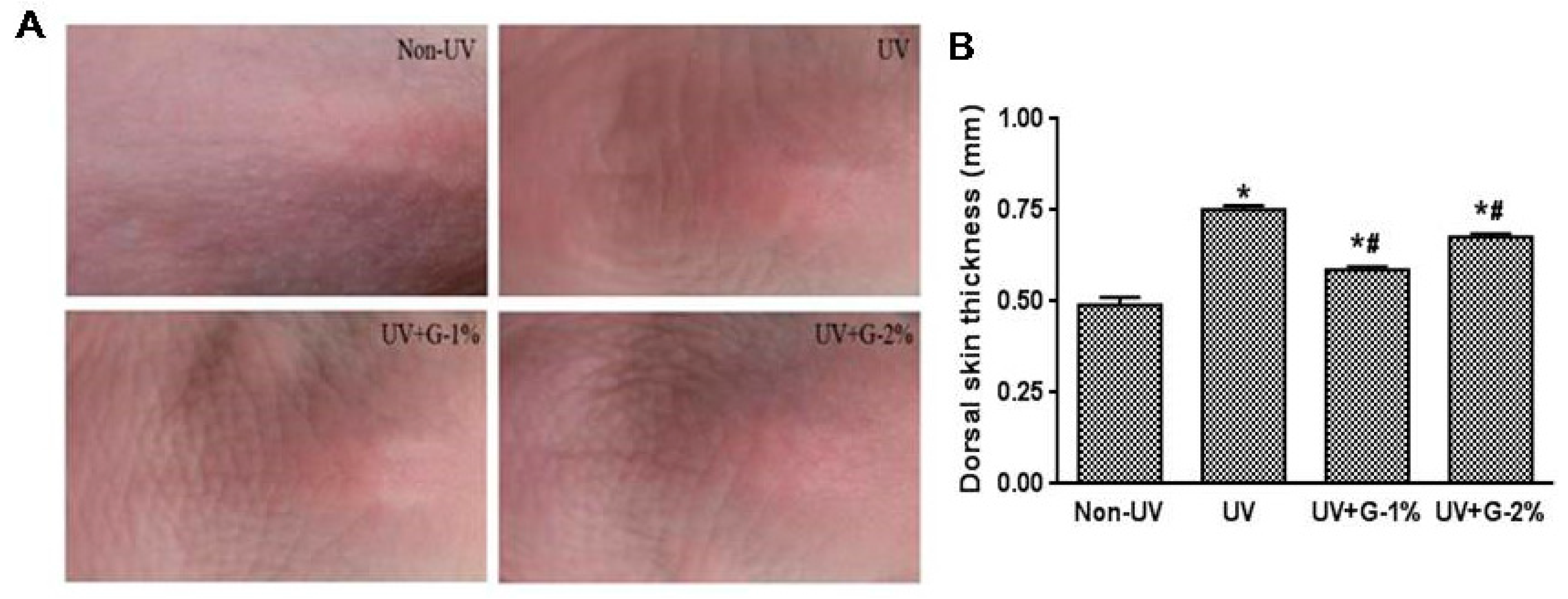
2.2. Wrinkle Formation and Skin Thickening
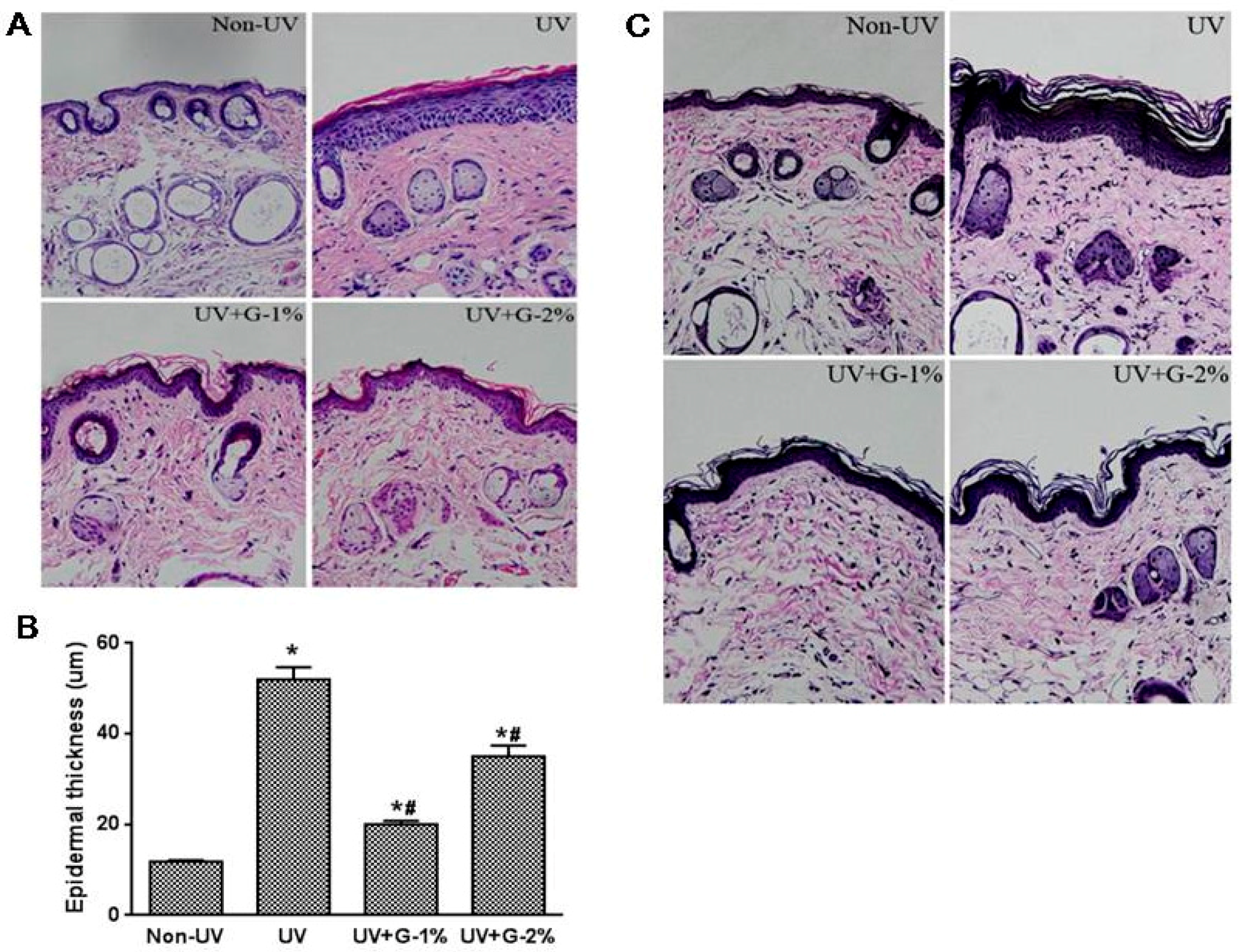
2.3. Histopathological Changes
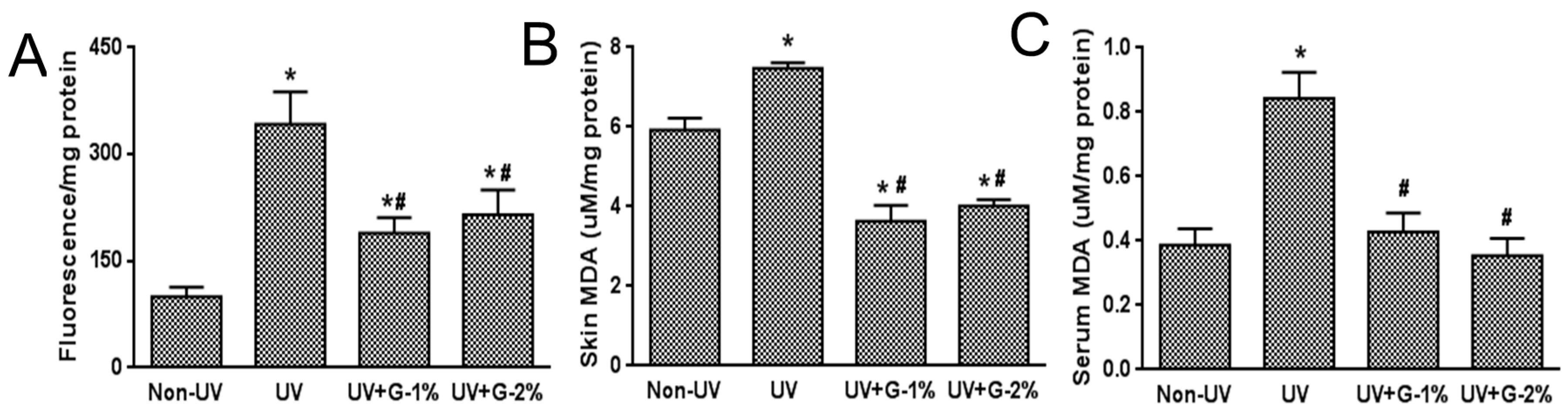
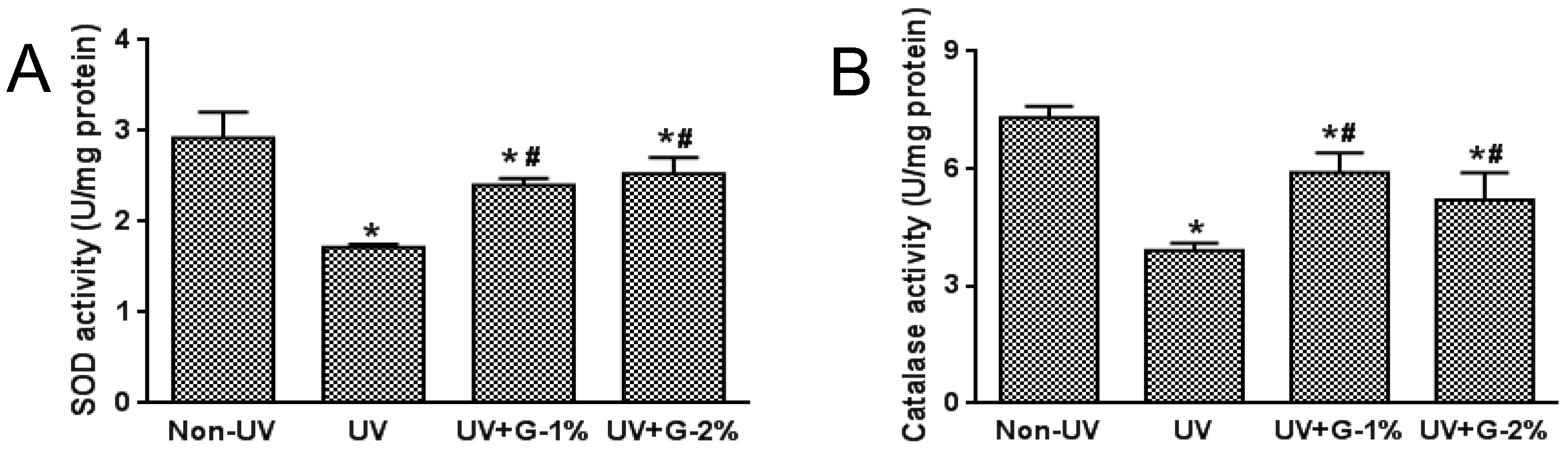
2.4. Antioxidant Status
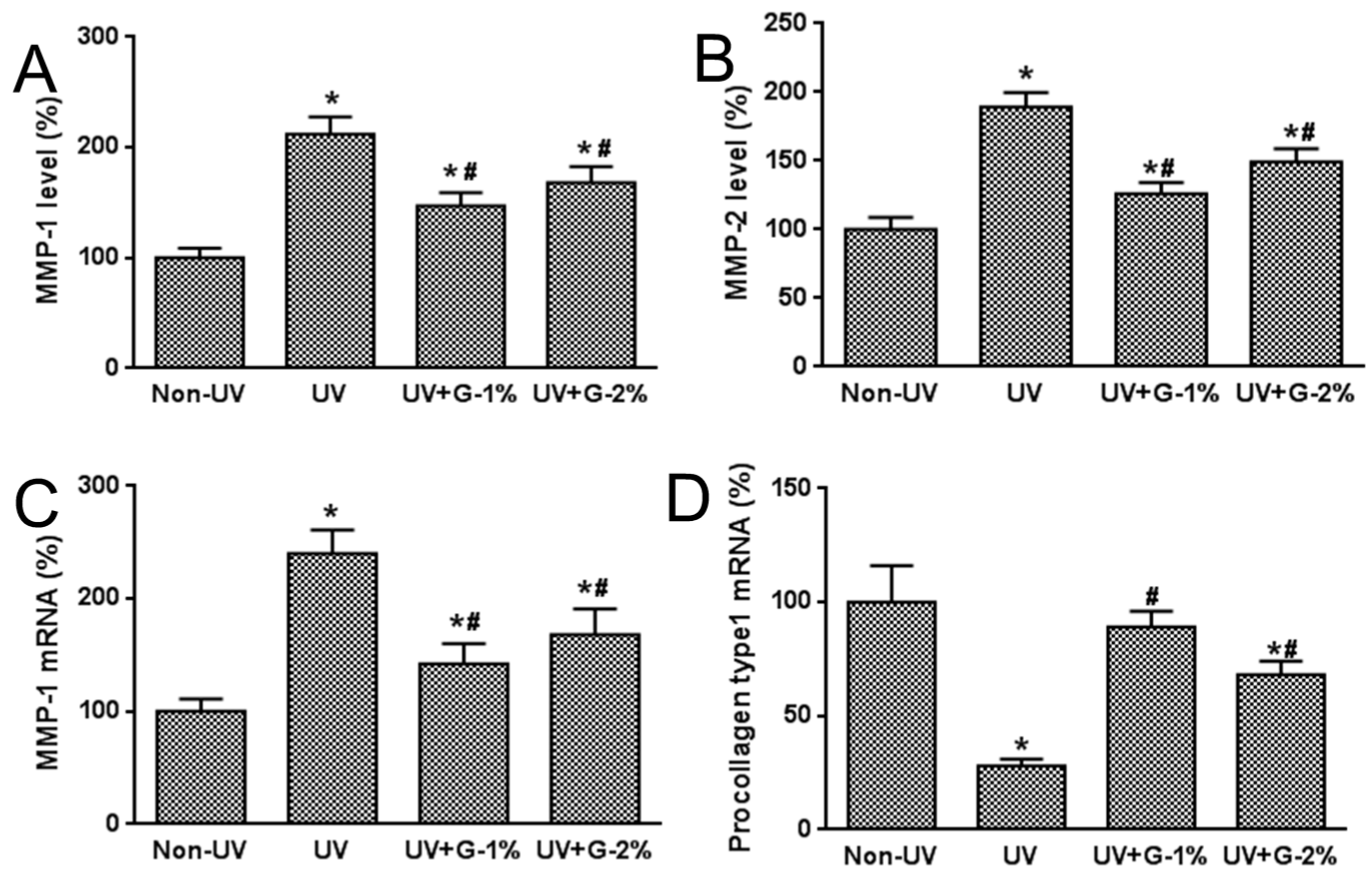
2.5. MMP Protein Expression
3. Discussion
4. Materials and Methods
4.1. Preparation of Garlic Powder
4.2. Experimental Animals and UV Irradiation
4.3. Evaluation of Skin Thickness
4.4. Serum Biochemical Analysis
4.5. Morphological and Histopathological Analysis
4.6. ROS Production
4.7. Lipid Peroxidation and Antioxidant Enzyme Activity
4.8. MMP-1 and MMP-2 Determination
4.9. Quantitative Real Time RT-PCR
4.10. Statistical Analysis
5. Conclusions
Acknowledgments
Conflicts of Interest
References
- Scharffetter-Kochanek, K.; Brenneisen, P.; Wenk, J.; Herrmann, G.; Ma, W.; Kuhr, L.; Meewes, C.; Wlaschek, M. Photoaging of the skin from phenotype to mechanisms. Exp. Gerontol. 2000, 35, 307–316. [Google Scholar] [CrossRef]
- Leyden, J.J. Clinical features of ageing skin. Br. J. Dermatol. 1990, 122. [Google Scholar] [CrossRef]
- Podhaisky, H.P.; Riemschneider, S.; Wohlrab, W. UV light and oxidative damage of the skin. Pharmazie 2002, 57, 30–33. [Google Scholar] [PubMed]
- Sander, C.S.; Chang, H.; Salzmann, S.; Müller, C.S.; Ekanayake-Mudiyanselage, S.; Elsner, P.; Thiele, J.J. Photoaging is associated with protein oxidation in human skin in vivo. J. Investig. Dermatol. 2002, 118, 618–625. [Google Scholar] [CrossRef] [PubMed]
- Jang, J.; Ye, B.R.; Heo, S.J.; Oh, C.; Kang, D.H.; Kim, J.H.; Affan, A.; Yoon, K.T.; Choi, Y.U.; Park, S.C.; et al. Photo-oxidative stress by ultraviolet-B radiation and antioxidative defense of eckstolonol in human keratinocytes. Environ.Toxicol. Pharmacol. 2012, 34, 926–934. [Google Scholar] [CrossRef] [PubMed]
- Wölfle, U.; Seelinger, G.; Bauer, G.; Meinke, M.C.; Lademann, J.; Schempp, C.M. Reactive molecule species and antioxidative mechanisms in normal skin and skin aging. Skin Pharmacol. Physiol. 2014, 27, 316–332. [Google Scholar] [CrossRef] [PubMed]
- Dissemond, J.; Schneider, L.A.; Brenneisen, P.; Briviba, K.; Wenk, J.; Wlaschek, M.; Scharffetter-Kochanek, K. Protective and determining factors for the overall lipid peroxidation in ultraviolet A1-irradiated fibroblasts: In vitro and in vivo investigations. Br. J. Dermatol. 2003, 149, 341–349. [Google Scholar] [CrossRef] [PubMed]
- Brenneisen, P.; Sies, H.; Scharffetter-Kochanek, K. Ultraviolet-B irradiation and matrix metalloproteinases: From induction via signaling to initial events. Ann. N. Y. Acad. Sci. 2002, 973, 31–43. [Google Scholar] [CrossRef] [PubMed]
- Inomata, S.; Matsunaga, Y.; Amano, S.; Takada, K.; Kobayashi, K.; Tsunenaga, M.; Nishiyama, T.; Kohno, Y.; Fukuda, M. Possible involvement of gelatinases in basement membrane damage and wrinkle formation in chronically ultraviolet B-exposed hairless mouse. J. Investig. Dermatol. 2003, 120, 128–134. [Google Scholar] [CrossRef] [PubMed]
- Cellini, L.; di Campli, E.; Masulli, M.; di Bartolomeo, S.; Allocati, N. Inhibitions of Helicobacter pylori by garlic extract (Allium sativum). FEMS Immunol. Med. Microbiol. 1996, 13, 273–277. [Google Scholar] [CrossRef] [PubMed]
- Durak, I.; Kavutcu, M.; Aytac, B.; Avc, A.; Devrim, E.; Ozbek, H.; Oztürk, H.S. Effects of garlic extract consumption on blood lipid and oxidant/antioxidant parameters in humans with high blood cholesterol. J. Nutr. Biochem. 2004, 15, 373–377. [Google Scholar] [CrossRef] [PubMed]
- Kaschula, C.H.; Hunter, R.; Hassan, H.T.; Stellenboom, N.; Cotton, J.; Zhai, X.Q.; Parker, M.I. Anti-proliferation activity of synthetic ajoene analogues on cancer cell-lines. Anticancer Agents Med. Chem. 2011, 11, 260–266. [Google Scholar] [CrossRef] [PubMed]
- Lau, B.H. Suppression of LDL oxidation by garlic compounds is a possible mechanism of cardiovascular health benefit. J. Nutr. 2006, 136, 765S–768S. [Google Scholar] [PubMed]
- Xiao, H.; Parkin, K.L. Antioxidant functions of selected allium thiosulfinates and S-alk(en)yl-l-cysteine sulfoxides. J. Agric. Food Chem. 2002, 50, 2488–2493. [Google Scholar] [CrossRef] [PubMed]
- Yeh, Y.Y.; Yeh, S.M. Garlic reduces plasma lipids by inhibiting hepatic cholesterol and triacylglycerol synthesis. Lipids 1994, 29, 189–193. [Google Scholar] [CrossRef] [PubMed]
- Matsumura, Y.; Ananthaswamy, H.N. Toxic effects of ultraviolet radiation on the skin. Toxicol. Appl. Pharmacol. 2004, 195, 298–308. [Google Scholar] [CrossRef] [PubMed]
- Pillai, S.; Oresajo, C.; Hayward, J. Ultraviolet radiation and skin aging: Roles ofreactive oxygen species, inflammation and protease activation, and strategiesfor prevention of inflammation-induced matrix degradation—A review. Int. J. Cosmet. Sci. 2005, 27, 17–34. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.H.; Cho, S.; Lee, S.; Kim, K.H.; Cho, K.H.; Eun, H.C.; Chung, J.H. Photoprotective and anti-skin-aging effects of eicosapentaenoic acid in human skin in vivo. J. Lipid Res. 2006, 47, 921–930. [Google Scholar] [CrossRef] [PubMed]
- Helen, A.; Rajasree, C.R.; Krishnakumar, K.; Augusti, K.T.; Vijayammal, P.L. Antioxidant role of oilsisolated from garlic (Allium sativum Linn) and onion (Allium cepa Linn) on nicotine-induced lipid peroxidation. Vet. Hum. Toxicol. 1999, 41, 316–319. [Google Scholar] [PubMed]
- Pedraza-Chaverri, J.; Maldonado, P.D.; Medina-Campos, O.N.; Olivares-Corichi, I.M.; Granados-Silvestre, M.A.; Hernandez-Pando, R.; Ibarra-Rubio, M.E. Garlic ameliorates gentamicin nephrotixicity: Relation to antioxidant enzymes. Free Radic. Biol. Med. 2000, 29, 602–611. [Google Scholar] [CrossRef]
- Hadshiew, I.M.; Eller, M.S.; Gilchrest, B.A. Skin aging and photoaging: The role of DNA damage and repair. Am. J. Contact Dermatol. 2000, 11, 19–25. [Google Scholar] [CrossRef]
- LeBel, C.P.; Ischiropoulos, H.; Bondy, S.C. Evaluation of the probe 2′,7′-dichlorofluorescin as an indicator of reactive oxygen species formation and oxidative stress. Chem. Res.Toxicol. 1992, 5, 227–231. [Google Scholar] [CrossRef] [PubMed]
- Wlaschek, M.; Tantcheva-Poor, I.; Naderi, L.; Ma, W.; Schneider, L.A.; Razi-Wolf, Z.; Schuller, J.; Scharffetter-Kochanek, K. Solar UV irradiation and dermal photoaging. J. Photochem. Photobiol. B 2001, 63, 41–51. [Google Scholar] [CrossRef]
- Bissett, D.L.; Chatterjee, R.; Hannon, D.P. Photoprotective effect of superoxide-scavenging antioxidants against ultraviolet radiation-induced chronic skin damage in the hairless mouse. Photodermatol. Photoimmunol. Photomed. 1990, 7, 56–62. [Google Scholar] [PubMed]
- Rittié, L.; Fisher, G.J. UV-light-induced signal cascades and skin aging. Ageing Res. Rev. 2002, 1, 705–720. [Google Scholar] [CrossRef]
- Yaar, M.; Gilchrest, B.A. Skin aging: Postulated mechanisms andconsequent changes in structure and function. Clin. Geriatr. Med. 2001, 17, 617–630. [Google Scholar] [CrossRef]
- Okada, K.; Takahashi, Y.; Ohnishi, K.; Ishikawa, O.; Miyachi, Y. Timedependenteffect of chronic UV irradiation on superoxide dismutaseand catalase activity in hairless mice skin. J. Dermatol. Sci. 1994, 8, 183–186. [Google Scholar] [CrossRef]
- Tsuji, N.; Moriwaki, S.; Suzuki, Y.; Takema, Y.; Imokawa, G. The role of elastases secreted by fibroblasts in wrinkle formation: Implication through selective inhibition of elastase activity. Photochem. Photobiol. 2001, 74, 283–290. [Google Scholar] [CrossRef]
- Ohnishi, Y.; Tajima, S.; Akiyama, M.; Ishibashi, A.; Kobayashi, R.; Horii, I. Expression ofelastin-related proteins and matrix metalloproteinases in actinic elastosis of sun damaged skin. Arch. Dermatol. Res. 2000, 292, 27–31. [Google Scholar] [CrossRef] [PubMed]
- El-Domyati, M.; Attia, S.; Saleh, F.; Brown, D.; Birk, D.E.; Gasparro, F.; Ahmad, H.; Uitto, J. Intrinsic agingvs. photoaging: A comparative histopathological, immunohistochemical, and ultrastructural study of skin. Exp. Dermatol. 2002, 11, 398–405. [Google Scholar] [CrossRef] [PubMed]
- Philips, N.; Conte, J.; Chen, Y.J.; Natrajan, P.; Taw, M.; Keller, T.; Givant, J.; Tuason, M.; Dulaj, L.; Leonardi, D.; et al. Beneficial regulation of matrix metalloproteinases and their inhibitors, fibrillar collagens and transforming growth factor-beta by Polypodiumleucotomos, directly or in dermal fibroblasts, ultraviolet radiated fibroblasts, and melanoma cells. Arch. Dermatol. Res. 2009, 301, 487–495. [Google Scholar] [CrossRef] [PubMed]
- Rahman, M.S. Allicin and other functional active components in garlic: Health benefits and bioavailability. Int. J. Food Prop. 2007, 10, 245–268. [Google Scholar] [CrossRef]
- Chung, L.Y. The antioxidant properties of garlic compounds: Allyl cysteine, alliin, allicin, and allyl disulfide. J. Med. Food 2006, 9, 205–213. [Google Scholar] [CrossRef] [PubMed]
- Prasad, K.; Laxdal, V.A.; Yu, M.; Raney, B.L. Antioxidant activity of allicin, an active principle in garlic. Mol. Cell. Biochem. 1995, 148, 183–189. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.R.; Jung, Y.R.; An, H.J.; Kim, D.H.; Jang, E.J.; Choi, Y.J.; Moon, K.M.; Park, C.H.; Chuang, K.W.; Bae, H.R.; et al. Anti-Wrinkle and Anti-Inflammatory Effects of Active Garlic Components and the Inhibition of MMPs via NF-κB Signaling. PLoS ONE 2013, 8, e73877. [Google Scholar] [CrossRef] [PubMed]
- Rahman, K. Garlic and aging: New insights into an old remedy. Ageing Res. Rev. 2003, 2, 39–56. [Google Scholar] [CrossRef]
- Chen, J.P.; Cheong, K.l.; Song, Z.; Shi, Y.; Huang, X. Structure and protective effect on UVB-induced keratinocyte damage of fructan from white garlic. Carbohydr. Polym. 2013, 92, 200–205. [Google Scholar] [CrossRef] [PubMed]
- Nagae, S.; Ushijima, M.; Hatono, S.; Imai, J.; Kasuga, S.; Matsuura, H.; Itakura, Y.; Higashi, Y. Pharmacokinetics of the garlic compound S-allyl cysteine. Planta Med. 1994, 60, 214–217. [Google Scholar] [CrossRef] [PubMed]
- Lawson, L.D.; Hughes, B.G. Characterization of the formation of allicin and other thiosulfinates from garlic. Planta Med. 1992, 58, 345–350. [Google Scholar] [CrossRef] [PubMed]
- Ide, N.; Nelson, A.B.; Lau, B.H.S. Aged Garlic Extract and its constituents inhibit Cu12-induced oxidative modification of low density lipoprotein. Planta Med. 1997, 63, 263–264. [Google Scholar] [CrossRef] [PubMed]
- Imai, J.; Ide, N.; Nagae, S.; Moriguchi, T.; Matsuura, H.; Itakura, Y. Antioxidant and radical scavenging effects of aged garlic extract and its constituents. Planta Med. 1994, 60, 417–420. [Google Scholar] [CrossRef] [PubMed]
- Amagase, H.; Milner, J. Impact of various sources of garlic and their constituents on 7,12-dimethylbenz[α]anthracene binding to mammary cell DNA. Carcinogenesis 1994, 14, 1627–1631. [Google Scholar] [CrossRef]
- Li, G.; Qiao, C.H.; Lin, R.I.; Pinto, J.; Osborne, M.P.; Tiwari, R.K. Anti-proliferative effects of garlic constituents in cultured human breast cancer cells. Oncol. Rep. 1995, 2, 787–791. [Google Scholar] [PubMed]
- Jandke, J.; Spiteller, G. Unusual conjugates in biological profiles originating from consumption of onions and garlic. J. Chromatogr. 1987, 421. [Google Scholar] [CrossRef]
- Steiner, M.; Li, W. Aged garlic extract, a modulator of cardiovascular risk factors. J. Nutr. 2001, 131, 980S–984S. [Google Scholar] [PubMed]
- Pintana, H.; Sripetchwandee, J.; Supakul, L.; Apaijai, N.; Chattipakorn, N.; Chattipakorn, S. Garlic extract attenuates brain mitochondrial dysfunction and cognitive deficit in obese-insulin resistant rats. Appl. Physiol. Nutr. Metab. 2014, 39, 1373–1379. [Google Scholar] [CrossRef] [PubMed]
- Aebi, H.E. Catalase. In Methods of Enzymatic Analysis, 3rd ed.; Bergmeyer, H.U., Bergmeyer, J., Graßl, M., Eds.; VerlagChemie: Weinheim, Germany, 1986; Volume VIII, pp. 273–286. [Google Scholar]
- Sample Availability: Samples and extracts are available from the authors.
© 2016 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons by Attribution (CC-BY) license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kim, H.K. Garlic Supplementation Ameliorates UV-Induced Photoaging in Hairless Mice by Regulating Antioxidative Activity and MMPs Expression. Molecules 2016, 21, 70. https://doi.org/10.3390/molecules21010070
Kim HK. Garlic Supplementation Ameliorates UV-Induced Photoaging in Hairless Mice by Regulating Antioxidative Activity and MMPs Expression. Molecules. 2016; 21(1):70. https://doi.org/10.3390/molecules21010070
Chicago/Turabian StyleKim, Hye Kyung. 2016. "Garlic Supplementation Ameliorates UV-Induced Photoaging in Hairless Mice by Regulating Antioxidative Activity and MMPs Expression" Molecules 21, no. 1: 70. https://doi.org/10.3390/molecules21010070
APA StyleKim, H. K. (2016). Garlic Supplementation Ameliorates UV-Induced Photoaging in Hairless Mice by Regulating Antioxidative Activity and MMPs Expression. Molecules, 21(1), 70. https://doi.org/10.3390/molecules21010070




