Removal of Indoor Volatile Organic Compounds via Photocatalytic Oxidation: A Short Review and Prospect
Abstract
:1. Introduction
2. VOCs in Indoor Environment
2.1. Sources of VOCs Indoors
| VOCs | Possible Sources |
|---|---|
| Formaldehyde | Pesticides, flooring materials, insulating materials, wood-based materials, machine, coatings and paints |
| Toluene | Pesticides, flooring materials, insulating materials, wood-based materials, paints, adhesives, gasoline, combustion sources |
| Acetaldehyde | Wood-based materials, flooring materials, HVAC system |
| Paradichlorobenzene | Ceiling materials, wood-based materials, pesticides |
| Ethylbenzene | Furniture, paints, adhesives, gasoline, combustion sources |
| Methylene chloride | Flooring materials, furniture, HVAC system, coatings and paints |
| Chloroethylene | Flooring materials, coatings and paints, dry-cleaned clothes |
| Carbon tetrachloride | Coatings and paints, industrial strength cleaners |
| Chloroform | Pesticide, glue |
| Naphthalene | Insulating materials, mixed materials, wall painting |
| Other VOCs (e.g., esters and ketones) | Plastics, resins, plasticizers, solvents usage, flavors, perfumes, paints, disinfectants, adhesives |
2.1.1. Indoor Sources
2.1.2. Outdoor Sources
| Techniques | Principle | By-Product | Advantage | Disadvantage | Ref. |
|---|---|---|---|---|---|
| Botanical purification | Air is passed through a planted soil or directly on the plants. The contaminants are then degraded by microorganisms and/or plants, the precise mechanisms being unclear. | CO2, organic and amino acids | Low cost, no secondary pollution, beautifying the indoor environment | The purification effect is bad for high concentration pollutants | [28,62] |
| Catalytic combustion | Combustion of VOCs at low temperature with the help of a catalyst. | CO2, H2O | Wide range of application coverage, high efficiency, no secondary pollution | Not suitable for gas containing dust particles and droplets | [63,64] |
| Bio-filtration | Bio-filtration is a process in which contaminated airs passed through a biological stuffing medium that supports many kinds microorganism that biodegrade the VOCs. | Biomass | Little or no energy needs to be added in the form of heat or radiation to support this process | The equipment is big, long residence time, easy to jam | [65,66] |
| Absorption | Absorption is used to remove VOCs from gas streams by contacting the contaminated air with a liquid solvent. | Wastewater | Product recovery can offset annual operating costs | High demands on absorbent, complex process, high cost | [24] |
| Zeolite based adsorption | Air pollutants are adsorbed onto zeolites, often as filtration post-treatment. | Spent zeolite and collected organics | Effective in more than 90% RH as the adsorbent might be too specific | Pollutant reemission | [67] |
| Activated carbon based adsorption | VOCs are removed from the inlet air by physical adsorption onto the surface of the carbon. | Spent carbon and collected organics | Recovery of compounds, which may offset annual operating costs | They are flammable, difficult to regenerate for high boiling solvents, promote polymerization or oxidation of some solvents to toxic or insoluble compounds, and require humidity control. | [68] |
| Membrane Separation | Pollutants are passed through a membrane into another fluid by affinity separation. | Exhausted membrane | No further treatment, simple process, small energy consumption, no secondary pollution | The stability of the membrane was poor | [69] |
3. Traditional Removal Approaches
4. Removal of Indoor VOCs and Formaldehyde via Photocatalytic Oxidation
4.1. Removal of VOCs by Photocatalytic Oxidation
| Target VOC | Concentration (ppm) | Light Source | Main Intermediates | Chemical | Analytical Method | Ref. |
|---|---|---|---|---|---|---|
| Benzene | 3000–6000 | 4000 W Xe lamp | Benzaldehyde, benzoic acid | - | GC/MS | [83] |
| 614 | White fluorescent lamp | Phenol | Hydroquinone, 1,4-benzoquinone | GC/MS | [84] | |
| - | - | Phenol, hydro-quinone, benzoic acid | Malonic acid, benzoquinone | GC/MS/FTIR | [85] | |
| Toluene | 10 | Black light lamp | Benzaldehyde, benzoic acid | Benzyl alcohol | FTIR | [86] |
| 50–800 | 365 nm UV | Acetone, acetaldehyde, formaldehyde | Acrolein, butanone | TDS-GC/MS/FID, HPLC/UV/FTIR | [87] | |
| 370 | >400 nm | Benzaldehyde, benzoic acid | - | DRIFTS | [88] | |
| Xylene | 3000–6000 | 4000 W Xe lamp | Benzaldehyde, Methyl-benzaldehydes | 2,5-Furandione, 1,3-isobenzofurandione | GC/MS | [83] |
| 25–75 | UV | o-Tolualdehyde, o-toluic acid, benzoate ion | - | FTIR | [89] |
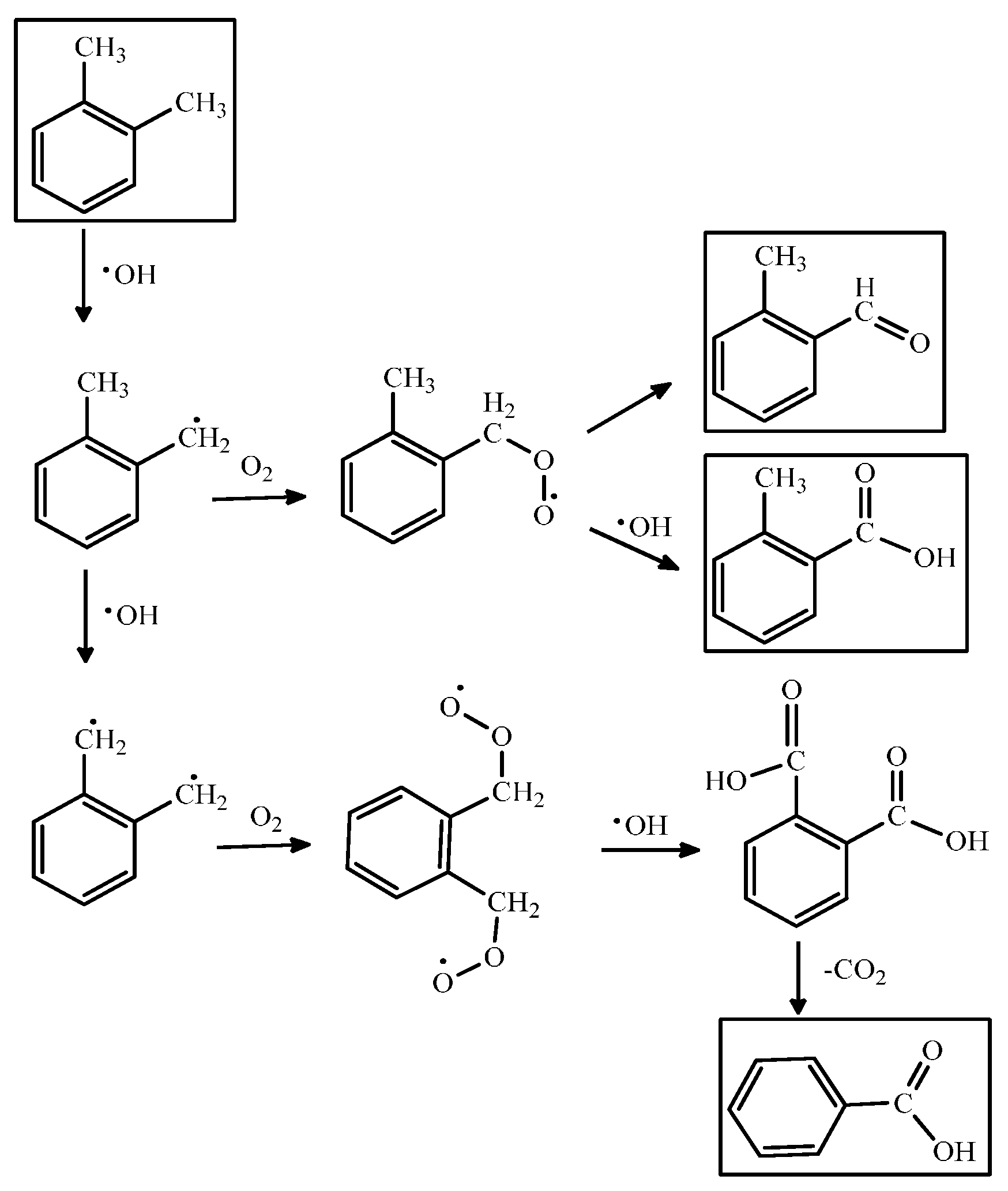
| Photocatalyst | Preparation/Coating Method | Configuration | Compounds | Light Source | ηremoval (%) | Ref. |
|---|---|---|---|---|---|---|
| TiO2 | Sol-gel | F | Acetone, toluene p-xylene | UV lamp, 254 nm | 77–62 (3 L/min) | [95] |
| TiO2 | Electrochemical | F | Acetaldehyde | UV | 99+ (110 min) | [93] |
| TiO2 | Sol-gel | F | Toluene | Black light | 52 (3.6 L/min) | [86] |
| TiO2 | Plasma deposited | F | m-Xylene | UV lamp | 99+ (30 min) | [94] |
| TiO2−xNx | Calcination | P | Toluene | Visible light | 99+ (3000 min) | [82] |
| TiO2−xNx | Hydrothermal | P | Acetaldehyde | Fluorescence | - | [98] |
| C-TiO2 | Hydrothermal | P | Toluene | Visible light | 60+ (120 min) | [106] |
| C-TiO2 | Hydrothermal | P | Toluene | Visible light | 20 (120 min) | [107] |
| CNT-TiO2 | Hydrothermal | P | Styrene | UV-LED, 365 nm | 50 (20 mL/min) | [108] |
| Pt/TiO2 | Photo-deposition | P | Benzene | Black light, 300–420 nm | 100 (100 mL/min) | [99] |
| Ln3+-TiO2 | Sol-gel | P | Benzene, toluene, ethylbenzene, o-xylene | UV, 365 nm | 22–79 | [109] |
| Ce-TiO2 | Sol-gel | F | Toluene | Visible light | 90 | [110] |
| Fe-TiO2 | Sol-gel | P | p-Xylene | Visible light—LED | 22 (5 min) | [111] |
| Fe-TiO2 | Sol-gel | P | Toluene | Visible light | 99+ (120 min) | [88] |
| In(OH)3 | Ultrasound radiation | P | Acetone, Benzene, Toluene | UV lamp, 254 nm | 99+ (5 h) | [104] |
| β-Ga2O3 | Chemical deposition | P | Benzene | UV-lamp, 254 nm | 60 (20 mL/min) | [105] |
| Ag4V2O7/Ag3VO4 | Hydrothermal | P | Benzene | White fluorescent lamp | 99+ (120 min) | [84] |
| Pt/WO3 | Photo-deposition | P | DCA, 4-CP, TMA | Visible light, >420 nm | 99+ (3 h) | [112] |
| Pd/WO3 | Calcination | P | Acetaldehyde, toluene | Fluorescence/visible light | 99+ (3 h) | [26] |
4.2. Removal of Formaldehyde by Photocatalytic Oxidation
| Catalyst | Preparation Method | HCHO Concentration | Light Source | Conversion Efficiency | Ref. |
|---|---|---|---|---|---|
| Mesoporous TiO2 | Evaporation-induced self-assembly | 30 ppm | UV light | 95.8% | [114] |
| Amorphous TiO2 film | CVD method | 50–55 ppm | UV light | 80% | [115] |
| PEG modified TiO2 film | Sol-gel method | 20 ppm | UV light | 95% | [116] |
| TiO2 coating on polyester fiber | Spray coating | 24.6 ± 2.8 ppm | UV light | 90% | [117] |
| UV/TiO2/O3 | Sol-gel | 18 ppm | UV light | 79.4% | [118] |
| Ag/TiO2 | Incipient wet impregnation | 500 ppm | UV light | Above 95% | [119] |
| Pt@TiO2 | Reverse micelle sol-gel | 10 ppm | Vis light | 98.3% | [120] |
| Ce/TiO2 | Sol-gel | 1 ppb | UV light | Above 70% | [121] |
| Pd-TiO2 film | Sol-gel dip coating | 500 ppb | UV light | Above 95% | [122] |
| Acrylic-silicon/nano-TiO2 | Emulsion blend | 0.8 ppm | Vis light | 83.4% | [123] |
| N-doped TiO2 film | Precipitation-peptization | 0.24 ppm | Vis light | 90% | [124] |
| AC loading TiO2 | Microwave-assisted synthetic | 30 ppm | UV light | 58.68% | [22] |
| Pt@SnO2 | Sol-gel method | — | Vis light | 93.2% | [125] |
| α-Bi2O3 | Calcination of hydrothermally prepared (BiO)2CO3 | 100 ppm | Vis light | 62.5% | [25] |
| Nano-ZnO | Mixing-calcination | 2.5–25 ppb | UV light | 73% | [126] |
| Zr0.08Ti0.92O2 | Sol-gel method | 0.08 ppb | UV-vis light | 92% | [127] |
| Zn2SnO4 | Hydrothermal method | 2 ppm | UV-vis light | 70% | [128] |
4.3. Influencing Factors
| Pollutants | Reactor Design | Initial Reaction Conditions | Deactivation | Ref. | ||||
|---|---|---|---|---|---|---|---|---|
| RT | Photocatalyst | [VOC] Gas (ppm) | PW(nm)/I (mW·cm−2) | RH (%) | T (°C) | |||
| Styrene | CR | CNT-TiO2 | 25 ± 1.5 | 365/70 | - | - | Y | [108] |
| Benzene | CR | Pt/TiO2 | 80 | 300–420/- | 65 | Ambient | n.r. | [99] |
| CR | In(OH)3 | 920 | 245/- | - | 25 | n.r. | [104] | |
| Acetone | CR | In(OH)3 | 420 | 245/- | - | 30 ± 1 | n.r. | [104] |
| Toluene | CR | TiO2 | 10 | >300/0.7 | 0–40 | Ambient | Y | [86] |
| CR | TiO2 | 17–35 | 365/2.34 | 47 | 25 | n.r. | [104] | |
| CR | P25 | 50–800 | 365/10 ± 1 | 0–50 | 25 | n.r. | [87,104] | |
| CR | Ce-TiO2 | 0.15–0.6 | Visible/- | <3–75 | 42 | n.r. | [110] | |
| CR | Fe-TiO2 | 370 | >400/- | 60 | 25 | Y + N | [88] | |
| CR | Ln3+-TiO2 | 23 ± 2 | 365/0.75 | - | - | n.r. | [109] | |
| CR | In(OH)3 | 1220 | 245/- | - | 25 | n.r. | [104] | |
| CR | TiO2 fibers | 200 | 365/9 | 20–60 | - | n.r. | [133] | |
| Xylene | CR | P25 | 25–75 | UV/1.5 | 30–90 | - | Y | [89] |
5. Summary and Outlook
Acknowledgments
Author Contributions
Conflicts of Interest
References
- World Health Organization (WHO). Air Quality Guidelines for Europe; World Health Organization: Geneva, Switzerland, 2000. [Google Scholar]
- Weschler, C.J. Changes in indoor pollutants since the 1950s. Atmos. Environ. 2009, 43, 153–169. [Google Scholar] [CrossRef]
- Bolden, A.L.; Kwiatkowski, C.F.; Colborn, T. New look at BTEX: Are ambient levels a problem? Environ. Sci. Technol. 2015, 49, 5261–5276. [Google Scholar] [CrossRef] [PubMed]
- Salthammer, T.; Mentese, S.; Marutzky, R. Formaldehyde in the indoor environment. Chem. Rev. 2010, 110, 2536–2572. [Google Scholar] [CrossRef] [PubMed]
- Jedrychowski, W.; Perera, F.; Mrozek-Budzyn, D.; Mroz, E.; Flak, E.; Spengler, J.D.; Edwards, S.; Jacek, R.; Kaim, I.; Skolicki, Z. Gender differences in fetal growth of newborns exposed prenatally to airborne fine particulate matter. Environ. Res. 2009, 109, 447–456. [Google Scholar] [CrossRef] [PubMed]
- U.S. Environmental Protection Agency. The Inside Story: A Guide to Air Quality; Office of Radiation and Indoor Air (6609J): Washington, DC, USA, 2009.
- U.S. Environmental Protection Agency. Compendium Method TO-11A: Determination of Formaldehyde in Ambient Air Using Adsorbent cartridge Followed by High Performance Liquid Chromatography; Center for Environmental Research Information, Office of Research and Development, U.S. Environmental Protection Agency: Cincinnati, OH, USA, 1999.
- Ho, S.S.H.; Yu, J.Z. Determination of airborne carbonyls: Comparison of a thermal desorption/GC method with the standard DNPH/HPLC method. Environ. Sci. Technol. 2004, 38, 862–870. [Google Scholar] [CrossRef] [PubMed]
- Guo, H.; Murray, F.; Wilkinson, S. Evaluation of total volatile organic compound emissions from adhesives based on chamber tests. J. Air Waste Manag. Assoc. 2000, 50, 199–206. [Google Scholar] [CrossRef] [PubMed]
- Guo, H.; Kwok, N.H.; Cheng, H.R.; Lee, S.C.; Hung, W.T.; Li, Y.S. Formaldehyde and volatile organic compounds in Hong Kong homes: Concentrations and impact factors. Indoor Air 2009, 19, 206–217. [Google Scholar] [CrossRef] [PubMed]
- Wolkoff, P. Volatile organic compounds—Sources, measurements, emissions, and the impact on indoor air quality. Indoor Air 1995, 5, 1–73. [Google Scholar] [CrossRef]
- Hodgson, A.; Beal, D.; McIlvaine, J. Sources of formaldehyde, other aldehydes and terpenes in a new manufactured house. Indoor Air 2002, 12, 235–242. [Google Scholar] [CrossRef] [PubMed]
- Brinke, J.T.; Selvin, S.; Hodgson, A.; Fisk, W.; Mendell, M.; Koshland, C.; Daisey, J. Development of new volatile organic compound (VOC) exposure metrics and their relationship to “sick building syndrome” symptoms. Indoor Air 1998, 8, 140–152. [Google Scholar] [CrossRef]
- Mishra, S.; Ajello, L.; Ahearn, D.; Burge, H.; Kurup, V.; Pierson, D.; Price, D.; Samson, R.; Sandhu, R.; Shelton, B. Environmental mycology and its importance to public health. J. Med. Vet. Mycol. 1992, 30, 287–305. [Google Scholar] [CrossRef] [PubMed]
- Tang, X.; Bai, Y.; Duong, A.; Smith, M.T.; Li, L.; Zhang, L. Formaldehyde in China: Production, consumption, exposure levels, and health effects. Environ. Int. 2009, 35, 1210–1224. [Google Scholar] [CrossRef] [PubMed]
- Ewlad-Ahmed, A.M.; Morris, M.A.; Patwardhan, S.V.; Gibson, L.T. Removal of formaldehyde from air using functionalized silica supports. Environ. Sci. Technol. 2012, 46, 13354–13360. [Google Scholar] [CrossRef] [PubMed]
- Bakheet, A.A.; Mohd Zain, M.F.; Kadhum, A.A.; Abdalla, Z. Photocatalytic oxidation performance to removal of volatile organic compounds in indoor environment. Environ. Res. Eng. Manag. 2011, 58, 27–33. [Google Scholar] [CrossRef] [Green Version]
- Fujishima, A.; Honda, K. Photolysis-decomposition of water at the surface of an irradiated semiconductor. Nature 1972, 238, 37–38. [Google Scholar] [CrossRef] [PubMed]
- Lim, M.; Zhou, Y.; Wang, L.; Rudolph, V.; Lu, G.Q. Development and potential of new generation photocatalytic systems for air pollution abatement: An overview. Asia Pac. J. Chem. Eng. 2009, 4, 387–402. [Google Scholar] [CrossRef]
- Mo, J.; Zhang, Y.; Xu, Q.; Lamson, J.J.; Zhao, R. Photocatalytic purification of volatile organic compounds in indoor air: A literature review. Atmos. Environ. 2009, 43, 2229–2246. [Google Scholar] [CrossRef]
- Schneider, J.; Matsuoka, M.; Takeuchi, M.; Zhang, J.; Horiuchi, Y.; Anpo, M.; Bahnemann, D.W. Understanding TiO2 photocatalysis: Mechanisms and materials. Chem. Rev. 2014, 114, 9919–9986. [Google Scholar] [CrossRef] [PubMed]
- Tian, F.; Wu, Z.; Yan, Y.; Ge, X.; Tong, Y. Photodegradation of formaldehyde by activated carbon loading TiO2 synthesized via microwave irradiation. Korean J. Chem. Eng. 2015, 32, 1333–1339. [Google Scholar] [CrossRef]
- Chan-Soo, K.; Jung-Woo, S.; Sang-Hun, A.; Hee-Dong, J.; Tae-Oh, K. Photodegradation of volatile organic compounds using Zirconium-doped TiO2/SiO2 visible light photocatalysts. Chem. Eng. J. 2012, 204–206, 40–47. [Google Scholar]
- Khan, F.I.; Ghoshal, A.K. Removal of volatile organic compounds from polluted air. J. Loss Prev. Process Ind. 2000, 13, 527–545. [Google Scholar] [CrossRef]
- Ai, Z.H.; Huang, Y.; Lee, S.C.; Zhang, L.Z. Monoclinic α-Bi2O3 photocatalyst for efficient removal of gaseous NO and HCHO under visible light irradiation. J. Alloys Compd. 2011, 509, 2044–2049. [Google Scholar] [CrossRef]
- Arai, T.; Horiguchi, M.; Yanagida, M.; Gunji, T.; Sugihara, H.; Sayama, K. Complete oxidation of acetaldehyde and toluene over a Pd/WO3 photocatalyst under fluorescent- or visible-light irradiation. Chem. Commun. 2008, 43, 5565–5567. [Google Scholar] [CrossRef] [PubMed]
- Dong, F.; Wang, Z.; Li, Y.; Ho, W.-K.; Lee, S.C. Immobilization of polymeric g-C3N4 on structured ceramic foam for efficient visible light photocatalytic air purification with real indoor illumination. Environ. Sci. Technol. 2014, 48, 10345–10353. [Google Scholar] [CrossRef] [PubMed]
- Soreanu, G.; Dixon, M.; Darlington, A. Botanical biofiltration of indoor gaseous pollutants—A mini review. Chem. Eng. J. 2013, 229, 585–594. [Google Scholar] [CrossRef]
- Tokumura, M.; Wada, Y.; Usami, Y.; Yamaki, T.; Mizukoshi, A.; Noguchi, M.; Yanagisawa, Y. Method of removal of volatile organic compounds by using wet scrubber coupled with photo-fenton reaction—Preventing emission of by-products. Chemosphere 2012, 89, 1238–1242. [Google Scholar] [CrossRef] [PubMed]
- Biard, P.F.; Couvert, A.; Renner, C.; Levasseur, J.P. Assessment and optimisation of VOC mass transfer enhancement by advanced oxidation process in a compact wet scrubber. Chemosphere 2009, 77, 182–187. [Google Scholar] [CrossRef] [PubMed]
- Dewulf, J.; Langenhove, H.V.; Smedt, E.D.; Geuens, S. Combination of advanced oxidation processes and gas absorption for the treatment of chlorinated solvents in waste gases. Water Sci. Technol. 2001, 44, 173–180. [Google Scholar] [PubMed]
- Kabir, E.; Kim, K.H. A review of some representative techniques for controlling the indoor volatile organic compounds. Asian J. Atmos. Environ. 2012, 6, 137–146. [Google Scholar] [CrossRef]
- Peral, J.; Domenech, X.; Ollis, D.F. Heterogeneous photocatalysis for purification, decontamination and deodorization of air. J. Chem. Technol. Biotechnol. 1997, 70, 117–140. [Google Scholar] [CrossRef]
- Adriana, Z.; Andreas, H.; Michal, N. Photocatalytic air purification. Recent Pat. Eng. 2010, 4, 200–216. [Google Scholar]
- Wang, S.; Ang, H.M.; Tade, M.O. Volatile organic compounds in indoor environment and photocatalytic oxidation: State of the art. Environ. Int. 2007, 33, 694–705. [Google Scholar] [CrossRef] [PubMed]
- Zhong, L.; Haghighat, F. Photocatalytic air cleaners and materials technologies—Abilities and limitations. Build. Environ. 2015, 91, 191–203. [Google Scholar] [CrossRef]
- Hay, S.O.; Obee, T.; Luo, Z.; Jiang, T.; Meng, Y.; He, J.; Murphy, S.C.; Suib, S. The viability of photocatalysis for air purification. Molecules 2015, 20, 1319–1356. [Google Scholar] [CrossRef] [PubMed]
- Li, W.; Wang, J.; Gong, H. Catalytic combustion of VOCs on non-noble metal catalysts. Catal. Today 2009, 148, 81–87. [Google Scholar] [CrossRef]
- Salthammer, T.; Fuhrmann, F.; Kaufhold, S.; Meyer, B.; Schwarz, A. Effects of climatic parameters on formaldehyde concentrations in indoor air. Indoor Air 1995, 5, 120–128. [Google Scholar] [CrossRef]
- Quiroz Torres, J.; Royer, S.; Bellat, J.P.; Giraudon, J.M.; Lamonier, J.F. Formaldehyde: Catalytic oxidation as a promising soft way of elimination. ChemSusChem 2013, 6, 578–592. [Google Scholar] [CrossRef] [PubMed]
- Wieslander, G.; Norbäck, D.; Björnsson, E.; Janson, C.; Boman, G. Asthma and the indoor environment: The significance of emission of formaldehyde and volatile organic compounds from newly painted indoor surfaces. Int. Arch. Occup. Environ. Health 1996, 69, 115–124. [Google Scholar] [CrossRef]
- Hoffmann, K.; Krause, C.; Seifert, B.; Ullrich, D. The German environmental survey 1990/92 (GerES II): Sources of personal exposure to volatile organic compounds. J. Expo. Anal. Environ. Epidemiol. 1999, 10, 115–125. [Google Scholar] [CrossRef]
- Benner, C.L.; Bayona, J.M.; Caka, F.M.; Tang, H.; Lewis, L.; Crawford, J.; Lamb, J.D.; Lee, M.L.; Lewis, E.A. Chemical composition of environmental tobacco smoke. 2. Particulate-phase compounds. Environ. Sci. Technol. 1989, 23, 688–699. [Google Scholar] [CrossRef]
- Fenske, J.D.; Paulson, S.E. Human breath emissions of vocs. J. Air Waste Manag. 1999, 49, 594–598. [Google Scholar] [CrossRef]
- Gupta, K.; Ulsamer, A.; Preuss, P. Formaldehyde in indoor air: Sources and toxicity. Environ. Int. 1982, 8, 349–358. [Google Scholar] [CrossRef]
- Zinn, T.W.; Cline, D.; Lehmann, W.F. Long-term study of formaldehyde emission decay from particleboard. For. Prod. J. 1990, 40, 15–18. [Google Scholar]
- Balakrishnan, K.; Ramaswamy, P.; Sankar, S. Biomass smoke and health risks—The situation in developing countries. In Air Pollution; Springer: Berlin, Germany, 2004; pp. 219–239. [Google Scholar]
- Bruce, N.; Perez-Padilla, R.; Albalak, R. Indoor air pollution in developing countries: A major environmental and public health challenge. Bull. World Health Organ. 2000, 78, 1078–1092. [Google Scholar] [PubMed]
- Raiyani, C.; Shah, S.; Desai, N.; Venkaiah, K.; Patel, J.; Parikh, D.; Kashyap, S. Characterization and problems of indoor pollution due to cooking stove smoke. Atmos. Environ. A Gen. Top. 1993, 27, 1643–1655. [Google Scholar] [CrossRef]
- Li, S.; Banyasz, J.; Parrish, M.; Lyons-Hart, J.; Shafer, K. Formaldehyde in the gas phase of mainstream cigarette smoke. J. Anal. Appl. Pyrolysis 2002, 65, 137–145. [Google Scholar] [CrossRef]
- Sherman, M.H.; Hodgson, A. Formaldehyde as a basis for residential ventilation rates. Indoor Air 2004, 14, 2–8. [Google Scholar] [CrossRef] [PubMed]
- Andreae, M.O.; Crutzen, P.J. Atmospheric aerosols: Biogeochemical sources and role in atmospheric chemistry. Science 1997, 276, 1052–1058. [Google Scholar] [CrossRef]
- Di Carlo, P.; Brune, W.H.; Martinez, M.; Harder, H.; Lesher, R.; Ren, X.; Thornberry, T.; Carroll, M.A.; Young, V.; Shepson, P.B. Missing OH reactivity in a forest: Evidence for unknown reactive biogenic VOCs. Science 2004, 304, 722–725. [Google Scholar] [CrossRef] [PubMed]
- Karl, T.; Guenther, A.; Yokelson, R.J.; Greenberg, J.; Potosnak, M.; Blake, D.R.; Artaxo, P. The tropical forest and fire emissions experiment: Emission, chemistry, and transport of biogenic volatile organic compounds in the lower atmosphere over amazonia. J. Geophys. Res. Atmos. 2007, 112. [Google Scholar] [CrossRef]
- Williams, J.; Yassaa, N.; Bartenbach, S.; Lelieveld, J. Mirror image hydrocarbons from tropical and boreal forests. Atmos. Chem. Phys. 2007, 7, 973–980. [Google Scholar] [CrossRef]
- Kuhn, U.; Andreae, M.; Ammann, C.; Araújo, A.; Brancaleoni, E.; Ciccioli, P.; Dindorf, T.; Frattoni, M.; Gatti, L.; Ganzeveld, L. Isoprene and monoterpene fluxes from central amazonian rainforest inferred from tower-based and airborne measurements, and implications on the atmospheric chemistry and the local carbon budget. Atmos. Chem. Phys. 2007, 7, 2855–2879. [Google Scholar] [CrossRef]
- Fehsenfeld, F.; Calvert, J.; Fall, R.; Goldan, P.; Guenther, A.B.; Hewitt, C.N.; Lamb, B.; Liu, S.; Trainer, M.; Westberg, H. Emissions of volatile organic compounds from vegetation and the implications for atmospheric chemistry. Glob. Biogeochem. Cycles 1992, 6, 389–430. [Google Scholar] [CrossRef]
- Atkinson, R.; Arey, J. Gas-phase tropospheric chemistry of biogenic volatile organic compounds: A review. Atmos. Environ. 2003, 37, 197–219. [Google Scholar] [CrossRef]
- Carlier, P.; Hannachi, H.; Mouvier, G. The chemistry of carbonyl compounds in the atmosphere—A review. Atmos. Environ. 1986, 20, 2079–2099. [Google Scholar] [CrossRef]
- Possanzini, M.; di Palo, V.; Cecinato, A. Sources and photodecomposition of formaldehyde and acetaldehyde in rome ambient air. Atmos. Environ. 2002, 36, 3195–3201. [Google Scholar] [CrossRef]
- Hoekman, S.K. Speciated measurements and calculated reactivities of vehicle exhaust emissions from conventional and reformulated gasolines. Environ. Sci. Technol. 1992, 26, 1206–1216. [Google Scholar] [CrossRef]
- Kim, K.J.; Kil, M.J.; Song, J.S.; Yoo, E.H.; Son, K.C.; Kays, S.J. Efficiency of volatile formaldehyde removal by indoor plants: Contribution of aerial plant parts versus the root zone. J. Am. Soc. Hortic. Sci. 2008, 133, 521–526. [Google Scholar]
- Sahle-Demessie, E.; Devulapelli, V.G. Oxidation of methanol and total reduced sulfur compounds with ozone over V2O5/TiO2 catalyst: Effect of humidity. Appl. Catal. A Gen. 2009, 361, 72–80. [Google Scholar] [CrossRef]
- Kilham, L.B.; Dodd, R.M. The Application of Ozone for Air Treatment: Case Study of a Bingo Hall HAVC System; International Ozone Association World Congress: Dearborn, MI, USA, 1999. [Google Scholar]
- Lau, S.; Groody, K.; Chan, A.; Ragib, G. Control of reduced sulphur and VOC emissions via biofiltration. Pulp Pap. Can. 2006, 107, 57–63. [Google Scholar]
- Kumar, T.P.; Rahul, M.; Chandrajit, B. Biofiltration of volatile organic compounds (VOCs)—An overview. Res. J. Chem. Sci. 2011, 1, 83–92. [Google Scholar]
- Wong, C.T. Removal of Volatile Organic Compound (VOC) from Air Using Zeolite Based Adsorption-Catalytic Combustion System. Master’s Thesis, Universiti Sains Malaysia, Penang, Malaysia, 2007. [Google Scholar]
- Mofidi, A.; Asilian, H.; Jafari, A.J. Adsorption of volatile organic compounds on fluidized activated carbon bed. Health Scope 2013, 2, 84–89. [Google Scholar] [CrossRef]
- Liu, Y.; Feng, X.; Lawless, D. Separation of gasoline vapor from nitrogen by hollow fiber composite membranes for VOC emission control. J. Membr. Sci. 2006, 271, 114–124. [Google Scholar] [CrossRef]
- Raso, R.A.; Zeltner, M.; Stark, W.J. Indoor air purification using activated carbon adsorbers: Regeneration using catalytic combustion of intermediately stored VOC. Ind. Eng. Chem. Res. 2014, 53, 19304–19312. [Google Scholar] [CrossRef]
- Jiao, Z.; Luo, P.; Wu, Y.; Ding, S.; Zhang, Z. Absorption of lean formaldehyde from air with Na2SO3 solution. J. Hazard. Mater. 2006, 134, 176–182. [Google Scholar] [CrossRef] [PubMed]
- Kim, D.I.; Park, J.H.; Do Kim, S.; Lee, J.Y.; Yim, J.H.; Jeon, J.K.; Park, S.H.; Park, Y.K. Comparison of removal ability of indoor formaldehyde over different materials functionalized with various amine groups. J. Ind. Eng. Chem. 2011, 17, 1–5. [Google Scholar] [CrossRef]
- Nuasaen, S.; Opaprakasit, P.; Tangboriboonrat, P. Hollow latex particles functionalized with chitosan for the removal of formaldehyde from indoor air. Carbohydr. Polym. 2014, 101, 179–187. [Google Scholar] [CrossRef] [PubMed]
- Shimizu, T.; Matsui, K.; Kitayama, I.; Nishijima, T. Development of aldehyde capture material to cabin air filter. Mazda Tech. Rev. 2000, 18, 104–111. [Google Scholar]
- Chang, M.B.; Lee, C.C. Destruction of formaldehyde with dielectric barrier discharge plasmas. Environ. Sci. Technol. 1995, 29, 181–186. [Google Scholar] [CrossRef] [PubMed]
- Zuo, H.; Zhang, H.; Zhang, X.; Han, N. Removal of formaldehyde from overactivated-carbon-fiber-loaded biological enzyme. J. Appl. Polym. Sci. 2013, 130, 2619–2623. [Google Scholar] [CrossRef]
- Ohko, Y.; Hashimoto, K.; Fujishima, A. Kinetics of photocatalytic reactions under extremely low-intensity UV illumination on titanium dioxide thin films. J. Phys. Chem. A 1997, 101, 8057–8062. [Google Scholar] [CrossRef]
- Zhang, T.; Zhu, D.B.; Wang, C.G.; He, X. Antibacterial behavior of Ag-Ce-TiO2 composite films in the different irradiations. Mater. Sci. Forum 2009, 610–613, 463–466. [Google Scholar] [CrossRef]
- Zaleska, A. Doped-TiO2: A review. Recent Pat. Eng. 2008, 2, 157–164. [Google Scholar] [CrossRef]
- Sun, Y.; Fang, L.; Wyon, D.P.; Wisthaler, A.; Lagercrantz, L.; Strøm-Tejsen, P. Experimental research on photocatalytic oxidation air purification technology applied to aircraft cabins. Build. Environ. 2008, 43, 258–268. [Google Scholar] [CrossRef]
- Maira, A.; Coronado, J.; Augugliaro, V.; Yeung, K.L.; Conesa, J.; Soria, J. Fourier transform infrared study of the performance of nanostructured TiO2 particles for the photocatalytic oxidation of gaseous toluene. J. Catal. 2001, 202, 413–420. [Google Scholar] [CrossRef]
- Irokawa, Y.; Morikawa, T.; Aoki, K.; Kosaka, S.; Ohwaki, T.; Taga, Y. Photodegradation of toluene over TiO2-xNx under visible light irradiation. Phys. Chem. Chem. Phys. 2006, 8, 1116–1121. [Google Scholar] [CrossRef] [PubMed]
- Blanco, J.; Avila, P.; Bahamonde, A.; Alvarez, E.; Sanchez, B.; Romero, M. Photocatalytic destruction of toluene and xylene at gas phase on a titania based monolithic catalyst. Catal. Today 1996, 29, 437–442. [Google Scholar] [CrossRef]
- Chen, L.C.; Pan, G.T.; Yang, T.C.K.; Chung, T.W.; Huang, C.M. In situ drift and kinetic studies of photocatalytic degradation on benzene vapor with visible-light-driven silver vanadates. J. Hazard. Mater. 2010, 178, 644–651. [Google Scholar] [CrossRef] [PubMed]
- Zhong, J.; Wang, J.; Tao, L.; Gong, M.; Zhimin, L.; Chen, Y. Photocatalytic degradation of gaseous benzene over TiO2/Sr2CeO4: Kinetic model and degradation mechanisms. J. Hazard. Mater. 2007, 139, 323–331. [Google Scholar] [CrossRef] [PubMed]
- Cao, L.; Gao, Z.; Suib, S.L.; Obee, T.N.; Hay, S.O.; Freihaut, J.D. Photocatalytic oxidation of toluene on nanoscale TiO2 catalysts: Studies of deactivation and regeneration. J. Catal. 2000, 196, 253–261. [Google Scholar] [CrossRef]
- Debono, O.; Thevenet, F.; Gravejat, P.; Hequet, V.; Raillard, C.; Lecoq, L.; Locoge, N. Toluene photocatalytic oxidation at ppbv levels: Kinetic investigation and carbon balance determination. Appl. Catal. B Environ. 2011, 106, 600–608. [Google Scholar] [CrossRef]
- Sun, S.; Ding, J.; Bao, J.; Gao, C.; Qi, Z.; Yang, X.; He, B.; Li, C. Photocatalytic degradation of gaseous toluene on Fe-TiO2 under visible light irradiation: A study on the structure, activity and deactivation mechanism. Appl. Surf. Sci. 2012, 258, 5031–5037. [Google Scholar] [CrossRef]
- Ameen, M.M.; Raupp, G.B. Reversible catalyst deactivation in the photocatalytic oxidation of diluteo-xylene in air. J. Catal. 1999, 184, 112–122. [Google Scholar] [CrossRef]
- Einaga, H.; Mochiduki, K.; Teraoka, Y. Photocatalytic oxidation processes for toluene oxidation over TiO2 catalysts. Catalysts 2013, 3, 219–231. [Google Scholar] [CrossRef]
- Nakajima, A.; Obata, H.; Kameshima, Y.; Okada, K. Photocatalytic destruction of gaseous toluene by sulfated TiO2 powder. Catal. Commun. 2005, 6, 716–720. [Google Scholar] [CrossRef]
- Daghrir, R.; Drogui, P.; Robert, D. Modified TiO2 for environmental photocatalytic applications: A review. Ind. Eng. Chem. Res. 2013, 52, 3581–3599. [Google Scholar]
- Liu, Z.; Zhang, X.; Nishimoto, S.; Murakami, T.; Fujishima, A. Efficient photocatalytic degradation of gaseous acetaldehyde by highly ordered TiO2 nanotube arrays. Environ. Sci. Technol. 2008, 42, 8547–8551. [Google Scholar] [CrossRef] [PubMed]
- Sumitsawan, S.; Cho, J.; Sattler, M.L.; Timmons, R.B. Plasma surface modified TiO2 nanoparticles: Improved photocatalytic oxidation of gaseous m-xylene. Environ. Sci. Technol. 2011, 45, 6970–6977. [Google Scholar] [CrossRef] [PubMed]
- Liang, W.J.; Li, J.; Jin, Y.Q. Photocatalytic degradation of gaseous acetone, toluene, and p-xylene using a TiO2 thin film. J. Environ. Sci. Health A 2010, 45, 1384–1390. [Google Scholar] [CrossRef] [PubMed]
- Yu, J.C.; Ho, W.K.; Yu, J.G.; Yip, H.; Wong, P.K.; Zhao, J.C. Efficient visible-light-induced photocatalytic disinfection on sulfur-doped nanocrystalline titania. Environ. Sci. Technol. 2005, 39, 1175–1179. [Google Scholar] [CrossRef] [PubMed]
- Tan, T.T.Y.; Yip, C.K.; Beydoun, D.; Amal, R. Effects of nano-Ag particles loading on TiO2 photocatalytic reduction of selenate ions. Chem. Eng. J. 2003, 95, 179–186. [Google Scholar] [CrossRef]
- Li, H.; Yin, S.; Wang, Y.; Sato, T. Persistent fluorescence-assisted TiO2−xNy-based photocatalyst for gaseous acetaldehyde degradation. Environ. Sci. Technol. 2012, 46, 7741–7745. [Google Scholar] [CrossRef] [PubMed]
- Einaga, H.; Futamura, S.; Ibusuki, T. Complete oxidation of benzene in gas phase by platinized titania photocatalysts. Environ. Sci. Technol. 2001, 35, 1880–1884. [Google Scholar] [CrossRef] [PubMed]
- Reddy, B.M.; Sreekanth, P.M.; Reddy, E.P.; Yamada, Y.; Xu, Q.A.; Sakurai, H.; Kobayashi, T. Surface characterization of La2O3-TiO2 and V2O5/ La2O3-TiO2 catalysts. J. Phys. Chem. B 2002, 106, 5695–5700. [Google Scholar] [CrossRef]
- Cappelletti, G.; Pifferi, V.; Mostoni, S.; Falciola, L.; di Bari, C.; Spadavecchia, F.; Meroni, D.; Davoli, E.; Ardizzone, S. Hazardous o-toluidine mineralization by photocatalytic bismuth doped ZnO slurries. Chem. Commun. 2015, 51, 10459–10462. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Hu, J.S.; Ren, L.L.; Guo, Y.G.; Liang, H.P.; Cao, A.M.; Wan, L.J.; Bai, C.L. Mass production and high photocatalytic activity of ZnS nanoporous nanoparticles. Angew. Chem. Int. Ed. 2005, 44, 1269–1273. [Google Scholar] [CrossRef] [PubMed]
- Zeng, X.; Wu, J.; Zhang, D.; Li, G.; Zhang, S.; Zhao, H.; An, T.; Wang, X.; Fu, J.; Sheng, G. Degradation of toluene gas at the surface of ZnO/SnO2 photocatalysts in a baffled bed reactor. Res. Chem. Intermed. 2009, 35, 827–838. [Google Scholar] [CrossRef]
- Yan, T.; Long, J.; Shi, X.; Wang, D.; Li, Z.; Wang, X. Efficient photocatalytic degradation of volatile organic compounds by porous indium hydroxide nanocrystals. Environ. Sci. Technol. 2010, 44, 1380–1385. [Google Scholar] [CrossRef] [PubMed]
- Hou, Y.; Wang, X.; Wu, L.; Ding, Z.; Fu, X. Efficient decomposition of benzene over a beta-Ga2O3 photocatalyst under ambient conditions. Environ. Sci. Technol. 2006, 40, 5799–5803. [Google Scholar] [CrossRef] [PubMed]
- Dong, F.; Guo, S.; Wang, H.; Li, X.; Wu, Z. Enhancement of the visible light photocatalytic activity of C-doped TiO2 nanomaterials prepared by a green synthetic approach. J. Phys. Chem. C 2011, 115, 13285–13292. [Google Scholar] [CrossRef]
- Dong, F.; Wang, H.; Wu, Z. One-step “green” synthetic approach for mesoporous C-doped titanium dioxide with efficient visible light photocatalytic activity. J. Phys. Chem. C 2009, 113, 16717–16723. [Google Scholar] [CrossRef]
- An, T.; Chen, J.; Nie, X.; Li, G.; Zhang, H.; Liu, X.; Zhao, H. Synthesis of carbon nanotube-anatase TiO2 sub-micrometer-sized sphere composite photocatalyst for synergistic degradation of gaseous styrene. ACS Appl. Mater. Interfaces 2012, 4, 5988–5996. [Google Scholar] [CrossRef] [PubMed]
- Li, F.B.; Li, X.Z.; Ao, C.H.; Lee, S.C.; Hou, M.F. Enhanced photocatalytic degradation of VOCs using In3+-TiO2 catalysts for indoor air purification. Chemosphere 2005, 59, 787–800. [Google Scholar] [CrossRef] [PubMed]
- Sidheswaran, M.; Tavlarides, L.L. Visible light photocatalytic oxidation of toluene using a Cerium-doped titania catalyst. Ind. Eng. Chem. Res. 2008, 47, 3346–3357. [Google Scholar] [CrossRef]
- Luu, C.L.; Nguyen, Q.T.; Ho, S.T. Synthesis and characterization of Fe-doped TiO2 photocatalyst by the sol-gel method. Adv. Nat. Sci. Nanosci. Nanotechnol. 2010, 1, 015008. [Google Scholar] [CrossRef]
- Kim, J.; Lee, C.W.; Choi, W. Platinized WO3 as an environmental photocatalyst that generates oh radicals under visible light. Environ. Sci. Technol. 2010, 44, 6849–6854. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Chen, Q.; Zhang, Y. Study of ESR on the photocatalytic oxidation of methanal. Chin. J. Chem. Phys. 1998, 11, 05. [Google Scholar]
- Li, C.Y.; Jia, Y.R.; Zhang, X.C.; Zhang, S.Y.; Tang, A.D. Photocatalytic degradation of formaldehyde using mesoporous TiO2 prepared by evaporation-induced self-assembly. J. Cent. South Univ. 2014, 21, 4066–4070. [Google Scholar] [CrossRef]
- Sun, Z.G.; Li, X.S.; Zhu, X.; Deng, X.Q.; Chang, D.L.; Zhu, A.M. Facile and fast deposition of amorphous TiO2 film under atmospheric pressure and at room temperature, and its high photocatalytic activity under UVC light. Chem. Vap. Depos. 2014, 20, 8–13. [Google Scholar] [CrossRef]
- Wang, X.; Han, F.; Wang, X.; Li, Y. Effect of aluminum foam support and polyethylene glycol on surface morphology and photocatalytic behavior of TiO2 films. Mater.Chem. Phys. 2014, 145, 68–74. [Google Scholar] [CrossRef]
- Han, Z.; Chang, V.W.; Zhang, L.; Tse, M.S.; Tan, O.K.; Hildemann, L.M. Preparation of TiO2-coated polyester fiber filter by spray-coating and its photocatalytic degradation of gaseous formaldehyde. Aerosol Air Qual. Res. 2012, 12, 1327–1335. [Google Scholar] [CrossRef]
- Hong, Q.; SUN, D.Z.; CHI, G.Q. Formaldehyde degradation by UV/TiO2/O3 process using continuous flow mode. J. Environ. Sci. 2007, 19, 1136–1140. [Google Scholar]
- Shie, J.L.; Lee, C.H.; Chiou, C.S.; Chang, C.T.; Chang, C.C.; Chang, C.Y. Photodegradation kinetics of formaldehyde using light sources of UVA, UVC and UV-LED in the presence of composed silver titanium oxide photocatalyst. J. Hazard. Mater. 2008, 155, 164–172. [Google Scholar] [CrossRef] [PubMed]
- Zhu, Z.; Wu, R.J. The degradation of formaldehyde using a Pt@TiO2 nanoparticles in presence of visible light irradiation at room temperature. J. Taiwan Inst. Chem. Eng. 2015, 50, 276–281. [Google Scholar] [CrossRef]
- Liang, W.; Li, J.; Jin, Y. Photo-catalytic degradation of gaseous formaldehyde by TiO2/UV, Ag/TiO2/UV and Ce/TiO2/UV. Build. Environ. 2012, 51, 345–350. [Google Scholar] [CrossRef]
- Fu, P.; Zhang, P.; Li, J. Photocatalytic degradation of low concentration formaldehyde and simultaneous elimination of ozone by-product using palladium modified TiO2 films under UV irradiation. Appl. Catal. B Environ. 2011, 105, 220–228. [Google Scholar] [CrossRef]
- Xiao, G.; Huang, A.; Su, H.; Tan, T. The activity of acrylic-silicon/nano-TiO2 films for the visible-light degradation of formaldehyde and NO2. Build. Environ. 2013, 65, 215–221. [Google Scholar] [CrossRef]
- Liu, W.X.; Jiang, P.; Shao, W.N.; Zhang, J.; Cao, W.B. A novel approach for the synthesis of visible-light-active nanocrystalline N-doped TiO2 photocatalytic hydrosol. Solid State Sci. 2014, 33, 45–48. [Google Scholar] [CrossRef]
- Chang, Y.C.; Yan, C.Y.; Wu, R.J. Preparation of Pt@SnO2 Core-Shell nanoparticles for photocatalytic degradation of formaldehyde. J. Chin. Chem. Soc. 2014, 61, 345–349. [Google Scholar] [CrossRef]
- Rezaee, A.; Rangkooy, H.; Khavanin, A.; Jafari, A.J. High photocatalytic decomposition of the air pollutant formaldehyde using nano-ZnO on bone char. Environ. Chem. Lett. 2014, 12, 353–357. [Google Scholar] [CrossRef]
- Huang, Q.; Ma, W.; Yan, X.; Chen, Y.; Zhu, S.; Shen, S. Photocatalytic decomposition of gaseous hcho by ZrxTi1−xO2 catalysts under UV-vis light irradiation with an energy-saving lamp. J. Mol. Catal. A Chem. 2013, 366, 261–265. [Google Scholar] [CrossRef]
- Ai, Z.H.; Lee, S.C.; Huang, Y.; Ho, W.K.; Zhang, L.Z. Photocatalytic removal of NO and HCHO over nanocrystalline Zn2SnO4 microcubes for indoor air purification. J. Hazard. Mater. 2010, 179, 141–150. [Google Scholar] [CrossRef] [PubMed]
- Egerton, T.; King, C. The influence of light intensity on photoactivity in TiO2 pigmented systems. J. Oil Colour Chem. Assoc. 1979, 62, 386–391. [Google Scholar]
- Obee, T.N. Photooxidation of sub-parts-per-million toluene and formaldehyde levels on titania using a glass-plate reactor. Environ. Sci. Technol. 1996, 30, 3578–3584. [Google Scholar] [CrossRef]
- Okamoto, K.I.; Yamamoto, Y.; Tanaka, H.; Itaya, A. Kinetics of heterogeneous photocatalytic decomposition of phenol over anatase TiO2 powder. Bull. Chem. Soc. Jpn. 1985, 58, 2023–2028. [Google Scholar] [CrossRef]
- Qu, X.G.; Liu, W.X.; Ma, J.; Cao, W. Research on photodegradation of formaldehyde by nanocrystalline N-TiO2 powders under visible light irradiation. Res. Chem. Intermed. 2009, 35, 313–320. [Google Scholar] [CrossRef]
- Le Bechec, M.; Kinadjan, N.; Ollis, D.; Backov, R.; Lacombe, S. Comparison of kinetics of acetone, heptane and toluene photocatalytic mineralization over TiO2 microfibers and Quartzel® mats. Appl. Catal. B Environ. 2015, 179, 78–87. [Google Scholar] [CrossRef]
- Yang, R.; Zhang, Y.; Xu, Q.; Mo, J. A mass transfer based method for measuring the reaction coefficients of a photocatalyst. Atmos. Environ. 2007, 41, 1221–1229. [Google Scholar] [CrossRef]
- Obee, T.N.; Hay, S.O. Effects of moisture and temperature on the photooxidation of ethylene on titania. Environ. Sci. Tchnol. 1997, 31, 2034–2038. [Google Scholar] [CrossRef]
- Liotta, L. Catalytic oxidation of volatile organic compounds on supported noble metals. Appl. Catal. B Environ. 2010, 100, 403–412. [Google Scholar] [CrossRef]
- Ordóñez, S.; Bello, L.; Sastre, H.; Rosal, R.; Dı́ez, F.V. Kinetics of the deep oxidation of benzene, toluene, n-hexane and their binary mixtures over a platinum on γ-alumina catalyst. Appl. Catal. B Environ. 2002, 38, 139–149. [Google Scholar] [CrossRef]
- Sarigiannis, D.A.; Karakitsios, S.P.; Gotti, A.; Liakos, I.L.; Katsoyiannis, A. Exposure to major volatile organic compounds and carbonyls in European indoor environments and associated health risk. Environ. Int. 2011, 37, 743–765. [Google Scholar] [CrossRef] [PubMed]
- Wu, M.S.; Xu, X.Y.; Xu, X.; Wang, X.T.; Zhao, X.T.; Pei, M.; Ma, X.H.; Wang, M.Z.; Wang, F.; Li, J.X. Comparison of removal efficiency of formaldehyde by chlorine dioxide, photocatalyst and active carbon. Appl. Mech. Mater. 2015, 723, 648–651. [Google Scholar] [CrossRef]
- Park, D.R.; Zhang, J.; Ikeue, K.; Yamashita, H.; Anpo, M. Photocatalytic oxidation of ethylene to CO2 and H2O on ultrafine powdered TiO2 photocatalysts in the presence of O2 and H2O. J. Catal. 1999, 185, 114–119. [Google Scholar] [CrossRef]
- Han, Z.; Chang, V.W.; Wang, X.; Lim, T.T.; Hildemann, L. Experimental study on visible-light induced photocatalytic oxidation of gaseous formaldehyde by polyester fiber supported photocatalysts. Chem. Eng. J. 2013, 218, 9–18. [Google Scholar] [CrossRef]
- Méndez-Román, R.; Cardona-Martı́nez, N. Relationship between the formation of surface species and catalyst deactivation during the gas-phase photocatalytic oxidation of toluene. Catal. Today 1998, 40, 353–365. [Google Scholar] [CrossRef]
© 2016 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons by Attribution (CC-BY) license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Huang, Y.; Ho, S.S.H.; Lu, Y.; Niu, R.; Xu, L.; Cao, J.; Lee, S. Removal of Indoor Volatile Organic Compounds via Photocatalytic Oxidation: A Short Review and Prospect. Molecules 2016, 21, 56. https://doi.org/10.3390/molecules21010056
Huang Y, Ho SSH, Lu Y, Niu R, Xu L, Cao J, Lee S. Removal of Indoor Volatile Organic Compounds via Photocatalytic Oxidation: A Short Review and Prospect. Molecules. 2016; 21(1):56. https://doi.org/10.3390/molecules21010056
Chicago/Turabian StyleHuang, Yu, Steven Sai Hang Ho, Yanfeng Lu, Ruiyuan Niu, Lifeng Xu, Junji Cao, and Shuncheng Lee. 2016. "Removal of Indoor Volatile Organic Compounds via Photocatalytic Oxidation: A Short Review and Prospect" Molecules 21, no. 1: 56. https://doi.org/10.3390/molecules21010056
APA StyleHuang, Y., Ho, S. S. H., Lu, Y., Niu, R., Xu, L., Cao, J., & Lee, S. (2016). Removal of Indoor Volatile Organic Compounds via Photocatalytic Oxidation: A Short Review and Prospect. Molecules, 21(1), 56. https://doi.org/10.3390/molecules21010056









