On-Line Organic Solvent Field Enhanced Sample Injection in Capillary Zone Electrophoresis for Analysis of Quetiapine in Beagle Dog Plasma
Abstract
:1. Introduction
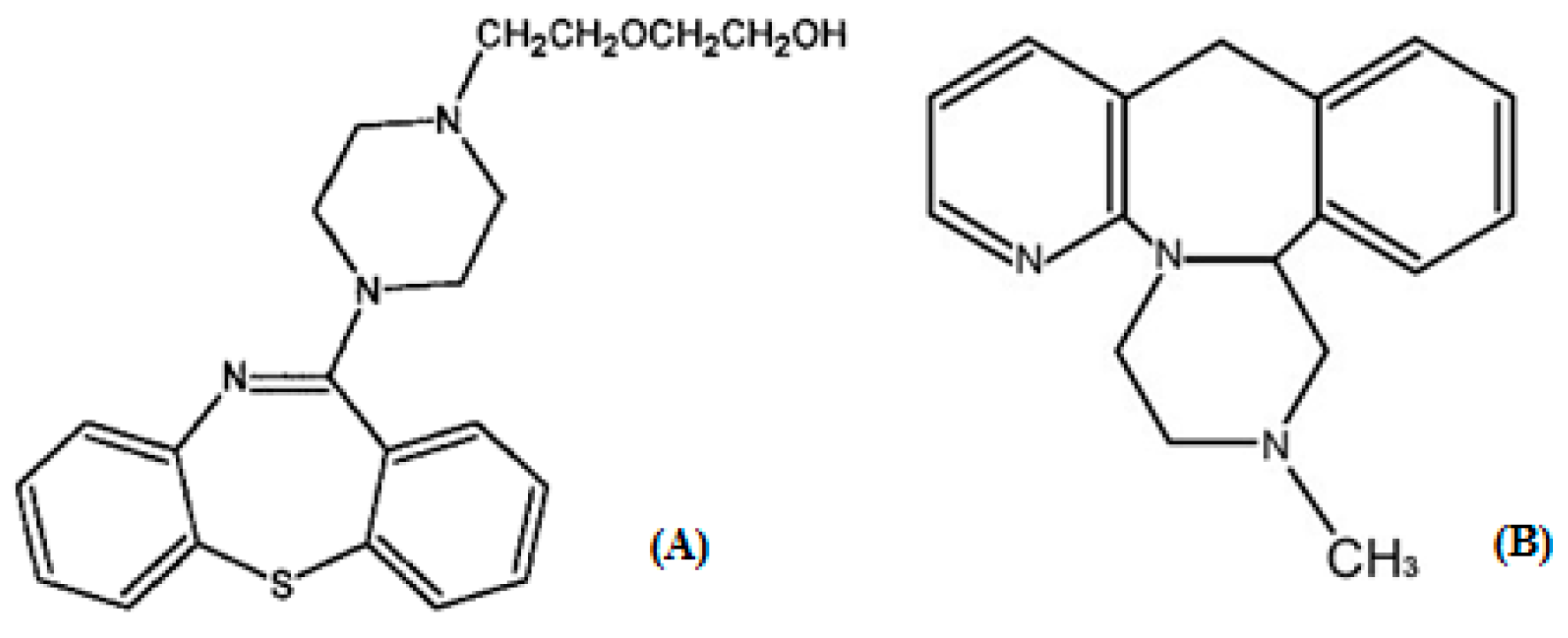
2. Results and Discussion
2.1. Method Development
2.1.1. Optimization of Buffer Specie and Buffer Concentration and pH and Voltage
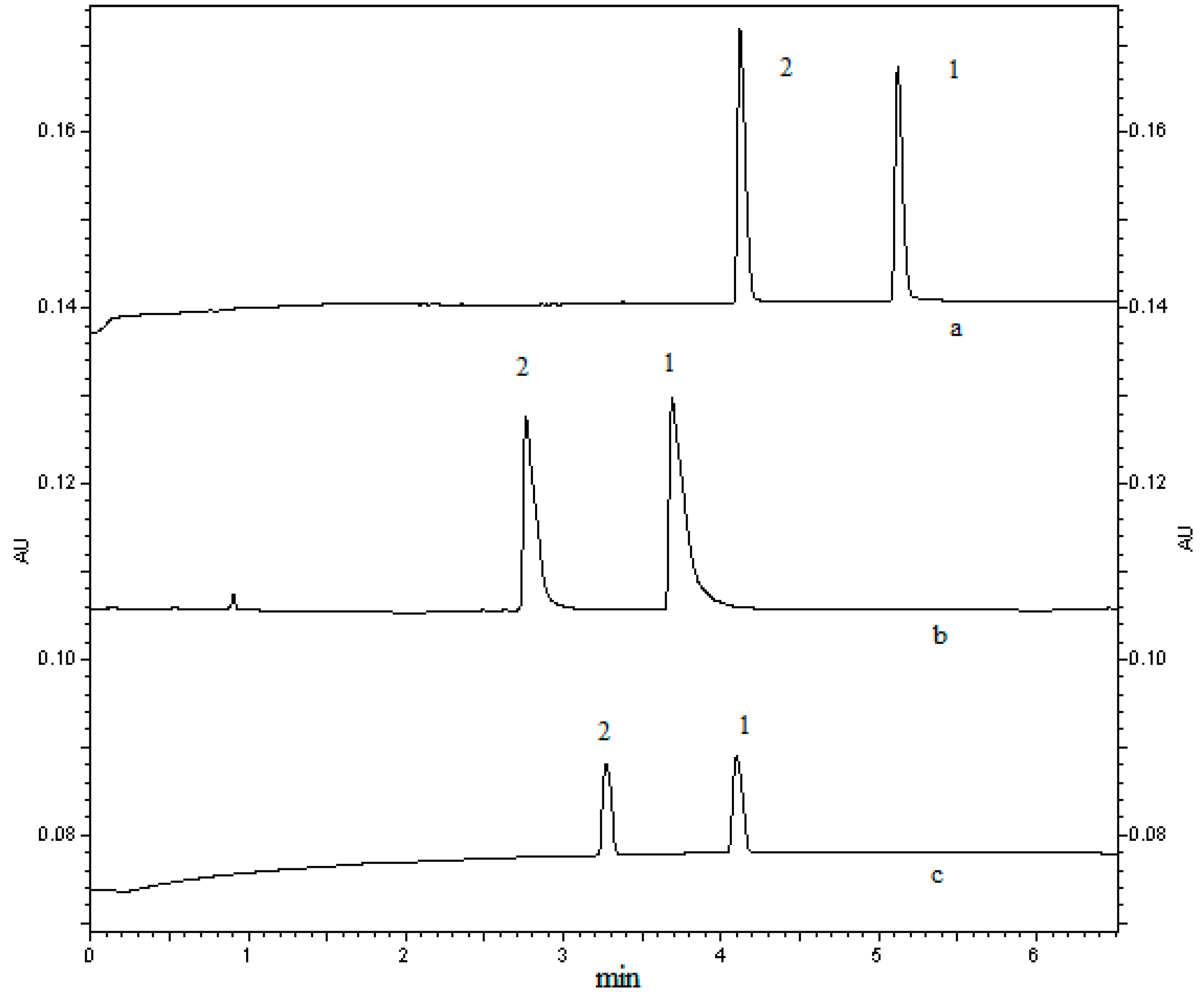
2.1.2. Selection of Injection Mode and Sample Solvent
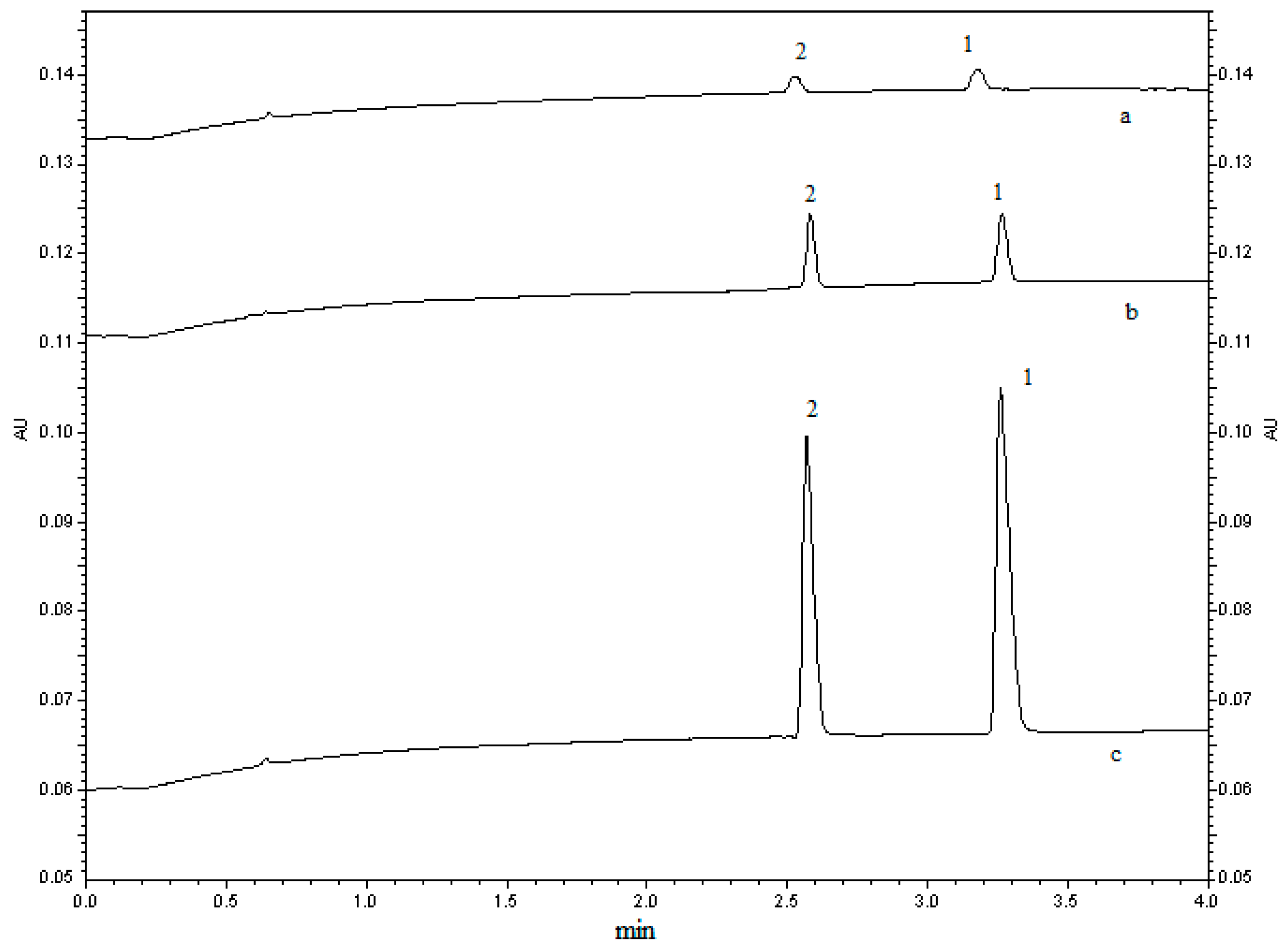
2.1.3. Pre-Treatment of Sample
2.2. Method Validation
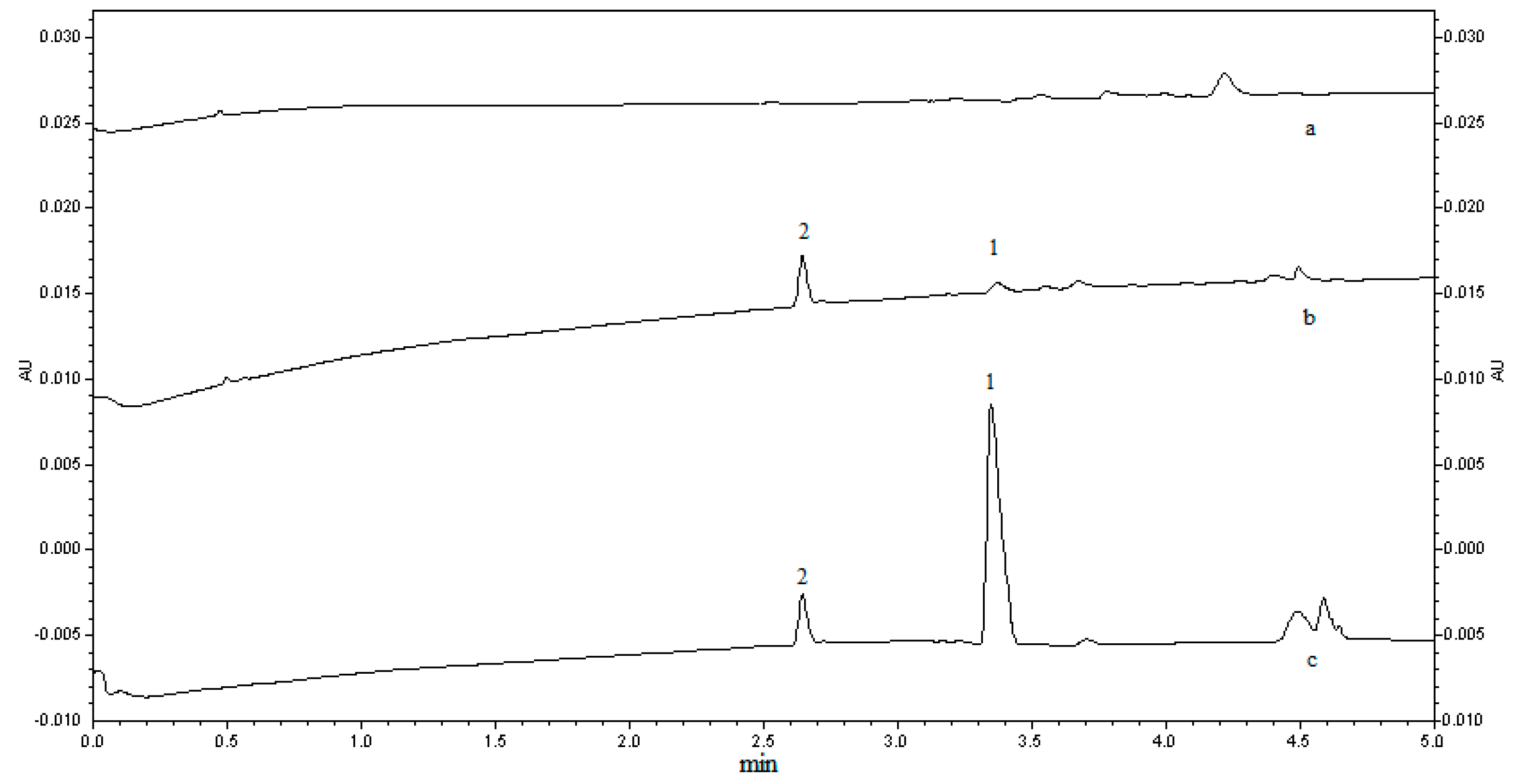
2.2.1. Specificity
2.2.2. Linearity of Calibration Curves and Lower Limit of Quantification (LLOQ)
2.2.3. Accuracy, Precision and Extraction Recovery
| Sample Level | Low QC | Medium QC | High QC |
|---|---|---|---|
| 2 ng/mL | 50 ng/mL | 800 ng/mL | |
| Within-run accuracy and precision | |||
| Validation run 1 | |||
| Mean ± SD (ng/mL) | 2.12 ± 0.13 | 49.63 ± 3.10 | 804.65 ± 43.08 |
| Accuracy ± SD (%) | 107.6 ± 6.7 | 101.0 ± 6.3 | 102.3 ± 5.5 |
| RSD (%) | 6.2 | 6.3 | 5.4 |
| N | 5 | 5 | 5 |
| Validation run 2 | |||
| Mean ± SD (ng/mL) | 1.94 ± 0.20 | 50.38 ± 4.55 | 812.36 ± 52.30 |
| Accuracy ± SD (%) | 98.2 ± 9.9 | 102.5 ± 9.3 | 103.3 ± 6.7 |
| RSD (%) | 10.1 | 9.0 | 6.4 |
| N | 5 | 5 | 5 |
| Validation run 3 | |||
| Mean ± SD (ng/mL) | 2.10 ± 0.15 | 51.09 ± 3.34 | 825.25 ± 39.85 |
| Accuracy ± SD (%) | 106.9 ± 7.5 | 104.0 ± 6.8 | 104.9 ± 5.1 |
| RSD (%) | 7.0 | 6.5 | 4.8 |
| N | 5 | 5 | 5 |
| Between-run accuracy and precision | |||
| Mean ± SD (ng/mL) | 2.05 ± 0.17 | 50.36 ± 3.50 | 814.09 ± 42.93 |
| Accuracy ± SD (%) | 104.2 ± 8.7 | 102.5 ± 7.1 | 103.5 ± 5.5 |
| RSD (%) | 8.4 | 6.9 | 5.3 |
| N | 15 | 15 | 15 |
| Compound | Concentration | Recovery (%) | RSD (%) |
|---|---|---|---|
| (ng/mL) | (mean ± SD) | ||
| Quetiapine (n = 5) | 2 | 79.5 ± 9.1 | 11.4 |
| 50 | 84.4 ± 7.3 | 8.6 | |
| 800 | 89.9 ± 4.9 | 5.5 | |
| IS (n = 15) | 100 | 85.6 ± 7.6 | 8.9 |
2.2.4. Stability
| Sample Condition | Nominal Concentration (ng/mL) | Measured Concentration (ng/mL) (Mean ± SD) | Accuracy (%) | RSD (%) |
|---|---|---|---|---|
| Freeze-thaw stability a | 2 | 2.04 ± 0.16 | 103.7 | 7.7 |
| 50 | 49.94 ± 1.25 | 101.6 | 2.5 | |
| 800 | 831.55 ± 21.89 | 105.7 | 2.6 | |
| 30-day stability b | 2 | 1.89 ± 0.17 | 96.3 | 8.9 |
| 50 | 51.26 ± 2.54 | 104.3 | 5.0 | |
| 800 | 825.86 ± 39.96 | 105.0 | 4.8 | |
| Bench top stability c | 2 | 1.85 ± 0.14 | 94.2 | 7.3 |
| 50 | 51.04 ± 2.68 | 103.8 | 5.3 | |
| 800 | 835.51 ± 29.61 | 106.3 | 3.5 | |
| Autosampler stability d | 2 | 1.93 ± 0.21 | 98.1 | 11.1 |
| 50 | 51.30 ± 2.65 | 104.4 | 5.2 | |
| 800 | 782.29 ± 35.95 | 99.5 | 4.6 |
2.3. Application to Preclinical Pharmacokinetic Study
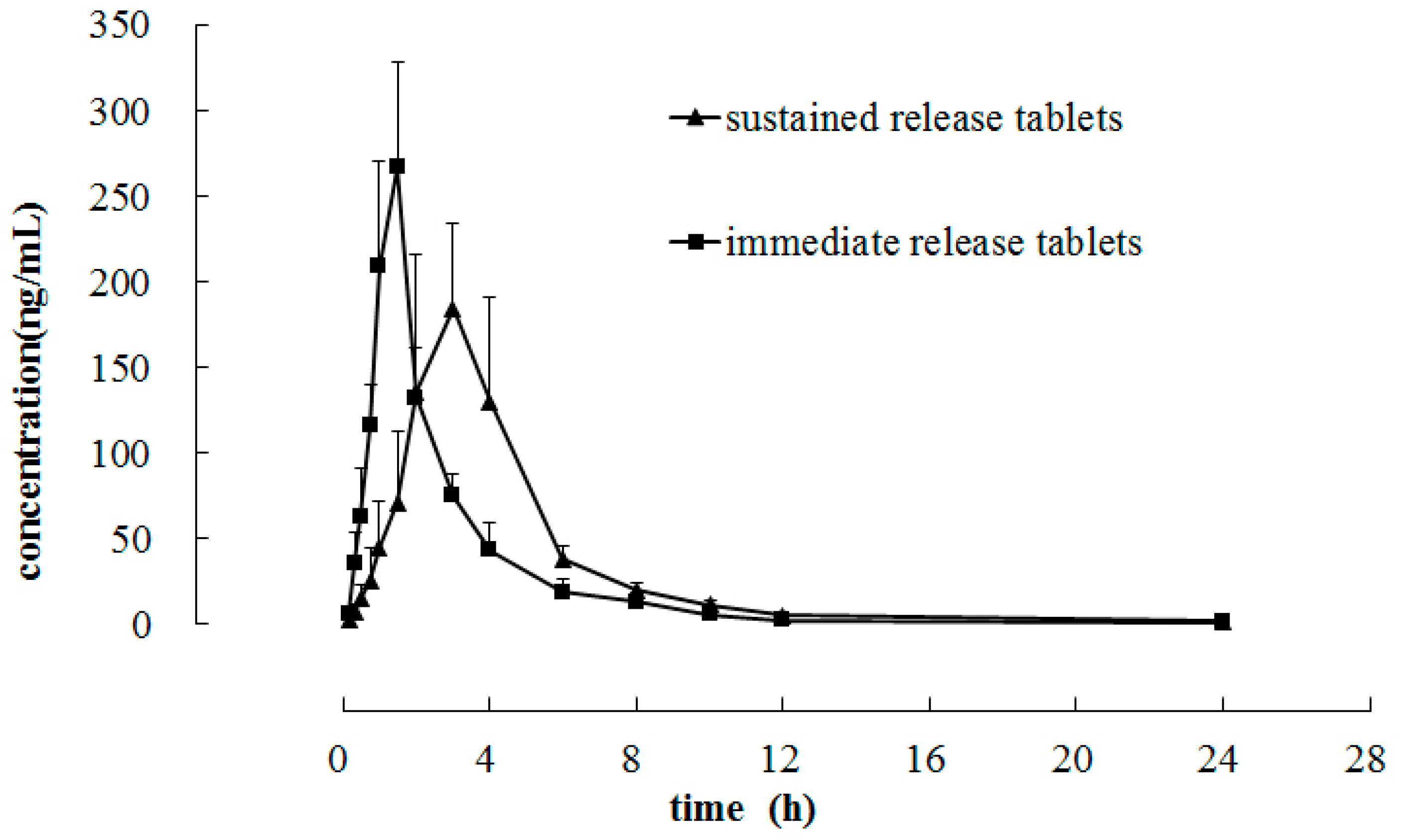
3. Experimental Section
3.1. Chemicals and Reagents
3.2. CE Instrumentation
3.3. Capillary Electrophoretic Conditions
3.4. Preparation of Stock Solutions, Calibration Samples and Quality Control Samples
3.5. Extraction Procedure
3.6. Pharmacokinetic Study in Beagle Dogs
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Heres, S.; Davis, J.; Maino, K.; Jetzinger, E.; Kissling, W.; Leucht, S. Why Olanzapine Beats Risperidone, Risperidone Beats Quetiapine, and Quetiapine Beats Olanzapine An Exploratory Analysis of Head-to-Head Comparison Studies of Second-Generation Antipsychotics. Am. J. Psychiatry 2006, 163, 185–194. [Google Scholar] [CrossRef] [PubMed]
- Dettlin, M.; Anghelescu, I.-G. Antipsychotic Drugs and Schizophrenia. N. Engl. J. Med. 2006, 354, 298–300. [Google Scholar]
- Arvanitis, L.A.; Miller, B.G. Multiple fixed doses of “Seroquel” (quetiapine) in patients with acute exacerbation of schizophrenia: A comparison with haloperidol and placebo. Soc. Biol. Psychiatry 1997, 42, 233–246. [Google Scholar] [CrossRef]
- Peuskens, J.; Link, C.G.G. A comparison of quetiapine and chlorpromazine in the treatment of schizophrenia. Acta Psychiatr. Scand. 1997, 96, 265–273. [Google Scholar] [CrossRef] [PubMed]
- DeVane, C.L.; Nemeroff, C.B. Clinical Pharmacokinetics of Quetiapine: An Atypical Antipsychotic. Clin. Pharmacokinet. 2001, 40, 509–522. [Google Scholar] [CrossRef] [PubMed]
- Jensen, N.H.; Rodriguiz, R.M.; Caron, M.G.; Wetsel, W.C.; Rothman, R.B.; Roth, B.L. N-Desalkylquetiapine, a Potent orepinephrine Reuptake Inhibitor and Partial 5-HT1A Agonist, as a Putative Mediator of Quetiapine’s Antidepressant Activity. Neuropsychopharmacology 2008, 33, 2303–2312. [Google Scholar] [CrossRef] [PubMed]
- Baune, B.T. New developments in the management of major depressive disorder and generalized anxiety disorder role of quetiapine. Neuropsychiatric Dis. Treat. 2008, 4, 1181–1192. [Google Scholar] [CrossRef]
- Wetzel, H.; Szegedi, A.; Hain, C.; Wiesner, J.; Schlegel, S.; Benkert, O. Seroquel (ICl 204 636), a putative atypical antipsychotic, in schizophrenia with positive symptomatology results of an open clinical trial and changes of neuroendocrinological and EEG parameters. Psychopharmacology 1995, 119, 231–238. [Google Scholar] [CrossRef] [PubMed]
- Prior, T.I.; Chue, P.S.; Tibbo, P.; Baker, G.B. Drug metabolism and atypical antipsychotics. Eur. Neuropsychopharmacol. 1999, 9, 301–309. [Google Scholar] [CrossRef]
- Sachse, J.; Köller, J.; artter, S.H.; Hiemke, C. Automated analysis of quetiapine and other antipsychotic drugs in human blood by high performance-liquid chromatography with column-switching and spectrophotometric detection. J. Chromatogr. B 2006, 830, 342–348. [Google Scholar] [CrossRef] [PubMed]
- Mercolini, L.; Grillo, M.; Bartoletti, C.; Boncompagni, G.; Raggi, M.A. Simultaneous analysis of classical neuroleptics, atypical antipsychotics and their metabolites in human plasma. Anal. Bioanal. Chem. 2007, 388, 235–243. [Google Scholar] [CrossRef] [PubMed]
- Saracino, M.A.; Mercolini, L.; Flotta, G.; Albers, L.J.; Merli, R.; Raggi, M.A. Simultaneous determination of fluvoxamine isomers and quetiapine in human plasma by means of high-performance liquid chromatography. J. Chromatogr. B 2006, 843, 227–233. [Google Scholar] [CrossRef] [PubMed]
- Trivedi, R.K.; Patel, M.C. Development and Validation of a Stability Indicating RP-UPLC Method for Determination of Quetiapine in Pharmaceutical Dosage Form. Sci. Pharm. 2011, 79, 97–111. [Google Scholar] [CrossRef] [PubMed]
- Davis, P.C.; Bravob, O.; Gehrke, M.; Azumaya, C.T. Development and validation of an LC-MS/MS method for the determination of quetiapine and four related metabolites in human plasma. J. Pharm. Biomed. Anal. 2010, 51, 1113–1119. [Google Scholar] [CrossRef] [PubMed]
- Fisher, D.S.; Partridge, S.J.; Handley, S.A.; Couchman, L.; Morgan, P.E.; Flanagan, R.J. LC-MS/MS of someatypicalantipsychotics inhumanplasma, serum, oralfluid and haemolysed whole blood. Forensic Sci. Int. 2013, 229, 145–150. [Google Scholar] [CrossRef] [PubMed]
- Barrett, B.; capek, M.H.; Huclováa, J.; Bořek-Dohalskýa, V.; Fejta, P.; Němeca, B.; Jelínekc, I. Validated HPLC-MS/MS method for determination of quetiapine in human plasma. J. Pharm. Biomed. Anal. 2007, 44, 498–505. [Google Scholar] [CrossRef] [PubMed]
- Pucci, V.; Mandrioli, R.; Ferranti, A.; Furlanetto, S.; Raggia, M.A. Quality control of commercial tablets containing the novel antipsychotic quetiapine. J. Pharm. Biomed. Anal. 2003, 32, 1037–1044. [Google Scholar] [CrossRef]
- Wang, Y.-R.; Yang, Y.-H.; Lu, C.-Y.; Lin, S.-J.; Chen, S.-H. Trace analysis of acetylcholinesterase inhibitors with antipsychotic drugs for Alzheimer’s disease by capillary electrophoresis with on column field-amplified sample injection. Anal. Bioanal. Chem. 2013, 405, 3233–3242. [Google Scholar] [CrossRef] [PubMed]
- Breadmore, M.C.; Tubaon, R.M.; Shallan, A.I.; Phung, S.C.; Keyon, A.S.A.; Gstoettenmayr, D.; Prapatpong, P.; Alhusban, A.A.; Ranjbar, L.; See, H.H.; et al. Recent advances in enhancing the sensitivity of electrophoresis and electrochromatography in capillaries and microchips (2012–2014). Electrophoresis 2015, 36, 36–61. [Google Scholar] [CrossRef] [PubMed]
- Simpson, S.L., Jr.; Quirino, J.P.; Terabe, S. On-line sample preconcentration in capillary electrophoresis Fundamentals and applications. J. Chromatogr. A 2008, 1184, 504–541. [Google Scholar] [CrossRef] [PubMed]
- Breadmore, M.C. Ionic liquid-based liquid phase microextraction with direct injection for capillary electrophoresis. J. Chromatogr. A 2011, 1218, 1347–1352. [Google Scholar] [CrossRef] [PubMed]
- Quirino, J.P.; Terabe, S. Approaching a Million-Fold Sensitivity Increase in Capillary Electrophoresis with Direct Ultraviolet Detection Cation-Selective Exhaustive Injection and Sweeping. Anal. Chem. 2000, 72, 1023–1030. [Google Scholar] [CrossRef] [PubMed]
- Krivácsy, Z.; Gelencsér, A.; Hlavay, J.; Kiss, G.; Sárvári, Z. Electrokinetic injection in capillary electrophoresis and its application to the analysis of inorganic compounds. J. Chromatogr. A 1999, 834, 21–44. [Google Scholar] [CrossRef]
- Quirino, J.P.; Terabe, S. Sample stacking of cationic and anionic analytes in capillary electrophoresis. J. Chromatogr. A 2000, 902, 119–135. [Google Scholar] [CrossRef]
- Li, J.; Bi, Y.; Wang, L.; Sun, F.; Chen, Z.; Xu, G.; Fan, G. β-Cyclodextrin enhanced on-line organic solvent field-amplified sample stacking in capillary zone electrophoresis for analysis of ambroxol in human plasma, following liquid-liquid extraction in the 96-well format. J. Pharm. Biomed. Anal. 2012, 66, 218–224. [Google Scholar] [CrossRef] [PubMed]
- US Food and Drug Administration (FDA). Bioanalytical Method Validation. Guidance for Industry, Center for Drug Evaluation and Research (CDER), Center for Veternary Medicine (CVM); FDA: Maryland, MD, USA, 2001.
- Sample Availability: Samples of Quetiapine Fumarate are available from the authors.
© 2016 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons by Attribution (CC-BY) license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cao, Y.; Wen, J.; Zhou, T.; Fan, G. On-Line Organic Solvent Field Enhanced Sample Injection in Capillary Zone Electrophoresis for Analysis of Quetiapine in Beagle Dog Plasma. Molecules 2016, 21, 121. https://doi.org/10.3390/molecules21010121
Cao Y, Wen J, Zhou T, Fan G. On-Line Organic Solvent Field Enhanced Sample Injection in Capillary Zone Electrophoresis for Analysis of Quetiapine in Beagle Dog Plasma. Molecules. 2016; 21(1):121. https://doi.org/10.3390/molecules21010121
Chicago/Turabian StyleCao, Yuqing, Jun Wen, Tingting Zhou, and Guorong Fan. 2016. "On-Line Organic Solvent Field Enhanced Sample Injection in Capillary Zone Electrophoresis for Analysis of Quetiapine in Beagle Dog Plasma" Molecules 21, no. 1: 121. https://doi.org/10.3390/molecules21010121
APA StyleCao, Y., Wen, J., Zhou, T., & Fan, G. (2016). On-Line Organic Solvent Field Enhanced Sample Injection in Capillary Zone Electrophoresis for Analysis of Quetiapine in Beagle Dog Plasma. Molecules, 21(1), 121. https://doi.org/10.3390/molecules21010121






