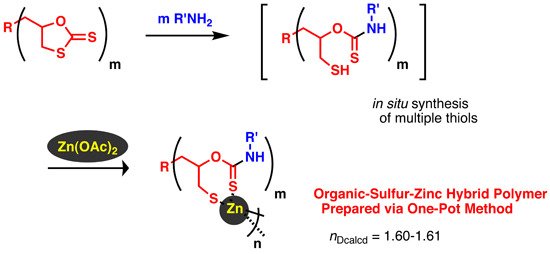
One-Pot Synthesis of Organic-Sulfur-Zinc Hybrid Materials via Polycondensation of a Zinc Salt and Thiols Generated in Situ from Cyclic Dithiocarbonates
Abstract
:1. Introduction
2. Results and Discussion
2.1. Polycondensation of Zn(OAc)2 with in Situ Generated Trithiol
| Run | Solvent | Temperature (°C) | R-NH2 (Equivalent to TDT) | Yield (%) a | Mn (Mw/Mn) b | Zn/S c |
|---|---|---|---|---|---|---|
| 1 | DMSO | 30 | 3.0 | 45 | 5800 (1.4) | 0.29 |
| 2 | THF | 30 | 3.0 | 31 | 2300 (1.4) | 0.31 |
| 3 | DMF | 30 | 3.0 | 42 | 2000 (1.4) | 0.15 |
| 4 | DMSO | 40 | 3.0 | 45 | 2300 (5.7) | 0.32 |
| 5 | DMSO | 60 | 3.0 | 47 | 4000 (39) | 0.35 |
| 6 | DMSO | 30 | 6.0 | 56 | 8400 (1.6) d | 0.34 |
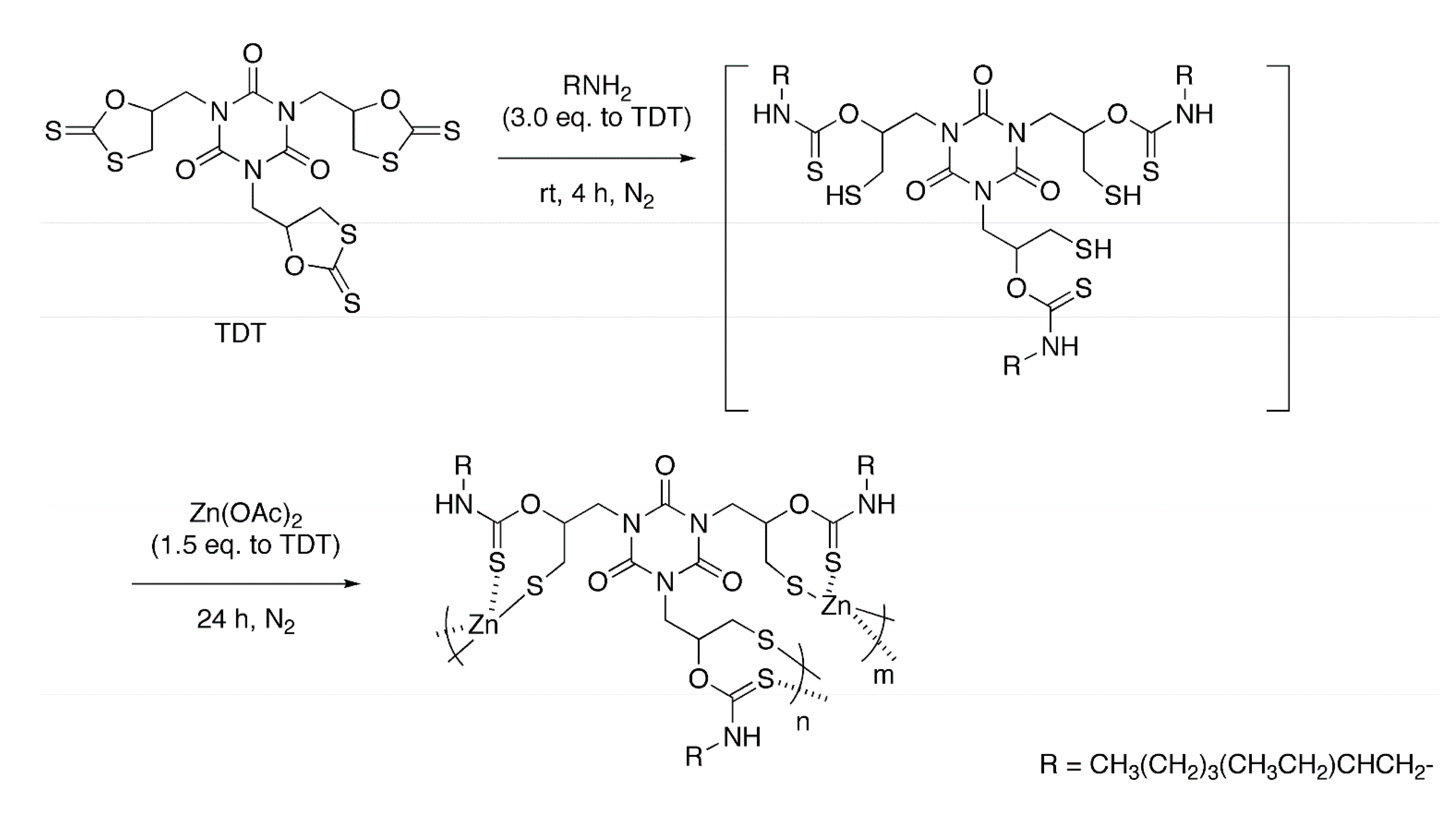
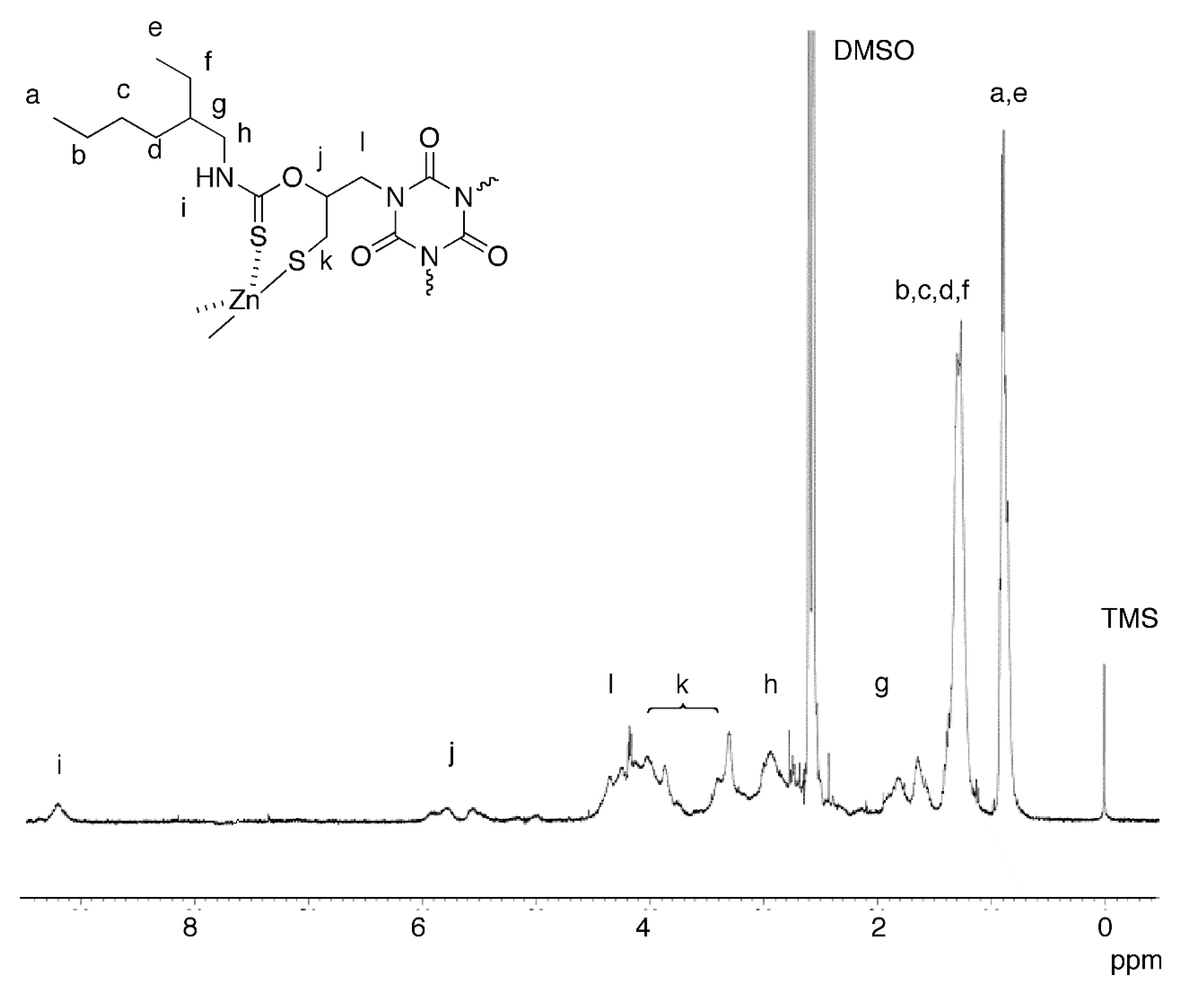
2.2. Polycondensation of Zn(OAc)2 with in Situ Generated Dithiol
| Run | Solvent | Temperature (°C) | R-NH2 (Equivalent to C=S) | Yield (%) a | Mn (Mw/Mn) b | Zn/S c |
|---|---|---|---|---|---|---|
| 1 | DMSO | 30 | 3.0 | 59 | 6600 (1.3) | 0.53 |
| 2 | THF | 30 | 3.0 | 39 | Insoluble | 0.48 |
| 3 | DMF | 30 | 3.0 | 58 | 1300 (1.4) | 0.40 |
| 4 | DMSO | 60 | 3.0 | 28 | 3200 (1.7) | 0.44 |
| 5 | DMSO | 30 | 6.0 | 63 | Insoluble | 0.39 |
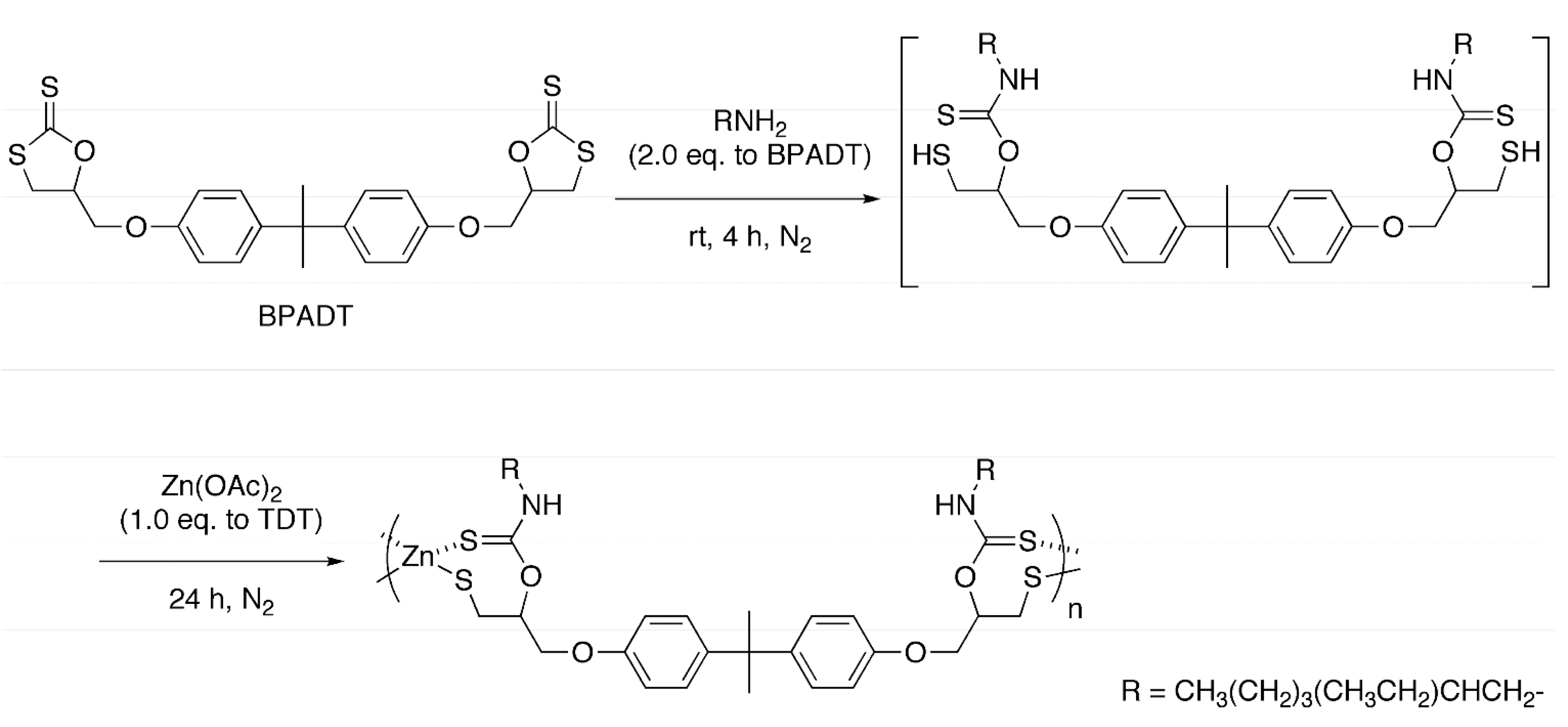
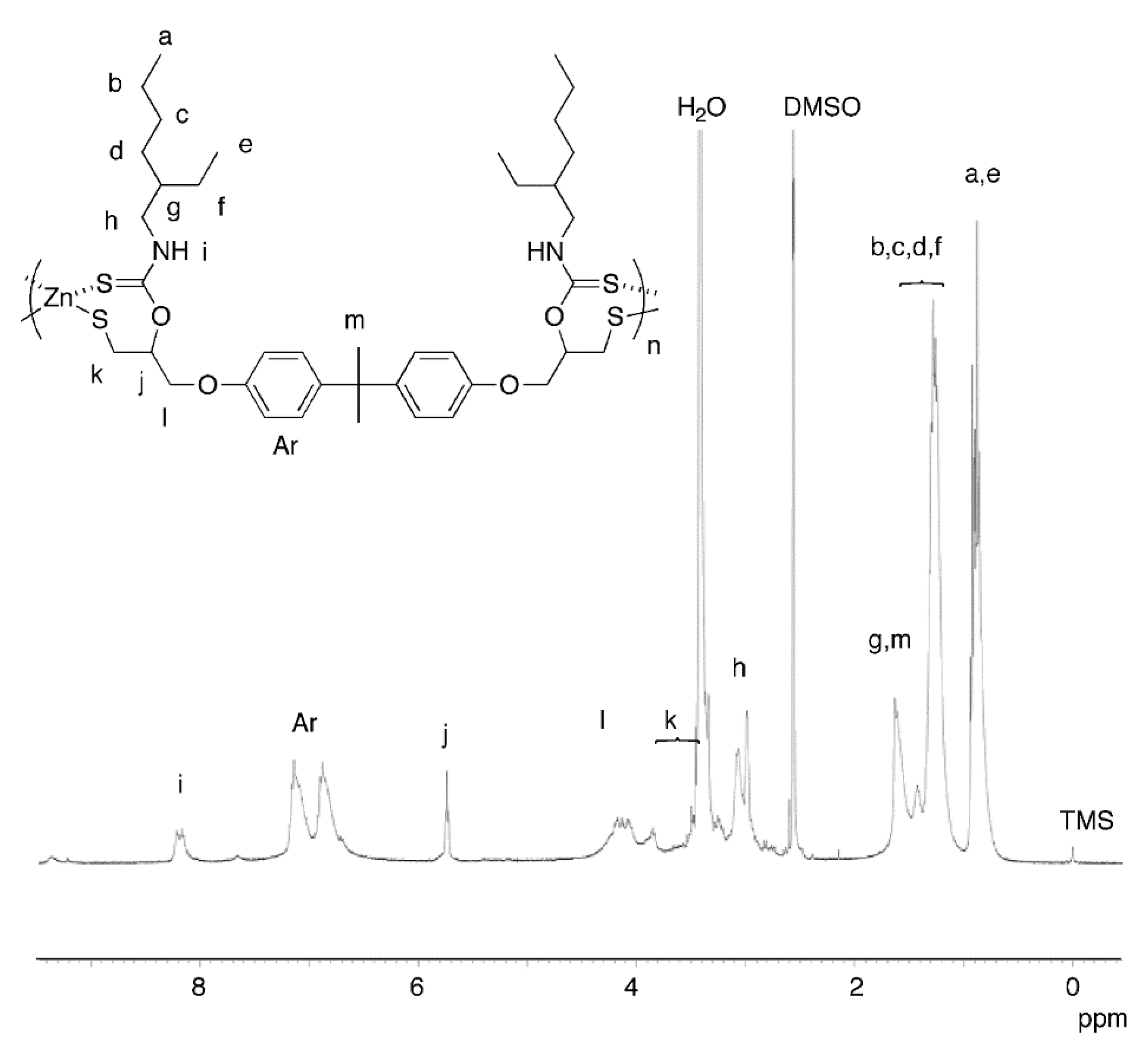
2.3. Properties of Organic-Sulfur-Zinc Hybrid Materials Obtained by the One-Pot Method
2.3.1. Films with PMMA and PS

| BZn | BZn10/PS90 | BZn10/PMMA90 | TZn | TZn10/PS90 | TZn10/PMMA90 | PS | PMMA | |
|---|---|---|---|---|---|---|---|---|
| Td5 (°C) a | 192 | 272 | 161 | 170 | 113 | 139 | 380 | 281 |
| Tg (°C) b | 40 | 86 | – c | – c | – c | – c | 100 | 105 |
2.3.2. Refractive Properties of Hybrid Polymers
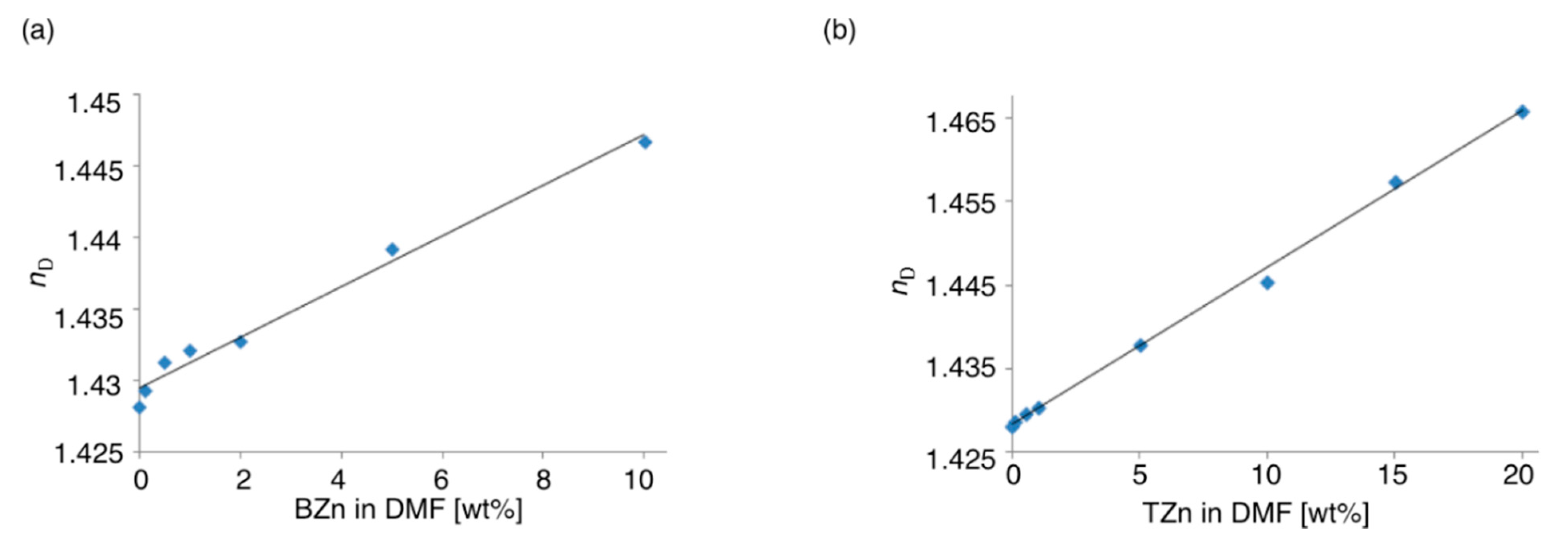
3. Experimental Section
3.1. Materials
3.2. Measurements
3.3. One-Pot Synthesis of Organic-Sulfur-Zinc Hybrid via Sequential Ring-Opening Addition of EHA and TDT and Polycondensation of the in Situ Generated Thiol with Zn(OAc)2 (Typical Procedure)
3.4. One-Pot Synthesis of Organic-Sulfur-Zinc Hybrid via Sequential Ring-Opening Addition of EHA and BPADT and Polycondensation of the in Situ Generated Thiol with Zn(OAc)2 (Typical Procedure)
3.5. Preparation of Composite Films (Typical Procedure)
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Takebe, H.; Kitagawa, R.; Hewak, D.W. Non-toxic sulfide glasses and thin films for optical applications. J. Ceram. Soc. Jpn. 2005, 113, 37–43. [Google Scholar] [CrossRef]
- Kricheldorf, H.R.; Schwarz, G. Poly(thioester)s. J. Macromol. Sci. Part A Pure Appl. Chem. 2007, 44, 625–649. [Google Scholar] [CrossRef]
- Jha, G.S.; Seshadri, G.; Mohan, A.; Khandal, R.K. Sulfur containing optical plastics and its ophthalmic lenses applications. e-Polymers 2008, 035, 1–27. [Google Scholar] [CrossRef]
- Houberttz, R.; Fröhlich, L.; Popall, M.; Streppel, U.; Dannberg, P.; Bräuer, A.; Serbin, J.; Chichkov, B.N. Inorganic–organic hybrid polymers for information technology: From planar technology to 3D nanostructures. Adv. Eng. Mater. 2003, 5, 551–555. [Google Scholar] [CrossRef]
- Althues, H.; Henle, J.; Kaskel, S. Functional inorganic nanofillers for transparent polymers. Chem. Soc. Rev. 2007, 36, 1454–1465. [Google Scholar] [CrossRef] [PubMed]
- Lebeau, B.; Innocenzi, P. Hybrid materials for optics and photonics. Chem Soc. Rev. 2007, 40, 886–906. [Google Scholar] [CrossRef] [PubMed]
- Lu, C.; Yang, B. High refractive index organic-inorganic nanocomposites: Design, synthesis, and application. J. Mater. Chem. 2009, 19, 2884–2901. [Google Scholar] [CrossRef]
- Jeon, I.Y.; Baek, J.B. Nanocomposites derived from polymers and inorganic nanoparticles. Materials 2010, 3, 3654–3674. [Google Scholar] [CrossRef]
- Macdonald, E.K.; Shaver, M.P. Intrinsic high refractive index polymers. Polym. Int. 2015, 64, 6–14. [Google Scholar] [CrossRef]
- Bhagat, S.D.; Chatterjee, J.; Chen, B.; Stiegman, A.E. High refractive index polymers based on thiol-ene cross-linking using polarizable inorganic/organic monomers. Macromolecules 2012, 45, 1174–1181. [Google Scholar] [CrossRef]
- Ochiai, B.; Konta, H. Organic-sulfur-zinc hybrid nanoparticle for optical applications synthesized via polycondensation of trithiol and Zn(OAc)2. Nanoscale Res. Lett. 2013. [Google Scholar] [CrossRef] [PubMed]
- Ochiai, B.; Kato, Y.; Endo, T. One-pot synthesis of graft copolymer by combination of free radical polymerization and polyaddition. Macromolecules 2009, 42, 8001–8002. [Google Scholar] [CrossRef]
- Ochiai, B.; Ogihara, T.; Mashiko, M.; Endo, T. synthesis of rare-metal absorbing polymer by three-component polyaddition through combination of chemo-selective nucleophilic and radical additions. J. Am. Chem. Soc. 2009, 131, 1636–1637. [Google Scholar] [CrossRef] [PubMed]
- Ochiai, B.; Endo, T. Carbon dioxide and carbon disulfide as resources for functional polymers. Prog. Polym. Sci. 2005, 30, 183–215. [Google Scholar] [CrossRef]
- Kihara, N.; Tochigi, H.; Endo, T. Synthesis and reaction of polymers bearing 5-membered cyclic dithiocarbonate group. J. Polym. Sci. Part A Polym. Chem. 1995, 33, 1005–1010. [Google Scholar] [CrossRef]
- Moriguchi, T.; Endo, T. Polyaddition of bifunctional dithiocarbonates derived from epoxides and carbon-disulfide—Synthesis of novel poly(thiourethanes). Macromolecules 1995, 28, 5386–5387. [Google Scholar] [CrossRef]
- Choi, W.; Nakajima, M.; Sanda, F.; Endo, T. Novel one-pot polymer syntheses based on dithiols obtained from a five-membered cyclic dithiocarbonate and diamines. Macromol. Chem. Phys. 1998, 199, 1909–1915. [Google Scholar] [CrossRef]
- Suzuki, A.; Nagai, D.; Ochiai, B.; Endo, T. Facile synthesis and crosslinking reaction of trifunctional five-membered cyclic carbonate and dithiocarbonate. J. Polym. Sci. Part A Polym. Chem. 2004, 42, 5983–5989. [Google Scholar] [CrossRef]
- Hirata, M.; Watanabe, T.; Ochiai, B.; Endo, T. Synthesis of graft terpolymers by addition reaction of amino-terminated polyether to poly(methacrylate)s bearing five-membered cyclic dithiocarbonate moieties and application of the graft terpolymers as modifiers for wool. J. Polym. Sci. Part A Polym. Chem. 2012, 50, 3259–3268. [Google Scholar] [CrossRef]
- Nagai, D.; Sato, M.; Ochiai, B.; Endo, T. Synthesis and properties of the poly(thiourethane)s obtained by cationic ring-opening polymerization of cyclic thiourethanes. J. Polym. Sci. Part A Polym. Chem. 2006, 44, 4795–4803. [Google Scholar] [CrossRef]
- Tamaki, R.; Samura, K.; Chujo, Y. Synthesis of polystyrene and silica gel polymer hybrids via π-π interactions. Chem. Commun. 1998, 1131–1132. [Google Scholar] [CrossRef]
- An, Y.C.; Jang, N.Y.; Lee, S.W.; Lim, J.H.; Kim, K.M. Organic-inorganic polymer hybrids of polystyrene with ladderlike polyphenylsilsesquioxanes. Polym. J. 2008, 40, 538–542. [Google Scholar] [CrossRef]
- Sample Availability: Samples of the compounds BZn and TZn are available from the authors.
© 2015 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ochiai, B.; Konta, H. One-Pot Synthesis of Organic-Sulfur-Zinc Hybrid Materials via Polycondensation of a Zinc Salt and Thiols Generated in Situ from Cyclic Dithiocarbonates. Molecules 2015, 20, 15049-15059. https://doi.org/10.3390/molecules200815049
Ochiai B, Konta H. One-Pot Synthesis of Organic-Sulfur-Zinc Hybrid Materials via Polycondensation of a Zinc Salt and Thiols Generated in Situ from Cyclic Dithiocarbonates. Molecules. 2015; 20(8):15049-15059. https://doi.org/10.3390/molecules200815049
Chicago/Turabian StyleOchiai, Bungo, and Hirohisa Konta. 2015. "One-Pot Synthesis of Organic-Sulfur-Zinc Hybrid Materials via Polycondensation of a Zinc Salt and Thiols Generated in Situ from Cyclic Dithiocarbonates" Molecules 20, no. 8: 15049-15059. https://doi.org/10.3390/molecules200815049
APA StyleOchiai, B., & Konta, H. (2015). One-Pot Synthesis of Organic-Sulfur-Zinc Hybrid Materials via Polycondensation of a Zinc Salt and Thiols Generated in Situ from Cyclic Dithiocarbonates. Molecules, 20(8), 15049-15059. https://doi.org/10.3390/molecules200815049