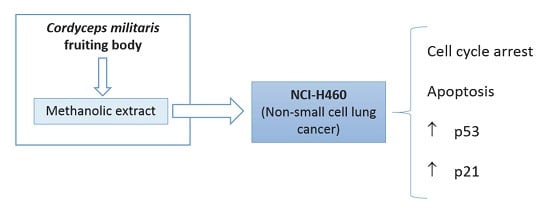
Cordyceps militaris (L.) Link Fruiting Body Reduces the Growth of a Non-Small Cell Lung Cancer Cell Line by Increasing Cellular Levels of p53 and p21
Abstract
:1. Introduction
2. Results and Discussion
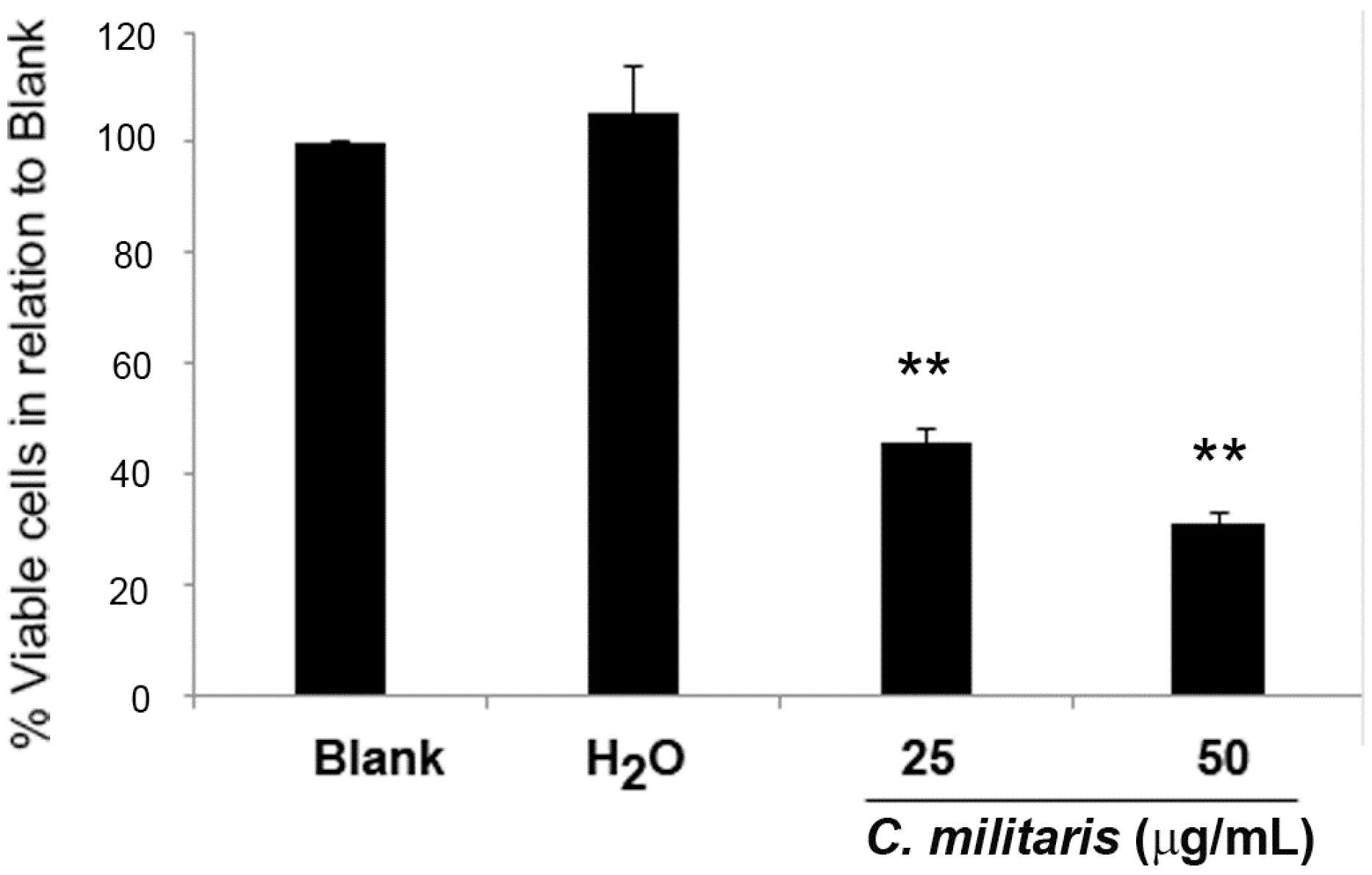
2.1. Effect of C. militaris Methanolic Extract in NCI-H460 Cell Viability
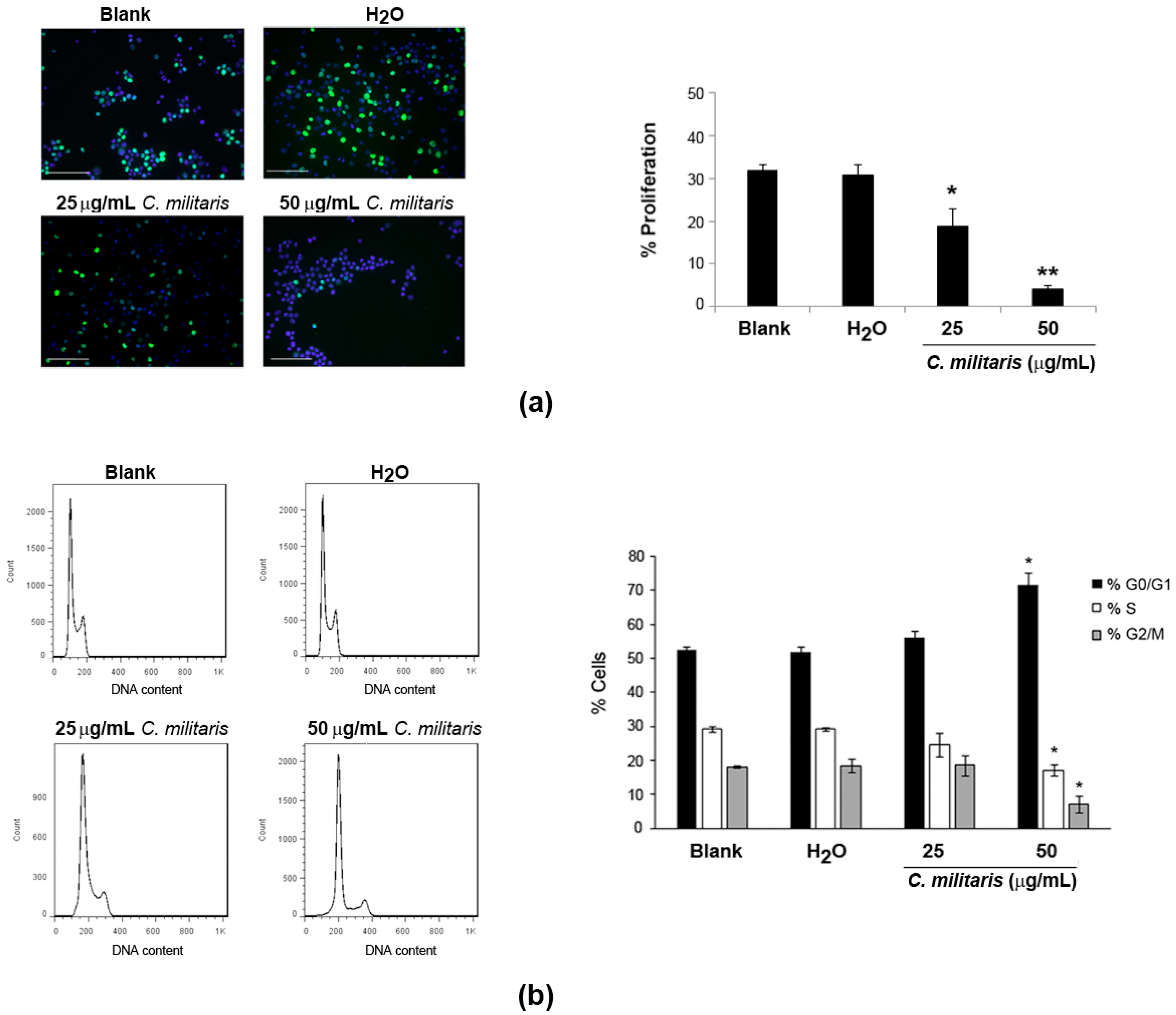
2.2. Effect of the Extract on NCI-H460 Cellular Proliferation and Cycle Profile
2.3. Effect of the Extract on NCI-H460 Cellular Apoptosis
| % Apoptosis | |||
|---|---|---|---|
| Early | Late | ||
| Blank | 5.9% ± 1.2% | 1.5% ± 0.8% | |
| H2O | 6.3% ± 2.0% | 2.2% ± 0.5% | |
| C. militaris | 25 µg/mL | 13.7% ± 1.4% * | 3.4% ± 0.3% |
| 50 µg/mL | 21.4% ± 4.2% * | 5.0% ± 5.3% | |
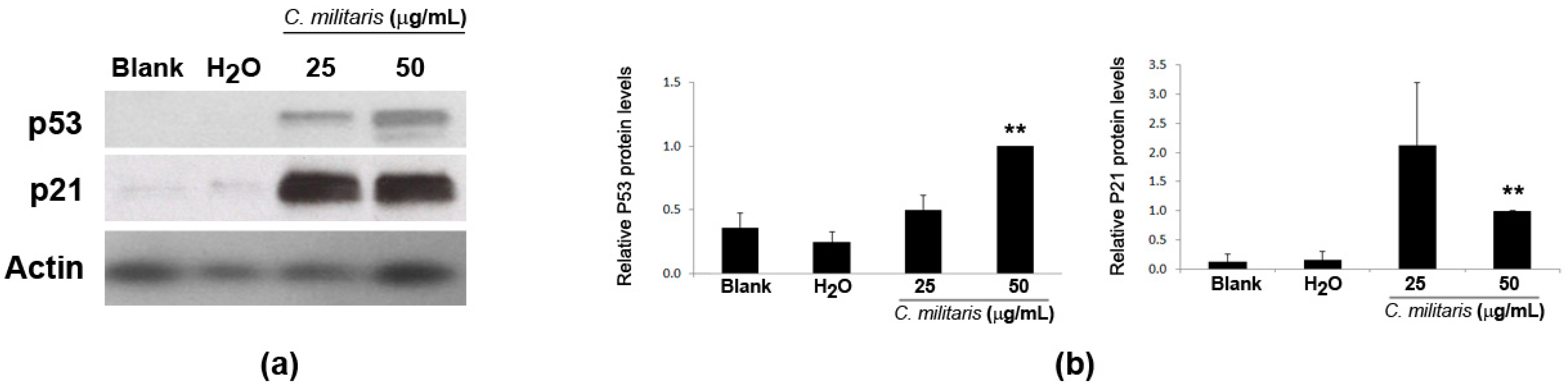
2.4. Effect of the Extract in p53 and p21 Protein Levels in NCI-H460 Cells
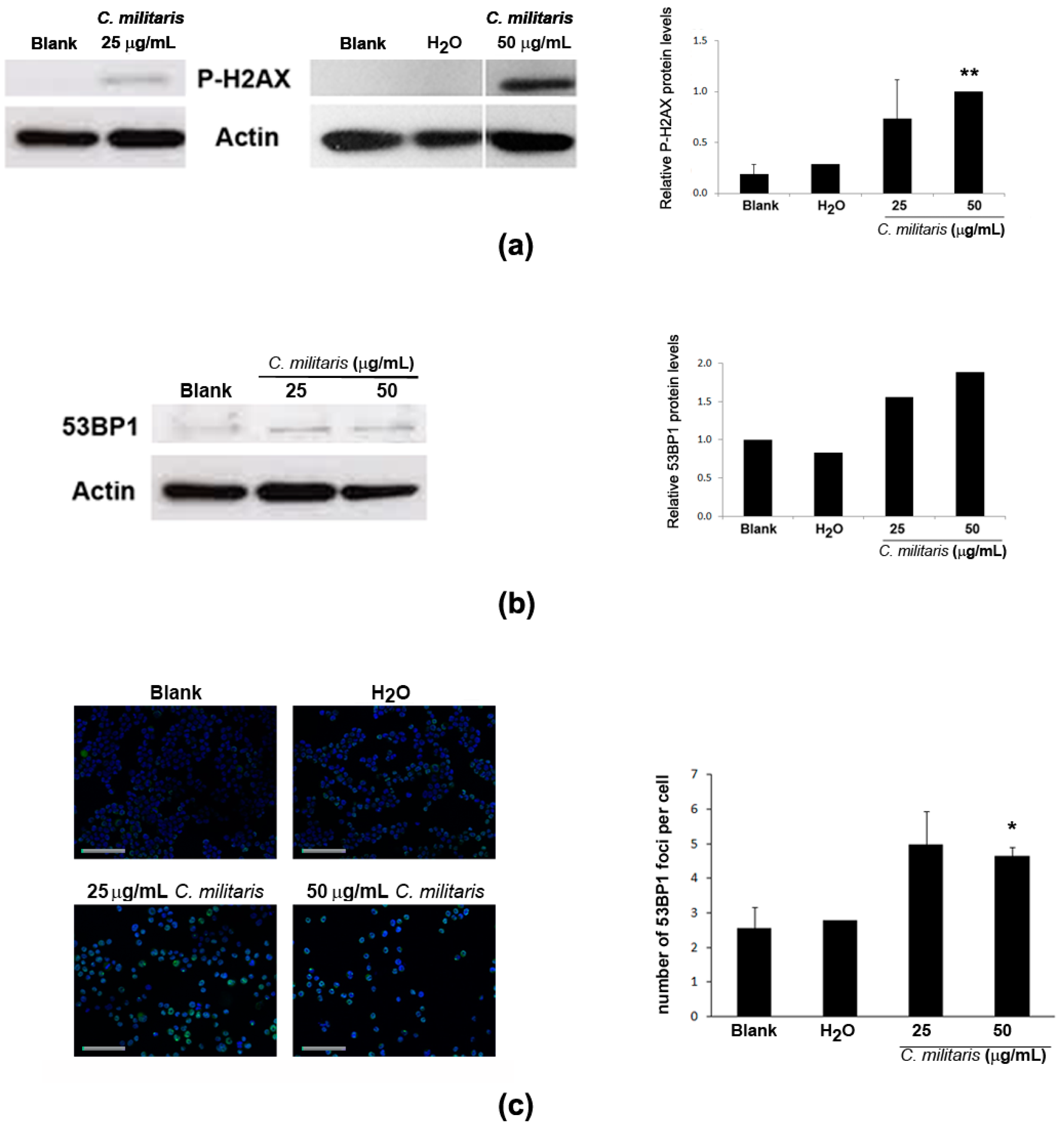
2.5. Effect of the Extract in DNA Damage of NCI-H460 Cells
3. Experimental Section
3.1. Extract
3.2. Cell Culture
3.3. Cell Treatments with C. militaris Extract
3.3.1. Analysis of Cell Cycle Profile by Flow Cytometry
3.3.2. Analysis of Cellular Proliferation with the BrdU Incorporation Assay
3.3.3. Analysis of Apoptosis by Flow Cytometry
3.3.4. Analysis of Protein Expression by Western Blot
3.3.5. Analysis of 53BP1 Expression by Immunofluorescence
3.4. Statistical Analysis
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Das, S.K.; Masuda, M.; Sakurai, A.; Sakakibara, M. Medicinal uses of the mushroom Cordyceps militaris: Current state and prospects. Fitoterapia 2010, 81, 961–968. [Google Scholar] [CrossRef] [PubMed]
- Park, N.S.; Lee, K.S.; Sohn, H.D.; Kim, D.H.; Lee, S.M.; Park, E.; Kim, I.; Je, Y.H.; Jin, B.R. Molecular cloning, expression, and characterization of the Cu, Zn superoxide dismutase (SOD1) gene from the entomopathogenic fungus Cordyceps militaris. Mycologia 2005, 97, 130–138. [Google Scholar] [CrossRef] [PubMed]
- Park, S.E.; Kim, J.; Lee, Y.W.; Yoo, H.S.; Cho, C.K. Antitumor activity of water extracts from Cordyceps militaris in NCI-H460 cell xenografted nude mice. J. Acupunct. Meridian Stud. 2009, 2, 294–300. [Google Scholar] [CrossRef]
- Yoo, H.S.; Shin, J.W.; Cho, J.H.; Son, C.G.; Lee, Y.W.; Park, S.Y.; Cho, C.K. Effects of Cordyceps militaris extract on angiogenesis and tumor growth. Acta Pharmacol. Sin. 2004, 25, 657–665. [Google Scholar] [PubMed]
- Yue, K.; Ye, M.; Zhou, Z.; Sun, W.; Lin, X. The genus Cordyceps: A chemical and pharmacological review. J. Pharm. Pharmacol. 2013, 65, 474–493. [Google Scholar] [CrossRef] [PubMed]
- Park, C.; Hong, S.H.; Lee, J.Y.; Kim, G.Y.; Choi, B.T.; Lee, Y.T.; Park, D.I.; Park, Y.M.; Jeong, Y.K.; Choi, Y.H. Growth inhibition of U937 leukemia cells by aqueous extract of Cordyceps militaris through induction of apoptosis. Oncol. Rep. 2005, 13, 1211–1216. [Google Scholar] [CrossRef] [PubMed]
- Lim, H.; Kwon, Y.; Cho, S.; Kim, J.; Yoon, G.; Lee, S.; Kim, H. Antitumor activity of Cordyceps militaris on human cancer cell line. Korean J. Pharmacogn. 2004, 35, 364–367. [Google Scholar]
- Jin, C.Y.; Kim, G.Y.; Choi, Y.H. Induction of apoptosis by aqueous extract of Cordyceps militaris through activation of caspases and inactivation of Akt in human breast cancer MDA-MB-231 Cells. J. Microbiol. Biotechnol. 2008, 18, 1997–2003. [Google Scholar] [PubMed]
- Lee, H.; Kim, Y.J.; Kim, H.W.; Lee, D.H.; Sung, M.K.; Park, T. Induction of apoptosis by Cordyceps militaris through activation of caspase-3 in leukemia HL-60 cells. Biol. Pharm. Bull. 2006, 29, 670–674. [Google Scholar] [CrossRef] [PubMed]
- Baik, J.S.; Kwon, H.Y.; Kim, K.S.; Jeong, Y.K.; Cho, Y.S.; Lee, Y.C. Cordycepin induces apoptosis in human neuroblastoma SK-N-BE(2)-C and melanoma SK-MEL-2 cells. Indian J. Biochem. Biophys. 2012, 49, 86–91. [Google Scholar] [PubMed]
- Tuli, H.S.; Sharma, A.K.; Sandhu, S.S.; Kashyap, D. Cordycepin: A bioactive metabolite with therapeutic potential. Life Sci. 2013, 93, 863–839. [Google Scholar] [CrossRef] [PubMed]
- Shrestha, B.; Zhang, W.; Zhang, Y.; Liu, X. The medicinal fungus Cordyceps militaris: Research and development. Mycol. Prog. 2012, 11, 599–614. [Google Scholar] [CrossRef]
- Rao, Y.K.; Fang, S.H.; Tzeng, Y.M. Evaluation of the anti-inflammatory and anti-proliferation tumoral cells activities of Antrodia camphorata, Cordyceps sinensis, and Cinnamomum osmophloeum bark extracts. J. Ethnopharmacol. 2007, 114, 78–85. [Google Scholar] [CrossRef] [PubMed]
- Huang, H.; Wang, H.; Luo, R.C. Inhibitory effects of cordyceps extract on growth of colon cancer cells. Zhong Yao Cai J. Chin. Med. Mater. 2007, 30, 310–313. [Google Scholar]
- Liu, X.; Huang, K.; Zhou, J. Composition and Antitumor Activity of the Mycelia and Fruiting Bodies of Cordyceps militaris. J. Food Nutr. Res. 2014, 2, 74–79. [Google Scholar] [CrossRef]
- Reis, F.S.; Barros, L.; Calhelha, R.C.; Ciric, A.; van Griensven, L.J.; Sokovic, M.; Ferreira, I.C. The methanolic extract of Cordyceps militaris (L.) Link fruiting body shows antioxidant, antibacterial, antifungal and antihuman tumor cell lines properties. Food Chem.Toxicol. Int. J. Publ. Br. Ind. Biol. Res. Assoc. 2013, 62, 91–98. [Google Scholar] [CrossRef] [PubMed]
- Jayat, C.; Ratinaud, M.H. Cell cycle analysis by flow cytometry: Principles and applications. Biol. Cell Auspices Eur. Cell Biol. Organ. 1993, 78, 15–25. [Google Scholar] [CrossRef]
- Yang, C.H.; Kao, Y.H.; Huang, K.S.; Wang, C.Y.; Lin, L.W. Cordyceps militaris and mycelial fermentation induced apoptosis and autophagy of human glioblastoma cells. Cell Death Dis. 2012, 3. [Google Scholar] [CrossRef] [PubMed]
- He, W.; Zhang, M.F.; Ye, J.; Jiang, T.T.; Fang, X.; Song, Y. Cordycepin induces apoptosis by enhancing JNK and p38 kinase activity and increasing the protein expression of Bcl-2 pro-apoptotic molecules. J. Zhejiang Univ. Sci. B 2010, 11, 654–660. [Google Scholar] [CrossRef] [PubMed]
- Park, S.E.; Yoo, H.S.; Jin, C.Y.; Hong, S.H.; Lee, Y.W.; Kim, B.W.; Lee, S.H.; Kim, W.J.; Cho, C.K.; Choi, Y.H. Induction of apoptosis and inhibition of telomerase activity in human lung carcinoma cells by the water extract of Cordyceps militaris. Food Chem. Toxicol. Int. J. Publ. Br. Ind. Biol. Res. Assoc. 2009, 47, 1667–1675. [Google Scholar] [CrossRef] [PubMed]
- Jeong, J.W.; Jin, C.Y.; Park, C.; Hong, S.H.; Kim, G.Y.; Jeong, Y.K.; Lee, J.D.; Yoo, Y.H.; Choi, Y.H. Induction of apoptosis by cordycepin via reactive oxygen species generation in human leukemia cells. Toxicol. Int. J. Publ. Assoc. BIBRA 2011, 25, 817–824. [Google Scholar] [CrossRef] [PubMed]
- Lakin, N.D.; Jackson, S.P. Regulation of p53 in response to DNA damage. Oncogene 1999, 18, 7644–7655. [Google Scholar] [CrossRef] [PubMed]
- Abukhdeir, A.M.; Park, B.H. p21 and p27: Roles in carcinogenesis and drug resistance. Expert Rev. Mol. Med. 2008, 10. [Google Scholar] [CrossRef] [PubMed]
- Amundson, S.A.; Myers, T.G.; Fornace, A.J., Jr. Roles for p53 in growth arrest and apoptosis: Putting on the brakes after genotoxic stress. Oncogene 1998, 17, 3287–3299. [Google Scholar] [CrossRef] [PubMed]
- Soussi, T.; Dehouche, K.; Beroud, C. p53 website and analysis of p53 gene mutations in human cancer: Forging a link between epidemiology and carcinogenesis. Hum. Mutat. 2000, 15, 105–113. [Google Scholar] [CrossRef]
- Vassilev, L.T. p53 Activation by small molecules: Application in oncology. J. Med. Chem. 2005, 48, 4491–4499. [Google Scholar] [CrossRef] [PubMed]
- Athar, M.; Elmets, C.A.; Kopelovich, L. Pharmacological activation of p53 in cancer cells. Curr. Pharm. Des. 2011, 17, 631–639. [Google Scholar] [CrossRef] [PubMed]
- Kuribayashi, K.; El-Deiry, W.S. Regulation of programmed cell death by the p53 pathway. Adv. Exp. Med. Biol. 2008, 615, 201–221. [Google Scholar] [PubMed]
- Mah, L.J.; El-Osta, A.; Karagiannis, T.C. gammaH2AX: A sensitive molecular marker of DNA damage and repair. Leukemia 2010, 24, 679–686. [Google Scholar] [CrossRef] [PubMed]
- Wang, B.; Matsuoka, S.; Carpenter, P.B.; Elledge, S.J. 53BP1, a mediator of the DNA damage checkpoint. Science 2002, 298, 1435–1438. [Google Scholar] [CrossRef] [PubMed]
- Mallette, F.A.; Richard, S. K48-linked ubiquitination and protein degradation regulate 53BP1 recruitment at DNA damage sites. Cell Res. 2012, 22, 1221–1223. [Google Scholar] [CrossRef] [PubMed]
- Fadlalla, K.; Watson, A.; Yehualaeshet, T.; Turner, T.; Samuel, T. Ruta graveolens extract induces DNA damage pathways and blocks Akt activation to inhibit cancer cell proliferation and survival. Anticancer Res. 2011, 31, 233–241. [Google Scholar] [PubMed]
- Abreu, R.M.; Ferreira, I.C.; Calhelha, R.C.; Lima, R.T.; Vasconcelos, M.H.; Adega, F.; Chaves, R.; Queiroz, M.J. Anti-hepatocellular carcinoma activity using human HepG2 cells and hepatotoxicity of 6-substituted methyl 3-aminothieno[3,2-b]pyridine-2-carboxylate derivatives: In vitro evaluation, cell cycle analysis and QSAR studies. Eur. J. Med. Chem. 2011, 46, 5800–5806. [Google Scholar] [CrossRef] [PubMed]
- Preto, A.; Goncalves, J.; Rebocho, A.P.; Figueiredo, J.; Meireles, A.M.; Rocha, A.S.; Vasconcelos, H.M.; Seca, H.; Seruca, R.; Soares, P.; et al. Proliferation and survival molecules implicated in the inhibition of BRAF pathway in thyroid cancer cells harbouring different genetic mutations. BMC Cancer 2009, 9, 387. [Google Scholar] [CrossRef] [PubMed]
- Queiroz, M.J.; Peixoto, D.; Calhelha, R.C.; Soares, P.; dos Santos, T.; Lima, R.T.; Campos, J.F.; Abreu, R.M.; Ferreira, I.C.; Vasconcelos, M.H. New di(hetero)arylethers and di(hetero)arylamines in the thieno[3,2-b]pyridine series: Synthesis, growth inhibitory activity on human tumor cell lines and non-tumor cells, effects on cell cycle and on programmed cell death. Eur. J. Med. Chem. 2013, 69, 855–862. [Google Scholar] [CrossRef] [PubMed]
- Oliveira, M.; Reis, F.S.; Sousa, D.; Tavares, C.; Lima, R.T.; Ferreira, I.C.; Santos, T.D.; Vasconcelos, M.H. A methanolic extract of Ganoderma lucidum fruiting body inhibits the growth of a gastric cancer cell line and affects cellular autophagy and cell cycle. Food Funct. 2014, 5, 1389–1394. [Google Scholar] [CrossRef] [PubMed]
- Lima, R.T.; Martins, L.M.; Guimaraes, J.E.; Sambade, C.; Vasconcelos, M.H. Chemosensitization effects of XIAP downregulation in K562 leukemia cells. J. Chemother. 2006, 18, 98–102. [Google Scholar] [CrossRef] [PubMed]
- Markova, E.; Schultz, N.; Belyaev, I.Y. Kinetics and dose-response of residual 53BP1/gamma-H2AX foci: Co-localization, relationship with DSB repair and clonogenic survival. Int. J. Radiat. Biol. 2007, 83, 319–329. [Google Scholar] [CrossRef] [PubMed]
- Seca, H.; Lima, R.T.; Almeida, G.M.; Sobrinho-Simoes, M.; Bergantim, R.; Guimaraes, J.E.; Vasconcelos, M.H. Effect of MiR-128 in DNA Damage of HL-60 Acute Myeloid Leukemia Cells. Curr. Pharm. Biotechnol. 2014, 15, 492–502. [Google Scholar] [CrossRef] [PubMed]
- Aramwit, P.; Bang, N.; Ratanavaraporn, J.; Nakpheng, T.; Srichana, T. An Anti-Cancer Cordycepin Produced by Cordyceps militaris Growing on the Dead Larva of Bombyx mori Silkworm. J. Agric. Sci. 2014, 6, 41–54. [Google Scholar] [CrossRef]
- Vaz, J.A.; Almeida, G.M.; Ferreira, D.F.; Ferreira, I.C.F.R.; Martins, A.; Vasconcelos, M.H. Clitocybe alexandri extract induces cell cycle arrest and apoptosis in a lung cancer cell line: Identification of phenolic acids with cytotoxic potential. Food Chem. 2012, 132, 482–486. [Google Scholar] [CrossRef]
- Sample Availability: Samples of the studied mushrooms are available from the authors.
© 2015 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bizarro, A.; Ferreira, I.C.F.R.; Soković, M.; Van Griensven, L.J.L.D.; Sousa, D.; Vasconcelos, M.H.; Lima, R.T. Cordyceps militaris (L.) Link Fruiting Body Reduces the Growth of a Non-Small Cell Lung Cancer Cell Line by Increasing Cellular Levels of p53 and p21. Molecules 2015, 20, 13927-13940. https://doi.org/10.3390/molecules200813927
Bizarro A, Ferreira ICFR, Soković M, Van Griensven LJLD, Sousa D, Vasconcelos MH, Lima RT. Cordyceps militaris (L.) Link Fruiting Body Reduces the Growth of a Non-Small Cell Lung Cancer Cell Line by Increasing Cellular Levels of p53 and p21. Molecules. 2015; 20(8):13927-13940. https://doi.org/10.3390/molecules200813927
Chicago/Turabian StyleBizarro, Ana, Isabel C. F. R. Ferreira, Marina Soković, Leo J. L. D. Van Griensven, Diana Sousa, M. Helena Vasconcelos, and Raquel T. Lima. 2015. "Cordyceps militaris (L.) Link Fruiting Body Reduces the Growth of a Non-Small Cell Lung Cancer Cell Line by Increasing Cellular Levels of p53 and p21" Molecules 20, no. 8: 13927-13940. https://doi.org/10.3390/molecules200813927
APA StyleBizarro, A., Ferreira, I. C. F. R., Soković, M., Van Griensven, L. J. L. D., Sousa, D., Vasconcelos, M. H., & Lima, R. T. (2015). Cordyceps militaris (L.) Link Fruiting Body Reduces the Growth of a Non-Small Cell Lung Cancer Cell Line by Increasing Cellular Levels of p53 and p21. Molecules, 20(8), 13927-13940. https://doi.org/10.3390/molecules200813927