Caesalpinia decapetala Extracts as Inhibitors of Lipid Oxidation in Beef Patties
Abstract
:1. Introduction
2. Results and Discussion
2.1. Antioxidant Capacity Assays (AOC)
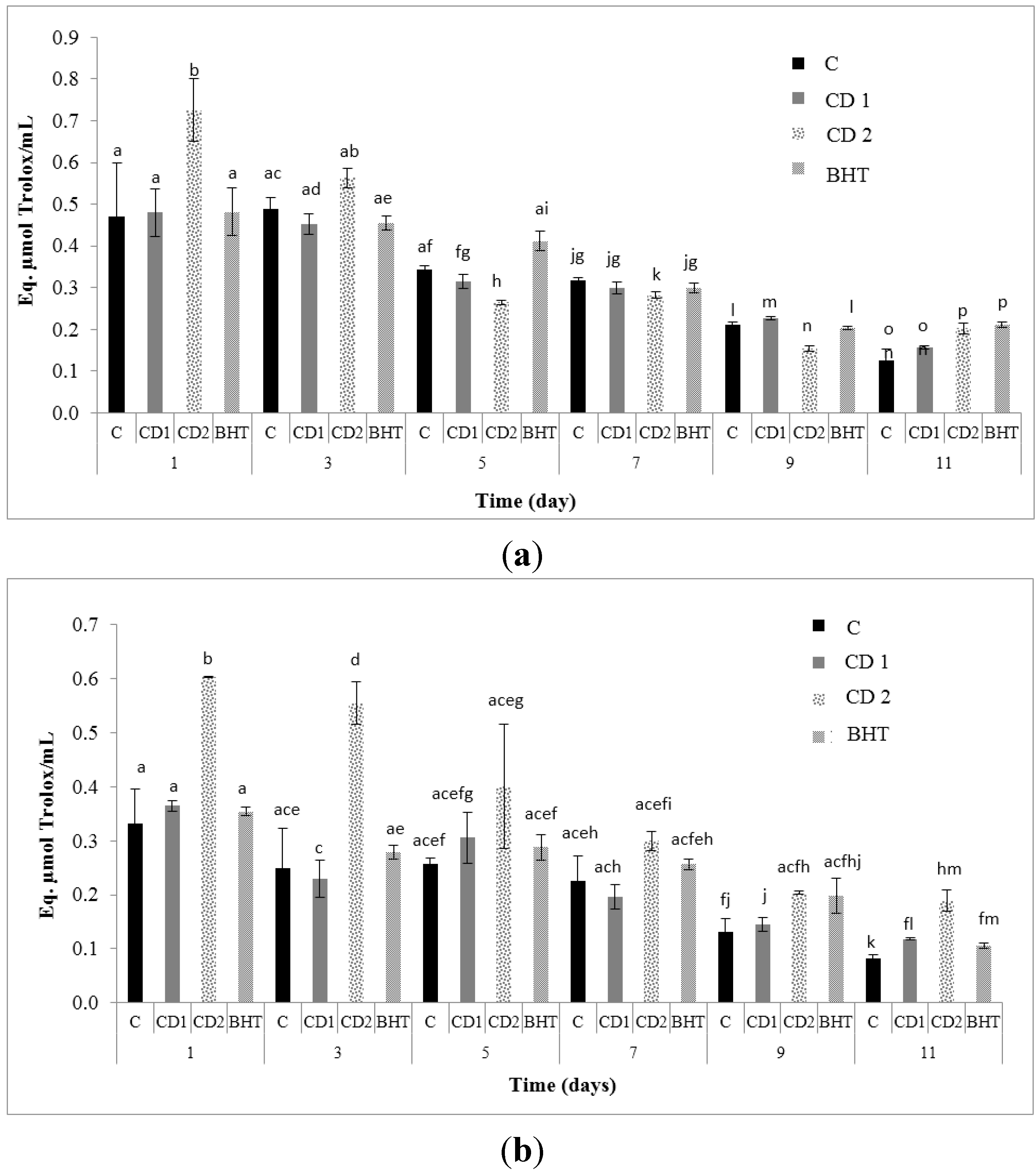
| Sample | Antioxidant Activities (µmol Trolox/mL Sample) | ||
|---|---|---|---|
| Hydrophylic | Lipophilic | Total | |
| Control | 0.13 (0.03) a | 0.08 (0.01) a | 0.21 (0.19) a |
| CD1 | 0.16 (0.003) b | 0.12 (0.002) b | 0.28 (0.07) b |
| CD2 | 0.20 (0.01) c | 0.19 (0.02) c | 0.39 (0.03) c |
| BHT | 0.21 (0.01) c | 0.11(0.004) b | 0.32 (0.01) d |
2.2. Effects on Metmyoglobin Formation
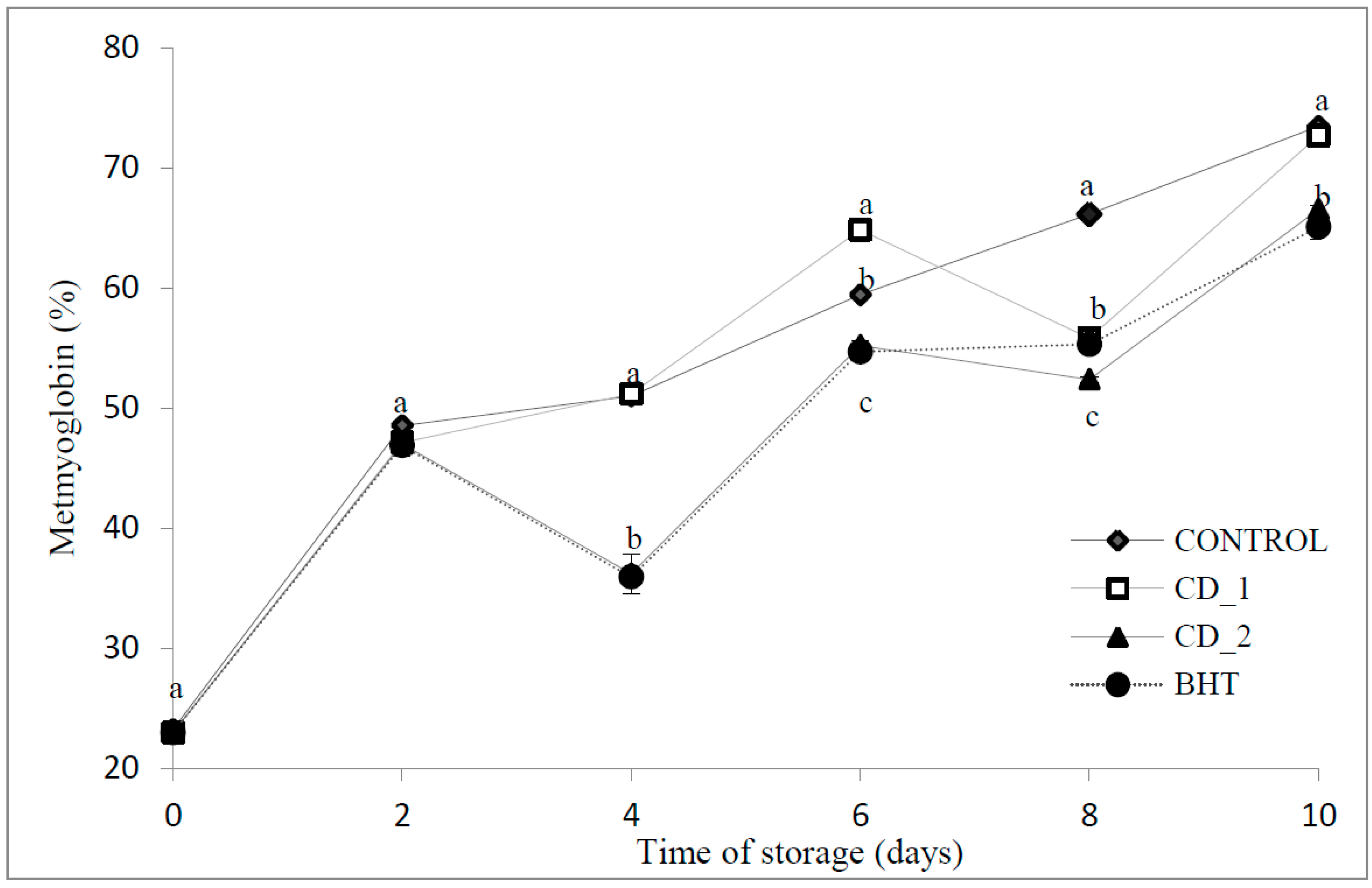
2.3. Volatile Compounds
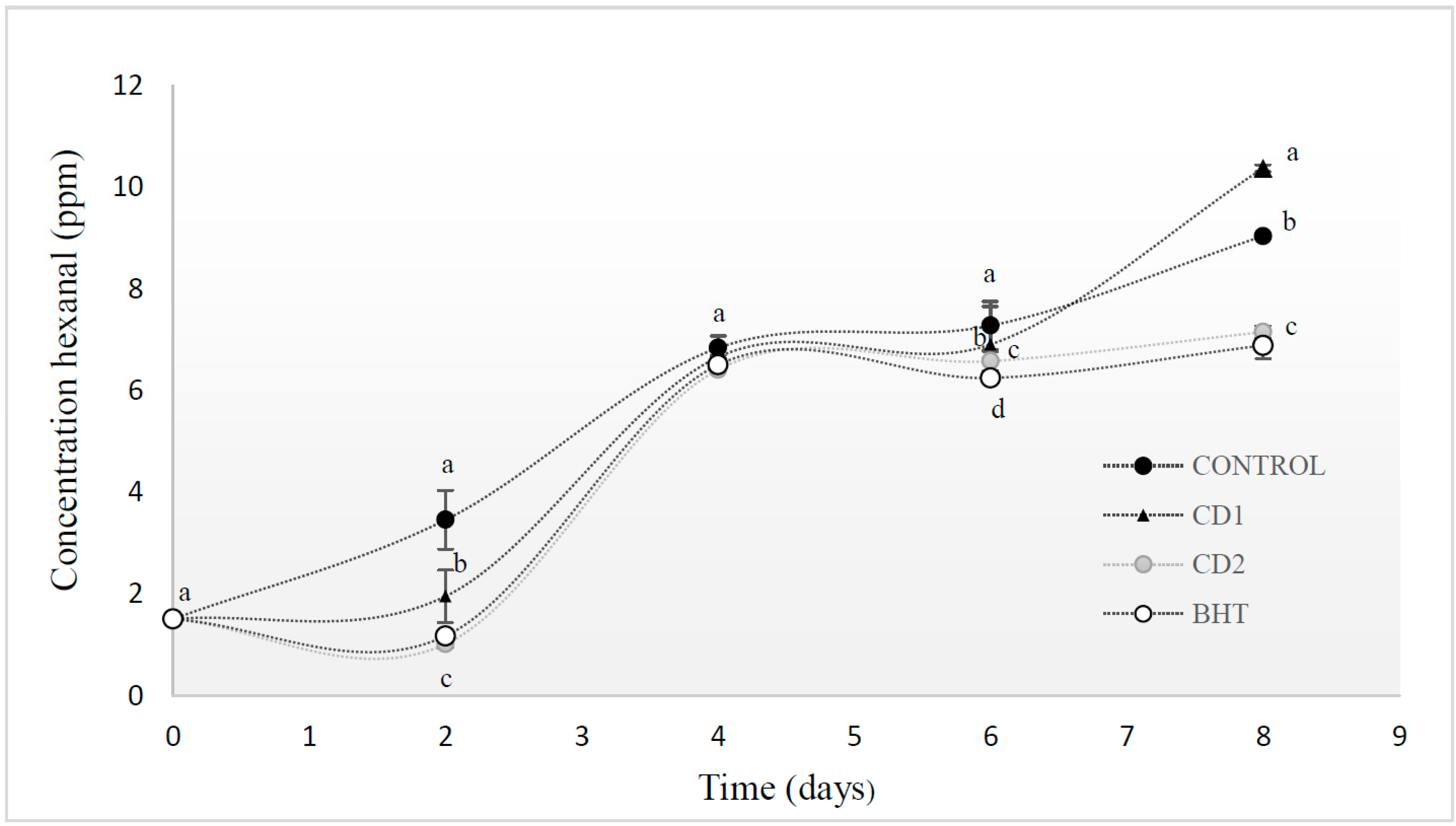
2.4. Effect on Lipid Oxidation and the Color of Beef Patties
2.4.1. Thiobarbituric Acid Reactive Substance (TBARS) Value
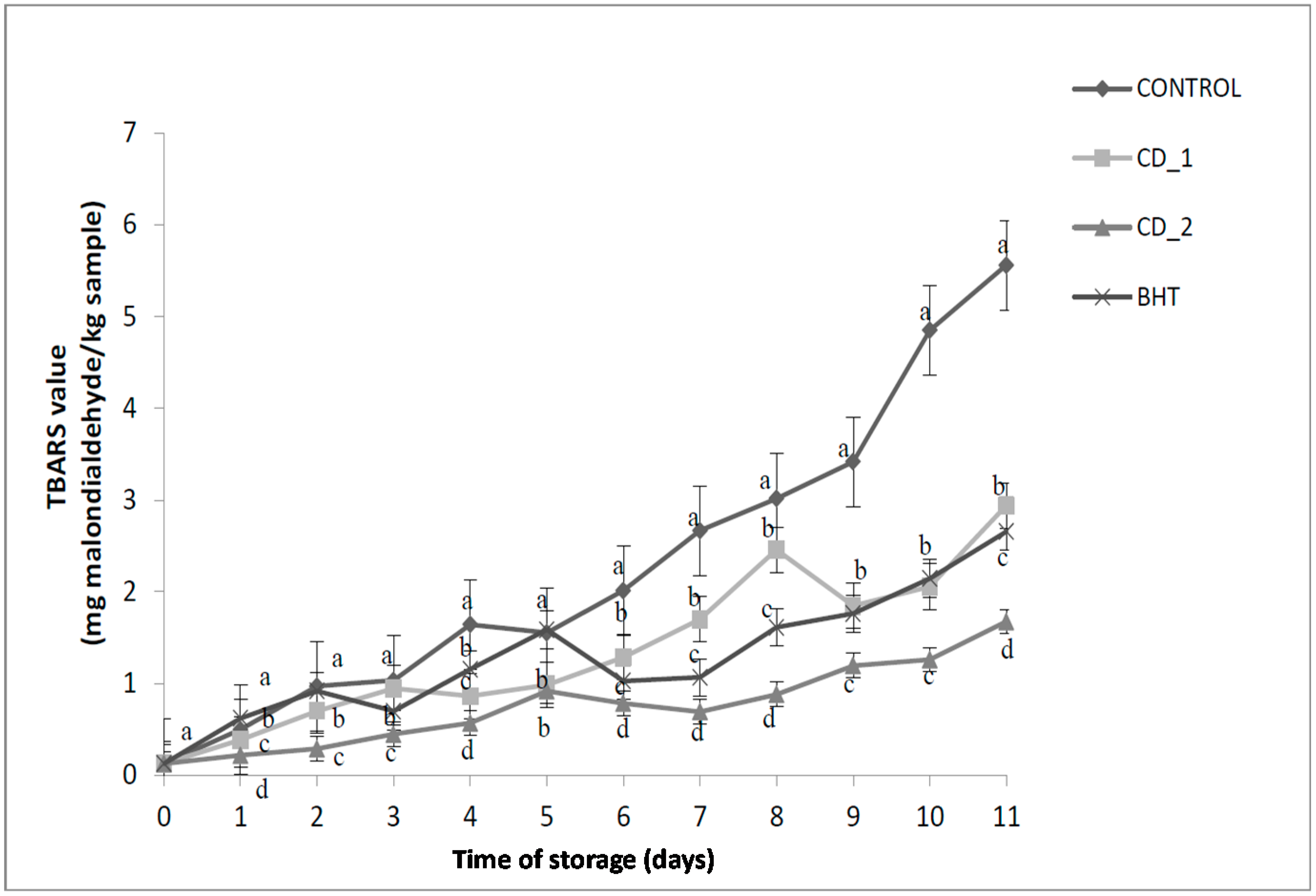
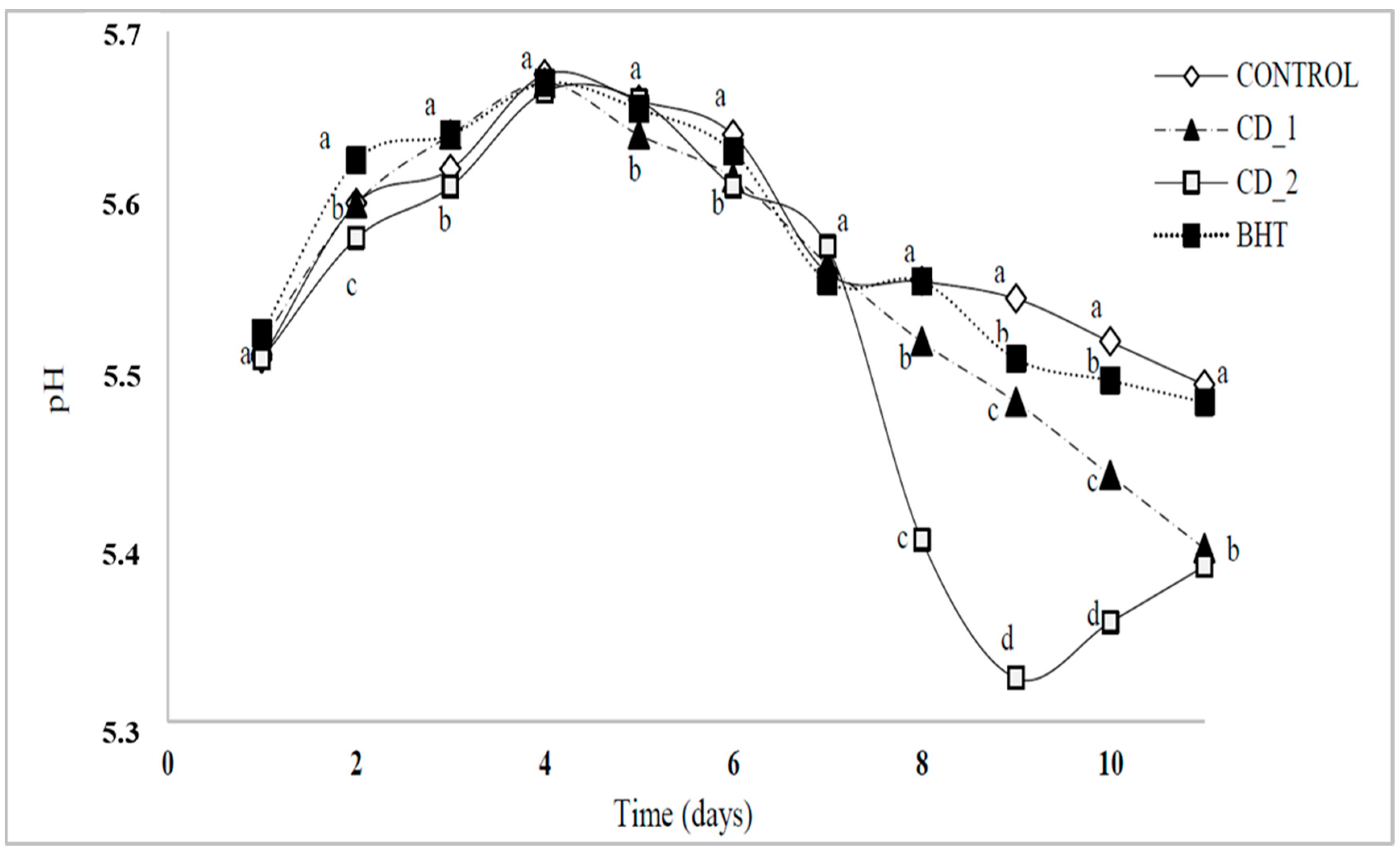
2.4.2. Changes in pH of Raw Beef Patties
2.4.3. Color Changes
| Trait | Days | Control | CD1 | CD2 | BHT | ||||
|---|---|---|---|---|---|---|---|---|---|
| Lightness (L*) | 0 | 37.53 | (3.19) a | ||||||
| 1 | 37.84 | (3.24) a | 38.86 | (1.75) a | 37.42 | (2.47) a | 42.42 | (5.14) b | |
| 2 | 36.42 | (2.22) a | 39.73 | (2.18) b | 39.35 | (1.77) b | 38.71 | (2.90) b | |
| 3 | 39.37 | (5.18) a | 38.74 | (3.55) a | 40.17 | (3.08) a | 40.93 | (3.41) a | |
| 4 | 38.47 | (5.18) a | 40.92 | (2.60) b,a | 34.53 | (2.35) c | 38.52 | (2.99) b | |
| 5 | 42.43 | (2.66) a | 41.34 | (3.05) a | 36.78 | (2.06) b | 37.29 | (4.37) b | |
| 6 | 44.33 | (3.36) a | 41.74 | (3.22) b | 40.05 | (1.74) b | 44.23 | (2.59) a | |
| 7 | 44.38 | (4.94) a,d | 46.36 | (2.85) b,d | 40.47 | (1.76) c | 45.94 | (1.88) d | |
| 8 | 42.85 | (2.50) a | 44.83 | (3.30) b,a | 40.59 | (2.56) c | 35.58 | (4.99) d | |
| 9 | 45.71 | (3.32) a | 48.14 | (3.92) b,a | 42.17 | (2.65) c | 44.63 | (2.12) a | |
| 10 | 44.31 | (3.35) a | 48.68 | (2.14) b,a | 46.76 | (3.76) b,a | 43.07 | (3.03) a,c | |
| 11 | 43.80 | (3.66) a | 50.90 | (1.88) b | 43.01 | (4.15) a | 44.46 | (7.98) a,b | |
| Redness (a*) | 0 | 3.33 | (0.58) a | ||||||
| 1 | 5.08 | (1.73) a | 5.92 | (1.88) b | 4.33 | (1.82) c | 6.42 | (2.08) d | |
| 2 | 6.20 | (0.63) a | 4.63 | (1.18) b | 2.35 | (1.05) c | 6.30 | (1.34) a | |
| 3 | 4.41 | (1.67) a | 6.52 | (1.63) b | 4.56 | (1.03) a | 4.20 | (1.33) a | |
| 4 | 5.04 | (2.14) a | 4.48 | (1.73) b,a | 2.57 | (0.88) c | 3.81 | (1.23) d | |
| 5 | 4.15 | (0.43) a | 2.43 | (0.96) b | 1.86 | (0.59) c,b | 0.87 | (0.59) d | |
| 6 | 2.68 | (0.87) a | 1.75 | (0.81) b | 0.83 | (0.59) c | 2.11 | (0.29) d | |
| 7 | 1.24 | (0.28) a | 0.44 | (0.24) b | 0.82 | (0.45) b,a | 0.72 | (0.36) b,a | |
| 8 | 1.48 | (0.35) a | 0.53 | (0.20) b | 0.38 | (0.29) c,b | 0.66 | (0.24) d,b | |
| 9 | 1.27 | (0.38) a | 0.95 | (0.49) a,b | 0.80 | (0.40) a,b | 0.63 | (0.27) b | |
| 10 | 0.58 | (0.44)a | 0.21 | (0.19) b | −0.58 | (0.50) b | 0.50 | (0.40) a,b | |
| 11 | −0.47 | (0.31) a | −0.78 | (0.34) b | −0.70 | (0.27) a,b | 0.41 | (0.23) a,b | |
| Yellowness (b*) | 0 | 3.33 | (0.82) a | ||||||
| 1 | 6.57 | (1.73) a | 6.89 | (2.45) a | 6.44 | (2.17) a | 8.20 | (1.85) b | |
| 2 | 8.21 | (2.08) a | 6.46 | (2.56) b | 8.39 | (1.07) a | 7.00 | (2.01) c | |
| 3 | 8.15 | (2.86) a | 10.50 | (1.75) b | 8.71 | (1.47) a | 5.89 | (2.05) c | |
| 4 | 6.62 | (2.36) a,b | 7.38 | (2.28) a,b | 8.48 | (2.43) a | 5.56 | (2.68) b | |
| 5 | 8.34 | (0.97) a | 7.38 | (2.50) b | 7.60 | (1.61) b,a | 3.32 | (2.25) c | |
| 6 | 9.60 | (2.01) a | 10.25 | (1.99) b,a | 6.17 | (1.04) c | 9.11 | (1.83) a,b | |
| 7 | 7.02 | (3.36) a | 6.75 | (2.09) b | 9.49 | (2.27) c | 9.00 | (1.61) c | |
| 8 | 10.46 | (0.98) a | 9.89 | (1.55) a | 9.55 | (1.30) a | 2.90 | (2.00) b | |
| 9 | 7.76 | (2.59) a | 8.74 | (2.99) b | 8.56 | (1.42) b | 7.39 | (1.97) a | |
| 10 | 7.69 | (1.16) a | 8.90 | (1.87) b,a | 11.59 | (3.01) c | 9.70 | (1.50) d,b | |
| 11 | 4.52 | (1.35) a | 12.30 | (2.42) b | 10.57 | (3.04) c,b | 6.23 | (4.06) d,a | |
3. Experimental Section
3.1. Plant Material
3.2. Chemicals
3.3. Sample Extraction
3.4. Preparation of Beef Patties
3.5. Antioxidant Capacity Assay (AOC)
FRAP Assay
3.6. Determination of Metmyoglobin
3.7. Headspace Volatile Analysis
3.8. Determination of Secondary Oxidation by TBARS
3.9. pH Measurement
3.10. Color Measurements
3.11. Statistical Analysis
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Lagerstedt, A.; Lundstrom, K.; Lindahl, G. Influence of vacuum or highoxygen modified atmosphere packaging on quality of beef M. longissimus dorsi steaks after different ageing times. Meat Sci. 2011, 87, 101–106. [Google Scholar] [CrossRef] [PubMed]
- Jayathilakan, K.; Sharma, G.K.; Radhakrishna, K.; Bawa, A.S. Antioxidant potential of synthetic and natural antioxidants and its effect on warmed-over-flavour in different species of meat. Food Chem. 2007, 105, 908–916. [Google Scholar] [CrossRef]
- Hinneburg, I.; Dorman, H.J.D.; Hiltunen, R. Antioxidant activities of extracts from selected culinary herbs and spices. Food Chem. 2006, 97, 122–129. [Google Scholar] [CrossRef]
- Zhang, Q.; Liu, X.T.; Liang, J.Y.; Min, Z.D. Chemical constituents from the stems of Caesalpinia decapetala. Chin. J. Nat. Med. 2008, 6, 168–172. [Google Scholar] [CrossRef]
- Kiem, P.V.; Minh, C.V.; Huong, H.T.; Lee, J.L.; Kim, Y.H. Caesaldecan, a cassane diterpenoid from the leaves of Caesalpinia decapetala. Chem. Pharm. Bull. 2005, 53, 428–430. [Google Scholar] [CrossRef] [PubMed]
- Benzie, I.F.F.; Strain, J.J. The ferric reducing ability of plasma (FRAP) as a measure of “antioxidant power”: The FRAP assay. Anal. Biochem. 1996, 239, 70–76. [Google Scholar] [CrossRef] [PubMed]
- Topuz, O.; Yerlikaya, P.; Ucak, I.; Gumus, B.; Aydan, H. Effects of olive oil and olive oil-pomegranate juice sauces on chemical oxidative and sensorial quality of marinated anchovy. Food Chem. 2014, 154, 63–70. [Google Scholar] [CrossRef] [PubMed]
- Bubonja-Sonje, M.; Giacometti, J.; Abram, M. Antioxidant and antilisterial activity of olive oil, cocoa and rosemary extract polyphenols. Food Chem. 2011, 127, 1821–1827. [Google Scholar] [CrossRef]
- Servili, M.; Selvaggini, R.; Esposto, S.; Taticchi, A.; Montedoro, G.; Morozzi, G. Heaalth and sensory properties of virgin olive oil hydrophilic phenols: Agronomic and technological aspects of production that affect their ocurrence in the oil. J. Chromatogr. A 2004, 1054, 113–127. [Google Scholar] [CrossRef]
- Bianco, A.; Uccella, N. Biophenolic components of olives. Food Res. Int. 2000, 33, 475–485. [Google Scholar] [CrossRef]
- Lindahl, G. Colour stability of steaks from large beef cuts aged under vacuum or high oxygen modified atmosphere. Meat Sci. 2011, 87, 428–435. [Google Scholar] [CrossRef] [PubMed]
- Sánchez-Escalante, A.; Djenane, D.; Torrescano, G.; Beltrán, J.A.; Roncalés, P. The effects of ascorbic acid, taurine, carnosine and rosemary powder on colour and lipid stability of beef patties packaged in modified atmosphere. Meat Sci. 2001, 58, 421–429. [Google Scholar] [CrossRef]
- MacLeod, G. The flavour of beef. In Flavor of Meat and Meat Products; Shahidi, F., Ed.; Blackie Academic and Professional: Glasgow, UK, 1994; pp. 4–37. [Google Scholar]
- Im, S.; Hayakawa, F.; Kurata, T. Identification and sensory evaluation of volatile compounds in oxidized porcine liver. J. Agric. Food Chem. 2004, 52, 300–305. [Google Scholar] [CrossRef] [PubMed]
- Meynier, A.; Genot, C.; Gandemer, G. Volatile compounds of oxidized pork phospholipids. J. Am. Oil Chem. Soc. 1998, 75, 1–7. [Google Scholar] [CrossRef]
- Juntachote, T.; Berghofer, E.; Siebenhandl, S.; Bauer, F. The effect of dried galangal powder and its ethanolic extracts on oxidative stability in cooked ground pork. LWT Food Sci. Technol. 2007, 40, 324–330. [Google Scholar] [CrossRef]
- Sampaio, G.R.; Salsanha, T.; Soares, R.A.M.; Torres, E.A.F.S. Effect of natural antioxidant combinations on lipid oxidation in cooked chicken meat during refrigerated storage. Meat Sci. 2012, 93, 715–722. [Google Scholar]
- Fasseas, M.K.; Mountzouris, K.C.; Tarantilis, P.A.; Polissiou, M.; Zervas, G. Antioxidant activity in meat treated with oregano and sage essential oils. Food Chem. 2008, 106, 1188–1194. [Google Scholar] [CrossRef]
- Mitsumoto, M.; O’Grady, M.N.; Kerry, J.P.; Buckley, D.J. Addition of tea catechins and vitamin C on sensory evaluation, colour and lipid stability during chilled storage in cooked or raw beef and chicken patties. Meat Sci. 2005, 69, 773–779. [Google Scholar] [CrossRef] [PubMed]
- McCarthy, T.L.; Kerry, J.P.; Kerry, J.F.; Lynch, P.B.; Buckley, D.J. Assessment of the antioxidant potential of natural food and plant extracts in fresh and previously frozen pork patties. Meat Sci. 2001, 57, 177–184. [Google Scholar] [CrossRef]
- Formanek, Z.; Kerry, J.P.; Higgins, F.M.; Buckley, D.J.; Morrison, P.A.; Farkas, J. Addition of synthetic antioxidants to α-tocopherol acetate supplemented beef patties: Effects of antioxidants and packaging on lipid oxidation. Meat Sci. 2001, 58, 337–341. [Google Scholar] [CrossRef]
- Han, J.; Rhee, K.S. Antioxidant properties of selected Oriental non-culinary/nutraceutical herb extracts as evaluated in raw and cooked meat. Meat Sci. 2005, 70, 25–33. [Google Scholar] [CrossRef] [PubMed]
- Bingol, E.B.; Ergun, O. Effects of modified atmosphere packaging (MAP) on the microbiological quality and shelf life of ostrich meat. Meat Sci. 2011, 88, 774–785. [Google Scholar] [CrossRef] [PubMed]
- Esmer, O.K.; Irkin, R.; Degirmencioglu, N.; Degirmencioglu, A. The effects of modified atmosphere gas composition on microbiological criteria, color and oxidation values of minced beef meat. Meat Sci. 2011, 88, 221–226. [Google Scholar] [CrossRef] [PubMed]
- Cachaldora, A.; García, G.; Lorenzo, J.M.; García-Fontán, M.C. Effect of modified atmosphere and vacuum packaging on some quality characteristics and the shelf-life of “morcilla”, a typical cooked blood sausage. Meat Sci. 2013, 93, 220–225. [Google Scholar] [CrossRef] [PubMed]
- Linden, S. α-Tocoperol, β-Carotin und l-Ascorbinsäure in der Rindermast—Einfluss auf Qualitätsparameter. Ph.D. Thesis, Universität Hamburg, Hamburg, Germany, 2003. [Google Scholar]
- Krzywicki, K. The determination of heam pigments in meat. Meat Sci. 1982, 7, 29–36. [Google Scholar] [CrossRef]
- Grau, A.; Guardiola, F.; Boatella, J.; Barroeta, A.; Codony, R. Measurement of 2-thiobarbituric acid values in dark chicken meat through derivative spectrophotometry: Influence of various parameters. J. Agric. Food Chem. 2000, 48, 1155–1159. [Google Scholar] [CrossRef] [PubMed]
- Sample Availability: Samples of the CD extracts are not available from the authors.
© 2015 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gallego, M.G.; Gordon, M.H.; Segovia, F.J.; Almajano, M.P. Caesalpinia decapetala Extracts as Inhibitors of Lipid Oxidation in Beef Patties. Molecules 2015, 20, 13913-13926. https://doi.org/10.3390/molecules200813913
Gallego MG, Gordon MH, Segovia FJ, Almajano MP. Caesalpinia decapetala Extracts as Inhibitors of Lipid Oxidation in Beef Patties. Molecules. 2015; 20(8):13913-13926. https://doi.org/10.3390/molecules200813913
Chicago/Turabian StyleGallego, Maria G., Michael H. Gordon, Francisco J. Segovia, and María P. Almajano. 2015. "Caesalpinia decapetala Extracts as Inhibitors of Lipid Oxidation in Beef Patties" Molecules 20, no. 8: 13913-13926. https://doi.org/10.3390/molecules200813913
APA StyleGallego, M. G., Gordon, M. H., Segovia, F. J., & Almajano, M. P. (2015). Caesalpinia decapetala Extracts as Inhibitors of Lipid Oxidation in Beef Patties. Molecules, 20(8), 13913-13926. https://doi.org/10.3390/molecules200813913







