Enzyme Inhibitory Radicinol Derivative from Endophytic fungus Bipolaris sorokiniana LK12, Associated with Rhazya stricta
Abstract
:1. Introduction
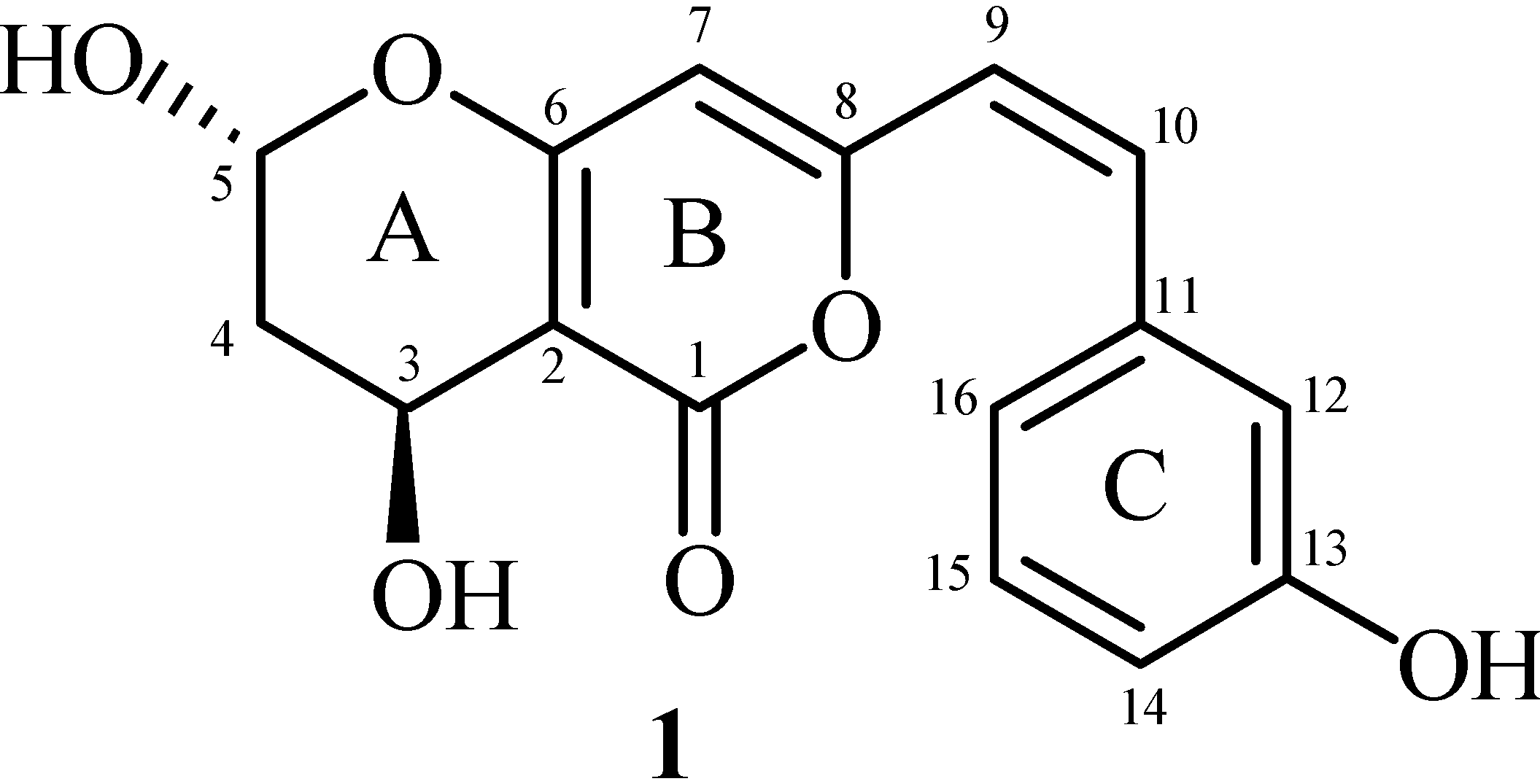
2. Results and Discussion
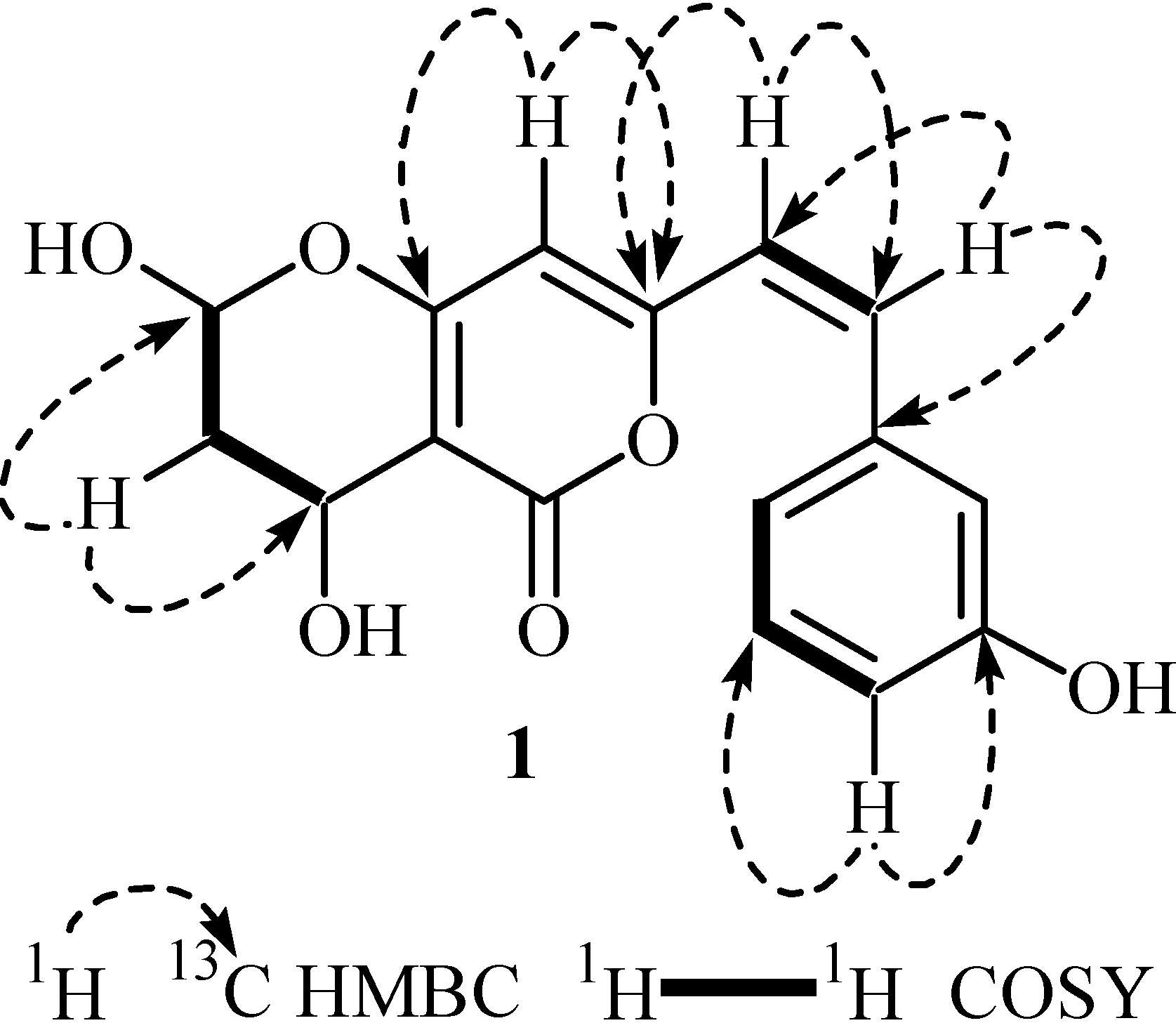
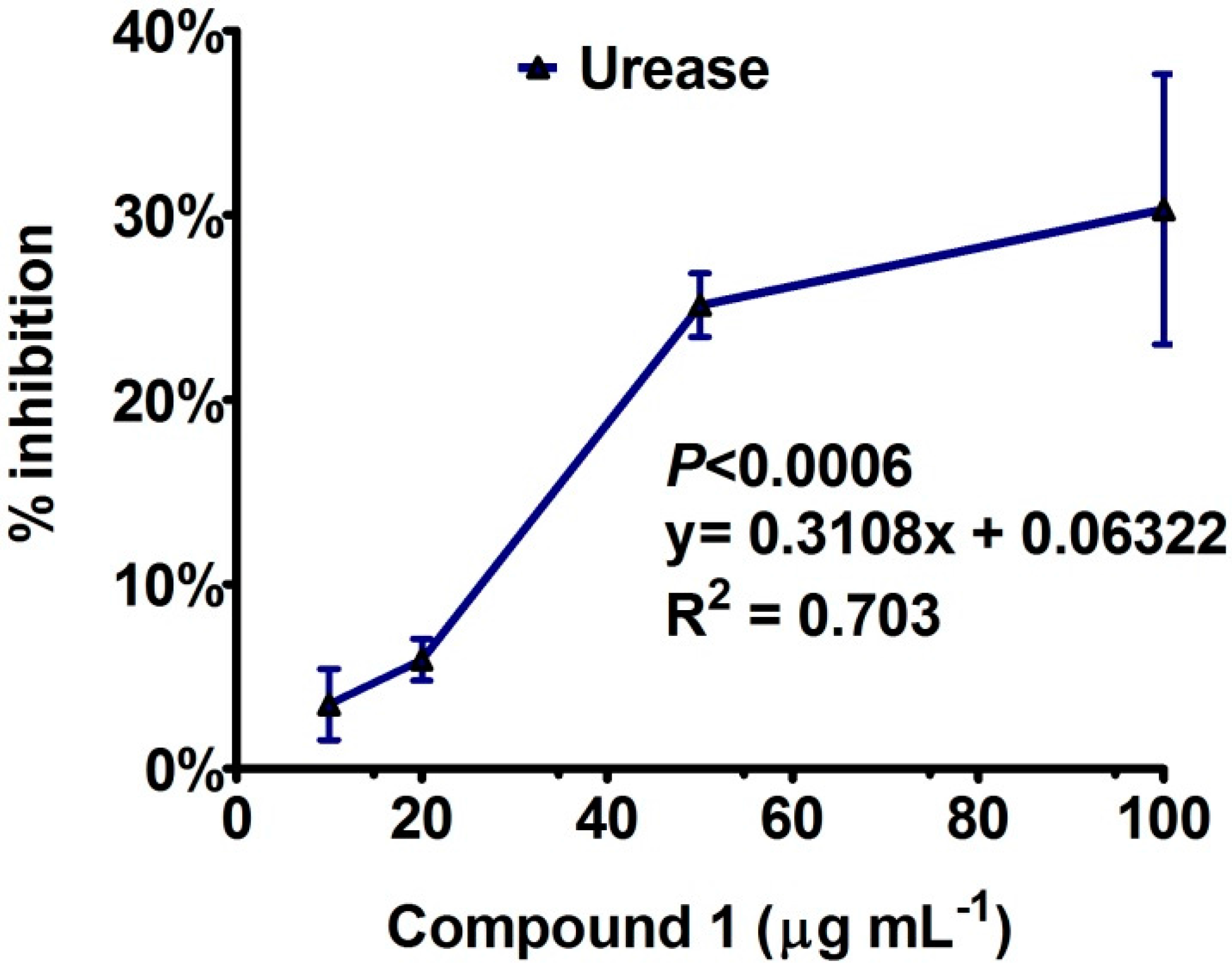
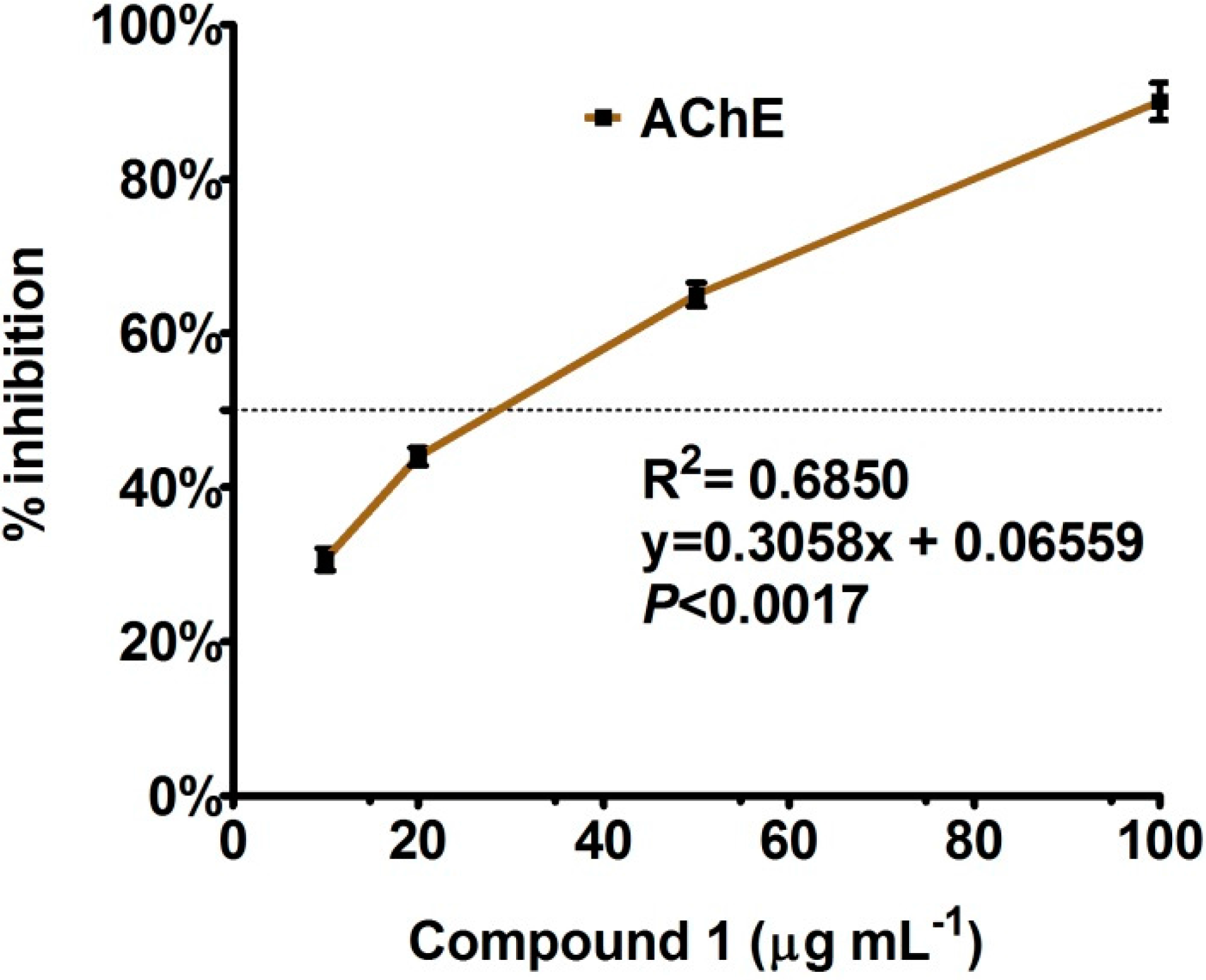
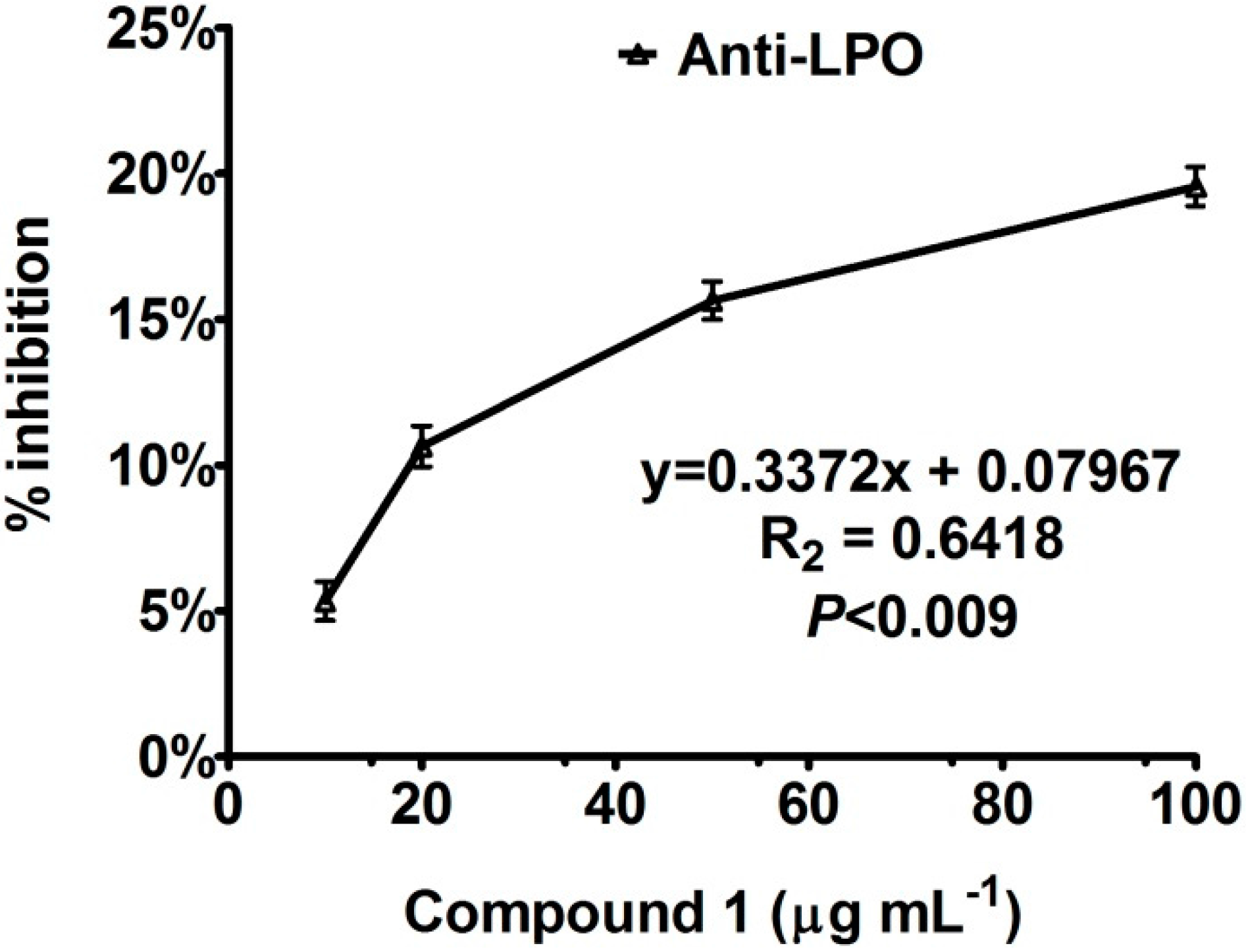
Enzyme Inhibition and Anti-Lipid Peroxidation Activities
3. Experimental Section
3.1. Endophyte Isolation and Identification
3.2. Secondary Metabolite Structure Elucidation
3.3. Extraction and Isolation
3.4. Anti-Lipid Peroxidation and Enzyme Inhibition Assays
3.5. Statistical Analysis
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Schulz, B.; Boyle, C. The endophytic continuum. Mycol. Res. 2005, 109, 661–686. [Google Scholar] [CrossRef] [PubMed]
- Guo, B.; Wang, Y.; Sun, X.; Tang, K. Bioactive natural products from endophytes: A review. Appl. Biochem. Microbiol. 2008, 44, 136–142. [Google Scholar] [CrossRef]
- Kusari, P.; Kusari, S.; Spiteller, M.; Kayser, O. Endophytic fungi harbored in Cannabis sativa L.: Diversity and potential as biocontrol agents against host plant-specific phytopathogens. Fungal Divers. 2013, 60, 137–151. [Google Scholar] [CrossRef]
- Gao, J.M.; Xiao, J.; Zhang, Q.; Tang, J.J.; Zhang, A.L.; Gao, Y.Q. Secondary Metabolites from the Endophytic Botryosphaeria dothidea of Melia azedarach and their antifungal, antibacterial, antioxidant, and cytotoxic activities. J. Agric. Food Chem. 2014, 62, 3584–3590. [Google Scholar]
- Khan, A.L.; Hussain, J.; Al-Harrasi, A.; Al-Rawahi, A.; Lee, I.J. Endophytic fungi: Resource for gibberellins and crop resistance to abiotic stress. Crit. Rev. Biotechnol. 2015, 35, 62–74. [Google Scholar] [CrossRef] [PubMed]
- Schulz, B.; Boyle, C.; Draeger, S.; Römmert, K.; Krohn, K. Endophytic fungi: A source of novel biologically active secondary metabolites. Mycol. Res. 2002, 106, 996–1004. [Google Scholar] [CrossRef]
- Khan, A.L.; Hamayun, M.; Kim, Y.H.; Kang, S.M.; Lee, I.J. Ameliorative symbiosis of endophyte (Penicillium funiculosum LHL06) under salt stress elevated plant growth of Glycine max L. Plant Physiol. Biochem. 2011, 49, 852–862. [Google Scholar] [CrossRef] [PubMed]
- Strobel, G.; Daisy, B.; Castillo, U.; Harper, J. Natural products from endophytic microorganisms. J. Nat. Prod. 2004, 67, 257–268. [Google Scholar] [CrossRef] [PubMed]
- Tan, R.X.; Zou, W.X. Endophytes: A rich source of functional metabolites. Nat. Prod. Rep. 2001, 18, 448–459. [Google Scholar] [CrossRef] [PubMed]
- Zaman, K.; Perveen, S.; Muzaffar, A.; Choudhary, M.I.; Pervin, A. Alkaloids from Rhazya stricta. Phytochemistry 1991, 30, 1285–1293. [Google Scholar]
- Ali, B.H.; Al-Qarawi, A.; Bashir, A.K.; Tanira, M. Phytochemistry, pharmacology and toxicity of Rhazya stricta Decne: A review. Phytother. Res. 2000, 14, 229–234. [Google Scholar] [CrossRef]
- Gilani, S.A.; Kikuchi, A.; Shinwari, Z.K.; Khattak, Z.I.; Watanabe, K.N. Phytochemical, pharmacological and ethnobotanical studies of Rhazya stricta Decne. Phytother. Res. 2007, 4, 301–307. [Google Scholar] [CrossRef] [PubMed]
- Sheridan, H.; Canning, A.M. Novel radicinol derivatives from long-term cultures of Alternaria chrysanthemi. J. Nat. Prod. 1999, 62, 1568–1569. [Google Scholar] [CrossRef] [PubMed]
- Giridharan, P.; Verekar, S.A.; Gohil, A.R.; Mishra, P.D.; Khanna, A.; Deshmukh, S.K. Antiproliferative activity of hamigerone and radicinol isolated from Bipolaris papendorfii. BioMed Res. Int. 2014, 2014, 890904. [Google Scholar] [CrossRef] [PubMed]
- Shaheen, F.; Ali, L.; Ali, S.; Erdemoglu, N.; Sener, B. Antioxidant flavonoids from Tamus communis ssp. Cretica. Chem. Nat. Compd. 2009, 45, 346–349. [Google Scholar] [CrossRef]
- Dixon, N.E.; Gazzola, C.; Watters, J.J.; Blakeley, R.L.; Zerner, B. Inhibition of Jack Bean urease (EC 3.5.1.5) by acetohydroxamic acid and by phosphoramidate: Equivalent weight for urease. J. Am. Chem. Soc. 1975, 97, 4130–4131. [Google Scholar] [CrossRef] [PubMed]
- Krajewska, B. Ureases I. Functional, catalytic and kinetic properties: A review. J. Mol. Catal. B 2009, 59, 9–21. [Google Scholar] [CrossRef]
- Burne, R.A.; Chen, Y.Y.M. Bacterial ureases in infectious diseases. Microbes Infect. 2000, 2, 533–542. [Google Scholar] [CrossRef]
- Krajewska, B.; Zaborska, W. Jack bean urease: The effect of active-site binding inhibitors on the reactivity of enzyme thiol groups. Bioorg. Chem. 2007, 35, 355–365. [Google Scholar] [CrossRef] [PubMed]
- Covacci, A.; Telford, J.L.; del Giudice, G.; Parsonnet, J.; Rappuoli, R. Helicobacter pylori virulence and genetic geography. Science 1999, 284, 1328–1333. [Google Scholar] [CrossRef] [PubMed]
- Shabana, S.; Kawai, A.; Kai, K.; Akiyama, K.; Hayashi, H. Inhibitory activity against urease of quercetin glycosides isolated from Allium cepa and Psidium guajava. Biosci. Biotechnol. Biochem. 2010, 74, 878–880. [Google Scholar] [CrossRef] [PubMed]
- Bae, E.A.; Han, M.J.; Kim, D.H. In vitro anti-Helicobacter pylori activity of some flavonoids and their metabolites. Planta Med. 1999, 65, 442–443. [Google Scholar] [CrossRef] [PubMed]
- Davies, P.; Maloney, A.J. Selective loss of central cholinergic neurons in Alzheimer’s disease. Lancet 1976, 2, 1403. [Google Scholar] [CrossRef]
- Mangialasche, F.M.; Solomon, A.; Winblad, B.; Mecocci, P.; Kivipelto, M. Alzheimer’s disease: Clinical trials and drug development. Lancet Neurol. 2010, 9, 702–716. [Google Scholar] [CrossRef]
- Mukherjee, P.K.; Kumar, V.; Mal, M.; Houghton, P.J. Acetylcholinesterase inhibitors from plants. Phytomedicine 2007, 14, 289–300. [Google Scholar] [CrossRef] [PubMed]
- Murray, A.P.; Faraoni, M.B.; Castro, M.J.; Alza, N.P.; Cavallaro, V. Natural AChE inhibitors from plants and their contribution to Alzheimer’s disease therapy. Curr. Neuropharmacol. 2013, 11, 388–413. [Google Scholar] [CrossRef] [PubMed]
- Arnold, A.E.; Lutzoni, F. Diversity and host range of foliar fungal endophytes: Are tropical leaves biodiversity hotspots. Ecology 2007, 88, 541–549. [Google Scholar] [CrossRef] [PubMed]
- Torres, M.S.; White, J.F.; Zhang, X.; Hinton, D.M.; Bacon, C.W. Endophyte-mediated adjustments in host morphology and physiology and effects on host fitness traits in grasses. Fungal Ecol. 2012, 5, 322–330. [Google Scholar] [CrossRef]
- Tamura, K.; Peterson, D.; Peterson, N.; Stecher, G.; Nei, M.; Kumar, S. MEGA5: Molecular evolutionary genetics analysis using maximum likelihood, evolutionary distance, and maximum parsimony methods. Mol. Biol. Evol. 2011, 28, 2731–2739. [Google Scholar] [CrossRef] [PubMed]
- Gulati, V.; Harding, I.H.; Palombo, E.A. Enzyme inhibitory and antioxidant activities of traditional medicinal plants: Potential application in the management of hyperglycemia. BMC Complement. Altern. Med. 2012, 12, 77. [Google Scholar] [CrossRef] [PubMed]
- Golbabaei, S.; Bazl, R.; Golestanian, S.; Nabati, R.; Omrany, R.B.; Yousefi, B.; Hajiaghaee, R.; Rezazadeh, S.; Amanlou, M. Urease inhibitory activities of β-boswellic acid derivatives. Daru 2013, 21. [Google Scholar] [CrossRef] [PubMed]
- Ingkaninan, K.; Temkitthawon, P.; Chuenchom, K.; Yuyaem, T.; Thongnoi, W. Screening for acetylcholinesterase inhibitory activity in plants used in Thai traditional rejuvenating and neurotonic remedies. J. Ethnopharmacol. 2003, 89, 261–264. [Google Scholar] [CrossRef] [PubMed]
- Sample Availability: Samples of the compound 1 is available from the authors.
© 2015 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Khan, A.L.; Ali, L.; Hussain, J.; Rizvi, T.S.; Al-Harrasi, A.; Lee, I.-J. Enzyme Inhibitory Radicinol Derivative from Endophytic fungus Bipolaris sorokiniana LK12, Associated with Rhazya stricta. Molecules 2015, 20, 12198-12208. https://doi.org/10.3390/molecules200712198
Khan AL, Ali L, Hussain J, Rizvi TS, Al-Harrasi A, Lee I-J. Enzyme Inhibitory Radicinol Derivative from Endophytic fungus Bipolaris sorokiniana LK12, Associated with Rhazya stricta. Molecules. 2015; 20(7):12198-12208. https://doi.org/10.3390/molecules200712198
Chicago/Turabian StyleKhan, Abdul Latif, Liaqat Ali, Javid Hussain, Tania Shamim Rizvi, Ahmed Al-Harrasi, and In-Jung Lee. 2015. "Enzyme Inhibitory Radicinol Derivative from Endophytic fungus Bipolaris sorokiniana LK12, Associated with Rhazya stricta" Molecules 20, no. 7: 12198-12208. https://doi.org/10.3390/molecules200712198
APA StyleKhan, A. L., Ali, L., Hussain, J., Rizvi, T. S., Al-Harrasi, A., & Lee, I.-J. (2015). Enzyme Inhibitory Radicinol Derivative from Endophytic fungus Bipolaris sorokiniana LK12, Associated with Rhazya stricta. Molecules, 20(7), 12198-12208. https://doi.org/10.3390/molecules200712198








