Quantitative 1H-NMR Method for the Determination of Tadalafil in Bulk Drugs and its Tablets
Abstract
:1. Introduction

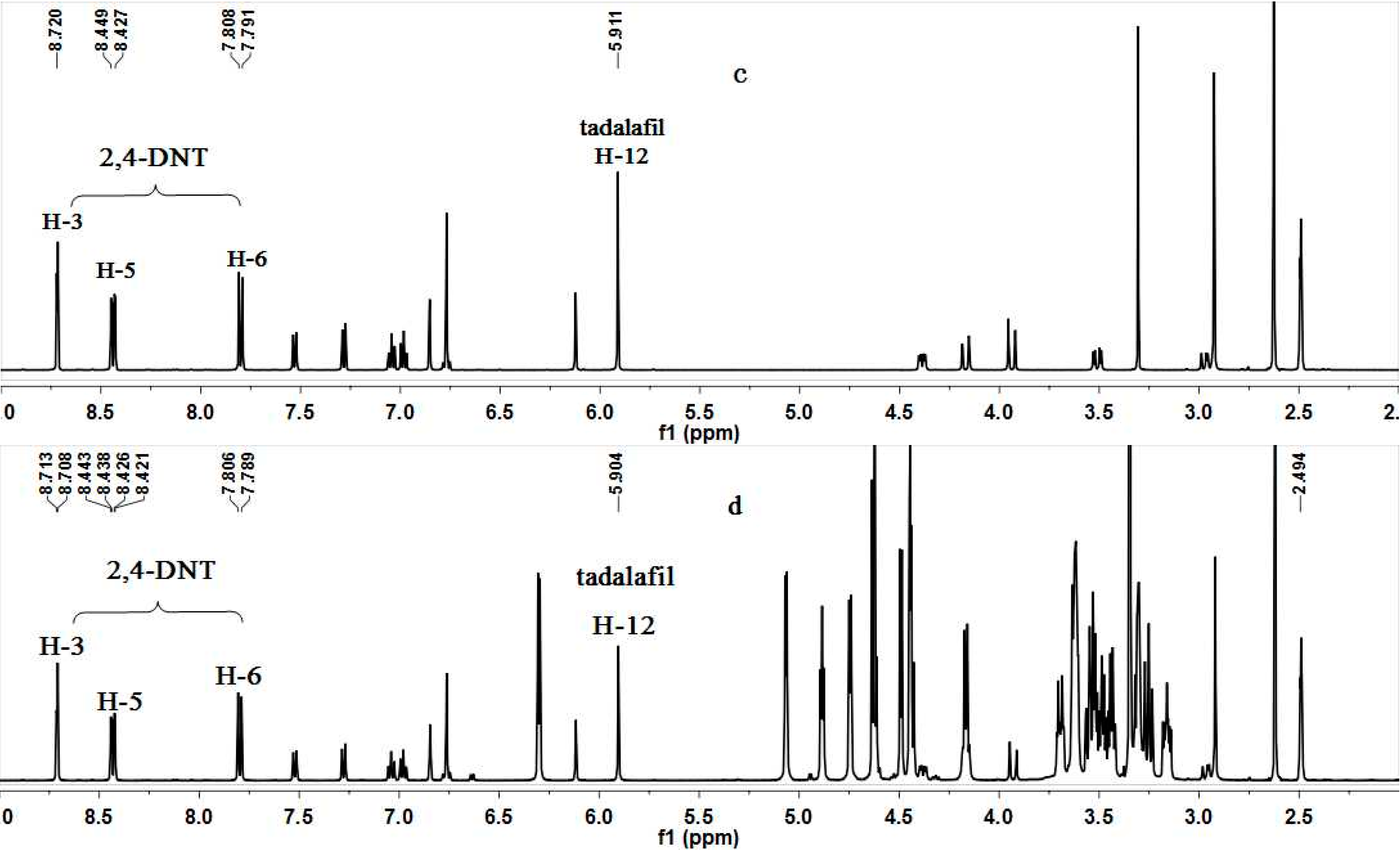
2. Results and Discussion
2.1. qNMR Methods
2.2. Validation
2.2.1. Linearity and Range
| No. | mstd (mg) | mx (mg) | mx/mstd | Ax/Astd | ||
|---|---|---|---|---|---|---|
| δ * 8.72 | δ 8.45 | δ 7.82 | ||||
| 1 | 5.18 | 2.41 | 0.47 | 0.44 | 0.44 | 0.44 |
| 2 | 4.83 | 3.45 | 0.71 | 0.67 | 0.67 | 0.67 |
| 3 | 5.56 | 5.14 | 0.92 | 0.87 | 0.87 | 0.87 |
| 4 | 6.46 | 11.72 | 1.81 | 1.68 | 1.68 | 1.67 |
| 5 | 5.48 | 14.56 | 2.66 | 2.46 | 2.45 | 2.45 |
| 6 | 5.00 | 19.40 | 3.88 | 3.64 | 3.64 | 3.64 |
| Linear equation | y = 0.9347x − 0.0021 | y = 0.9339x − 0.0030 | y = 0.9344x − 0.0052 | |||
| R2 | 0.9999 | 0.9999 | 0.9999 | |||
2.2.2. Limit of Quantification (LOQ)
2.2.3. Precision and Stability
| No. | mstd (mg) | mx (mg) | Px (%) | |||
|---|---|---|---|---|---|---|
| δ * 8.72 | δ 8.45 | δ 7.82 | ||||
| Precision (n = 3) | 1 | 4.67 | 5.11 | 99.87 | 100.04 | 100.21 |
| 2 | 5.29 | 5.37 | 100.13 | 100.13 | 99.81 | |
| 3 | 5.13 | 5.73 | 99.51 | 99.51 | 99.17 | |
| 4 | 5.02 | 5.61 | 100.84 | 100.84 | 100.32 | |
| 5 | 5.95 | 5.43 | 99.59 | 99.59 | 99.17 | |
| 6 | 5.56 | 5.14 | 100.11 | 99.82 | 99.82 | |
| Average value | / | / | 100.01 | 99.99 | 99.75 | |
| RSD% | / | / | 0.48 | 0.48 | 0.49 | |
| Repeatability | 1 | 100.71 | 100.71 | 100.19 | ||
| 2 | 100.21 | 100.21 | 100.21 | |||
| 3 | 99.70 | 100.21 | 100.21 | |||
| 4 | 99.70 | 99.70 | 100.21 | |||
| 5 | 99.70 | 100.21 | 99.70 | |||
| 6 | 99.70 | 100.21 | 100.21 | |||
| Average value | 99.95 | 100.21 | 100.12 | |||
| RSD% | 0.43 | 0.32 | 0.21 | |||
| Stability | 0 # | 100.11 | 99.62 | 99.62 | ||
| 1 | 101.10 | 100.11 | 99.62 | |||
| 2 | 100.11 | 100.11 | 99.62 | |||
| 4 | 99.62 | 100.11 | 99.62 | |||
| 8 | 99.62 | 100.11 | 99.62 | |||
| 24 | 100.11 | 100.11 | 99.62 | |||
| Average value | 100.11 | 100.03 | 99.62 | |||
| RSD% | 0.54 | 0.20 | 0.00 | |||
2.2.4. Recovery Tests
| No. | Tadalafil | Recovery (%) | ||||||
|---|---|---|---|---|---|---|---|---|
| In Tablets (mg) | Added (mg) | Found (mg) | ||||||
| δ * 8.72 | δ 8.45 | Δ 7.82 | δ 8.72 | δ 8.45 | δ 7.82 | |||
| 1 | 2.51 | 2.00 | 4.48 | 4.53 | 4.48 | 98.60 | 101.04 | 98.60 |
| 2 | 2.45 | 1.84 | 4.30 | 4.31 | 4.30 | 100.75 | 101.56 | 100.75 |
| 3 | 2.54 | 2.02 | 4.53 | 4.54 | 4.51 | 98.50 | 99.17 | 97.50 |
| 4 | 2.46 | 2.48 | 4.96 | 5.00 | 4.95 | 101.10 | 102.65 | 100.72 |
| 5 | 2.50 | 2.59 | 5.11 | 5.12 | 5.04 | 100.71 | 101.16 | 98.04 |
| 6 | 2.52 | 2.52 | 4.96 | 4.96 | 5.04 | 96.80 | 96.80 | 99.90 |
| 7 | 2.52 | 3.00 | 5.60 | 5.54 | 5.44 | 102.65 | 100.66 | 97.26 |
| 8 | 2.50 | 2.93 | 5.44 | 5.45 | 5.44 | 100.37 | 100.78 | 100.37 |
| 9 | 2.52 | 3.01 | 5.58 | 5.61 | 5.48 | 101.60 | 102.54 | 98.38 |
| Average value | / | / | / | / | / | 100.12 | 100.71 | 99.06 |
| RSD% | / | / | / | / | / | 1.81 | 1.78 | 1.40 |
2.3. Sample Analysis
| Samples | Batch No. | Specifications (mg) | qNMR Method (n = 6) | HPLC Method | |||
|---|---|---|---|---|---|---|---|
| δ * | % | RSD% | % | RSD% | |||
| Tadalafil | 140501 | / | 8.72 | 100.38 | 1.31 | 99.99 | 0.66 |
| 8.45 | 99.73 | 0.30 | |||||
| 7.82 | 99.38 | 0.45 | |||||
| 140502 | / | 8.72 | 100.06 | 0.19 | 99.90 | 0.94 | |
| 8.45 | 99.83 | 0.39 | |||||
| 7.82 | 99.77 | 0.25 | |||||
| 140503 | / | 8.72 | 99.91 | 0.38 | 100.05 | 1.02 | |
| 8.45 | 100.06 | 0.30 | |||||
| 7.82 | 99.09 | 0.54 | |||||
| Tadalafil tablets | 140710 | 5 | 8.72 | 99.01 | 0.90 | 99.73 | 1.15 |
| 8.45 | 99.31 | 0.88 | |||||
| 7.82 | 100.22 | 0.98 | |||||
| C219428 | 5 | 8.72 | 99.10 | 1.33 | 99.23 | 0.84 | |
| 8.45 | 99.27 | 0.68 | |||||
| 7.82 | 99.53 | 0.68 | |||||
| 140725 | 20 | 8.72 | 99.70 | 2.01 | 100.36 | 1.05 | |
| 8.45 | 98.98 | 1.80 | |||||
| 7.82 | 100.37 | 0.74 | |||||
| C023347 | 20 | 8.72 | 99.87 | 1.05 | 101.98 | 1.16 | |
| 8.45 | 100.62 | 1.13 | |||||
| 7.82 | 101.54 | 1.06 | |||||
3. Experimental Section
3.1. Materials
3.2. Instrumentation
3.3. Sample Preparation and Calculations
3.3.1. qNMR Analysis of Tadalafil
3.3.2. qNMR Analysis of Tadalafil in Tablets
3.3.3. HPLC Analysis of Tadalafil
3.3.4. HPLC Analysis of Tadalafil in Tablets
4. Conclusions
Supplementary Materials
Author Contributions
Conflicts of Interest
References
- Schoenberger, T. Determination of standard sample purity using the high-precision 1H-NMR process. Anal. Bioanal. Chem. 2012, 403, 247–254. [Google Scholar] [CrossRef] [PubMed]
- Liu, S.Y.; Hu, C.Q. A comparative uncertainty study of the calibration of macrolide antibiotic reference standards using quantitative nuclear magnetic resonance and mass balance methods. Anal. Chim. Acta 2007, 602, 114–121. [Google Scholar] [CrossRef] [PubMed]
- Simmler, C.; Napolitano, J.G.; McAlpine, J.B.; Chen, S.N.; Pauli, G.F. Universal quantitative NMR analysis of complex natural samples. Curr. Opin. Biotech. 2014, 25, 51–59. [Google Scholar] [CrossRef] [PubMed]
- Holzgrabe, U.; Deubner, R.; Schollmayer, C.; Waibel, B. Quantitative NMR spectroscopy—Applications in drug analysis. J. Pharm. Biomed. Anal. 2005, 38, 806–812. [Google Scholar] [CrossRef] [PubMed]
- Ohtsuki, T.; Sato, K.; Sugimoto, N.; Akiyama, H.; Kawamura, Y. Absolute quantification for benzoic acid in processed foods using quantitative proton nuclear magnetic resonance spectroscopy. Talanta 2012, 99, 342–348. [Google Scholar] [CrossRef] [PubMed]
- Salem, A.A.; Mossa, H.A. Method validation and determination of levofloxacin, metronidazole and sulfamethoxazole in an aqueous pharmaceutical, urine and blood plasma samples using quantitative nuclear magnetic resonance spectrometry. Talanta 2012, 88, 104–114. [Google Scholar] [CrossRef] [PubMed]
- Garrido, R.; Puyada, A.; Fernandez, A.; Gonzalez, M.; Ramirez, U.; Cardoso, F.; Valdes, Y.; Gonzalez, D.; Fernandez, V.; Verez, V.; Velez, H. Quantitative proton nuclear magnetic resonance evaluation and total assignment of the capsular polysaccharide Neisseria meningitides serogroup X. J. Pharm. Biomed. Anal. 2012, 70, 295–300. [Google Scholar] [CrossRef] [PubMed]
- Staneva, J.; Denkova, P.; Todorova, M.; Evstatieva, L. Quantitative analysis of sesquiterpene lactones in extract of Arnica montana L. by 1H-NMR spectroscopy. J. Pharm. Biomed. Anal. 2011, 54, 94–99. [Google Scholar] [CrossRef] [PubMed]
- Goger, N.G.; Parlatan, H.K.; Basan, H.; Berkkan, A.; Ozden, T. Quantitative determination of azathioprine in tablets by 1H-NMR spectroscopy. J. Pharm. Biomed. Anal. 1999, 21, 685–689. [Google Scholar] [CrossRef]
- Zoppi, A.; Linares, M.; Longhi, M. Quantitative analysis of enalapril by 1H-NMR spectroscopy in tablets. J. Pharm. Biomed. Anal. 2005, 37, 627–630. [Google Scholar] [CrossRef] [PubMed]
- Trefi, S.; Routaboul, C.; Hamieh, S.; Gilard, V.; Martino, M.M.; Martino, R. Analysis of illegally manufactured formulations of tadalafil (Cialis) by 1H-NMR, 2D DOY 1H-NMR and Raman spectroscopy. J. Pharm. Biomed. Anal. 2008, 47, 103–113. [Google Scholar] [CrossRef] [PubMed]
- The United States Pharmacopeia (USP); United States PharmacopeiaL Convention Inc.: Rockville, MD, USA, 2013; pp. 5261–6265.
- Chen, Y.L.; Tao, L.H.; Hou, H.; Zhang, Y.; Zhou, X.D.; Fan, X.J. Quantitative determination of berberine hydrochloride by proton nuclear magnetic resonance spectroscopy with internal standard method. J. Third Mil. Med. Uni. 2009, 31, 2217–2219. [Google Scholar]
- Malz, F.; Jancke, H. Validation of quantitative NMR. J. Pharm. Biomed. Anal. 2005, 38, 813–823. [Google Scholar] [CrossRef] [PubMed]
- Sample Availability: Samples of the compounds tadalafil and 2,4-DNT are available from the authors.
© 2015 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yang, Q.; Qiu, H.; Guo, W.; Wang, D.; Zhou, X.; Xue, D.; Zhang, J.; Wu, S.; Wang, Y. Quantitative 1H-NMR Method for the Determination of Tadalafil in Bulk Drugs and its Tablets. Molecules 2015, 20, 12114-12124. https://doi.org/10.3390/molecules200712114
Yang Q, Qiu H, Guo W, Wang D, Zhou X, Xue D, Zhang J, Wu S, Wang Y. Quantitative 1H-NMR Method for the Determination of Tadalafil in Bulk Drugs and its Tablets. Molecules. 2015; 20(7):12114-12124. https://doi.org/10.3390/molecules200712114
Chicago/Turabian StyleYang, Qingyun, Hui Qiu, Wei Guo, Dongmei Wang, Xingning Zhou, Dan Xue, Jinlan Zhang, Song Wu, and Yinghong Wang. 2015. "Quantitative 1H-NMR Method for the Determination of Tadalafil in Bulk Drugs and its Tablets" Molecules 20, no. 7: 12114-12124. https://doi.org/10.3390/molecules200712114
APA StyleYang, Q., Qiu, H., Guo, W., Wang, D., Zhou, X., Xue, D., Zhang, J., Wu, S., & Wang, Y. (2015). Quantitative 1H-NMR Method for the Determination of Tadalafil in Bulk Drugs and its Tablets. Molecules, 20(7), 12114-12124. https://doi.org/10.3390/molecules200712114







