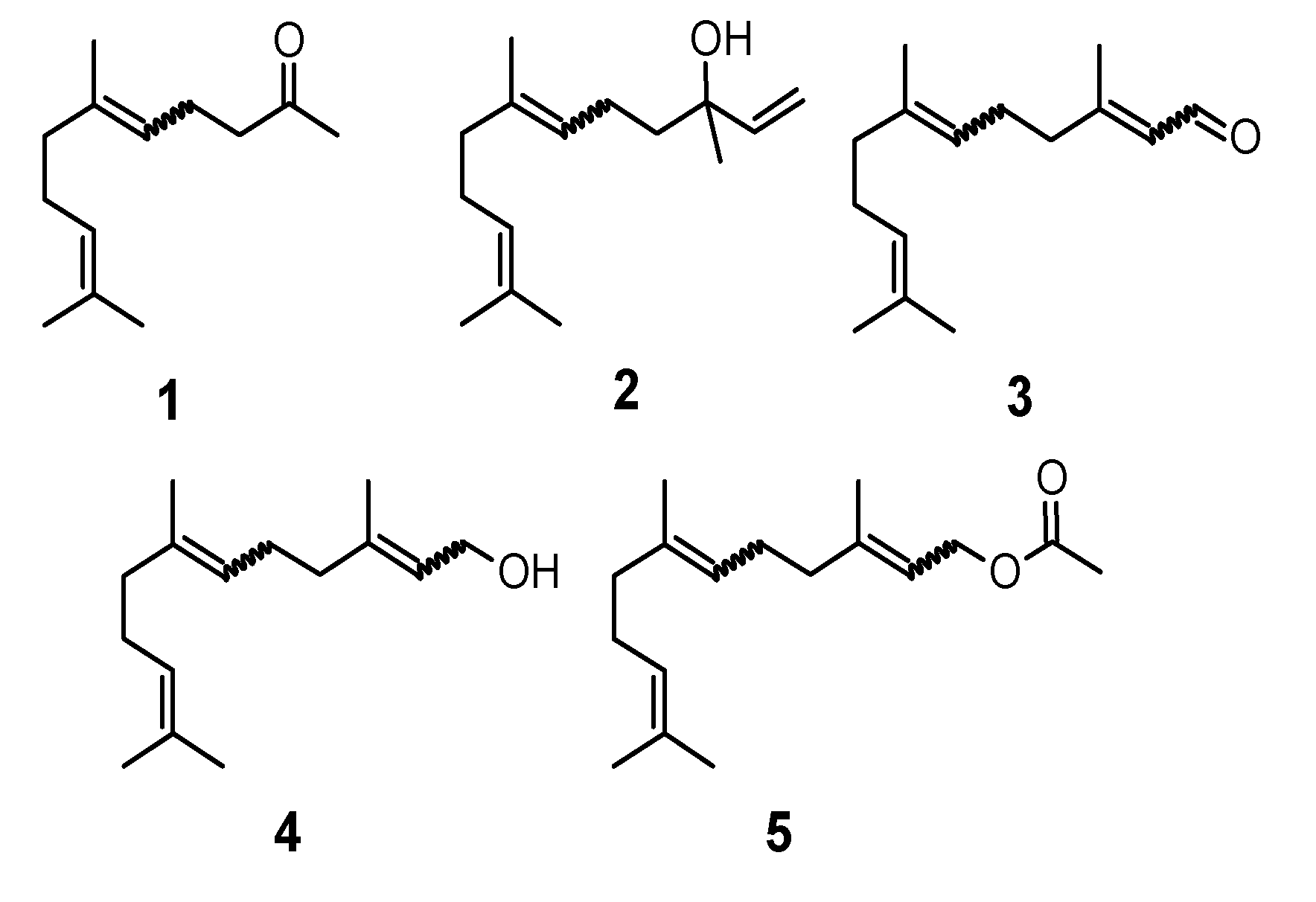
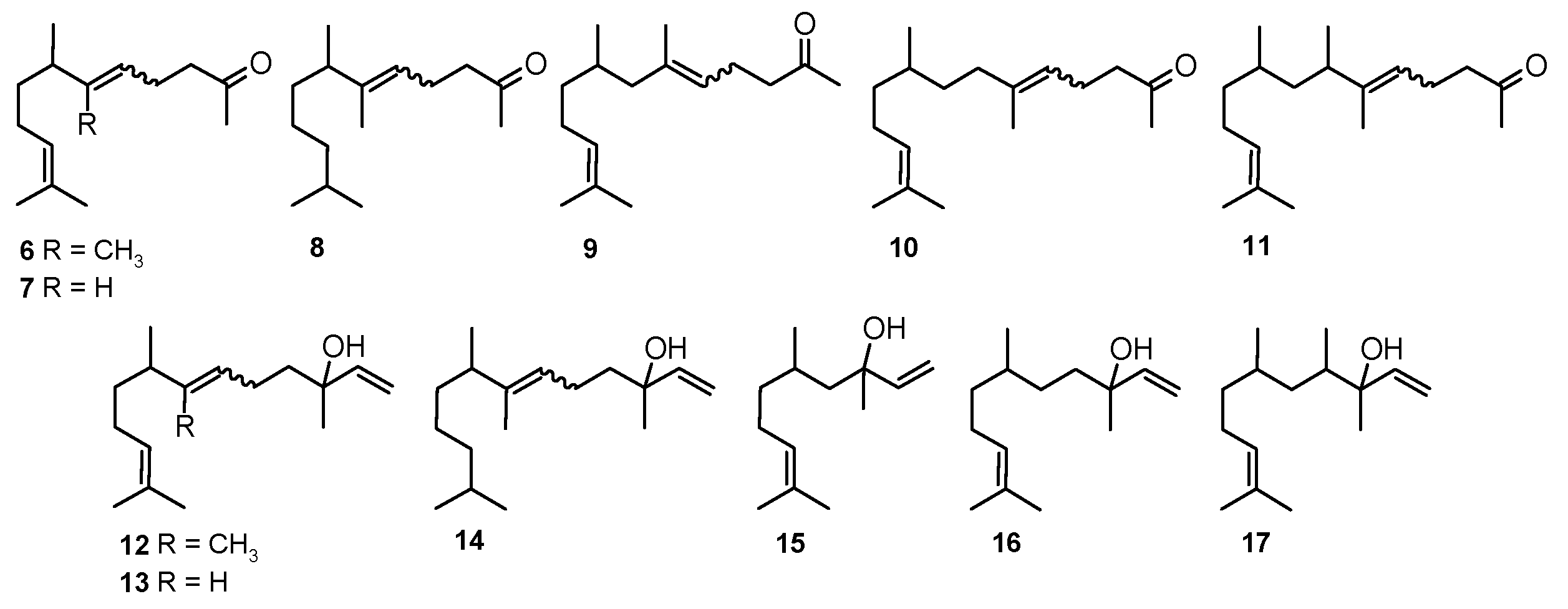
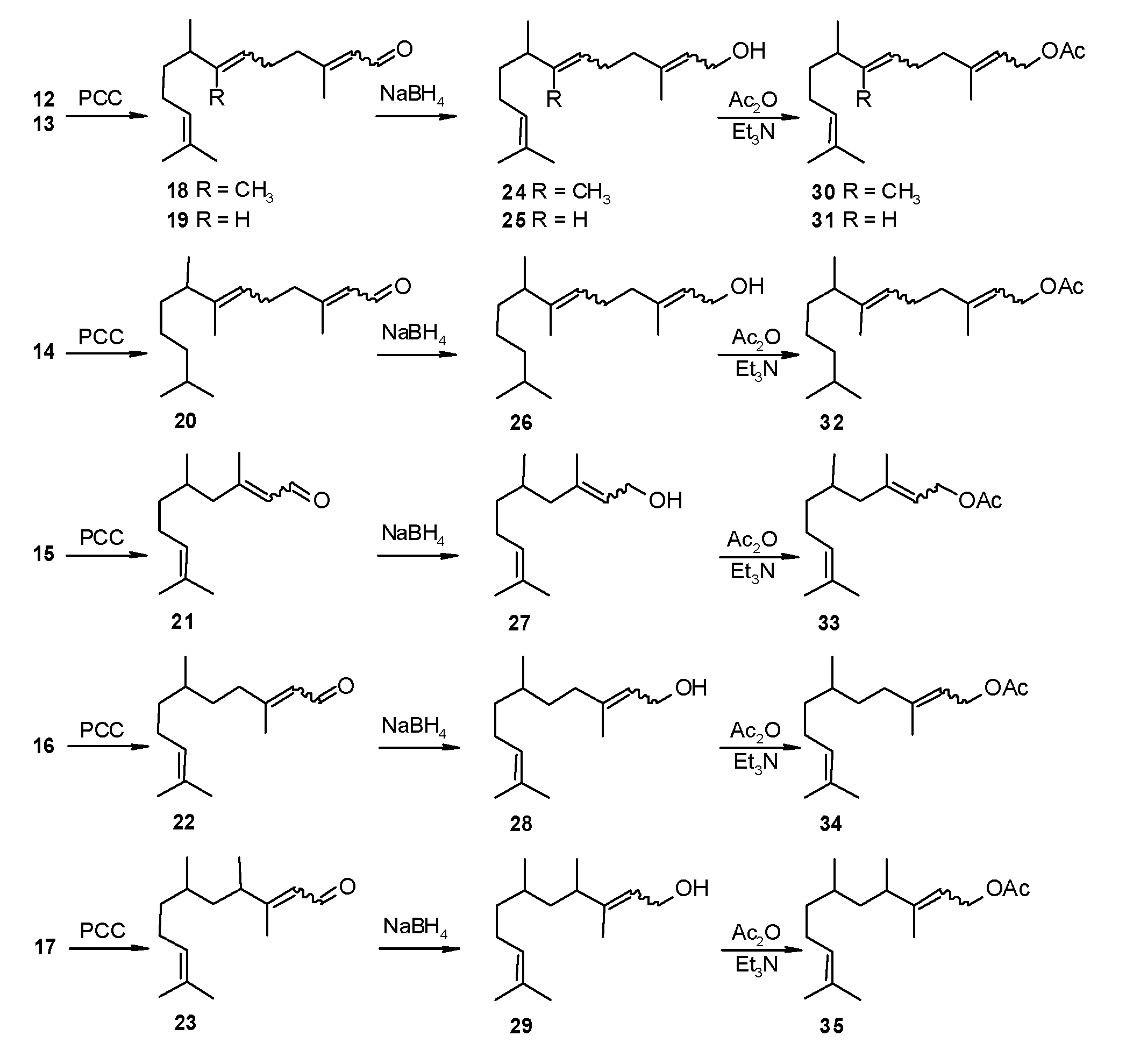
3.2. General Method for Rearrangement of the Tertiary Allylic Alcohols Combined with Oxidation to Aldehydes-Synthesis of Compounds Structurally Related to Farnesal (18–23)
A total of 340 mmol of the appropriate allylic alcohol (12–17), 150 mL of dichloromethane and 50 g of silica gel was placed into a flask. During vigorous stirring, 410 mmol of pyridinium chlorochromate (PCC) was added portion-wise. The temperature was maintained at 10 °C. After addition, the stirring was continued for another 12 h. After that time, 10 mL of methanol was added and the mixture was stirred again for 1 h. The products were isolated by column chromatography with diethyl ether as eluent. Purity of the products according to the GC analysis was 88%–97%. The obtained products (18–23) were additionally distilled or purified by column chromatography (see below).
3.2.1. 3,7,8,12-Tetramethyltrideca-2,6,11-trienal (18)
Fractional distillation (boiling point 131–134 °C/0.1 Torr) gave the product with a yield 43%. When the product was isolated by column chromatography on silica (hexane:ethyl acetate 96:4) the yield was 72%. IR (ATR): 2961, 2922, 2856, 2765, 1674, 1633, 1445, 1377, 1156, 1121, 830 cm−1; 1H-NMR (250 MHz, CDCl3) Isomer (2Z,6Z) δ: 9.90 (d, J = 8.2 Hz, 1H), 5.85 (dd, J = 8.2 Hz, 1.2 Hz, 1H), 5.14–5.00 (m, 2H), 2.56 (dd, J = 15.1, 7.5 Hz, 2H), 2.57–2.45 (m, 1H), 2.28–2.16 (m, 2H), 1.95 (d, J = 1.2 Hz, 3H), 1.80 (dd, J = 14.6 Hz, 7.1 Hz, 2H), 1.64 (s, 3H), 1.54 (s, 3H), 1.53 (s, 3H), 1.37–1.20 (m, 2H), 0.92 (d, J = 6.9 Hz, 3H); Isomer (2Z,6E) δ: 9.88 (d, J = 8.2 Hz, 1H), 5.85 (dd, J = 8.2 Hz, 1.2 Hz, 1H), 5.14–5.00 (m, 2H), 2.57 (t, J = 7.5 Hz, 3H), 2.28–2.16 (m, 2H), 2.05 (dd, J = 14.4 Hz, 6.9 Hz, 1H), 1.96 (d, J = 1.2 Hz, 3H), 1.80 (dd, J = 14.6 Hz, 7.1 Hz, 2H), 1.64 (s, 3H), 1.54 (s, 3H), 1.48 (s, 3H), 1.37–1.20 (m, 2H), 0.92 (d, J = 6.9 Hz, 3H); Isomer (2E,6Z) δ: 9.95 (d, J = 8.1 Hz, 1H), 5.85 (dd, J = 8.1 Hz, 1.1 Hz, 1H), 5.10–5.00 (m, 2H), 2.62–2.48 (m, 1H), 2.27–2.15 (m, 4H), 2.14 (d, J = 1.1 Hz, 3H), 1.81 (dd, J = 14.9 Hz, 7.1 Hz, 2H), 1.64 (s, 3H), 1.54 (s, 3H), 1.53 (s, 3H), 1.39–1.16 (m, 2H), 0.92 (d, J = 6.9 Hz, 3H); Isomer (2E,6E) δ: 9.95 (d, J = 8.1 Hz, 1H), 5.85 (dd, J = 8.1 Hz, 1.1 Hz, 1H), 5.10–5.00 (m, 2H), 2.28–2.15 (m, 4H), 2.14 (d, J = 1.1 Hz, 3H), 2.05 (dd, J = 14.5 Hz, 6.9 Hz, 1H), 1.81 (dd, J = 14.9 Hz, 7.1 Hz, 2H), 1.64 (s, 3H), 1.54 (s, 3H), 1.48 (s, 3H), 1.39–1.16 (m, 2H), 0.92 (d, J = 6.9 Hz, 3H); 13C-NMR (63 MHz, CDCl3) Isomer (2Z,6Z) δ: 190.5 (d), 163.6 (s), 141.0 (s), 131.2 (s), 128.5 (d), 124.5 (d), 122.8 (d), 34.7 (t), 33.4 (d), 32.8 (t), 26.2 (t), 26.1 (t), 25.5 (q), 24.9 (q), 19.1 (q), 17.8 (q), 17.5 (q); Isomer (2Z,6E) δ: 190.6 (d), 163.8 (s), 141.2 (s), 130.9 (s), 128.5 (d), 124.6 (d), 121.5 (d), 42.2 (d), 34.9 (t), 32.4 (t), 26.7 (t), 26.0 (t), 25.5 (q), 25.3 (q), 19.5 (q), 17.5 (q), 12.2 (q); Isomer (2E,6Z) δ: 191.0 (d), 163.6 (s), 140.4 (s), 131.1 (s), 127.3 (d), 124.5 (d), 123.0 (d), 40.8 (t), 34.7 (t), 33.4 (d), 26.1 (t), 25.5 (q), 24.9 (t), 19.1 (q), 17.8 (q), 17.5 (q), 17.4 (q); Isomer (2E,6E) δ: 191.0 (d), 163.6 (s), 140.5 (s), 130.9 (s), 127.3 (d), 124.6 (d), 121.9 (d), 42.2 (d), 40.5 (t), 34.9 (t), 26.0 (t), 25.5 (q), 25.3 (t), 19.6 (q), 17.5 (q), 17.4 (q), 12.1 (q); MS (70 eV) m/z (%) Isomer (2Z,6Z) 248 [M]+ (1), 82 (100), 69 (53), 95 (45), 41 (38), 109 (38), 55 (36), 67 (33), 81 (25), 83 (25), 149 (12), 121 (11), 137 (10), 167 (5), 205 (4); Isomer (2Z,6E) 248 [M]+ (1), 82 (100), 69 (45), 95 (30), 41 (29), 109 (29), 55 (28), 67 (27), 83 (21), 81 (20), 149 (8), 121 (7), 137 (6), 167 (5), 205 (2); Isomer (2E,6Z) 248 [M]+ (1), 82 (100), 69 (53), 95 (35), 41 (33), 109 (30), 67 (30), 55 (29), 81 (22), 83 (22) 149 (10), 121 (8), 137 (7), 167 (5), 205 (2); Isomer (2E,6E) 248 [M]+ (1), 82 (100), 69 (40), 41 (26), 67 (26), 95 (25), 55 (24), 109 (23), 83 (19), 81 (17), 167 (7), 149 (6), 121 (5), 205 (1). Anal. Calcd for: C17H28O: C, 82.20; H, 11.36. Found: C, 82.48; H, 11.32.
3.2.2. (6E)-3,8,12-Trimethyltrideca-2,6,11-trienal (19)
Column chromatography on silica gel (hexane:ethyl acetate 98:2) gave a yield of 71%. IR (ATR) 2965, 2915, 2853, 2764, 1673, 1633, 1449, 1377, 1193, 1123, 970, 841 cm−1; 1H-NMR (250 MHz, CDCl3) Isomer (2Z,6E) δ: 9.92 (d, J = 8.2 Hz, 1H), 5.88 (dd, J = 8.2 Hz, 1.2 Hz, 1H), 5.36–5.29 (m, 2H), 5.08 (dqq, J = 7.1, 1.1, 1.1 Hz, 1H), 2.63 (t, J = 7.5 Hz, 2H), 2.30–2.19 (m, 2H), 2.11–2.01 (m, 1H), 1.97 (d, J = 1.2 Hz, 3H), 1.96–1.86 (m, 2H), 1.67 (d, J = 1.1 Hz, 3H), 1.58 (s, 3H), 1.26 (dd, J = 15.3 Hz, 7.1 Hz, 2H), 0.94 (d, J = 6.7 Hz, 3H); Isomer (2E,6E) δ: 9.98 (d, J = 8.1 Hz, 1H), 5.87 (dd, J = 8.1 Hz, 1.2 Hz, 1H), 5.35-5.28 (m, 2H), 5.07 (dqq, J = 7.1, 1.0, 1.0 Hz, 1H), 2.32–2.18 (m, 4H), 2.15 (d, J = 1.2 Hz, 3H), 2.10–2.00 (m, 1H), 1.95–1.84 (m, 2H), 1.67 (d, J = 1.0 Hz, 3H), 1.57 (s, 3H), 1.25 (dd, J = 15.4 Hz, 7.1 Hz, 2H), 0.93 (d, J = 6.8 Hz, 3H); 13C-NMR (63 MHz, CDCl3) Isomer (2Z,6E) δ: 190.3 (d), 163.1 (s), 138.1 (d), 130.7 (s), 128.5 (d), 125.9 (d), 124.4 (d), 36.8 (t), 35.9 (d), 32.5 (t), 31.3 (t), 25.5 (t), 25.4 (q), 20.7 (q), 20.4 (q), 17.4 (q); Isomer (2E,6E) δ: 190.7 (d), 162.9 (s), 137.5 (d), 130.7 (s), 127.3 (d), 126.3 (d), 124.4 (d), 40.4 (t), 36.8 (t), 36.0 (d), 29.9 (t), 25.5 (t), 25.4 (q), 20.5 (q), 17.4 (q), 17.2 (q); MS (70 eV) m/z (%) Isomer (2Z,6E) 234 [M]+ (2), 69 (100), 82 (82), 41 (81), 81 (77), 95 (69), 84 (61), 55 (57), 67 (53), 107 (34), 109 (32), 135 (22), 123 (21), 121 (20), 150 (16), 219 (6); Isomer (2E,6E) 234 [M]+ (1), 69 (100), 84 (96), 81 (83), 82 (71), 41 (71), 95 (60), 55 (59), 67 (53), 107 (29), 109 (28), 150 (25), 151 (24), 135 (17), 121 (13). Anal. Calcd for: C16H26O: C, 81.99; H, 11.18. Found: C, 81.69; H, 11.15.
3.2.3. 3,7,8,12-Tetramethyltrideca-2,6-dienal (20)
Boiling point 120–124 °C/0.1 Torr. Yield 36%. Isolation by column chromatography gave a product with a yield of 83%. IR (ATR) 2955, 2926, 2868, 2775, 1675, 1456, 1381, 1192, 1121, 852 cm−1; 1H-NMR (250 MHz, CDCl3) Isomer (2Z,6Z) δ: 9.91 (d, J = 8.3 Hz, 1H), 5.87 (d, J = 8.3 Hz, 1H), 5.15–5.05 (m, 1H), 2.77–2.64 (m, 2H), 2.59 (t, J = 7.4 Hz, 2H), 2.30–2.17 (m, 2H), 1.97 (s, 3H), 1.57–1.40 (m, 1H), 1.54 (s, 3H), 1.30–1.06 (m, 6H), 0.93 (d, J = 6.9 Hz, 3H), 0.86–0.81 (m, 6H); Isomer (2Z,6E) δ: 9.92 (d, J = 8.3 Hz, 1H), 5.87 (d, J = 8.3 Hz, 1H), 5.15–5.05 (m, 1H), 2.59 (t, J = 7.4 Hz, 2H), 2.30–2.17 (m, 2H), 2.10–1.92 (m, 2H), 1.97 (s, 3H), 1.57–1.40 (m, 1H), 1.50 (s, 3H), 1.30–1.06 (m, 6H), 0.93 (d, J = 6.9 Hz, 3H), 0.86–0.81 (m, 6H); Isomer (2E,6Z) δ: 9.98 (d, J = 8.1 Hz, 1H), 5.85 (d, J = 8.1 Hz, 1H), 5.11–4.99 (m, 1H), 2.77–2.64 (m, 2H), 2.30–2.17 (m, 3H), 1.97 (s, 3H), 1.57–1.40 (m, 1H), 1.54 (s, 3H), 1.30–1.06 (m, 6H), 0.93 (d, J = 6.9 Hz, 3H), 0.86–0.81 (m, 6H); Isomer (2E,6E) δ: 9.98 (d, J = 8.1 Hz, 1H), 5.85 (d, J = 8.1 Hz, 1H), 5.11–4.99 (m, 1H), 2.30–2.17 (m, 3H), 2.10–1.92 (m, 2H), 1.97 (s, 3H), 1.57–1.40 (m, 1H), 1.50 (s, 3H), 1.30–1.06 (m, 6H), 0.93 (d, J = 6.9 Hz, 3H), 0.86–0.81 (m, 6H); 13C-NMR (63 MHz, CDCl3) Isomer (2Z,6Z) δ: 190.6 (d), 163.7 (s), 141.3 (s), 128.5 (d), 122.5 (d), 39.1 (t), 34.9 (t), 33.9 (d), 32.9 (t), 27.8 (d), 26.3 (t), 25.4 (t), 24.9 (q), 22.6 (q), 22.5 (q), 19.2 (q), 17.9 (q); Isomer (2Z,6E) δ: 190.7 (d), 163.9 (s), 141.5 (s), 128.5 (d), 121.2 (d), 42.6 (d), 39.0 (t), 35.0 (t), 32.5 (t), 27.8 (d), 26.7 (t), 25.2 (t), 24.9 (q), 22.6 (q), 22.5 (q), 19.5 (q), 12.5 (q); Isomer (2E,6Z) δ: 191.1 (d), 163.7 (s), 140.7 (s), 127.3 (d), 122.8 (d), 40.9 (t), 39.1 (t), 34.8 (t), 33.9 (d), 27.8 (d), 25.4 (t), 25.0 (t), 22.6 (q), 22.5 (q), 19.2 (q), 17.9 (q), 17.4 (q); Isomer (2E,6E) δ: 191.1 (d), 163.8 (s), 140.8 (s), 127.3 (d), 122.6 (d), 42.6 (d), 40.5 (t), 38.9 (t), 35.0 (t), 27.8 (d), 25.3 (t), 25.2 (t), 22.6 (q), 22.5 (q), 19.7 (q), 17.4 (q), 12.2 (q); MS (70 eV) m/z (%) Isomer (2Z,6Z) 250 [M]+ (1), 69 (100), 55 (87), 95 (70), 109 (58), 81 (47), 82 (47), 83 (47), 41 (43), 137 (43), 43 (41), 123 (20), 157 (12), 166 (11), 147 (11), 235 (3); Isomer (2Z,6E) 250 [M]+ (4), 69 (100), 55 (62), 111 (36), 83 (36), 81 (35), 97 (35), 41 (28), 43 (25), 165 (17), 137 (15), 221 (4); Isomer (2E,6Z) 250 [M]+ (1), 69 (100), 55 (84), 95 (64), 109 (51), 81 (46), 83 (46), 137 (39), 41 (39), 43 (37), 123 (15), 157 (12), 165 (11), 147 (10), 235 (3); Isomer (2E,6E) 250 [M]+ (2), 69 (100), 55 (70), 111 (38), 97 (34), 83 (34), 81 (30), 57 (30), 41 (28), 43 (25), 137 (14), 165 (11), 221 (3). Anal. Calcd for: C17H30O: C, 81.54; H, 12.07. Found: C, 81.96; H, 12.15.
3.2.4. 3,5,9-Trimethyldeca-2,8-dienal (21)
B.P. 90–92 °C/0.17 Torr. Yield 54%. IR (ATR) 2964, 2918, 2853, 2767, 1671, 1629, 1449, 1378, 1195, 1122, 867 cm−1; 1H-NMR (250 MHz, CDCl3) Isomer Z δ: 9.91 (d, J = 8.3 Hz, 1H), 5.83 (dd, J = 8.3 Hz, 1.2 Hz, 1H), 5.05 (dqq, J = 7.1, 1.3, 1.3 Hz, 1H), 2.45 (d, J = 7.4 Hz, 2H), 2.01 (dd, J = 16.0 Hz, 7.1 Hz, 3H), 1.92 (d, J = 1.2 Hz, 3H), 1.73 (dd, J = 13.3 Hz, 6.8 Hz, 1H), 1.66 (d, J = 1.3 Hz, 3H), 1.58 (s, 3H), 1.43–1.12 (m, 2H), 0.89 (d, J = 6.8 Hz, 3H); Isomer E δ: 9.96 (d, J = 8.1 Hz, 1H), 5.83 (dd, J = 8.1 Hz, 1.2 Hz, 1H), 5.05 (dqq, J = 7.1, 1.3, 1.3 Hz, 1H), 2.21 (dd, J = 13.3 Hz, 6.1 Hz, 1H), 2.10 (d, J = 1.2 Hz, 3H), 2.02–1.90 (m, 3H), 1.72 (dd, J = 13.4 Hz, 6.7 Hz, 1H), 1.66 (d, J = 1.3 Hz, 3H), 1.58 (s, 3H), 1.40–1.05 (m, 2H), 0.84 (d, J = 6.7 Hz, 3H); 13C-NMR (63 MHz, CDCl3) Isomer Z δ: 190.8 (d), 163.5 (s), 131.6 (s), 129.7 (d), 124.0 (d), 39.8 (t), 36.9 (t), 31.5 (d), 25.6 (q), 25.4 (t), 25.1 (q), 19.3 (q), 17.6 (q); Isomer E δ: 191.1 (d), 163.2 (s), 131.5 (s), 128.6 (d), 124.1 (d), 48.5 (t), 36.8 (t), 30.5 (d), 25.6 (q), 25.3 (t), 19.3 (q), 17.6 (q), 17.3 (q); MS (70 eV) m/z (%) Isomer Z 194 [M]+ (3), 109 (100), 95 (88), 69 (87), 41 (83), 55 (56), 111 (49), 151 (43), 67 (37), 81 (35), 84 (35), 43 (31), 123 (28), 138 (25), 179 (18); Isomer E 194 [M]+ (3), 69 (100), 41 (77), 95 (69), 109 (67), 55 (53), 111 (50), 67 (32), 84 (30), 179 (25), 81 (25), 107 (24), 123 (23), 137 (22), 151 (16). Anal. Calcd for: C13H22O: C, 80.35; H, 11.41. Found: C, 79.96; H, 11.49.
3.2.5. 3,6,10-Trimethylundeca-2,9-dienal (22)
B.P. 90–94 °C/0.04 Torr. Yield 46%. IR (ATR) 2961, 2917, 2855, 2764, 1673, 1633, 1448, 1378, 1129, 839 cm−1; 1H-NMR (250 MHz, CDCl3) Isomer Z δ: 9.96 (d, J = 8.3 Hz, 1H), 5.83 (dd, J = 8.3 Hz, 1.2 Hz, 1H), 5.08 (t, J = 7.1 Hz, 1H), 2.56 (t, J = 7.4 Hz, 2H), 2.02-1.92 (m, 2H), 1.97 (d, J = 1.2 Hz, 3H), 1.68 (s, 3H), 1.60 (s, 3H), 1.50–1.42 (m, 1H), 1.40–1.32 (m, 2H), 1.20–1.13 (m, 2H), 0.93 (d, J = 6.6 Hz, 3H); Isomer E δ: 9.98 (d, J = 8.1 Hz, 1H), 5.85 (dd, J = 8.1 Hz, 1.1 Hz, 1H), 5.08 (t, J = 7.1 Hz, 1H), 2.22–2.16 (m, 2H), 2.10 (d, J = 1.1 Hz, 3H), 1.98 (dd, J = 14.5 Hz, 7.1 Hz, 2H), 1.68 (s, 3H), 1.60 (s, 3H), 1.50–1.42 (m, 1H), 1.40–1.32 (m, 2H), 1.20–1.13 (m, 2H), 0.95 (d, J = 6.5 Hz, 3H); 13C-NMR (63 MHz, CDCl3) Isomer Z δ: 190.8 (d), 165.7 (s), 131.3 (s), 127.9 (d), 124.4 (d), 36.6 (t), 36.2 (t) 32.4 (d), 30.2 (t), 25.6 (q), 25.3 (t), 25.0 (q), 19.3 (q), 17,5 (q); Isomer E δ: 190.7 (d), 165.3 (s), 130.9 (s), 127.9 (d), 124.8 (d), 36.7 (t), 36.3 (t), 36.0 (t), 32.7 (d), 25.6 (q), 25.4 (t), 19.2 (q), 17.5 (q), 17.5 (q); MS (70 eV) m/z (%) Isomer Z: 208 [M]+ (4), 69 (100), 97 (90), 41 (81), 84 (67), 109 (64), 95 (57), 55 (47), 82 (35), 67 (34), 123 (27), 193 (21); Isomer E: 208 [M]+ (8), 69 (100), 97 (97), 41 (77), 84 (52), 55 (49), 109 (48), 95 (44), 67 (30), 81 (28), 123 (26), 193 (13). Anal. Calcd for: C14H24O: C, 80.71; H, 11.61. Found: C, 80.62; H, 11.71.
3.2.6. 3,4,6,10-Tetramethylundeca-2,9-dienal (23)
Boiling point 115–117 °C/0.16 Torr. Yield 67%. IR (ATR) 2964, 2916, 2873, 2851, 2756, 1672, 1632, 1455, 1378, 1198, 1135, 858 cm−1; 1H-NMR (250 MHz, CDCl3) Z Isomers δ: 10.06 (d, J = 8.3 Hz, 1H), 5.83 (d, J = 8.3 Hz, 1H), 5.13–5.00 (m, 1H), 3.63–3.50 (m, 1H), 2.04–1.89 (m, 2H), Racemate 1 and 2: 1.87 and 1.85 (d, J = 1.2 Hz, 3H), 1.67 (s, 3H), 1.59 (s, 3H), 1.55–1.22 (m, 5H), Racemate 1 and 2: 1.12 and 1.07 (d, J = 6.7 Hz, 3H), 0.91–0.86 (m, 3H); E Isomers δ: 10.01 (d, J = 8.0 Hz, 1H), 5.89 (dd, J = 8.0 Hz, 1.2 Hz, 1H), 5.06 (dqq, J = 7.0, 1.2, 1.2 Hz, 1H), 2.38 (m, 1H), Raemate 4: 2.12 (d, J = 1.2 Hz, 3H), Racemate 3: 2.09 (d, J = 1.2 Hz, 3H), 2.03–1.90 (m, 2H), 1.67 (d, J = 1.2 Hz, 3H), 1.59 (d, J = 1.2 Hz, 3H), 1.52–1.15 (m, 5H), Racemate 3: 1.07 (d, J = 6.8 Hz, 3H), Racemate 4: 1.85 (d, J = 6.8 Hz, 3H), 0.87 (d, J = 6.3 Hz, 3H); 13C-NMR (63 MHz, CDCl3) Z Racemate 1 and 2 δ: 189.9 (d), 189.8 (d), 168.9 (s), 168.4 (s), 131.3 (s), 131.2 (s), 129.1 (d), 128.4 (d), 124.6 (d), 124.4 (d), 42.3 (t), 41.8 (t), 37.5 (t), 36.7 (t), 32.3 (d), 30.2 (d), 30.2 (d), 25.6 (q), 25.3 (t), 19.8 (q), 19.4 (q), 19.2 (q), 19.0 (q), 18.6 (q), 18.6 (q), 17.6 (q); E Racemate 3 δ: 191.6 (d), 168.5 (s), 131.3 (s), 127.0 (d), 124.5 (d), 41.9 (t), 41.2 (d), 37.3 (t), 30.1 (d), 25.6 (q), 25.3 (t), 19.7 (q), 19.5 (q), 17.6 (q), 13.8 (q); E Racemate 4 δ: 191.6 (d), 169.1 (s), 131.3 (s), 126.5 (d), 124.5 (d), 42.2 (t), 41.1 (d), 36.9 (t), 30.0 (d), 25.6 (q), 25.2 (t), 19.6 (q), 18.7 (q), 17.6 (q), 14.6 (q); MS (70 eV) m/z (%) Racemate 1 (Z) 222 [M]+ (2), 69 (100), 41 (77), 109 (70), 111 (60), 55 (47), 95 (44), 85 (41), 83 (40), 98 (36), 43 (33), 149 (24), 152 (30), 123 (19), 166 (10), 207 (6); Racemate 2 (Z) 222 [M]+ (3), 69 (100), 41 (87), 111 (77), 109 (70), 55 (50), 98 (47), 95 (47), 83 (41), 152 (40), 43 (33), 67 (32), 149 (26), 123 (19), 166 (12), 207 (7); Racemate 3 (E) 222 [M]+ (3), 69 (100), 111 (91), 41 (77), 109 (60), 98 (43), 55 (42), 95 (37), 83 (32), 152 (29), 81 (28), 67 (27), 43 (21), 149 (19), 123 (14), 207 (6); Racemate 4 (E) 222 [M]+ (4), 69 (100), 111 (78), 41 (77), 109 (60), 98 (39), 55 (39), 95 (34), 83 (30), 81 (27), 67 (26), 152 (24), 43 (21), 149 (20), 123 (14), 166 (6). Anal. Calcd for: C15H26O: C, 81.02; H, 11.79. Found: C, 80.94; H, 11.65.
3.3. Sodium Borohydride Reduction of Aldehydes Structurally Related to Farnesol (24–29)
A total of 0.11 g (3 mmol) of sodium borohydride was added portion-wise to the stirred solution of 5.5 mmol of appropriate aldehyde (18–23) dissolved in 10 mL of isopropanol. After addition, the reaction mixture was stirred for 24 h. Next isopropanol was removed by evaporation and the residue was transferred to the separatory funnel, after which 20 mL of water and 30 mL of hexane was added. After separation of the organic layer, the water layer was extracted with hexane (2 × 20 mL). The combined extracts were washed with brine and dried over anhydrous magnesium sulphate. The product was purified by column chromatography on silica gel using hexane:ethyl acetate (97:3), or distilled (see below).
3.3.1. 3,7,8,12-Tetramethyltrideca-2,6,11-trien-1-ol (24)
Yield 87%. IR (ATR) 3306, 2962, 2919, 2856, 1667, 1448, 1376, 1001, 829 cm−1. 1H-NMR (250 MHz, CDCl3) Isomer (2Z,6Z) and (2Z,6E) δ: 5.58–5.39 (m, 1H), 5.15–5.00 (m, 2H), 4.11 (d, J = 7.1 Hz, 2H), 2.18–1.98 (m, 5H), 1.85 (dd, J = 14.7 Hz, 7.2 Hz, 2H), 1.75 (d, J = 1.3 Hz, 3H), 1.67 (d, J = 0.9 Hz, 3H), 1.58 (s, 3H), 1.51 (d, J = 1.0 Hz, 3H), 1.40–1.15 (m, 2H), 0.96 (d, J = 6.9 Hz, 3H); Isomer (2E,6Z) and (2E,6E) δ: 5.45–5.37 (m, 1H), 5.15–5.05 (m, 2H), 4.14 (d, J = 6.9 Hz, 2H), 2.16–2.00 (m, 5H), 1.85 (dd, J = 14.7 Hz, 7.1 Hz, 2H), 1.68 (s, 6H), 1.58 (s, 3H), 1.51 (d, J = 0.9 Hz, 3H), 1.42–1.21 (m, 2H), 0.96 (d, J = 6.9 Hz, 3H); 13C-NMR (63 MHz, CDCl3) Isomer (2Z, 6Z) δ: 140.0 (s), 139.6 (s), 131.2 (s), 124.7 (d), 124.4 (d), 124.4 (d), 58.9 (t), 34.9 (t), 33.5 (d), 32.3 (t), 26.2 (t), 25.7 (t), 25.6 (q), 23.4 (q), 19.2 (q), 17.9 (q), 17.6 (q); Isomer (2Z,6E) δ: 139.8 (s), 139.8 (s), 131.0 (s), 124.8 (d), 124.3 (d), 123.0 (d), 58.9 (t), 42.3 (d), 35.0 (t), 32.0 (t), 26.3 (t), 26.1 (t), 25.6 (q), 23.4 (q), 19.6 (q), 17.6 (q), 12.2 (q); Isomer (2E,6Z) δ: 139.6 (s), 139.4 (s), 131.1 (s), 124.8 (d), 124.4 (d), 123.4 (d), 59.2 (t), 39.9 (t), 34.9 (t), 33.5 (d), 26.2 (t), 25.7 (t), 25.6 (q), 19.2 (q), 17.9 (q), 17.6 (q), 16.2 (q); Isomer (2E,6E) δ: 139.6 (s), 139.4 (s), 131.0 (s), 124.8 (d), 123.4 (d), 123.3 (d), 59.2 (t), 42.3 (d), 39.5 (t), 35.0 (t), 26.1 (t), 26.0 (t), 25.6 (q), 19.7 (q), 17.6 (q), 16.2 (q), 12.2 (q); MS (70 eV) m/z (%) 250 [M]+ (1), 232 ([M]+-18, 2), 82 (100), 69 (69), 109 (40), 95 (33), 81 (27), 41 (26), 55 (25), 83 (25), 121 (16), 168 (4), 217 (2). Anal. Calcd for: C17H30O: C, 81.54; H, 12.07. Found: C, 81.46; H, 12.02.
3.3.2. (6E)-3,8,12-Trimethyltrideca-2,6,11-trien-1-ol (25)
Yield 74%. IR (ATR): 3340, 2965, 2916, 2855, 1670, 1450, 1377, 997, 968, 911, 733 cm−1. 1H-NMR (250 MHz, CDCl3) Isomer (2Z,6E) δ: 5.47–5.38 (m, 1H), 5.35–5.20 (m, 2H), 5.12–5.04 (m, 1H), 4.10 (d, J = 7.2 Hz, 2H), 2.18–2.00 (m, 5H), 1.92 (dd, J = 15.3 Hz, 7.3 Hz, 2H), 1.73 (d, J = 1.0 Hz, 3H), 1.67 (s, 3H), 1.58 (s, 3H), 1.26 (dd, J = 15.4 Hz, 7.3 Hz, 2H), 0.94 (d, J = 6.7 Hz, 3H); Isomer (2E,6E) δ: 5.44–5.38 (m, 1H), 5.34–5.18 (m, 2H), 5.12–5.04 (m, 1H), 4.12 (d, J = 7.1 Hz, 2H), 2.15–2.00 (m, 5H), 1.92 (dd, J = 15.3 Hz, 7.2 Hz, 2H), 1.67 (s, 3H), 1.66 (d, J = 0.9 Hz, 3H), 1.58 (s, 3H), 1.25 (dd, J = 14.8 Hz, 7.2 Hz, 2H), 0.94 (d, J = 6.7 Hz, 3H); 13C-NMR (63 MHz, CDCl3) Isomer (2Z,6E) δ: 138.7 (s), 136.7 (d), 130.8 (s), 127.5 (d), 124.6 (d), 124.5 (d), 58.6 (t), 37.0 (t), 36.1 (d), 32.0 (t), 31.0 (t), 25.6 (t), 25.5 (q), 23.2 (q), 20.6 (q), 17.4 (q); Isomer (2E,6E) δ: 138.5 (s), 136.4 (d), 130.8 (s), 127.7 (d), 124.6 (d), 123.6 (d), 58.8 (t), 39.5 (t), 37.0 (t), 36.1 (d), 30.7 (t), 25.6 (t), 25.5 (q), 20.7 (q), 17.4 (q), 16.0 (q); MS (70 eV) m/z (%) Isomer (2Z,6E) 236 [M]+ (0), 218 ([M]+-18, 1), 69 (100), 41 (51), 82 (47), 81 (45), 55 (36), 95 (34), 67 (30), 93 (26), 107 (21), 109 (17); Isomer (2E,6E) 236 [M]+ (0), 218 ([M]+-18, 1), 69 (100), 82 (66), 41 (59), 81 (58), 95 (50), 55 (42), 67 (38), 93 (27), 107 (26), 109 (24), 135 (14). Anal. Calcd for: C16H28O: C, 81.29; H, 11.94. Found: C, 81.12; H, 11.89.
3.3.3. 3,7,8,12-Tetramethyltrideca-2,6-dien-1-ol (26)
Yield 95%. IR (ATR) 3309, 2955, 2925, 2869, 1667, 1456, 1380, 1368, 1092, 1002, 955, 836 cm−1. 1H-NMR (250 MHz, CDCl3) Isomer (2Z,6Z) and (2Z,6E) δ: 5.48–5.38 (m, 1H), 5.14–5.04 (m, 1H), 4.11 (d, J = 7.1 Hz, 2H), 2.13–2.00 (m, 5H), 1.75 (d, J = 1.0 Hz, 3H), 1.59–1.46 (m, 1H), 1.51 (d, J = 1.2 Hz, 3H), 1.35–1.10 (m, 6H), 0.95 (d, J = 6.9 Hz, 3H), 0.86 (d, J = 6.6 Hz, 6H); Isomer (2E,6Z) and (2E,6E) δ: 5.46–5.38 (m, 1H), 5.10 (t, J = 5.7 Hz, 1H), 4.15 (d, J = 7.0 Hz, 2H), 2.18–1.97 (m, 5H), 1.68 (d, J = 0.6 Hz, 3H), 1.62–1.44 (m, 1H), 1.50 (d, J = 1.2 Hz, 3H), 1.33–1.05 (m, 6H), 0.95 (d, J = 6.9 Hz, 3H), 0.85 (d, J = 6.6 Hz, 3H), 0.84 (d, J = 6.6 Hz, 3H); 13C-NMR (63 MHz, CDCl3) Isomer (2Z,6Z) δ: 140.3 (s), 139.7 (s), 124.5 (d), 124.2 (d), 58.9 (t), 39.2 (t), 35.0 (t), 33.9 (d), 32.3 (t), 27.9 (d), 25.8 (t), 25.5 (t), 23.4 (q), 22.7 (q), 22.6 (q), 19.4 (q), 17.9 (q); Isomer (2Z,6E) δ: 140.2 (s), 139.9 (s), 124.3 (d), 122.7 (d), 59.0 (t), 42.7 (d), 39.1 (t), 35.1 (t), 32.0 (t), 27.9 (d), 26.3 (t), 25.3 (t), 23.4 (q), 22.7 (q), 22.6 (q), 19.7 (q), 12.2 (q); Isomer (2E,6Z) δ: 139.7 (s), 139.6 (s), 124.2 (d), 123.3 (d), 62.3 (t), 39.9 (t), 39.2 (t), 35.0 (t), 33.9 (d), 27.9 (d), 25.6 (t), 25.5 (t), 22.6 (q), 22.6 (q), 19.4 (q), 17.9 (q), 16.2 (q); Isomer (2E,6E) δ: 149.7 (s), 139.6 (s), 123.3 (d), 123.0 (d), 62.3 (t), 42.7 (d), 39.6 (t), 39.1 (t), 35.1 (t), 27.9 (d), 26.1 (t), 25.2 (t), 22.7 (q), 22.6 (q), 19.8 (q), 16.2 (q), 12.1 (q); MS (70 eV) m/z (%) Isomer (2Z,6Z) 252 [M]+ (4), 69 (100), 55 (74), 111 (44), 83 (41), 84 (39), 97 (37), 57 (33), 93 (32), 41 (30), 43 (28), 121 (24), 221 (4); Isomer (2Z,6E) 252 [M]+ (3), 69 (100), 55 (67), 111 (54), 83 (36), 97 (35), 57 (34), 41 (28), 43 (25), 81 (25), 93 (24), 121 (15), 221 (4); Isomer (2E,6Z) 252 [M]+ (3), 69 (100), 55 (75), 111 (50), 83 (41), 97 (40), 57 (36), 84 (33), 93 (31), 41 (31), 43 (28), 121 (22), 221 (3); Isomer (2E,6E) 252 [M]+ (2), 69 (100), 55 (61), 111 (53), 83 (34), 97 (32), 57 (31), 41 (24), 43 (21), 81 (21), 93 (18), 121 (11), 221 (3). Anal. Calcd for: C17H32O: C, 80.89; H, 12.78. Found: C, 81.03; H, 12.85.
3.3.4. 3,5,9-Trimethyldeca-2,8-dien-1-ol (27)
Boiling point 94–96 °C/0.5 Torr. Yield 69%. IR (ATR) 3312, 2964, 2913, 2872, 1668, 1449, 1377, 1063, 996, 889 cm−1; 1H-NMR (250 MHz, CDCl3) Isomer Z: δ: 5.42–5.34 (m, 1H), 5.14–5.04 (m, 1H), 4.15 (d, J = 6.9 Hz, 2H), 2.08–1.97 (m, 4H), 1.70 (d, J = 1.2 Hz, 3H), 1.68 (d, J = 1.1 Hz, 3H), 1.65–1.52 (m, 1H), 1.60 (s, 3H), 1.40–1.24 (m, 1H), 1.18–1.04 (m, 1H), 0.82 (d, J = 6.6 Hz, 3H); Isomer E: δ: 5.52–5.43 (m, 1H), 5.14–5.04 (m, 1H), 4.11 (d, J = 7.1 Hz, 2H), 2.09–1.99 (m, 2H), 1.92 (dd, J = 13.1 Hz , 8.3 Hz, 1H), 1.78 (dd, J = 13.1 Hz, 8.3 Hz, 1H), 1.68 (d, J = 1.1 Hz, 3H), 1.65–1.52 (m, 1H), 1.64 (s, 3H), 1.60 (s, 3H), 1.40–1.24 (m, 1H), 1.18–1.04 (m, 1H), 0.83 (d, J = 6.5 Hz, 3H); 13C-NMR (63 MHz, CDCl3) Isomer Z: δ: 137.7 (s), 130.8 (s), 125.5 (d), 125.0 (d), 58.6 (t), 39.3 (t), 36.8 (t), 30.6 (d), 25.4 (q), 25.3 (t), 23.4 (q), 19.1 (q), 17.3 (q); Isomer E: δ: 137.4 (s), 130.7 (s), 124.6 (d), 124.5 (d), 58.7 (t), 47.4 (t), 36.7 (t), 30.0 (d), 25.4 (q), 25.3 (t), 19.1 (q), 17.3 (q), 15.7 (q); MS (70 eV) m/z (%) Isomer Z: 196 [M]+ (2), 69 (100), 109 (66), 41 (63), 95 (62), 55 (46), 111 (42), 82 (42), 67 (40), 107 (36), 81 (28), 93 (25), 163 (15); Isomer E: 196 [M]+ (2), 69 (100), 41 (65), 95 (64), 109 (63), 82 (55), 55 (41), 67 (38), 81 (26), 107 (23), 111 (20). Anal. Calcd for: C13H24O: C, 79.53; H, 12.32. Found: C, 79.44; H, 12.36.
3.3.5. 3,6,10-Trimethylundeca-2,9-dien-1-ol (28)
Yield 97%. IR (ATR) 3319, 2963, 2915, 2855, 1669, 1452, 1377, 1114, 1075, 998, 954 cm−1; 1H-NMR (700 MHz, CDCl3) Isomer Z: δ: 5.39 (t, J = 6.9 Hz, 1H), 5.09 (t, J = 6.9 Hz, 1H), 4.12 (d, J = 6.9 Hz, 1H), 2.10–2.01 (m, 2H), 1.99 (dd, J = 14.8, 6.9 Hz, 1H), 1.96–1.89 (m, 1H), 1.73 (s, 3H), 1.68 (s, 3H), 1.60 (s, 3H), 1.40 (dd, J = 12.3, 6.5 Hz, 1H), 1.38–1.35 (m, 1H), 1.34–1.30 (m, 1H), 1.21–1.18 (m, 1H), 1.17–1.12 (m, 1H), 0.89 (d, J = 6.5 Hz, 3H); Isomer E: δ: 5.43–5.39 (m, 1H), 5.12–5.08 (m, 1H), 4.14 (s. 2H), 2.07–2.02 (m, 1H), 2.01–1.96 (m, 2H), 1.93 (dd, J = 14.5, 7.1 Hz, 1H), 1.68 (d, J = 1.2 Hz, 3H), 1.67 (s, 3H), 1.60 (s, 3H), 1.46–1.41 (m, 1H), 1.39 (dd, J = 12.8, 6.6 Hz, 1H), 1.36–1.31 (m, 1H), 1.26–1.20 (m, 1H), 1.18–1.12 (m, 1H), 0.88 (d, J = 6.6 Hz, 3H); 13C-NMR (176 MHz, CDCl3) Isomer Z: δ: 140.4 (s), 131.0 (s), 124.7 (d), 123.8 (d), 58.8 (t), 36.8 (t), 35.6 (t), 32.4 (d), 29.4 (t), 25.6 (q), 25.4 (t), 23.4 (q), 19.4 (q), 17.5 (q); Isomer E: δ: 140.0 (s), 130.9 (s), 124.8 (d), 123.0 (d), 59.2 (t), 37.0 (t), 36.9 (t), 34.9 (t), 32.1 (d), 25.6 (q), 25.4 (t), 19.4 (q), 17.5 (q), 16.1 (q); MS (70 eV) m/z (%) Isomer Z: 210 [M]+ (3), 69 (100), 109 (65), 81 (59), 41 (59), 55 (44), 82 (43), 107 (37), 67 (35), 84 (33), 177 (12); Isomer E: 210 [M]+ (1), 69 (100), 109 (77), 81 (67), 41 (58), 82 (40), 55 (40), 67 (32), 95 (27), 71 (25), 107 (22). Anal. Calcd for: C14H26O: C, 79.94; H, 12.46. Found: C, 80.11; H, 12.53.
3.3.6. 3,4,6,10-Tetramethylundeca-2,9-dien-1-ol (29)
Yield 49%. IR (ATR) 3322, 2963, 2914, 2872, 1664, 1455, 1377, 1161, 1129, 1028, 998, 953, 818 cm−1; 1H-NMR (250 MHz, CDCl3) Z Isomers δ: 5.35 (td, J = 6.9 Hz, 1.3 Hz, 1H), 5.12-5.02 (m, 1H), 4.18–4.11 (m, 2H), 2.75 (dd, J = 14.3 Hz, 6.8 Hz, 1H), 2.05–1.85 (m, 2H), 1.67 (d, J = 1.4 Hz, 3H), 1.62 (d, J = 1.3 Hz, 3H), 1.59 (s, 3H), 1.42–1.10 (m, 5H), Racemate 2: 0.97 (d, J = 6.8 Hz, 3H), Racemate 1: 0.94 (d, J = 6.8 Hz, 3H), 0.86 (d, J = 6.2 Hz, 3H); E Isomers δ: 5.41 (td, J = 6.8 Hz, 0.6 Hz, 1H), 5.13–5.03 (m, 1H), 4.14 (d, J = 6.8 Hz, 2H), 2.31–2.16 (m, 1H), 2.02–1.88 (m, 2H), 1.67 (s, 3H), 1.59 (s, 3H), 1.56 (d, J = 0.6 Hz, 3H), 1.42–1.10 (m, 5H), Racemate 3: 0.97 (d, J = 6.8 Hz, 3H), Racemate 4: 0.95 (d, J = 6.8 Hz, 3H), 0.84 (d, J = 6.2 Hz, 3H); 13C-NMR (63 MHz, CDCl3) Racemate 1 (Z) δ: 144.3 (s), 131.1 (s), 124.8 (d), 123.8 (d), 58.5 (t), 42.4 (t), 37.0 (t), 31.6 (d), 30.1 (d), 25.6 (q), 25.4 (t), 19.9 (q), 19.2 (q), 18.1 (q), 17.6 (q); Racemate 2 (Z) δ: 143.5 (s), 131.0 (s), 124.8 (d), 124.4 (d), 58.6 (t), 41.9 (t), 37.8 (t), 31.6 (d), 30.2 (d), 25.6 (q), 25.4 (t), 20.1 (q), 19.6 (q), 17.8 (q), 17.6 (q); Racemate 3 (E) δ: 143.5 (s), 131.0 (s), 124.9 (d), 123.2 (d), 59.2 (t), 42.1 (t), 40.0 (d), 37.5 (t), 30.0 (d), 25.6 (q), 25.4 (t), 20.2 (q), 19.6 (q), 17.6 (q), 11.9 (q); Racemate 4 (E) δ: 144.3 (s), 131.0 (s), 124.9 (d), 123.6 (d), 59.2 (t), 42.5 (t), 39.8 (d), 37.0 (t), 29.9 (d), 25.6 (q), 25.3 (t), 19.7 (q), 19.2 (q), 17.6 (q), 12.8 (q); MS (70 eV) m/z (%) Racemate 1 and 2 (Z) 224 [M]+ (2), 69 (100), 82 (77), 109 (65), 81 (55), 41 (55), 95 (44), 55 (44), 67 (40) 98 (39), 43 (33) 123 (20), 135 (11), 151 (10), 191 (6); Racemate 3 and 4 (E) 224 [M]+ (4), 69 (100), 82 (71), 109 (61), 81 (60), 41 (50), 55 (40), 95 (39), 67 (37), 43 (33), 83 (24), 71 (19), 123 (16), 151 (15), 191 (6). Anal. Calcd for: C15H28O: C, 80.29; H, 12.58. Found: C, 80.17; H, 12.63.
3.4. Preparation of Compounds Structurally Related to Farnesyl Acetate (30–35)
To the stirred and cooled (ca. 0 °C) solution of 6.7 mmol of appropriate compound structurally related to farnesol (24–29) and 0.82 g (8 mmol) of acetic anhydride, 0.81 g (8 mmol) of triethylamine was added. After addition, the mixture was stirred for 2 h. Next, 2 mL of methanol was added and stirring was continued for another 0.5 h. Afterwards, 30 mL of hexane and 20 mL of water was added and the hexane layer was separated, washed with brine and dried over anhydrous magnesium sulphate. The obtained acetates were purified by column chromatography on silica with hexane: ethyl acetate (98:2) as the mobile phase. Purity of the products according to GC-MS was 92%–99%.
3.4.1. 3,7,8,12-Tetramethyltrideca-2,6,11-trien-1-yl acetate (30)
Yield 90%. IR (ATR) 2964, 2923, 2857, 1740, 1666, 1450, 1377, 1230, 1022, 953 cm−1. 1H-NMR (250 MHz, CDCl3) Isomer (2Z,6Z) and (2Z,6E) δ: 5.40–5.31 (m, 1H), 5.14–5.04 (m, 2H), 4.56 (d, J = 7.2 Hz, 2H), 2.15–2.02 (m, 5H), 2.05 (s, 3H), 1.85 (dd, J = 14.7 Hz, 7.3 Hz, 2H), 1.77 (d, J = 1.1 Hz, 3H), 1.67 (s, 3H), 1.58 (s, 3H), 1.51 (d, J = 1.0 Hz, 3H), 1.43–1.18 (m, 2H), 0.95 (d, J = 6.9 Hz, 3H); Isomer (2E,6Z) and (2E,6E) δ: 5.38–5.30 (m, 1H), 5.13–5.05 (m, 2H), 4.58 (d, J = 7.1 Hz, 2H), 2.17–2.00 (m, 5H), 2.05 (s, 3H), 1.85 (dd, J = 14.7 Hz, 7.1 Hz, 3H), 1.70 (s, 3H), 1.67 (s, 3H), 1.58 (s, 3H), 1.50 (d, J = 0.8 Hz, 3H), 1.44–1.16 (m, 2H), 0.95 (d, J = 6.9 Hz, 3H); 13C-NMR (63 MHz, CDCl3) Isomer (2Z,6Z) δ: 170.8 (s), 142.3 (s), 139.6 (s), 131.0 (s), 124.7 (d), 124.1 (d), 119.1 (d), 60.9 (t), 34.8 (t), 33.4 (d), 32.4 (t), 26.1 (t), 25.8 (t), 25.5 (q), 23.4 (q), 20.8 (q), 19.1 (q), 17.8 (q), 17.5 (q); Isomer (2Z,6E) δ: 170.8 (s), 142.4 (s), 139.7 (s), 130.8 (s), 124.8 (d), 122.8 (d), 119.1 (d), 60.9 (t), 42.3 (d), 35.0 (t), 32.0 (t), 26.3 (t), 26.0 (t), 25.5 (q), 23.3 (q), 20.8 (q), 19.6 (q), 17.5 (q), 12.1 (q); Isomer (2E,6Z) δ: 170.8 (s), 141.9 (s), 139.3 (s), 131.0 (s), 124.7 (d), 124.3 (d), 118.2 (d), 61.2 (t), 39.8 (t), 34.8 (t), 33.4 (d), 26.1 (t), 25.5 (q), 25.4 (t), 20.8 (q), 19.1 (q), 17.8 (q), 17.5 (q), 16.3 (q); Isomer (2E,6E) δ: 170.8 (s), 141.9 (s), 139.3 (s), 130.8 (s), 124.8 (d), 123.1 (d), 118.3 (d), 61.2 (t), 42.2 (d), 39.4 (t), 34.9 (t), 26.0 (t), 25.8 (t), 25.5 (q), 20.8 (q), 19.7 (q), 17.5 (q), 16.2 (q), 12.1 (q); MS (70 eV) m/z (%) Isomer (2Z,6Z) 292 [M]+ (0), 82 (100), 69 (56), 109 (32), 95 (27), 41 (24), 55 (24), 67 (24), 81 (24), 43 (21), 83 (20), 121 (15); Isomer (2Z,6E) and (2E,6Z) 292 [M]+ (0), 82 (100), 69 (57), 109 (30), 95 (27), 67 (23), 81 (23), 41 (22), 55 (22), 43 (21), 83 (20), 121 (12); Isomer (2E,6E) 292 [M]+ (0), 82 (100), 69 (52), 109 (29), 95 (26), 43 (23), 67 (22), 81 (21), 41 (19), 55 (18), 83 (17), 121 (11). Anal. Calcd for: C19H32O2: C, 78.03; H, 11.03. Found: C, 77.87; H, 11.08.
3.4.2. (E)-3,8,12-Trimethyltrideca-2,6,11-trien-1-yl acetate (31)
Yield 83%. IR (ATR) 2963, 2916, 2854, 1740, 1671, 1450, 1376, 1230, 1022, 968 cm−1; 1H-NMR (250 MHz, CDCl3) Isomer (2Z,6E) δ: 5.39–5.18 (m, 3H), 5.12–5.02 (m, 1H), 4.54 (d, J = 7.2 Hz, 2H), 2.19–1.97 (m, 5H), 1.92 (dd, J = 15.1 Hz, 7.3 Hz, 2H), 1.74 (d, J = 1.3 Hz, 3H), 1.66 (s, 3H), 1.57 (s, 3H), 1.25 (dd, J = 15.4 Hz, 7.3 Hz, 2H), 0.94 (d, J = 6.7 Hz, 3H); Isomer (2E,6E) δ: 5.40–5.18 (m, 3H), 5.12–5.03 (m, 1H), 4.56 (d, J = 7.0 Hz, 2H), 2.18–1.98 (m, 5H), 1.91 (dd, J = 15.1 Hz, 7.1 Hz, 2H), 1.68 (s, 3H), 1.67 (d, J = 0.9 Hz, 3H), 1.57 (s, 3H), 1.25 (dd, J = 15.4 Hz, 7.1 Hz, 2H), 0.93 (d, J = 6.7 Hz, 3H); 13C-NMR (63 MHz, CDCl3) Isomer (2Z,6E) δ: 169.9 (s), 141.0 (s), 136.4 (d), 130.2 (s), 126.9 (d), 124.3 (d), 119.2 (d), 60.3 (t), 36.7 (t), 35.8 (d), 31.7 (t), 30.6 (t), 25.3 (t), 25.1 (q), 22.8 (q), 20.2 (q), 20.1 (q), 17.0 (q); Isomer (2E,6E) δ: 169.8 (s), 140.7 (s), 136.2 (d), 130.1 (s), 127.2 (d), 124.3 (d), 118.4 (d), 60.5 (t), 39.1 (t), 36.7 (t), 35.8 (d), 30.2 (t), 25.3 (t), 25.1 (q), 20.4 (q), 20.1 (q), 17.0 (q), 15.7 (q); MS (70 eV) m/z (%) Isomer (2Z,6E) 278 [M]+ (1), 69 (100), 82 (70), 43 (65), 41 (59), 95 (54), 81 (53), 94 (52), 55 (48), 93 (46), 67 (45), 107 (40), 175 (15), 203 (7), 218 (5); Isomer (2E,6E) 278 [M]+ (1), 69 (100), 43 (83), 82 (66), 41 (62), 95 (56), 81 (53), 67 (44), 94 (43), 55 (42), 93 (41), 107 (40), 150 (20), 203 (10), 218 (4). Anal. Calcd for: C18H30O2: C, 77.65; H, 10.86. Found: C, 77.48; H, 10.97.
3.4.3. 3,7,8,12-Tetramethyltrideca-2,6-dien-1-yl acetate (32)
Yield 69%. IR (ATR) 2956, 2926, 2869, 1741, 1665, 1455, 1378, 1366, 1230, 1022, 954 cm−1; 1H-NMR (250 MHz, CDCl3) Isomer (2Z,6Z) and (2Z,6E) δ: 5.37–5.27 (m, 1H), 5.12–5.01 (m, 1H), 4.54 (d, J = 7.2 Hz, 2H), 2.11–1.98 (m, 5H), 2.01 (s, 3H), 1.73 (s, 3H), 1.55–1.40 (m, 1H), 1.47 (s, 3H), 1.32–1.06 (m, 6H), 0.92 (d, J = 6.9 Hz, 3H), 0.82 (d, J = 6.6 Hz, 6H); Isomer (2E,6Z) and (2E,6E) δ: 5.36–5.27 (m, 1H), 5.10–5.00 (m, 1H), 4.56 (d, J = 7.1 Hz, 2H), 2.15–1.95 (m, 5H), 2.03 (s, 3H), 1.68 (s, 3H), 1.56–1.41 (m, 1H), 1.47 (d, J = 1.1 Hz, 3H), 1.32–1.05 (m, 6H), 0.92 (d, J = 6.6 Hz, 3H), 0.83 (d, J = 6.6 Hz, 3H), 0.82 (d, J = 6.6 Hz, 3H); 13C-NMR (63 MHz, CDCl3) Isomer (2Z,6Z) δ: 170.9 (s), 142.4 (s), 139.9 (s), 123.9 (d), 119.2 (d), 61.0 (t), 39.2 (t), 35.0 (t), 33.9 (d), 32.5 (t), 27.8 (d), 25.9 (t), 25.5 (t), 23.4 (q), 22.6 (q), 22.5 (q), 20.9 (q), 19.3 (q), 17.9 (q); Isomer (2Z,6E) δ: 170.9 (s), 142.5 (s), 140.0 (s), 122.6 (d), 119.1 (d), 61.0 (t), 42.6 (d), 39.1 (t), 35.1 (t), 32.1 (t), 27.9 (d), 26.3 (t), 25.3 (t), 23.4 (q), 22.6 (q), 22.5 (q), 20.9 (q), 19.7 (q), 12.2 (q); Isomer (2E,6Z) δ: 170.9 (s), 142.1 (s), 139.7 (s), 124.1 (d), 118.3 (d), 61.3 (t), 39.9 (t), 39.2 (t), 35.0 (t), 33.9 (d), 27.9 (d), 25.5 (t), 25.5 (t), 22.6 (q), 22.5 (q), 20.9 (q), 19.3 (q), 17.9 (q), 16.3 (q); Isomer (2E,6E) δ: 170.9 (s), 142.1 (s), 139.7 (s), 122.8 (d), 118.3 (d), 61.3 (t), 42.6 (d), 39.5 (t), 39.1 (t), 35.1 (t), 27.9 (d), 25.9 (t), 25.3 (t), 22.6 (q), 22.5 (q), 20.9 (q), 19.7 (q), 16.3 (q), 12.2 (q); MS (70 eV) m/z (%) Isomer (2Z,6Z) 294 [M]+ (0), 69 (100), 55 (64), 80 (53), 93 (52), 43 (51), 121 (43), 111 (34), 83 (33), 97 (32), 149 (13), 234 (8), 166 (7); Isomer (2Z,6E) and (2E,6Z) 294 [M]+ (0), 69 (100), 55 (63), 43 (49), 93 (44), 80 (44), 111 (42), 121 (35), 97 (35), 83 (34), 57 (33), 149 (15), 166 (9), 234 (8); Isomer (2E,6E) 294 [M]+ (0), 69 (100), 55 (61), 43 (52), 111 (46), 93 (34), 83 (33), 97 (32), 57 (32), 121 (24), 149 (14), 166 (14), 234 (6). Anal. Calcd for: C19H34O2: C, 77.50; H, 11.64. Found: C, 77.38; H, 11.73.
3.4.4. 3,5,9-Trimethyldeca-2,8-dien-1-yl acetate (33)
Yield 82%. IR (ATR) 2966, 2916, 2849, 1740, 1668, 1453, 1378, 1229, 1022, 953 cm−1; 1H-NMR (700 MHz, CDCl3) Isomer Z: δ: 5.41 (t, J = 7.1 Hz, 1H), 5.10–5.06 (m, 1H), 4.56 (d, J = 7.1 Hz, 2H), 2.06–2.00 (m, 2H), 2.04 (s, 3H), 1.95 (dd, J = 13.3 Hz, 8.5 Hz, 2H), 1.73 (s, 3H), 1.68 (s, 3H), 1.63 (dd, J = 13.3 Hz, 6.5 Hz, 1H), 1.60 (s, 3H), 1.34–1.28 (m, 1H), 1.16–1.09 (m, 2H), 0.83 (d, J = 6.5 Hz, 3H); Isomer E: δ: 5.33–5.30 (m, 1H), 5.10–5.06 (m, 1H), 4.59 (d, J = 7.1 Hz, 2H), 2.05 (s, 3H), 2.01 (dd, J = 15.4 Hz, 7.1 Hz, 2H), 1.95 (dd, J = 13.3 Hz, 8.4 Hz, 1H), 1.80 (dd, J = 13.3 Hz, 8.4 Hz, 1H), 1.68 (s, 3H), 1.66 (s, 3H), 1.63 (dd, J = 13.3 Hz, 6.6 Hz, 1H), 1.60 (s, 3H), 1.34–1.28 (m, 1H), 1.15–1.08 (m, 2H), 0.82 (d, J = 6.6 Hz, 3H); 13C-NMR (176 MHz, CDCl3) Isomer Z: δ: 171.0 (s), 141.7 (s), 131.1 (s), 124.6 (d), 120.0 (d), 61.1 (t), 39.5 (t), 36.9 (t), 30.8 (d), 25.6 (q), 25.6 (t), 23.6 (q), 20.9 (q), 19.2 (q), 17.6 (q); Isomer E: δ: 171.0 (s), 141.2 (s), 131.1 (s), 124.7 (d), 119.7 (d), 61.3 (t), 47.5 (t), 36.8 (t), 30.1 (d), 25.6 (q), 25.4 (t), 20.9 (q), 19.3 (q), 17.5 (q), 16.1 (q); MS (70 eV) m/z (%) Isomer Z: 238 [M]+ (1), 69 (100), 95 (84), 109 (78), 107 (69), 43 (58), 41 (54), 82 (43), 55 (43), 67 (42), 163 (41), 93 (40), 81 (38), 122 (25), 135 (23), 178 (16); Isomer E: 238 [M]+ (1), 69 (100), 95 (81), 109 (71), 107 (66), 43 (60), 41 (50), 82 (46), 67 (42), 55 (39), 163 (38), 93 (38), 81 (33), 122 (22), 135 (20), 178 (16). Anal. Calcd for: C15H26O2: C, 75.58; H, 10.99. Found: C, 75.77; H, 10.83.
3.4.5. 3,6,10-Trimethylundeca-2,9-dien-1-yl acetate (34)
Yield 86%. IR (ATR) 2965, 2916, 2856, 1740, 1671, 1453, 1378, 1229, 1122, 853 cm−1; 1H-NMR (700 MHz, CDCl3) Isomer Z: δ: 5.34–5.31 (m, 1H), 5.11–5.08 (m, 1H), 4.56 (d, J = 7.1 Hz, 1H), 2.10–2.02 (m, 2H), 2.05 (s, 3H), 2.02–1.97 (m, 1H), 1.97–1.91 (m, 1H), 1.75 (s, 3H), 1.68 (s, 3H), 1.60 (s, 3H), 1.46–1.36 (m, 2H), 1.35–1.30 (m, 1H), 1.26–1.18 (m, 1H), 1.18–1.12 (m, 1H), 0.90 (d, J = 6.5 Hz, 3H); Isomer E: δ: 5.36–5.31 (m, 1H), 5.11–5.08 (m, 1H), 4.58 (d, J = 7.1 Hz, 1H), 2.09–2.03 (m, 1H), 2.05 (s, 3H), 2.02–1.97 (m, 2H), 1.96–1.90 (m, 1H), 1.69 (s, 3H), 1.68 (s, 3H), 1.60 (s, 3H), 1.46–1.41 (m, 1H), 1.39 (dd, J = 12.6, 6.5 Hz, 1H), 1.36–1.30 (m, 1H), 1.26–1.20 (m, 1H), 1.18–1.12 (m, 1H), 0.88 (d, J = 6.5 Hz, 3H); 13C-NMR (176 MHz, CDCl3) Isomer Z: δ: 170.9 (s), 143.4 (s), 131.0 (s), 124.7 (d), 118.5 (d), 60.9 (t), 36.9 (t), 35.4 (t), 32.4 (d), 29.6 (t), 25.6 (q), 25.4 (t), 23.4 (q), 20.9 (q), 19.4 (q), 17.5 (q); Isomer E: δ: 170.9 (s), 142.8 (s), 130.9 (s), 124.8 (d), 117.9 (d), 61.3 (t), 37.0 (t), 36.9 (t), 34.8 (t), 32.1 (d), 25.6 (q), 25.4 (t), 20.9 (q), 19.4 (q), 17.5 (q), 16.3 (q); MS (70 eV) m/z (%) Isomer Z: 252 [M]+ (1), 69 (100), 81 (72), 109 (68), 107 (60), 41 (52), 43 (51), 82 (50), 67 (39), 55 (36), 93 (31), 121 (28), 136 (25), 177 (23), 149 (20); Isomer E: 252 [M]+ (2), 69 (100), 81 (77), 109 (67), 43 (56), 107 (54), 41 (51), 82 (45), 67 (36), 55 (34), 93 (28), 121 (26), 136 (20), 149 (19), 177 (18). Anal. Calcd for: C16H28O2: C, 76.14; H, 11.18. Found: C, 76.31; H, 11.23.
3.4.6. 3,4,6,10-Tetramethylundec-2,9-dien-1-yl acetate (35)
Yield 91%. IR (ATR) 2956, 2915, 2869, 1741, 1665, 1455, 1375, 1230, 1022, 953 cm−1; 1H-NMR (250 MHz, CDCl3) Racemate 1 and 2 (Z) δ: 5.34–5.25 (m, 1H), 5.12–5.02 (m, 1H), 4.62–4.54 (m, 2H), 2.83–2.71 (m, 1H), 2.03 (s, 3H), 2.00–1.86 (m, 2H), 1.67 (s, 3H), 1.64 (d, J = 1.4 Hz, 3H), 1.59 (s, 3H), 1.47–1.06 (m, 5H), Racemate 2: 0.96 (d, J = 6.9 Hz, 3H), Racemate 1: 0.94 (d, J = 6.9 Hz, 3H), 0.85 (t, J = 6.4 Hz, 3H); Racemate 3 and 4 (E) δ: 5.34 (t, J = 6.9 Hz, 1H), 5.12–5.02 (m, 1H), 4.58 (d, J = 6.9 Hz, 2H), 2.33–2.18 (m, 1H), 2.04 (s, 3H), 2.00–1.86 (m, 2H), 1.67 (s, 3H), 1.61 (d, J = 0.6 Hz, 3H), 1.59 (s, 3H), 1.47–1.06 (m, 5H), Racemate 3: 0.97 (d, J = 6.9 Hz, 3H), 0.84 (d, J = 6.3 Hz, 3H Racemate 4: 0.96 (d, J = 6.9 Hz, 3H), 0.84 (d, J = 6.9 Hz, 3H); 13C-NMR (63 MHz, CDCl3) Racemate 1 and 2: 170.9 (s), 147.0 (s), 146.3 (s), 130.8 (s), 124.8 (d), 119.0 (d), 118.5 (d), 60.4 (t), 42.2 (t), 41.8 (t), 37.7 (t), 31.7 (d), 30.1 (d), 30.0 (d), 25.6 (q), 25.3 (t), 20.9 (q), 19.9 (q), 19.8 (q), 19.6 (q), 18.9 (q), 18.1 (q), 17.8 (q), 17.5 (q); Racemate 3: 170.8 (s), 145.7 (s), 130.8 (s), 124.8 (d), 118.0 (d), 61.2 (t), 42.0 (t), 40.0 (d), 37.4 (t), 30.0 (d), 25.6 (q), 25.3 (t), 20.9 (q), 20.0 (q), 19.5 (q), 17.5 (q), 12.1 (q); Racemate 4: 170.9 (s), 146.6 (s), 130.8 (s), 124.8 (d), 117.5 (d), 61.3 (t), 42.4 (t), 39.8 (d), 37.0 (t), 29.9 (d), 25.6 (q), 25.3 (t), 20.9 (q), 19.7 (q), 19.1 (q), 17.5 (q), 12.8 (q); MS (70 eV) m/z (%) Racemate 1 and 2 (Z) 266 [M]+ (1), 82 (100), 69 (76), 43 (57), 109 (55), 81 (52), 95 (44), 67 (38), 41 (37), 55 (32), 107 (28), 121 (26), 123 (25), 135 (15), 191 (12), 206 (6); Racemate 3 (E) 266 [M]+ (2), 82 (100), 69 (94), 43 (71), 109 (65), 81 (54), 95 (53), 41 (47), 67 (40), 121 (36), 55 (33), 107 (31), 123 (27), 135 (20), 191 (12), 206 (7); Racemate 4 (E) 266 [M]+ (1), 69 (100), 82 (97), 43 (71), 81 (67), 109 (65), 95 (53), 41 (47), 67 (40), 121 (38), 55 (34), 107 (30), 123 (26), 135 (19), 191 (11), 206 (6). Anal. Calcd for: C17H30O2: C, 76.41; H, 11.35. Found: C, 76.49; H, 11.28.