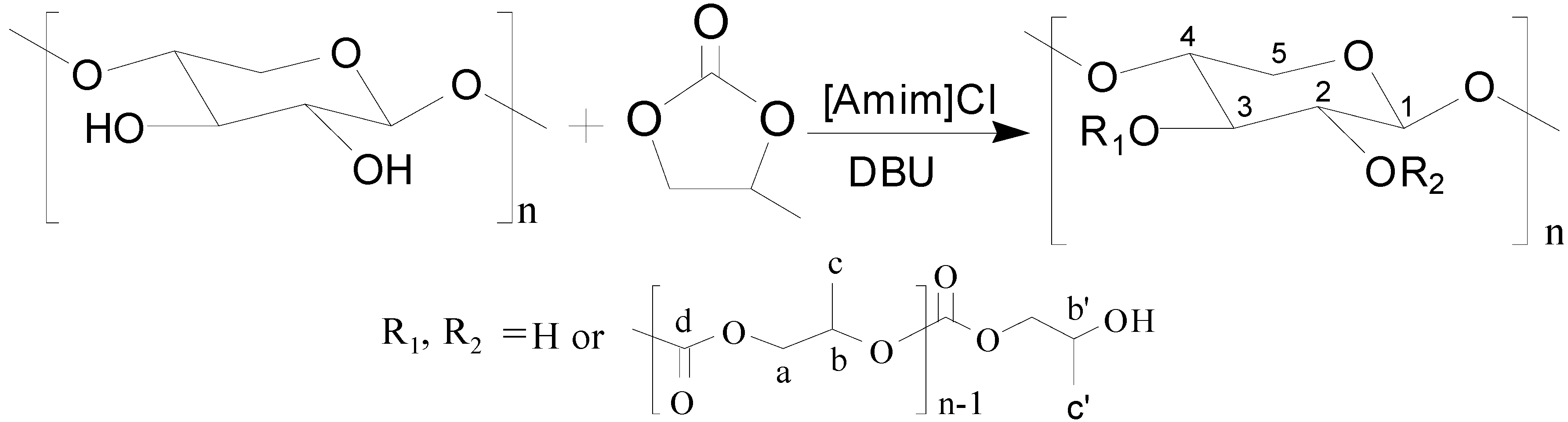
Ring-Opening Graft Polymerization of Propylene Carbonate onto Xylan in an Ionic Liquid
Abstract
:1. Introduction
2. Results and Discussion
2.1. Effects of Reaction Parameters on the Properties of Xylan-g-PPC Copolymers
| Sample | Temp. | Time | AXU:PC | DS | DP | WPG a (%) |
|---|---|---|---|---|---|---|
| No | (°C) | (h) | ||||
| 1 | 120 | 24 | 1:1 | 0.26 | 1.35 | 7.69 |
| 2 | 120 | 24 | 1:5 | 0.31 | 1.42 | 15.38 |
| 3 | 120 | 24 | 1:10 | 0.33 | 1.47 | 26.92 |
| 4 | 120 | 24 | 1:15 | 0.38 | 1.65 | 38.46 |
| 5 | 120 | 24 | 1:20 | 0.29 | 1.49 | 23.07 |
| 6 | 90 | 24 | 1:10 | 0.25 | 1.38 | 3.85 |
| 7 | 100 | 24 | 1:10 | 0.32 | 1.43 | 23.07 |
| 8 | 110 | 24 | 1:10 | 0.48 | 1.62 | 46.15 |
| 9 | 130 | 24 | 1:10 | 0.30 | 1.41 | 23.07 |
| 10 | 120 | 3 | 1:10 | 0.24 | 1.31 | 7.69 |
| 11 | 120 | 9 | 1:10 | 0.31 | 1.40 | 30.77 |
| 12 | 120 | 12 | 1:10 | 0.47 | 1.73 | 57.69 |
| 13 | 120 | 36 | 1:10 | 0.26 | 1.34 | 7.69 |
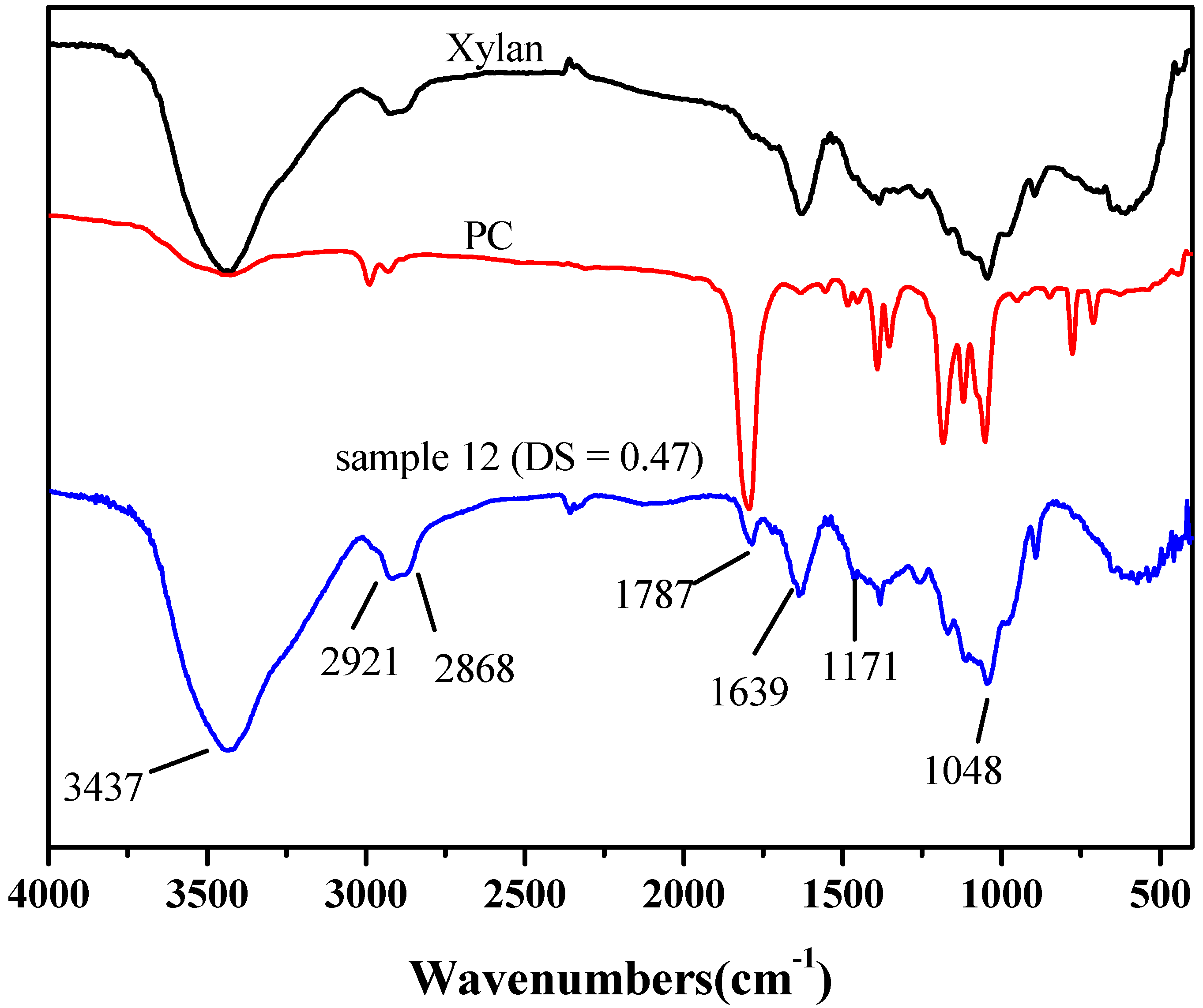
2.2. FT-IR Spectra
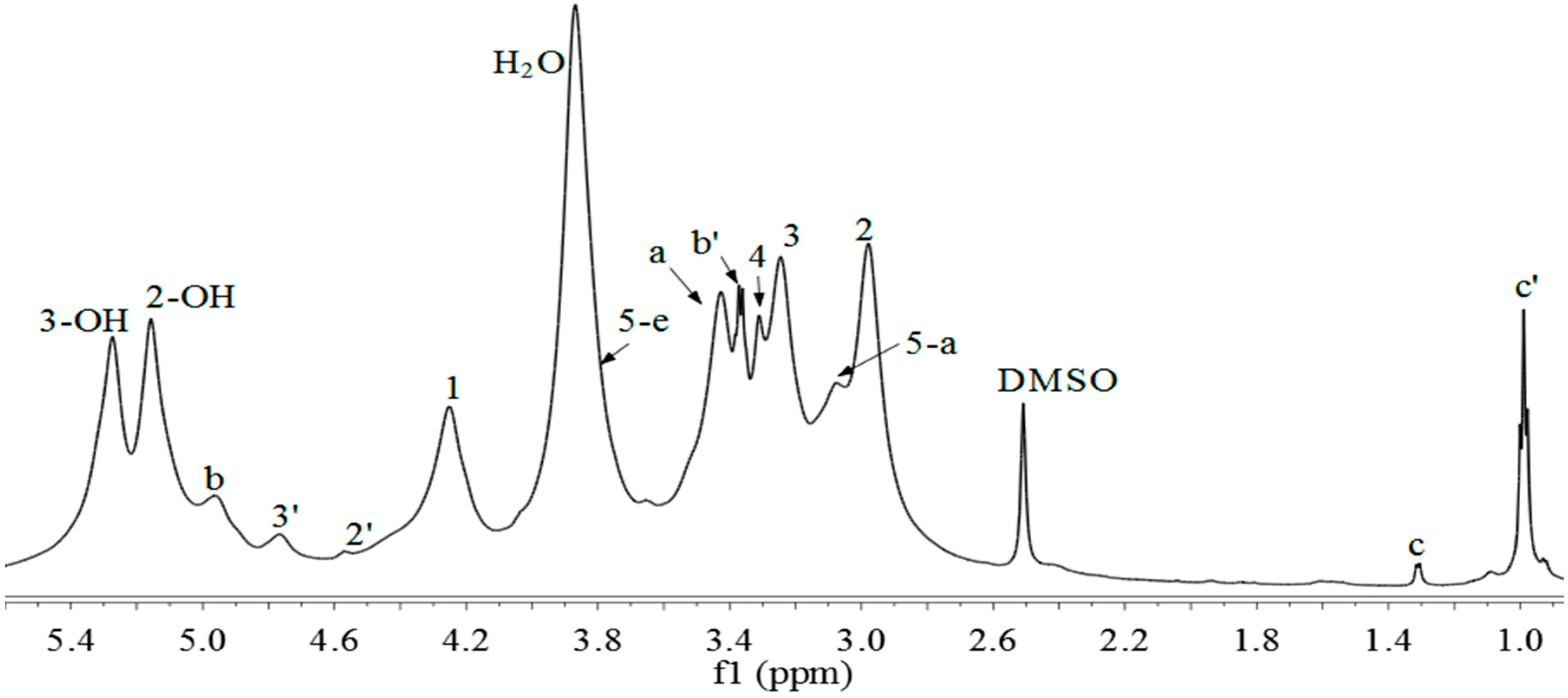
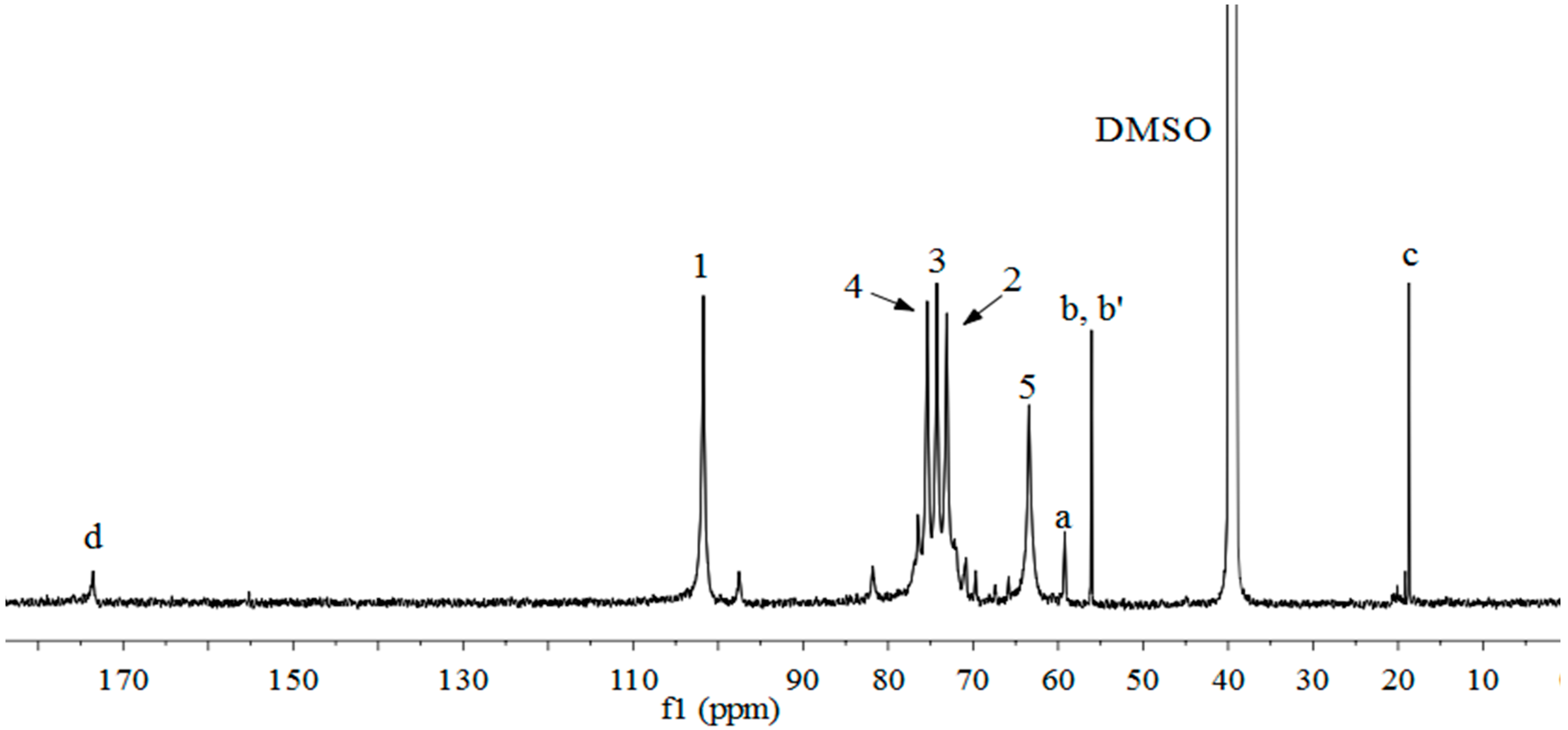
2.3. 1H-NMR, 13C-NMR and 1H-13C HSQC Spectra
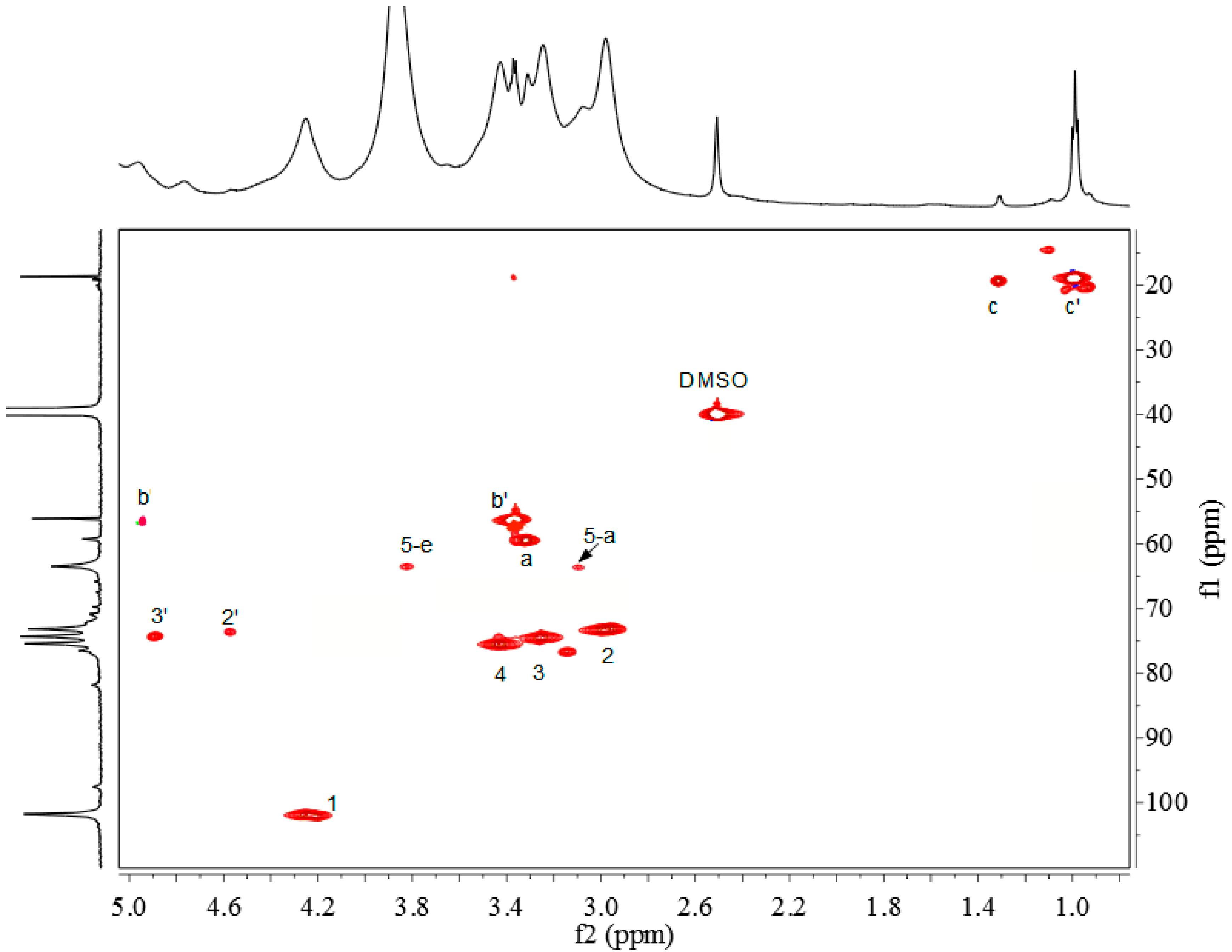
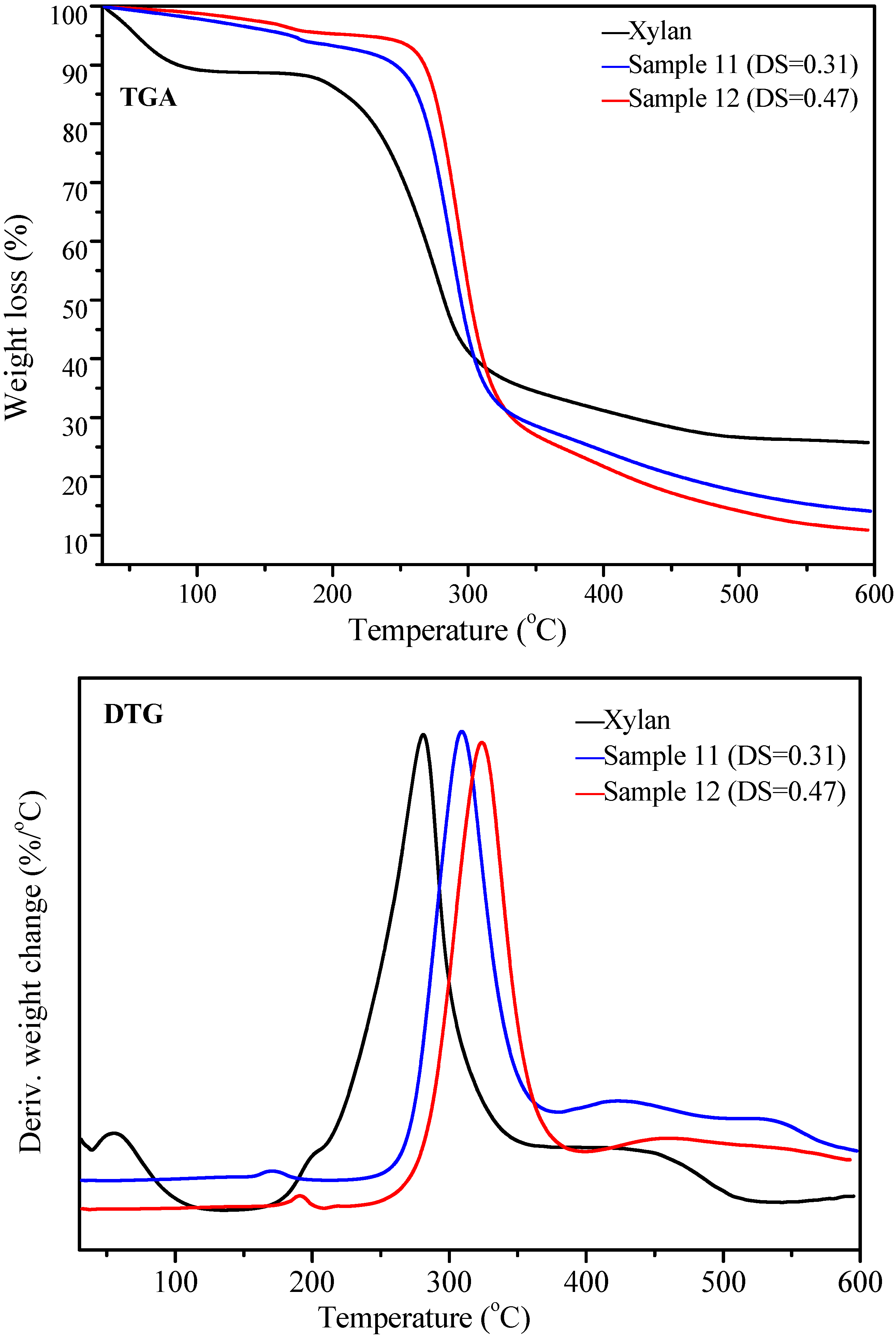
2.4. Thermal Analysis
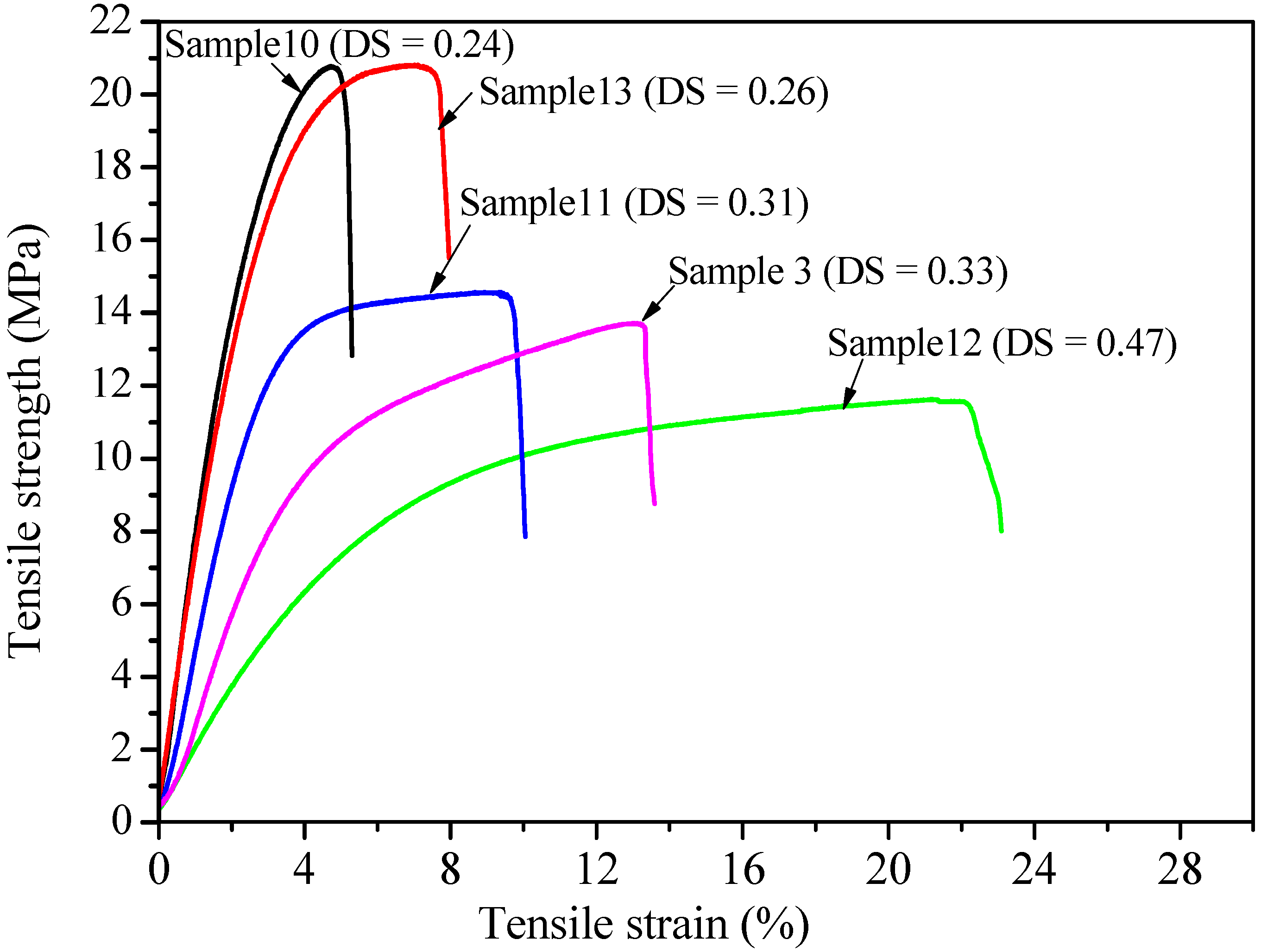
2.5. Effect of DS and DP on the Mechanical Properties of Xylan-g-PPC Copolymer Films
| Sample (S) | DS | Tensile Strength (MPa) | Tensile Strain at Break (%) | Young’s Modulus (MPa) |
|---|---|---|---|---|
| 10 | 0.23 | 20.78 ± 3.2 | 4.75 ± 1.1 | 658 ± 38 |
| 13 | 0.24 | 20.76 ± 2.9 | 7.73 ± 2.5 | 632 ± 36 |
| 11 | 0.30 | 14.48 ± 2.8 | 9.79 ± 2.7 | 343 ± 31 |
| 3 | 0.32 | 13.11 ± 2.3 | 13.23 ± 3.7 | 329 ± 29 |
| 12 | 0.45 | 11.32 ± 2.1 | 22.13 ± 3.8 | 126 ± 22 |
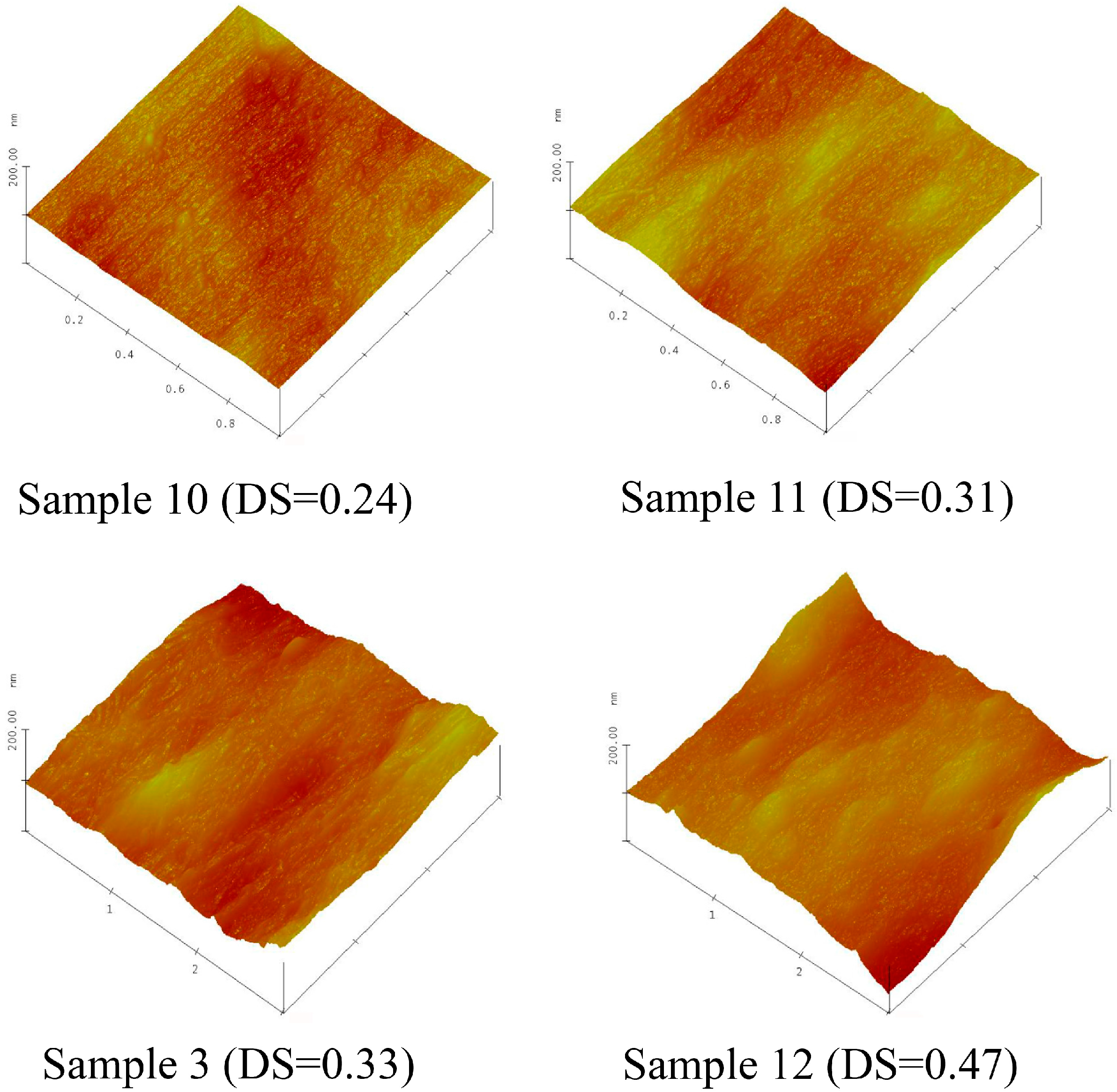
2.6. Morphology of Xylan-g-PPC Copolymer Film Surface
3. Experimental Section
3.1. Materials
3.2. Homogeneous Preparation of Xylan-g-PPC Copolymers with Different Reaction Parameters
3.3. Characterization
3.4. Film Preparation
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Lindblad, M.S.; Albertsson, A.C.; Ranucci, E.; Laus, M.; Giani, E. Biodegradable Polymers from Renewable Sources: Rheological Characterization of Hemicellulose-Based Hydrogels. Biomacromolecules 2005, 6, 684–690. [Google Scholar] [CrossRef] [PubMed]
- John, M.J.; Thomas, S. Biofibres and biocomposites. Carbohydr. Polym. 2008, 71, 343–364. [Google Scholar] [CrossRef]
- Choi, E.J.; Kim, C.H.; Park, J.K. Synthesis and Characterization of Starch-g-Polycaprolactone Copolymer. Macromolecules 1999, 32, 7402–7408. [Google Scholar] [CrossRef]
- Chen, L.; Qiu, X.; Deng, M.; Hong, Z.; Luo, R.; Chen, X.; Jing, X. The starch grafted poly(l-lactide) and the physical properties of its blending composites. Polymer 2005, 46, 5723–5729. [Google Scholar] [CrossRef]
- Fundador, N.G.V.; Enomoto-Rogers, Y.; Takemura, A.; Iwata, T. Syntheses and characterization of xylan esters. Polymer 2012, 53, 3885–3893. [Google Scholar] [CrossRef]
- González, D.; Campos, A.R.; Cunha, A.M.; Santos, V.; Parajó, J.C. Manufacture of fibrous reinforcements for biodegradable biocomposites from Citysus scoparius. J. Chem. Technol. Biot. 2011, 86, 575–583. [Google Scholar] [CrossRef]
- Lönnberg, H.; Zhou, Q.; Brumer, H.; Teeri, T.T.; Malmström, E.; Hult, A. Grafting of Cellulose Fibers with Poly(ε-caprolactone) and Poly(l-lactic acid) via Ring-Opening Polymerization. Biomacromolecules 2006, 7, 2178–2185. [Google Scholar] [CrossRef] [PubMed]
- Chung, Y.L.; Olsson, J.V.; Li, R.J.; Frank, C.W.; Waymouth, R.M.; Billington, S.L.; Sattely, E.S. A renewable lignin-lactide copolymer and application in biobased composites. ACS Sustain. Chem. Eng. 2013, 1, 1231–1238. [Google Scholar] [CrossRef]
- Saha, B. Hemicellulose bioconversion. J. Ind. Microbiol. Biot. 2003, 30, 279–291. [Google Scholar] [CrossRef]
- Peng, H.; Yang, A.S.; Xiong, J.H. Green, microwave-assisted synthesis of silver nanoparticles using bamboo hemicelluloses and glucose in an aqueous medium. Carbohydr. Polym. 2013, 91, 348–355. [Google Scholar] [CrossRef] [PubMed]
- Peng, F.; Peng, P.; Xu, F.; Sun, R.-C. Fractional purification and bioconversion of hemicelluloses. Biotechnol. Adv. 2012, 30, 879–903. [Google Scholar] [CrossRef] [PubMed]
- Gurbuz, E.I.; Gallo, J.M.R.; Alonso, D.M.; Wettstein, S.G.; Lim, W.Y.; Dumesic, J.A. Conversion of Hemicellulose into Furfural Using Solid Acid Catalysts in gamma-Valerolactone. Angew. Chem. Int. Ed. 2013, 52, 1270–1274. [Google Scholar] [CrossRef]
- Huda, M.S.; Drzal, L.T.; Misra, M.; Mohanty, A.K.; Williams, K.; Mielewski, D.F. A Study on Biocomposites from Recycled Newspaper Fiber and Poly(lactic acid). Ind. Eng. Chem. Res. 2005, 44, 5593–5601. [Google Scholar] [CrossRef]
- Mohanty, A.K.; Wibowo, A.; Misra, M.; Drzal, L.T. Effect of process engineering on the performance of natural fiber reinforced cellulose acetate biocomposites. Compos. Part A-Appl. Sci. Manuf. 2004, 35, 363–370. [Google Scholar] [CrossRef]
- Bhattacharya, A.; Misra, B.N. Grafting: A versatile means to modify polymers: Techniques, factors and applications. Prog. Polym. Sci. 2004, 29, 767–814. [Google Scholar] [CrossRef]
- Carlmark, A.; Larsson, E.; Malmström, E. Grafting of cellulose by ring-opening polymerisation—A review. Eur. Polym. J. 2012, 48, 1646–1659. [Google Scholar] [CrossRef]
- Mohanty, A.K.; Misra, M.; Hinrichsen, G. Biofibres, biodegradable polymers and biocomposites: An overview. Macromol. Mater. Eng. 2000, 276–277, 1–24. [Google Scholar]
- Riedel, U.; Nickel, J. Natural fibre-reinforced biopolymers as construction materials—New discoveries. Angew. Makromol. Chem. 1999, 272, 34–40. [Google Scholar] [CrossRef]
- Ge, X.C.; Li, X.H.; Zhu, Q.; Li, L.; Meng, Y.Z. Preparation and properties of biodegradable poly(propylene carbonate)/starch composites. Polym. Eng. Sci. 2004, 44, 2134–2140. [Google Scholar] [CrossRef]
- Kricheldorf, H.R. Syntheses and application of polylactides. Chemosphere 2001, 43, 49–54. [Google Scholar] [CrossRef] [PubMed]
- Endo, T.; Shibasaki, Y.; Sanda, F. Controlled ring-opening polymerization of cyclic carbonates and lactones by an activated monomer mechanism. J. Polym. Sci. Pol. Chem. 2002, 40, 2190–2198. [Google Scholar] [CrossRef]
- Guillaume, S.M.; Carpentier, J.-F. Recent advances in metallo/organo-catalyzed immortal ring-opening polymerization of cyclic carbonates. Catal. Sci. Technol. 2012, 2, 898–906. [Google Scholar] [CrossRef]
- Ma, X.; Yu, J.; Wang, N. Compatibility characterization of poly(lactic acid)/poly(propylene carbonate) blends. J. Polym. Sci. Pol. Phys. 2006, 44, 94–101. [Google Scholar] [CrossRef]
- Yan, C.; Zhang, J.; Lv, Y.; Yu, J.; Wu, J.; Zhang, J.; He, J. Thermoplastic Cellulose-graft-poly(l-lactide) Copolymers Homogeneously Synthesized in an Ionic Liquid with 4-Dimethylaminopyridine Catalyst. Biomacromolecules 2009, 10, 2013–2018. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.; Zheng, L.; Li, C.; Zhang, D.; Xiao, Y.; Guan, G.; Zhu, W. A novel and simple procedure to synthesize chitosan-graft-polycaprolactone in an ionic liquid. Carbohydr. Polym. 2013, 94, 505–510. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.-Q.; Chen, M.-J.; Liu, C.-F.; Sun, R.-C. Dual-component system dimethyl sulfoxide/licl as a solvent and catalyst for homogeneous ring-opening grafted polymerization of ε-caprolactone onto xylan. J. Agric. Food Chem. 2014, 62, 682–690. [Google Scholar] [CrossRef] [PubMed]
- Mani, R.; Tang, J.; Bhattacharya, M. Synthesis and characterization of starch-graft-polycaprolactone as compatibilizer for starch/polycarprolactone blends. Macromol. Rapid Commun. 1998, 19, 283–286. [Google Scholar] [CrossRef]
- Ren, G.; Sheng, X.; Qin, Y.; Chen, X.; Wang, X.; Wang, F. Toughening of poly(propylene carbonate) using rubbery non-isocyanate polyurethane: Transition from brittle to marginally tough. Polymer 2014, 55, 5460–5468. [Google Scholar] [CrossRef]
- Xing, C.; Wang, H.; Hu, Q.; Xu, F.; Cao, X.; You, J.; Li, Y. Mechanical and thermal properties of eco-friendly poly(propylene carbonate)/cellulose acetate butyrate blends. Carbohydr. Polym. 2013, 92, 1921–1927. [Google Scholar] [CrossRef] [PubMed]
- Ge, X.C.; Xu, Y.; Meng, Y.Z.; Li, R.K.Y. Thermal and mechanical properties of biodegradable composites of poly(propylene carbonate) and starch–poly(methyl acrylate) graft copolymer. Compos. Sci. Technol. 2005, 65, 2219–2225. [Google Scholar] [CrossRef]
- Ma, X.; Chang, P.R.; Yu, J.; Wang, N. Preparation and properties of biodegradable poly(propylene carbonate)/thermoplastic dried starch composites. Carbohydr. Polym. 2008, 71, 229–234. [Google Scholar] [CrossRef]
- Nederberg, F.; Connor, E.F.; Möller, M.; Glauser, T.; Hedrick, J.L. New Paradigms for Organic Catalysts: The First Organocatalytic Living Polymerization. Angew. Chem. Int. Ed. 2001, 40, 2712–2715. [Google Scholar] [CrossRef]
- Becker, J.M.; Tempelaar, S.; Standord, M.J.; Pounder, R.J.; Covington, J.A.; Dove, A.P. Development of amino-oxazoline and amino-thiazoline organic catalysts for the ring opening polymerization of lactide. Chem.-Eur. J. 2009, 16, 6099–6105. [Google Scholar] [CrossRef]
- Kamber, N.E.; Jeong, W.; Waymouth, R.M.; Pratt, R.C.; Lohmeijer, B.G.G.; Hedrick, J.L. Organocatalytic Ring-Opening Polymerization. Chem. Rev. 2007, 107, 5813–5840. [Google Scholar] [CrossRef] [PubMed]
- Nederberg, F.; Lohmeijer, B.G.G.; Leibfarth, F.; Pratt, R.C.; Choi, J.; Dove, A.P.; Waymouth, R.M.; Hedrick, J.L. Organocatalytic Ring Opening Polymerization of Trimethylene Carbonate. Biomacromolecules 2006, 8, 153–160. [Google Scholar] [CrossRef]
- Brown, H.A.; de Crisci, A.G.; Hedrick, J.L.; Waymouth, R.M. Amidine-Mediated Zwitterionic Polymerization of Lactide. ACS Macro Lett. 2012, 1, 1113–1115. [Google Scholar] [CrossRef]
- Chen, C.-Y.; Chen, M.-J.; Zhang, X.-Q.; Liu, C.-F.; Sun, R.-C. Per-O-acetylation of cellulose in dimethyl sulfoxide with catalyzed transesterification. J. Agric. Food Chem. 2014, 62, 3446–3452. [Google Scholar] [CrossRef] [PubMed]
- Cao, X.F.; Sun, S.N.; Peng, X.W.; Zhong, L.X.; Sun, R.C. Synthesis and characterization of cyanoethyl hemicelluloses and their hydrated products. Cellulose 2013, 20, 291–301. [Google Scholar] [CrossRef]
- Peng, X.W.; Ren, J.L.; Zhong, L.X.; Sun, R.C. Synthesis and Characterization of Amphoteric Xylan-type Hemicelluloses by Microwave Irradiation. J. Agric. Food Chem. 2012, 60, 1695–1702. [Google Scholar] [CrossRef] [PubMed]
- Peng, X.W.; Ren, J.L.; Zhong, L.X.; Sun, R.C. Homogeneous synthesis of hemicellulosic succinates with high degree of substitution in ionic liquid. Carbohydr. Polym. 2011, 86, 1768–1774. [Google Scholar] [CrossRef]
- Sample Availability: Samples are available from the authors.
© 2015 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhang, X.; Chen, M.; Liu, C.; Zhang, A.; Sun, R. Ring-Opening Graft Polymerization of Propylene Carbonate onto Xylan in an Ionic Liquid. Molecules 2015, 20, 6033-6047. https://doi.org/10.3390/molecules20046033
Zhang X, Chen M, Liu C, Zhang A, Sun R. Ring-Opening Graft Polymerization of Propylene Carbonate onto Xylan in an Ionic Liquid. Molecules. 2015; 20(4):6033-6047. https://doi.org/10.3390/molecules20046033
Chicago/Turabian StyleZhang, Xueqin, Mingjie Chen, Chuanfu Liu, Aiping Zhang, and Runcang Sun. 2015. "Ring-Opening Graft Polymerization of Propylene Carbonate onto Xylan in an Ionic Liquid" Molecules 20, no. 4: 6033-6047. https://doi.org/10.3390/molecules20046033
APA StyleZhang, X., Chen, M., Liu, C., Zhang, A., & Sun, R. (2015). Ring-Opening Graft Polymerization of Propylene Carbonate onto Xylan in an Ionic Liquid. Molecules, 20(4), 6033-6047. https://doi.org/10.3390/molecules20046033








