Design, Synthesis, and Insecticidal Activity of Some Novel Diacylhydrazine and Acylhydrazone Derivatives
Abstract
:1. Introduction
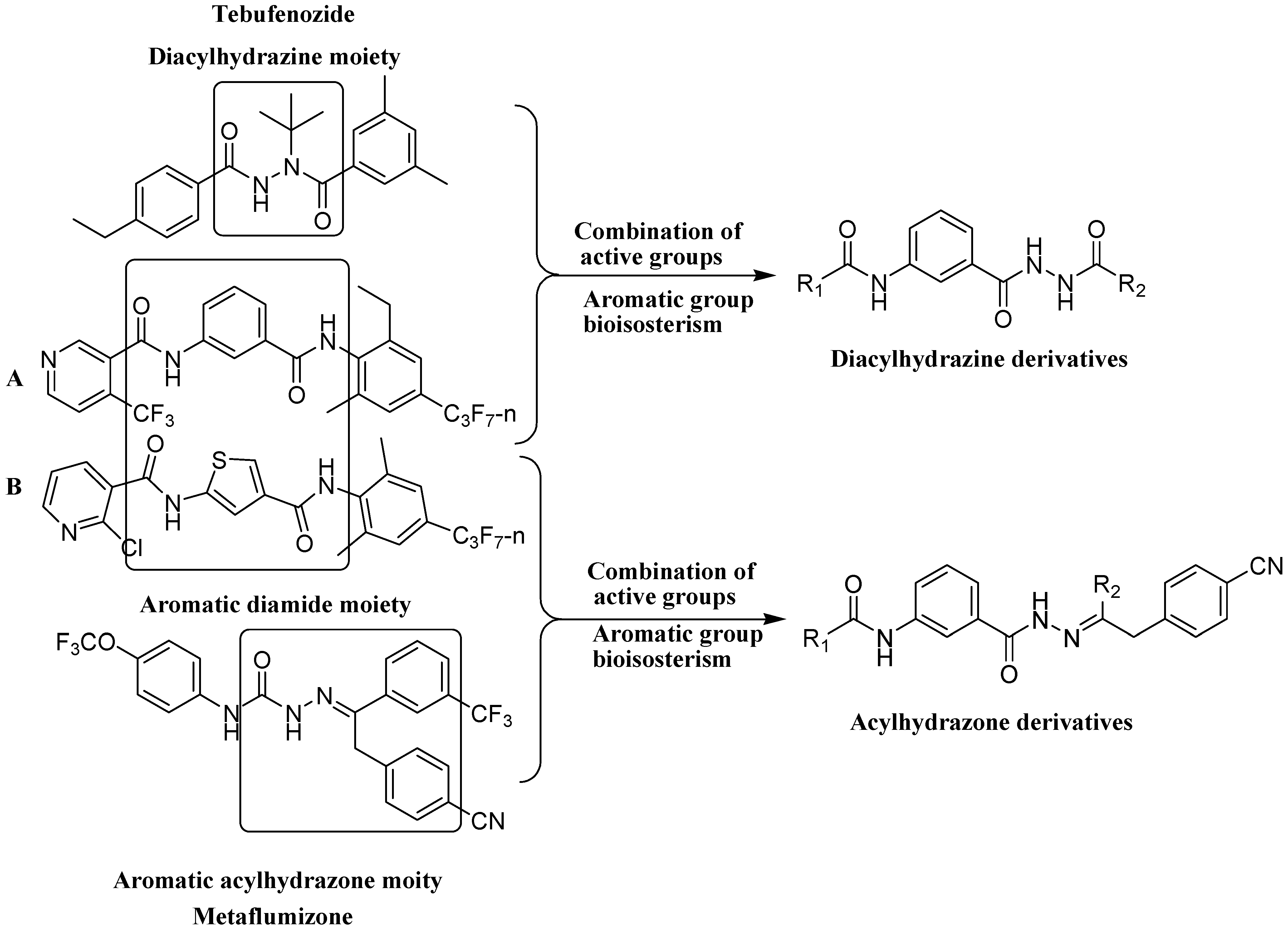
2. Results and Discussion
2.1. Chemistry
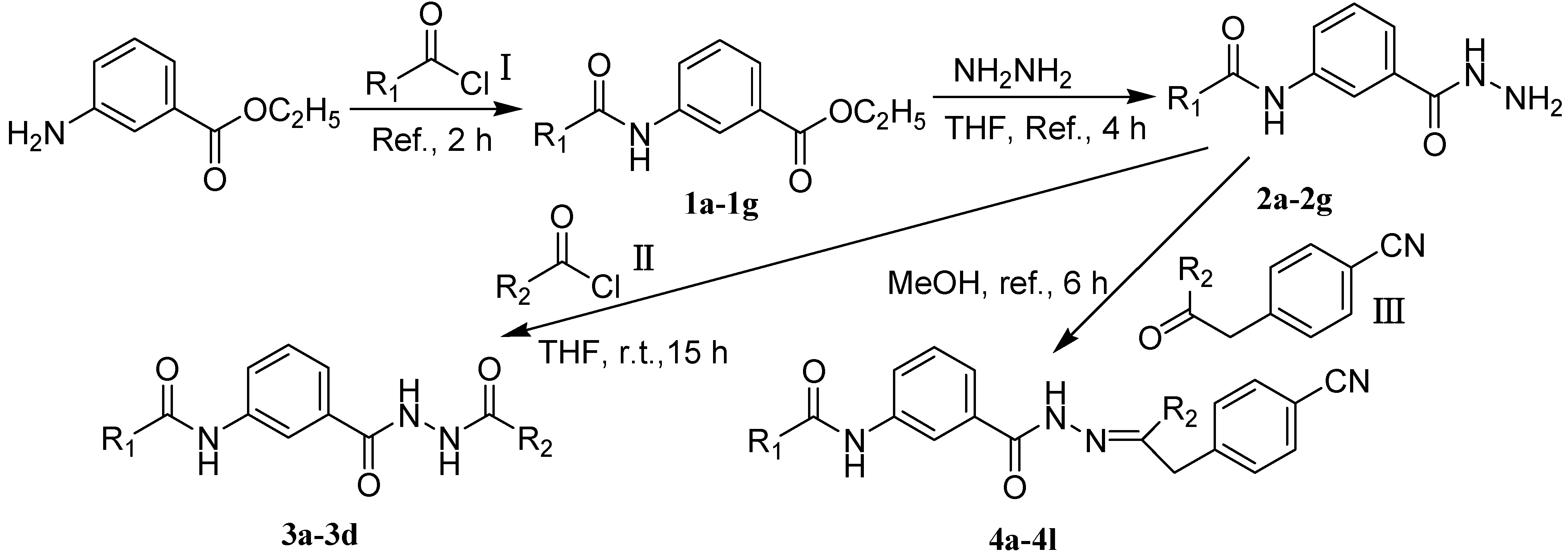
2.2. Insecticidal Activities
| Compound | R1 | R2 | Mortality (%) | ||
|---|---|---|---|---|---|
| 24 h | 48 h | 72 h | |||
| 3a |  | 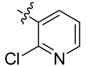 | 12.50 | 29.17 | 70.83 |
| 3b | 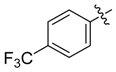 | 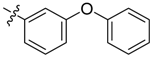 | 45.83 | 66.67 | 95.83 |
| 3c | 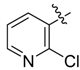 | 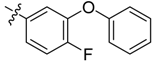 | 16.67 | 37.50 | 79.17 |
| 3d | 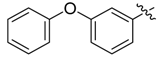 | 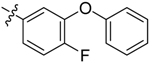 | 41.67 | 66.67 | 91.67 |
| 4a | 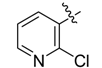 |  | 45.83 | 66.67 | 87.50 |
| 4b | 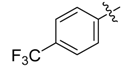 | 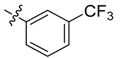 | 50.00 | 75.00 | 100.00 |
| 4c | 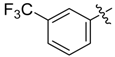 | 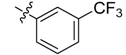 | 50.00 | 79.17 | 95.83 |
| 4d | 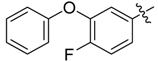 | 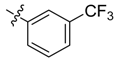 | 54.17 | 79.17 | 100.00 |
| 4e | 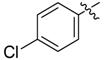 | 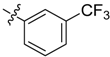 | 45.83 | 66.67 | 91.67 |
| 4f | 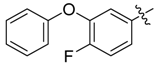 | 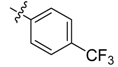 | 54.17 | 70.83 | 95.83 |
| 4g | 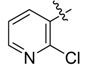 | 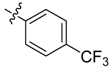 | 41.67 | 70.83 | 91.67 |
| 4h | 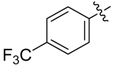 | 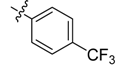 | 37.50 | 75.00 | 91.67 |
| 4i | 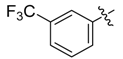 | 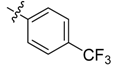 | 45.83 | 70.83 | 91.67 |
| 4j |  | 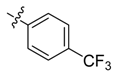 | 37.50 | 66.67 | 87.50 |
| 4k | 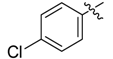 |  | 41.67 | 70.83 | 87.50 |
| 4l | 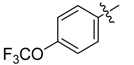 | 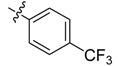 | 58.33 | 83.33 | 100.00 |
| Reference Compounds | |||||
| Tebufenozide | 12.50 | 37.50 | 66.67 | ||
| Metaflumizone | 37.50 | 45.83 | 66.67 | ||
| Tolfenpyrad | 45.83 | 66.67 | 79.17 | ||
| Compound | Mortality(%) | ||
|---|---|---|---|
| H. armigera | P. xyllostella | P. rapae | |
| 4a | 87.50 | 79.17 | 91.67 |
| 4b | 91.67 | 95.83 | 100.0 |
| 4c | 91.67 | 87.50 | 100.0 |
| 4d | 95.83 | 87.50 | 100.0 |
| 4e | 79.17 | 83.33 | 91.67 |
| 4f | 95.83 | 100.0 | 100.0 |
| 4g | 87.50 | 87.50 | 95.83 |
| 4h | 83.33 | 87.50 | 95.83 |
| 4i | 87.50 | 75.50 | 91.67 |
| 4j | 83.33 | 83.33 | 91.67 |
| 4k | 91.67 | 70.83 | 91.67 |
| 4l | 79.17 | 95.83 | 100.0 |
| Metaflumizone | 87.50 | 83.33 | 87.50 |
| Tolfenpyrad | 91.67 | 91.67 | 95.83 |
3. Experimental Section
3.1. General Procedures
3.2. General Procedure for the Preparation of 1a–1g
3.3. General Procedure for the Preparation of 2a–2g
3.4. General Procedure for Preparation of 3a–3d and 4a–4l
3.5. Insecticidal Activity Bioassays
4. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Seiber, J.N.; Kleinschmidt, L.A. Contribution of pesticide residue chemistry to improving food and environmental safety: past and present accomplishments and future challenges. J. Agric. Food Chem. 2011, 59, 7536–7543. [Google Scholar] [CrossRef] [PubMed]
- Chatterjee, N.S.; Gupta, S.; Varghese, E. Degradation of metaflumizone in soil: Impact of varying moisture, light, temperature, atmospheric CO2 level, soil type and soil sterilization. Chemosphere 2013, 90, 729–736. [Google Scholar] [CrossRef] [PubMed]
- Luo, M.; Chen, Q.C.; Wang, J.; Hu, C.Y.; Lu, J.; Luo, X.M.; Sun, D.Q. Novel chlorantraniliprole derivatives as potential insecticides and probe to chlorantraniliprole binding site on ryanodine receptor. Bioorg. Med. Chem. Lett. 2014, 24, 1987–1992. [Google Scholar] [CrossRef] [PubMed]
- Tohnishi, M.; Nakao, H.; Kohno, E.; Nishida, T.; Furuya, T.; Shimizu, T.; Seo, A.; Sakata, K.; Fujioka, S. Phthalic Acid Diamide Derivatives, Fluorine-Containing Aniline Compounds as Starting Material, Agricultural and Horticultural Insecticides, and A Method for Application of the Insecticides. U.S. Patent 6,559,341, 20 March 2003. [Google Scholar]
- Masali, T.; Yasokawa, N.; Tohnishi, M.; Nishimatsu, T.; Tsubata, K.; Inoue, K.; Motoba, K.; Hirooka, T. Flubendiamide, a novel Ca2+ channel modulator, reveals evidence for functional cooperation between Ca2+ pumps and Ca2+ release. Mol. Pharmacol. 2006, 69, 1733–1739. [Google Scholar] [CrossRef] [PubMed]
- Lahm, G.P.; Cordova, D.; Barry, J.D. New and selective ryanodine receptor activators for insect control. Bioorg. Med. Chem. 2009, 17, 4127–4133. [Google Scholar] [CrossRef] [PubMed]
- Hughes, K.A.; Lahm, G.P.; Selby, T.P.; Stevenson, T.M. Cyano Anthranilamide Insecticides. WO 2004/067528, 12 August 2004. [Google Scholar]
- Akihiko, Y.; Yukiyishi, W.; Katsuaki, W.; Tetsuya, M.; Katsuhiko, S.; Eiichi, S.; Akira, E. Insecticidal 3-Acylaminobenzanilides. WO 2007/017075, 15 February 2007. [Google Scholar]
- Pierre, J.; Patricia, D.; William, L.; Peter, M.; Thomas, P.; Peter, R.; Werner, Z. Insecticidal Compounds. WO 2007/128410, 15 November 2007. [Google Scholar]
- Zhang, Y.B. The insecticedes of ryanodine receptor inhibitors and varieties structural character and mechanism. Agrochemicals 2012, 51, 157–162. [Google Scholar]
- Li, Y.X.; Mao, M.Z.; Li, Y.M.; Xiong, L.X.; Li, Z.M.; Xu, J.Y. Modulations of high-voltage activated Ca2+channels in the central neurones of Spodoptera exigua by chlorantraniliprole. Physiol. Entomol. 2011, 36, 230–234. [Google Scholar] [CrossRef]
- Cordova, D.; Benner, E.A.; Sacher, M.D.; Rauh, J.J.; Sopa, J.S.; Lahm, G.P.; Selby, T.P.; Stevenson, T.M.; Flexner, L.; Gutteridge, S.; et al. Anthranilicdiamides: A new class of insecticides with a novel mode of action, ryanodine receptor activation. Pestic. Biochem. Physiol. 2006, 84, 196–214. [Google Scholar]
- Wing, K.D. RH-5849, a nonsteroidal ecdysone agonist: effects on a drosophila cell line. Science 1988, 241, 467–469. [Google Scholar] [CrossRef] [PubMed]
- Sun, R.F.; Zhang, Y.L.; Chen, L.; Li, Y.Q.; Li, Q.S.; Song, H.B.; Huang, R.Q.; Bi, F.C.; Wang, Q.M. Design, synthesis, and insecticidal activities of new N-benzoyl-N'-phenyl-N'-sulfenylureas. J. Agric. Food Chem. 2009, 57, 3661–3668. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.; Yang, Z.K.; Fan, Z.J.; Wu, Q.J.; Zhang, Y.J.; Mi, N.; Wang, S.X.; Zhang, Z.C.; Song, H.B.; Liu, F. Synthesis and insecticidal activity of N-tert-Butyl-N,N'-diacylhydrazines containing 1,2,3-Thiadiazoles. J. Agric. Food Chem. 2011, 59, 628–634. [Google Scholar] [CrossRef] [PubMed]
- Yanagi, M.; Tsukamoto, Y.; Watanabe, T.; Kawagishi, A. Development of a novel lepidopteran insect control agent chromafenozide. J. Pestic. Sci. 2006, 31, 163–164. [Google Scholar] [CrossRef]
- Aguirre, O.U.; Martínez, A.M.; Campos-García, J.; Hernández, L.; Figueroa, J.I.; Lobit, P.; Viñuela, E.; Chavarrieta, J.M.; Smagghe, G.; Pineda1, S. Foliar persistence and residual activity of methoxyfenozide against beet armyworm (Lepidoptera: Noctuidae). Insect Sci. 2013, 20, 734–742. [Google Scholar] [CrossRef] [PubMed]
- Salgado, V.L.; Hayashi, J.H. Metaflumizone is a novel sodium channel blocker insecticide. Vet. Parasitol. 2007, 150, 182–189. [Google Scholar] [CrossRef] [PubMed]
- Hempel, K.; Hess, F.G.; Bogi, C.; Fabian, E.; Hellwig, J.; Fegert, I. Toxicological properties of metaflumizone. Vet. Parasitol. 2007, 150, 190–195. [Google Scholar] [CrossRef] [PubMed]
- Che, Z.P.; Zhang, S.Y.; Shao, Y.H.; Fan, L.L.; Xu, H.; Yu, X.; Zhi, X.Y.; Yao, X.J.; Zhang, R. Synthesis and quantitative structure-activity relationship (QSAR) study of novel N-arylsulfonyl-3-acylindole arylcarbonyl hydrazone derivatives as nematicidal agents. J. Agric. Food Chem. 2013, 61, 5696–5705. [Google Scholar] [CrossRef] [PubMed]
- Qu, H.; Yu, Y.; Zhi, X.Y.; Lv, M.; Xu, H. Natural-product-based insecticidal agents 14.Semisynthesis and insecticidal activity of new piperine-based hydrazone derivatives against Mythimna separata Walker in vivo. Bioorg. Med. Chem. Lett. 2013, 23, 5552–5557. [Google Scholar]
- Cao, G.; Han, Z. Tebufenozide resistance selected in Plutella xylostella and its cross-resistance and fitness cost. Pest. Manag. Sci. 2006, 62, 746–751. [Google Scholar] [CrossRef] [PubMed]
- Mao, C.H.; Wang, Q.M.; Huang, R.Q.; Bi, F.C.; Chen, L.; Liu, Y.X.; Shang, J. Synthesis and insecticidal evaluation of novel N-oxalyl derivatives of tebufenozide. J. Agric. Food Chem. 2004, 52, 6737–6741. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.W.; Hu, Z.; Li, S.K.; Wu, W.J. Synthesis and insecticidal activities of new ester-derivatives of Celangulin-V. Int. J. Mol. Sci. 2011, 12, 9596–9604. [Google Scholar] [CrossRef] [PubMed]
- Sample Availability: Samples of the compounds 3a–3d and 4a–4l are available from the authors.
© 2015 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sun, J.; Zhou, Y. Design, Synthesis, and Insecticidal Activity of Some Novel Diacylhydrazine and Acylhydrazone Derivatives. Molecules 2015, 20, 5625-5637. https://doi.org/10.3390/molecules20045625
Sun J, Zhou Y. Design, Synthesis, and Insecticidal Activity of Some Novel Diacylhydrazine and Acylhydrazone Derivatives. Molecules. 2015; 20(4):5625-5637. https://doi.org/10.3390/molecules20045625
Chicago/Turabian StyleSun, Jialong, and Yuanming Zhou. 2015. "Design, Synthesis, and Insecticidal Activity of Some Novel Diacylhydrazine and Acylhydrazone Derivatives" Molecules 20, no. 4: 5625-5637. https://doi.org/10.3390/molecules20045625
APA StyleSun, J., & Zhou, Y. (2015). Design, Synthesis, and Insecticidal Activity of Some Novel Diacylhydrazine and Acylhydrazone Derivatives. Molecules, 20(4), 5625-5637. https://doi.org/10.3390/molecules20045625





