Abstract
Through the incorporation of a thiophene functionality, a novel solution-processable small organic chromophore was designed, synthesized and characterized for application in bulk-heterojunction solar cells. The new chromophore, (2Z,2′Z)-2,2′-(1,4-phenylene)bis(3-(5-(4-(diphenylamino)phenyl)thiophen-2-yl)acrylonitrile) (coded as AS2), was based on a donor–acceptor–donor (D–A–D) module where a simple triphenylamine unit served as an electron donor, 1,4-phenylenediacetonitrile as an electron acceptor, and a thiophene ring as the π-bridge embedded between the donor and acceptor functionalities. AS2 was isolated as brick-red, needle-shaped crystals, and was fully characterized by 1H- and 13C-NMR, IR, mass spectrometry and single crystal X-ray diffraction. The optoelectronic and photovoltaic properties of AS2 were compared with those of a structural analogue, (2Z,2′Z)-2,2′-(1,4-phenylene)bis(3-(4-(diphenylamino)phenyl)-acrylonitrile) (AS1). Benefiting from the covalent thiophene bridges, compared to AS1 thin solid film, the AS2 film showed: (1) an enhancement of light-harvesting ability by 20%; (2) an increase in wavelength of the longest wavelength absorption maximum (497 nm vs. 470 nm) and (3) a narrower optical band-gap (1.93 eV vs. 2.17 eV). Studies on the photovoltaic properties revealed that the best AS2-[6,6]-phenyl-C61-butyric acid methyl ester (PC61BM)-based device showed an impressive enhanced power conversion efficiency of 4.10%, an approx. 3-fold increase with respect to the efficiency of the best AS1-based device (1.23%). These results clearly indicated that embodiment of thiophene functionality extended the molecular conjugation, thus enhancing the light-harvesting ability and short-circuit current density, while further improving the bulk-heterojunction device performance. To our knowledge, AS2 is the first example in the literature where a thiophene unit has been used in conjunction with a 1,4-phenylenediacetonitrile accepting functionality to extend the π-conjugation in a given D–A–D motif for bulk-heterojunction solar cell applications.
1. Introduction
The development of renewable energy technologies is pivotal for accommodating the ever increasing energy demands of the modern society. Such technologies are also important for lowering environmental pollution and greenhouse gas emissions. Towards this objective, many approaches to harvest solar energy have been investigated. The fabrication of bulk-heterojunction (BHJ) devices is one such promising strategy that has attracted considerable attention over the past two decades due to their advantages of being lightweight, low cost and their flexibility in large-area applications [1,2,3,4,5,6,7]. Such devices are comprised of an interpenetrating network of organic donor and acceptor domains that is formed during their fabrication via solution processing. Conventionally, semiconducting donor polymers such as poly(3-hexylthiophene) (P3HT) and acceptors such as soluble fullerene derivatives, PC61BM and its C71 analogue (PC71BM), have been used to obtain a deeper understanding of device design and morphology [8,9,10,11,12,13]. Apart from archetypal P3HT, conjugated polymers have also been developed and significant progress has been attained with promising BHJ architecture. Power conversion efficiency (PCE) values above 10% has been reported with such polymeric donors [14,15,16,17]. In the interim, solution-processed small molecular donor-based BHJ devices have also aroused interest, mainly due to their advantages of well-defined chemical structure, convenient purification methods, such as simple column chromatography, and monodisperse molecular weight [18,19,20,21,22,23]. These advantages allow and encourage researchers to exert efforts for the design and development of small molecular donors. Recently, immense efforts have been dedicated to developing small molecular-based solution-processable organic solar cells [1,21] and so far, the highest PCE of 9.95% was achieved by Kan et al. [24] which is analogous to those of the polymer-based solution-processable devices. Thus, in view of such reports and the fact that BHJ devices incorporating small molecular donors can compete with polymer-based devices, there is an overwhelming interest in developing small molecular donors.
Recent years have seen a dramatic surge not only in terms of device efficiency using small molecular donors but also in their design and efficient synthetic development. A variety of small molecule donor materials based on donor–acceptor (D–A) combinations such as D–A–D [25,26,27], A–D–A [28,29], D–π–A [18,19,30] and star-shaped architectures [31] have been reported. The D–A–D design in particular is one of the most promising and successful modules based on which various donor and acceptor units have been explored for high-performance solution-processable photovoltaic devices. A finite number of central accepting units, such as naphthalene diimide [25], diketopyrrolopyrrole [27], 2-pyran-4-ylidenemalononitrile [32] and thiazolothiazole [33] have been reported to suit the D–A–D module. Not only that the availability and selection of such accepting blocks is limited, it is furthermore imperative that the target chromophore must possess a low optical band gap, broad absorption profile, high mobility and appropriately tuned highest occupied molecular orbital (HOMO) and lowest unoccupied molecular orbital (LUMO) energy levels using such blocks. Such requirements do possess a challenge for an organic chemist who must consider such factors while designing a new chromophore based on the D–A–D module. Therefore, it is not surprising that there exists an enormous scope for the design and development of new light-harvesting materials based on the challenging D–A–D module and is an aspiration for most of the researchers.
In our own studies of small molecule chromophores and charge transport materials based on a variety of D–A combinations, we have reported examples of successful solution-processable BHJ photovoltaic devices [18,19,25,34,35,36,37]. Furthermore, we are highly interested to extend our efforts on the design and development of new chromophores that are inspired by the D–A–D module. In this study, we report the design, facile synthesis and characterization of the optoelectronic and photovoltaic properties of two small organic chromophores, AS2 and AS1, (shown in Figure 1), and their direct comparison. Both materials are based on a D–A–D structural motif where a triphenyl-amine functionality has been chosen as a common donor at both ends of the central acceptor unit, 1,4-phenylenediacetonitrile (PDA), so as to get symmetrical AS2 and AS1. AS2 is a structural analogue of AS1 where a thiophene ring has been introduced between the donor and acceptor functionalities in order to vary the optoelectronic and photovoltaic properties (Figure 1). When compared with the commonly used accepting groups, such as dicyanovinylidene, aromatizable cyanopyridone, indenedione or oxoindenemalononitrile [38,39], PDA can be an acceptor of choice for extended π-conjugation over the whole molecular backbone, mainly due to its bidentate nature. It is notable to mention that the use of the PDA unit for electroluminescent conjugated polymers has been reported in the literature [40,41] as well as one- and two-photon spectroscopy studies [42,43,44,45,46]. However, its suitability as an acceptor in small molecular chromophores, particularly D–A–D modular, for BHJ applications is still unknown [4,21]. This provides an encouragement and some strong incentive for its investigation. Owing to the exploration of PDA acceptor unit in the D–A–D module, this study continues our search for the generation of new organic chromophores for BHJ applications.
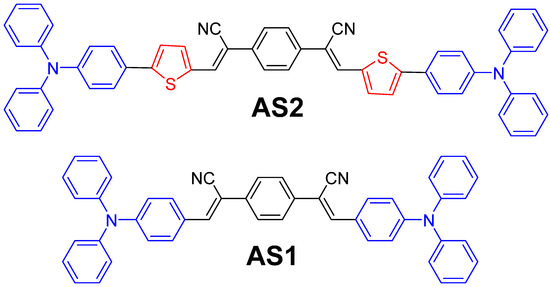
Figure 1.
Molecular structures of the newly designed AS2 and reference AS1 materials investigated in this study.
2. Results and Discussion
2.1. Design Strategy, Synthesis
The materials AS2 and AS1 were synthesized via the Knoevenagel condensation of appropriate aldehydes with active methylene groups of the PDA acceptor unit and their chemical structures were confirmed by 1H- and 13C-NMR, mass spectrometry, and, where possible (AS2 only), by single crystal X-ray diffraction (XRD). The synthetic methodology for synthesizing AS1 was similar to an old literature report [42], albeit dissimilar base and solvent were used to deal with a homogeneous reaction solution. Knoevenagel condensation reaction of an aldehyde with active methylene group is an efficient way of generating a double bond between a π-bridge and acceptor functionality. The use of such chemistry is a common strategy to generate organic sensitizers for dye-sensitized solar cells [47]. However, the use of same strategy to develop small molecular chromophores for BHJ applications is still limited [4,21]. Herein, not only we are demonstrating the Knoevenagel condensation reaction of PDA acceptor unit but also the fabrication of solution-processable BHJ devices incorporating a fullerene acceptor (PC61BM) and either AS2 or AS1 as a donor component (Figure 1). To the best of our knowledge, this is the first time PDA has been used to generate D–A–D modular small molecular chromophores for BHJ applications. Initial screening of the BHJ devices revealed that greater PCE was achieved for AS2 (4.10% for AS2 compared with 1.23% for AS1), as confirmed by the increased short-circuit current density (8.01 mA·cm−2 for AS2 and 3.15 mA·cm−2 for AS1), under simulated AM 1.5 illumination (100 mW·cm−2).
Both materials were based on the D–A–D module and the central acceptor moiety was directly linked to the terminal donor functionalities in order to create a conjugated structure. The development of these target materials incorporates the use of two identical donor units (triphenylamine) on each side of the central core, resulting in symmetrical chromophores. Insertion of a planar, conjugated functionality, such as thiophene in AS2, between the donor and acceptor components of a target material can provide greater absorption over visible light spectrum when compared with otherwise structurally similar compounds [38,48]. Moreover, the selection of thiophene over highly aromatic, conjugating functionalities, such as phenyl, was based on the earlier work reported by Würthner et al. [49] and Gupta et al. [50] where it has been demonstrated that replacement of a phenyl group with thiophene can provide significant spectral red-shifts and is advantageous for superior charge delocalization. As a result, AS2 is deemed to exhibit a large red shift of lambda maximum when compared with the reference compound AS1. Both of the materials were synthesized per the reaction shown in Scheme 1 and were purified by simple column chromatography. Brick-red, needle-shaped crystals, suitable for single crystal XRD, were prepared by diffusing methanol into a dichloromethane solution of AS2, over approximately three days. However, no crystal growth was observed for AS1. Both materials were synthesized in moderate to high yields (AS2 = 63% and AS1 = 86%) and were highly soluble in a variety of common organic solvents, for example chlorobenzene, chloroform, and toluene. The solubility of organic p-type materials is paramount for fabricating solution-processable BHJ devices and both the materials fulfill this criterion. In fact, the solubility of AS2 was found to be higher by 50% w/v when compared with AS1.
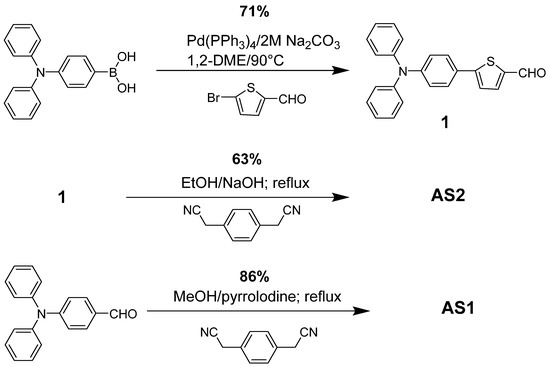
Scheme 1.
Reaction scheme for the synthesis of AS2 and AS1.
The precursor aldehyde 1 of AS2 was also crystallized by diffusion of methanol into chloroform to obtain yellow, needle-shaped crystals. Diffraction measurements were performed at 200 K on a Bruker Apex II CCD diffractometer using Mo Kα radiation(λ = 0.71073 Å) The structure were solved using dual space methods using the program SHELX-2014/7 [51] using the Olex2 1.2 GUI [52], with anisotropic thermal parameters for all non-hydrogen atoms. All non-hydrogen atoms were refined anisotropically by full-matric least-squares methods SHELX-2014/7. Molecular drawings were obtained using Mercury [53]. The utility of 1 for dye-sensitized solar cells has been reported [54], however, its use to generate small molecular chromophores for BHJ applications is seldom reported [4,21]. This state of affairs encourages us and provides a strong incentive to report its synthesis and crystal growth strategy.
The compound 1 was crystallized in the monoclinic space group (P 21/c) with four asymmetric units in one cell. The thiophene group and the adjacent phenyl groups are planar with pendant phenyl groups displaced around nitrogen. The packing consist of a two-fold screw axis with centers of inversion between sulfur molecules as well as a glide plane perpendicular to the thiophene plane. The packing is dominated by π-π face-to-face stacking between the thiophene and phenyl groups. The pendant phenyl groups are stabilized by π-π edge-to-face stacking with distances in the range 2.771–3.283 Å as shown in Figure 2.
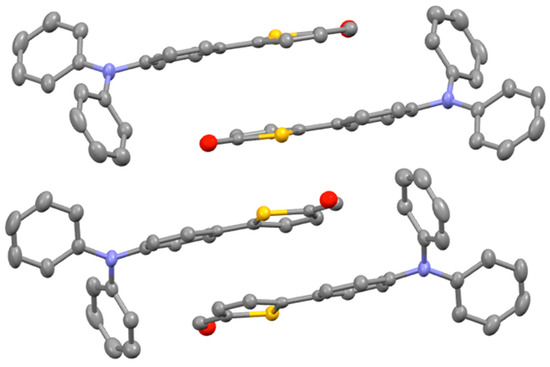
Figure 2.
Packing of 1 along the b axis.
AS2 co-crystallizes with chloroform in the triclinic space group (P-1) with a center of symmetry around the central phenyl group. The packing is dominated by face-to-face π-π stacking between central phenyls and thiophenes, and by edge-to-face π‑π‑stacking between pendant phenyl groups with the distance 2.772 Å as shown in Figure 3. The details of packing structure, formula, crystal size of 1 and AS2 are described in Table 1.

Table 1.
Details of crystal data and structure refinement parameters of 1 and AS2.
| Identification Code | CCDC: 1420377 (1) | CCDC: 1420378 (AS2) |
|---|---|---|
| Empirical formula | C23H17NOS | C31H22OS |
| Formula weight | 355.43 | 442.55 |
| Temperature/K | 200(2) | 200(2) |
| Crystal system | monoclinic | monoclinic |
| Space group | P21/c | P21 |
| a/Å | 19.916(3) | 5.5812(7) |
| b/Å | 6.6680(9) | 47.120(6) |
| c/Å | 13.4336(16) | 9.2435(11) |
| α/° | 90 | 90 |
| β/° | 96.613(3) | 103.343(3) |
| γ/° | 90 | 90 |
| Volume/Å3 | 1772.1(4) | 2365.3(5) |
| Z | 4 | 4 |
| ρcalcg/cm3 | 1.332 | 1.243 |
| μ/mm−1 | 0.194 | 0.158 |
| F(000) | 744.0 | 928.0 |
| Crystal size/mm3 | 0.292 × 0.076 × 0.063 | 0.667 × 0.137 × 0.087 |
| Radiation | MoKα (λ = 0.71073) | MoKα (λ = 0.71073) |
| 2Θ range for data collection/ | 4.118 to 48.588 | 3.458 to 67.842 |
| Index ranges | −22 ≤ h ≤ 23, −7 ≤ k ≤ 7, −15 ≤ l ≤ 15 | −8 ≤ h ≤ 8, −73 ≤ k ≤ 73, −14 ≤ l ≤ 12 |
| Reflections collected | 15255 | 83,300 |
| Independent reflections | 2883 [Rint = 0.0707, Rsigma = 0.0447] | 19,180 [Rint = 0.0616, Rsigma = 0.0596] |
| Data/restraints/parameters | 2883/0/235 | 19180/1/595 |
| Goodness-of-fit on F2 | 0.949 | 1.028 |
| Final R indexes [I ≥ 2σ (I)] | R1 = 0.0412, wR2 = 0.1095 | R1 = 0.0659, wR2 = 0.1531 |
| Final R indexes [all data] | R1 = 0.0720, wR2 = 0.1274 | R1 = 0.0972, wR2 = 0.1684 |
| Largest diff. peak/hole/e Å−3 | 0.17/−0.26 | 0.29/−0.39 |
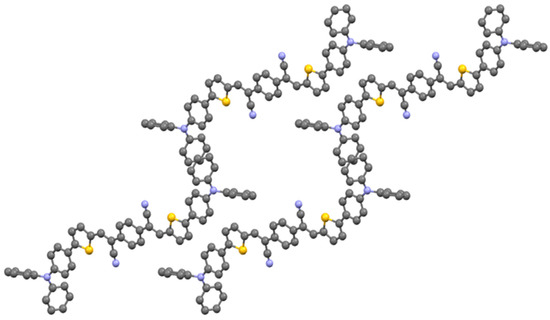
Figure 3.
Packing of AS2 along the c axis.
2.2. Optoelectronic Properties
The optical properties of AS2 and AS1 were investigated by measuring their ultraviolet–visible (UV–Vis) absorption spectra in chloroform solution and in pristine spin-cast films (Figure 4). The longest wavelength absorption maximum (λmax) exhibited by AS2 in solution form was at 459 nm which was red-shifted by 23 nm when compared with the solution λmax of AS1. Both the absorption maximum and extinction coefficient (AS2 = 59,000 M−1·cm−1; AS1 = 49,000 M−1·cm−1) increased with the insertion of thiophene functionality. With the insertion of thiophene ring we found enhancement to the peak molar absorptivity of >20% of AS2 compared with AS1. This enhanced profile allows a larger amount of the solar spectrum to be absorbed, thus exhibiting greater intramolecular charge transfer (ICT) transition. We observed a similar bathochromic absorption shift in the thin film spectrum of AS2 compared with that of AS1 (Figure 4). The strong red-shift is attributed to the extended π-conjugation within the molecular backbone of AS2 that became possible with the insertion of thiophene functionality. This type of control over the absorption profile through the insertion of a strongly conjugated unit can help to fine tune optical energy levels, to enhance light harvesting and BHJ device performance.
Density functional theory (DFT) calculations using the Gaussian 09 suite of programs [55] and the B3LYP/6-311 + G(d,p)//B3LYP/6-31G(d) level of theory indicated that the HOMO orbital densities of both AS2 and AS1 have a major distribution over the whole molecular backbone and the LUMO densities were delocalized through the central acceptor functionality and adjacent rings (Figure 5). Inserted thiophene rings in case of AS2 can accommodate LUMO density with almost equal contribution as the central PDA, thus sparing the adjoining phenyl rings of the donor triphenylamine for an efficient segregation of HOMO and LUMO densities. Such separation is ideal for the ICT transition and is attributed to the presence of a strong conjugated unit, of which thiophene is an example, in the given D–A–D system. Experimental estimation of the HOMO energies was carried out using photo electron spectroscopy in air (PESA) and the LUMO energies were calculated by adding the optical band gap (film spectra) to the HOMO values (see Figure 6 for energy level diagram and Figure S1 [see Supplementary Material] for the PESA curve). Film spectra indicated that insertion of thiophene reduces the band gap of AS2 by 0.24 eV when compared with AS1. Furthermore, the estimation of HOMO using PESA revealed that the HOMO energy level of AS2 was raised by 0.18 eV when compared with the HOMO level of AS1. This is in agreement with the DFT calculations that the presence of thiophene indeed plays a crucial role for: (1) density segregation; (2) tuning the optical energy levels; and (3) theoretical and experimental band gap reduction. These measurements and calculations provide a strong rational for our design strategy that the induction of a conjugated functionality can indeed improve the optoelectronic properties of a given chromophore. The energy level diagram advised that the band gaps of these materials are all in the range required of donor materials for BHJ devices. AS2 optical band gap is somewhat narrower in magnitude than 2.0 eV measured for the conventional P3HT. The optical and electrochemical properties of both the materials in solution and film form are summarized in Supplementary Material Table S1.
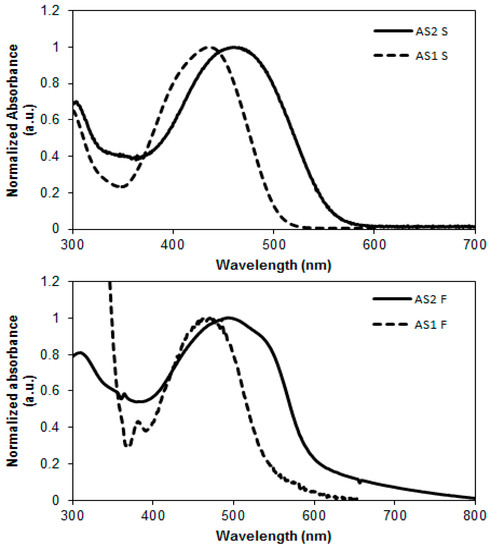
Figure 4.
Normalized absorption spectra of compounds AS2 and AS1 in CHCl3 solutions (upper) and for pristine as-casted films (lower); (films of AS2 and AS1 were spin-coated at 2000 rpm for 1 min to give a film thickness of ~70 nm).
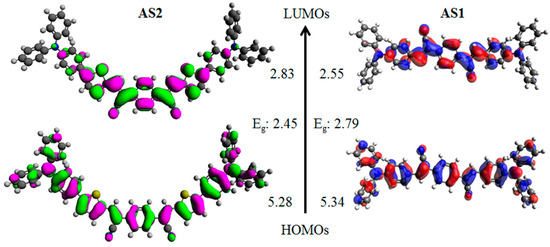
Figure 5.
Orbital density distribution for the frontier molecular orbitals of AS2 and AS1. DFT calculations were performed using the Gaussian 09 suite of programs and the B3LYP/6-311 + G(d,p)//B3LYP/6-31G(d) level of theory. Theoretical HOMO/LUMO energy levels and band-gaps (vs. Vac scale) are also shown.

Figure 6.
Energy level diagram depicting the band gaps of AS2 and AS1 in comparison with P3HT and PC61BM. Experimental estimation of the HOMO energies was carried out using PESA and the LUMO energies were calculated by adding the optical band gap (film spectra) to the HOMO values.
Encouraging though the optoelectronic properties are, the compounds must display thermal stability given the rigorous conditions used in device fabrication such as annealing at temperature in the excess of 100 °C. In line with this requirement and to determine the thermal stability of AS2 and AS1, thermogravimetric analysis (TGA) was conducted. TGA (Supplementary Material Figure S2) indicated that both AS2 and AS1 are thermally stable and will not degrade during the annealing of BHJ devices.
After correlating the optoelectronic properties of AS2 and AS1 with those of the conventional PC61BM acceptor (see Figure 6), we screened their potency as donor materials (p-type) in solution-processable BHJ devices under simulated sunlight and monochromatic light illumination. The blend solutions of both the materials and PC61BM were used to cast an active layer on top of the PEDOT:PSS surface. The BHJ device architecture used was ITO/PEDOT:PSS (38 nm)/active layer/Ca (20 nm)/Al (100 nm) where the active layer was a solution processed blend of either AS2 or AS1 and PC61BM. For AS2, a promising PCE of 4.10% was achieved when the film was spin-coated from a chlorobenzene solution as a 1:1 blend with PC61BM. By contrast, the maximum PCE obtained for a device based on AS1 was 1.23%, when fabricated under similar conditions. The comparative current–voltage curves for the optimized blends of AS2 and AS1 with PC61BM are shown in Figure 7. High boiling solvents such as chlorobenzene are better not only from processing point of view but also for achieving smoother films without crystallization occurring on the active surfaces. Latter was particularly true as our efforts to construct BHJ devices using a low-boiling solvent, such as chloroform, afforded either uneven surfaces or minor cracks on the active surfaces.
The optimized devices based on AS2 exhibited a decreased open circuit voltage (Voc) than the devices based on AS1. This in fact is consistent with the measured HOMO values where a higher HOMO for AS2 would predict a lower Voc. On the other hand, the short circuit current density (Jsc) for the devices fabricated using AS2 was higher than the Jsc extracted from the devices based on AS1. This was in agreement with the observed bathochromic shift in the absorption spectrum of AS2 compared with AS1. The photovoltaic cell parameters for AS2-based devices were 0.88 V [open circuit voltage, Voc], 8.01 mA·cm−2 [current density, (Jsc)], 0.58 [fill factor, (FF)] and 4.10% [power conversion efficiency, (PCE)]. Initial screening of the BHJ devices based on AS1:PC61BM showed moderate device performance with a high Voc of 0.90 V, FF of 43% Jsc of 3.15 mA/cm2 and an overall PCE of 1.23%. Taken as a whole, the insertion of thiophene functionality into the studied D–A–D structural motif incorporating PDA acceptor functionality resulted in significant enhancement of Jsc and PCE values by factors >2 and >3, respectively, thus promoting the use of a smaller conjugated functionality, such as thiophene, as an interesting structural concept for the design and development of highly efficient BHJ materials. Furthermore, it is notable to mention that AS2 showed optimal performance with inexpensive PC61BM and simple device architecture, thus providing some strong incentive to apply the design concept reported in this work to the new generations of BHJ materials.
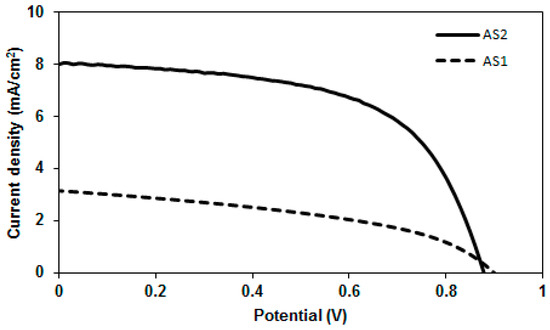
Figure 7.
Current–voltage curves for the optimized devices based on AS2 and AS1 in blends with PC61BM (1:1 w/w) under simulated sunlight (AM 1.5, 1000 W·m−2). Device structure was: ITO/PEDOT-PSS (38 nm)/active layer/Ca (20 nm)/Al (100 nm) where the active layers were the blends of either AS2 or AS1 and PC61BM spun on top of the films of PEDOT:PSS using chlorobenzene solvent.
The incident photon-to-current-conversion efficiency (IPCE) spectra of these BHJ devices are shown in Figure 8. The IPCE measurement of these BHJ devices was broad spectrum, typically covering most of the visible range, from 350 to 750 nm. AS2 and AS1 exhibited high plateaus at ~40% and ~27% for the best BHJ devices respectively. The IPCE spectrum of AS2, which carried a thiophene functionality, was red-shifted when compared with AS1, a finding that is consistent with the result of thin film absorption spectrum. The significantly higher peak IPCE of AS2 compared to AS1 indicated that the superior performance of AS2 can be rationalized in terms of enhanced light-harvesting and appropriately tuned optical energy levels, thereby corroborating the design principle.
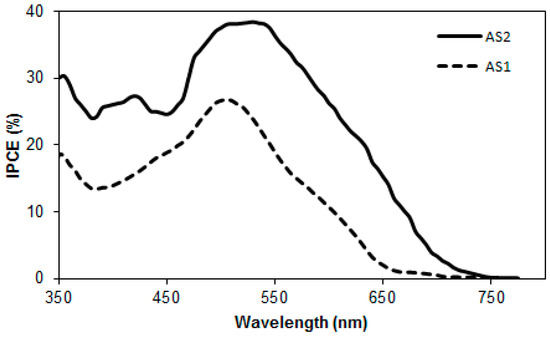
Figure 8.
IPCE spectra of AS2 and AS1 with PC61BMblends.
To examine the physical microstructure of the blend surface, we used atomic force microscopy (AFM) in tapping mode. The actual surface morphology of the blend films of AS2/AS1:PC61BM (1:1 w/w) is shown in Figure 9. Physically, both the blends were found to be smooth and the root mean square roughness of 0.41 nm and 0.35 nm was observed for AS2 and AS1 respectively. No cracks were observed on the film surfaces when the films were spin-casted using chlorobenzene (3000 rpm). The processing of active films of BHJ devices using a high boiling solvent such as chlorobenzene is advantageous over low boiling solvent such as chloroform and is in agreement with the AFM morphology. Our attempts to fabricate BHJ devices using chloroform resulted in very poor photovoltaic performance. This was mainly due to inferior film quality. Though AS2 exerted promising PCE in this preliminary work, ample scope still exists to explore device strategies to enhance PCE. The performance might be improved by (1) using PC71BM or (2) effective interlayer, such as metal oxide interlayer, which can facilitate the efficient charge extraction, and (3) devising processing methods, such as use of additives. Work towards some of such strategies is the subject of on-going work in our laboratories. The discovery of potential materials, such as AS2, exhibiting promising optoelectronic and photovoltaic properties opens up the way to develop D–A–D modular small organic chromophores, with the use of PDA acceptor in particular, and paves the way for such materials to be used for other organic electronic applications.

Figure 9.
AFM images of 1:1 blends of AS2 (top) and AS1 (bottom) with PC61BM as-casted from chlorobenzene solution. Topographic (left) and phase images (right) are shown.
3. Experimental Section
3.1. Materials and Instruments
All reagents and chemicals used, unless otherwise specified, were purchased from Sigma-Aldrich (Sydney, Australia). The solvents used for reactions were obtained from Merck Speciality Chemicals (Sydney, Australia) and were used as received. Unless otherwise specified, all 1H- and 13C-NMR spectra were recorded using a Bruker AV300 spectrometer at 300 and 75 MHz or a Bruker AV400 spectrometer at 400 and 100 MHz, respectively (Bruker Corporation, Billerica, MA, USA). Chemical shifts (δ) are reported in parts per million (ppm). Thin-layer chromatography (TLC) was performed using 0.25 mm thick plates precoated with Kieselgel 60 F254 silica gel (Merck, Darmstadt, Germany) and visualized using UV light (254 nm and 365 nm). Melting points were measured using a MPD350 digital melting point apparatus (Gallenkamp, Sanyo, Osaka, Japan) and are uncorrected. High-resolution mass spectra experiments were carried out on a Q-Exactive FTMS (Thermo Scientific, Bremen, Germany) ionizing by atmospheric-pressure chemical ionization (APCI) from an ASAP probe. All UV-Vis absorption spectra were recorded on a Hewlett Packard (HP) 8453 diode array UV-Vis spectrophotometer (Agilent Technologies, Mulgrave Victoria, Australia). Thin films were spin-coated from chlorobenzene at a spin speed of 2000 rpm for 1 min onto cleaned glass slides. PESA measurement was recorded using a Riken Keiki AC-2 PESA spectrometer (RKI Instruments, Union City, CA, USA) with a power setting of 5 nW and a power number of 0.5. Samples for PESA were prepared on clean glass substrates. Fabrication and characterization of BHJ devices, and preparation of thin-film transistors has been reported previously [18,30].
3.2. Synthesis and Characterization of Target Molecules
3.2.1. Synthesis of AS1
1,4-Phenylenediacetonitrile (500 mg, 3.21 mmol) was added to the mixture of 4-(diphenyl-amino)benzaldehyde (1.84 g, 6.74 mmol) in methanol (50.0 mL) at room temperature and the resulting mixture was heated at reflux overnight. The precipitated solid was collected by filtration, washed with methanol and dried under vacuum to give 1.85 g (86.3%) of AS1 as an orange powder. m. p. 242–245 °C; HPLC (5% H2O/ACN): 97.6%; IR (solid film, cm−1) 3061, 3035 (Ar -CH str), 2250, 2206 (-CN str), 1580, 1504, 1486 (Ar C=C str), 1330, 1285, 1192, 1179; 1H-NMR (400 MHz, CDCl3): δ = 7.80–7.76 (m, 4 H), 7.68 (s, 4 H), 7.45 (s, 2 H), 7.33–7.28 (m, 8 H), 7.16–7.10 (m, 12 H), 7.05–7.02 (m, 4 H); 13C-NMR (400 MHz, CDCl3): δ = 150.2, 146.5, 141.8, 135.1, 130.8, 129.6, 126.2, 126.1, 125.8, 124.5, 120.7, 118.5, 106.6; HRMS (APCI): calculated for C48H35N4 [M+H]+ 667.2856; found 667.2851.
3.2.2. Synthesis of 5-(4-(Diphenylamino)phenyl)thiophene-2-carbaldehyde (1)
A solution of 1,2-dimethoxyethane (DME, 40.0 mL) and 2M Na2CO3 (20.0 mL) is degassed with nitrogen (N2) for 30 min. To this solution 5-bromothiophene-2-carbaldehyde (191 mg, 1.00 mmol) and (4-(diphenylamino)phenyl)boronic acid (433 mg, 1.50 mmol) were added and the mixture was heated at 60 °C for 30 min. [Pd(PPh3)4] (110 mg, 0.10 mmol) was added and the resulting mixture was stirred at 90 °C overnight. The reaction mixture was extracted with diethyl ether (3 × 50 mL). The organic layers were combined, washed with brine (100 mL) and dried over anhydrous magnesium sulfate. The solvent was evaporated to afford crude yellow solid, which was crystallized from hexane and chloroform to yield 378 mg (71%) of 1 as yellow needle like crystals. m. p. 100–102 °C; IR (solid film, cm−1): 3313, 3023, 2794, 1956, 1887, 1720, 1659, 1584, 1527, 1485, 1465, 1324, 1261, 1226, 1177, 1153, 1075, 1054; 1H-NMR (300 MHz, CD2Cl2): δ = 9.87 (s, 1H), 7.75 (d, J = 6.8 Hz, 1H), 7.60–7.57 (m, 2H), 7.37–7.31 (m, 5H), 7.71–7.15 (m, 6H), 7.13–7.06 (m, 2H); 13C-NMR (300 MHz, CDCl3): δ = 182.9, 154.5, 149.5, 147.4, 141.8, 138.1, 129.8, 127.6, 126.5, 125.6, 124.3, 123.4, 122.6; LRMS (ESI; 2% Formic acid): m/z = 356 (M + H)+; HRMS (APCI): calculated for C23H17NOS [M]+ 355.1031; found 355.1026.
3.2.3. Synthesis of AS2
A solution of sodium ehtoxide was prepared by dissolving one pellet (approx. 200 mg) of sodium hydroxide (NaOH) in ethanol (EtOH, 20 mL) with mild heating (40 °C). To this solution PDA (78 mg, 0.50 mmol) was added and allowed to dissolve. A solution of 1 (390 mg, 1.1 mmol) in EtOH (10 mL) was added dropwise and the resulting solution was refluxed for 6 h. The mixture was allowed to cool to room temperature and placed in an ice-bath for 30 min. The separated red solid was filtered off, washed with cold EtOH (50.0 mL) followed by cold hexane (50.0 mL), and dried under high vacuum at 40 °C. The solid was crystallized using CHCl3/hexane to afford 574 mg of AS2 (63%) as a brick-red solid. m. p. >240 °C (decomposed at 240 °C); IR (solid film, cm−1) 3674, 2988, 2208 (-CN str), 1578, 1486, 1440, 1421, 1324, 1271, 1238, 1179, 1064, 922, 829. 1H-NMR (300 MHz, CD2Cl2): δ = 7.77–7.76 (m, 3H), 7.65–7.63 (m, 1H), 7.60–7.59 (m, 2H), 7.36–7.31 (m, 5H) 7.18–7.15 (m, 5H), 7.12–7.08 (m, 3H); 13C-NMR (300 MHz, CDCl3) δ = 149.9, 148.6, 147.1, 136.1, 134.8, 134.7, 134.4, 129.4, 127.0, 126.7, 126.0, 124.9, 123.6, 122.8, 122.7, 118.1, 105.7; LRMS (MALDI-TOF): m/z = 830.229; HRMS (APCI): calculated for C56H39N4S2 [M + H]+ 831.2611; found 831.2602.
3.2.4. X-ray Crystallography
CCDC’s 1420377 for 1 and 1420378 for AS2 contain the supplementary crystallographic data for this paper. These data can be obtained free of charge via http://www.ccdc.cam.ac.uk/conts/retrieving.html (or from the CCDC, 12 Union Road, Cambridge CB2 1EZ, UK; Fax: +44 1223 336033; E-mail: deposit@ccdc.cam.ac.uk).
4. Conclusions
In conclusion, we have demonstrated the first use of the PDA acceptor functionality in conjunction with a thiophene unit for the design and development of a BHJ chromophore, AS2, where PDA was used in the D–A–D modular arrangement. The incorporation of this strong conjugating thiophene unit helped to improve light-harvesting, photocurrent density and PCE of AS2 when compared with an analogue, AS1. The incorporation of the thiophene functionality was of clear benefit in improving the BHJ performance and indicates a potential to be broadly applicable in the design and development of future high performance BHJ chromophores.
Supplementary Materials
Supplementary materials can be accessed at: http://www.mdpi.com/1420-3049/20/12/19798/s1.
Acknowledgments
S.V.B. (RMIT) acknowledges financial support from the Australian Research Council (ARC), Australia, under a Future Fellowship Scheme (FT110100152). The CSIRO Division of Materials Science and Engineering, Clayton, Victoria is acknowledged for providing support through a visiting fellow position (A.G.). P.S. is thankful to the ARC Future Fellowship Scheme (FT130101337) at Queensland University of Technology, Brisbane, Queensland.
Author Contributions
A.M.R. synthesis, characterization, properties of materials and fabrication of devices; S.L.J and C.M.P.: single crystal structure determinations; A.G. and H.P. device fabrication and characterization; A.B.: performance of DFT calculation; P.S: results and discussion part, and S.V.B.: design, supervision and analysis of data. All the authors contributed for the manuscript preparation.
Conflicts of Interest
The authors declare no conflict of interests.
References
- Chen, Y.; Wan, X.; Long, G. High performance photovoltaic applications using solution-processed small molecules. Acc. Chem. Res. 2013, 46, 2645–2655. [Google Scholar] [CrossRef] [PubMed]
- Duan, C.; Huang, F.; Cao, Y. Recent development of push–pull conjugated polymers for bulk-heterojunction photovoltaics: Rational design and fine tailoring of molecular structures. J. Mater. Chem. 2012, 22, 10416–10434. [Google Scholar] [CrossRef]
- Günes, S.; Neugebauer, H.; Sariciftci, N.S. Conjugated polymer-based organic solar cells. Chem. Rev. 2007, 107, 1324–1338. [Google Scholar] [CrossRef] [PubMed]
- Lin, Y.; Li, Y.; Zhan, X. Small molecule semiconductors for high-efficiency organic photovoltaics. Chem. Soc. Rev. 2012, 41, 4245–4272. [Google Scholar] [CrossRef] [PubMed]
- Huang, Y.; Kramer, E.J.; Heeger, A.J.; Bazan, G.C. Bulk heterojunction solar cells: Morphology and performance relationships. Chem. Rev. 2014, 114, 7006–7043. [Google Scholar] [CrossRef] [PubMed]
- Heegar, A.J. 25th Anniversary article: Bulk heterojunction solar cells: Understanding the mechanism of operation. Adv. Mater. 2014, 26, 10–28. [Google Scholar] [CrossRef] [PubMed]
- Scharber, M.C.; Sariciftci, N.S. Efficiency of bulk-heterojunction organic solar cells. Prog. Polym. Sci. 2013, 38, 1929–1940. [Google Scholar] [CrossRef] [PubMed]
- Boudreault, P.-L.T.; Najari, A.; Leclerc, M. Processable Low-Bandgap Polymers for Photovoltaic Applications. Chem. Mater. 2011, 23, 456–469. [Google Scholar] [CrossRef]
- Facchetti, A. π-Conjugated polymers for organic electronics and photovoltaic cell applications. Chem. Mater. 2011, 23, 733–758. [Google Scholar] [CrossRef]
- Gupta, A.; Watkins, S.E.; Scully, A.D.; Singh, T.B.; Wilson, G.J.; Rozanski, L.J.; Evans, R.A. Band-gap tuning of pendant polymers for organic light-emitting devices and photovoltaic applications. Synth. Met. 2011, 161, 856–863. [Google Scholar] [CrossRef]
- He, Y.; Li, Y. Fullerene derivative acceptors for high performance polymer solar cells. Phys. Chem. Chem. Phys. 2011, 13, 1970–1983. [Google Scholar] [CrossRef] [PubMed]
- Laquai, F.; Andrienko, D.; Mauer, R.; Blom, P.W.M. Charge carrier transport and photogeneration in P3HT:PCBM photovoltaic blends. Macromol. Rapid Commun. 2015, 36, 1001–1025. [Google Scholar] [CrossRef] [PubMed]
- Dang, M.T.; Hirsch, L.; Wantz, G.; Wuest, J.D. Controlling the morphology and performance of bulk heterojunctions in solar cells. Lessons learned from the benchmark poly(3-hexylthiophene):[6,6]-phenyl-C61-butyric acid methyl ester system. Chem. Rev. 2013, 113, 3734–3765. [Google Scholar] [CrossRef] [PubMed]
- You, J.; Chen, C.-C.; Hong, Z.; Yoshimura, K.; Ohya, K.; Xu, R.; Ye, S.; Gao, J.; Li, G.; Yang, Y. 10.2% Power conversion efficiency polymer tandem solar cells consisting of two identical sub-cells. Adv. Mater. 2013, 25, 3973–3978. [Google Scholar] [CrossRef] [PubMed]
- You, J.; Dou, L.; Yoshimura, K.; Kato, T.; Ohya, K.; Moriarty, T.; Emery, K.; Chen, C.-C.; Gao, J.; Li, G.; et al. A polymer tandem solar cell with 10.6% power conversion efficiency. Nat. Commun. 2013, 4. [Google Scholar] [CrossRef] [PubMed]
- Lu, L.; Zheng, T.; Wu, Q.; Schneider, A.M.; Zhao, D.; Yu, L. Recent advances in bulk heterojunction polymer solar cells. Chem. Rev. 2015. [Google Scholar] [CrossRef] [PubMed]
- Guo, X.; Baumgarten, M.; Muellen, K. Designing π-conjugated polymers for organic electronics. Prog. Polym. Sci. 2013, 38, 1832–1908. [Google Scholar] [CrossRef]
- Gupta, A.; Ali, A.; Bilic, A.; Gao, M.; Hegedus, K.; Singh, B.; Watkins, S.E.; Wilson, G.J.; Bach, U.; Evans, R.A. Absorption enhancement of oligothiophene dyes through the use of a cyanopyridone acceptor group in solution-processed organic solar cells. Chem. Commun. 2012, 48, 1889–1891. [Google Scholar] [CrossRef] [PubMed]
- Kumar, R.J.; Churches, Q.I.; Subbiah, J.; Gupta, A.; Ali, A.; Evans, R.A.; Holmes, A.B. Enhanced photovoltaic efficiency via light-triggered self-assembly. Chem. Commun. 2013, 49, 6552–6554. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Guo, Q.; Li, Z.; Pei, J.; Tian, W. Solution processable D-A small molecules for bulk-heterojunction solar cells. Energy Environ. Sci. 2010, 3, 1427–1436. [Google Scholar] [CrossRef]
- Mishra, A.; Bäuerle, P. Small molecule organic semiconductors on the move: Promises for future solar energy technology. Angew. Chem. Int. Ed. 2012, 51, 2020–2067. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Walker, B.; Nguyen, T.-Q. Solution-processed molecular bulk heterojunction solar cells. In Organic Photovoltaics, 2nd ed.; Brabec, C.J., Scherf, U., Dyakonov, V., Eds.; Wiley-VCH Verlag GmbH & Co. KGaA: Weinheim, Germany, 2014; pp. 95–137. [Google Scholar]
- Roncali, J.; Leriche, P.; Blanchard, P. Molecular materials for organic photovoltaics: Small is beautiful. Adv. Mater. 2014, 26, 3821–3838. [Google Scholar] [CrossRef] [PubMed]
- Kan, B.; Zhang, Q.; Li, M.; Wan, X.; Ni, W.; Long, G.; Wang, Y.; Yang, X.; Feng, H.; Chen, Y. Solution-processed organic solar cells based on dialkylthiol-substituted benzodithiophene unit with efficiency near 10%. J. Am. Chem. Soc. 2014, 136, 15529–15532. [Google Scholar] [CrossRef] [PubMed]
- Patil, H.; Gupta, A.; Bilic, A.; Jackson, S.L.; Latham, K.; Bhosale, S.V. Donor-acceptor-donor modular small organic molecules based on the naphthalene diimide acceptor unit for solution-processable photovoltaic devices. J. Electron. Mater. 2014, 43, 3243–3254. [Google Scholar] [CrossRef]
- Sonar, P.; Williams, E.T.; Singh, S.P.; Manzhos, S.; Dodabalapur, A. A benzothiadiazole end capped donor-acceptor based small molecule for organic electronics. Phys. Chem. Chem. Phys. 2013, 15, 17064–17069. [Google Scholar] [CrossRef] [PubMed]
- Walker, B.; Tamayo, A.B.; Dang, X.-D.; Zalar, P.; Seo, J.H.; Garcia, A.; Tantiwiwat, M.; Nguyen, T.-Q. Nanoscale phase separation and high photovoltaic efficiency in solution-processed, small-molecule bulk heterojunction solar cells. Adv. Funct. Mater. 2009, 19, 3063–3069. [Google Scholar] [CrossRef]
- Loser, S.; Bruns, C.J.; Miyauchi, H.; Ortiz, R.O.P.; Facchetti, A.; Stupp, S.I.; Marks, T.J. A naphthodithiophene-diketopyrrolopyrrole donor molecule for efficient solution-processed solar cells. J. Am. Chem. Soc. 2011, 133, 8142–8145. [Google Scholar] [CrossRef] [PubMed]
- Yong, W.; Zhang, M.; Xin, X.; Li, Z.; Wu, Y.; Guo, X.; Yang, Z.; Hou, J. Solution-processed indacenodithiophene-based small molecule for bulk heterojunction solar cells. J. Mater. Chem. A 2013, 1, 14214–14220. [Google Scholar] [CrossRef]
- Gupta, A.; Ali, A.; Gao, M.; Singh, T.B.; Bilic, A.; Watkins, S.E.; Bach, U.; Evans, R.A. Small molecules containing rigidified thiophenes and a cyanopyridone acceptor unit for solution-processable bulk-heterojunction solar cells. Dyes Pigm. 2015, 119, 122–132. [Google Scholar] [CrossRef]
- Yuan, M.-S.; Wang, Q.; Wang, W.-J.; Li, T.-B.; Wang, L.; Deng, W.; Du, Z.-T.; Wang, J.-R. Symmetrical and asymmetrical (multi)branched truxene compounds: Structure and photophysical properties. Dyes Pigm. 2012, 95, 236–243. [Google Scholar] [CrossRef]
- Li, Z.; Dong, Q.; Li, Y.; Xu, B.; Deng, M.; Pei, J.; Zhang, J.; Chen, F.; Wen, S.; Gao, Y.; et al. Design and synthesis of solution processable small molecules towards high photovoltaic performance. J. Mater. Chem. 2011, 21, 2159–2168. [Google Scholar] [CrossRef]
- Shi, Q.; Cheng, P.; Li, Y.; Zhan, X. A solution processable D-A-D molecule based on thiazolothiazole for high performance organic solar cells. Adv. Energy Mater. 2012, 2, 63–67. [Google Scholar] [CrossRef]
- Gupta, A.; Hangarge, R.V.; Wang, X.; Alford, B.; Chellapan, V.; Jones, L.A.; Rananaware, A.; Bilic, A.; Sonar, P.; Bhosale, S.V. Crowning of dibenzosilole with a naphthalenediimide functional group to prepare an electron acceptor for organic solar cells. Dyes Pigm. 2015, 120, 314–321. [Google Scholar] [CrossRef]
- Patil, H.; Gupta, A.; Alford, B.; Ma, D.; Privér, S.H.; Bilic, A.; Sonar, P.; Bhosale, S.V. Conjoint use of dibenzosilole and indan-1,3-dione functionalities to prepare an efficient non-fullerene acceptor for solution-processable bulk-heterojunction solar cells. Asian J. Org. Chem. 2015. [Google Scholar] [CrossRef]
- Patil, H.; Zu, W.X.; Gupta, A.; Chellappan, V.; Bilic, A.; Sonar, P.; Rananaware, A.; Bhosale, S.V.; Bhosale, S.V. A non-fullerene electron acceptor based on fluorene and diketopyrrolopyrrole building blocks for solution-processable organic solar cells with an impressive open-circuit voltage. Phys. Chem. Chem. Phys. 2014, 16, 23837–23842. [Google Scholar] [CrossRef] [PubMed]
- Raynor, A.M.; Gupta, A.; Patil, H.; Bilic, A.; Bhosale, S.V. A diketopyrrolopyrrole and benzothiadiazole based small molecule electron acceptor: Design, synthesis, characterization and photovoltaic properties. RSC Adv. 2014, 4, 57635–57638. [Google Scholar] [CrossRef]
- Gupta, A.; Ali, A.; Singh, T.B.; Bilic, A.; Bach, U.; Evans, R.A. Molecular engineering for panchromatic absorbing oligothiophene donor-π-acceptor organic semiconductors. Tetrahedron 2012, 68, 9440–9447. [Google Scholar] [CrossRef]
- Kronenberg, N.M.; Deppisch, M.; Würthner, F.; Lademann, H.W.A.; Deing, K.; Meerholz, K. Bulk heterojunction organic solar cells based on merocyanine colorants. Chem. Commun. 2008, 48, 6489–6491. [Google Scholar] [CrossRef] [PubMed]
- Grimsdale, A.C.; Chan, K.L.; Martin, R.E.; Jokisz, P.G.; Holmes, A.B. Synthesis of light-emitting conjugated polymers for applications in electroluminescent devices. Chem. Rev. 2009, 109, 897–1091. [Google Scholar] [CrossRef] [PubMed]
- Halliday, D.A.; Burn, P.L.; Friend, R.H.; Bradley, D.D.C.; Holmes, A.B. Determination of the average molecular weigth of poly(p-phenylenevinylene). Synth. Met. 1993, 55, 902–907. [Google Scholar] [CrossRef]
- Pond, S.J.K.; Rumi, M.; Levin, M.D.; Parker, T.C.; Beljonne, D.; Day, M.W.; Brédas, J.-L.; Marder, S.R.; Perry, J.W. One- and two-photon spectroscopy of donor−acceptor−donor distyrylbenzene derivatives: Effect of cyano substitution and distortion from planarity. J. Phys. Chem. A 2002, 106, 11470–11480. [Google Scholar] [CrossRef]
- Maurin, M.; Vurth, L.; Vial, J.-C.; Baldeck, P.; Marder, S.R.; van der Sanden, B.; Stephan, O. Pluronic organic fluorescent probes for two-photon vascularisation imaging. Nonlinear Opt. Quantum Opt. 2010, 40, 175–181. [Google Scholar]
- Fang, H.-H.; Chen, Q.-D.; Yang, J.; Xia, H.; Gao, B.-R.; Feng, J.; Ma, Y.-G.; Sun, H.-B. Two-photon pumped amplified spontaneous emission from cyano-substituted oligo(p-phenylenevinylene) crystals with aggregation-induced emission enhancement. J. Phys. Chem. C 2010, 114, 11958–11961. [Google Scholar] [CrossRef]
- Yan, Z.-Q.; Yang, Z.-Y.; Wang, H.; Li, A.-W.; Wang, L.-P.; Yang, H.; Gao, B.-R. Study of aggregation induced emission of cyano-substituted oligo(p-phenylenevinylene) by femtosecond time resolved fluorescence. Spectrochim Acta A Mol Biomol Spectrosc. 2011, 78, 1640–1645. [Google Scholar] [CrossRef] [PubMed]
- Maurin, M.; Stephan, O.; Vial, J.-C.A.; Marder, S.R.; van der Sanden, B.P.J. Deep in vivo two-photon imaging of blood vessels with a new dye encapsulated in pluronic nanomicelles. J. Biomed. Opt. 2011, 16, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Xiang, W.; Gupta, A.; Kashif, M.K.; Duffy, N.; Bilic, A.; Evans, R.A.; Spiccia, L.; Bach, U. Cyanomethylbenzoic acid: An acceptor for donor–π–acceptor chromophores used in dye-sensitized solar cells. ChemSusChem 2013, 6, 256–260. [Google Scholar] [CrossRef] [PubMed]
- Bobe, S.R.; Gupta, A.; Rananaware, A.; Bilic, A.; Bhosale, S.V.; Bhosale, S.V. Improvement of optoelectronic and photovoltaic properties through the insertion of a naphthalenediimide unit in donor–acceptor oligothiophenes. RSC Adv. 2015, 5, 4411–4415. [Google Scholar] [CrossRef]
- Würthner, F.; Wortmann, R.; Matschiner, R.; Lukaszuk, K.; Meerholz, K.; DeNardin, Y.; Bittner, R.; Brauchle, C.; Sens, R. Merocyanine dyes in the cyanine limit: A new class of chromophores for photorefractive materials. Angew. Chem. Int. Ed. 1997, 36, 2765–2768. [Google Scholar] [CrossRef]
- Gupta, A.; Armel, V.; Xiang, W.; Fanchini, G.; Watkins, S.E.; MacFarlane, D.R.; Bach, U.; Evans, R.A. The effect of direct amine substituted push-pull oligothiophene chromophores on dye-sensitized and bulk heterojunction solar cells performance. Tetrahedron 2013, 69, 3584–3592. [Google Scholar] [CrossRef]
- Sheldrick, G.M. A short history of SHELX. Acta. Cryst. 2008, 64, 112–122. [Google Scholar] [CrossRef] [PubMed]
- Dolomanov, O.V.; Bourhis, L.J.; Gildea, R.J.; Howard, J.A.K.; Puschmann, H. OLEX2: A complete structure solution, refinement and analysis program. J. Appl. Cryst. 2009, 42, 339–341. [Google Scholar] [CrossRef]
- Sheldrick, G.M. Crystal structure refinement with SHELXL. Struct. Chem. 2014, 71, 3–8. [Google Scholar]
- Hagberg, D.P.; Marinado, T.; Karlsson, K.M.; Nonomura, K.; Qin, P.; Boschloo, G.; Brinck, T.; Hagfeldt, A.; Sun, L. Tuning the HOMO and LUMO energy levels of organic chromophores for dye sensitized solar cells. J. Org. Chem. 2007, 72, 9550–9556. [Google Scholar] [CrossRef] [PubMed]
- Frisch, M.J.; Trucks, G.W.; Schlegel, H.B.; Scuseria, G.E.; Robb, M.A.; Cheeseman, J.R.; Scalmani, G.; Barone, V.; Mennucci, B.; Petersson, G.A.; et al. Gaussian 09, revision D.01; Gaussian Incorporation: Wallingford, CT, USA, 2013. [Google Scholar]
- Sample Availability: Samples of the compounds S10 and S11 are available from the authors.
© 2015 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons by Attribution (CC-BY) license ( http://creativecommons.org/licenses/by/4.0/).