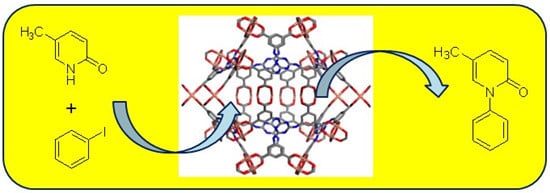
A Copper-Based Metal-Organic Framework as an Efficient and Reusable Heterogeneous Catalyst for Ullmann and Goldberg Type C–N Coupling Reactions
Abstract
:1. Introduction
2. Results and Discussion
2.1. Cu-TDPAT as a Solid Catalyst for the N-Arylation of 5-Methyl-2-(1H)-Pyridone
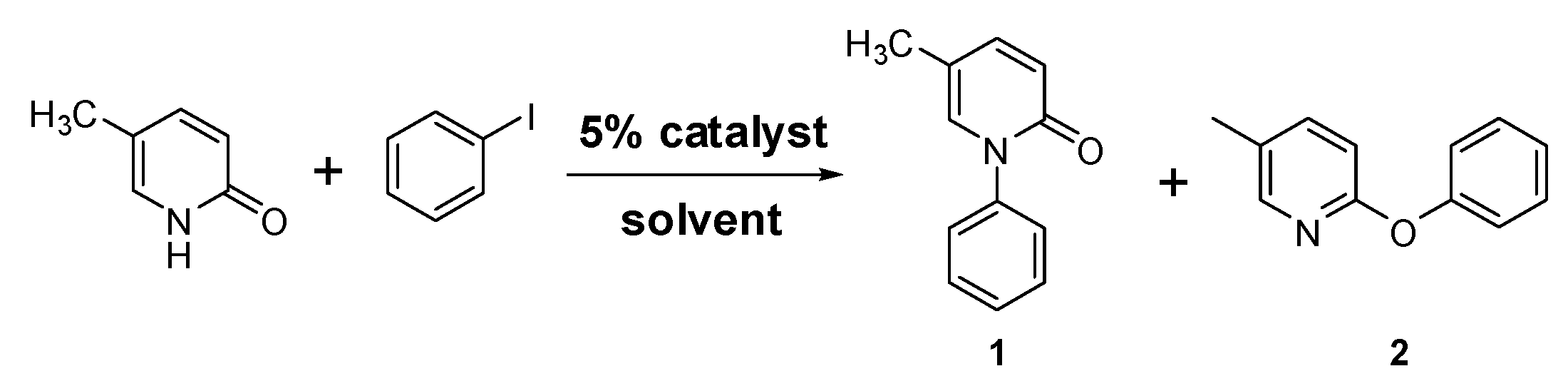
| Entry | Catalyst | Solvent | Base | T (°C) | 1 (%) | 2 (%) |
|---|---|---|---|---|---|---|
| 1 | \ | DMSO | K2CO3 | 120 | n.r | n.r |
| 2 | Cu-TDPAT | DMSO | K2CO3 | 80 | n.r | n.r |
| 3 | Cu-TDPAT | DMSO | K2CO3 | 100 | 9 | 1 |
| 4 | Cu-TDPAT | DMSO | K2CO3 | 120 | 63 (90 b) | 3 (4 b) |
| 5 | Cu-TDPAT | DMSO | K2CO3 | 140 | 70 | 3 |
| 6 | Cu-TDPAT | DMSO | K2CO3 | 160 | 14 | 1 |
| 7 | Cu-TDPAT | Toluene | K2CO3 | 120 | n.r | n.r |
| 8 | Cu-TDPAT | 1,2-Dichlorobenzene | K2CO3 | 120 | n.r | n.r |
| 9 | Cu-TDPAT | DMF | K2CO3 | 120 | 34 | 2 |
| 10 | Cu-TDPAT | 1,4-Dioxane | K2CO3 | 120 | n.r | n.r |
| 11 | Cu-TDPAT | DMSO | KOH | 120 | 61 | 3 |
| 12 | Cu-TDPAT | DMSO | Cs2CO3 | 120 | 57 | 3 |
| 13 | Cu-TDPAT | DMSO | Et3N | 120 | n.r | n.r |
| 14 | Cu-TDPAT | DMSO | NaOMe | 120 | 49 | 4 |
| 15 | CuI | DMSO | K2CO3 | 120 | 80 | 4 |
| 16 | CuCl | DMSO | K2CO3 | 120 | 71 | 5 |
| 17 | Cu(OAc)2 | DMSO | K2CO3 | 120 | 38 | 4 |
| 18 | Cu(NO3)2 | DMSO | K2CO3 | 120 | 29 | 2 |
| 19 | CuBTC | DMSO | K2CO3 | 120 | 88 | 5 |
| 20 | Cu(NO3)2 + BTC | DMSO | K2CO3 | 120 | 43 | 2 |
| 21 | Cu(NO3)2 + TDPAT | DMSO | K2CO3 | 120 | 31 | 2 |
2.2. Heterogeneity of the Reaction
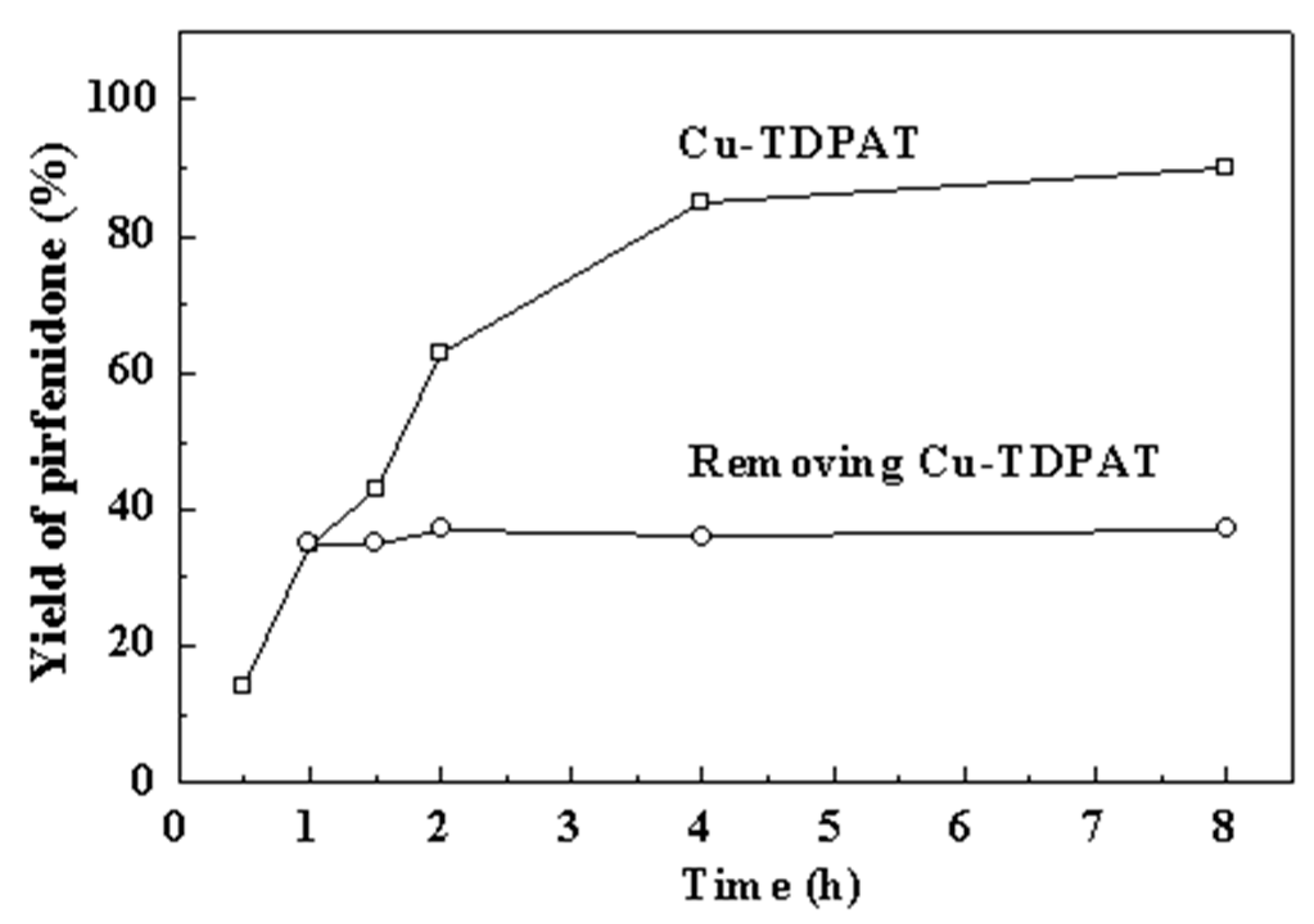
2.3. Comparison with Other Homogeneous Copper Catalysts
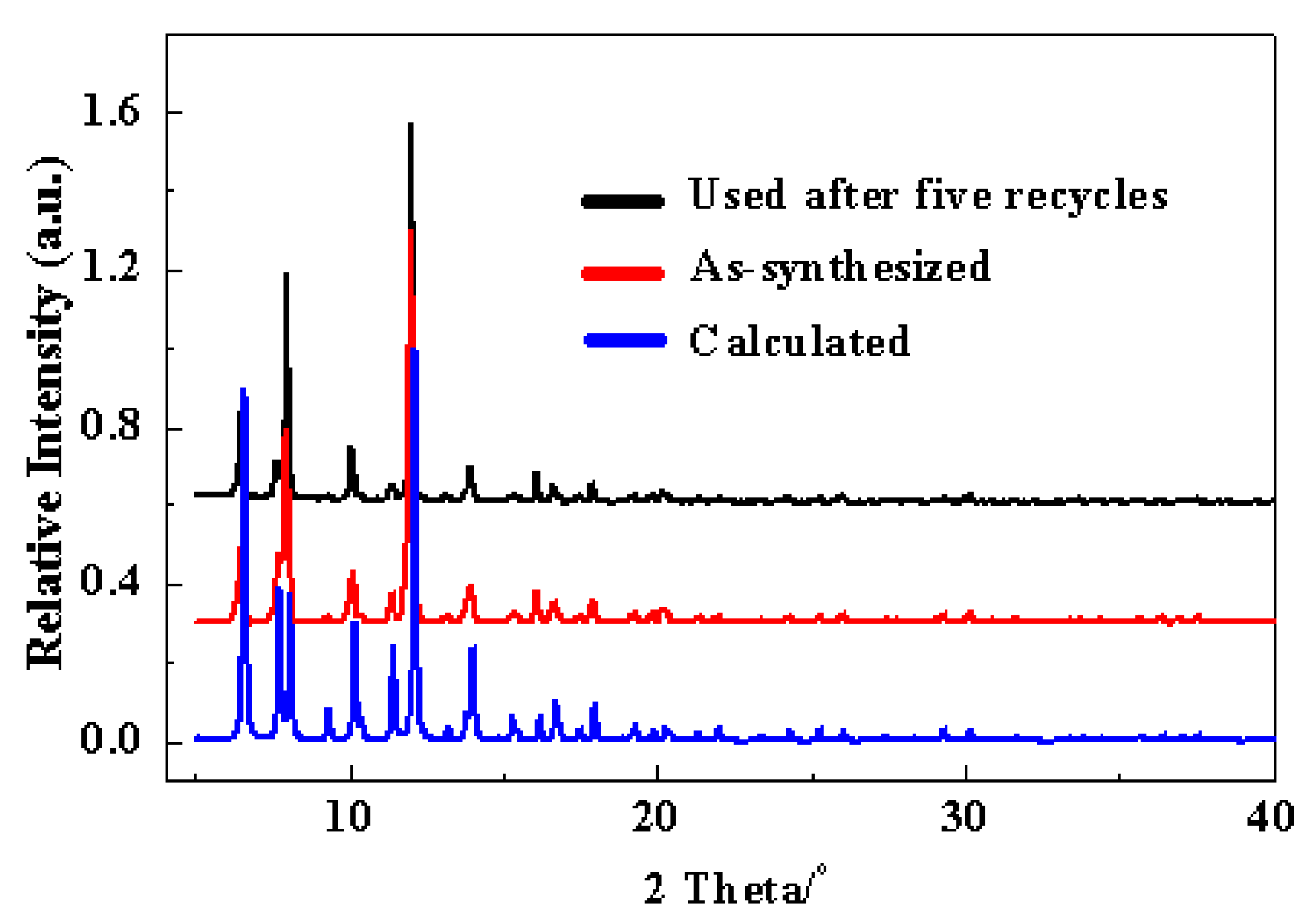
2.4. Reusability of the Cu-TDPAT Catalyst
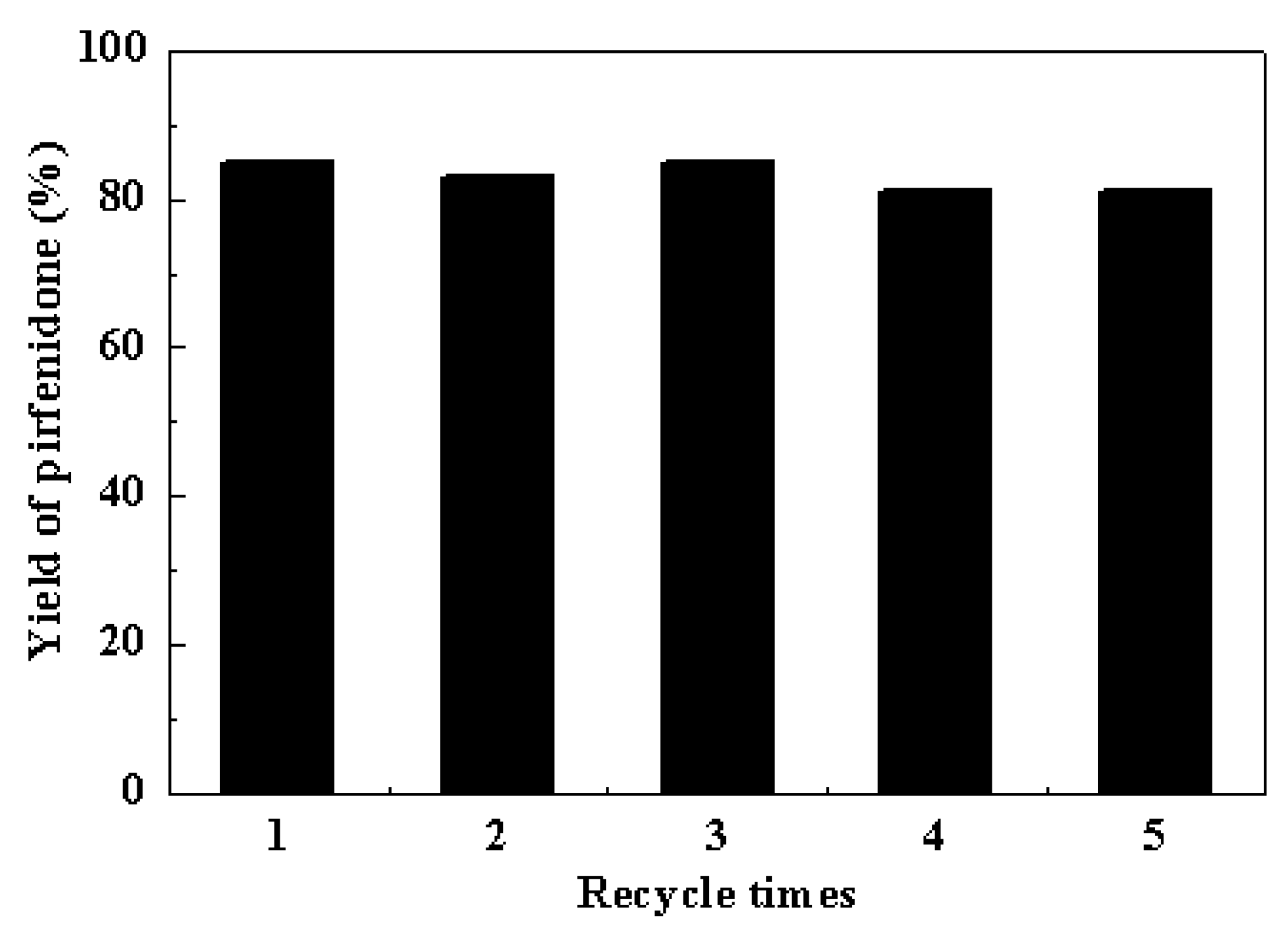
2.5. Generality of the N-Arylation Reaction
| Entry | (R1R2)NH | ArX | Yield (%) |
|---|---|---|---|
| 1 |  | 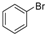 | 39 (2 a) |
| 2 |  |  | 37 |
| 3 | 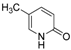 | 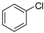 | n.r |
| 4 | 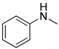 | 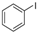 | 51 |
| 5 |  |  | 54 |
| 6 |  |  | 43 |
| 7 | 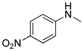 |  | 35 |
| 8 | 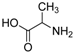 |  | 59 |
| 9 | 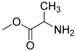 |  | 55 |
| 10 |  |  | 56 |
| 11 | 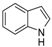 |  | 72 (93 b) |
| 12 | 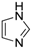 |  | 61 (91 b) |
| Catalyst * | Reaction Conditions | Yield | Reference |
|---|---|---|---|
| CuI/Meso-N-C-1 | DMSO, KOH, 125 °C, 24 h | 88% | [42] |
| CuI/PSP | H2O, K3PO4, 120 °C, 8 h | 65% | [48] |
| Cu-MPTA-1 | H2O, KOH, 120 °C, 12 h | 91% | [52] |
| Cu/SiO2 | Toluene, Cs2CO3, 100 °C, 8 h | 23%–92% | [44] |
| Cu-TDPAT | DMSO, K2CO3, 120 °C, 8 h | 91% | This study |
2.6. Mechanistic Considerations
3. Experimental Section
3.1. General Information
3.2. Catalytic Reactions
3.3. Reuse of Cu-TDPAT
4. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Bikker, J.A.; Brooijmans, N.; Wissner, A.; Mansour, T.S. Kinase domain mutations in cancer: Implications for small molecule drug design strategies. J. Med. Chem. 2009, 52, 1493–1509. [Google Scholar] [CrossRef] [PubMed]
- Balle, T.; Perregaard, J.; Ramirez, M.T.; Larsen, A.K.; Soby, K.K.; Liljefors, T.; Andersen, K. Synthesis and structure-affinity relationship investigations of 5-heteroaryl-substituted analogues of the antipsychotic sertindole. A new class of highly selective α1 adrenoceptor antagonists. J. Med. Chem. 2003, 46, 265–283. [Google Scholar] [CrossRef] [PubMed]
- Popowycz, F.; Routier, S.; Joseph, B.; Merour, J.Y. Synthesis and reactivity of 7-azaindole (1H-pyrrolo[2,3-b]pyridine). Tetrahedron 2007, 63, 1031–1064. [Google Scholar] [CrossRef]
- Surry, D.S.; Buchwald, S.L. Diamine ligands in copper-catalyzed reactions. Chem. Sci. 2010, 1, 13–31. [Google Scholar] [CrossRef] [PubMed]
- Monnier, F.; Taillefer, M. Catalytic C–C, C–N, and C–O Ullmann-type coupling reactions. Angew. Chem. Int. Ed. 2009, 48, 6954–6971. [Google Scholar] [CrossRef] [PubMed]
- Ma, D.W.; Cai, Q. Copper/amino acid catalyzed cross-couplings of aryl and vinyl halides with nucleophiles. Acc. Chem. Res. 2008, 41, 1450–1460. [Google Scholar] [CrossRef] [PubMed]
- Casitas, A.; Ribas, X. The role of organometallic copper(III) complexes in homogeneous catalysis. Chem. Sci. 2013, 4, 2301–2318. [Google Scholar] [CrossRef]
- Klapars, A.; Antilla, J.C.; Huang, X.H.; Buchwald, S.L. A general and efficient copper catalyst for the amidation of aryl halides and the N-arylation of nitrogen heterocycles. J. Am. Chem. Soc. 2001, 123, 7727–7729. [Google Scholar] [CrossRef] [PubMed]
- Taillefer, M.; Cristau, H.J.; Cellier, P.; Spindler, J.F. Method for Forming a Carbon-Carbon or Carbon-Heteroatom Linkage. WO Patent 053,885-A, 20 December 2002. [Google Scholar]
- Klapars, A.; Huang, X.; Buchwald, S.L. A general and efficient copper catalyst for the amidation of aryl halides. J. Am. Chem. Soc. 2002, 124, 7421–7428. [Google Scholar] [CrossRef] [PubMed]
- Audisio, D.; Messaoudi, S.; Peyrat, J.B.; Brion, J.D.; Alami, M. A general copper powder-catalyzed Ullmann-type reaction of 3-halo-4(1H)-quinolones with various nitrogen-containing nucleophiles. J. Org. Chem. 2011, 76, 4995–5005. [Google Scholar] [CrossRef] [PubMed]
- Zhou, F.T.; Guo, J.J.; Liu, J.G.; Ding, K.; Yu, S.Y.; Cai, Q. Copper-catalyzed desymmetric intramolecular Ullmann C–N coupling: An enantioselective preparation of indulines. J. Am. Chem. Soc. 2012, 134, 14326–14329. [Google Scholar] [CrossRef] [PubMed]
- Antila, J.C.; Klapars, A.; Buchwald, S.L. The copper-catalyzed N-arylation of indoles. J. Am. Chem. Soc. 2002, 124, 11684–11688. [Google Scholar] [CrossRef]
- Ma, D.; Zhang, Y.; Yao, J.; Wu, S.; Tao, F. Accelerating effect induced by the structure of R-amino acid in the copper-catalyzed coupling reaction of aryl halides with R-amino acids. Synthesis of benzolactam-V8. J. Am. Chem. Soc. 1998, 120, 12459–12467. [Google Scholar] [CrossRef]
- Kim, J.; Chang, S. Ammonium salts as an inexpensive and convenient nitrogen source in the Cu-catalyzed amination of aryl halides at room temperature. Chem. Commun. 2008, 26, 3052–3054. [Google Scholar] [CrossRef] [PubMed]
- Hammoud, H.; Schmitt, M.; Bihel, F.; Antheaume, C.; Bourguignon, J.J. Direct guanidinylation of aryl and heteroarylhalides via copper-catalyzed cross-coupling reaction. J. Org. Chem. 2012, 77, 417–423. [Google Scholar] [CrossRef] [PubMed]
- Lu, Z.; Twieg, R.J.; Huang, S.D. Copper-catalyzed amination of aromatic halides with 2-N,N-dimethylaminoethanol as solvent. Tetrahedron Lett. 2003, 44, 6289–6292. [Google Scholar] [CrossRef]
- Lu, Z.; Twieg, R.J. A mild and practical copper catalyzed amination of halothiophenes. Tetrahedron 2005, 61, 903–918. [Google Scholar] [CrossRef]
- Kwong, F.Y.; Klapars, A.; Buchwald, S.L. Copper-catalyzed coupling of alkylamines and aryl iodides: An efficient system even in an air atmosphere. Org. Lett. 2002, 4, 581–584. [Google Scholar] [CrossRef] [PubMed]
- Wang, N.; Chen, B.; Ma, S.M. Studies on electrophilic cyclization of N-(buta-2,3-dienyl)amides with N-bromosuccinimide and its applications. Adv. Synth. Catal. 2014, 356, 485–492. [Google Scholar] [CrossRef]
- Tye, J.W.; Weng, Z.; Johns, A.M.; Incarvito, C.D.; Hartwig, J.F. Copper complexes of anionic nitrogen ligands in the amidation and imidation of aryl halides. J. Am. Chem. Soc. 2008, 130, 9971–9983. [Google Scholar] [CrossRef] [PubMed]
- Goodbrand, H.B.; Hu, N.X. Ligand-accelerated catalysis of the Ullmann condensation: Application to hole conducting triarylamines. J. Org. Chem. 1999, 64, 670–674. [Google Scholar] [CrossRef]
- Gujadhur, R.K.; Bates, C.G.; Venkataraman, D. Formation of aryl-nitrogen, aryl-oxygen, and aryl-carbon bonds using well-defined copper(I)-based catalysts. Org. Lett. 2001, 3, 4315–4317. [Google Scholar] [CrossRef] [PubMed]
- Yang, K.; Qiu, Y.T.; Li, Z.; Wang, Z.Y.; Jiang, S. Ligands for copper-catalyzed C–N bond forming reactions with 1 mol % CuBr as Catalyst. J. Org. Chem. 2011, 76, 3151–3159. [Google Scholar] [CrossRef] [PubMed]
- Shafir, A.; Buchwald, S.L. Highly selective room-temperature copper-catalyzed C–N coupling reactions. J. Am. Chem. Soc. 2006, 128, 8742–8743. [Google Scholar] [CrossRef] [PubMed]
- Shafir, A.; Lichtor, P.A.; Buchwald, S.L. N-versus O-arylation of aminoalcohols: Orthogonal selectivity in copper-based catalysts. J. Am. Chem. Soc. 2007, 129, 3490–3491. [Google Scholar] [CrossRef] [PubMed]
- Xia, N.; Taillefer, M. A very simple copper-catalyzed synthesis of anilines by employing aqueous ammonia. Angew. Chem. Int. Ed. 2009, 48, 337–339. [Google Scholar] [CrossRef] [PubMed]
- Chen, H.; Lei, M.; Hu, L.H. Synthesis of 1-aryl indoles via coupling reaction of indoles and aryl halides catalyzed by CuI/metformin. Tetrahedron 2014, 70, 5626–5631. [Google Scholar] [CrossRef]
- Ghorbani-Vaghei, R.; Hemmati, S.; Veisi, H. An in situ generated CuI/metformin complex as a novel and efficient catalyst for C–N and C–O cross-coupling reactions. Tetrahedron Lett. 2013, 54, 7095–7099. [Google Scholar] [CrossRef]
- Yang, S.L.; Wu, C.Q.; Ruan, M.B.; Yang, Y.Q.; Zhao, Y.X.; Niu, J.J.; Yang, W.; Xu, J.W. Metal- and ligand-free Ullmann-type C–O and C–N coupling reactions promoted by potassium tert-butoxide. Tetrahedron Lett. 2012, 53, 4288–4292. [Google Scholar] [CrossRef]
- Yuan, Y.; Thome, I.; Kim, S.H.; Chen, D.; Beyer, A.; Bonnamour, J.; Zuidema, E.; Chang, S.; Bolm, C. Dimethyl sulfoxide/potassium hydroxide: A superbase for the transition metal-free preparation of cross-coupling products. Adv. Synth. Catal. 2010, 352, 2892–2898. [Google Scholar] [CrossRef]
- Cano, R.; Ramon, D.J.; Yus, M. Transition-metal-free O-, S-, and N-arylation of alcohols, thiols, amides, amines, and related heterocycles. J. Org. Chem. 2011, 76, 654–660. [Google Scholar] [CrossRef] [PubMed]
- Creutz, S.E.; Lotito, K.J.; Fu, G.C.; Peters, J.C. Photoinduced Ullmann C–N coupling: Demonstrating the viability of a radical pathway. Science 2012, 338, 647–651. [Google Scholar] [CrossRef] [PubMed]
- Ziegler, D.T.; Choi, J.; Munoz-Molina, J.M.; Bissember, A.C.; Peters, J.C.; Fu, G.C. A versatile approach to Ullmann C–N couplings at room temperature: New families of nucleophiles and electrophiles for photoinduced, copper-catalyzed processes. J. Am. Chem. Soc. 2013, 135, 13107–13112. [Google Scholar] [CrossRef] [PubMed]
- Majek, M.; von Jacobi Wangelin, A. Ambient-light-mediated copper-catalyzed C–C and C–N bond formation. Angew. Chem. Int. Ed. 2013, 52, 5919–5921. [Google Scholar] [CrossRef] [PubMed]
- Strieter, E.R.; Bhayana, B.; Buchwald, S.L. Mechanistic studies on the copper-catalyzed N-arylation of amides. J. Am. Chem. Soc. 2009, 131, 78–88. [Google Scholar] [CrossRef] [PubMed]
- Yu, H.Z.; Jiang, Y.Y.; Fu, Y.; Liu, L. Alternative mechanistic explanation for ligand-dependent selectivities in copper-catalyzed N- and O-arylation reactions. J. Am. Chem. Soc. 2010, 132, 18078–18091. [Google Scholar] [CrossRef] [PubMed]
- Jones, G.O.; Liu, P.; Houk, K.N.; Buchwald, S.L. Computational explorations of mechanisms and ligand-directed selectivities of copper-catalyzed Ullmann-type reactions. J. Am. Chem. Soc. 2010, 132, 6205–6213. [Google Scholar] [CrossRef] [PubMed]
- Satish, G.; Reddy, K.H.V.; Ramesh, K.; Kumar, B.S.P.A.; Nageswar, Y.V.D. An elegant protocol for the synthesis of N-substituted pyrroles through C–N cross coupling/aromatization process using CuFe2O4 nanoparticles as catalyst under ligand-free conditions. Tetrahedron Lett. 2014, 55, 2596–2599. [Google Scholar] [CrossRef]
- Costa, M.V.; Viana, G.M.; de Souza, T.M.; Malta, L.F.B.; Aguiar, L.C.S. Copper-catalyzed C–N cross-coupling reactions for the preparation of aryl diamines applying mild conditions. Tetrahedron Lett. 2013, 54, 2332–2335. [Google Scholar] [CrossRef]
- Reddy, K.H.V.; Satish, G.; Ramesh, K.; Karnakar, K.; Nageswar, Y.V.D. An efficient synthesis of N–substituted indoles from indoline/indoline carboxylic acid via aromatization followed by C–N cross-coupling reaction by using nano copper oxide as a recyclable catalyst. Tetrahedron Lett. 2012, 53, 3061–3065. [Google Scholar] [CrossRef]
- Zhang, P.F.; Yuan, J.Y.; Li, H.R.; Liu, X.F.; Xu, X.; Antonietti, M.; Wang, Y. Mesoporous nitrogen-doped carbon for copper mediated Ullmann-type C–O/–N/–S cross-coupling reactions. RSC Adv. 2013, 3, 1890–1895. [Google Scholar] [CrossRef]
- Xiao, R.; Zhao, H.; Cai, M. MCM-41-immobilized bidentate nitrogen copper(I) complex: A highly efficient and recyclable catalyst for Buchwald N-arylation of indoles. Tetrahedron 2013, 69, 5444–5450. [Google Scholar] [CrossRef]
- Likhar, P.R.; Roy, S.; Roy, M.; Kantam, M.L.; De, R.L. Silica immobilized copper complexes: Efficient and reusable catalysts for N-arylation of N(H)-heterocycles and benzyl amines with aryl halides and arylboronic acids. J. Mol. Catal. A: Chem. 2007, 271, 57–62. [Google Scholar] [CrossRef]
- Choudary, B.M.; Sridhar, C.; Kantam, M.L.; Venkanna, G.T.; Sreedhar, B. Design and evolution of copper apatite catalysts for N-arylation of heterocycles with chloro- and fluoroarenes. J. Am. Chem. Soc. 2005, 127, 9948–9949. [Google Scholar] [CrossRef] [PubMed]
- Chouhan, G.; Wang, D.; Alper, H. Magnetic nanoparticle-supported proline as a recyclable and recoverable ligand for the CuI catalyzed arylation of nitrogen nucleophiles. Chem. Commun. 2007, 45, 4809–4811. [Google Scholar] [CrossRef] [PubMed]
- Mostafalu, R.; Kaboudin, B.; Kazemi, F.; Yokomatsu, T. N-arylation of amines: C–N coupling of amines with arylboronic acids using Fe3O4 magnetic nanoparticles-supported EDTA-Cu(II) complex in water. RSC Adv. 2014, 4, 49273–49279. [Google Scholar] [CrossRef]
- Huang, L.; Yu, R.; Zhu, X.; Wan, Y. A recyclable Cu-catalyzed C–N coupling reaction in water and its application to synthesis of imidazo[1,2-a]quinoxaline. Tetrahedron 2013, 69, 8974–8977. [Google Scholar] [CrossRef]
- Huang, Y.B.; Yang, C.T.; Yi, J.; Deng, X.J.; Fu, Y.; Liu, L. Cu-catalyzed carbon-heteroatom coupling reactions under mild conditions promoted by resin-bound organic ionic bases. J. Org. Chem. 2011, 76, 800–810. [Google Scholar] [CrossRef] [PubMed]
- Islam, S.M.; Mondal, S.; Mondal, P.; Roy, A.S.; Tuhina, K.; Salam, N.; Mobarak, M. A reusable polymer supported copper catalyst for the C–N and C–O bond cross-coupling reaction of aryl halides as well as arylboronic acids. J. Org. Chem. 2012, 696, 4264–4274. [Google Scholar] [CrossRef]
- Yang, B.; Mao, Z.X.; Zhu, X.H.; Wan, Y.Q. Functionalised chitosan as a green, recyclable, supported catalyst for the copper-catalysed Ullmann C–N coupling reaction in water. Catal. Commun. 2015, 60, 92–95. [Google Scholar] [CrossRef]
- Salam, N.; Kundu, S.K.; Roy, A.S.; Mondal, P.; Roy, S.; Bhaumik, A.; Islam, S.M. Cu-grafted mesoporous organic polymer: A new recyclable nanocatalyst for multi-component, N-arylation and S-arylation reactions. Catal. Sci. Technol. 2013, 3, 3303–3316. [Google Scholar]
- Corma, A.; Garcia, H.; Llabres, I.; Xamena, F.X. Engineering metal organic frameworks for heterogeneous catalysis. Chem. Rev. 2010, 110, 4606–4655. [Google Scholar] [CrossRef] [PubMed]
- Chughtai, A.H.; Ahmad, N.; Younus, H.A.; Laypkov, A.; Verpoort, F. Metal-organic frameworks: Versatile heterogeneous catalysts for efficient catalytic organic transformations. Chem. Soc. Rev. 2015, 44, 6804–6849. [Google Scholar] [CrossRef] [PubMed]
- Dhakshinamoorthy, A.; Opanasenko, M.; Cejka, J.; Garcia, H. Metal organic frameworks as heterogeneous catalysts for the production of fine chemicals. Catal. Sci. Technol. 2013, 3, 2509–2540. [Google Scholar] [CrossRef]
- Gomez-Lor, B.; Gutierrez-Puebla, E.; Iglesias, M.; Monge, M.A.; Ruiz-Valero, C.; Snejko, N. In2(OH)3(BDC)1.5 (BDC = 1,4-Benzendicarboxylate): An In(III) supramolecular 3D framework with catalytic activity. Inorg. Chem. 2002, 41, 2429–2432. [Google Scholar] [CrossRef] [PubMed]
- Llabresi Xamena, F.X.; Abad, A.; Corma, A.; Garcia, H. MOFs as catalysts: Activity, reusability and shape-selectivity of a Pd-containing MOF. J. Catal. 2007, 250, 294–298. [Google Scholar] [CrossRef]
- Dhakshinamoorthy, A.; Alvaro, M.; Chevreau, H.; Horcajada, P.; Devic, T.; Serre, C.; Garcia, H. Iron(III) metal-organic frameworks as solid Lewis acids for the isomerization of α-pinene oxide. Catal. Sci. Technol. 2012, 2, 324–330. [Google Scholar] [CrossRef]
- Gu, J.M.; Kim, W.S.; Huh, S. Size-dependent catalysis by DABCO-functionalized Zn-MOF with one-dimensional channels. Dalton Trans. 2011, 40, 10826–10829. [Google Scholar] [CrossRef] [PubMed]
- Dang, D.; Wu, P.; He, C.; Xie, Z.; Duan, C. Homochiral metal-organic frameworks for heterogeneous asymmetric catalysis. J. Am. Chem. Soc. 2010, 132, 14321–14323. [Google Scholar] [CrossRef] [PubMed]
- Llabres i Xamena, F.X.; Casanova, O.; Tailleur, R.G.; Garcia, H.; Corma, A. Metal organic frameworks (MOFs) as catalysts: A combination of Cu2+ and Co2+ MOFs as an efficient catalyst for tetralin oxidation. J. Catal. 2008, 255, 220–227. [Google Scholar] [CrossRef]
- Dhakshinamoorthy, A.; Alvaro, M.; Garcia, H. Aerobic oxidation of benzylic alcohols catalyzed by metal-organic frameworks assisted by TEMPO. ACS Catal. 2011, 1, 48–53. [Google Scholar] [CrossRef]
- Marx, S.; Kleist, W.; Baiker, A. Synthesis, structural properties, and catalytic behavior of Cu-BTC and mixed-linker Cu-BTC-PyDC in the oxidation of benzene derivatives. J. Catal. 2011, 281, 76–87. [Google Scholar] [CrossRef]
- Fei, H.H.; Shin, J.; Meng, Y.S.; Adelhardt, M.; Sutter, J.; Meyer, K.; Cohen, S.M. Reusable oxidation catalysis using metal-monocatecholato species in a robust metal-organic framework. J. Am. Chem. Soc. 2014, 136, 4965–4973. [Google Scholar] [CrossRef] [PubMed]
- Maksimchuk, N.V.; Kovalenko, K.A.; Fedin, V.P.; Kholdeeva, O.A. Cyclohexane selective oxidation over metal-organic frameworks of MIL-101 family: Superior catalytic activity and selectivity. Chem. Commun. 2012, 48, 6812–6814. [Google Scholar] [CrossRef] [PubMed]
- Laurier, K.G.M.; Vermoortele, F.; Ameloot, R.; de Vos, D.E.; Hofkens, J.; Roeffaers, M.B.J. Iron(III)-based metal-organic frameworks as visible light photocatalysts. J. Am. Chem. Soc. 2013, 135, 14488–14491. [Google Scholar] [CrossRef] [PubMed]
- Wang, D.K.; Huang, R.K.; Liu, W.J.; Sun, D.R.; Li, Z.H. Fe-based MOFs for photocatalytic CO2 reduction: Role of coordination unsaturated sites and dual excitation pathways. ACS Catal. 2014, 4, 4254–4260. [Google Scholar] [CrossRef]
- Deng, M.; Ling, Y.; Xia, B.; Chen, Z.; Zhou, Y.; Liu, X.; Yue, B.; He, H. Synthesis of isoreticular zinc(II)-phosphonocarboxylate frameworks and their application in the Friedel-Crafts benzylation reaction. Chem. Eur. J. 2011, 17, 10323–10328. [Google Scholar] [CrossRef] [PubMed]
- Phan, N.T.S.; Le, K.K.A.; Phan, T.D. MOF-5 as an efficient heterogeneous catalyst for Friedel-Crafts alkylation reactions. Appl. Catal. A Gen. 2010, 382, 246–253. [Google Scholar] [CrossRef]
- Ravon, U.; Savonnet, M.; Aguado, S.; Domine, M.E.; Janneau, E.; Farrusseng, D. Engineering of coordination polymers for shape selective alkylation of large aromatics and the role of defects. Microporous Mesoporous Mater. 2010, 129, 319–329. [Google Scholar] [CrossRef]
- Dhakshinamoorthy, A.; Opanasenko, M.; Cejka, J.; Garcia, H. Metal organic frameworks as solid catalysts in condensation reactions of carbonyl groups. Adv. Synth. Catal. 2013, 355, 247–268. [Google Scholar] [CrossRef]
- Phan, N.T.S.; Nguyen, T.T.; Nguyen, C.V.; Nguyen, T.T. Ullmann-type coupling reaction using metal-organic framework MOF-199 as an efficient recyclable solid catalyst. Appl. Catal. A: Gen. 2013, 457, 69–77. [Google Scholar] [CrossRef]
- Wang, M.; Yuan, B.Z.; Ma, T.M.; Jiang, H.F.; Li, Y.W. Ligand-free coupling of phenols and alcohols with aryl halides by arecyclable heterogeneous copper catalyst. RSC Adv. 2012, 2, 5528–5530. [Google Scholar] [CrossRef]
- Dang, T.T.; Zhu, Y.H.; Ghosh, S.C.; Chen, A.Q.; Chai, C.L.L.; Seayad, A.M. Atmospheric pressure aminocarbonylation of aryl iodides using palladium nanoparticles supported on MOF-5. Chem. Commun. 2012, 48, 1805–1807. [Google Scholar] [CrossRef]
- Dhakshinamoorthy, A.; Alvaro, M.; Concepcion, P.; Garcia, H. Chemical instability of Cu3(BTC)2 by reaction with thiols. Catal. Commun. 2011, 12, 1018–1021. [Google Scholar] [CrossRef]
- Li, B.Y.; Zhang, Z.J.; Li, Y.; Yao, K.X.; Zhu, Y.H.; Deng, Z.Y.; Yang, F.; Zhou, X.J.; Li, G.H.; Wu, H.H.; et al. Enhanced binding affinity, remarkable selectivity, and high capacity of CO2 by dual functionalization of a rht-type metal-organic framework. Angew. Chem. Int. Ed. 2012, 51, 1412–1415. [Google Scholar] [CrossRef] [PubMed]
- Gant, T.G.; Sarshar, S. New Substituted N-Arylpyridone Derivatives Useful for Treating e.g., Idiopathic Pulmonary Fibrosis, Cirrhosis, Diffuse Parenchymal Lung Disease, Mediastinal Fibrosis, Tuberculosis, and Rheumatoid Arthritis. U.S. Patent 0,319,026 A1, 20 June 2008. [Google Scholar]
- Okazaki, A.; Ohkura, N.; Fujimura, M.; Katayama, N.; Kasahara, K. Effects of pirfenidone on increased cough reflex sensitivity in guinea pigs. Pulm. Pharmacol. Ther. 2013, 26, 603–608. [Google Scholar] [CrossRef] [PubMed]
- Yadav, D.K.T.; Rajak, S.S.; Bhanage, B.M. N-arylation of indoles with aryl halides using copper/glycerolas a mild and highly efficient recyclable catalytic system. Tetrahedron Lett. 2014, 55, 931–935. [Google Scholar] [CrossRef]
- Ricordi, V.G.; Freitas, C.S.; Perin, G.; Lenardao, E.J.; Jacob, R.G.; Savegnago, L.; Alves, D. Glycerol as a recyclable solvent for copper-catalyzed cross-coupling reactionsof diaryldiselenides with aryl boronic acids. Green Chem. 2012, 14, 1030–1034. [Google Scholar] [CrossRef]
- Frank, J.; Katritzky, A.R. Tautornericpyridines. Part XV. Pyridone-hydroxypyridine equilibriain solvents of differing polarity. J. Chem. Soc. Perkin Trans. 1976, 2, 1428–1431. [Google Scholar] [CrossRef]
- Alaerts, L.; Seguin, E.; Poelman, H.; Thibault-Starzyk, F.; Jacobs, P.A.; de Vos, D.E. Probing the Lewis acidity and catalytic activity of the metal-organic framework [Cu3(btc)2] (BTC = Benzene-1,3,5-tricarboxylate). Chem. Eur. J. 2006, 12, 7353–7363. [Google Scholar] [CrossRef] [PubMed]
- Schlichte, K.; Kratzke, T.; Kaskel, S. Improved synthesis, thermal stability and catalytic properties of the metal-organic framework compound Cu3(BTC)2. Microporous Mesoporous Mater. 2004, 73, 81–88. [Google Scholar] [CrossRef]
- Priyadarshini, S.; Amal Joseph, P.J.; Lakshmi Kantam, M.; Sreedhar, B. Copper MOF: Scope and limitation in catalytic hydroxylation and nitration of aryl halides. Tetrahedron 2013, 69, 6409–6414. [Google Scholar] [CrossRef]
- Wee, L.H.; Bajpe, S.R.; Janssens, N.; Hermans, I.; Houthoofd, K.; Kirschhock, C.E. A.; Martens, J.A. Convenient synthesis of Cu3(BTC)2 encapsulated Keggin heteropolyacid nanomaterial for application in catalysis. Chem. Commun. 2010, 46, 8186–8188. [Google Scholar] [CrossRef] [PubMed]
- Li, C.Q.; Qiu, W.G.; Long, W.; Deng, F.; Bai, G.M.; Zhang, G.Z.; Zi, X.H.; He, H. Synthesis of porphyrin@MOFs type catalysts through “one-pot” self-assembly. J. Mol. Catal. A Chem. 2014, 393, 166–170. [Google Scholar] [CrossRef]
- Yin, J.; Buchwald, S.L. Palladium-catalyzed intermolecular coupling of aryl halides and amides. Org. Lett. 2000, 2, 1101–1104. [Google Scholar] [CrossRef] [PubMed]
- Quan, Z.J.; Xia, H.D.; Zhang, Z.; Da, Y.X.; Wang, X.C. An efficient copper-catalyzed N-arylation of amides: Synthesis of N-arylacrylamides and 4-amido-N-phenylbenzamides. Tetrahedron 2013, 69, 8368–8374. [Google Scholar] [CrossRef]
- Sperotto, E.; Klink, G.P. M.V.; Koten, G.V.; Vries, J.G. D. The mechanism of the modified Ullmann reaction. Dalton Trans. 2010, 39, 10338–10351. [Google Scholar] [CrossRef] [PubMed]
- Sambiagio, C.; Marsden, S.P.; John Blacker, A.; McGowan, P.C. Copper catalyzed Ullmann type chemistry: From mechanistic aspects to modern development. Chem. Soc. Rev. 2014, 43, 3525–3550. [Google Scholar] [CrossRef] [PubMed]
- Mkami, H.E.L.; Mohideen, M.I.H.; Pal, C.; McKinlay, A.; Scheimann, O.; Morris, R.E. EPR and magnetic studies of a novel copper metal organic framework (STAM-I). Chem. Phys. Lett. 2012, 544, 17–21. [Google Scholar] [CrossRef]
- Simenas, M.; Kobalz, M.; Mendt, M.; Eckold, P.; Krautscheid, H.; Banys, J.; Poppl, A. Synthesis, structure, and electron paramagnetic resonance study of a mixed valent metal-organic framework containing Cu2 paddle-wheel units. J. Phys. Chem. C 2015, 119, 4898–4907. [Google Scholar] [CrossRef]
- Sample Availability: Sample of the compound Cu-TDPAT is available from the authors.
© 2015 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons by Attribution (CC-BY) license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Long, W.; Qiu, W.; Guo, C.; Li, C.; Song, L.; Bai, G.; Zhang, G.; He, H. A Copper-Based Metal-Organic Framework as an Efficient and Reusable Heterogeneous Catalyst for Ullmann and Goldberg Type C–N Coupling Reactions. Molecules 2015, 20, 21178-21192. https://doi.org/10.3390/molecules201219756
Long W, Qiu W, Guo C, Li C, Song L, Bai G, Zhang G, He H. A Copper-Based Metal-Organic Framework as an Efficient and Reusable Heterogeneous Catalyst for Ullmann and Goldberg Type C–N Coupling Reactions. Molecules. 2015; 20(12):21178-21192. https://doi.org/10.3390/molecules201219756
Chicago/Turabian StyleLong, Wei, Wenge Qiu, Chongwei Guo, Chuanqiang Li, Liyun Song, Guangmei Bai, Guizhen Zhang, and Hong He. 2015. "A Copper-Based Metal-Organic Framework as an Efficient and Reusable Heterogeneous Catalyst for Ullmann and Goldberg Type C–N Coupling Reactions" Molecules 20, no. 12: 21178-21192. https://doi.org/10.3390/molecules201219756
APA StyleLong, W., Qiu, W., Guo, C., Li, C., Song, L., Bai, G., Zhang, G., & He, H. (2015). A Copper-Based Metal-Organic Framework as an Efficient and Reusable Heterogeneous Catalyst for Ullmann and Goldberg Type C–N Coupling Reactions. Molecules, 20(12), 21178-21192. https://doi.org/10.3390/molecules201219756