Solvatochromic and Single Crystal Studies of Two Neutral Triarylmethane Dyes with a Quinone Methide Structure
Abstract
:1. Introduction
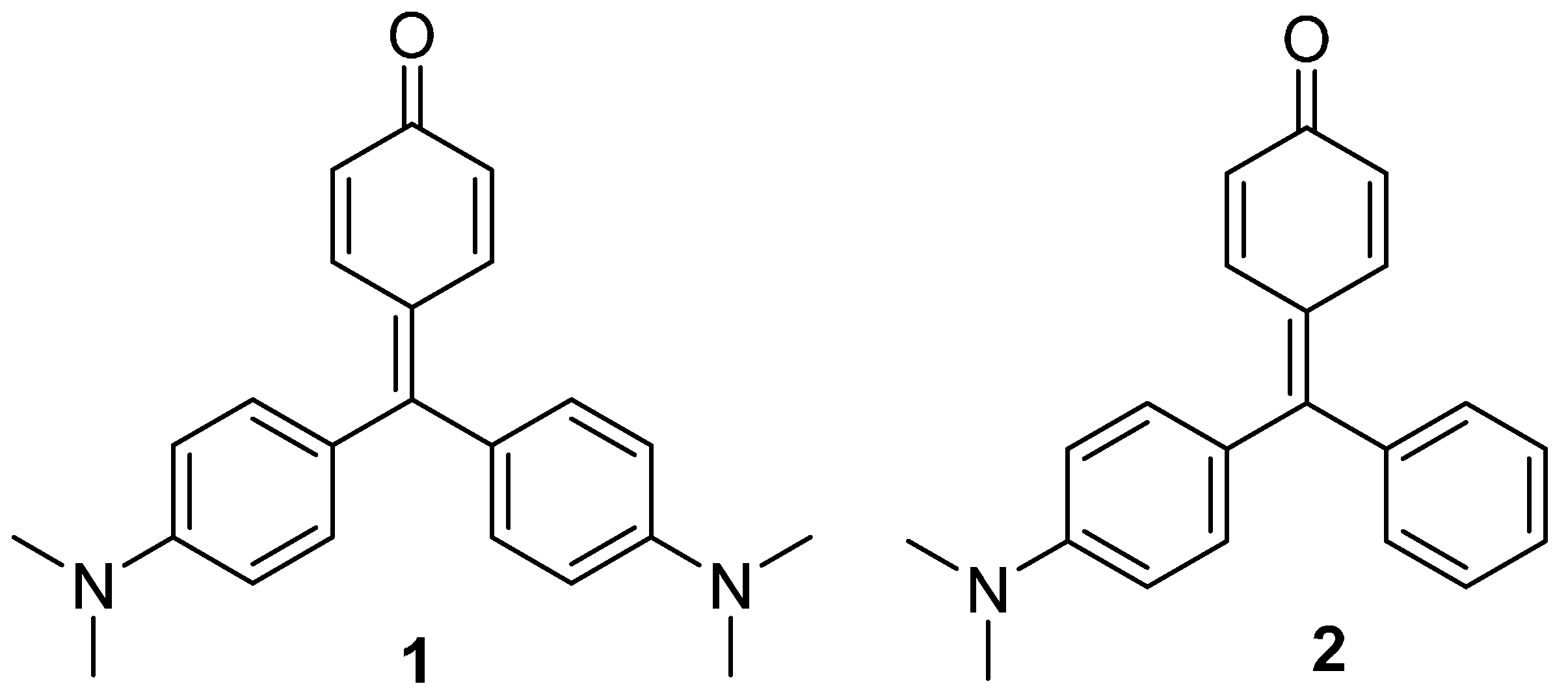
2. Results and Discussion
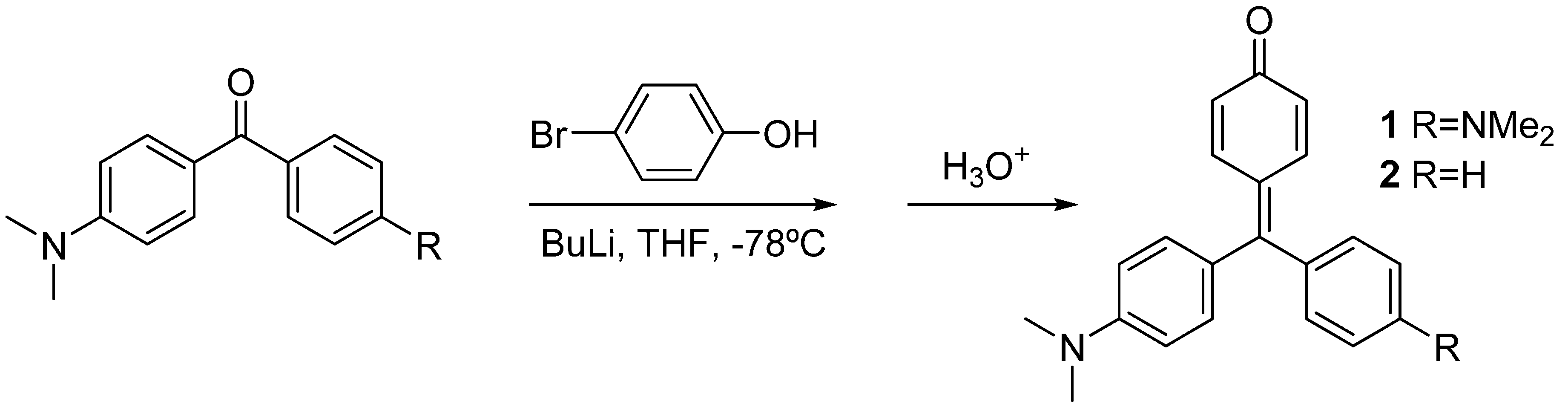
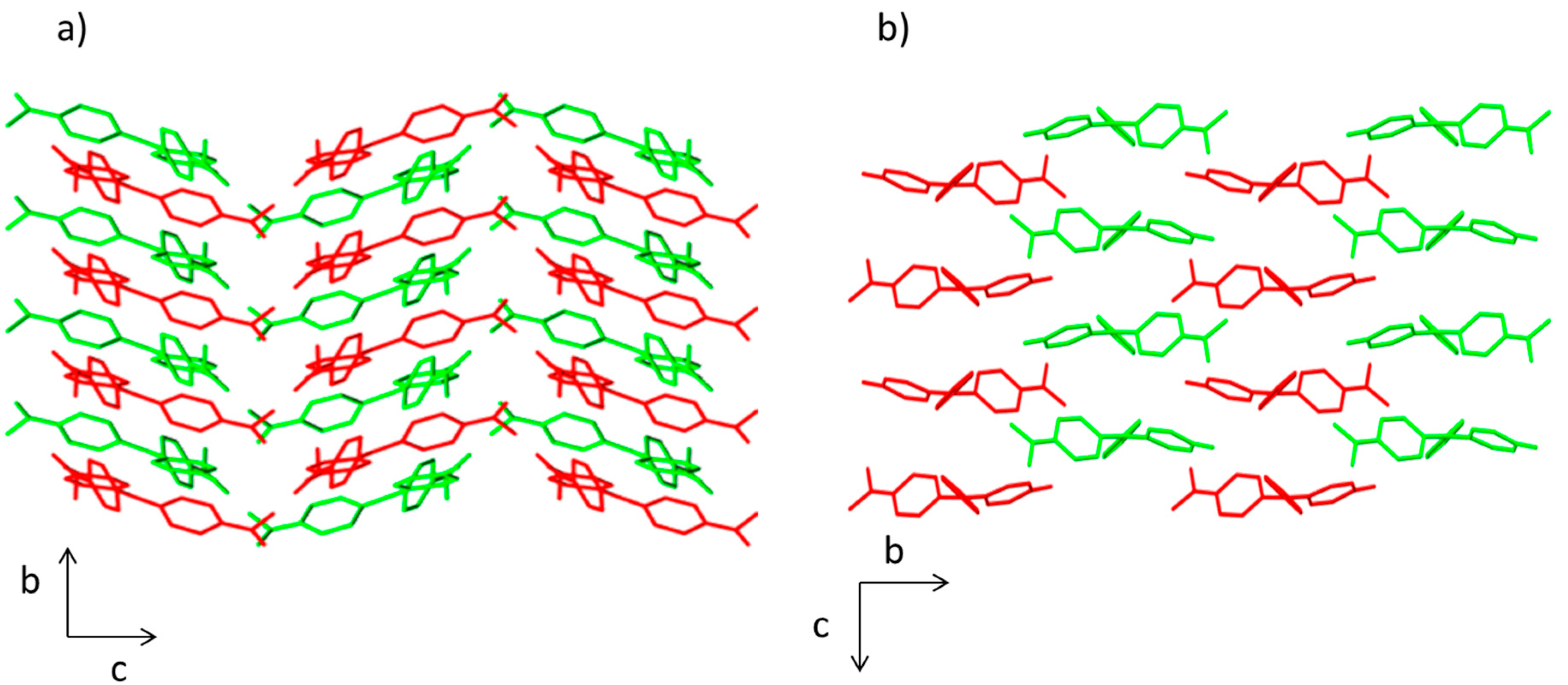
2.1. Synthesis and X-ray Diffraction Studies
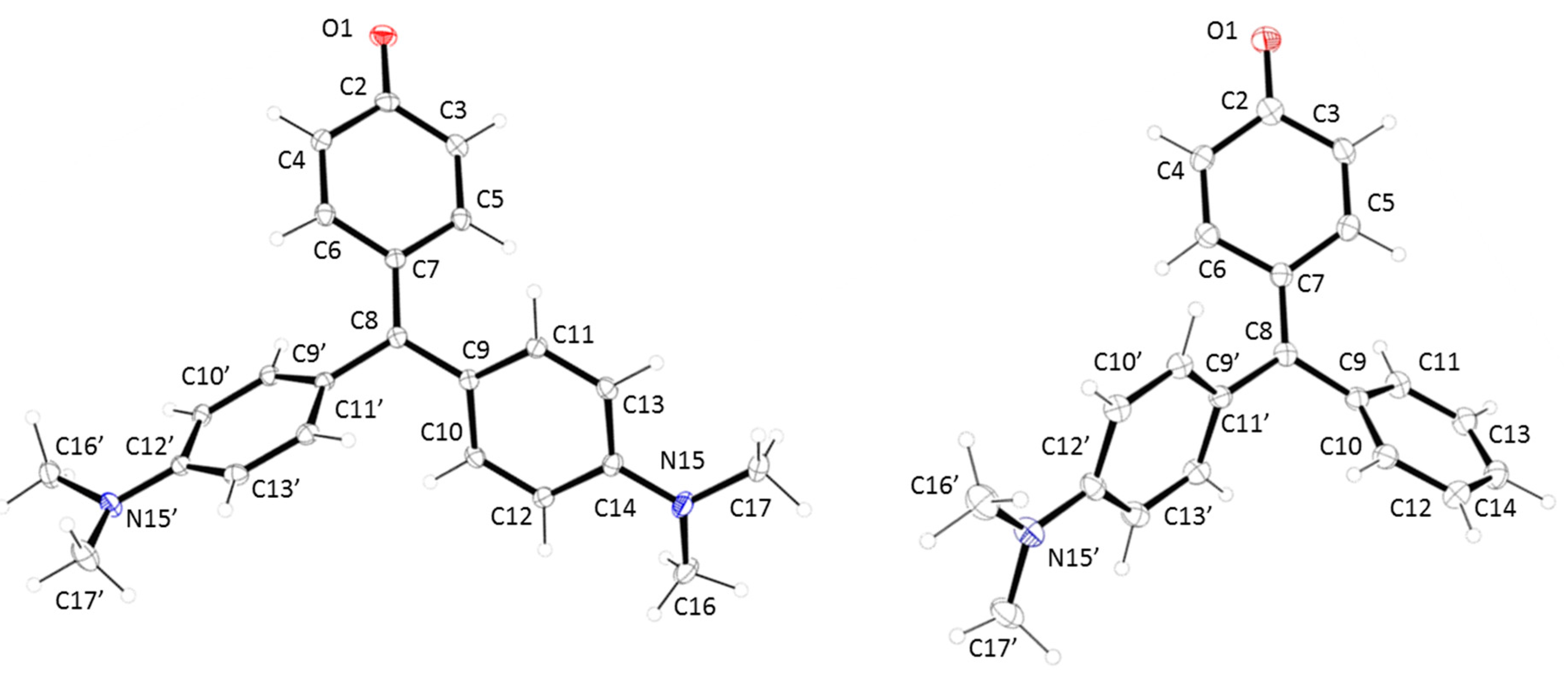
| Atomic Bond Distances (Å) | 1 | 2 | Angles (°) | 1 | 2 |
|---|---|---|---|---|---|
| O1-C2 | 1.248(3) | 1.249(3) | C8-C7-C5 | 123.1(2) | 123.4(3) |
| C2-C4 | 1.445(3) | 1.446(4) | C8-C7-C6 | 121.4(2) | 121.3(3) |
| C2-C3 | 1.451(3) | 1.451(4) | C7-C8-C9 | 123.25(19) | 121.4(3) |
| C3-C5 | 1.384(3) | 1.342(4) | C7-C8-C9′ | 120.0(2) | 121.7(3) |
| C4-C6 | 1.340(3) | 1.346(4) | C9-C8-C9′ | 116.71(19) | 116.8(3) |
| C5-C7 | 1.440(3) | 1.440(4) | |||
| C6-C7 | 1.441(3) | 1.452(4) | |||
| C7-C8 | 1.396(3) | 1.381(4) | |||
| C8-C9 | 1.454(3) | 1.482(4) | |||
| C8-C9′ | 1.469(3) | 1.466(4) |
| Angles (°) | Atomic Bond Distances (Å) | Torsion Angles (°) | ||||
|---|---|---|---|---|---|---|
| C14′-N15′-C16′ | C14′-N15′-C17′ | C16′-N15′-C17′ | C14′-N15′ | C12′-C14′-N15′-C16′ | C13′-C14′-N15′-C17′ | |
| 1 | 118.6(2) | 119.8(2) | 116.7(2) | 1.373(2) | −21.3(3) | 5.1(3) |
| 2 | 120.6(3) | 121.6(3) | 117.7(3) | 1.372(4) | −1.5(5) | 2.8(5) |
| C14-N15-C16 | C14-N15-C17 | C16-N15-C17 | C14-N15 | C12-C14-N15-C16 | C13-C14-N15-C17 | |
| 1 | 118.6(2) | 121.5(2) | 116.1(2) | 1.380(3) | 13.9(4) | −9.6(4) |
| Torsion Angles (°) | |||
|---|---|---|---|
| C6-C7-C8-C9′ | C7-C8-C9-C11 | C7-C8-C9′-C11′ | |
| 1 | 18.1(3) | 35.8(3) | −134.6(2) |
| 2 | −20.4(5) | −40.5(5) | 139.2(3) |
2.2. Solvatochromic Studies
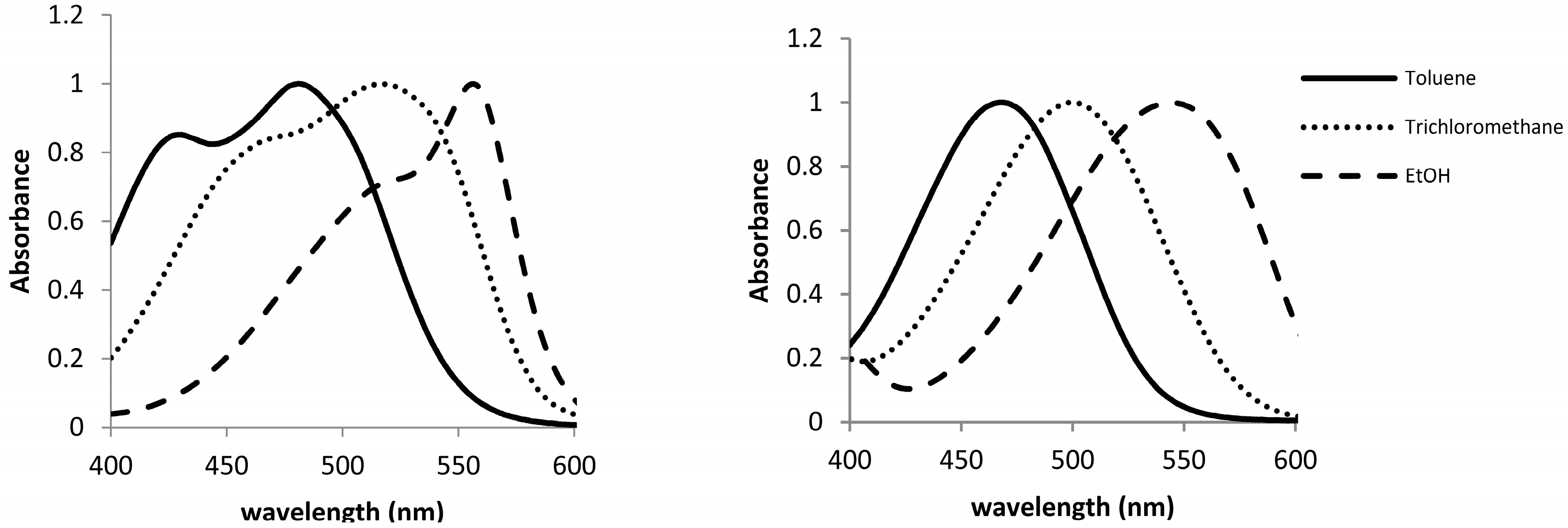
| Solvent | Type of Solvent | ET(30) | π* | β | α | λmax(1) | λmax(2) | ET(1) | ET(2) |
|---|---|---|---|---|---|---|---|---|---|
| Toluene | NHB-weak HBA | 33.9 | 0.54 | 0.11 | 0.0 | 480.5 | 467 | 59.50 | 61.22 |
| Benzene | NHB-weak HBA | 34.3 | 0.59 | 0.10 | 0.0 | 483 | 469.5 | 59.20 | 60.90 |
| Diethylether | HBA | 34.5 | 0.27 | 0.47 | 0.0 | 475 | 456.5 | 60.19 | 62.63 |
| Bromobenzene | NHB-weak HBA | 36.6 | 0.79 | 0.06 | 0.0 | 504 | 488.5 | 56.73 | 58.53 |
| Chlorobenzene | NHB-weak HBA | 36.8 | 0.71 | 0.07 | 0.0 | 503 | 484.5 | 56.84 | 59.01 |
| THF | HBA | 37.4 | 0.58 | 0.55 | 0.0 | 487.5 | 474.5 | 58.65 | 60.26 |
| Ethyl acetate | HBA | 38.1 | 0.55 | 0.45 | 0.0 | 482.5 | 471 | 59.26 | 60.70 |
| Methyl acetate | HBA | 38.9 | 0.60 | 0.42 | 0.0 | 490 | 474.5 | 58.35 | 60.26 |
| Trichloromethane | weak HBD | 39.1 | 0.58 | 0.0 | 0.44 | 515 | 498.5 | 55.52 | 57.35 |
| Pyridine | HBA | 40.5 | 0.87 | 0.64 | 0.0 | 512.5 | 499 | 55.79 | 57.30 |
| Dichloromethane | NHB | 40.7 | 0.82 | 0.0 | 0.30 | 510.5 | 494 | 56.01 | 57.88 |
| 2-Butanone | HBA | 41.3 | 0.67 | 0.48 | 0.0 | 503.5 | 483 | 56.79 | 59.20 |
| Acetone | HBA | 42.2 | 0.71 | 0.48 | 0.0 | 501 | 487 | 57.07 | 58.71 |
| N,N-Dimethylacetamide | HBA | 42.9 | 0.88 | 0.76 | 0.0 | 510 | 497.5 | 56.06 | 57.47 |
| DMF | HBA | 43.2 | 0.88 | 0.69 | 0.0 | 514 | 500.5 | 55.62 | 57.13 |
| 2-Methylpropan-2-ol | HBA-D | 43.3 | 0.41 | 1.01 | 0.68 | 541 | 507.5 | 52.85 | 56.34 |
| DMSO | HBA | 45.1 | 1.00 | 0.76 | 0.0 | 525 | 510.5 | 54.46 | 56.06 |
| Acetonitrile | weak HBA | 45.6 | 0.75 | 0.31 | 0.19 | 507.5 | 491.5 | 56.34 | 58.17 |
| 1-Butanol | HBA-D | 49.7 | 0.47 | 0.88 | 0.79 | 555.5 | 537 | 51.47 | 53.24 |
| 1-Propanol | HBA-D | 50.7 | 0.52 | 0.78 | 0.78 | 555.5 | 534.5 | 51.47 | 53.49 |
| Ethanol | HBA-D | 51.9 | 0.54 | 0.77 | 0.83 | 556 | 543 | 51.42 | 52.65 |
| Methanol | HBA-D | 55.4 | 0.60 | 0.62 | 0.93 | 555 | 552 | 51.52 | 51.80 |
| H2O | HBA-D | 63.1 | 1.09 | 0.18 | 1.17 | 572 | 581 | 49.98 | 49.21 |
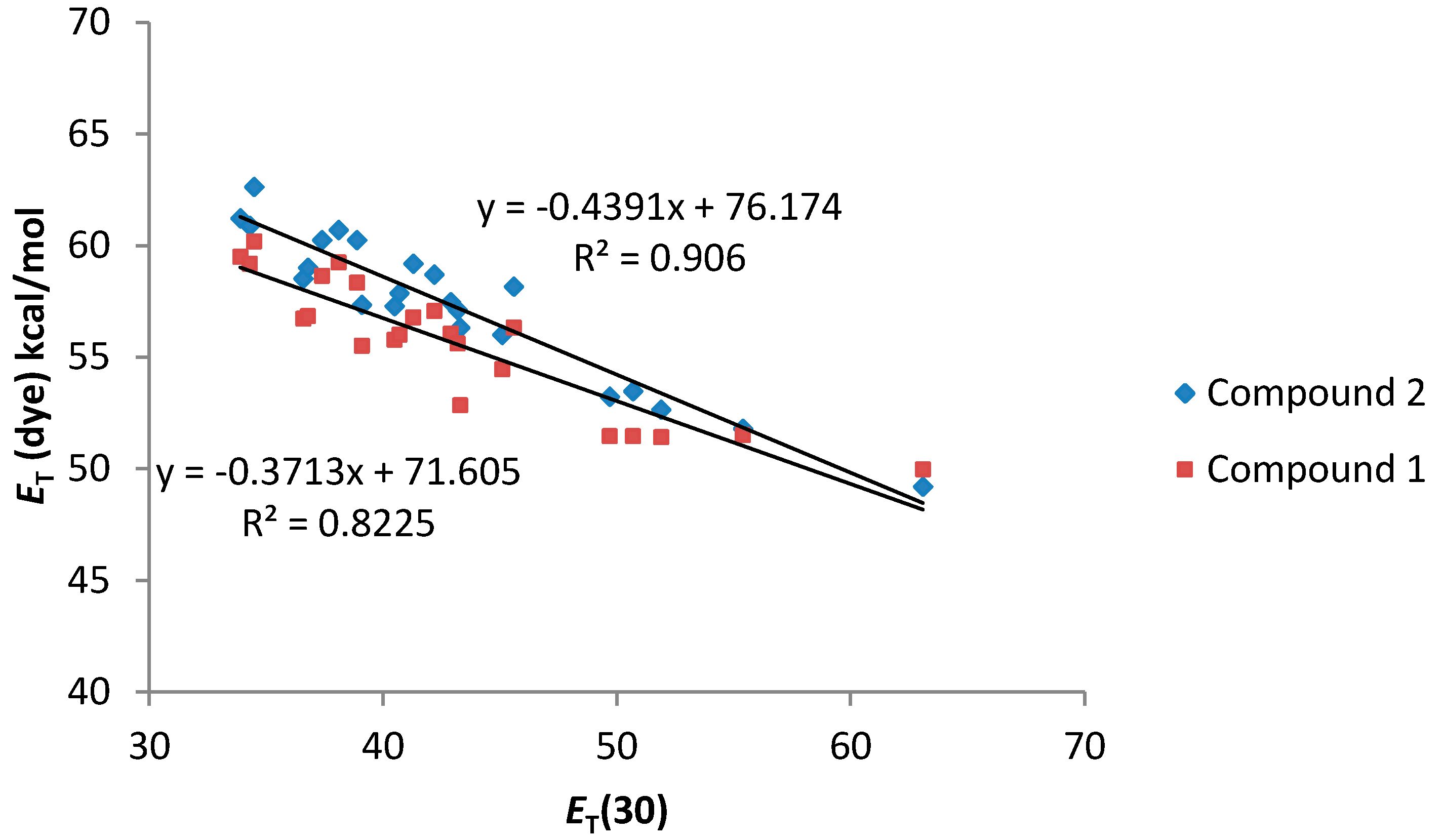
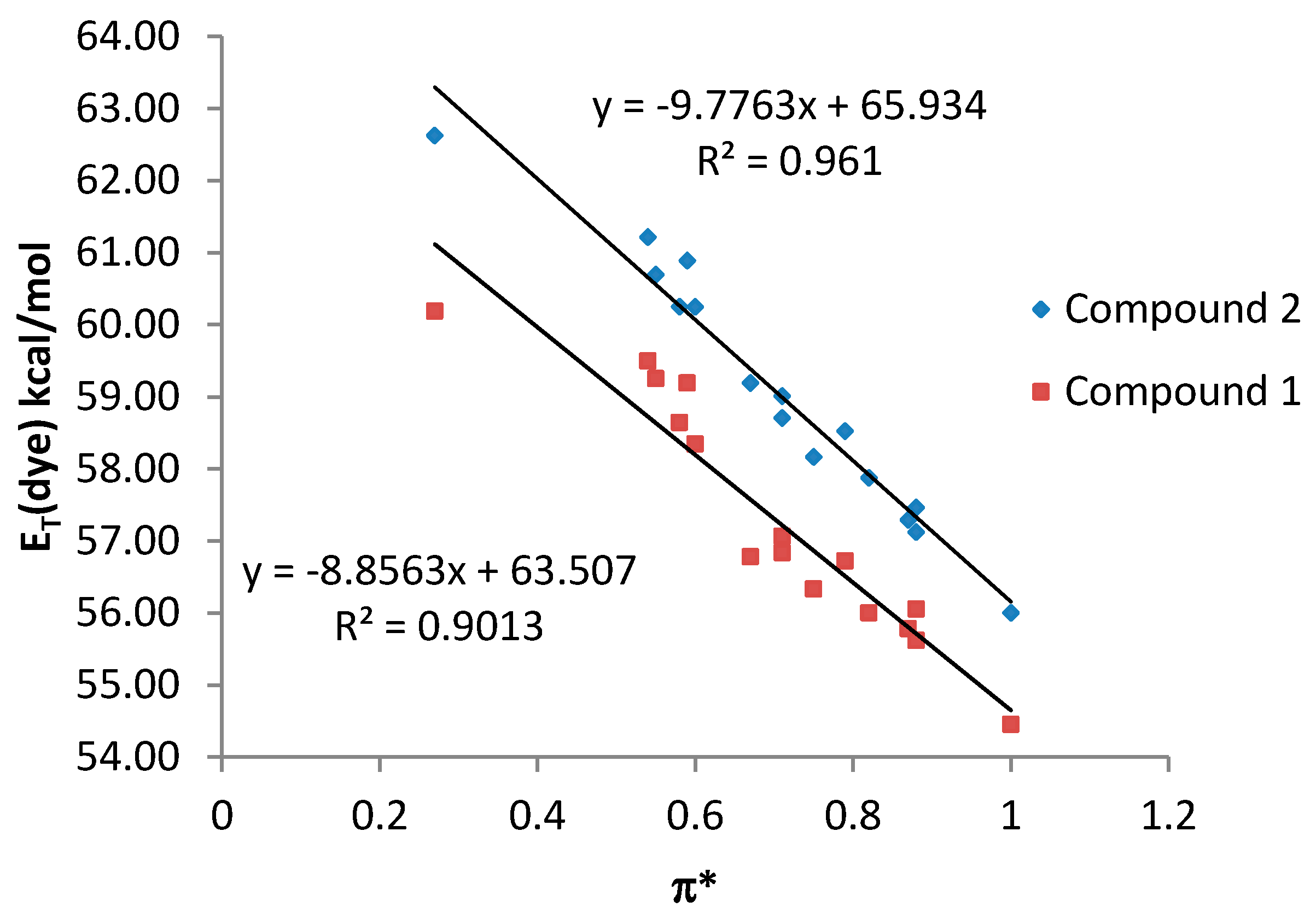
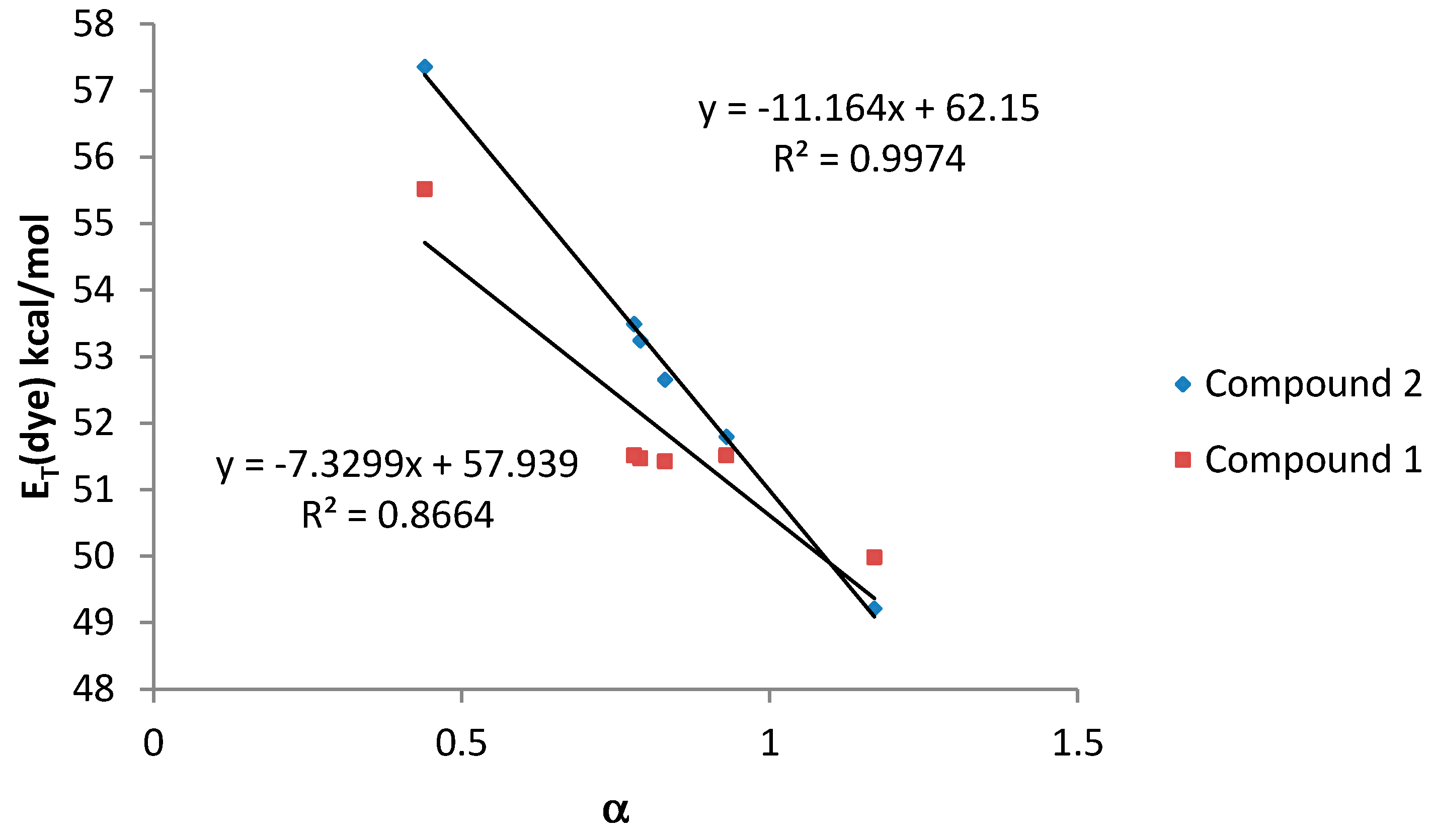
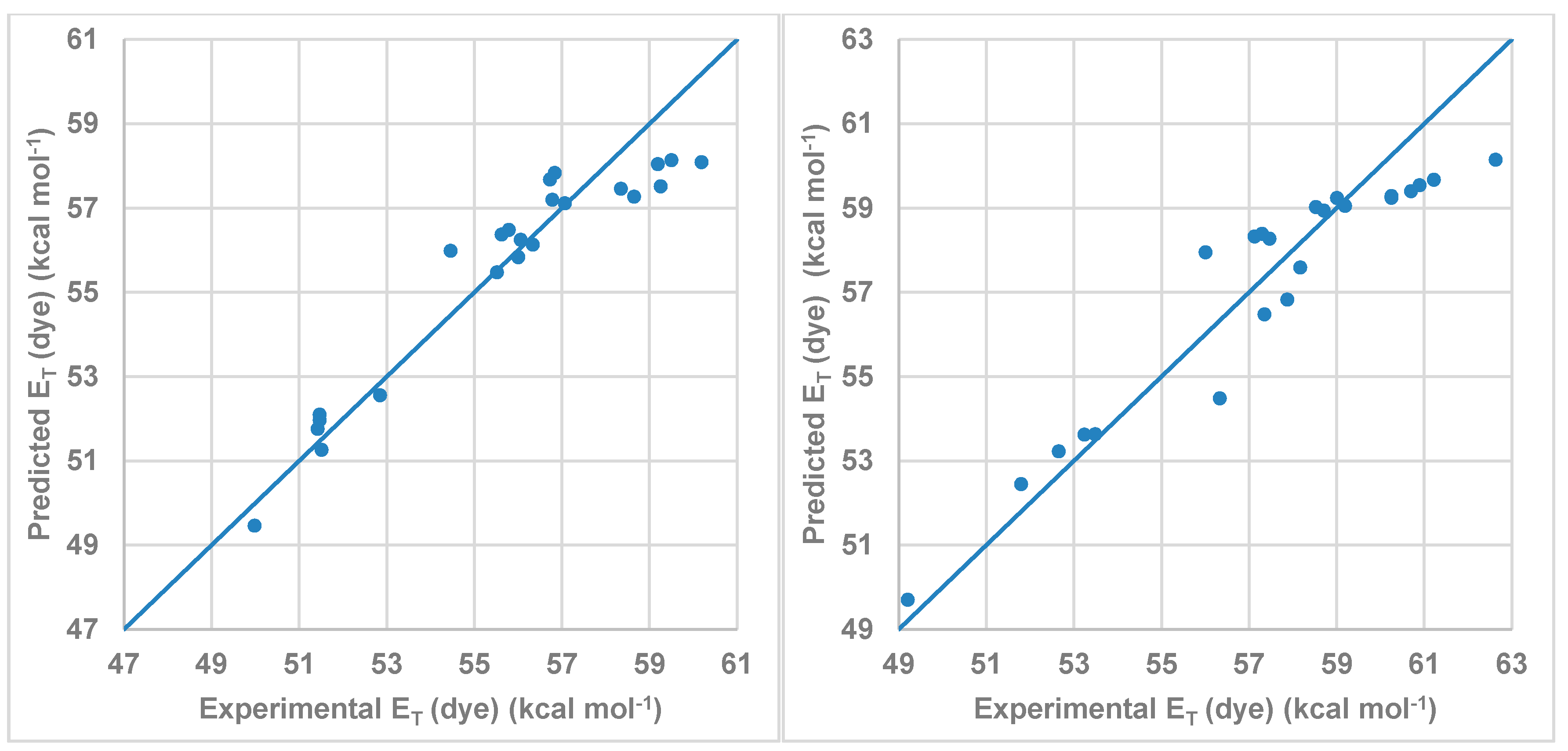
| Compound | Cπ* | Cα | Cβ | RMSE | r |
|---|---|---|---|---|---|
| 1 | −2.177 | −6.282 | −1.763 | 0.9344 | 0.95 |
| 2 | −2.728 | −7.192 | −0.722 | 1.1094 | 0.95 |
3. Experimental Section
| Compound 1 | Compound 2 | |||
|---|---|---|---|---|
| Empirical formula | C23H24N2O | C21H19NO | ||
| Formula weight | 344.44 | 301.37 | ||
| Temperature | 120(2) K | 120(2) K | ||
| Wavelength | 0.71073 Å | 0.71073 Å | ||
| Crystal system | Orthorhombic | Monoclinic | ||
| Space group | P b c a | P 21/c | ||
| Unit cell dimensions | a = 17.3098(12) Å | α = 90° | a = 9.8522(8) Å a = 90° | α = 90° |
| b = 9.8125(10) Å | β = 90° | b = 17.4626(8) Å | β = 111.916(9)° | |
| c = 21.5541(19) Å | γ = 90° | c = 10.0550(7) Å | γ = 90° | |
| Volume | 3661.0(6) Å3 | 1604.9(2) Å3 | ||
| Z | 8 | 4 | ||
| Density (calculated) | 1.250 Mg/m3 | 1.247 Mg/m3 | ||
| Absorption coefficient | 0.077 mm−1 | 0.076 mm−1 | ||
| F(000) | 1472 | 640 | ||
| Crystal size | 0.480 × 0.360 × 0.110 mm3 | 0.110 × 0.060 × 0.020 mm3 | ||
| θ range for data collection | 3.019 to 24.996° | 3.398 to 24.999° | ||
| Index ranges | −20 ≤ h ≤ 20, −10 ≤ k ≤ 11, −25 ≤ l ≤ 25 | −11 ≤ h ≤ 11, −12 ≤ k ≤ 20, −8 ≤ l ≤ 11 | ||
| Reflections collected | 9574 | 6095 | ||
| Independent reflections | 3213 [R(int) = 0.0591] | 2814 [R(int) = 0.0616] | ||
| Completeness to θ = 24.996° | 99.9% | 99.7% | ||
| Refinement method | Full-matrix least-squares on F2 | Full-matrix least-squares on F2 | ||
| Data/restraints/parameters | 3213/0/332 | 2814/0/285 | ||
| Goodness-of-fit on F2 | 1.026 | 0.999 | ||
| Final R indices [I > 2sigma(I)] | R1 = 0.0529, wR2 = 0.0843 | R1 = 0.0615, wR2 = 0.0945 | ||
| R indices (all data) | R1 = 0.0901, wR2 = 0.1027 | R1 = 0.1312, wR2 = 0.1287 | ||
| Extinction coefficient | 0.0020(2) | 0.0040(7) | ||
| Largest diff. peak and hole | 0.194 and −0.237 e·Å−3 | 0.228 and −0.207 e·Å−3 | ||
4. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Bunzli, J.-C.G.; Piguet, C. Taking advantage of luminescent lanthanide ions. Chem. Soc. Rev. 2005, 34, 1048. [Google Scholar] [CrossRef] [PubMed]
- Costero, A.M.; Gil, S.; Parra, M.; Mancini, P.M.E.; Martinez-Mañez, R.; Sancenon, F.; Royo, S. Chromogenic detection of nerve agent mimics. Chem. Commun. 2008, 6002–6004. [Google Scholar] [CrossRef] [PubMed]
- Dale, T.J.; Rebek, J. Hydroxy Oximes as Organophosphorus Nerve Agent Sensors. Angew. Chem. Int. Ed. 2009, 48, 7850. [Google Scholar] [CrossRef] [PubMed]
- Han, S.; Xue, Z.; Wang, Z.; Wen, T.B. Visual and fluorogenic detection of a nerve agent simulant via a Lossen rearrangement of rhodamine–hydroxamate. Chem. Commun. 2010, 46, 8413. [Google Scholar] [CrossRef] [PubMed]
- Xi, C.; Liu, Z.; Kong, L.; Hu, X.; Liu, S. Effects of interaction of folic acid with uranium (VI) and basic triphenylmethane dyes on resonance Rayleigh scattering spectra and their analytical applications. Anal. Chim. Acta 2008, 613, 83–90. [Google Scholar] [CrossRef] [PubMed]
- Eldem, Y.; Özer, I. Electrophilic reactivity of cationic triarylmethane dyes towards proteins and protein-related nucleophiles. Dyes Pigm. 2004, 60, 49–54. [Google Scholar] [CrossRef]
- Jang, M.-S.; Kang, N.-Y.; Kim, K.-S.; Kim, C.H.; Lee, J.-H.; Lee, Y.-C. Mutational analysis of NADH-binding residues in triphenylmethane reductase from Citrobacter sp. strain KCTC 18061P. FEMS Microbiol. Lett. 2007, 271, 78–82. [Google Scholar] [CrossRef] [PubMed]
- Gotor, R.; Costero, A.M.; Gil, S.; Parra, M.; Martínez-Máñez, R.; Sancenón, F.; Gaviña, P. Selective and sensitive chromogenic detection of cyanide and HCN in solution and in gas phase. Chem. Commun. 2013, 49, 5669–5671. [Google Scholar] [CrossRef] [PubMed]
- Gotor, R.; Costero, A.M.; Gil, S.; Parra, M.; Martínez-Máñez, R.; Sancenón, F. A Molecular Probe for the Highly Selective Chromogenic Detection of DFP, a Mimic of Sarin and Soman Nerve Agents. Chem.-Eur. J. 2011, 17, 11994–11997. [Google Scholar] [CrossRef] [PubMed]
- Borisov, S.M.; Klimant, I. A versatile approach for ratiometric time-resolved read-out of colorimetric chemosensors using broadband phosphors as secondary emitters. Anal. Chim. Acta 2013, 787, 219–225. [Google Scholar] [CrossRef] [PubMed]
- Uda, R.M.; Oue, M.; Kimura, K. Specific behavior of crowned crystal violet in cation complexation and photochromism. J. Supramol. Chem. 2002, 2, 311–316. [Google Scholar] [CrossRef]
- Ramkumar, S.; Anandan, S. Bibridged bianchoring metal-free dyes based on phenoxazine and triphenyl amine as donors for dye-sensitized solar cell applications. RSC Adv. 2013, 3, 21535–21543. [Google Scholar] [CrossRef]
- Lewis, G.N.; Magel, T.T.; Lipkin, D. Isomers of Crystal Violet Ion. Their Absorption and Re-emission of Light. J. Am. Chem. Soc. 1942, 64, 1774–1782. [Google Scholar] [CrossRef]
- Duxbury, D.F. The photochemistry and photophysics of triphenylmethane dyes in solid and liquid media. Chem. Rev. 1993, 93, 381–433. [Google Scholar] [CrossRef]
- Lueck, H.B.; McHale, J.L.; Edwards, W.D. Symmetry-breaking solvent effects on the electronic structure and spectra of a series of triphenylmethane dyes. J. Am. Chem. Soc. 1992, 114, 2342. [Google Scholar] [CrossRef]
- Maruyama, Y.; Ishikawa, M.; Satozono, H. Femtosecond Isomerization of Crystal Violet in Alcohols. J. Am. Chem. Soc. 1996, 118, 6257–6263. [Google Scholar] [CrossRef]
- Ishikawa, M.; Maruyama, Y. Femtosecond spectral hole-burning of crystal violet in methanol. New evidence for ground state conformers. Chem. Phys. Lett. 1994, 219, 416–420. [Google Scholar] [CrossRef]
- Angeloni, L.; Smulevich, G.; Marzocchi, M.P. Resonance Raman spectra of conjugated chromophores. Evidence for electronic and vibrational couplings in crystal violet. J. Mol. Struct. 1980, 61, 331–336. [Google Scholar] [CrossRef]
- Clark, F.T.; Drickamer, H.G. High-pressure study of triphenylmethane dyes in polymeric and aqueous media. J. Phys. Chem. 1986, 90, 589–592. [Google Scholar] [CrossRef]
- McKay, R.B.; Hillson, P.J. Metachromatic behaviour of dyes in solution. Interpretation on the basis of interaction between dye ions and counter-ionsMetachromatic behaviour of dyes in solution. Interpretation on the basis of interaction between dye ions and counter-ions. J. Chem. Soc. Trans. Faraday Soc. 1965, 11, 1800–1810. [Google Scholar] [CrossRef]
- Oliveira, C.S.; Branco, K.P.; Baptista, M.S.; Indig, G.L. Solvent and concentration effects on the visible spectra of tri-para-dialkylamino-substituted triarylmethane dyes in liquid solutions. Spectrochim. Acta Part A 2002, 58, 2971–2982. [Google Scholar] [CrossRef]
- Lewis, L.M.; Indig, G.L. Solvent effects on the spectroscopic properties of triarylmethane dyes. Dyes Pigm. 2000, 46, 145–154. [Google Scholar] [CrossRef]
- Kamlet, M.J.; Abboud, J.L.; Taft, R.W. The solvatochromic comparison method. 6. The π* scale of solvent polarities. J. Am. Chem. Soc. 1977, 99, 6027–6038. [Google Scholar] [CrossRef]
- Kamlet, M.J.; Abboud, J.-L.M.; Abraham, M.H.; Taft, R.W. Linear solvation energy relationships. 23. A comprehensive collection of the solvatochromic parameters, π*, α, and β, and some methods for simplifying the generalized solvatochromic equation. J. Org. Chem. 1983, 48, 2877–2887. [Google Scholar] [CrossRef]
- Reichardt, C. Solvatochromic Dyes as Solvent Polarity Indicators. Chem. Rev. 1994, 94, 2319–2358. [Google Scholar] [CrossRef]
- Filarowski, A.; Lopatkova, M.; Lipkowski, P.; Van der Auwearaer, M.; Leen, V.; Dehaen, W. Solvatochromism of BODIPY-Schiff Dye. J. Phys. Chem. B 2015, 119, 2576–2584. [Google Scholar] [CrossRef] [PubMed]
- Agilent (2014) and Agilent (2010). In CrysAlisPRO, Versions 1.171.37.35 and 1.171.34.41; Agilent Technologies, UK Ltd.: Yarnton, UK, 2010/2014.
- Sheldrick, G.M. Crystal structure refinement with SHELXL. Acta Crystallogr. Sect. C 2015, 71, 3–8. [Google Scholar] [CrossRef] [PubMed]
- Farrugia, L.J. WinGX and ORTEP for Windows: An update. J. Appl. Cryst. 2012, 45, 849–854. [Google Scholar] [CrossRef]
- Macrae, C.F.; Bruno, I.J.; Chisholm, J.A.; Edgington, P.R.; McCabe, P.; Pidcock, E.; Rodriguez-Monge, L.; Taylor, R.; van de Streek, J.; Wood, P.A. Mercury CSD 2.0—New features for the visualization and investigation of crystal structures. J. Appl. Crystallogr. 2008, 41, 466–470. [Google Scholar] [CrossRef]
- Sample Availability: Samples of the compounds are not available.
© 2015 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons by Attribution (CC-BY) license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chulvi, K.; Costero, A.M.; Ochando, L.E.; Gil, S.; Vivancos, J.-L.; Gaviña, P. Solvatochromic and Single Crystal Studies of Two Neutral Triarylmethane Dyes with a Quinone Methide Structure. Molecules 2015, 20, 20688-20698. https://doi.org/10.3390/molecules201119724
Chulvi K, Costero AM, Ochando LE, Gil S, Vivancos J-L, Gaviña P. Solvatochromic and Single Crystal Studies of Two Neutral Triarylmethane Dyes with a Quinone Methide Structure. Molecules. 2015; 20(11):20688-20698. https://doi.org/10.3390/molecules201119724
Chicago/Turabian StyleChulvi, Katherine, Ana M. Costero, Luis E. Ochando, Salvador Gil, José-Luis Vivancos, and Pablo Gaviña. 2015. "Solvatochromic and Single Crystal Studies of Two Neutral Triarylmethane Dyes with a Quinone Methide Structure" Molecules 20, no. 11: 20688-20698. https://doi.org/10.3390/molecules201119724
APA StyleChulvi, K., Costero, A. M., Ochando, L. E., Gil, S., Vivancos, J.-L., & Gaviña, P. (2015). Solvatochromic and Single Crystal Studies of Two Neutral Triarylmethane Dyes with a Quinone Methide Structure. Molecules, 20(11), 20688-20698. https://doi.org/10.3390/molecules201119724







