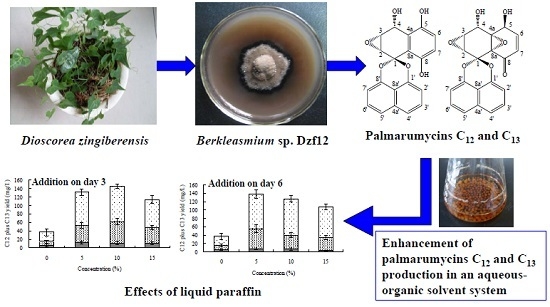
Enhancement of Palmarumycin C12 and C13 Production by the Endophytic Fungus Berkleasmium sp. Dzf12 in an Aqueous-Organic Solvent System
Abstract
:1. Introduction

2. Results and Discussion
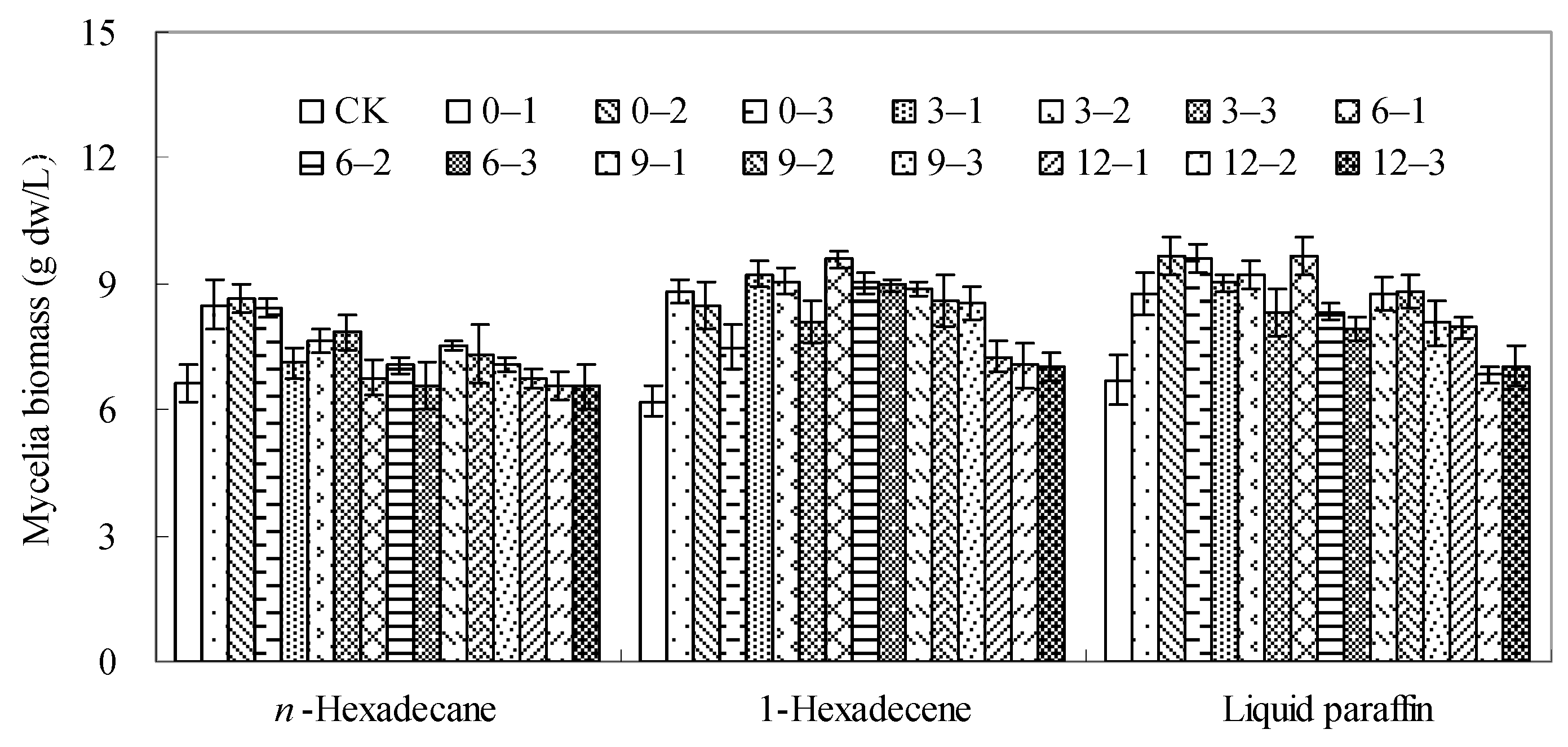
2.1. Effects of the Organic Solvents on Mycelial Growth of Berkleasmium sp. Dzf12
| Organic Solvent | Molecular Weight (Da) | Density (g/mL) | Boiling Point (°C) | Log p |
|---|---|---|---|---|
| n-Dodecane | 170 | 0.75 | 216 | 6.6 |
| n-Hexadecane | 226 | 0.89 | 287 | 8.8 |
| 1-Hexadecene | 224 | 0.78 | 285 | none |
| Liquid paraffin | 150–250 | 0.83–0.86 | 185–250 | none |
| Dibutyl phthalate | 278 | 1.05 | 340 | 5.4 |
| Butyl oleate | 339 | 0.88 | 227–228 | 9.8 |
| Oleic acid | 282 | 0.89 | 360 | 7.7 |
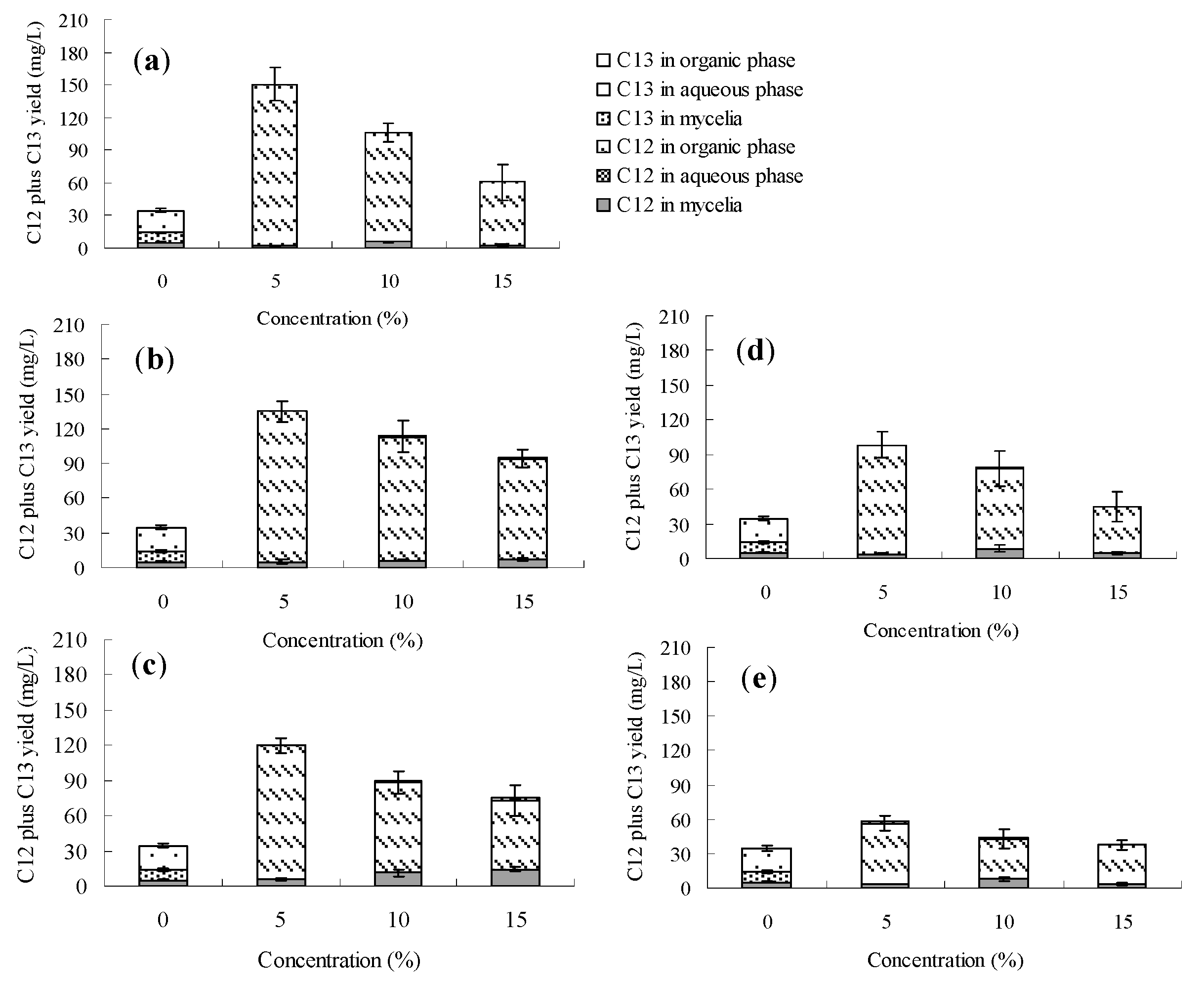
2.2. Effects of the Organic Solvents on Palmarumycin C12 Production on Berkleasmium sp. Dzf12
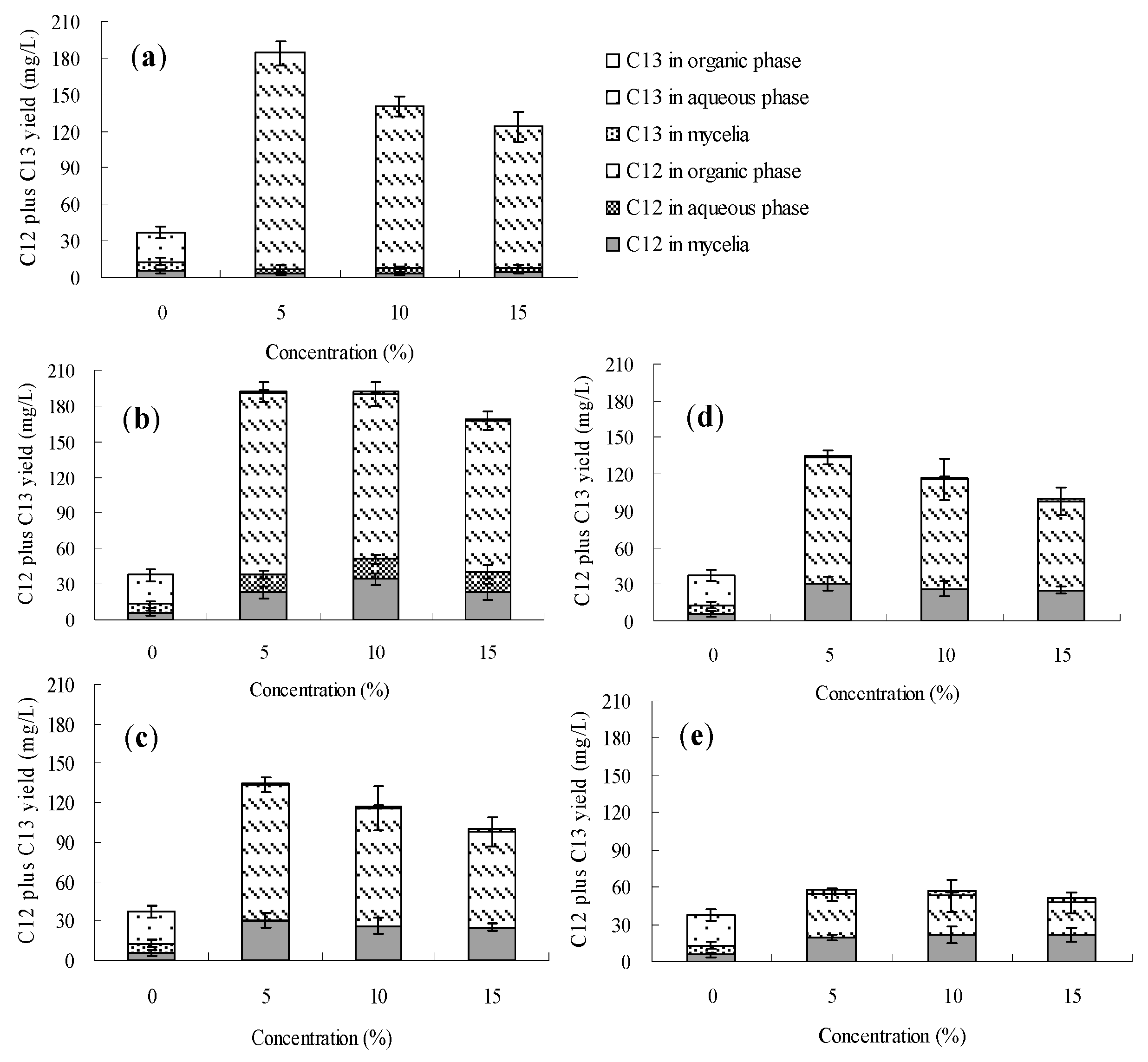
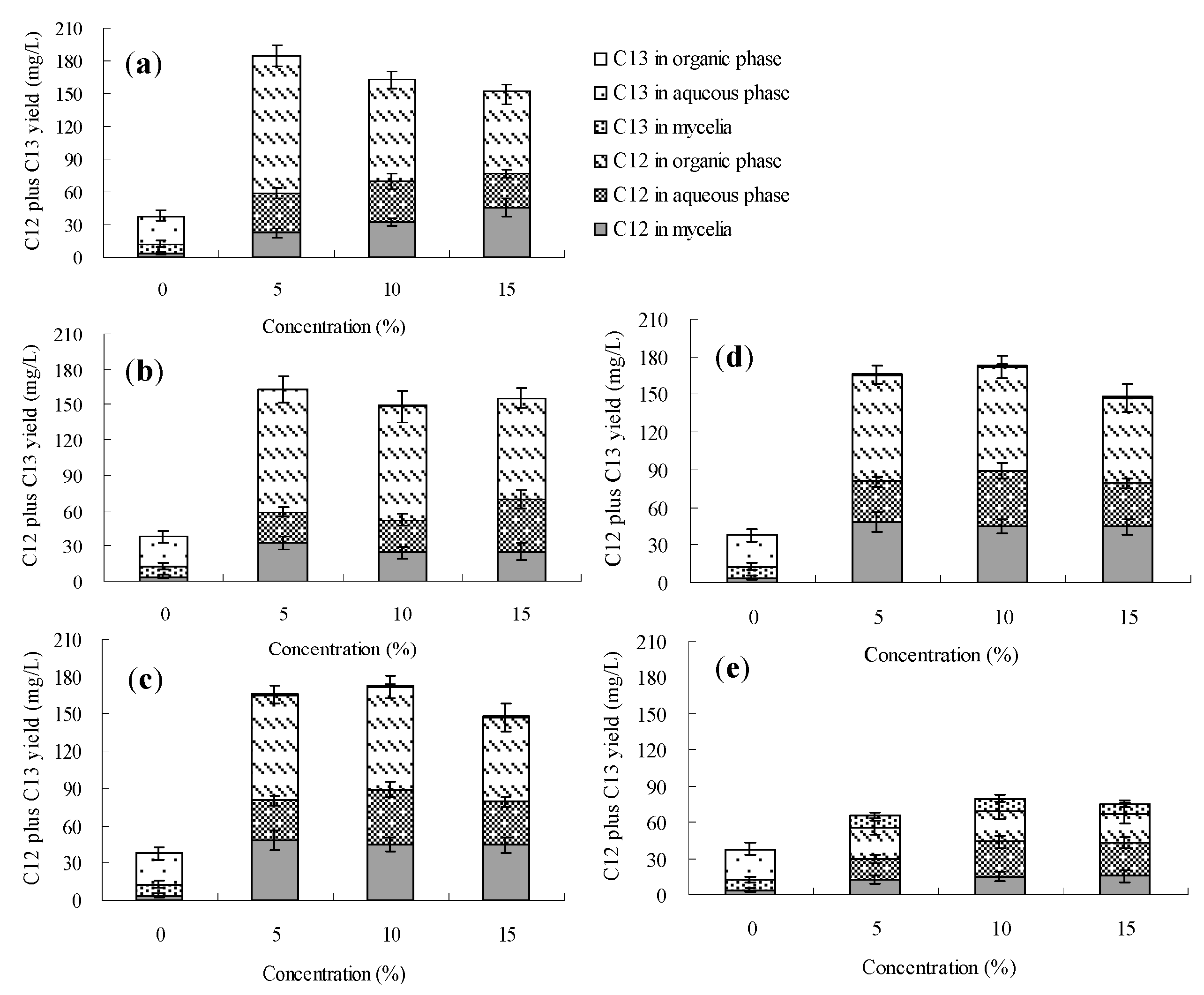
2.3. Effects of the Organic Solvents on Palmarumycin C13 Production on Berkleasmium sp. Dzf12
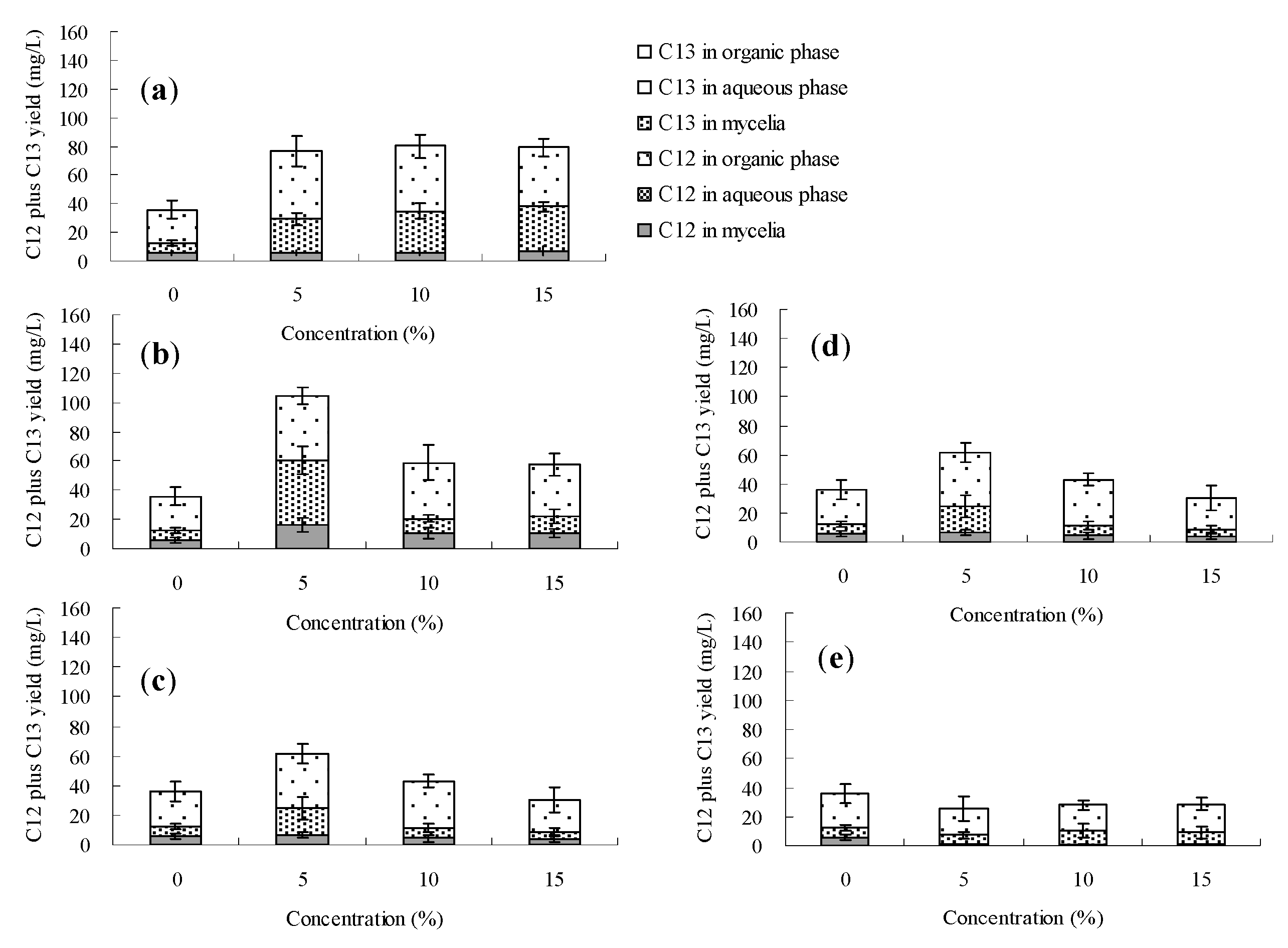
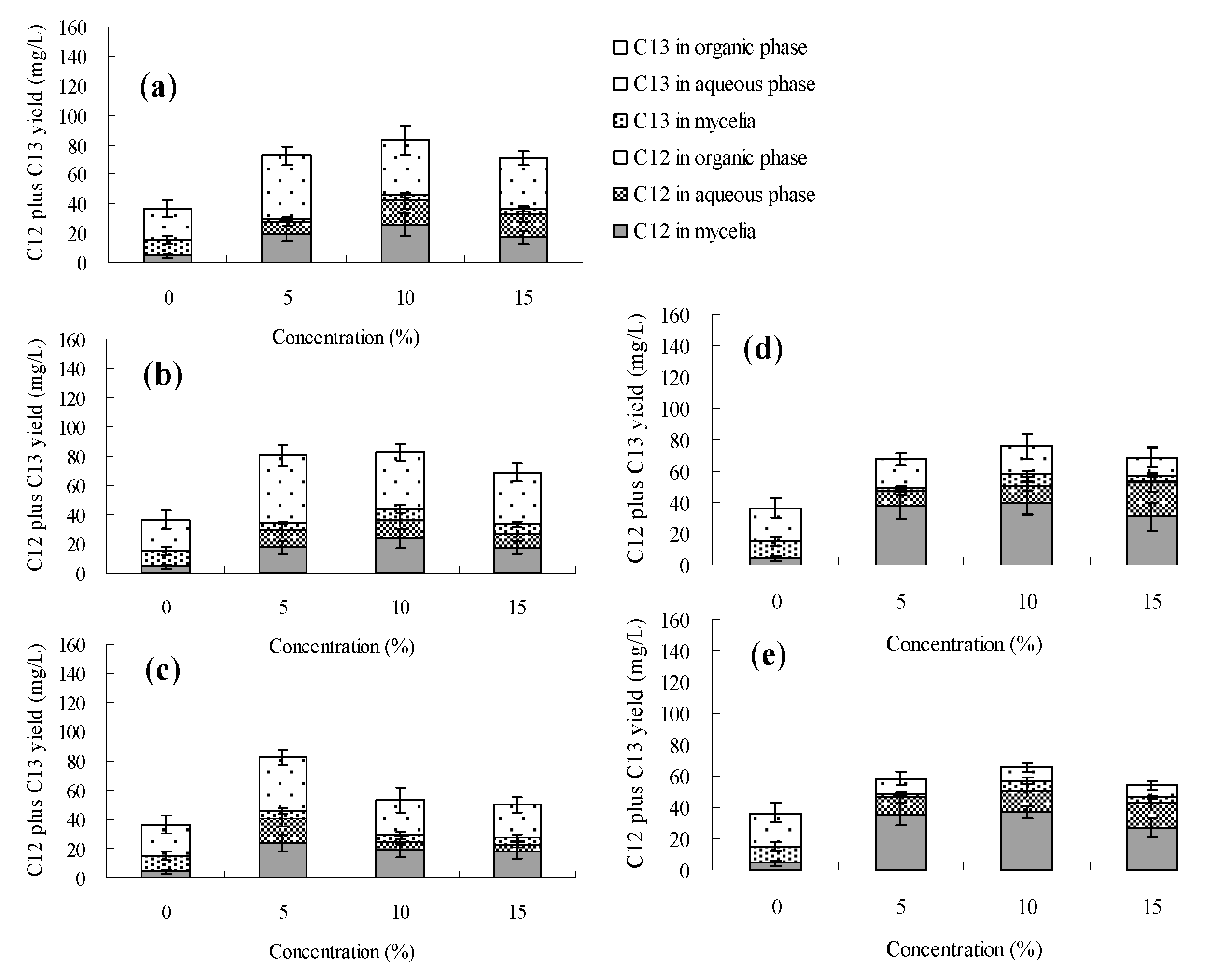

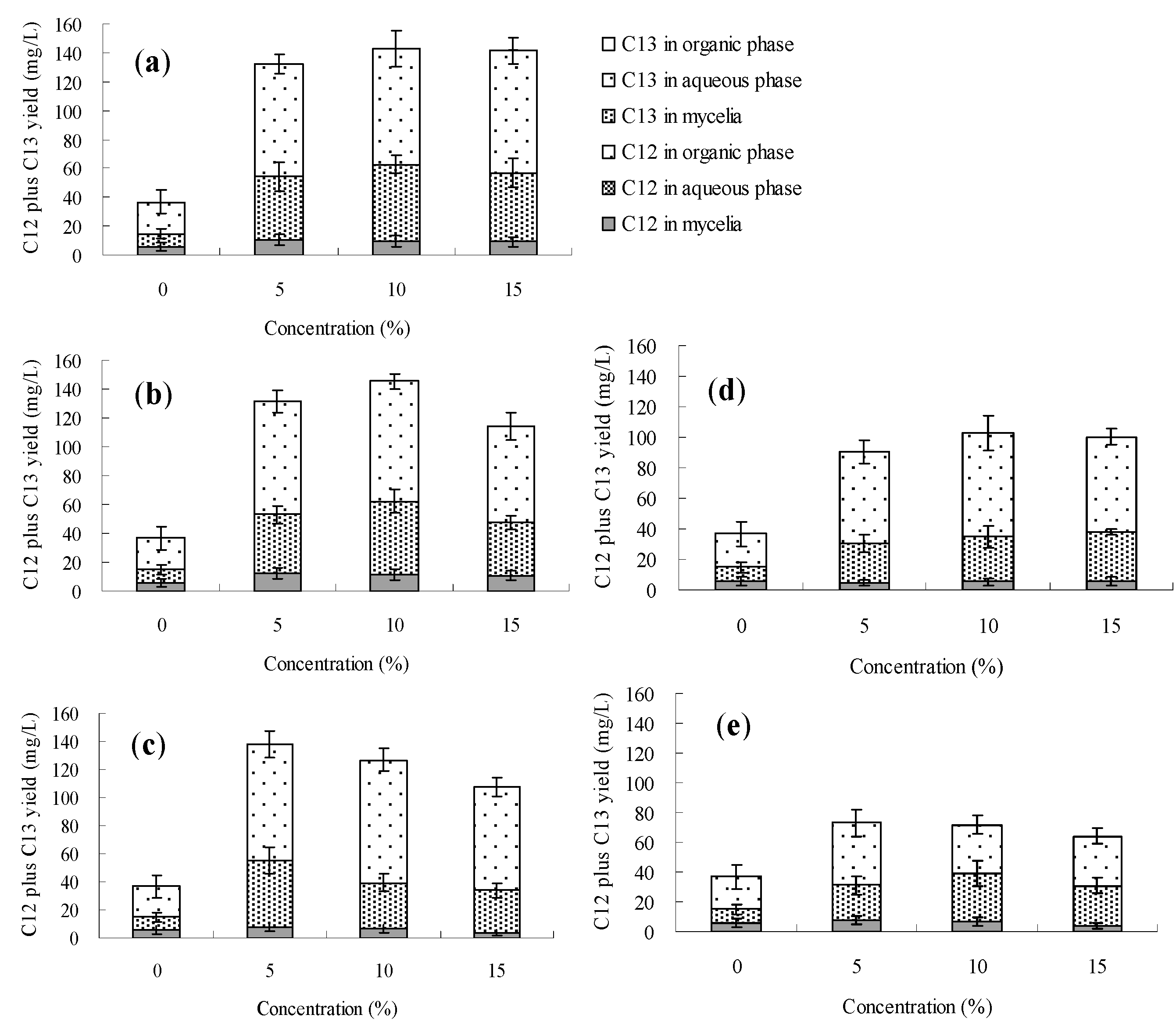
3. Experimental Section
3.1. Endophytic Fungus and Culture Conditions
3.2. Organic Solvents and Two-phase Culture
3.3. Measurement of Mycelia Biomass and Palmarumycins C12 and C13 Yield
3.4. Statistical Analysis
4. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Arnold, A.E.; Mejia, L.C.; Kyllo, D.; Rojas, E.I.; Maynard, Z.; Robbins, N.; Herre, E.A. Fungal endophyte limit pathogen damage in a tropical tree. Proc. Natl. Acad. Sci. USA 2003, 100, 15649–15654. [Google Scholar] [CrossRef] [PubMed]
- Aly, A.H.; Debbab, A.; Kjer, J.; Proksch, P. Fungal endophytes from higher plants: A prolific source of phytochemicals and other bioactive natural products. Fungal Divers. 2010, 41. [Google Scholar] [CrossRef]
- Kusari, S.; Singh, S.; Jayabaskaran, C. Biotechnological potential of plant-associated endophytic fungi: Hope vs. hype. Trends Biotechnol. 2014, 32, 297–303. [Google Scholar] [CrossRef] [PubMed]
- Kour, A.; Shawl, A.S.; Reham, S.; Sultan, P.; Qazi, P.H.; Suden, P.; Khajuria, R.K.; Verma, V. Isolation and identification of an endophytic strain of Fusarium oxysporum producing podophyllotoxin from Juniperus recurva. World J. Microbiol. Biotechnol. 2008, 24, 1115–1121. [Google Scholar] [CrossRef]
- Li, X.-B.; Xie, F.; Liu, S.-S.; Ying, Li.; Zhou, J.-C.; Liu, Y.-Q.; Yuan, H.-Q.; Lou, H.-X. Naphtho-γ-pyrones from endophyte Aspergillus niger occurring in the liverwort Heteroscyphus tener (Steph.) Schiffn. Chem. Biodivers. 2013, 10, 1193–1200. [Google Scholar] [CrossRef] [PubMed]
- Li, D.-M.; Zhang, Y.-H.; Ji, H.-X.; Wu, X.; Pei, Y.-H.; Bai, J. Tricycloalternarene derivatives from endophytic fungus Alternaria tenuissima SY-P-07. Nat. Prod. Res. 2013, 27, 1877–1881. [Google Scholar] [CrossRef] [PubMed]
- Zhou, L.; Zhao, J.; Shan, T.; Cai, X.; Peng, Y. Spirobisnaphthalenes from fungi and their biological activities. Mini-Rev. Med. Chem. 2010, 10, 977–989. [Google Scholar] [CrossRef] [PubMed]
- Cai, Y.S.; Guo, Y.W.; Krohn, K. Structure, bioactivities, biosynthetic relationships and chemical synthesis of the spirodioxynaphthalenes. Nat. Prod. Rep. 2010, 27, 1840–1870. [Google Scholar] [CrossRef] [PubMed]
- Krohn, K.; Michel, A.; Floerke, U.; Aust, H.J.; Draeger, S.; Schulz, B. Palmarumycins C1–C16 from Coniothyrium sp.: Isolation, structure elucidation, and biological activity. Liebigs Ann. Chem. 1994, 11, 1099–1108. [Google Scholar] [CrossRef]
- Cai, X.; Shan, T.; Li, P.; Huang, Y.; Xu, L.; Zhou, L.; Wang, M.; Jiang, W. Spirobisnaphthalenes from the endophytic fungus Dzf12 of Dioscorea zingiberensis and their antimicrobial activities. Nat. Prod. Commun. 2009, 4, 1469–1472. [Google Scholar] [PubMed]
- Petersen, F.; Moerker, T.; Vanzanella, F.; Peter, H.H. Production of cladosporine bisepoxide, a new fungal metabolite. J. Antibiot. 1994, 47, 1098–1103. [Google Scholar] [CrossRef] [PubMed]
- Shan, T.; Tian, J.; Wang, X.; Mou, Y.; Mao, Z.; Lai, D.; Dai, J.; Peng, Y.; Zhou, L.; Wang, M. Bioactive spirobisnaphthalenes from the endophytic fungus Berkleasmium sp. J. Nat. Prod. 2014, 77, 2151–2160. [Google Scholar] [CrossRef] [PubMed]
- Schlingmann, G.; West, R.R.; Milne, L.; Pearce, C.J.; Carter, G.T. Diepoxins, novel fungal metabolites with antibiotic activity. Tetrahedron Lett. 1993, 34, 7225–7228. [Google Scholar] [CrossRef]
- Chu, M.; Truumees, I.; Patel, M.G.; Gullo, V.P.; Blood, C.; King, I.; Pai, J.K.; Puar, M.S. A novel class of antitumor metabolites from the fungus Nattrassia mangiferae. Tetrahedron Lett. 1994, 35, 1343–1346. [Google Scholar] [CrossRef]
- Chu, M.; Truumees, I.; Patel, M.G.; Gullo, V.P.; Puar, M.S.; McPhail, A.T. Structure of Sch 49209: A novel antitumor agent from the fungus Nattrassia mangiferae. J. Org. Chem. 1994, 59, 1222–1223. [Google Scholar] [CrossRef]
- Bode, H.B.; Zeeck, A. UV mutagenesis and enzyme inhibitors as tools to elucidate the late biosynthesis of the spirobisnaphthalenes. Phytochemistry 2000, 55, 311–316. [Google Scholar] [CrossRef]
- Zhao, J.; Wang, X.; Sun, W.; Mou, Y.; Peng, Y.; Zhou, L. Medium optimization for palmarumycin C13 production in liquid culture of endophytic fungus Berkleasmium sp. Dzf12 using response surface methodology. Electron. J. Biotechnol. 2013, 16, 16–16. [Google Scholar] [CrossRef]
- Mou, Y.; Luo, H.; Mao, Z.; Shan, T.; Sun, W.; Zhou, K.; Zhou, L. Enhancement of palmarumycins C12 and C13 production in liquid culture of endophytic fungus Berkleasmium sp. Dzf12 after treatments with metal ions. Int. J. Mol. Sci. 2013, 14, 979–998. [Google Scholar] [CrossRef] [PubMed]
- Ruhland, C.T.; Day, T.A. Effects of ultraviolet-B radiation on leaf elongation, production and phenylpropanoid concentrations of Deschampsia antarctica and Colobanthus quitensis in Antarctica. Physiol. Plantarum 2000, 109, 244–251. [Google Scholar] [CrossRef]
- Wu, J.; Ge, X. Oxidative burst, jasmonic acid biosynthesis, and taxol production induced by low-energy ultrasound in Taxus chinensis cell suspension cultures. Biotechnol. Bioeng. 2004, 85, 714–721. [Google Scholar] [CrossRef] [PubMed]
- Quinn, L.; Dempsey, R.; Casey, E.; Kane, A.; Murphy, C.D. Production of drug metabolites by immobilized Cunnighamella elegans: From screening to scale up. J. Ind. Microbiol. Biotechnol. 2015, 42, 799–806. [Google Scholar] [CrossRef] [PubMed]
- Luo, H.; Liu, H.; Cao, Y.; Xu, D.; Mao, Z.; Mou, Y.; Meng, J.; Lai, D.; Liu, Y.; Zhou, L. Enhanced production of botrallin and TMC-264 with in situ macroporous resin adsorption in mycelial liquid culture of the endophytic fungus Hyalodendriella sp. Ponipodef12. Molecules 2014, 19, 14221–14234. [Google Scholar] [CrossRef] [PubMed]
- Ola, A.R.B.; Thomy, D.; Lai, D.; Brotz-Oesterhelt, H.; Proksch, P. Inducing secondary metabolites production by the endophytic fungus Fusarium tricinctum through coculture with Bacillus subtilis. J. Nat. Prod. 2013, 76, 2094–2099. [Google Scholar] [CrossRef] [PubMed]
- Whitt, J.; Shpley, S.M.; Newman, D.J.; Zuck, K.M. Tetramic acid analogues produced by coculture of Saccharopolyspora erythraea with Fusarium pallidoroseum. J. Nat. Prod. 2014, 77, 173–177. [Google Scholar] [CrossRef] [PubMed]
- Zhou, L.; Wu, J. Development and application of medicinal plant tissue cultures for production of drugs and herbal medicinals in China. Nat. Prod. Rep. 2006, 23, 789–810. [Google Scholar] [CrossRef] [PubMed]
- Malik, S.; Mirjalili, M.H.; Fett-Neto, A.G.; Mazzafera, P.; Bonfill, M. Living between two worlds: Two-phase culture systems for producing plant secondary metabolites. Crit. Rev. Biotechnol. 2013, 33. [Google Scholar] [CrossRef] [PubMed]
- Byun, S.Y.; Pedersen, H.; Chin, C.K. Two-phase culture for the enhanced production of benzophenanthridine alkaloids in cell suspensions of Eschscholtzia californica. Phytochemistry 1990, 29, 3135–3139. [Google Scholar] [CrossRef]
- Byun, S.Y.; Pedersen, H. Two-phase airlift fermentor operation with elicitation for the enhanced production of benzophenanthridine alkaloids in cell suspensions of Eschscholtzia californica. Biotechnol. Bioeng. 1994, 44, 14–20. [Google Scholar] [CrossRef] [PubMed]
- Dutta, A.; Pedersen, H.; Chin, C.-K. Two-phase culture system for plant cells. Ann. N. Y. Acad. Sci. 1994, 745, 251–260. [Google Scholar] [CrossRef] [PubMed]
- Staebler, A.; Cruz, A.; Goot, W.; Pinheiro, H.M.; Cabral, J.M.S.; Fernandes, P. Optimization of androstenedione production in an organic-aqueous two-liquid phase system. J. Mol. Catal. B Enzym. 2004, 29, 19–23. [Google Scholar] [CrossRef]
- Lin, J.X.; Bai, L.Q.; Deng, Z.X.; Zhong, J.J. Enhanced production of ansamitocin P-3 by addition of isobutanol in fermentation of Actinosynnema pretiosum. Bioresour. Technol. 2011, 102, 1863–1868. [Google Scholar] [CrossRef] [PubMed]
- Collins-Pavao, M.; Chinb, C.K.; Pedersen, H. Taxol partitioning in two-phase plant cell cultures of Taxus brevifolia. J. Biotechnol. 1996, 49, 95–100. [Google Scholar] [CrossRef]
- Zhang, J.G.; Wang, X.D.; Zhang, J.N.; Wei, D.Z. Oxygen vectors used for S-adenosylmethionine production in recombinant Pichia pastoris with sorbitol as supplemental carbon source. J. Biosci. Bioeng. 2008, 105, 335–340. [Google Scholar] [CrossRef] [PubMed]
- Lopes da Silva, T.; Reis, A.; Roseiro, J.C.; Hewitt, C.J. Physiological effects of the addition of n-dodecane as an oxygen vector during steady-state Bacillus licheniformis thermophillic fermentations perturbed by a starvation period or a glucose pulse. Biochem. Eng. J. 2008, 42, 208–216. [Google Scholar] [CrossRef]
- Zhang, D.; Feng, X.H.; Li, S.; Chen, F.; Xua, H. Effects of oxygen vectors on the synthesis and molecular weight of poly(γ-glutamic acid) and the metabolic characterization of Bacillus subtilis NX-2. Process Biochem. 2012, 47, 2103–2109. [Google Scholar] [CrossRef]
- Wang, J.L. Enhancement of citric acid production by Aspergillus niger using n-dodecane as an oxygen-vector. Process Biochem. 2000, 35, 1079–1083. [Google Scholar] [CrossRef]
- Galaction, A.I.; Cascaval, D.; Oniscu, C.; Turnea, M. Enhancement of oxygen mass transfer in stirred bioreactors using oxygen-vectors. 1. Simulated fermentation broths. Bioprocess Biosyst. Eng. 2004, 26, 231–238. [Google Scholar] [CrossRef] [PubMed]
- Li, M.H.; Meng, X.M.; Diao, E.J.; Dua, F.L.; Zhao, X.Y. Productivity enhancement of S-adenosylmethionine in Saccharomyces cerevisiae using n-hexadecane as oxygen vector. J. Chem. Technol. Biotechnol. 2012, 87, 1379–1384. [Google Scholar] [CrossRef]
- Wei, Z.; Yuan, Y.; Wu, Z.; Wu, J. Improved production of paclitaxel from suspension culture of Taxus chinensis var. mairei by in situ extraction with organic solvents. Chin. J. Chem. Eng. 2003, 11, 224–226. [Google Scholar]
- Zare, K.; Nazemiyeh, H.; Movafeghi, A.; Khosrowshahli, M.; Motallebi-Azar, A.; Dadpour, M.; Omidi, Y. Bioprocess engineering of Echium italicum L.: Induction of shikonin and alkanin derivatives by two liquid-phase suspension cultures. Plant Cell Tiss. Org. Cult. 2010, 100, 157–164. [Google Scholar] [CrossRef]
- Zhao, J.; Zheng, B.; Li, Y.; Shan, T.; Mou, Y.; Lu, S.; Li, P.; Zhou, L. Enhancement of diepoxin ζ production by yeast extract and its fractions in liquid culture of Berkleasmium-like endophytic fungus Dzf12 from Dioscorea zingiberensis. Molecules 2011, 16, 847–856. [Google Scholar] [CrossRef] [PubMed]
- Zhao, J.; Li, Y.; Shan, T.; Mou, Y.; Zhou, L. Enhancement of diepoxin ζ production with in situ resin adsorption in mycelial liquid culture of the endophytic fungus Berkleasmium sp. Dzf12 from Dioscorea zingiberensis. World J. Microbiol. Biotechnol. 2011, 27, 2753–2758. [Google Scholar] [CrossRef]
- Li, Y.; Shan, T.; Mou, Y.; Li, P.; Zhao, J.; Zhao, W.; Peng, Y.; Zhou, L.; Ding, C. Enhancement of palmarumycins C12 and C13 production in liquid culture of the endophytic fungus Berkleasmium sp. Dzf12 by oligosaccharides from its host plant Dioscorea zingiberensis. Molecules 2012, 17, 3761–3773. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Li, P.; Mou, Y.; Zhao, J.; Shan, T.; Ding, C.; Zhou, L. Enhancement of diepoxin ζ production in liquid culture of endophytic fungus Berkleasmium sp. Dzf12 by polysaccharides from its host plant Dioscorea zingiberensis. World J. Microbiol. Biotechnol. 2012, 28, 1407–1413. [Google Scholar] [CrossRef] [PubMed]
- Laane, C.; Boeren, S.; Vos, K.; Veeger, C. Rules for optimization of biocatalysis in organic solvents. Biotechnol. Bioeng. 1987, 30, 81–87. [Google Scholar] [CrossRef] [PubMed]
- Wang, C.G.; Wu, J.Y.; Mei, X.G. Enhanced taxol production and release in Taxus chinensis cell suspension cultures with selected organic solvents and sucrose feeding. Biotechnol. Prog. 2001, 17, 89–94. [Google Scholar] [CrossRef] [PubMed]
- Xu, F.; Yuan, Q.P.; Zhu, Y. Improved production of lycopene and β-carotene by Blakeslea trispora with oxygen-vectors. Process Biochem. 2007, 42, 289–293. [Google Scholar] [CrossRef]
- Giridhar, R.; Srivastava, A.K. Productivity enhancement in l-sorbose fermentation using oxygen vector. Enzyme Microb. Technol. 2000, 27, 537–541. [Google Scholar] [CrossRef]
- Lai, L.T.; Tsai, T.; Wang, T.C. Application of oxygen vectors to Aspergillus terreus cultivation. J. Biosci. Bioeng. 2002, 94, 453–459. [Google Scholar] [CrossRef]
- Wu, J.; Lin, L. Enhancement of taxol production and release in Taxus chinensis cell cultures by ultrasound, methyl jasmonate and in situ solvent extraction. Appl. Microbiol. Biotechnol. 2003, 62, 151–155. [Google Scholar] [CrossRef] [PubMed]
- Li, Z.; Yu, J.; Ren, X.; Ge, Y.; Chen, Q.; Zhou, L.; Xiao, Y.; Liu, W. Effect of different kinds of organic phase on the content of cordycepin from Paecilomyces verticillatus. Sci. Technol. Food Ind. 2011, 32, 226–227. [Google Scholar]
- Mou, Y.; Meng, J.; Fu, X.; Wang, X.; Tian, J.; Wang, M.; Peng, Y.; Zhou, L. Antimicrobial and antioxidant activities and effect of 1-hexadecene addition on palmarumycin C2 and C3 yields in liquid culture of endophytic fungus Berkleasmium sp. Dzf12. Molecules 2013, 18, 15587–15599. [Google Scholar] [CrossRef] [PubMed]
- Narta, U.; Roy, S.; Kanwar, S.S.; Azmi, W. Improved production of L-asparaginase by Bacillus brevis cultivated in the presence of oxygen-vectors. Bioresource Technol. 2011, 102, 2083–2085. [Google Scholar] [CrossRef] [PubMed]
- Sample Availability: Samples of palmarumycins C12 and C13 are available from the authors.
© 2015 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons by Attribution (CC-BY) license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mou, Y.; Xu, D.; Mao, Z.; Dong, X.; Lin, F.; Wang, A.; Lai, D.; Zhou, L.; Xie, B. Enhancement of Palmarumycin C12 and C13 Production by the Endophytic Fungus Berkleasmium sp. Dzf12 in an Aqueous-Organic Solvent System. Molecules 2015, 20, 20320-20333. https://doi.org/10.3390/molecules201119700
Mou Y, Xu D, Mao Z, Dong X, Lin F, Wang A, Lai D, Zhou L, Xie B. Enhancement of Palmarumycin C12 and C13 Production by the Endophytic Fungus Berkleasmium sp. Dzf12 in an Aqueous-Organic Solvent System. Molecules. 2015; 20(11):20320-20333. https://doi.org/10.3390/molecules201119700
Chicago/Turabian StyleMou, Yan, Dan Xu, Ziling Mao, Xuejiao Dong, Fengke Lin, Ali Wang, Daowan Lai, Ligang Zhou, and Bingyan Xie. 2015. "Enhancement of Palmarumycin C12 and C13 Production by the Endophytic Fungus Berkleasmium sp. Dzf12 in an Aqueous-Organic Solvent System" Molecules 20, no. 11: 20320-20333. https://doi.org/10.3390/molecules201119700
APA StyleMou, Y., Xu, D., Mao, Z., Dong, X., Lin, F., Wang, A., Lai, D., Zhou, L., & Xie, B. (2015). Enhancement of Palmarumycin C12 and C13 Production by the Endophytic Fungus Berkleasmium sp. Dzf12 in an Aqueous-Organic Solvent System. Molecules, 20(11), 20320-20333. https://doi.org/10.3390/molecules201119700