Cytotoxic Activities, SAR and Anti-Invasion Effects of Butylphthalide Derivatives on Human Hepatocellular Carcinoma SMMC7721 Cells
Abstract
:1. Introduction
2. Results and Discussion
2.1. Isolation and Structure Identification of Compounds 1–8
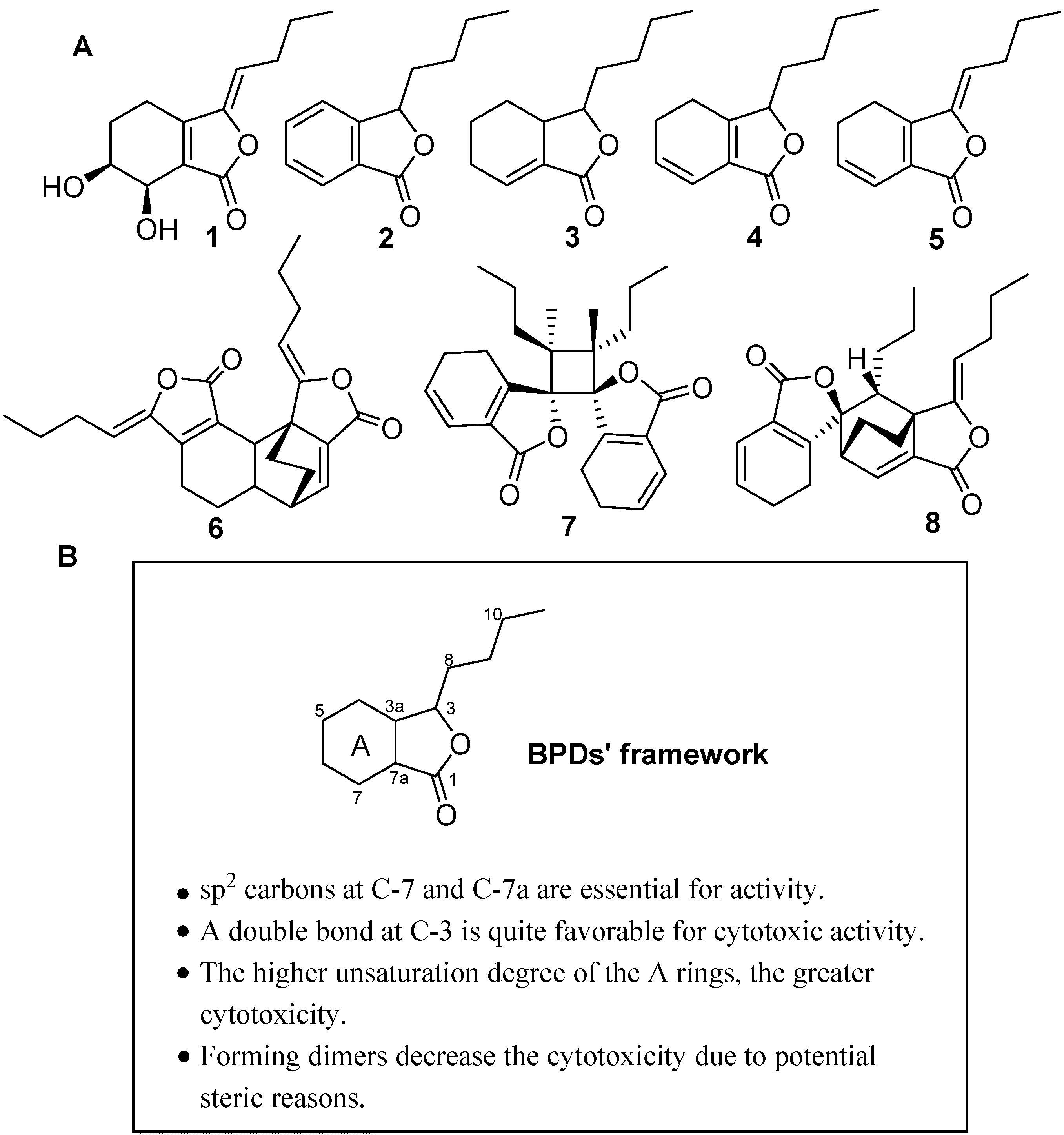
2.2. Cytotoxic Effect of BPDs on H460, SMMC7721 and BGC823 Cells
| Compound | H460 | SMMC7721 | BGC823 |
|---|---|---|---|
| 1 a | - | - | - |
| 2 | 32.0 ± 2.1 | 22.4 ± 1.0 | 25.5 ± 1.0 |
| 3 | 69.0 ± 5.3 | 53.3 ± 1.3 | 49.9 ± 2.5 |
| 4 | 86.1 ± 10.7 | 71.2 ± 10.1 | 80.7 ± 10.9 |
| 5 | 13.3 ± 0.7 | 3.2 ± 0.3 | 4.0 ± 0.1 |
| 6 | 26.9 ± 1.6 | 18.8 ± 1.2 | 55.1 ± 3.5 |
| 7 | 34.8 ± 0.5 | 72.5 ± 8.7 | 69.7 ± 10.3 |
| 8 | 6.4 ± 0.4 | 13.4 ± 1.0 | 19.1 ± 2.4 |
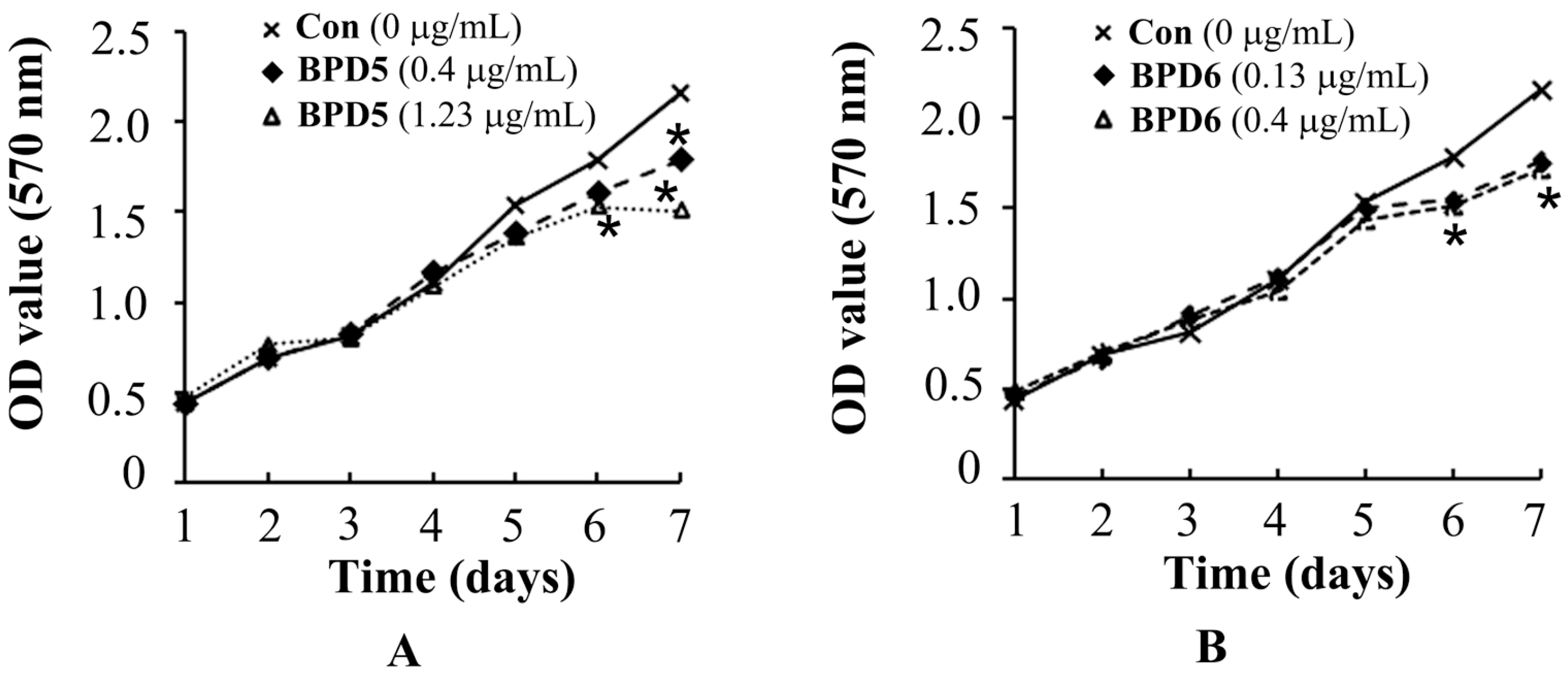
2.3. Effects of BPDs 5 and 6 on the Viability of SMMC7721 Cells
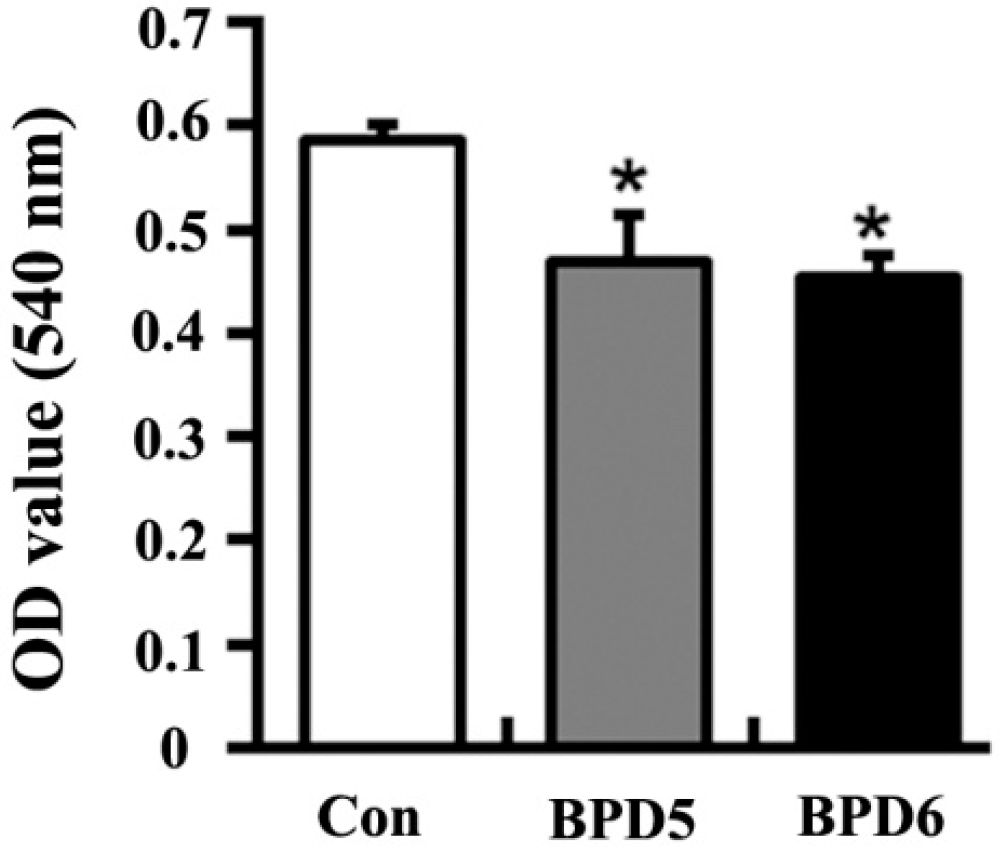
2.4. Effects of BPDs 5 and 6 on Adhesion, Migration and Invasion in SMMC7721 Cells
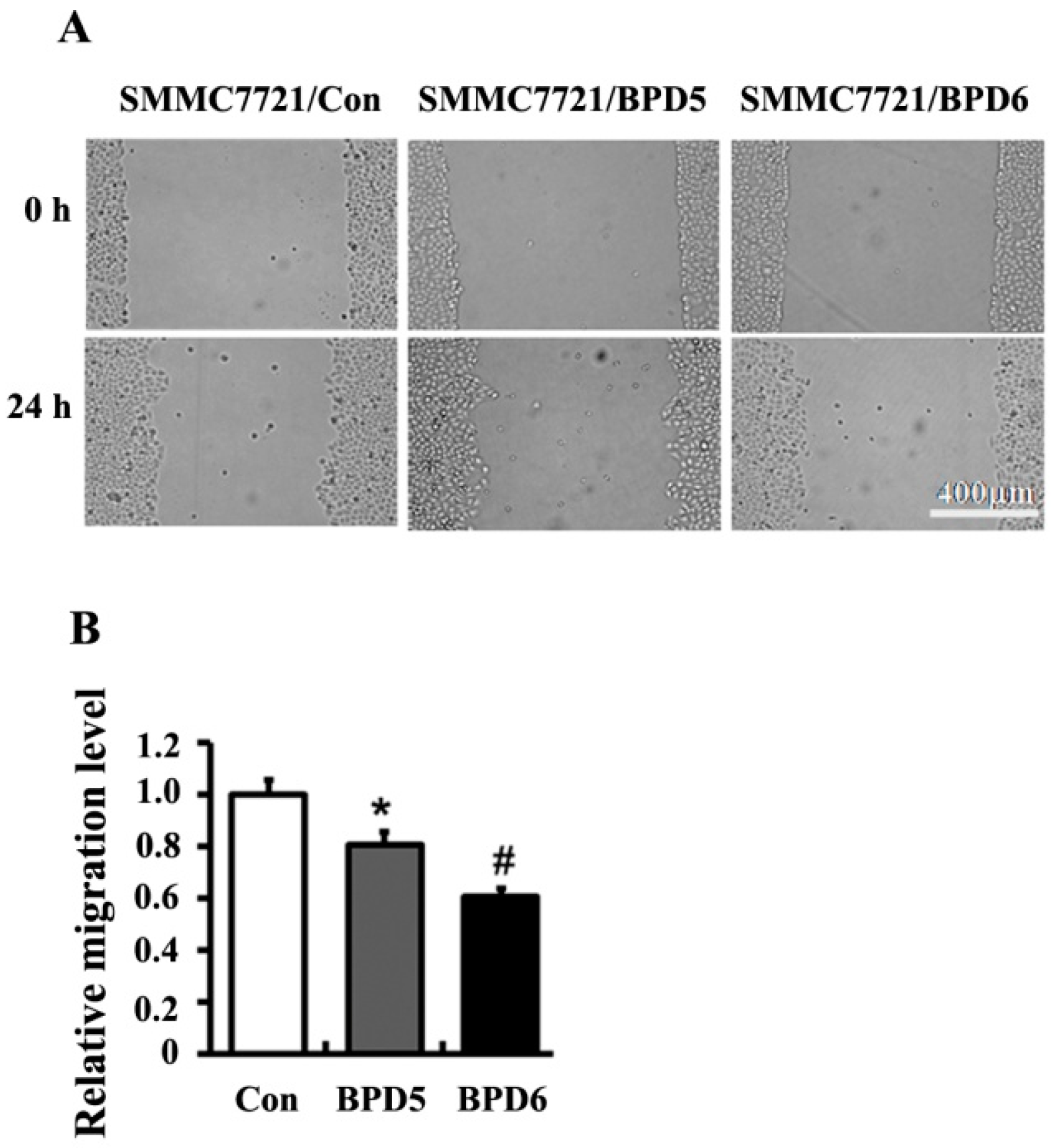
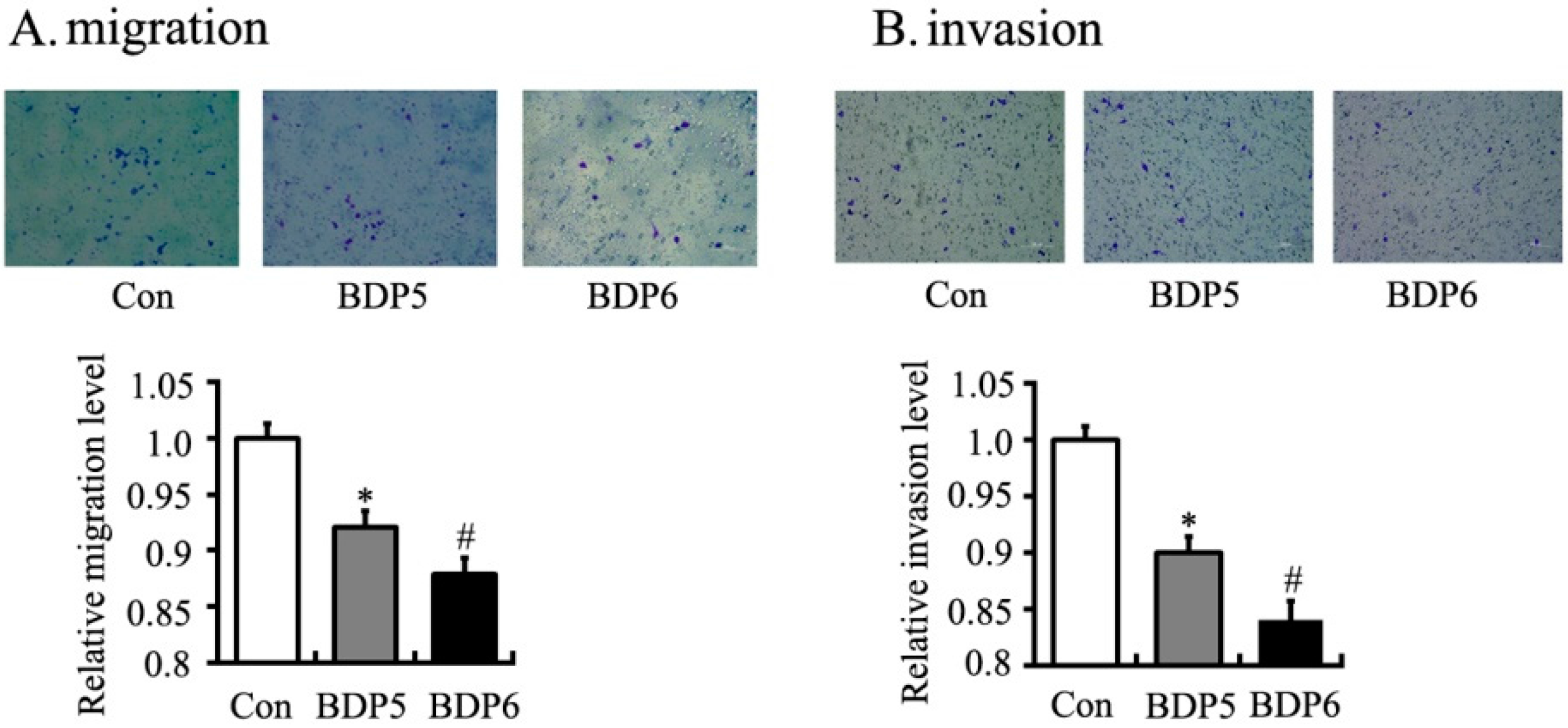
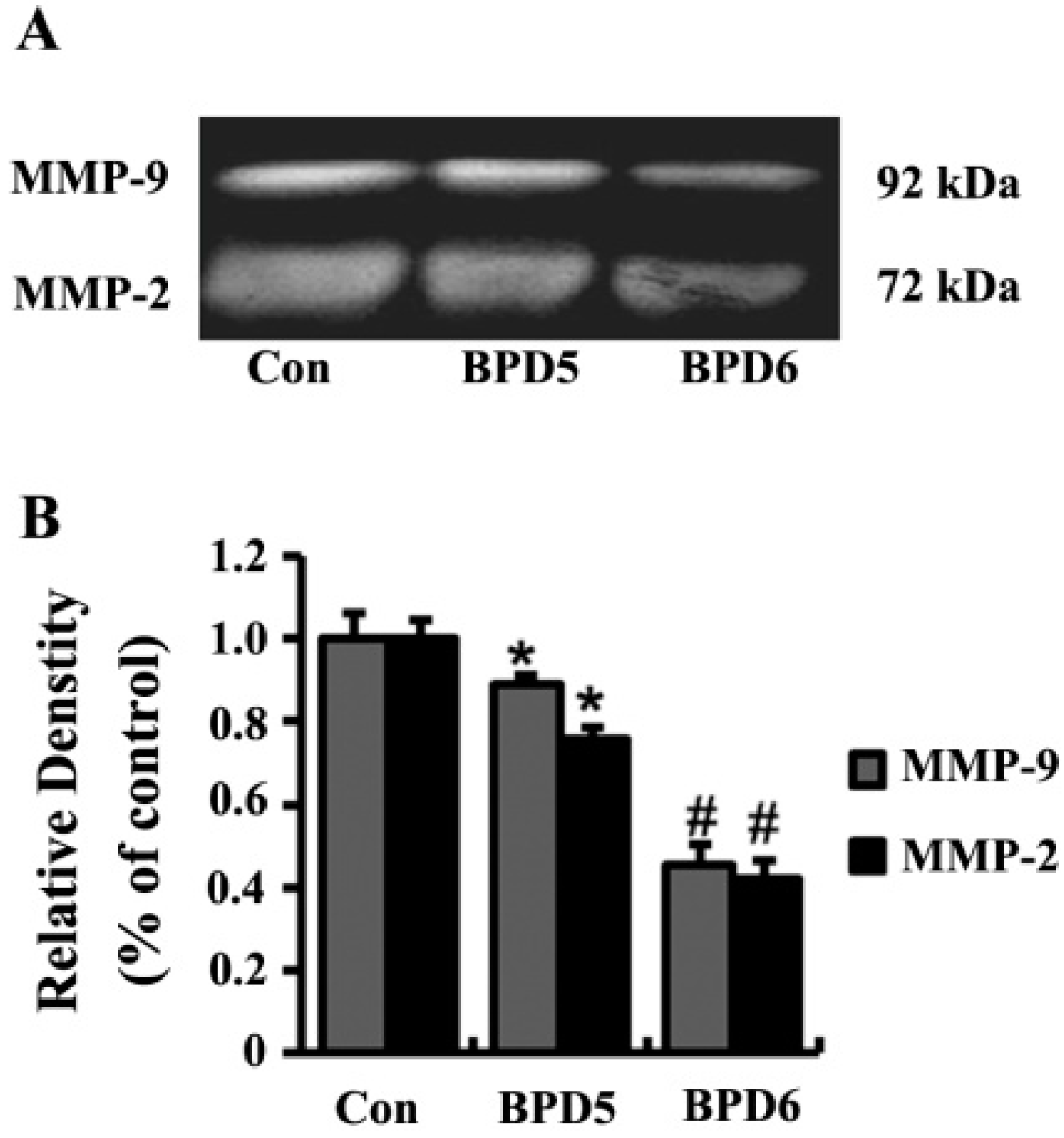
3. Experimental Section
3.1. General Experimental
3.2. Isolation and Preparation of Compounds
3.3. Cytotoxicity Assay
3.4. Cell Adhesion Assay
3.5. Wound Healing Assay
3.6. Transwell Chamber Assay
3.7. Gelatin Zymography
3.8. Statistical Analysis
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Momin, R.A.; Nair, M.G. Mosquitocidal, nematicidal, and antifungal compounds from Apium graveolens L. seeds. J. Agric. Food Chem. 2001, 49, 142–145. [Google Scholar] [CrossRef] [PubMed]
- Beck, J.J.; Chou, S.C. The structural diversity of phthalides from the Apiaceae. J. Nat. Prod. 2007, 70, 891–900. [Google Scholar] [CrossRef] [PubMed]
- Yi, T.; Leung, K.S.; Lu, G.H.; Zhang, H. Comparative analysis of Ligusticum chuanxiong and related umbelliferous medicinal plants by high performance liquid chromatography-electrospray ionization mass spectrometry. Planta Med. 2007, 73, 392–398. [Google Scholar] [CrossRef] [PubMed]
- Wedge, D.E.; Klun, J.A.; Tabanca, N.; Demirci, B.; Ozek, T.; Baser, K.H.C.; Liu, Z.; Zhang, S.; Cantrell, C.; Zhang, J. Bioactivity-guided fractionation and GC/MS fingerprinting of Angelica sinensis and Angelica archangelica root components for antifungal and mosquito deterrent activity. J. Agric. Food Chem. 2009, 57, 464–470. [Google Scholar] [CrossRef] [PubMed]
- Cao, Y.; Zhang, W.; He, J.; He, L.; Xu, C. Ligustilide induces vasodilatation via inhibiting voltage dependent calcium channel and receptor-mediated Ca2+ influx and release. Vasc. Pharmacol. 2006, 45, 171–176. [Google Scholar] [CrossRef] [PubMed]
- Chen, Q.; Lee, J.; Jin, W.; Youn, U.; Kim, H.; Lee, I.; Zhang, X.; Song, K.; Seong, Y.; Bae, K. Cytotoxic constituents from Angelicae sinensis Radix. Arch. Pharm. Res. 2007, 30, 565–569. [Google Scholar] [CrossRef] [PubMed]
- Tsai, N.; Chen, Y.; Lee, C.; Lin, P.; Cheng, Y.; Chang, W.; Lin, S.; Harn, H. The natural compound n-butylidenephthalide derived from Angelica sinensis inhibits malignant brain tumor growth in vitro and in vivo. J. Neurochem. 2006, 99, 1251–1262. [Google Scholar] [CrossRef] [PubMed]
- Huang, J.; Lu, X.Q.; Zhang, C.; Lu, J.; Li, G.Y.; Lin, R.C.; Wang, J.H. Anti-inflammatory ligustilides from Ligusticum chuanxiong Hort. Fitoterapia 2013, 91, 21–27. [Google Scholar] [CrossRef] [PubMed]
- Nam, K.; Kim, K.; Cho, K.; Jung, W.; Park, J.; Cho, S.; Park, S.; Park, T.; Kim, Y.; Lee, E. Prevention of inflammation-mediated neurotoxicity by butylidenephthalide and its role in microglial activation. Cell. Biochem. Funct. 2013, 31, 707–712. [Google Scholar] [CrossRef] [PubMed]
- Yin, J.; Wang, C.; Mody, A.; Bao, L.; Hung, S.; Svoronos, S.; Tseng, Y. The effect of Z-ligustilide on the mobility of human glioblastoma T98G cells. PLoS ONE 2013, 8, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Li, W.; Tang, Y.; Chen, Y.; Duan, J. Advances in the chemical analysis and biological activities of chuanxiong. Molecules 2012, 17, 10614–10651. [Google Scholar] [CrossRef] [PubMed]
- Naito, T.; Katsuhara, T.; Niitsu, K.; Ikeya, Y.; Okada, M.; Mitsuhashi, H. Two phthalides from Ligusticum chuanxiong. Phytochemistry 1992, 31, 639–642. [Google Scholar] [CrossRef]
- Kosaka, M.; Sekiguchi, S.; Naito, J.; Uemura, M.; Kuwahara, S.; Watanabe, M.; Harada, N.; Hiroi, K. Synthesis of enantiopurephthalides including 3-butylphthalide, a fragrance component of celery oil, and determination of their absolute configurations. Chirality 2005, 17, 218–232. [Google Scholar] [CrossRef] [PubMed]
- Mahadevegowda, S.H.; Khan, F.A. Total syntheses of (±)-cis- and (±)-trans-neocnidilides. Tetrahedron Lett. 2014, 55, 400–4403. [Google Scholar] [CrossRef]
- Kobayashi, M.; Mitsuhashi, H. Studies on the constituents of Umbelliferae plants. XVII. (1) Structures of three new ligustilide derivatives from Ligusticum wallichii. Chem. Pharm. Bull. 1987, 35, 4789–4792. [Google Scholar] [CrossRef]
- Beck, J.J.; Stermitz, F.R. Addition of methyl thioglycolate and benzylamine to (Z)-ligustilide, a bioactive unsaturated lactone constituent of several herbal medicines. An improved synthesis of (Z)-ligustilide. J. Nat. Prod. 1995, 58, 1047–1055. [Google Scholar] [CrossRef] [PubMed]
- Leon, A.; Chavez, M.; Delgado, G. 1H- and DOSY-NMR spectroscopy analysis of Ligusticum porter rhizome extracts. Magn. Reson. Chem. 2011, 49, 469–476. [Google Scholar] [CrossRef] [PubMed]
- Lu, X.; Zhang, J.; Zhang, X.; Liang, H.; Zhao, Y. Study on biligustilides from Angelica sinensis. Chin. J. Chin. Mater. Med. 2008, 33, 2196–2201. [Google Scholar]
- Sample Availability: Samples of the compounds 1–8 are available from the authors.
© 2015 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons by Attribution (CC-BY) license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hu, Y.; Bi, X.; Zhao, P.; Zheng, H.; Huang, X. Cytotoxic Activities, SAR and Anti-Invasion Effects of Butylphthalide Derivatives on Human Hepatocellular Carcinoma SMMC7721 Cells. Molecules 2015, 20, 20312-20319. https://doi.org/10.3390/molecules201119699
Hu Y, Bi X, Zhao P, Zheng H, Huang X. Cytotoxic Activities, SAR and Anti-Invasion Effects of Butylphthalide Derivatives on Human Hepatocellular Carcinoma SMMC7721 Cells. Molecules. 2015; 20(11):20312-20319. https://doi.org/10.3390/molecules201119699
Chicago/Turabian StyleHu, Yihan, Xiaoxu Bi, Pu Zhao, Huachuan Zheng, and Xueshi Huang. 2015. "Cytotoxic Activities, SAR and Anti-Invasion Effects of Butylphthalide Derivatives on Human Hepatocellular Carcinoma SMMC7721 Cells" Molecules 20, no. 11: 20312-20319. https://doi.org/10.3390/molecules201119699
APA StyleHu, Y., Bi, X., Zhao, P., Zheng, H., & Huang, X. (2015). Cytotoxic Activities, SAR and Anti-Invasion Effects of Butylphthalide Derivatives on Human Hepatocellular Carcinoma SMMC7721 Cells. Molecules, 20(11), 20312-20319. https://doi.org/10.3390/molecules201119699






