Potent Activities of Roemerine against Candida albicans and the Underlying Mechanisms
Abstract
:1. Introduction
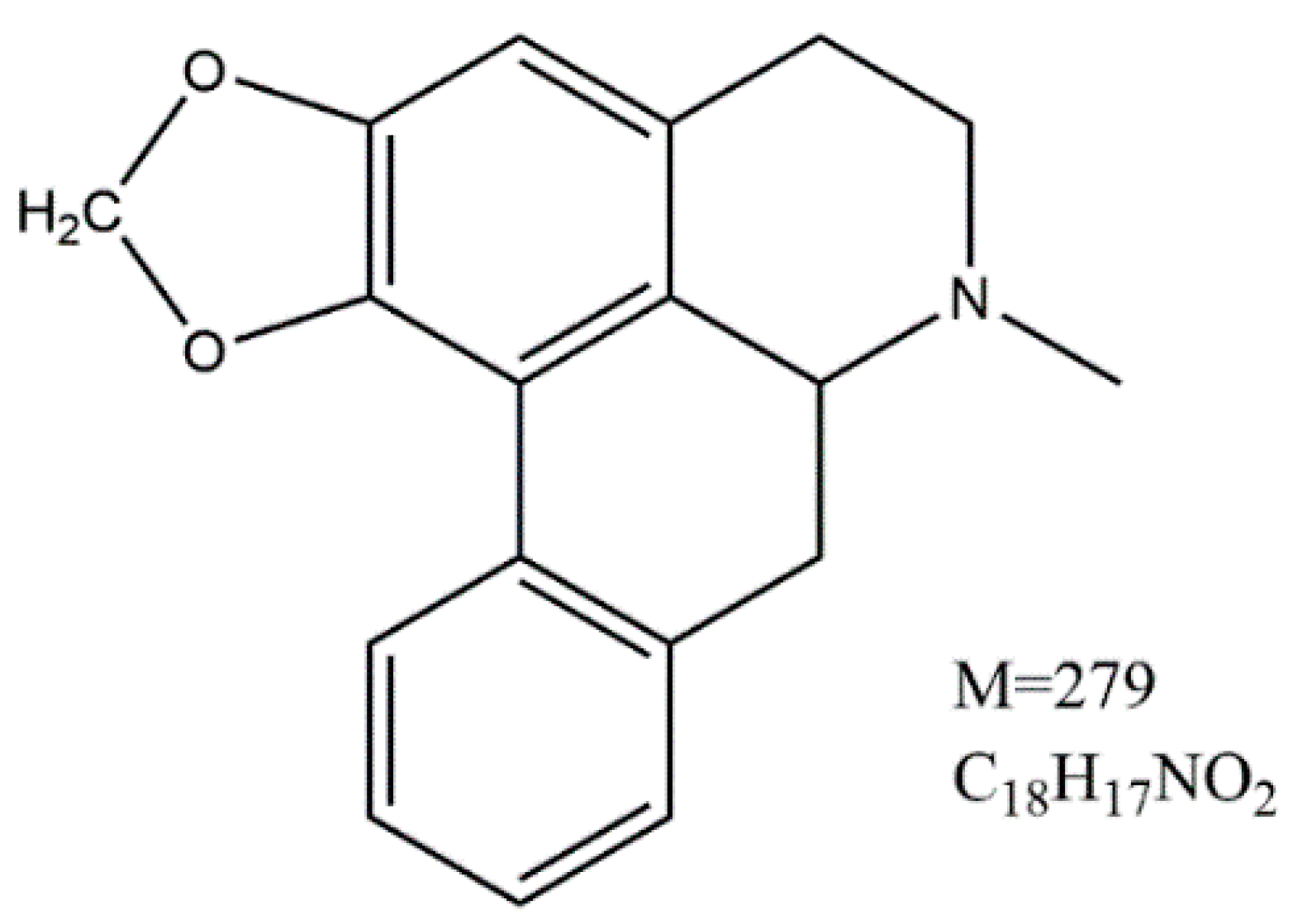
2. Results
2.1. Effect of RM against C. albicans Yeast Cells
| Strains | RM (MIC50s, mg/L) | RM (MIC80s, mg/L) |
|---|---|---|
| Candida albicans SC5314 | 128 | 256 |
| Candida glabrata 8535 | 32 | 64 |
| Candida krusei 4996 | 16 | 32 |
| Candida tropicalis 8915 | 32 | 64 |
| Candida parapsilosis 90018 | 16 | 32 |
| Aspergillus fumigatus 7544 | 16 | 32 |
| * MRSA 135 | 32 | 64 |
| MRSA 98 | 32 | 64 |
| MRSA 310 | 16 | 32 |
| MRSA 311 | 32 | 64 |
| MRSA 111 | 32 | 64 |
| MRSA 234 | 32 | 64 |
| MRSA 92 | 32 | 64 |
| MRSA 8 | 32 | 64 |
| Staphylococcus aureus ATCC 25913 | 32 | 64 |

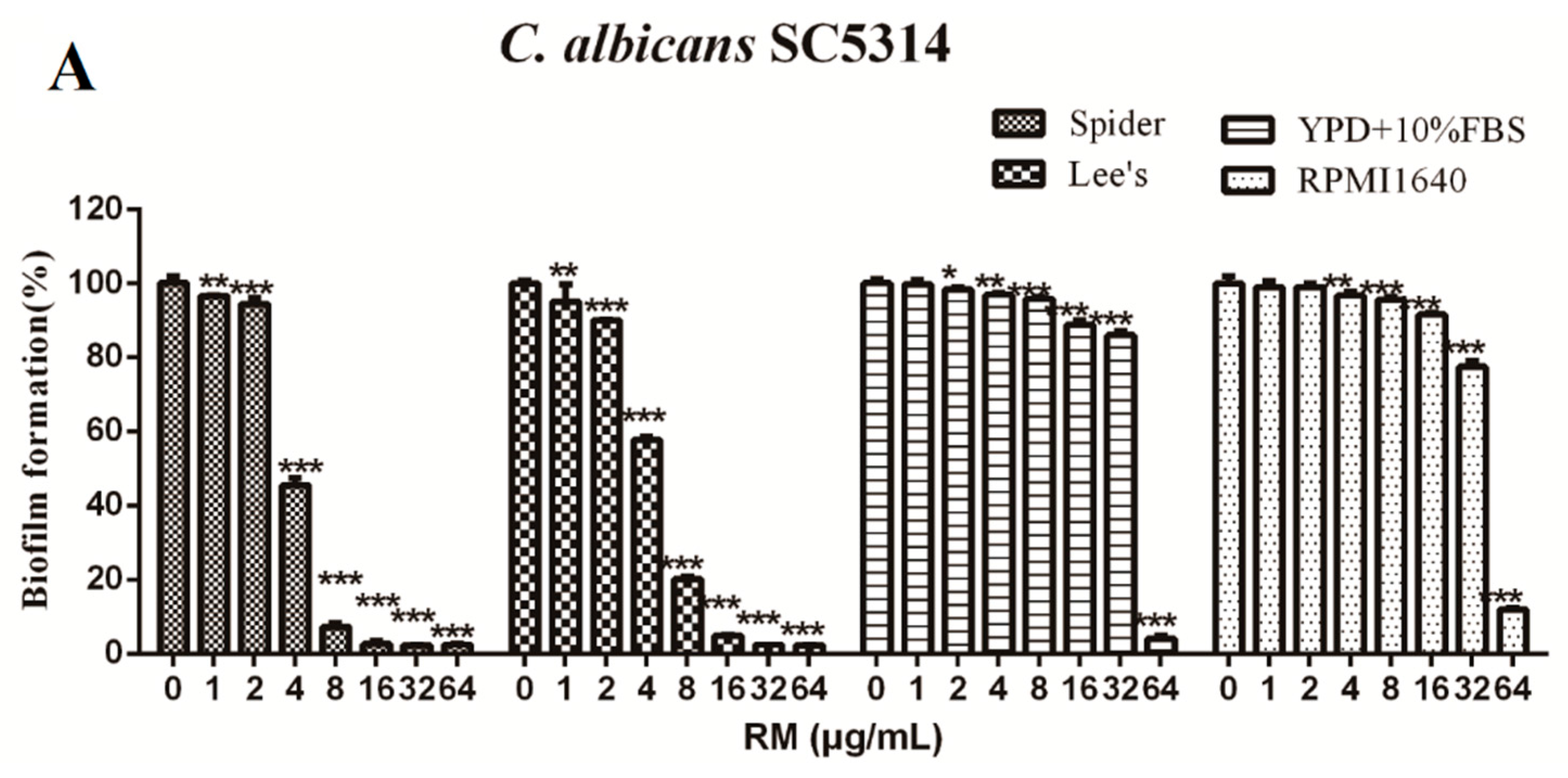
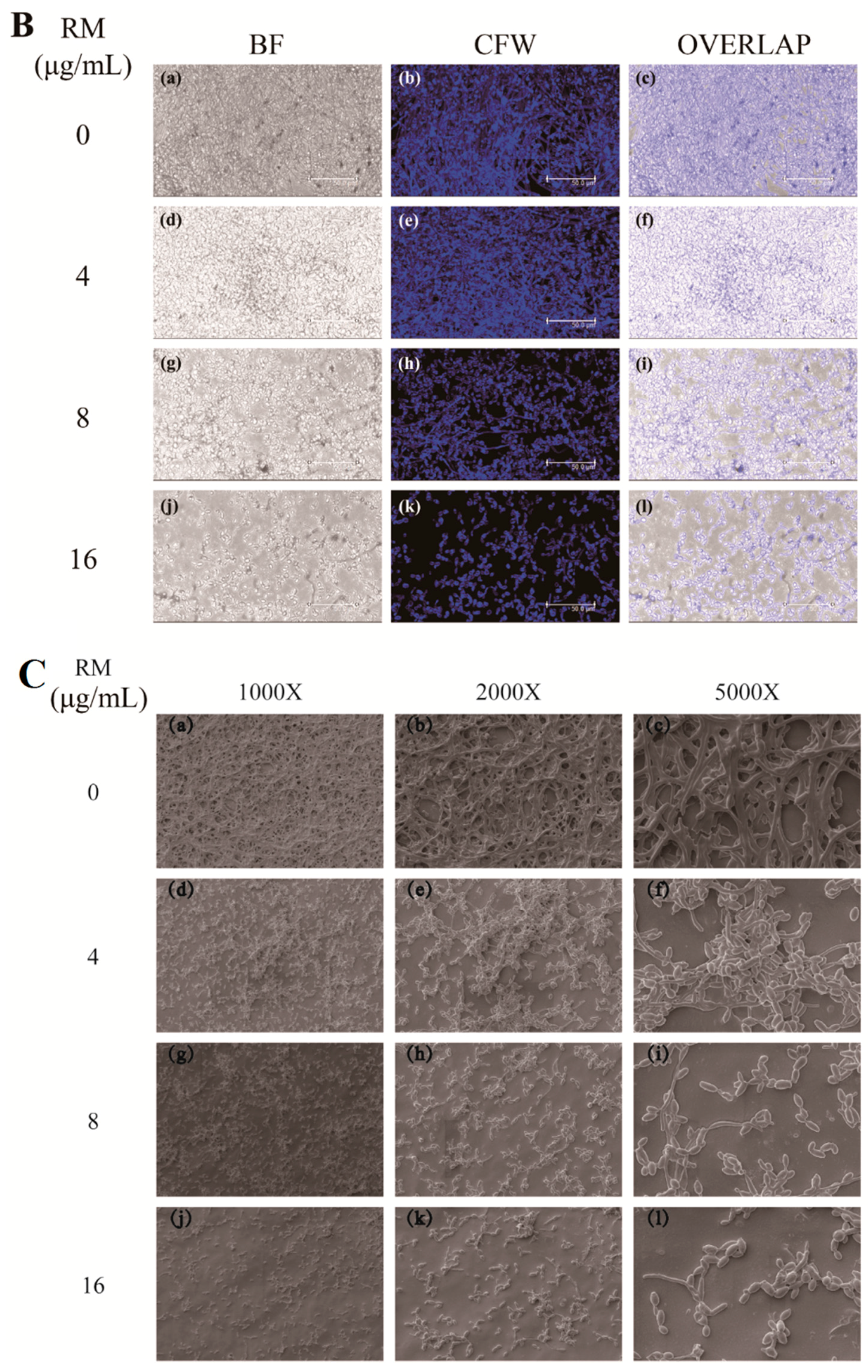
2.2. RM Inhibits the Formation of C. albicans Biofilms
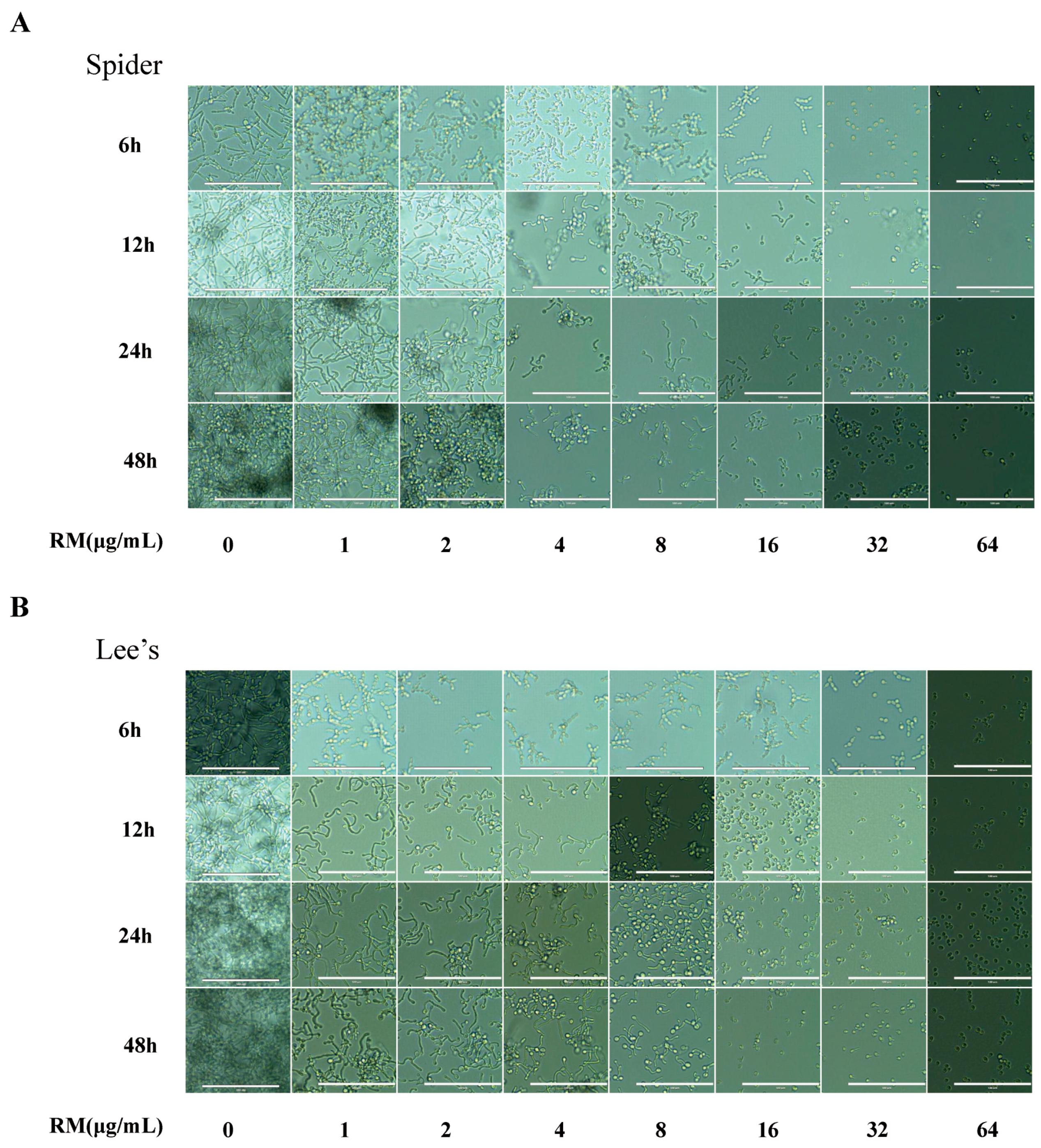
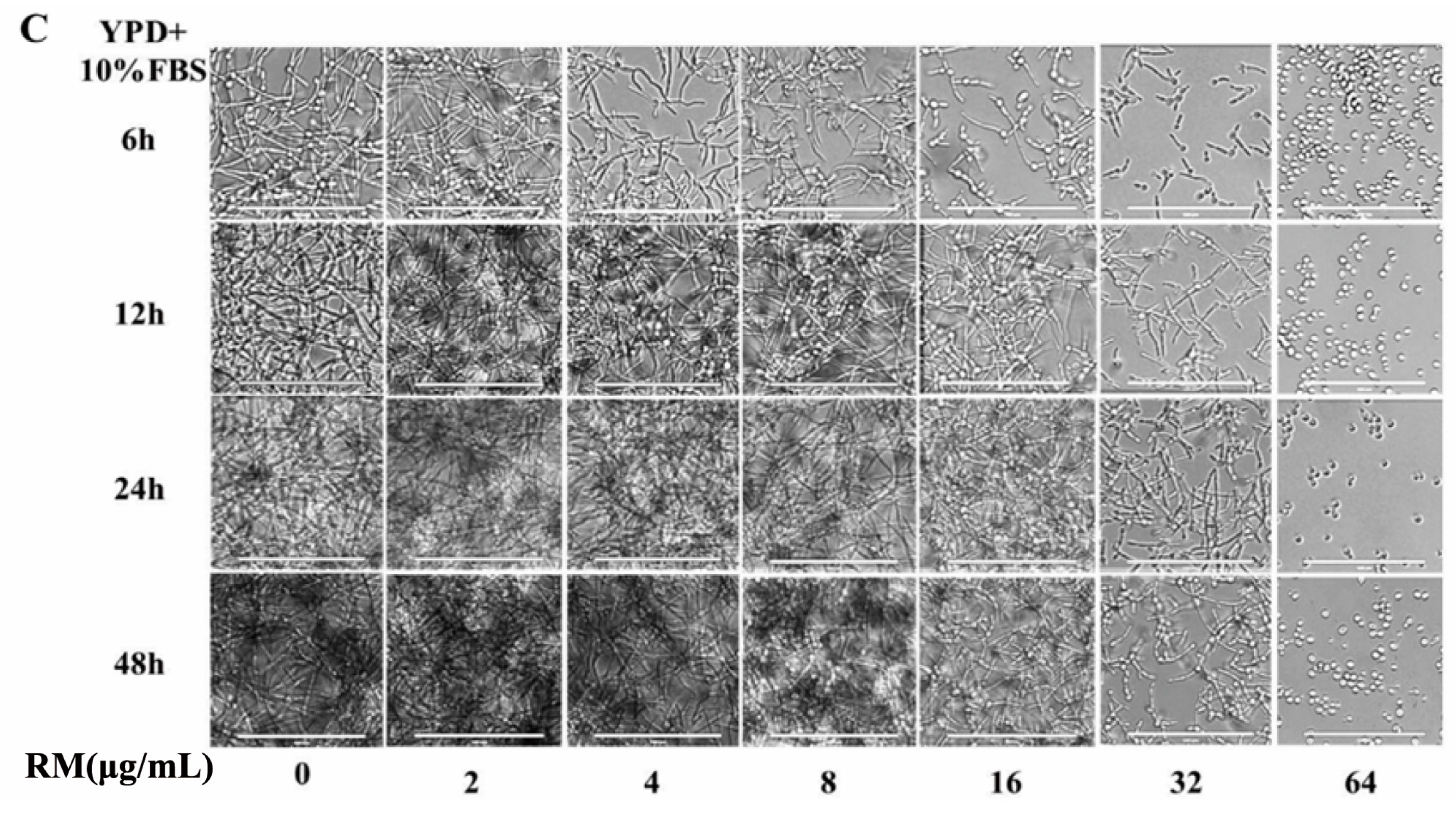
2.3. RM Inhibits the Yeast-to-Hypha Morphological Transition of C. albicans
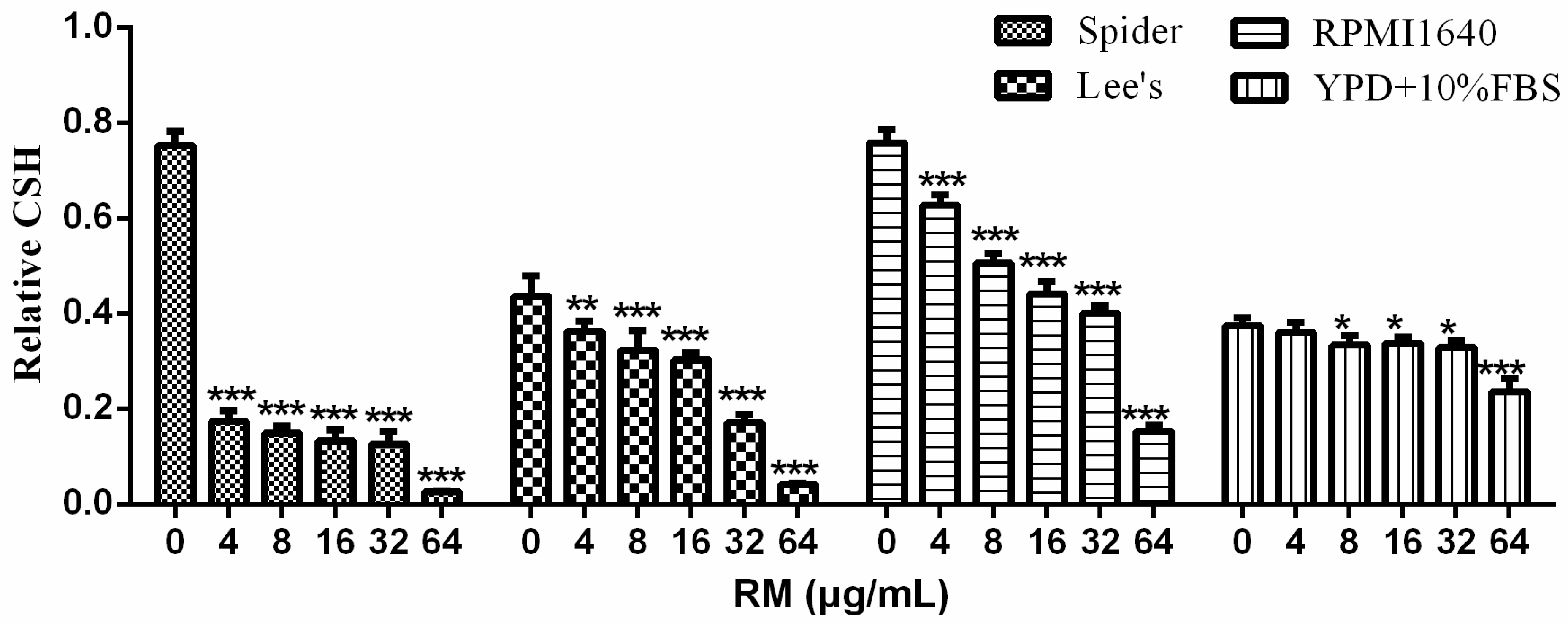
2.4. RM Decreases CSH of C. albicans Biofilm
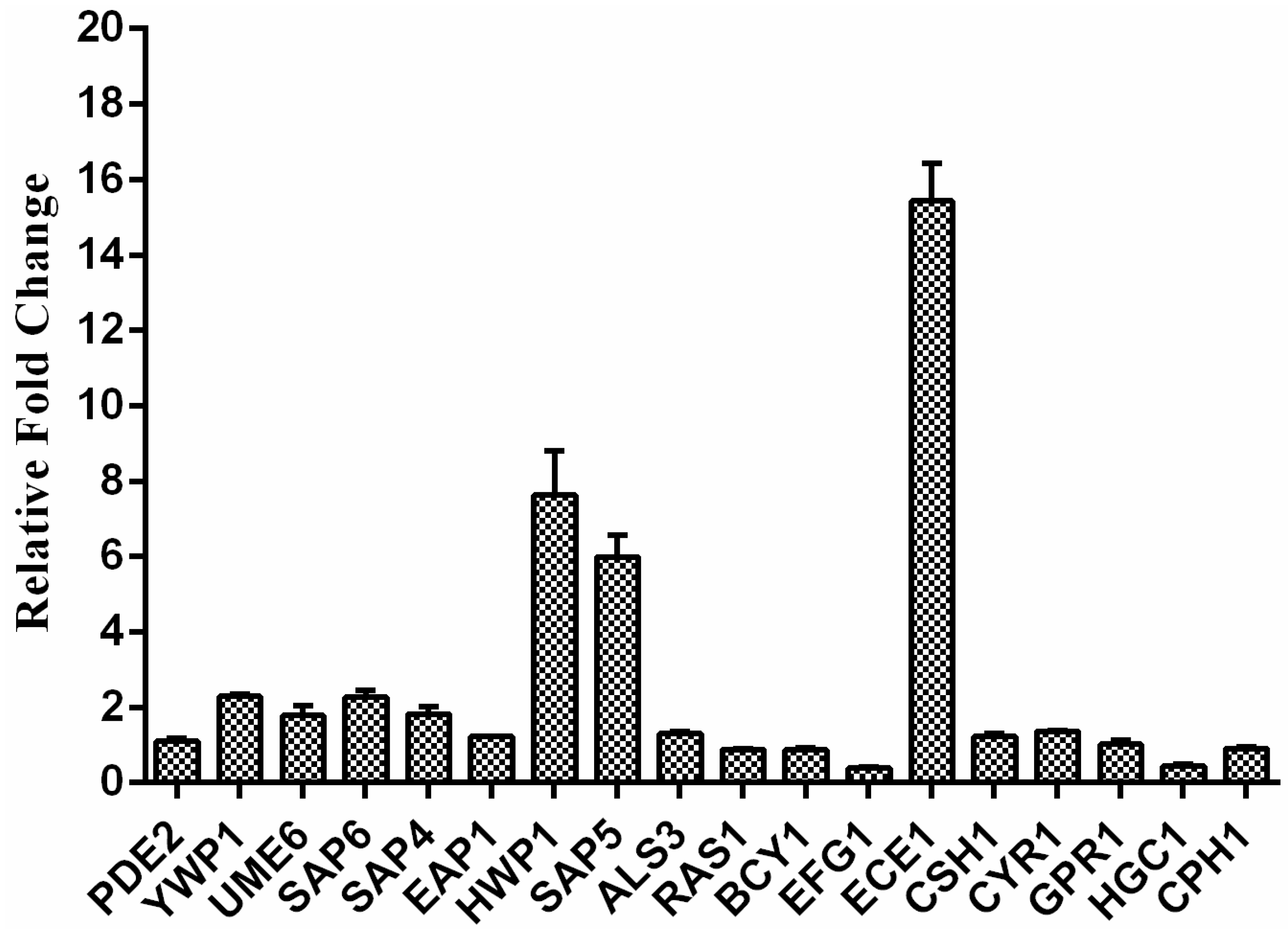
2.5. Exposure to RM Alters C. albicans Gene Transcription
2.6. Exogenous cAMP Reverts the Morphogenesis Defect Caused by RM
2.7. Cytotoxicity Studies

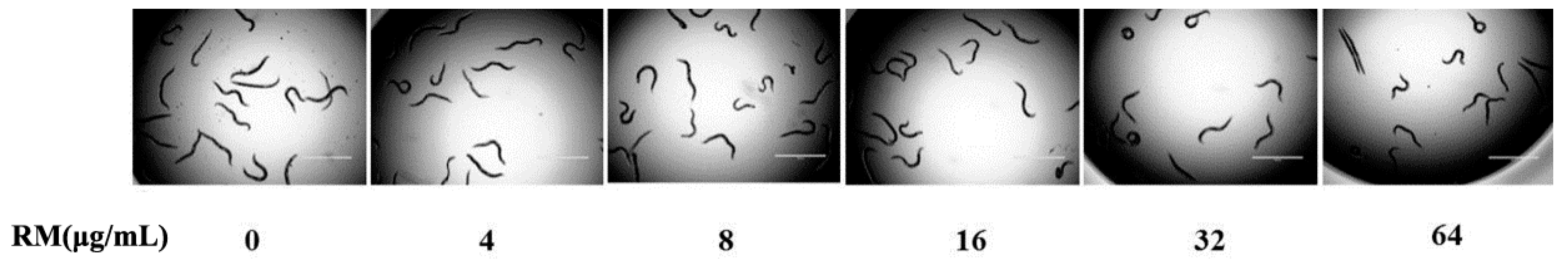
2.8. The Toxicity of RM on C. elegans Worms
3. Discussion
4. Experimental Section
4.1. Strains and Growth Condition
4.2. Antifungal Susceptibility Testing
4.3. Time-Growth Curves Assay
4.4. In Vitro Biofilm Formation Assay
4.5. Confocal Laser Scanning Microscopy (CLSM) Assay
4.6. Scanning Electron Microscopy (SEM) Assay
4.7. Yeast-to-Hyphae Transition Assays
4.8. Cellular Surface Hydrophobicity (CSH) Assay
4.9. Real-Time RT-PCR
4.10. Exogenous Cyclic AMP (cAMP) Supplement Assay
4.11. Cytotoxicity Studies
4.12. Toxicity Evaluation using C. elegans Worms
5. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Gow, N.A.; van de Veerdonk, F.L.; Brown, A.J.; Netea, M.G. Candida albicans morphogenesis and host defence: Discriminating invasion from colonization. Nat. Rev. Microbiol. 2011, 10, 112–122. [Google Scholar] [CrossRef] [PubMed]
- Li, D.D.; Xu, Y.; Zhang, D.Z.; Quan, H.; Mylonakis, E.; Hu, D.D.; Li, M.B.; Zhao, L.X.; Zhu, L.H.; Wang, Y.; et al. Fluconazole assists berberine to kill fluconazole-resistant Candida albicans. Antimicrob. Agents Chemother. 2013, 57, 6016–6027. [Google Scholar] [CrossRef] [PubMed]
- Sudbery, P.E. Growth of Candida albicans hyphae. Nat. Rev. Microbiol. 2011, 9, 737–748. [Google Scholar] [CrossRef] [PubMed]
- Zhao, L.X.; Li, D.D.; Hu, D.D.; Hu, G.H.; Yan, L.; Wang, Y.; Jiang, Y.Y. Effect of tetrandrine against Candida albicans biofilms. PLoS ONE 2013, 8, e79671. [Google Scholar] [CrossRef] [PubMed]
- Ramage, G.; Saville, S.P.; Thomas, D.P.; Lopez-Ribot, J.L. Candida biofilms: An update. Eukaryot. Cell 2005, 4, 633–638. [Google Scholar]
- Chandra, J.; Mukherjee, P.K.; Leidich, S.D.; Faddoul, F.F.; Hoyer, L.L.; Douglas, L.J.; Ghannoum, M.A. Antifungal resistance of candidal biofilms formed on denture acrylic in vitro. J. Dent. Res. 2001, 80, 903–908. [Google Scholar] [CrossRef] [PubMed]
- Valiente, M.; D’Ocon, P.; Noguera, MA.; Cassels, B.K.; Lugnier, C.; Ivorra, M.D. Vascular activity of (−)-anonaine, (−)-roemerine and (−)-pukateine, three natural 6a(R)-1, 2-methylenedioxyaporphines with different affinities for α1-adrenoceptor subtypes. Planta Med. 2004, 70, 603–609. [Google Scholar] [CrossRef] [PubMed]
- Lin, R.J.; Wu, M.H.; Ma, Y.H.; Chung, L.Y.; Chen, C.Y.; Yen, C.M. Anthelmintic activities of aporphine from Nelumbo nucifera Gaertn. cv. Rosa-plena against Hymenolepis nana. Int. J. Mol. Sci. 2014, 15, 3624–3639. [Google Scholar] [PubMed]
- Baghdikian, B.; Mahiou-Leddet, V.; Bory, S.; Bun, S.S.; Dumetre, A.; Mabrouki, F.; Hutter, S.; Azas, N.; Ollivier, E. New antiplasmodial alkaloids from Stephania rotunda. J. Ethnopharmacol. 2013, 145, 381–385. [Google Scholar] [CrossRef] [PubMed]
- You, M.; Wickramara, D.B.; Silva, G.L.; Chai, H.; Chagwedera, T.E.; Farnsworth, N.R.; Cordell, G.A.; Kinghorn, A.D.; Pezzuto, J.M. (−)-Roemerine, an aporphine alkaloid from Annona senegalensis that reverses the multidrug-resistance phenotype with cultured cells. J. Nat. Prod. 1995, 58, 598–604. [Google Scholar] [CrossRef] [PubMed]
- Munusamy, V.; Yap, B.K.; Buckle, M.J.; Doughty, S.W.; Chung, L.Y. Structure-based identification of aporphines with selective 5-HT (2A) receptor-binding activity. Chem. Biol. Drug Des. 2013, 81, 250–256. [Google Scholar] [CrossRef] [PubMed]
- Tsai, I.L.; Liou, Y.F.; Lu, S.T. Screening of isoquinoline alkaloids and their derivatives for antibacterial and antifungal activities. Kaohsiung J. Med. Sci. 1989, 5, 132–145. [Google Scholar]
- Rao, G.X.; Zhang, S.; Wang, H.M.; Li, Z.M.; Gao, S.; Xu, G.L. Antifungal alkaloids from the fresh rattan stem of Fibraurea recisa Pierre. J. Ethnopharmacol. 2009, 123, 1–5. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.Q.; He, G.H.; Li, H.L.; He, J.C.; Feng, E.F.; Bai, L.; Wang, C.Y.; Xu, G.L. Plasma pharmacokinetics and tissue distribution study of roemerine in rats by liquid chromatography with tandem mass spectrometry (LC-MS/MS). J. Chromatogr. B Anal. Technol. Biomed. Life Sci. 2014, 969, 249–255. [Google Scholar] [CrossRef] [PubMed]
- Cullen, P.J.; Sprague, G.J. The regulation of filamentous growth in yeast. Genetics 2012, 190, 23–49. [Google Scholar] [CrossRef] [PubMed]
- Lu, Y.; Su, C.; Liu, H. Candida albicans hyphal initiation and elongation. Trends Microbiol. 2014, 22, 707–714. [Google Scholar] [CrossRef] [PubMed]
- Bockmuhl, D.P.; Ernst, J.F. A potential phosphorylation site for an A-kinase in the Efg1 regulator protein contributes to hyphal morphogenesis of Candida albicans. Genetics 2001, 157, 1523–1530. [Google Scholar] [PubMed]
- Teodoro, J.G.; Heilman, D.W.; Parker, A.E.; Green, M.R. The viral protein Apoptin associates with the anaphase-promoting complex to induce G2/M arrest and apoptosis in the absence of p53. Genes Dev. 2004, 18, 1952–1957. [Google Scholar] [CrossRef] [PubMed]
- Liu, S.; Hou, Y.; Chen, X.; Gao, Y.; Li, H.; Sun, S. Combination of fluconazole with non-antifungal agents: A promising approach to cope with resistant Candida albicans infections and insight into new antifungal agent discovery. Int. J. Antimicrob. Agents 2014, 43, 395–402. [Google Scholar] [CrossRef] [PubMed]
- Chandra, J.; Kuhn, D.M.; Mukherjee, P.K.; Hoyer, L.L.; McCormick, T.; Ghannoum, M.A. Biofilm formation by the fungal pathogen Candida albicans: Development, architecture, and drug resistance. J. Bacteriol. 2001, 183, 5385–5394. [Google Scholar] [CrossRef] [PubMed]
- Mayer, F.L.; Wilson, D.; Hube, B. Candida albicans pathogenicity mechanisms. Virulence 2014, 4, 119–128. [Google Scholar] [CrossRef] [PubMed]
- Samaranayake, Y.H.; Wu, P.C.; Samaranayake, L.P.; So, M. Relationship between the cell surface hydrophobicity and adherence of Candida krusei and Candida albicans to epithelial and denture acrylic surfaces. APMIS 1995, 103, 707–713. [Google Scholar] [CrossRef] [PubMed]
- Fan, Y.; He, H.; Dong, Y.; Pan, H. Hyphae-specific genes HGC1, ALS3, HWP1, and ECE1 and relevant signaling pathways in Candida albicans. Mycopathologia 2013, 176, 329–335. [Google Scholar] [CrossRef] [PubMed]
- Nobile, C.J.; Schneider, H.A.; Nett, J.E.; Sheppard, D.C.; Filler, S.G.; Andes, D.R.; Mitchell, A.P. Complementary adhesin function in C. albicans biofilm formation. Curr. Biol. 2008, 18, 1017–1024. [Google Scholar] [CrossRef] [PubMed]
- Moazeni, M.; Khoramizadeh, M.R.; Teimoori-Toolabi, L.; Noorbakhsh, F.; Rezaie, S. The effect of EFG1 gene silencing on down-regulation of SAP5 gene, by use of RNAi technology. Acta Med. Iran. 2014, 52, 9–14. [Google Scholar] [PubMed]
- Tran, T.D.; Pham, N.B.; Fechner, G.; Quinn, R.J. Chemical investigation of drug-like compounds from the Australian tree, Neolitsea dealbata. Bioorg. Med. Chem. Lett. 2010, 20, 5859–5863. [Google Scholar] [CrossRef] [PubMed]
- Krause, J.; Geginat, G.; Tammer, I. Prostaglandin E2 from Candida albicans stimulates the growth of Staphylococcus aureus in mixed biofilms. PLoS ONE 2015, 10, e0135404. [Google Scholar] [CrossRef] [PubMed]
- Clinical and Laboratory Standards Institute (CLSI). M07-A9: Methods for Dilution Antimicrobial Susceptibility Tests for Bacteria That Grow Aerobically; Approved Standard—Ninth Edition; CLSI Publication: Wayne, PA, USA, 2012. [Google Scholar]
- Clinical and Laboratory Standards Institute (CLSI). M27-A3: Reference Method for Broth Dilution Antifungal Susceptibility Testing of Yeasts; Approved Standard—Third Edition; CLSI Publication: Wayne, PA, USA, 2009. [Google Scholar]
- Clinical and Laboratory Standards Institute (CLSI). M27-S4: Reference method for Broth Dilution Antifungal Susceptibility Testing of Yeasts; Fourth Informational Supplement; CLSI Publication: Wayne, PA, USA, 2012. [Google Scholar]
- Dhamgaye, S.; Devaux, F.; Vandeputte, P.; Khandelwal, N.K.; Sanglard, D.; Mukhopadhyay, G.; Prasad, R. Molecular mechanisms of action of herbal antifungal alkaloid berberine, in Candida albicans. PLoS ONE 2014, 9, e104554. [Google Scholar] [CrossRef] [PubMed]
- Li, D.D.; Zhao, L.X.; Mylonakis, E.; Hu, G.H.; Zou, Y.; Huang, T.K.; Yan, L.; Wang, Y.; Jiang, Y.Y. In vitro and in vivo activities of pterostilbene against Candida albicans biofilms. Antimicrob. Agents Chemother. 2014, 58, 2344–2355. [Google Scholar] [CrossRef] [PubMed]
- Daniels, K.J.; Park, Y.N.; Srikantha, T.; Pujol, C.; Soll, D.R. Impact of environmental conditions on the form and function of Candida albicans biofilms. Eukaryotic Cell 2013, 12, 1389–1402. [Google Scholar] [CrossRef] [PubMed]
- Maidan, M.M.; De Rop, L.; Serneels, J.; Exler, S.; Rupp, S.; Tournu, H.; Thevelein, J.M.; van Dijck, P. The G protein-coupled receptor Gpr1 and the Galpha protein Gpa2 act through the cAMP-protein kinase A pathway to induce morphogenesis in Candida albicans. Mol. Biol. Cell 2005, 16, 1971–1986. [Google Scholar] [CrossRef] [PubMed]
- Cao, Y.Y.; Dai, B.; Wang, Y.; Huang, S.; Xu, Y.; Cao, Y.; Gao, P.; Zhu, Z.; Jiang, Y. In vitro activity of baicalein against Candida albicans biofilms. Int. J. Antimicrob. Agents 2008, 32, 73–77. [Google Scholar] [CrossRef] [PubMed]
- Nobile, C.J.; Andes, D.R.; Nett, J.E.; Smith, F.J.; Yue, F.; Phan, Q.T.; Edwards, J.E.; Filler, S.G.; Mitchell, A.P. Critical role of Bcr1-dependent adhesins in C. albicans biofilm formation in vitro and in vivo. PLoS Pathog. 2006, 2, e63. [Google Scholar] [CrossRef] [PubMed]
- Klotz, S.A.; Drutz, D.J.; Zajic, J.E. Factors governing adherence of Candida species to plastic surfaces. Infect. Immun. 1985, 50, 97–101. [Google Scholar] [PubMed]
- Sun, L.M.; Cheng, A.X.; Wu, X.Z.; Zhang, H.J.; Lou, H.X. Synergistic mechanisms of retigeric acid B and azoles against Candida albicans. J. Appl. Microbiol. 2010, 108, 341–348. [Google Scholar] [CrossRef] [PubMed]
- Li, D.D.; Wang, Y.; Dai, B.D.; Li, X.X.; Zhao, L.X.; Cao, Y.B.; Yan, L.; Jiang, Y.Y. ECM17-dependent methionine/cysteine biosynthesis contributes to biofilm formation in Candida albicans. Fungal Genet. Biol. 2013, 51, 50–59. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.; Jang, J.H.; Kim, S.C.; Cho, J.H. De novo generation of short antimicrobial peptides with enhanced stability and cell specificity. J. Antimicrob. Chemother. 2014, 69, 121–132. [Google Scholar] [CrossRef] [PubMed]
- Sample Availability: Samples of the isolated compounds are available from the authors.
© 2015 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ma, C.; Du, F.; Yan, L.; He, G.; He, J.; Wang, C.; Rao, G.; Jiang, Y.; Xu, G. Potent Activities of Roemerine against Candida albicans and the Underlying Mechanisms. Molecules 2015, 20, 17913-17928. https://doi.org/10.3390/molecules201017913
Ma C, Du F, Yan L, He G, He J, Wang C, Rao G, Jiang Y, Xu G. Potent Activities of Roemerine against Candida albicans and the Underlying Mechanisms. Molecules. 2015; 20(10):17913-17928. https://doi.org/10.3390/molecules201017913
Chicago/Turabian StyleMa, Chaoyu, Faya Du, Lan Yan, Gonghao He, Jianchang He, Chengying Wang, Gaoxiong Rao, Yuanying Jiang, and Guili Xu. 2015. "Potent Activities of Roemerine against Candida albicans and the Underlying Mechanisms" Molecules 20, no. 10: 17913-17928. https://doi.org/10.3390/molecules201017913
APA StyleMa, C., Du, F., Yan, L., He, G., He, J., Wang, C., Rao, G., Jiang, Y., & Xu, G. (2015). Potent Activities of Roemerine against Candida albicans and the Underlying Mechanisms. Molecules, 20(10), 17913-17928. https://doi.org/10.3390/molecules201017913





